Biogenic Amines in Traditional Fiore Sardo PDO Sheep Cheese: Assessment, Validation and Application of an RP-HPLC-DAD-UV Method
Abstract
:1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Chemicals
2.3. Equipment
2.4. Biogenic Amine Analysis
2.4.1. Exctraction and Derivatisation
2.4.2. HPLC Separation
2.4.3. Wavelength Optimization
2.4.4. Identification and Quantification of the Biogenic Amines
2.4.5. Analytical Method Validation
2.5. Statistical Analysis
3. Results and Discussion
3.1. Assessment of the Chromatographic Method
3.2. Validation Parameters
3.3. Biogenic Amines in Fiore Sardo Sheep Cheese
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kelly, M.T.; Blaise, A.; Larroque, M. Rapid automated high performance liquid chromatography method for simultaneous determination of amino acids and biogenic amines in wine, fruit and honey. J. Chromatogr. A 2010, 1217, 7385–7392. [Google Scholar] [CrossRef]
- Mayer, H.K.; Fiechter, G.; Fischer, E. A new ultra-pressure liquid chromatography method for the determination of biogenic amines in cheese. J. Chromatogr. A 2010, 1217, 3251–3257. [Google Scholar] [CrossRef]
- Chong, C.Y.; Abu Bakar, F.; Russly, A.R.; Jamilah, B.; Mahyudin, N.A. The effects of food processing on biogenic amines formation. Int. Food Res. J. 2011, 18, 867–876. [Google Scholar]
- Linares, D.M.; del Rio, B.; Redruello, B.; Ladero, V.; Martin, M.C.; Fernandez, M.; Ruas-Madiedo, P.; Alvarez, M.A. Comparative analysis of the in vitro cytotoxicity of the dietary biogenic amines tyramine and histamine. Food Chem. 2016, 197, 658–663. [Google Scholar] [CrossRef]
- McCabe, B.J.; Wolfe, J.J.; Frankel, E.H. Handbook of Food-Drug Interactions; Taylor & Francis: Oxfordshire, UK, 2003; ISBN 9780849315312. [Google Scholar]
- Mohammed, G.I.; Bashammakh, A.S.; Alsibaai, A.A.; Alwael, H.; El-Shahawi, M.S. A critical overview on the chemistry, clean-up and recent advances in analysis of biogenic amines in foodstuffs. Trends Anal. Chem. 2016, 78, 84–94. [Google Scholar] [CrossRef]
- Vale, S.R.; Glória, M.B. Determination of biogenic amines in cheese. J. AOAC Int. 1997, 80, 1006–1012. [Google Scholar] [PubMed]
- Combarros-Fuertes, P.; Fernandez, D.; Arenas, R.; Diezhandino, I.; Tornadijo, M.E.; Fresno, J.M. Biogenic amines in Zamorano cheese: Factors involved in their accumulation. J. Sci. Food Agric. 2016, 96, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Ramani, D.; De Bandt, J.P.; Cynober, L. Aliphatic polyamines in physiology and diseases. Clin. Nutr. 2014, 33, 14–22. [Google Scholar] [CrossRef]
- Wang, C.; Ruan, P.; Zhao, Y.; Li, X.; Wang, J.; Wu, X.; Liu, T.; Wang, S.; Hou, J.; Li, W.; et al. Spermidine/spermine N(1)-acetyltransferase regulates cell growth and metastasis via AKT/β-catenin signaling pathways in hepatocellular and colorectal carcinoma cells. Oncotarget 2017, 8, 1092–1109. [Google Scholar] [CrossRef]
- Gosetti, F.; Mazzucco, E.; Gianotti, V.; Polati, S.; Gennaro, M.C. High performance liquid chromatography/tandem mass spectrometry determination of biogenic amines in typical Piedmont cheeses. J. Chromatogr. A 2007, 1149, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Innocente, N.; Biasutti, M.; Padovese, M.; Moret, S. Determination of biogenic amines in cheese using HPLC technique and direct derivatization of acid extract. Food Chem. 2007, 101, 1285–1289. [Google Scholar] [CrossRef]
- Linares, D.M.; Martin, M.M.; Ladero, V.; Alvarez, M.A.; Fernandez, M. Biogenic amines in dairy products. Crit. Rev. Food Sci. Nutr. 2011, 51, 691–703. [Google Scholar] [CrossRef]
- Švarc-Gajic, J.; Stojanovic, Z. Determination of histamine in cheese by chronopotentiometry on a thin film mercury electrode. Food Chem. 2011, 124, 1172–1176. [Google Scholar] [CrossRef]
- Ruiz-Capillas, C.; Moral, A. Production of biogenic amines and their potential use as quality control indices for hake (Merluccius merluccius, L.) stored in ice. J. Food Sci. 2001, 66, 1030–1032. [Google Scholar] [CrossRef]
