Estimation of Solute-Stationary Phase and Solute-Mobile Phase Interactions in the Presence of Ionic Liquids
Abstract
:1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Instrumentation
2.3. Experimental Designs
3. Results and Discussion
3.1. Solute-Stationary Phase and Solute-Mobile Phase Interactions
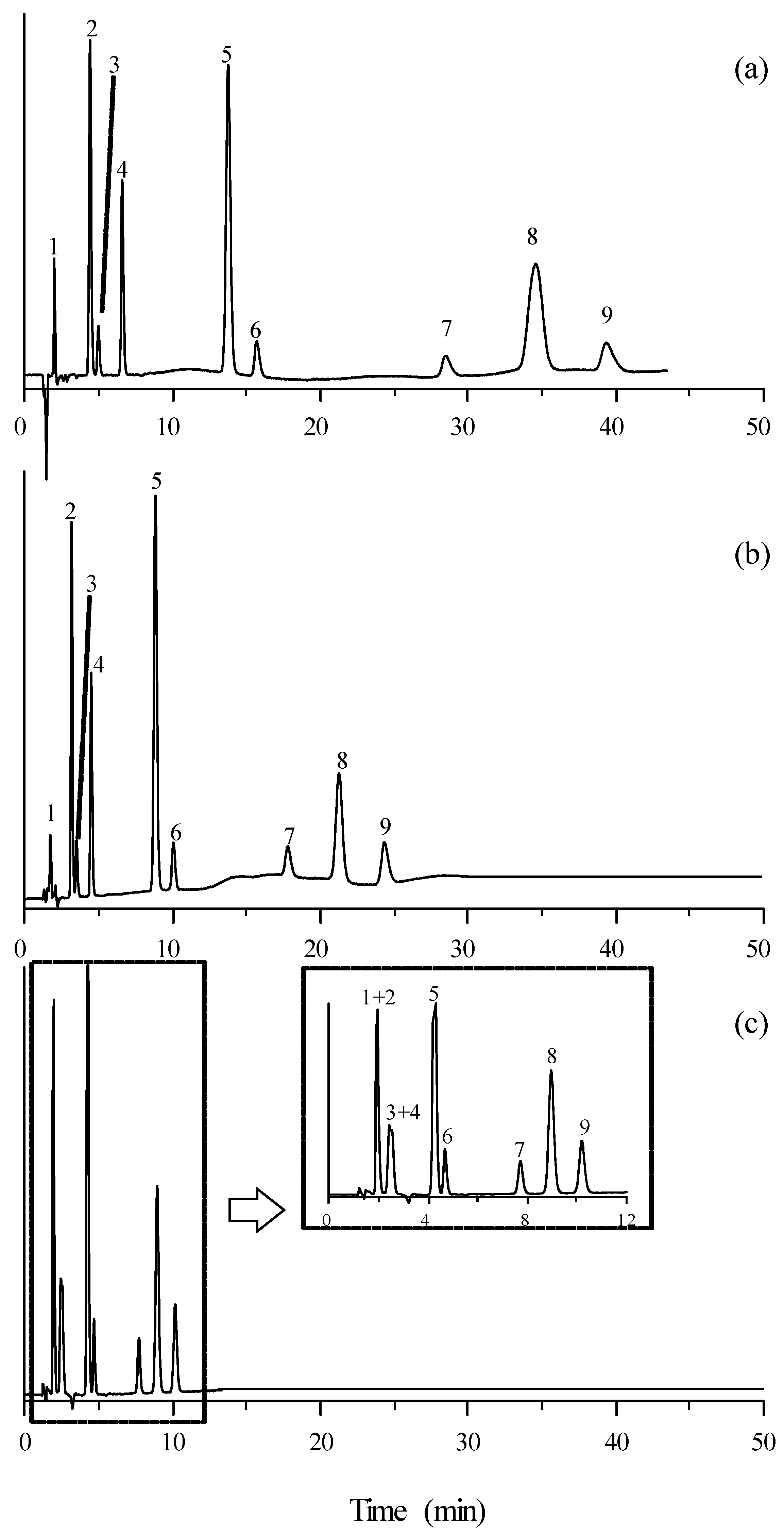
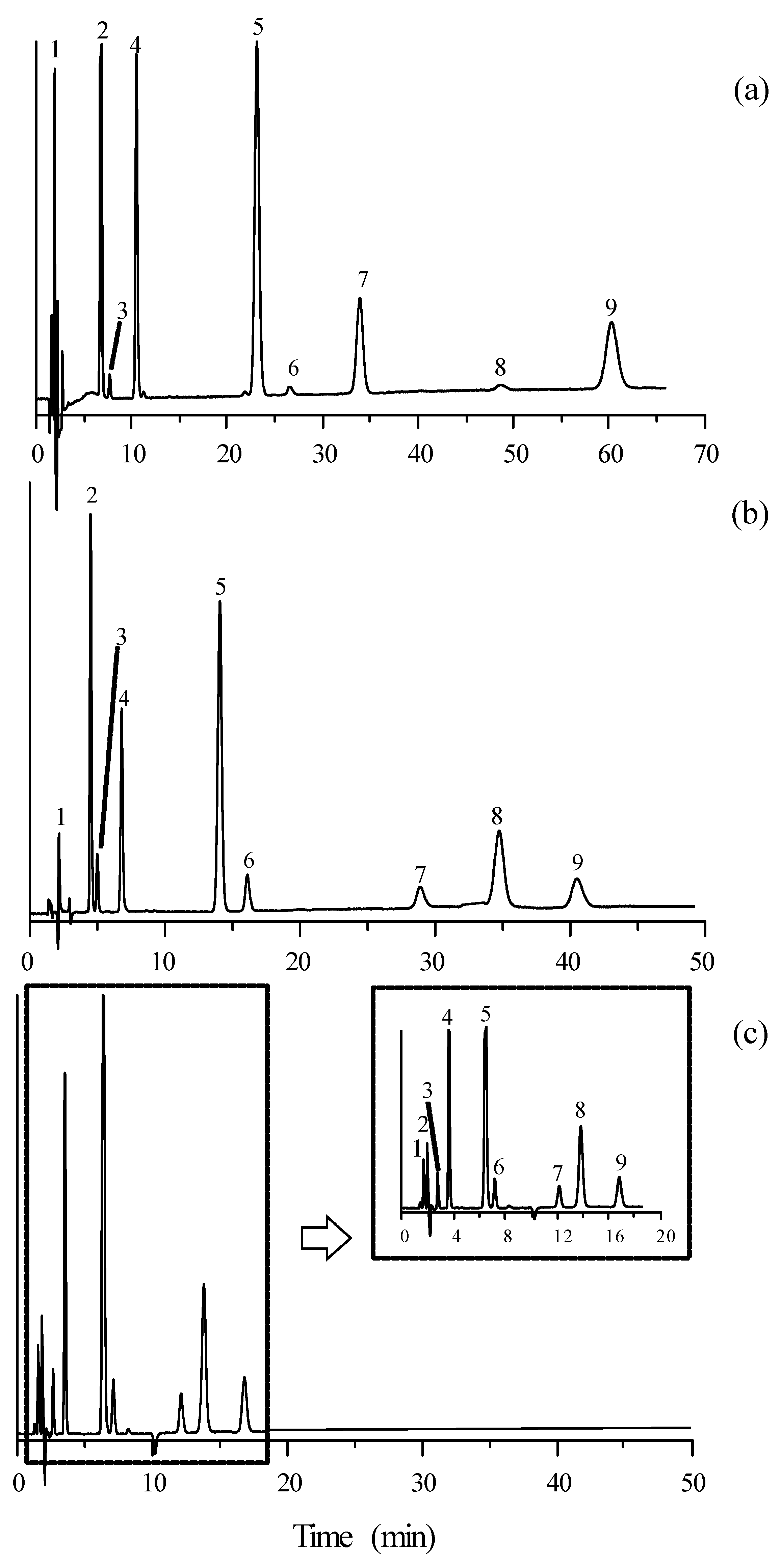
3.2. Separation of Mixtures of β-Adrenoceptor Antagonists
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Snyder, L.R.; Kirkland, J.J.; Dolan, J.W. Introduction to Modern Liquid Chromatography, 3rd ed.; Wiley: New York, NY, USA, 2010. [Google Scholar]
- García-Alvarez-Coque, M.C.; Ramis-Ramos, G.; Baeza-Baeza, J.J. Reversed phase liquid chromatography. In Analytical Separation Science Series; Anderson, J., Berthod, A., Pino, V., Stalcup, A.M., Eds.; Wiley: New York, NY, USA, 2015; Volume 1, pp. 159–198. [Google Scholar]
- Méndez, A.; Bosch, E.; Rosés, M.; Neue, U.D. Comparison of the acidity of residual silanol groups in several liquid chromatography columns. J. Chromatogr. A 2003, 986, 33–44. [Google Scholar] [CrossRef]
- Nawrocki, J. The silanol group and its role in liquid chromatography. J. Chromatogr. A 1997, 779, 29–71. [Google Scholar] [CrossRef]
- Reta, M.; Carr, P.W. Comparative study of divalent metals and amines as silanol-blocking agents in reversed-phase liquid chromatography. J. Chromatogr. A 1999, 855, 121–127. [Google Scholar] [CrossRef]
- Neue, U.D.; Tran, K.; Méndez, A.; Carr, P.W. The combined effect of silanols and the reversed-phase ligand on the retention of positively charged analytes. J. Chromatogr. A 2005, 1063, 35–45. [Google Scholar] [CrossRef] [PubMed]
