Total and Free Fatty Acids Analysis in Milk and Dairy Fat
Abstract
1. Introduction
2. An Overview of FA Analysis
2.1. Lipid Extraction
2.2. Fractionation of Lipid Classes
3. FA Derivatization
4. Separation and Analysis of FA
4.1. Separation and Analysis of FAME from TFA
4.2. Separation and Analysis of FFA
5. Identification and Quantification of FA
6. Complementary Methods for Analysis and Quantification of FA
7. Conclusions
Author Contributions
Conflicts of Interest
References
- Jensen, R.G. The composition of bovine milk lipids: January 1995 to december 2000. J. Dairy Sci. 2002, 85, 295–350. [Google Scholar] [CrossRef]
- Christie, W.W. Composition and structure of milk lipids. In Advanced Dairy Chemistry, Volume 2. Lipids, 2nd ed.; Fox, P.F., Ed.; Chapman & Hall: London, UK, 1995; pp. 1–36. [Google Scholar]
- Rodriguez-Alcala, L.M.; Fontecha, J. Major lipid classes separation of buttermilk, and cows, goats and ewes milk by high performance liquid chromatography with an evaporative light scattering detector focused on the phospholipid fraction. J. Chromatogr. A 2010, 1217, 3063–3066. [Google Scholar] [CrossRef] [PubMed]
- Fox, P.F.; McSweeney, P.L.H. Advanced Dairy Chemistry. Volume 2. Lipids, 3rd ed.; Springer Science: New York, NY, USA, 2006. [Google Scholar]
- Schröder, M.; Vetter, W. Detection of 430 fatty acid methyl esters from a transesterified butter sample. J. Am. Oil Chem. Soc. 2013, 90, 771–790. [Google Scholar] [CrossRef]
- de la Fuente, M.A.; Juarez, M. Fatty Acids. In Handbook of Dairy Foods Analysis; Nollet, L.M.L., Toldrá, F., Eds.; CRC Press: Portland, OR, USA, 2009; pp. 211–228. [Google Scholar]
- Liu, Z.; Ezernieks, V.; Rochfort, S.; Cocks, B. Comparison of methylation methods for fatty acid analysis of milk fat. Food Chem. 2018, 261, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Warnakula, S.; Kunutsor, S.; Crowe, F.; Ward, H.A.; Johnson, L.; Franco, O.H.; Butterworth, A.S.; Forouhi, N.G.; Thompson, S.G.; et al. Association of dietary, circulating, and supplement fatty acids with coronary risk: A systematic review and meta-analysis. Ann. Intern. Med. 2014, 160, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Michas, G.; Micha, R.; Zampelas, A. Dietary fats and cardiovascular disease: Putting together the pieces of a complicated puzzle. Atherosclerosis 2014, 234, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Chavarri, F.; Bustamante, M.A.; Santisteban, A.; Virto, M.; Barron, L.J.R.; De Renobales, M. Changes in free fatty acids during ripening of Idiazabal cheese manufactured at different times of the year. J. Dairy Sci. 1999, 82, 885–890. [Google Scholar] [CrossRef]
- Mannion, D.T.; Furey, A.; Kilcawley, K.N. Comparison and validation of 2 analytical methods for the determination of free fatty acids in dairy products by gas chromatography with flame ionization detection. J. Dairy Sci. 2016, 99, 5047–5063. [Google Scholar] [CrossRef] [PubMed]
- Mannion, D.T.; Furey, A.; Kilcawley, K.N. Free fatty acids quantification in dairy products. Int. J. Dairy Technol. 2016, 69, 1–12. [Google Scholar] [CrossRef]
- Antonelli, M.L.; Curini, R.; Scricciolo, D.; Vinci, G. Determination of free fatty acids and lipase activity in milk: Quality and storage markers. Talanta 2002, 58, 561–568. [Google Scholar] [CrossRef]
- Kilcawley, K.N.; Mannion, D.T. Free Fatty Acids Quantification in Dairy Products. In Fatty Acids; Catala, A., Ed.; IntechOpen: London, UK, 2017; pp. 209–220. [Google Scholar]
- Hamilton, S.; Hamilton, R.J.; Sewell, P.A. Extraction of lipids and derivatives formation. In Lipid Analysis. A Practical Approach; Hamilton, R.J., Hamilton, S., Eds.; Oxford University Press Inc.: New York, NY, USA, 1992; pp. 13–64. [Google Scholar]
- Folch, J.; Lees, M.; Stanley, G.H.S. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [PubMed]
- Bligh, E.G.; Dyer, W.J. A rapid method of total lipid extraction and purification. Can. J. Biochem. Phys. 1959, 37, 911–917. [Google Scholar] [CrossRef]
- Nichols, D.S. Principles of Lipid Analysis. In Chemical and Functional Properties of Food Lipids; Sikorski, Z.E., Kolakowska, A., Eds.; CRC Press: Boca Raton, FL, USA, 2003; pp. 167–188. [Google Scholar]
- Petrović, M.; Kezić, N.; Bolanča, V. Optimization of the GC method for routine analysis of the fatty acid profile in several food samples. Food Chem. 2010, 122, 285–291. [Google Scholar] [CrossRef]
- Aldai, N.; Kramer, J.K.G.; Cruz-Hernandez, C.; Santercole, V.; Delmonte, P.; Mossoba, M.M.; Dugan, M.E.R. Appropriate extraction and methylation techniques for lipid analysis. In Fats and Fatty Acids in Poultry Nutrition and Health; Cherian, G., Poureslami, R., Eds.; Context Products Ltd: Leicestershire, UK, 2012; pp. 249–290. [Google Scholar]
- Maxwell, R.J.; Mondimore, D.; Tobias, J. Rapid method for the quantitative extraction and simultaneous class separation of milk lipids. J. Dairy Sci. 1986, 69, 321–325. [Google Scholar] [CrossRef]
- Lepage, G.; Roy, C.C. Direct transesterification of all classes of lipids in a one-step reaction. J. Lipid Res. 1986, 27, 114–120. [Google Scholar] [PubMed]
- Reis, M.G.; dos Reis, M.M.; Leath, S.; Stelwagen, K. Direct analysis of fatty acid profile from milk by thermochemolysis–gas chromatography–mass spectrometry. J. Chromatogr. A 2011, 1218, 316–323. [Google Scholar] [CrossRef] [PubMed]
- Cruz-Hernandez, C.; Goeuriot, S.; Giuffrida, F.; Thakkar, S.K.; Destaillats, F. Direct quantification of fatty acids in human milk by gas chromatography. J. Chromatogr. A 2013, 1284, 174–179. [Google Scholar] [CrossRef] [PubMed]
- Amer, B.; Nebel, C.; Bertram, H.C.; Mortensen, G.; Hermansen, K.; Dalsgaard, T.K. Novel method for quantification of individual free fatty acids in milk using an in-solution derivatisation approach and gas chromatography-mass spectrometry. Int. Dairy J. 2013, 32, 199–203. [Google Scholar] [CrossRef]