- Sagratini, G.; Fernandez-Franzon, M.; Berardinis, F.D.; Font, G.; Vittori, S.; Manes, J. Simultaneous determination of eight underivatised biogenic amines in fish by solid phase extraction and liquid chromatography–tandem mass spectrometry. Food Chem. 2012, 132, 537–543. [Google Scholar] [CrossRef] [PubMed]
- Cohen, G.; Rudnik, D.D.; Laloush, M.; Yakir, D.; Karpas, Z. A Novel method for determination of histamine in tuna fish by ion mobility spectrometry. Food Anal. Methods 2015, 8, 2376–2382. [Google Scholar] [CrossRef]
- Draisci, R.; Volpe, P.G.; Lucentini, O.L.; Cecilia, A.; Federico, R.; Palleschi, G. Determination of biogenic amines with an electrochemical biosensor and its application to salted anchovies. Food Chem. 1998, 62, 225–232. [Google Scholar] [CrossRef]
- Bilgin, B.; Gençcelep, H. Determination of biogenic amines in fish products. Food Sci. Biotechnol. 2015, 24, 1907–1913. [Google Scholar] [CrossRef]
- Hernandez-Jover, T.; Izquierdo-Pulido, M.; Veciana-Nogués, M.T.; Vidal-Carou, M.C. Ion-pair high-performance liquid chromatographic determination of biogenic amines in meat and meat products. J. Agric. Food Chem. 1996, 44, 2710–2715. [Google Scholar] [CrossRef]
- Saccani, G.; Tanzi, E.; Pastore, P.; Cavalli, S.; Rey, M. Determination of biogenic amines in fresh and processed meat by suppressed ion chromatography-mass spectrometry using a cation-exchange column. J. Chromatogr. A 2005, 1082, 43–50. [Google Scholar] [CrossRef]
- Stojanovi, Z.S.; Svarc-Gaji, J.V. A simple and rapid method for histamine determination in fermented sausages by mediated chronopotentiometry. Food Contr. 2011, 22, 2013–2019. [Google Scholar] [CrossRef]
- Liu, M.; Li, Y.G.; Chou, G.X.; Cheng, X.M.; Zhang, M.; Wang, Z.T. Extraction and ultra-performance liquid chromatography of hydrophilic and lipophilic bioactive components in a Chinese herb Radix Salviae Miltiorrhizae. J. Chromatogr. A 2007, 1157, 51–55. [Google Scholar] [CrossRef] [PubMed]
- Moret, S.; Smela, D.; Populin, T.; Conte, L.S. A survey on free biogenic amine content of fresh and preserved vegetables. Food Chem. 2005, 89, 355–361. [Google Scholar] [CrossRef]
- Pastore, P.; Favaro, G.; Badacco, D.; Tapparo, A.; Cavalli, C.; Saccani, G. Determination of biogenic amines in chocolate by ion chromatographic separation and pulsed integrated amperometric detection with implemented wave-form at Au disposable electrode. J. Chromatogr. A 2005, 1098, 111–115. [Google Scholar] [CrossRef]
- Pena-Gallego, A.; Hernndez-Orte, P.; Cacho, J.; Ferreira, V. Biogenic amine determination in wines using solid-phase extraction: A comparative study. J. Chromatogr. A 2009, 1216, 3398–3401. [Google Scholar] [CrossRef] [PubMed]
- Beneduce, L.; Romano, A.; Capozzi, V.; Lucas, P.; Barnavon, L.; Bach, B.; Vuchot, F.; Grieco, G.; Spano, E. Biogenic amine in wines. Ann. Microbiol. 2010, 60, 573–578. [Google Scholar] [CrossRef]
- Pena-Gallego, A.; Hernandez-Orte, P.; Cacho, J.; Ferreira, A.V. High-Performance Liquid Chromatography Analysis of Amines in Must and Wine: A Review. Food Rev. Int. 2012, 28, 71–96. [Google Scholar] [CrossRef]
- Daniel, D.; dos Santos, V.B.; Vidal, D.T.R.; do Lago, C.L. Determination of biogenic amines in beer and wine by capillary electrophoresis–tandem mass spectrometry. J. Chromatogr. A 2015, 1416, 121–128. [Google Scholar] [CrossRef]
- Tuberoso, C.I.G.; Congiu, F.; Serreli, G.; Mameli, S. Determination of dansylated amino acids and biogenic amines in Cannonau and Vermentino wines by HPLC-FLD. Food Chem. 2015, 175, 29–35. [Google Scholar] [CrossRef]
- Huang, K.-L.; Jin, C.-X.; Song, S.-L.; Wei, C.-Y.; Liu, Y.-M.; Li, J. Development of an ionic liquid-based ultrasonic-assisted liquid-liquid microextraction method for sensitive determination of biogenic amines: application to the analysis of octopamine, tyramine and phenethylamine in beer samples. J. Chromatogr. B 2011, 879, 579–584. [Google Scholar] [CrossRef]
- Stratton, J.E.; Hutkins, R.W.; Taylor, S.L. Biogenic amines in cheese and other fermented foods: A review. J. Food Prot. 1991, 54, 460–470. [Google Scholar] [CrossRef]