- Engelhardt, H.; Blay, Ch.; Saar, J. Reversed phase chromatography: The mystery of surface silanols. Chromatographia 2005, 62, S19–S29. [Google Scholar] [CrossRef]
- Ruiz-Angel, M.J.; Carda-Broch, S.; Torres-Lapasió, J.R.; García-Álvarez-Coque, M.C. Retention mechanisms in micellar liquid chromatography, J. Chromatrogr. A 2009, 1216, 1798–1814. [Google Scholar] [CrossRef]
- McCalley, D.V. The challenges of the analysis of basic compounds by high performance liquid chromatography: Some possible approaches for improved separations. J. Chromatogr. A 2010, 1217, 858–880. [Google Scholar] [CrossRef]
- Calabuig-Hernández, S.; García-Alvarez-Coque, M.C.; Ruiz-Angel, M.J. Performance of amines as silanol suppressors in reversed-phase liquid chromatography. J. Chromatogr. 2016, 1465, 98–106. [Google Scholar] [CrossRef]
- Ruiz-Ángel, M.J.; Torres-Lapasió, J.R.; García-Álvarez-Coque, M.C.; Carda-Broch, S. Retention Mechanisms for Basic Drugs in the Submicellar and Micellar Reversed-Phase Liquid Chromatographic Modes. Anal. Chem. 2008, 80, 9705–9713. [Google Scholar] [CrossRef]
- Herrera-Herrera, A.V.; Hernández-Borges, J.; Rodríguez-Delgado, M.A. Ionic liquids as mobile phase additives for the high-performance liquid chromatographic analysis of fluoroquinolone antibiotics in water samples. Anal. Bioanal. Chem. 2008, 392, 1439–1446. [Google Scholar] [CrossRef]