- Innocente, N.; Moret, S.; Corradini, C.; Conte, L.S. A rapid method for the quantitative determination of short-chain free volatile fatty acids from cheese. J. Agric. Food Chem. 2000, 48, 3321–3323. [Google Scholar] [CrossRef]
- Wijesundera, C.; Drury, L.; Walsh, T. Determination of free fatty acids and lactones in cheese by solid phase microextraction (SPME). Aust. J. Dairy Technol. 1998, 53, 140. [Google Scholar]
- Pinho, O.; Ferreira, I.M.P.L.V.O.; Ferreira, M.A. Solid-phase microextraction in combination with GC/MS for quantification of the major volatile free fatty acids in ewe cheese. Anal. Chem. 2002, 74, 5199–5204. [Google Scholar] [CrossRef] [PubMed]
- Pinho, O.; Ferreira, I.M.P.L.V.O.; Ferreira, M.A. Quantification of short-chain free fatty acids in “terrincho” ewe cheese: Intravarietal comparison. J. Dairy Sci. 2003, 86, 3102–3109. [Google Scholar] [CrossRef]
- Gonzalez-Cordova, A.F.; Vallejo-Cordoba, B. Quantitative determination of short-chain free fatty acids in milk using solid-phase microextraction and gas chromatography. J. Agric. Food Chem. 2001, 49, 4603–4608. [Google Scholar] [CrossRef]
- Gonzalez-Cordova, A.F.; Vallejo-Cordoba, B. Detection and prediction of hydrolytic rancidity in milk by multiple regression analysis of short-chain free fatty acids determined by solid phase microextraction gas chromatography and quantitative flavor intensity assessment. J. Agric. Food Chem. 2003, 51, 7127–7131. [Google Scholar] [CrossRef] [PubMed]
- Rincon, A.A.; Pino, V.; Ayala, J.H.; Afonso, A.M. Multiple headspace solid-phase microextraction for quantifying volatile free fatty acids in cheeses. Talanta 2014, 129, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Henderson, J.; Tocher, D.R. Thin-layer chromatography. In Lipid Analysis. A Practical Approach; Hamilton, R.J., Hamilton, S., Eds.; Oxford University Press Inc.: New York, NY, USA, 1992; pp. 65–100. [Google Scholar]
- Christie, W.W.; Han, X. Lipid Analysis. Isolation, Separation and Lipidomic Analysis, 4th ed.; The Oily Press: Bridgwater, UK, 2010; pp. 71–73. [Google Scholar]
- Kaluzny, M.A.; Duncan, L.A.; Merritt, M.V.; Epps, D.E. Rapid separation of lipid classes in high-yield and purity using bonded phase columns. J. Lipid Res. 1985, 26, 135–140. [Google Scholar] [PubMed]
- Barron, L.J.R.; Hernandez, I.; Bilbao, A.; Flanagan, C.E.; Najera, A.I.; Virto, M.; Pérez-Elortondo, F.J.; Albisu, M.; de Renobales, M. Changes in lipid fractions and sensory properties of Idiazabal cheese induced by lipase addition. J. Dairy Res. 2004, 71, 372–379. [Google Scholar] [CrossRef] [PubMed]
- De Jong, C.; Badings, H.T. Determination of free fatty acids in milk and cheese procedures for extraction, clean up, and capillary gas chromatographic analysis. J. High Res. Chromatog. 1990, 13, 94–98. [Google Scholar] [CrossRef]
- De Jong, C.; Palma, K.; Neeter, R. Sample preparation before capillary gas-chromatographic estimation of free fatty-acids in fermented dairy-products. Neth. Milk Dairy J. 1994, 48, 151–156. [Google Scholar]
- Deeth, H.C.; Fitzgerald, C.H.; Snow, A.J. A gas chromatographic method for the quantitative determination of free fatty acids in milk and milk products. N. Z. J. Dairy Sci. 1983, 18, 13–20. [Google Scholar]
- De la Fuente, M.A.; Juarez, M. Review: Determination of free fatty acids in dairy products. Rev. Esp. Cienc. Tecnol. Aliment. 1993, 33, 247–269. [Google Scholar]
- Deeth, H.C.; Fitzgerald, C.H. Lipolytic enzymes and hydrolytic rancidity in milk and milk products. In Advanced Dairy Chemistry. Volume 2. Lipids, 2nd ed.; Fox, P.F., Ed.; Chapman & Hall: London, UK, 1995; pp. 247–308. [Google Scholar]
- Collomb, M.; Spahni, M. Review of the methods used for the determination of free fatty acids in milk and dairy products. Lebensm. Wiss. Technol. 1995, 28, 355–379. [Google Scholar] [CrossRef]
- Guler, Z. Quantification of free fatty acids and flavor characteristics of kasar cheeses. J. Food Lipids 2005, 12, 209–221. [Google Scholar] [CrossRef]
- Najera, A.I.; Barcina, Y.; de Renobales, M.; Barron, L.J.R. Determination of triacylglycerol composition of Idiazabal cheese. Chromatographia 1998, 47, 579–586. [Google Scholar] [CrossRef]
- Najera, A.I.; Perea, S.; Barcina, Y.; de Renobales, M.; Barron, L.J.R. A reverse-phase high-performance liquid chromatographic method for analyzing complex mixtures of triglycerides. Application to the fat fraction of an aged cheese. J. Am. Oil Chem. Soc. 1999, 76, 399–407. [Google Scholar] [CrossRef]
- Christie, W.W. Advances in Lipid Methodology-One; The Oily Press: Dundee, Scotland, 1992; pp. 181–237. [Google Scholar]
- Momchilova, S.M.; Nikolova-Damyanova, B.M. Advances in silver ion chromatography for the analysis of fatty acids and triacylglycerols-2001 to 2011. Anal. Sci. 2012, 28, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Iverson, J.L.; Sheppard, A.J. Determination of fatty acids in butter fat using temperature-programmed gas chromatography of the butyl esters. Food Chem. 1986, 21, 223–234. [Google Scholar] [CrossRef]
- Sasaki, R.; Umezawa, M.; Tsukahara, S.; Ishiguro, T.; Sato, S.; Watanabe, Y. Assignment of milk fat fatty acid propyl esters by GC-FID analysis with the aid of Ag-ion solid-phase extraction. J. Oleo Sci. 2015, 64, 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Kramer, J.K.C.; Fellner, V.; Dugan, M.E.R.; Sauer, F.D.; Mossoba, M.M.; Yurawecz, M.P. Evaluating acid and base catalysts in the methylation of milk and rumen fatty acids with special emphasis on conjugated dienes and total trans fatty acids. Lipids 1997, 32, 1219–1228. [Google Scholar] [CrossRef] [PubMed]
- Guinee, T.P.; McSweeney, P.L.H. Significance of milk fat in cheese. In Advanced Dairy Chemistry. Volume 2. Lipids, 3rd ed.; Fox, P.F., McSweeney, P.L.H., Eds.; Springer Science: New York, NY, USA, 2006; pp. 377–428. [Google Scholar]