- Rak, L. Biogenic amines in dairy products. Med. Weter. 2005, 61, 391–393. [Google Scholar]
- EFSA Panel on Biological Hazards (BIOHAZ). Scientific opinion on risk based control of biogenic amine formation in fermented foods. EFSA J. 2011, 9, 2393. [Google Scholar] [CrossRef]
- Linares, D.M.; Del Río, B.; Ladero, V.; Martinez, N.; Fernandez, M.; Martín, M.C.; Álvarez, M.A. Factors influencing biogenic amines accumulation in dairy products. Front. Microbiol. 2012, 3, 180. [Google Scholar] [CrossRef]
- Bonczar, G.; Filipczak-Fiutak, M.; Pluta-Kubica, A.; Duda, I. Biogenic amines present in cheese—Occurrence and threats. Med. Weter. 2017, 73, 136–143. [Google Scholar] [CrossRef]
- Novella-Rodríguez, S.; Veciana-Nogués, M.T.; Roig-Sagués, A.X.; Trujillo-Mesa, A.J.; Vidal-Carou, M.C. Influence of starter and nonstarter on the formation of biogenic amine in goat cheese during ripening. J. Dairy Sci. 2002, 85, 2471–2478. [Google Scholar] [CrossRef]
- Fernández, M.; Linares, D.M.; del Rio, B.; Ladero, V.; Álvarez, M.A. HPLC quantification of biogenic amines in cheeses: correlation with PCR-detection of tyramine-producing microorganisms. J. Dairy Res. 2007, 74, 276–282. [Google Scholar] [CrossRef] [PubMed]
- Naila, A.; Flint, S.; Fletcher, G.; Bremer, P.; Meerdink, G. Control of biogenic amines in food-existing and emerging approaches. J. Food Sci. 2010, 75, 139–150. [Google Scholar] [CrossRef]
- Innocente, N.; D’Agostin, P. Formation of Biogenic Amines in a Typical Semihard Italian Cheese. J. Food Prot. 2002, 65, 1498–1501. [Google Scholar] [CrossRef]
- Gardini, F.; Martuscelli, M.; Caruso, M.C.; Galgano, F.; Crudele, M.A.; Favati, F.; Guerzoni, M.E.; Suzzi, G. Effect of pH, temperature and NaCl concentration on the growth kinetic, proteolytic activity and biogenic amines production of Enterococcus faecalis. Int. J. Food Microbiol. 2001, 64, 105–117. [Google Scholar] [CrossRef]
- Fernández-García, E.; Tomillo, E.J.; Nuñez, M. Formation of biogenic amines in raw milk Hispanico cheese manufactured with proteinases and different levels of starter culture. J. Food Prot. 2000, 63, 1551–1555. [Google Scholar] [CrossRef] [PubMed]
- Bunková, L.; Bunka, F.; Mantlová, G.; Cablová, A.; Sedlácek, I.; Švec, P.; Pachlová, V.; Krácmar, S. The effect of ripening and storage conditions on the distribution of tyramine, putrescine and cadaverine in Edam-cheese. Food Microbiol. 2010, 27, 880–888. [Google Scholar] [CrossRef]
- Custódio, F.B.; Tavares, E.; Glória, M.B. Extraction of bioactive amines from grated Parmesan cheese using acid, alkaline and organic solvents. J. Food Composit. Anal. 2007, 20, 280–288. [Google Scholar] [CrossRef]
- Ladero, V.; Fernández, M.; Álvarez, M.A. Effect of post-ripening processing on the histamine and histamine-producing bacteria contents of different cheeses. Int. Dairy J. 2009, 19, 759–762. [Google Scholar] [CrossRef]
- Fernández, M.; Linares, D.M.; Rodríguez, A.; Álvarez, M.A. Factors affecting tyramine production in Enterococcus durans IPLA655. Appl. Microbiol. Biotechnol. 2007, 73, 1400–1406. [Google Scholar] [CrossRef]
- Pinho, O.; Ferreira, I.M.; Mendes, E.; Oliveira, B.M.; Ferreira, M. Effect of temperature on evolution of free amino acid and biogenic amine contents during storage of Azeitão cheese. Food Chem. 2001, 75, 287–291. [Google Scholar] [CrossRef]
- Mercogliano, R.; De Felice, A.; Chirollo, C.; Cortesi, M.L. Production of vasoactive amines during the ripening of Pecorino Carmasciano cheese. Vet. Res. Commun. 2010, 34, S175–S178. [Google Scholar] [CrossRef]
- Schirone, M.; Tofalo, R.; Mazzone, G.; Corsetti, A.; Suzzi, G. Biogenic amine content and microbiological profile of Pecorino di Farindola cheese. Food Microbiol. 2011, 28, 128–136. [Google Scholar] [CrossRef]
- Novella-Rodríguez, S.; Veciana-Nogués, M.T.; Izquerdo-Pulido, M.; Vidal-Carou, M.C. Distribution of biogenic amines and polyamines in cheese. J. Food Sci. 2003, 68, 750–755. [Google Scholar] [CrossRef]
- Rabie, M.A.; Elsaidy, S.; el-Badawy, A.A.; Siliha, H.; Malcata, F.X. Biogenic amine contents in selected Egyptian fermented foods as determined by ion-exchange chromatography. J. Food Prot. 2011, 74, 681–685. [Google Scholar] [CrossRef]