- Martín-Calero, A.; Pino, V.; Ayala, J.H.; González, V.; Afonso, A.M. Ionic liquids as mobile phase additives in high-performance liquid chromatography with electrochemical detection: Application to the determination of heterocyclic aromatic amines in meat-based infant foods. Talanta 2009, 79, 590–597. [Google Scholar] [CrossRef]
- Martín-Calero, A.; Tejral, G.; Ayala, J.H.; González, V.; Afonso, A.M. Suitability of ionic liquids as mobile-phase additives in HPLC with fluorescence and UV detection for the determination of heterocyclic aromatic amines. J. Sep. Sci. 2010, 33, 182–190. [Google Scholar] [CrossRef]
- Petruczynik, A. Effect of ionic liquid additives to mobile phase on separation and system efficiency for HPLC of selected alkaloids on different stationary phases. J. Chromatogr. Sci. 2012, 50, 287–293. [Google Scholar] [CrossRef]
- Tang, Y.; Sun, A.; Liu, R.; Zhang, Y. Simultaneous determination of fangchinoline and tetrandrine in Stephania tetrandra S. Moore by using 1-alkyl-3-methylimidazolium- based ionic liquids as the RP-HPLC mobile phase additives. Anal. Chim. Acta 2013, 767, 148–154. [Google Scholar] [CrossRef]
- García-Alvarez-Coque, M.C.; Ruiz-Angel, M.J.; Berthod, A.; Carda-Broch, S. On the use of ionic liquids as mobile phase additives in high-performance liquid chromatography. Anal. Chim. Acta 2015, 883, 1–21. [Google Scholar] [CrossRef]
- Poole, C.F. Chromatographic and spectroscopic methods for the determination of solvent properties of room temperature ionic liquids. J. Chromatogr. A 2004, 1037, 49–82. [Google Scholar] [CrossRef]
- Ho, T.D.; Zhang, C.; Hantao, L.W.; Anderson, J.L. Ionic liquids in analytical chemistry: Fundamentals, advances, and perspectives. Anal. Chem. 2014, 86, 262–285. [Google Scholar] [CrossRef]