- Virto, M.; Chavarri, F.; Bustamante, M.A.; Barron, L.J.R.; Aramburu, M.; Vicente, M.S.; Pérez-Elortondo, F.J.; Albisu, M.; de Renobales, M. Lamb rennet paste in ovine cheese manufacture. Lipolysis and flavour. Int. Dairy J. 2003, 13, 391–399. [Google Scholar] [CrossRef]
- Martin-Hernández, M.C.; Alonso, L.; Juárez, M.; Fontecha, J. Gas chromatographic method for determining free fatty acids in cheese. Chromatographia 1988, 25, 87–90. [Google Scholar] [CrossRef]
- Martínez-Castro, I.; Alonso, L.; Juárez, M. Gas chromatographic analysis of free fatty acids and glycerides of milk fat using tetramethylammonium hydroxide as catalyste. Chromatographia 1986, 21, 37–40. [Google Scholar] [CrossRef]
- Juárez, M.; de la Fuente, M.A.; Fontecha, J. Improved gas chromatographic method for the determination of the individual free fatty acids in cheese using a capillary column and a PTV injector. Chromatographia 1992, 33, 351–355. [Google Scholar] [CrossRef]
- Chavarri, F.; Virto, M.; Martin, C.; Najera, A.I.; Santisteban, A.; Barron, L.J.R.; De Renobales, M. Determination of free fatty acids in cheese: Comparison of two analytical methods. J. Dairy Res. 1997, 64, 445–452. [Google Scholar] [CrossRef]
- Zhou, T.; Leng, J.; Peng, Y.; Zhang, L.; Guo, Y. Mass spectrometric analysis of free fatty acids in infant milk powders by frozen pretreatment coupled with isotope-labeling derivatization. J. Sep. Sci. 2016, 39, 873–879. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, K.; Suzuki, T.; Itabashi, Y. Determination of free fatty acids in milk and dairy products by reversed-phase HPLC with fluorescence detection. J. Food Hyg. Soc. Jpn. 2002, 43, 230–233. [Google Scholar] [CrossRef]
- Lu, C.Y.; Wu, H.L.; Chen, S.H.; Kou, H.S.; Wu, S.M. Highly Sensitive Determination of Long-Chain Free Fatty Acids by Fluorogenic Derivatization and Liquid Chromatography. Chromatographia 2001, 53, S251–S252. [Google Scholar] [CrossRef]
- ISO-IDF 15885:2002. Milk Fat—Determination of the FA Composition by Gas-Chromatography; International Standard ISO: Geneva, Switzerland, 2002; IDF 184:2002. [Google Scholar]
- Christie, W.W.; Han, X. Lipid Analysis. Isolation, Separation and Lipidomic Analysis, 4th ed.; The Oily Press: Bridgwater, UK, 2010; pp. 159–180. [Google Scholar]
- Park, P.W.; Goins, R.E. In situ preparation of fatty acid methyl esters for analysis of fatty acid composition in foods. J. Food Sci. 1994, 59, 1262–1266. [Google Scholar] [CrossRef]
- Méndez-Cid, F.J.; Centeno, J.A.; Martínez, S.; Carballo, J. Changes in the chemical and physical characteristics of cow’s milk butter during storage: Effects of temperature and addition of salt. J. Food Compos. Anal. 2017, 63, 121–132. [Google Scholar] [CrossRef]
- Ezequiel, J.M.B.; Sancanari, J.B.D.; Machado Neto, O.R.; da Silva, Z.F.; Almeida, M.T.C.; Silva, D.A.V.; van Cleef, F.O.S.; van Cleef, E.H.C.B. Effects of high concentrations of dietary crude glycerin on dairy cow productivity and milk quality. J. Dairy Sci. 2015, 98, 8009–8017. [Google Scholar] [CrossRef] [PubMed]
- Perna, A.; Intaglietta, I.; Simonetti, A.; Gambacorta, E. The influence of casein haplotype on morphometric characteristics of fat globules and fatty acid composition of milk in Italian Holstein cows. J. Dairy Sci. 2016, 99, 2512–2519. [Google Scholar] [CrossRef] [PubMed]
- Trigueros, L.; Sendra, E. Fatty acid and conjugated linoleic acid (CLA) content in fermented milks as assessed by direct methylation. LWT-Food Sci. Technol. 2015, 60, 315–319. [Google Scholar] [CrossRef]
- Cattani, M.; Mantovani, R.; Schiavon, S.; Bittante, G.; Bailoni, L. Recovery of n-3 polyunsaturated fatty acids and conjugated linoleic acids in ripened cheese obtained from milk of cows fed different levels of extruded flaxseed. J. Dairy Sci. 2014, 97, 123–135. [Google Scholar] [CrossRef] [PubMed]
- Tzamaloukas, O.; Orford, M.; Miltiadou, D.; Papachristoforou, C. Partial suckling of lambs reduced the linoleic and conjugated linoleic acid contents of marketable milk in chios ewes. J. Dairy Sci. 2015, 98, 1739–1749. [Google Scholar] [CrossRef] [PubMed]
- Firl, N.; Kienberger, H.; Rychlik, M. Validation of the sensitive and accurate quantitation of the fatty acid distribution in bovine milk. Int. Dairy J. 2014, 35, 139–144. [Google Scholar] [CrossRef]
- Ariko, T.; Kass, M.; Henno, M.; Fievez, V.; Kaert, O.; Kaart, T.; Ots, M. The effect of replacing barley with glycerol in the diet of dairy cows on rumen parameters and milk fatty acid profile. Anim. Feed Sci. Technol. 2015, 209, 69–78. [Google Scholar] [CrossRef]
- Kramer, J.K.G.; Hernandez, M.; Cruz-Hernandez, C.; Kraft, J.; Dugan, M.E.R. Combining results of two GC separations partly achieves determination of all cis and trans 16:1, 18:1, 18:2 and 18:3 except CLA isomers of milk fat as demonstrated using Ag-ion SPE fractionation. Lipids 2008, 43, 259–273. [Google Scholar] [CrossRef] [PubMed]
- Precht, D.; Molkentin, J.; Vahlendieck, M. Influence of the heating temperature on the fat composition of milk fat with emphasis on cis-/trans-isomerization. Nahrung 1999, 43, 25–33. [Google Scholar] [CrossRef]
- Delmonte, P.; Fardin-Kia, A.R.; Kramer, J.K.G.; Mossoba, M.M.; Sidisky, L.; Tyburczy, C.; Rader, J.I. Evaluation of highly polar ionic liquid gas chromatographic column for the determination of the fatty acids in milk fat. J. Chromatogr. A 2012, 1233, 137–146. [Google Scholar] [CrossRef] [PubMed]
- Aldai, N.; de Renobales, M.; Barron, L.J.R.; Kramer, J.K.G. Highlight Article summary: What are the trans fatty acids issues in foods after discontinuation of industrially produced trans fats? Ruminant products, vegetable oils, and synthetic supplements. Eur. J. Lipid Sci. Technol. 2013, 115, 1257. [Google Scholar] [CrossRef]