- Erim, F.B. Recent analytical approaches to the analysis of biogenic amines in food samples. Trends Anal. Chem. 2013, 52, 239–247. [Google Scholar] [CrossRef]
- Papageorgiou, M.; Lambropoulou, D.; Morrison, C.; Kłodzińska, E.; Namieśnik, J.; Płotka-Wasylka, J. Literature update of analytical methods for biogenic amines determination in food and beverages. Trends Anal. Chem. 2018, 98, 128–142. [Google Scholar] [CrossRef]
- Moret, S.; Conte, L.S. High-performance liquid chromatographic evaluation of biogenic amines in foods an analysis of different methods of sample preparation in relation to food characteristics. J. Chromatogr. A 1996, 729, 363–369. [Google Scholar] [CrossRef]
- Dadáková, E.; Křížek, M.; Pelikánová, T. Determination of biogenic amines in foods using ultra-performance liquid chromatography (UPLC). Food Chem. 2009, 116, 365–370. [Google Scholar] [CrossRef]
- Jia, S.; Kang, Y.P.; Park, J.H.; Lee, J.; Kwon, S.W. Simultaneous determination of 23 amino acids and 7 biogenic amines in fermented food samples by liquid chromatography/quadrupole time-of-flight mass spectrometry. J. Chromatogr. A 2011, 1218, 9174–9182. [Google Scholar] [CrossRef] [PubMed]
- Gianotti, V.; Chiuminatto, U.; Mazzucco, E.; Gosetti, F.; Bottaro, M.; Frascarolo, P.; Gennaro, M.C. A new hydrophilic interaction liquid chromatography tandem mass spectrometry method for the simultaneous determination of seven biogenic amines in cheese. J. Chromatogr. A 2008, 1185, 296–300. [Google Scholar] [CrossRef] [PubMed]
- Palermo, C.; Muscarella, M.; Nardiello, D.; Iammarino, M.; Centonze, D. A multiresidual method based on ion-exchange chromatography with conductivity detection for the determination of biogenic amines in food and beverages. Anal. Bioanal. Chem. 2013, 405, 1015–1023. [Google Scholar] [CrossRef] [PubMed]
- Draisci, R.; Giannetti, L.; Boria, P.; Lucentini, L.; Palleschi, L.; Cavalli, S. Improved ion chromatography-integrated pulsed amperometric detection method for the evaluation of biogenic amines in food of vegetable or animal origin and in fermented foods. J. Chromatogr. A 1998, 798, 109–116. [Google Scholar] [CrossRef]
- Ali Awan, M.; Fleet, I.; Paul Thomas, C.L. Determination of biogenic diamines with a vaporisation derivatisation approach using solid-phase microextraction gas chromatography-mass spectrometry. Food Chem. 2008, 111, 462–468. [Google Scholar] [CrossRef]
- Aygün, O.; Schneider, E.; Scheuer, R.; Usleber, E.; Gareis, M.; Märtlbauer, E. Comparison of ELISA and HPLC for the determination of histamine in cheese. J. Agric. Food Chem. 1999, 47, 1961–1964. [Google Scholar] [CrossRef]
- Numanoǧlu, E.; Boyaci, I.H.; Topcu, A. Simple determination of histamine in cheese by capillary electrophoresis with diode array detection. J. Food Drug Anal. 2008, 16, 74–80. [Google Scholar]
- Calvo-Pérez, A.; Domínguez-Renedo, O.; Alonso-Lomillo, M.A.; Arcos-Martínez, M.J. Disposable amperometric biosensor for the determination of tyramine using plasma amino oxidase. Microchim. Acta 2013, 180, 253–259. [Google Scholar] [CrossRef]
- Costa, D.J.E.; Martínez, A.M.; Ribeiro, W.F.; Bichinho, K.M.; Di Nezio, M.S.; Pistonesi, M.F.; Araujo, M.C.U. Determination of Tryptamine in foods using square wave adsorptive stripping voltammetry. Talanta 2016, 154, 134–140. [Google Scholar] [CrossRef]
- Addis, M.; Pirisi, A.; Di Salvo, R.; Podda, F.; Piredda, G. The influence of the enzymatic composition of lamb rennet paste on some properties of experimentally produced PDO Fiore Sardo cheese. Int. Dairy J. 2005, 15, 1271–1278. [Google Scholar] [CrossRef]
- Pirisi, A.; Pinna, G.; Addis, M.; Piredda, G.; Mauriello, R.; De Pascale, S.; Caira, S.; Mamone, G.; Ferranti, P.; Addeo, F.; et al. Relationship between the enzymatic composition of lamb rennet paste and proteolytic, lipolytic pattern and texture of PDO Fiore Sardo ovine cheese. Int. Dairy J. 2007, 17, 143–156. [Google Scholar] [CrossRef]