- Berthod, A.; Ruiz-Ángel, M.J.; Carda-Broch, S. Recent advances on ionic liquid uses in separation techniques. J. Chromatogr. A 2018, 1559, 2–16. [Google Scholar] [CrossRef]
- Zhao, D.; Liao, Y.; Zhang, Z. Toxicity of ionic liquids. Clean 2007, 35, 42–48. [Google Scholar] [CrossRef]
- Cevasco, G.; Chiappe, C. Are ionic liquids a proper solution to current environmental challenges? Green Chem. 2014, 16, 2375–2385. [Google Scholar] [CrossRef]
- Fernández-Navarro, J.J.; García-Álvarez-Coque, M.C.; Ruiz-Ángel, M.J. The role of the dual nature of ionic liquids in the reversed-phase liquid chromatographic separation of basic drugs. J. Chromatogr. A 2011, 1218, 398–407. [Google Scholar] [CrossRef]
- Ubeda-Torres, M.T.; Ortiz-Bolsico, C.; García-Alvarez-Coque, M.C.; Ruiz-Angel, M.J. Gaining insight in the behaviour of imidazolium-based ionic liquids asadditives in reversed-phase liquid chromatography for the analysis of basic compounds. J. Chromatogr. A 2015, 1380, 96–103. [Google Scholar] [CrossRef]
- Mehvar, R.; Brocks, D.R. Stereospecific pharmacokinetics and pharmacodynamics of beta-adrenergic blockers in humans. J. Pharm. Sci. 2001, 4, 185–200. [Google Scholar]
- Berthod, A.; García-Álvarez-Coque, M.C. Micellar Liquid Chromatography; Marcel Dekker: New York, NY, USA, 2000. [Google Scholar]
- Armstrong, D.W.; Nome, F. Partitioning behavior of solutes eluted with micellar mobile phases in liquid chromatography. Anal. Chem. 1981, 53, 1662–1666. [Google Scholar] [CrossRef]