- Gómez-Cortés, P.; Rodríguez-Pino, V.; Juárez, M.; de la Fuente, M.A. Optimization of milk odd and branched-chain fatty acids analysis by gas chromatography using an extremely polar stationary phase. Food Chem. 2017, 231, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Bravo-Lamas, L.; Aldai, N.; Kramer, J.K.G.; Barron, L.J.R. Case study using commercial dairy sheep flocks: Comparison of the fat nutritional quality of milk produced in mountain and valley farms. LWT-Food Sci. Technol. 2018, 89, 374–380. [Google Scholar] [CrossRef]
- Precht, D.; Molkentin, J.; Destaillats, F.; Wolff, R.L. Comparative studies on individual isomeric 18:1 acids in cow, goat, and ewe milk fats by low-temperature high-resolution capillary gas-liquid chromatography. Lipids 2001, 36, 827–832. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Alcala, L.M.; Alonso, L.; Fontecha, J. Stability of fatty acid composition after thermal, high pressure, and microwave processing of cow milk as affected by polyunsaturated fatty acid concentration. J. Dairy Sci. 2014, 97, 7307–7315. [Google Scholar] [CrossRef] [PubMed]
- Villegas, C.; Zhao, Y.; Curtis, J.M. Two methods for the separation of monounsaturated octadecenoic acid isomers. J. Chromatogr. A 2010, 1217, 775–784. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Alcala, L.M.; Fontecha, J. Hot topic: Fatty acid and conjugated linoleic acid (CLA) isomer composition of commercial CLA-fortified dairy products: Evaluation after processing and storage. J. Dairy Sci. 2007, 90, 2083–2090. [Google Scholar] [CrossRef] [PubMed]
- Sehat, N.; Rickert, R.; Mossoba, M.M.; Kramer, J.K.G.; Yurawecz, M.P.; Roach, J.A.G.; Adlof, R.O.; Morehouse, K.M.; Fritsche, J.; Eulitz, K.D.; et al. Improved separation of conjugated fatty acid methyl esters by silver ion-high-performance liquid chromatography. Lipids 1999, 34, 407–413. [Google Scholar] [CrossRef] [PubMed]
- Delmonte, P.; Fardin Kia, A.; Kramer, J.K.G.; Mossoba, M.M.; Sidisky, L.; Rader, J.I. Separation characteristics of fatty acid methyl esters using SLB-IL111, a new ionic liquid coated capillary gas chromatographic column. J. Chromatogr. A 2011, 1218, 545–554. [Google Scholar] [CrossRef] [PubMed]
- Ragonese, C.; Tranchida, P.Q.; Dugo, P.; Dugo, G.; Sidisky, L.M.; Robillard, M.V.; Mondello, L. Evaluation of use of a dicationic liquid stationary phase in the fast and conventional gas chromatographic analysis of health-hazardous C-18 cis/trans fatty acids. Anal. Chem. 2009, 81, 5561–5568. [Google Scholar] [CrossRef] [PubMed]
- Fanali, C.; Micalizzi, G.; Dugo, P.; Mondello, L. Ionic liquids as stationary phases for fatty acid analysis by gas chromatography. Analyst 2017, 142, 4601–4612. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Cortés, P.; Cívico, A.; de la Fuente, M.A.; Juárez, M.; Sánchez, N.N.; Blanco, F.P.; Marín, A.L.M. Dietary linseed oil increases trans-10,cis-15 18:2 in caprine milk fat. J. Dairy Sci. 2017, 100, 4235–4240. [Google Scholar] [CrossRef] [PubMed]
- Vlaeminck, B.; Harynuc, J.; Fievez, V.; Marriott, P. Comprehensive two-dimensional gas chromatography for the separation of fatty acids in milk. Eur. J. Lipid Sci. Technol. 2007, 109, 757–766. [Google Scholar] [CrossRef]
- Zeng, A.X.; Chin, S.T.; Marriott, P.J. Integrated multidimensional and comprehensive 2D GC analysis of fatty acid methyl esters. J. Sep. Sci. 2013, 36, 878–885. [Google Scholar] [CrossRef] [PubMed]
- Tranchida, P.Q. Comprehensive two-dimensional gas chromatography: A perspective on processes of modulation. J. Chromatogr. A 2018, 1536, 2–5. [Google Scholar] [CrossRef] [PubMed]
- Bergamaschi, M.; Bittante, G. Detailed fatty acid profile of milk, cheese, ricotta and by products, from cows grazing summer highland pastures. J. Dairy Res. 2017, 84, 329–338. [Google Scholar] [CrossRef] [PubMed]
- Schiavon, S.; Cesaro, G.; Cecchinato, A.; Cipolat-Gotet, C.; Tagliapietra, F.; Bittante, G. The influence of dietary nitrogen reduction and conjugated linoleic acid supply to dairy cows on fatty acids in milk and their transfer to ripened cheese. J. Dairy Sci. 2016, 99, 8759–8778. [Google Scholar] [CrossRef] [PubMed]
- Kilcawley, N.K. High Performance Liquid Chromatographic and Gas Chromatographic Methods for Lipids Analysis. In Advanced Dairy Chemistry. Volume 2. Lipids, 3rd ed.; Fox, P.F., McSweeney, P.L.H., Eds.; Springer Science: New York, NY, USA, 2006; pp. 675–696. [Google Scholar]
- Reed, A.W.; Deeth, H.C.; Clegg, D.E. Liquid chromatographic method for quantitative determination of free fatty acids in butter. J. Assoc. Off. Ana. Chem. 1984, 67, 718–721. [Google Scholar]
- Elliott, J.M.; de Haan, B.; Parkin, K.L. An improved liquid chromatographic method for the quantitative determination of free fatty acids in milk products. J. Dairy Sci. 1989, 72, 2478–2482. [Google Scholar] [CrossRef]
- Miwa, H.; Yamamoto, M. Liquid chromatographic determination of free and total fatty acids in milk and milk products as their 2-nitrophenylhydrazides. J. Chromatogr. A 1990, 523, 235–246. [Google Scholar] [CrossRef]
- Lu, C.Y.; Wu, H.L.; Chen, S.H.; Kou, H.S.; Wu, S.M. Simple and sensitive analysis of long-chain free fatty acids in milk by fluorogenic derivatization and high-performance liquid chromatography. J. Agric. Food Chem. 2002, 50, 71–73. [Google Scholar] [CrossRef]
- Vallejo-Cordoba, B.; Mazorra-Manzano, M.A.; Gonzalez-Cordova, A.F. Determination of short-chain free fatty acids in lipolyzed milk fat by capillary electrophoresis. J. Capillary Electrop. 1998, 5, 111–114. [Google Scholar]
- Buchberger, W.; Winna, K. Determination of free fatty acids by capillary zone electrophoresis. Mikrochim. Acta 1996, 122, 45–52. [Google Scholar] [CrossRef]
- The NIST 17 Mass Spectral Library & Search Software (NIST 2017/2014/EPA/NIH). Available online: https://www.sisweb.com/software/ms/nist.htm (accessed on 20 December 2018).