- Mannu, L.; Comunian, R.; Scintu, M.F. Mesophilic lactobacilli in Fiore Sardo cheese: PCR-identification and evolution during cheese ripening. Int. Dairy J. 2000, 10, 383–389. [Google Scholar] [CrossRef]
- Pisano, M.B.; Fadda, M.E.; Deplano, M.; Corda, A.; Cosentino, S. Microbiological and chemical characterization of Fiore Sardo, a traditional Sardinian cheese made from ewe’s milk. Int. J. Dairy Technol. 2006, 59, 171–179. [Google Scholar] [CrossRef]
- Mangia, N.P.; Murgia, M.A.; Garau, G.; Sanna, M.G.; Deiana, P. Influence of selected lab cultures on the evolution of free amino acids, free fatty acids and Fiore Sardo cheese microflora during the ripening. Food Microbiol. 2008, 2, 366–377. [Google Scholar] [CrossRef]
- Comunian, R.; Paba, A.; Daga, E.; Dupré, I.; Scintu, M.F. Traditional and innovative production methods of Fiore Sardo cheese: a comparison of microflora with a PCR-culture technique. Int. J. Dairy Technol. 2010, 63, 224–233. [Google Scholar] [CrossRef]
- Manca, G.; Porcu, A.; Ru, A.; Salaris, M.; Franco, M.A.; de Santis, E.P.L. Comparison of gamma-aminobutyric acid and biogenic amine content of different types of ewe’s milk cheese produced in Sardinia, Italy. Ital. J. Food Saf. 2015, 4, 123–128. [Google Scholar] [CrossRef]
- Urgeghe, P.P.; Piga, C.; Addis, M.; Di Salvo, R.; Piredda, G.; Scintu, M.F.; Wolf, I.V.; Sanna, G. SPME/GC-MS Characterization of the Volatile Fraction of an Italian PDO Sheep Cheese to Prevalent Lypolitic Ripening: The Case of Fiore Sardo. Food Anal. Methods 2012, 5, 723–730. [Google Scholar] [CrossRef]
- Pulinas, L.; Spanu, C.; Idda, I.; Ibba, I.; Nieddu, G.; Virdis, S.; Scarano, C.; Piras, F.; Spano, N.; Sanna, G.; et al. Farmstead production of artisan lactose-free Pecorino di Osilo cheese and Ricotta cheeses from sheep’s milk. Ital. J. Food Saf. 2017, 6, 6353. [Google Scholar] [CrossRef] [PubMed]
- Idda, I.; Spano, N.; Addis, M.; Galistu, G.; Ibba, I.; Nurchi, V.M.; Pilo, M.I.; Scintu, M.F.; Piredda, G.; Sanna, G. Optimization of a newly established gas-chromatographic method for determining lactose and galactose traces: Application to Pecorino Romano cheese. J. Food Composit. Anal. 2018, 74, 89–94. [Google Scholar] [CrossRef]
- Mocak, J.; Bond, A.M.; Mitchell, S.; Schollary, G. A statistical overview of standard (IUPAC and ACS) and new procedures for determining the limits of detection and quantification: Application to voltammetric and stripping techniques. Pure Appl. Chem. 1997, 69, 297–328. [Google Scholar] [CrossRef]
- Horwitz, W. Evaluation of Analytical Methods Used for Regulation of Foods and Drugs. Anal. Chem. 1982, 54, 67A–76A. [Google Scholar] [CrossRef]
- Torracca, B.; Pedonese, F.; Turchi, B.; Fratini, F.; Nuvoloni, R. Qualitative and quantitative evaluation of biogenic amines in vitro production by bacteria isolated from ewes’ milk cheeses. Eur. Food Res. Technol. 2018, 244, 721–728. [Google Scholar] [CrossRef]
- Moret, S.; Bortolomeazzi, R.; Lercker, G. Improvement of extraction procedure for biogenic amines in foods and their high-performance liquid chromatographic determination. J. Chromatogr. A 1992, 591, 175–180. [Google Scholar] [CrossRef]
- Martuscelli, M.; Gardini, F.; Torriani, S.; Mastrocola, D.; Serio, A.; Chaves-Lòpez, C.; Schirone, M.; Suzzi, G. Production of biogenic amines during the ripening of Pecorino Abruzzese cheese. Int. Dairy J. 2005, 15, 571–578. [Google Scholar] [CrossRef]
- Torracca, B.; Nuvoloni, R.; Ducci, M.; Bacci, C.; Pedonese, F. Biogenic Amines Content of Four Types of “Pecorino” Cheese Manufactured in Tuscany. Int. J. Food Prop. 2015, 18, 999–1005. [Google Scholar] [CrossRef]
- Valsamaki, K.; Michaelidou, A.; Polychroniadou, A. Biogenic amine production in Feta cheese. Food Chem. 2000, 71, 259–266. [Google Scholar] [CrossRef]
- Gaya, P.; Sánchez, C.; Nuñez, M.; Fernández-García, E. Proteolysis during ripening of Manchego cheese made from raw or pasteurized ewes’ milk. Seasonal variation. J. Dairy Res. 2005, 72, 287–295. [Google Scholar] [CrossRef] [PubMed]