- Arunyanart, M.; Cline-Love, L.J. Model for micellar effects on liquid chromatography capacity factors and for determination of micelle-solute equilibrium constants. Anal. Chem. 1984, 56, 1557–1561. [Google Scholar] [CrossRef]
- Berthod, A.; Ruiz-Angel, M.J.; Huguet, S. Nonmolecular solvents in separation methods: Dual nature of room temperature ionic liquids. Anal. Chem. 2005, 77, 4071–4080. [Google Scholar] [CrossRef]
- Armstrong, D.W.; Stine, G.Y. Selectivity in pseudophase liquid chromatography. Anal. Chem. 1983, 55, 2317–2320. [Google Scholar] [CrossRef]
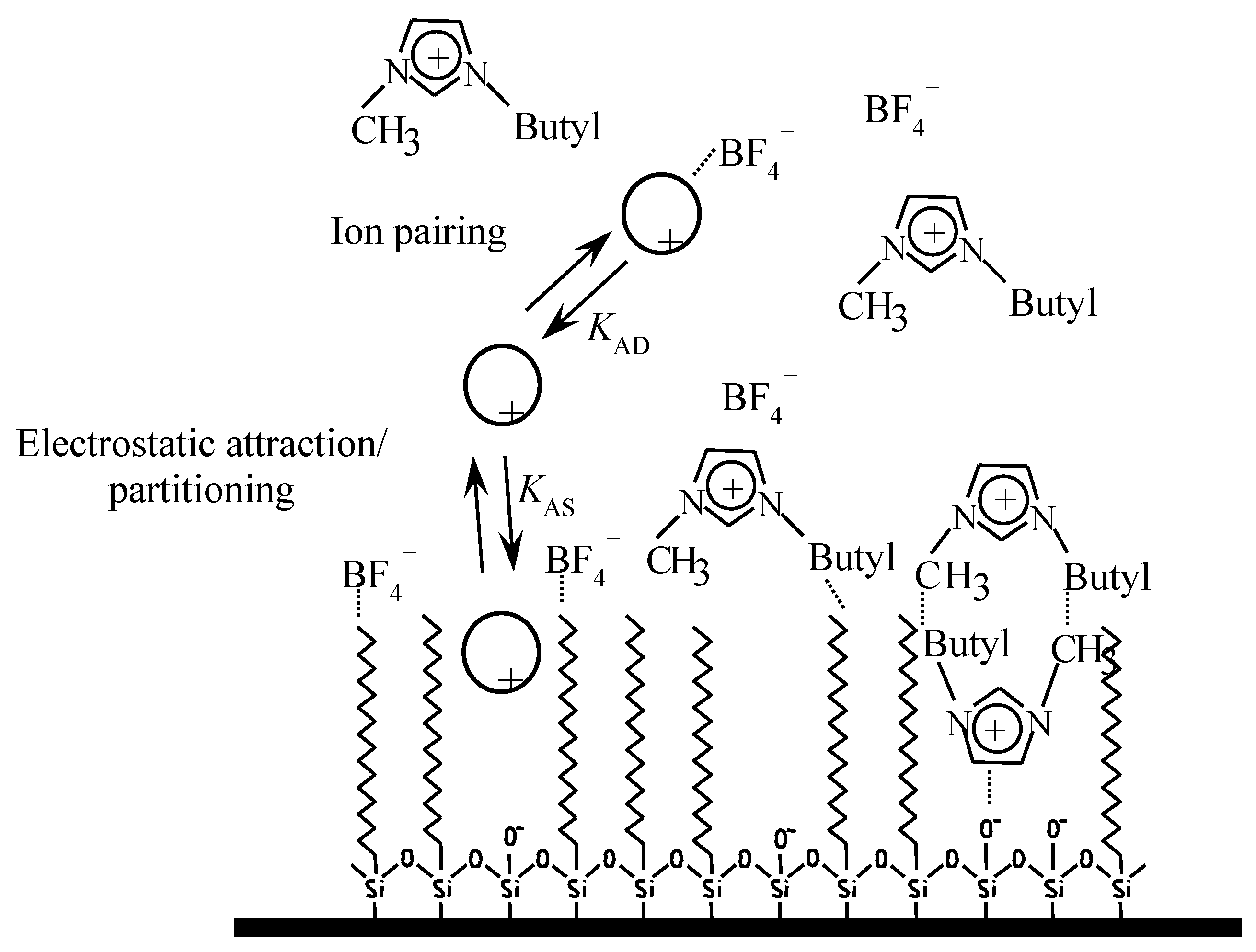
| Ionic Liquid | 1-R-3-Methylimidazolium Cation 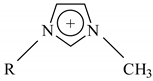 | Concentration Range (mM) | Anion | m.p. (°C) a | d (g/mL) a | Water Solubility a |
|---|---|---|---|---|---|---|
| [C2MIM][Cl] | Ethyl− | 10–40 | [Cl]− | 87 | – | Soluble |
| [C4MIM][Cl] | Butyl– | 10–40 | [Cl]− | 65 | 1.09 | Soluble |
| [C6MIM][Cl] | Hexyl– | 10–40 | [Cl]− | −70 | 1.03 | Soluble |
| [C2MIM][BF4] | Ethyl− | 5–30 | [BF4]− | 15 | 1.29 | Soluble |
| [C4MIM][BF4] | Butyl− | 10–40 | [BF4]− | −71 | 1.21 | Soluble |
| [C6MIM][BF4] | Hexyl− | 10–40 | [BF4]− | −81 | 1.15 | Non soluble |
| [C2MIM][Cl] | [C4MIM][Cl] | [C6MIM][Cl] | ||||
|---|---|---|---|---|---|---|
| Compound | KAS | KAD | KAS | KAD | KAS | KAD |
| Acebutolol | 6.3 ± 0.3 | 0.2 ± 1.8 | 4.4 ± 0.3 | 15.4 ± 9.0 | 1.30 ± 0.05 | 11.6 ± 1.4 |
| Atenolol | 0.300 ± 0.006 | −8.2 ± 0.8 | 0.50 ± 0.01 | 17.7 ± 0.6 | −0.100 ± 0.006 | −15.0 ± 9.9 |
| Carteolol | 1.40 ± 0.06 | −1.2 ± 0.6 | 0.80 ± 0.09 | 7.9 ± 0.9 | 0.20 ± 0.04 | 5.2 ± 0.5 |