- Kairenius, P.; Ärölä, A.; Leskinen, H.; Toivonen, V.; Ahvenjärvi, S.; Vanhatalo, A.; Huhtanen, P.; Hurme, T.; Griinari, J.M.; Shingfield, K.J. Dietary fish oil supplements depress milk fat yield and alter milk fatty acid composition in lactating cows fed grass silage-based diets. J. Dairy Sci. 2015, 98, 5653–5671. [Google Scholar] [CrossRef] [PubMed]
- Destaillats, F.; Trottier, J.P.; Galvez, J.M.G.; Angers, P. Analysis of α-linolenic acid biohydrogenation intermediates in milk fat with emphasis on conjugated linolenic acids. J. Dairy Sci. 2005, 88, 3231–3239. [Google Scholar] [CrossRef]
- Plourde, M.; Destaillats, F.; Chouinard, P.Y.; Angers, P. Conjugated α-linolenic acid isomers in bovine milk and muscle. J. Dairy Sci. 2007, 90, 5269–5275. [Google Scholar] [CrossRef] [PubMed]
- Thurnhofer, S.; Vetter, W. Application of ethyl esters and d(3)-methyl esters as internal standards for the gas chromatographic quantification of transesterified fatty acid methyl esters in food. J. Agric. Food Chem. 2006, 54, 3209–3214. [Google Scholar] [CrossRef] [PubMed]
- Teng, F.; Wang, P.; Yang, L.; Ma, Y.; Day, L. Quantification of fatty acids in human, cow, buffalo, goat, yak, and camel milk using an improved one-step GC-FID method. Food Anal. Method. 2017, 10, 2881–2891. [Google Scholar] [CrossRef]
- Wiking, L.; Lokke, M.M.; Kidmose, U.; Sundekilde, U.K.; Dalsgaard, T.K.; Larsen, T.; Feilberg, A. Comparison between novel and standard methods for analysis of free fatty acids in milk—Including relation to rancid flavour. Int. Dairy J. 2017, 75, 22–29. [Google Scholar] [CrossRef]
- Perea, S.; De Labastida, E.F.; Najera, A.I.; Chavarri, F.; Virto, M.; De Renobales, M.; Barron, L.J.R. Seasonal changes in the fat composition of lacha sheep’s milk used for Idiazabal cheese manufacture. Eur. Food Res. Technol. 2000, 210, 318–323. [Google Scholar] [CrossRef]
- Abilleira, E.; Collomb, M.; Schlichtherle-Cerny, H.; Virto, M.; de Renobales, M.; Barron, L.J.R. Winter/spring changes in fatty acid composition of farmhouse Idiazabal cheese due to different flock management systems. J. Agric. Food Chem. 2009, 57, 4746–4753. [Google Scholar] [CrossRef] [PubMed]
- Quigley, A.; Connolly, D.; Cummins, W. The application of dispersive liquid–liquid microextraction in the analyses of the fatty acid profile in bovine milk in response to changes in body condition score. J. Chromatogr. B 2018, 1073, 130–135. [Google Scholar] [CrossRef] [PubMed]
- Alves, S.P.; Bessa, R.J.B. Comparison of two gas–liquid chromatograph columns for the analysis of fatty acids in ruminant meat. J. Chromatogr. A 2009, 1216, 5130–5139. [Google Scholar] [CrossRef] [PubMed]