- Contreras, M.; Izquierdo, P.; Allara, M.; García, A.; Torres, G.; Céspedes, E. Determinación de aminas biógenas en quesos madurados. Rev. Cient. FCV-LUZ 2007, 17, 89–95. [Google Scholar]
- Poveda, J.M.; Chicón, R.; Cabezas, L. Biogenic amine content and proteolysis in Manchego cheese manufactured with Lactobacillus paracasei subsp. paracasei as adjunct and other autochthonous strains as starters. Int. Dairy J. 2015, 47, 94–101. [Google Scholar] [CrossRef]
- Lanciotti, R.; Patrignani, F.; Iucci, L.; Guerzoni, M.E.; Suzzi, G.; Belletti, N.; Gardini, F. Effects of milk high pressure homogenization on biogenic amine accumulation during ripening of ovine and bovine Italian cheeses. Food Chem. 2007, 104, 693–701. [Google Scholar] [CrossRef]
- Pintado, A.I.E.; Pinho, O.; Ferreira, I.M.P.L.V.O.; Pintado, M.M.E.; Gomes, A.M.P.; Malcata, F.X. Microbiological, biochemical and biogenic amine profiles of Terrincho cheese manufactured in several dairy farms. Int. Dairy J. 2008, 18, 631–640. [Google Scholar] [CrossRef]
- Mascaro, N.; Stocchi, R.; Ricciutelli, M.; Cammertoni, N.; Renzi, F.; Cecchini, S.; Loschi, A.R.; Rea, S. Biogenic amine content and chemical and physical features of italian formaggio di Fossa. Ital. J. Food Safety 2010, 8, 49–53. [Google Scholar] [CrossRef]
- Buňková, L.; Adamcová, G.; Hudcová, K.; Velichová, H.; Pachlová, V.; Lorencová, E.; Buňka, F. Monitoring of biogenic amines in cheeses manufactured at small-scale farms and in fermented dairy products in the Czech Republic. Food Chem. 2013, 141, 548–551. [Google Scholar] [CrossRef] [PubMed]
- Calzada, J.; Del Olmo, A.; Picon, A.; Gaya, P.; Nuñez, M. Proteolysis and biogenic amine buildup in high-pressure treated ovine milk blue-veined cheese. J. Dairy Sci. 2013, 96, 4816–4829. [Google Scholar] [CrossRef] [PubMed]
- Schirone, M.; Tofalo, R.; Fasoli, G.; Perpetuini, G.; Corsetti, A.; Manetta, A.C.; Ciarrocchi, A.; Suzzi, G. High content of biogenic amines in Pecorino cheeses. Food Microbiol. 2013, 34, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Pirisi, A.; Comunian, R.; Urgeghe, P.P.; Scintu, M.F. Sheep’s and goat’s dairy products in Italy: technological, chemical, microbiological, and sensory aspects. Small Rumin. Res. 2011, 101, 102–112. [Google Scholar] [CrossRef]
- Halász, S.; Bárath, A.; Simon-Sarkadi, L.; Holzapfel, W. Biogenic amines and their production by microorganism in food. Trends Food Sci. Technol. 1994, 5, 42–49. [Google Scholar] [CrossRef]
- Schirone, M.; Tofalo, R.; Visciano, P.; Corsetti, A.; Suzzi, G. Biogenic Amines in Italian Pecorino Cheese. Front. Microbiol. 2012, 3, 171. [Google Scholar] [CrossRef] [PubMed]
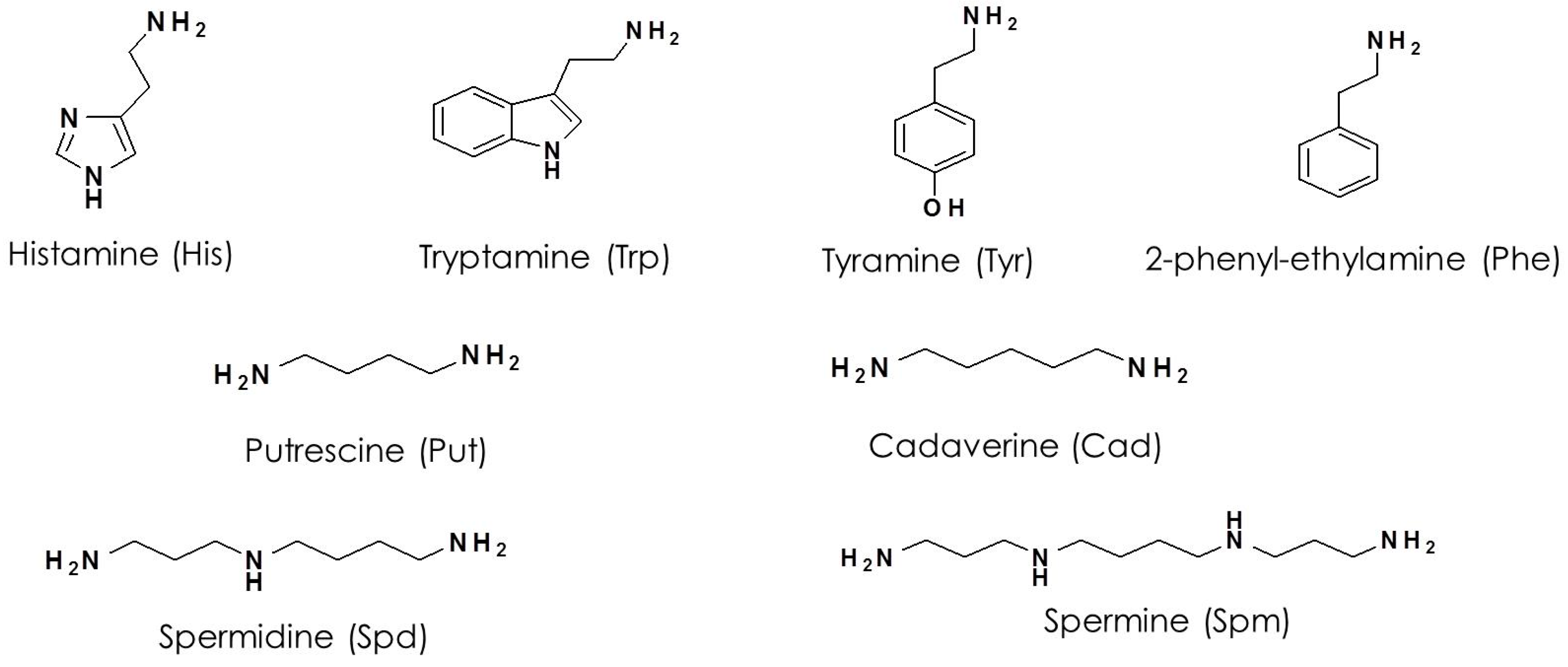
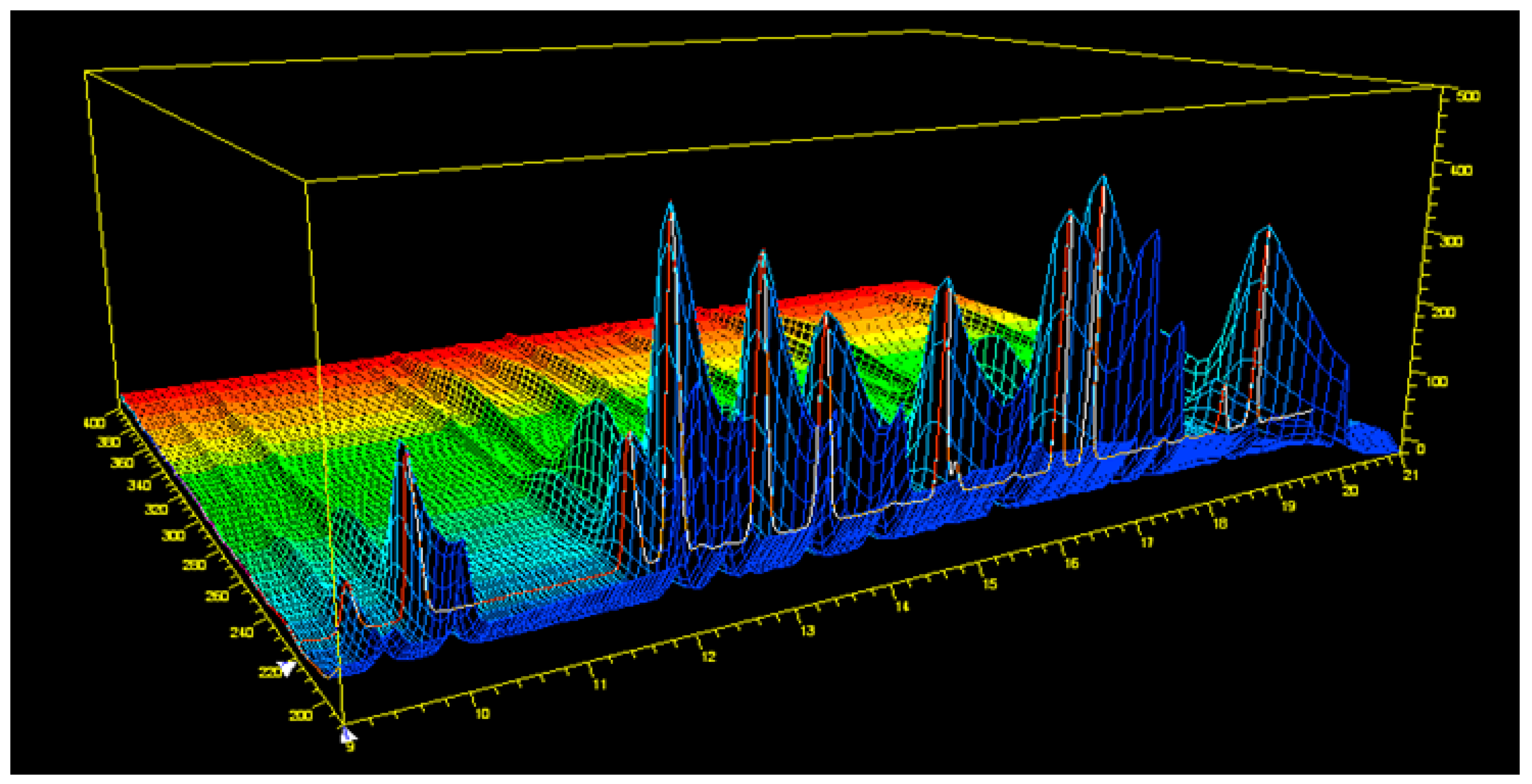
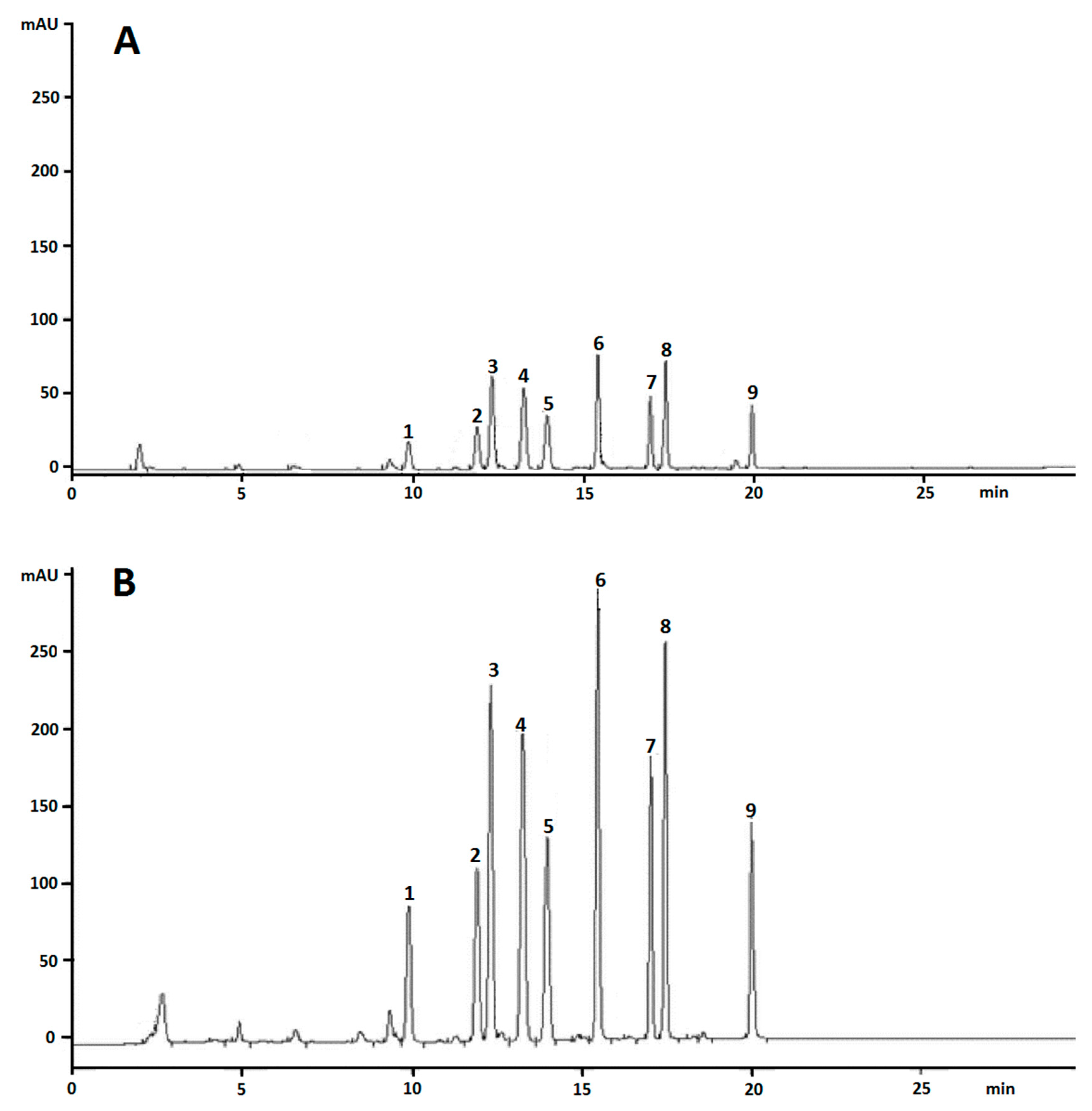
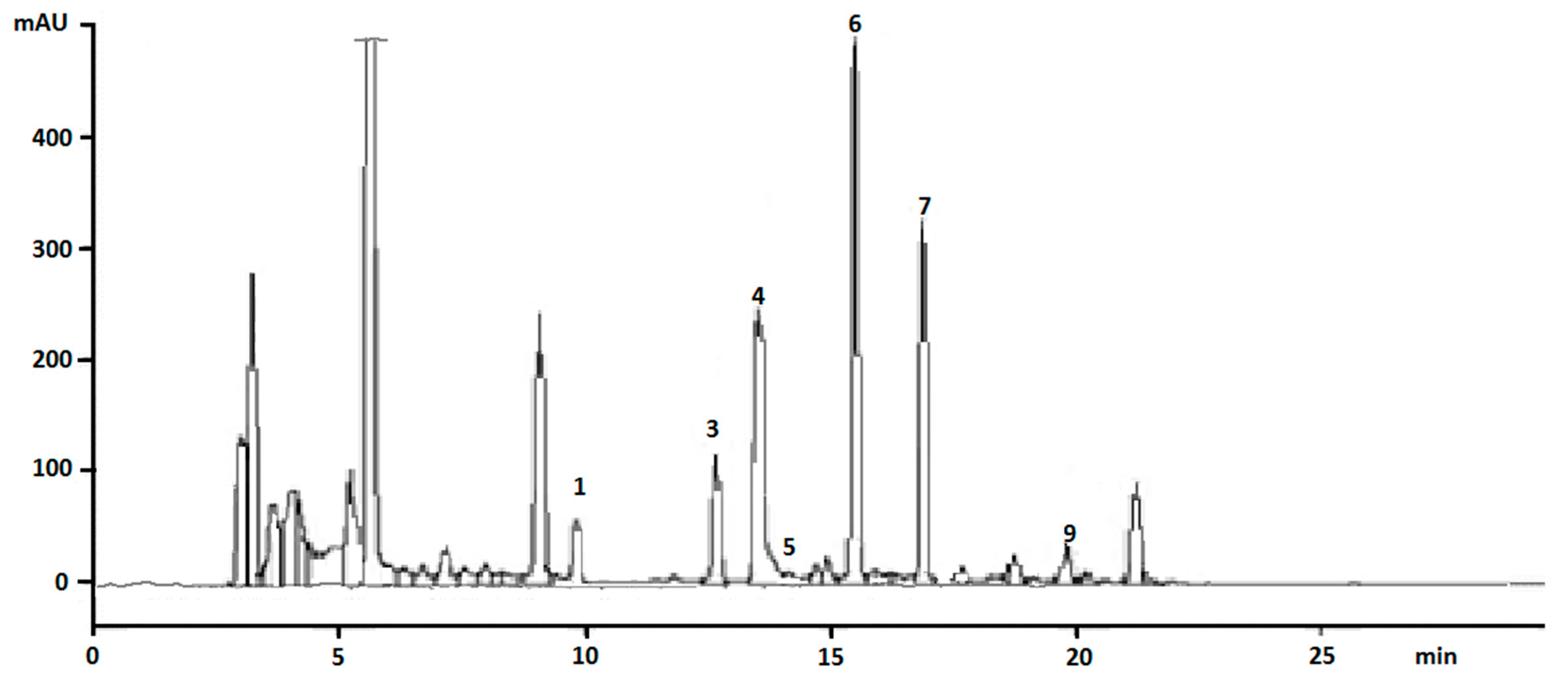
| Time (min) | Acetonitrile (%) | Water (%) |
|---|---|---|
| 0 | 65 | 35 |
| 1 | 65 | 35 |
| 10 | 80 | 20 |
| 12 | 90 | 10 |
| 16 | 100 | 0 |
| 30 | 100 | 0 |
| Post run | ||
| 30.1 | 65 | 30 |
| 35 | 65 | 30 |
| Linearity | Precision | Trueness | ||||
|---|---|---|---|---|---|---|
| Analyte | LoD (mg/kg) | LoQ (mg/kg) | Linearity Range, mg/kg, (R2) | Repeatability CV% exper(r) (HorRatr) | Intermediate Precision CV% exper(IP) (HorRatIP) | Recovery (%±SD) |
| Trp | 0.20 | 0.60 | 0.60–100 (0.9997) 2.5–1250 (0.9993) | 6.5 (0.4) | 14.3 (0.9) | 99 ± 2 |
| Phe | 0.10 | 0.30 | 0.30–125 (0.9998) 2.5–1250 (0.9996) | 6.8 (0.4) | 10.7 (0.7) | 94 ± 2 |
| Put | 0.07 | 0.20 | 0.20–125 (0.9960) 2.5–1250 (0.9905) | 4.4 (0.3) | 7.0 (0.4) | 87 ± 9 |
| Cad | 0.13 | 0.40 | 0.40–250 (0.9997) 2.5–1250 (0.9949) | 4.1 (0.3) | 6.4 (0.4) | 95 ± 12 |
| His | 0.20 | 0. 60 | 0.60–500 (0.9994) 2.5–1250 (0.9994) | 3.8 (0.2) | 9.2 (0.6) | 85 ± 5 |
| Tyr | 0.17 | 0.50 | 0.50–125 (0.9985) 2.5–1250 (0.9979) | 5.0 (0.3) | 4.9 (0.3) | 90 ± 6 |
| Spd | 0.23 | 0.70 | 0.70–125 (0.9980) 2.5–1250 (0.9945) | - | - | - |
| Spm | 0.07 | 0.20 | 0.25–100 (0.9999) 2.5–1250 (0.9949) | 8.6 (0.5) | 16.1 (1.0) | 82 ± 8 |
| Stores | Trp | Phe | Put | Cad | ||||
| Mean ± sd | range | Mean ± sd | range | Mean ± sd | range | Mean ± sd | range | |