| Celiprolol | 17.3 ± 0.6 | 1.5 ± 0.4 | 11.6 ± 2.0 | 14.0 ± 2.5 | 3.9 ± 0.3 | 14.9 ± 1.4 |
| Esmolol | 16.6 ± 1.8 | 7.0 ± 1.1 | 18.5 ± 1.4 | 58.5 ± 3.6 | 3.3 ± 0.3 | 10.4 ± 1.3 |
| Metoprolol | 7.9 ± 0.2 | 2.3 ± 0.9 | 4.8 ± 1.4 | 6.8 ± 2.1 | 1.6 ± 0.1 | 10.3 ± 1.5 |
| Nadolol | 1.7 ± 0.09 | −6.4 ± 3.5 | 1.3 ± 1.5 | 30.2 ± 3.5 | 0.50 ± 0.08 | 5.9 ± 2.6 |
| Oxprenolol | 20.800 ± 0.001 | 2.20 ± 0.05 | 16.0 ± 4.7 | 26.0 ± 7.7 | 4.7 ± 0.4 | 10.5 ± 1.5 |
| Pindolol | 2.8 ± 0.009 | 2.9 ± 0.3 | 1.7 ± 0.8 | 12.6 ± 6.2 | 0.40 ± 0.03 | 14.9 ± 2.6 |
| Timolol | 6.3 ± 0.004 | 1.6 ± 0.2 | 5.1 ± 1.5 | 29.2 ± 8.7 | 1.2 ± 0.2 | 7.4 ± 1.4 |
| [C2MIM][BF4] | [C4MIM][BF4] | [C6MIM][BF4] | ||||
|---|---|---|---|---|---|---|
| Compound | KAS | KAD | KAS | KAD | KAS | KAD |
| Acebutolol | 10.8 ± 0.5 | −8.0 ± 1.9 | 7.6 ± 0.6 | 9.3 ± 3.0 | 3.2 ± 0.06 | 14.7 ± 0.8 |
| Atenolol | 0.3 ± 0.06 | – | 0.3 ± 0.02 | 6.8 ± 2.4 | – | – |
| Carteolol | 2.60 ± 0.12 | −8.0 ± 1.7 | 1.8 ± 0.1 | 9.6 ± 3.0 | 0.6 ± 0.08 | 21.0 ± 5.5 |
| Celiprolol | 19.6 ± 0.8 | 3.5 ± 1.5 | 16.4 ± 1.1 | 1.9 ± 0.6 | 9.4 ± 0.3 | 30.7 ± 1.4 |
| Esmolol | 26.5 ± 1.9 | −4.3 ± 2.6 | 13.8 ± 0.9 | 1.4 ± 0.4 | 7.9 ± 0.3 | 19.4 ± 1.7 |
| Metoprolol | 14.1 ± 0.9 | −4.7 ± 2.5 | 7.4 ± 0.5 | 1.3 ± 0.4 | 3.8 ± 0.06 | 15.8 ± 0.7 |
| Nadolol | 3.4 ± 0.2 | −4.0 ± 2.4 | 1.7 ± 0.1 | 1.9 ± 0.4 | 0.6 ± 0.04 | 16.8 ± 2.8 |
| Oxprenolol | 37.0 ± 2.1 | −5.0 ± 2.0 | 23.8 ± 1.9 | 7.0 ± 3.0 | 10.8 ± 0.25 | 16.7 ± 0.9 |
| Pindolol | 4.4 ±10.5 | −12.0 ± 1.3 | 3.2 ± 0.2 | 5.5 ± 2.9 | – | – |
| Timolol | 11.5 ± 0.8 | −4.8 ± 2.5 | 6.1 ± 0.3 | 1.7 ± 2.0 | 3.2 ± 0.1 | 20.6 ± 1.5 |