- Wise, S.A.; Phillips, M.M. Evolution of reference materials for the determination of organic nutrients in food and dietary supplements-a critical review. Anal. Bioanal. Chem. 2019, 411, 97–127. [Google Scholar] [CrossRef] [PubMed]
- Tao, F.; Ngadi, M. Applications of spectroscopic techniques for fat and fatty acids analysis of dairy foods. Curr. Opin. Food Sci. 2017, 17, 100–112. [Google Scholar] [CrossRef]
- Andueza, D.; Rouel, J.; Chilliard, Y.; Leroux, C.; Ferlay, A. Prediction of the goat milk fatty acids by near infrared reflectance spectroscopy. Eur. J. Lipid Sci. Technol. 2013, 115, 612–620. [Google Scholar] [CrossRef]
- Coppa, M.; Ferlay, A.; Leroux, C.; Jestin, M.; Chilliard, Y.; Martin, B.; Andueza, D. Prediction of milk fatty acid composition by near infrared reflectance spectroscopy. Int. Dairy J. 2010, 20, 182–189. [Google Scholar] [CrossRef]
- Núñez-Sánchez, N.; Martínez-Marín, A.L.; Polvillo, O.; Fernández-Cabanás, V.M.; Carrizosa, J.; Urrutia, B.; Serradilla, J.M. Near Infrared Spectroscopy (NIRS) for the determination of the milk fat fatty acid profile of goats. Food Chem. 2016, 190, 244–252. [Google Scholar] [CrossRef] [PubMed]
- Lucas, A.; Andueza, D.; Ferlay, A.; Martin, B. Prediction of fatty acid composition of fresh and freeze-dried cheeses by visible–near-infrared reflectance spectroscopy. Int. Dairy J. 2008, 18, 595–604. [Google Scholar] [CrossRef]
- Meurens, M.; Baeten, V.; Yan, S.H.; Mignolet, E.; Larondelle, Y. Determination of the conjugated linoleic acids in cow’s milk fat by Fourier transform Raman spectroscopy. J. Agric. Food Chem. 2005, 53, 5831–5835. [Google Scholar] [CrossRef] [PubMed]
- Bernuy, B.; Meurens, M.; Mignolet, E.; Larondelle, Y. Performance comparison of UV and FT-Raman spectroscopy in the determination of conjugated linoleic acids in cow milk fat. J. Agric. Food Chem. 2008, 56, 1159–1163. [Google Scholar] [CrossRef] [PubMed]
- Stefanov, I.; Baeten, V.; Abbas, O.; Colman, E.; Vlaeminck, B.; De Baets, B.; Fievez, V. Analysis of Milk Odd- and Branched-Chain Fatty Acids Using Fourier Transform (FT)-Raman Spectroscopy. J. Agric. Food Chem. 2010, 58, 10804–10811. [Google Scholar] [CrossRef] [PubMed]
- Stefanov, I.; Baeten, V.; Abbas, O.; Colman, E.; Vlaeminck, B.; De Baets, B.; Fievez, V. Determining Milk Isolated and Conjugated trans-Unsaturated Fatty Acids Using Fourier Transform Raman Spectroscopy. J. Agric. Food Chem. 2011, 59, 12771–12783. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Beattie, R.J.; Fearon, A.M.; O’Donnell, C.P.; Downey, G. Prediction of naturally-occurring, industrially-induced and total trans fatty acids in butter, dairy spreads and Cheddar cheese using vibrational spectroscopy and multivariate data analysis. Int. Dairy J. 2015, 51, 41–51. [Google Scholar] [CrossRef]