| A | 0.4 ± 0.4 | <0.2–1.2 | 2 b ± 2 | <0.3–6.3 | <0.2 c | 6 b ± 4 | 1.3–13 | |
| B | 3 ± 3 | 0.2–8.2 | 16 a ± 8 | 1.8–25 | 450 a ± 200 | 88–730 | 20 ab ± 30 | 1.0–94 |
| C | 4.4 ± 3.5 | 0.3–10.5 | 15 a ± 10 | 9.3–42 | 130 b ± 70 | 36–210 | 40 a ± 20 | 6.7–70 |
| D | 5.5 ± 6.5 | <0.2–16.9 | 9 ab ± 10 | 1.3–27 | 20 bc ± 20 | <0.2–49 | 40 a ± 25 | 7.3–83 |
| Total | 3 ± 8 | <0.2–16.9 | 10 ± 20 | <0.3–42 | 150 ± 200 | <0.2–730 | 30 ± 50 | 1.0–94 |
| Store Effect | ns | ** | *** | * | ||||
| Stores | His | Tyr | Spm | ΣBAs | ||||
| Mean ± sd | range | Mean ± sd | range | Mean ± sd | range | Mean ± sd | ||
| A | 4 c ± 6 | <0.65–19 | 60 c ± 70 | <0.5–180 | 100 a ± 30 | 8–160 | 170 ± 80 | |
| B | 85 b ± 75 | 5.2–250 | 400 b ± 100 | 170–530 | 30 b ± 30 | 10–88 | 1000 ± 200 | |
| C | 210 a ± 20 | 190–250 | 700 a ± 100 | 445–800 | 30 b ± 30 | 17–115 | 1100 ± 100 | |
| D | 10 c ± 10 | <0.65–33 | 300 b ± 300 | 110–770 | 50 b ± 10 | 29–65 | 500 ± 300 | |
| Total | 80 ± 80 | <0.65–250 | 350 ± 300 | <0.5–800 | 50 ± 60 | 8–160 | 700 ± 400 | |
| Store Effect | *** | *** | *** | |||||
| Sheep Cheese | Key Features | Trp | Phe | Put | Cad | His | Tyr | Spm | Spd | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| Zamorano, Spain | Cheese made from raw milk; BAs have been measured between 1 day and 10 months of ripening. Data are inferred by the Figure 8 of the study. | 3–50 | 3–120 | 10–190 | 5–35 | 1–55 | 1–85 | 0–20 | 0–115 | [8] |
| Azeitão cheese | Cheese made from raw milk using an exctract of Cardoon as rennet. The length of ripening is at least of 20 days. | NM | NM | ND–137 | 161–260 | 414–818 | 72.3–445 | 17.2–81.6 | NM | [47] |
| Pecorino Carmasciano, Italy | Cheese obtained from raw milk, coagulated at 40–45 °C with lamb curdle and ripened for 201 days. Data are inferred by the Figure 4 of the study. | NM | NM | 100 | 120 | 65.5 | 136.41 | 350 | 110 | [48] |
| Pecorino di Farindola, Italy | Cheese obtained from raw milk, coagulated at 30–35 °C with pig rennet and ripened for 90 days. | NM | 0.0–127.1 | 9.9–394.1 | 26.8–276.1 | 0.0–21.8 | 52.3–1171.3 | NM | 36.3–143.9 | [49] |
| (1) Pecorino Sardo PDO (2) Pecorino (3) Casu Marzu | (1) Pecorino Sardo PDO is semi-cooked cheese made from thermised sheep milk inoculated with a starter culture and coagulated with calf rennet, ripening time between 1 and 12 months. (2) Pecorino is semi-cooked cheese made from raw sheep milk without a starter culture and coagulated with calf rennet, ripening time between 2 and 12 months. (3) Casu Marzu is a sheep’s cheese produced with the use of larvae of the cheese fly Piophila casei. Ripened for 2–3 months in warmer rooms. | (1) ND–9.3 (2) ND–13.5 (3) ND–41.8 | (1) ND (2) ND–8.6 (3) ND–90.9 | (1) 0.1–0.8 (2) ND–92.7 (3) 1.9–165.8 | (1) 0.1–9.7 (2) ND–137.0 (3) 3.1–470.7 | (1) ND–7.2 (2) ND–128.4 (3) ND–126 | (1) ND–19.3 (2) 1.6–93.0 (3) ND–231.4 | (1) ND–7.2 (2) ND (3) ND | (1) 0.2–5.4 (2) ND (3) ND | [71] |
| Pecorino Abruzzese, Italy | Two kinds of cheese: (a) raw milk, no starter culture; (b) Thermized milk with the addition of starter culture. Ripening time: 60 days.Data are inferred by the Figure 2 of the study. | (a) 25 a; 0–50 (b) 15 a; 10–20 | (a) 40 a; 35–45 (b) 300 a; 30–600 | (a) 60 a; 40–80 (b) 150 a; 40–250 | (a) 20 a; 10–35 (b) 50 a; 0–120 | (a) 270 a; 130–360 (b) 60 a; 30–100 | (a) 170 a; 20–330 (b) 240 a; 100–400 | (a) 30 a; 10–60 (b) 20 a 10–30 | (a) ND (b) ND | [79] |
| Pecorino Toscano, Italy | Four kinds of sheep’s cheeses, different each among other by the nature of thermal treatment (two from raw milk, two from pasteurized milk), the nature of the starter culture (two different types), the weight of the rind (between 1 and 2.4 kg) and the conditions and the length of the ripening. | 11–88 | 24–144 | 22–512 | 2–262 | ND–23 | 147–1132 | ND–2 | ND | [80] |
| Feta, Greece | Cheese from thermized milk constituted by 70% of sheep milk and 30% of goat milk, ripened and stored in brine. BAs have been measured during 4 months after production. | 4.39–5.74 | 3.51–4.94 | 1.62–193 | 0.27–82.8 | 2.40–84.6 | 0–246 | NM | NM | [81] |