| TEA | DMOA | |||
|---|---|---|---|---|
| Compound | KAS | KAD | KAS | KAD |
| Acebutolol | 6.9 ± 1.3 | 5.2 ± 2.1 | 2.1 ± 0.6 | 284 ± 85 |
| Atenolol | 0.3 ± 0.1 | −2.3 ± 0.5 | – | – |
| Carteolol | 1.3 ± 0.9 | −1.5 ± 0.9 | 0.300 ± 0.003 | 16.4 ± 0.4 |
| Celiprolol | 15.6 ± 7.8 | −2.0 ± 0.1 | 5.6 ± 1.0 | 198 ± 37 |
| Esmolol | 2.7 ± 1.7 | −1.6 ± 0.9 | 2.2 ± 0.5 | 4185 ± 707 |
| Metoprolol | 1.6 ± 0.3 | −0.2 ± 0.4 | 2.5 ± 0.6 | 241 ± 56 |
| Nadolol | 1.6 ± 0.2 | −1.6 ± 0.9 | 0.30 ± 0.03 | 12.8 ± 3.9 |
| Oxprenolol | 18.7 ± 0.3 | −1.8 ± 0.1 | 6.5 ± 0.9 | 173 ± 26 |
| Pindolol | 2.9 ± 0.5 | 6.3 ± 2.2 | 1.0 ± 0.4 | 557 ± 22 |
| Timolol | 5.6 ± 2.9 | −1.9 ± 1.0 | 2.0 ± 0.5 | 240 ± 55 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Ángel, M.J. Estimation of Solute-Stationary Phase and Solute-Mobile Phase Interactions in the Presence of Ionic Liquids. Separations 2019, 6, 40. https://doi.org/10.3390/separations6030040
Ruiz-Ángel MJ. Estimation of Solute-Stationary Phase and Solute-Mobile Phase Interactions in the Presence of Ionic Liquids. Separations. 2019; 6(3):40. https://doi.org/10.3390/separations6030040
Chicago/Turabian StyleRuiz-Ángel, María José. 2019. "Estimation of Solute-Stationary Phase and Solute-Mobile Phase Interactions in the Presence of Ionic Liquids" Separations 6, no. 3: 40. https://doi.org/10.3390/separations6030040
APA StyleRuiz-Ángel, M. J. (2019). Estimation of Solute-Stationary Phase and Solute-Mobile Phase Interactions in the Presence of Ionic Liquids. Separations, 6(3), 40. https://doi.org/10.3390/separations6030040