- Hu, F.; Furihata, K.; Kato, Y.; Tanokura, M. Nondestructive quantification of organic compounds in whole milk without pretreatment by two-dimensional NMR spectroscopy. J. Agric. Food Chem. 2007, 55, 4307–4311. [Google Scholar] [CrossRef] [PubMed]
- Andreotti, G.; Lamanna, R.; Trivellone, E.; Motta, A. 13C-NMR spectra of TAG: An easy way to distinguish milks from different animal species. J. Am. Oil Chem. Soc. 2002, 79, 123–127. [Google Scholar] [CrossRef]
- Andreotti, G.; Trivellone, E.; Lamanna, R.; Di Luccia, A.; Motta, A. Milk identification of different species: 13C-NMR spectroscopy of triacylglycerols from cows and buffaloes’ milks. J. Dairy Sci. 2000, 83, 2432–2437. [Google Scholar] [CrossRef]
- Schievano, E.; Pasini, G.; Cozzi, G.; Mammi, S. Identification of the production chain of Asiago d’Allevo cheese by nuclear magnetic resonance spectroscopy and principal component analysis. J. Agric. Food Chem. 2008, 56, 7208–7214. [Google Scholar] [CrossRef] [PubMed]
- Scano, P.; Anedda, R.; Melis, M.P.; Dessi’, M.A.; Lai, A.; Roggio, T. 1H- and 13C-NMR Characterization of the Molecular Components of the Lipid Fraction of Pecorino Sardo Cheese. J. Am. Oil Chem. Soc. 2011, 88, 1305–1316. [Google Scholar] [CrossRef]
- Prema, D.; Pilfold, J.L.; Krauchi, J.; Church, J.S.; Donkor, K.K.; Cinel, B. Rapid Determination of Total Conjugated Linoleic Acid Content in Select Canadian Cheeses by 1H NMR Spectroscopy. J. Agric. Food Chem. 2013, 61, 9915–9921. [Google Scholar] [CrossRef] [PubMed]
- Gómez-Gallego, C.; Manuel Morales, J.; Monleon, D.; du Toit, E.; Kumar, H.; Linderborg, K.M.; Zhang, Y.; Yang, B.; Isolauri, E.; Salminen, S.; et al. Human Breast Milk NMR Metabolomic Profile across Specific Geographical Locations and Its Association with the Milk Microbiota. Nutrients 2018, 10, 1355. [Google Scholar] [CrossRef] [PubMed]
- O’Callaghan, T.F.; Vazquez-Fresno, R.; Serra-Cayuela, A.; Dong, E.; Mandal, R.; Hennessy, D.; McAuliffe, S.; Dillon, P.; Wishart, D.S.; Stanton, C.; et al. Pasture Feeding Changes the Bovine Rumen and Milk Metabolome. Metabolites 2018, 8, 27. [Google Scholar] [CrossRef] [PubMed]
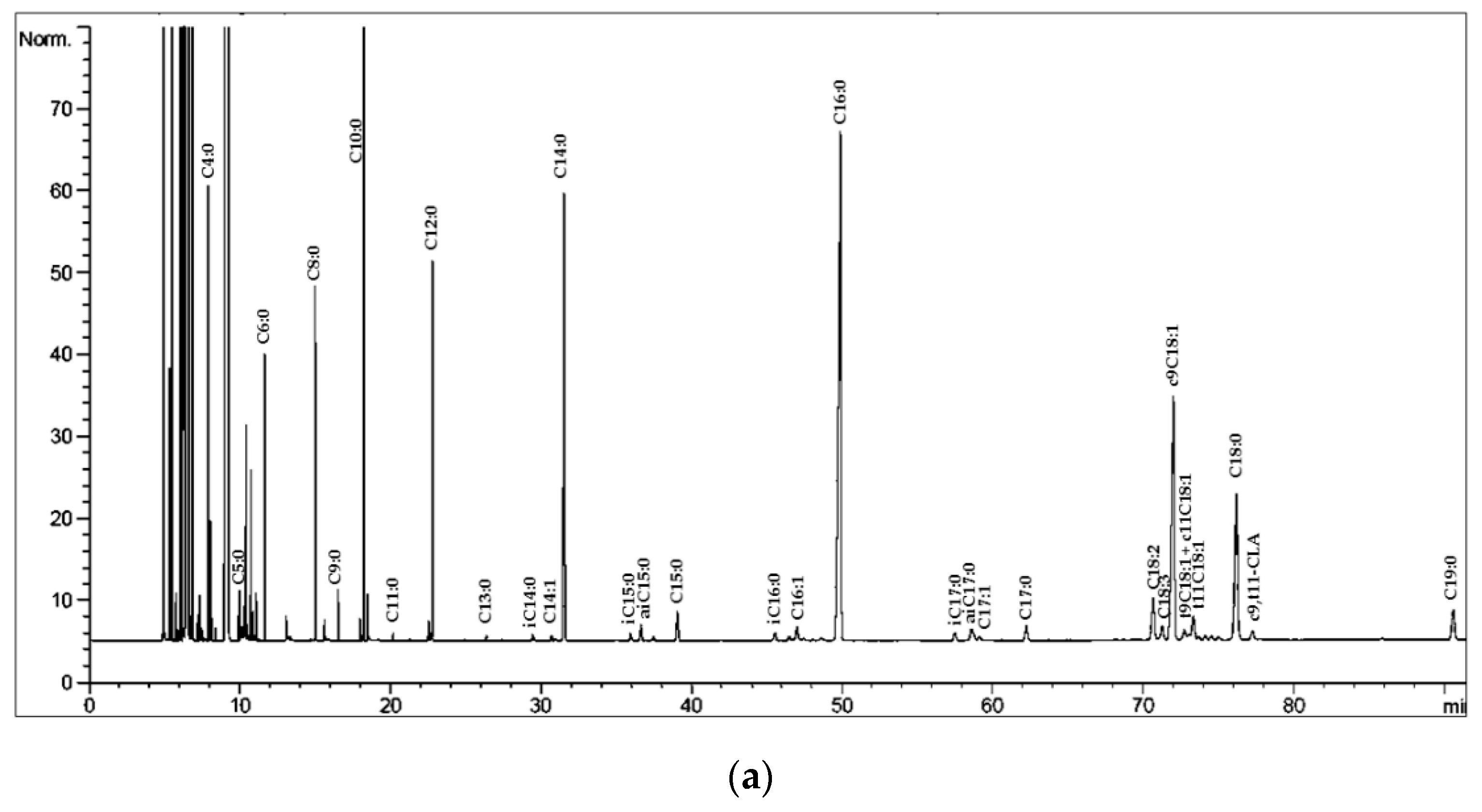
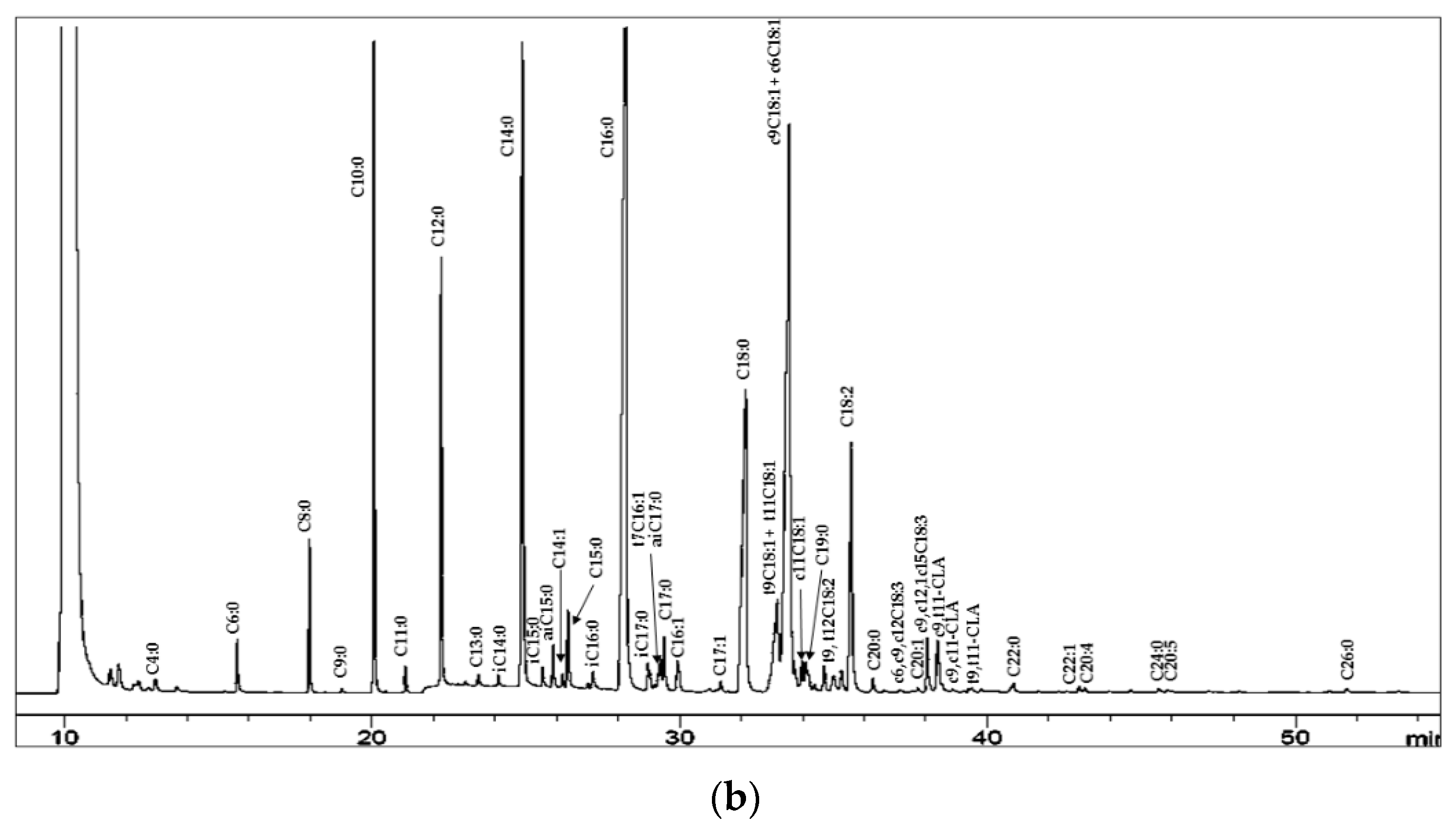
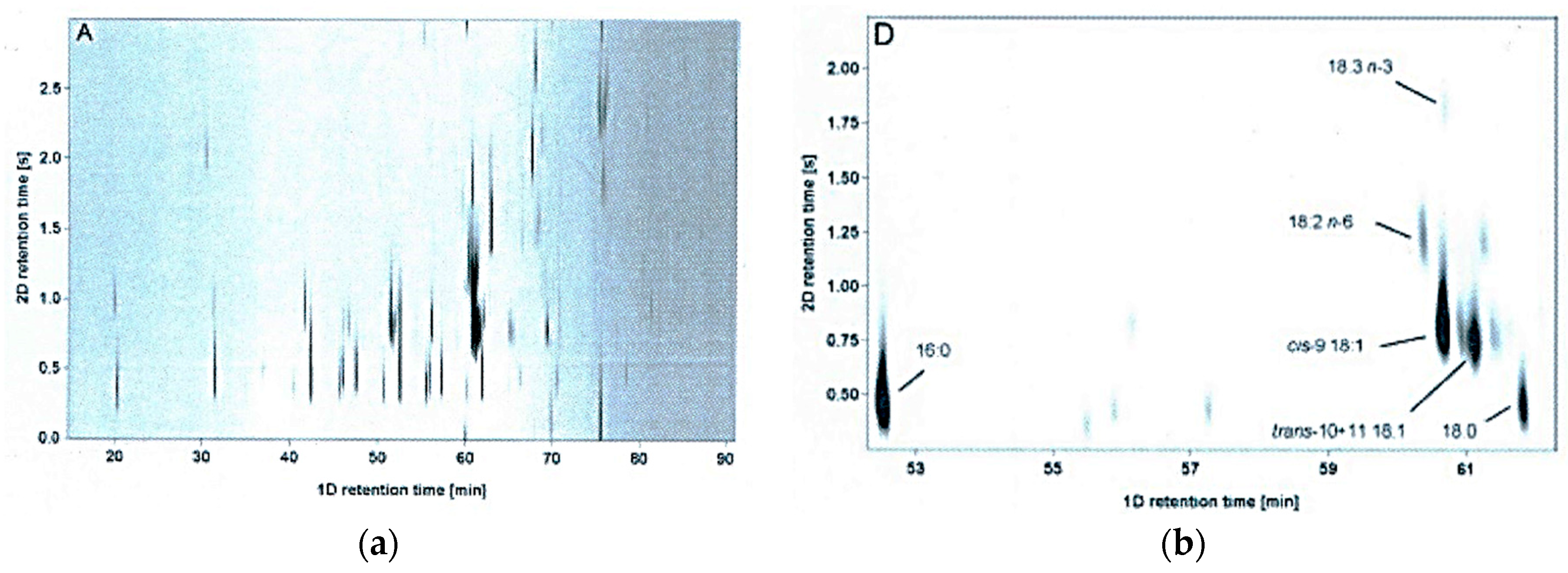
| Lipid Class | Cow 1 | Sheep 2 | Goat 2 | Buffalo 1 |
|---|---|---|---|---|
| Total (wt% of total milk) | 3.3–4.7 | 4.0–9.9 | 4.1–4.5 | 4.7 |
| Triacylglycerol | 97.5 | 98.1 | 97.3 | 98.6 |
| Diacylglycerol | 0.36 | 0.7 | ||
| Monoacylglycerol | 0.027 | 0.03 | 0.10 | T |
| Cholesteryl esters | T | 0.02 | 0.04 | 0.1 |
| Cholesterol | 0.31 | 0.3 | ||
| Free fatty acids | 0.027 | 0.5 | ||
| Chol + DAG + FFA 3 | 1.45 | 1.89 | ||
| Phospholipids | 0.6 | 0.38 | 0.65 | 0.5 |