| Manchego, Spain and Venezuela [82,83,84] | Cheese made from raw milk; BAs have been measured after 3 and 6 months of ripening Commercial samples, no details on the production phase Cheese made from raw milk; BAs have been measured between 3 and 8 months of ripening. | 3 months: <LoD–2.8 6 months: <LoD–15.3 53.78 a 44.22–63.35 ND | NM NM ND–49.8 | NM 104.66 a 101.05–108.28 215.4–668.3 | NM 52.48 a 38.25–66.72 289.9–803.1 | 3 months: 24.5–50.3 6 months: 54.1–229 98.93 a 76.70–121.18 60.0–100.1 | 3 months: 233–304.1 6 months: 402.4–533 43.10 a 38.57–47.62 109.4–326.8 | NM 110.36 a 93.05–127.67 NM | NM 120.22 a 117.40–123.05 NM | [82] [83] [84] |
| Pecorino, Italy | (a) high pressure homogenization of raw milk, (b) raw milk, (c) thermized milk. Ripening time: 21 days. | NM | (a) 19.5 (b) 63.3 (c) 155 | (a) 14.80 (b) 29.28 (c) 70.92 | (a) 20.3 (b) 107 (c) 257 | (a) 3.35 (b) 6.32 (c) 23.92 | (a) 62.8 (b) 162 (c) 350 | (a) 0 (b) 1.49 (c) 0 | (a) 10.4 (b) 9.03 (c) 15.9 | [85] |
| Terrincho; Portugal | Cheese obtained from raw milk, coagulated with calf rennet and ripened for 30 days. | 35.4–172.9 | 12.9–237.8 | 82.6–446.5 | 48.6–239.6 | 0.0–10.9 | 0.0–283.1 | NM | NM | [86] |
| Formaggio di fossa, Italy | Sheep cheese characterized by a ripening performed into pits dug in the sandstone for 90 days. | NM | 173.0 | 579.6 | 1302.86 | 24.11 | 461.62 | 0 | 16.49 | [87] |
| (1) Bryndza (2) Smoked cheese (3) Fresh cheese (4) Unripened (fresh) cheese (5) Pasta filata type cheese (6) Brined cheese (7) Flavored cheese | Cheeses 1–3 were from raw ewe’s milk, whereas cheeses 4–7 were from pasteurized ewe’s milk. | NM | NM | (1) ND–60.9 (2) 16.2–99.9 (3) ND–20.7 (4) ND–118.2 (5) ND (6) ND–229.5 (7) ND–108.8 | (1) ND–42.6 (2) ND–80.7 (3) ND–19.6 (4) ND–35.8 (5) ND (6) ND–125.6 (7) ND | (1) ND–24.2 (2) ND (3) ND (4) ND (5) ND (6) ND (7) ND | (1) 34.6–107.4 (2) 8.9–38.3 (3) ND (4) ND–11.1 (5) ND (6) 23.1–174.6 (7) ND–114.7 | (1) ND–9.7 (2) ND (3) ND (4) ND (5) ND–13.0 (6) ND–14.7 (7) ND | NM | [88] |
| Blue-veined cheese | Cheese made from pasteurized sheep’s milk and ripened for 6 to 12 months. | 61.8–71.11 | 13.25–61.44 | 17.28–33.46 | NM | NM | 7.15–52.20 | NM | NM | [89] |
| 12 cheeses from Abruzzo, Italy | (1) 3 cheeses were from sheep’s and cow’s milk, ripening time between 3 and 8 months. (2) 9 cheeses were only from sheep’s milk, ripening time between 3 and 10 months. | NM | (1) 26.0–232.4 (2) ND–44.4 | (1) 8.9–986.0 (2) ND–377.7 | (1) ND–2172.6 (2) ND–116.4 | (1) 200–743.3 (2) 10.3–761.4 | (1) 312.1–1771.3 (2) ND–702.4 | NM | NM | [90] |
| Fiore Sardo cheese, Italy | Cheese produced from raw milk, minimum ripening time: 3.5 months. | 3 a; <0.2–16.9 | 10 a; <0.3–42 | 150 a; <0.2–730 | 30 a; 1.0–94 | 80 a; <0.65–250 | 350 a; <0.5–800 | 50 a; 10–160 | <0.23 | This study |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zazzu, C.; Addis, M.; Caredda, M.; Scintu, M.F.; Piredda, G.; Sanna, G. Biogenic Amines in Traditional Fiore Sardo PDO Sheep Cheese: Assessment, Validation and Application of an RP-HPLC-DAD-UV Method. Separations 2019, 6, 11. https://doi.org/10.3390/separations6010011
Zazzu C, Addis M, Caredda M, Scintu MF, Piredda G, Sanna G. Biogenic Amines in Traditional Fiore Sardo PDO Sheep Cheese: Assessment, Validation and Application of an RP-HPLC-DAD-UV Method. Separations. 2019; 6(1):11. https://doi.org/10.3390/separations6010011
Chicago/Turabian StyleZazzu, Claudia, Margherita Addis, Marco Caredda, Maria Francesca Scintu, Giovanni Piredda, and Gavino Sanna. 2019. "Biogenic Amines in Traditional Fiore Sardo PDO Sheep Cheese: Assessment, Validation and Application of an RP-HPLC-DAD-UV Method" Separations 6, no. 1: 11. https://doi.org/10.3390/separations6010011
APA StyleZazzu, C., Addis, M., Caredda, M., Scintu, M. F., Piredda, G., & Sanna, G. (2019). Biogenic Amines in Traditional Fiore Sardo PDO Sheep Cheese: Assessment, Validation and Application of an RP-HPLC-DAD-UV Method. Separations, 6(1), 11. https://doi.org/10.3390/separations6010011