| Column | Length (m) | Time (min) | Analyzed Sample | FA Resolved | Observations | Ref. |
|---|---|---|---|---|---|---|
| Supelcowax 10 (Supelco Inc.) | 30 | 20 | Human milk | 23 | cis and trans isomers of unsaturated FA not resolved. | [62] |
| Innowax (Agilent Technologies) | 30 | 47 | Cow milk | 28 | cis and trans isomers of unsaturated FA not resolved. Detected CLA (without specifying the isomer). | [63] |
| Omegawax 250 (Supelco Inc.) | 30 | 57 | Cow milk | 30 | cis and trans isomers of unsaturated FA not resolved. Detected c9,t11-CLA. | [64] |
| Omegawax (Supelco Inc.) | 30 | 60 | Cow milk | 43 | Included BCFA of C14:0, C15:0 and C17:0. Separated c7 and c9-C16:1 and c9 and t9-C18:1. Detected CLA (without specifying the isomer). | [65] |
| DB-23 (Agilent J&W) | 30 | 40 | Fermented milk from cow | 21 | Analyzed only main FA, but resolved c9,t11 and t10,c12-CLA isomers. | [66] |
| RTX-2330 (Restek Corp.) | 40 | 60 | Cow milk | 37 | Resolved c9 and t11-C18:1 and c9,t11 and t10,c12-CLA isomers. | [67] |
| CP-Sil 88 (Agilent Technologies) | 50 | 82 | Ewes milk | 40 | Included BCFA, resolved c9 and t11-C18:1 (and “others”) and c9,t11- and t10,c12-CLA isomers. | [68] |
| SP-2560 (Supelco Inc.) | 100 | 80 | Cow milk | 180 | Among 180 resolved peaks, identified around 70 FA, but failed in resolving some cis and trans C18:1 and conjugated and not conjugated C18:2 isomers. | [50] |
| CP7420 (Agilent Technologies) | 100 | 90 | Cow milk | 50 | Column tuned for optimal cis/trans separations of FAME, especially the C18 isomers. Resolved t6, t9, t10 and t11, and c9, c11, c12 and c13-C18:1 and c9,t10; c10,t11 and t9, t11-CLA. | [69,70] |
| CP-Sil 88 (Varian Inc.) | 100 | 86 + 110.33 | Cow milk | 105 | Two separations conducted in the same column. Only few minor CLA isomers could not be resolved. | [71] |
| CP-Wax 58 CB + CP-Sil 88 (Chrompack) | 25 + 100 | 58 + 210 | Cow, goat and ewes milk | Resolved almost all cis/trans C18:1 isomers present in milk fat. | [72] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Amores, G.; Virto, M. Total and Free Fatty Acids Analysis in Milk and Dairy Fat. Separations 2019, 6, 14. https://doi.org/10.3390/separations6010014
Amores G, Virto M. Total and Free Fatty Acids Analysis in Milk and Dairy Fat. Separations. 2019; 6(1):14. https://doi.org/10.3390/separations6010014
Chicago/Turabian StyleAmores, Gustavo, and Mailo Virto. 2019. "Total and Free Fatty Acids Analysis in Milk and Dairy Fat" Separations 6, no. 1: 14. https://doi.org/10.3390/separations6010014
APA StyleAmores, G., & Virto, M. (2019). Total and Free Fatty Acids Analysis in Milk and Dairy Fat. Separations, 6(1), 14. https://doi.org/10.3390/separations6010014






