Lanthanum Nitrate Modification of Soybean Protein Activated Carbon for Enhanced Fluoride Adsorption
Abstract
1. Introduction
2. Materials and Methods
2.1. Experimental Materials
2.2. Characterization Techniques
2.3. Activated Carbon Modification Procedure
2.4. Fluoride Ion Measurement and Adsorption Methods
2.4.1. Fluoride Ion Measurement Method
2.4.2. Adsorption Method
2.4.3. Data Fitting Models
3. Results and Discussion
3.1. Effect of Modifier and Pyrolysis Temperature on Fluoride Ion Adsorption
3.2. Materials Characterization (SEM-EDS and BET Analysis)
3.3. Effect of pH on Fluoride Ion Adsorption
3.4. Adsorption Time and Kinetics
3.5. Adsorption Isotherm Models
3.6. Adsorption Mechanism
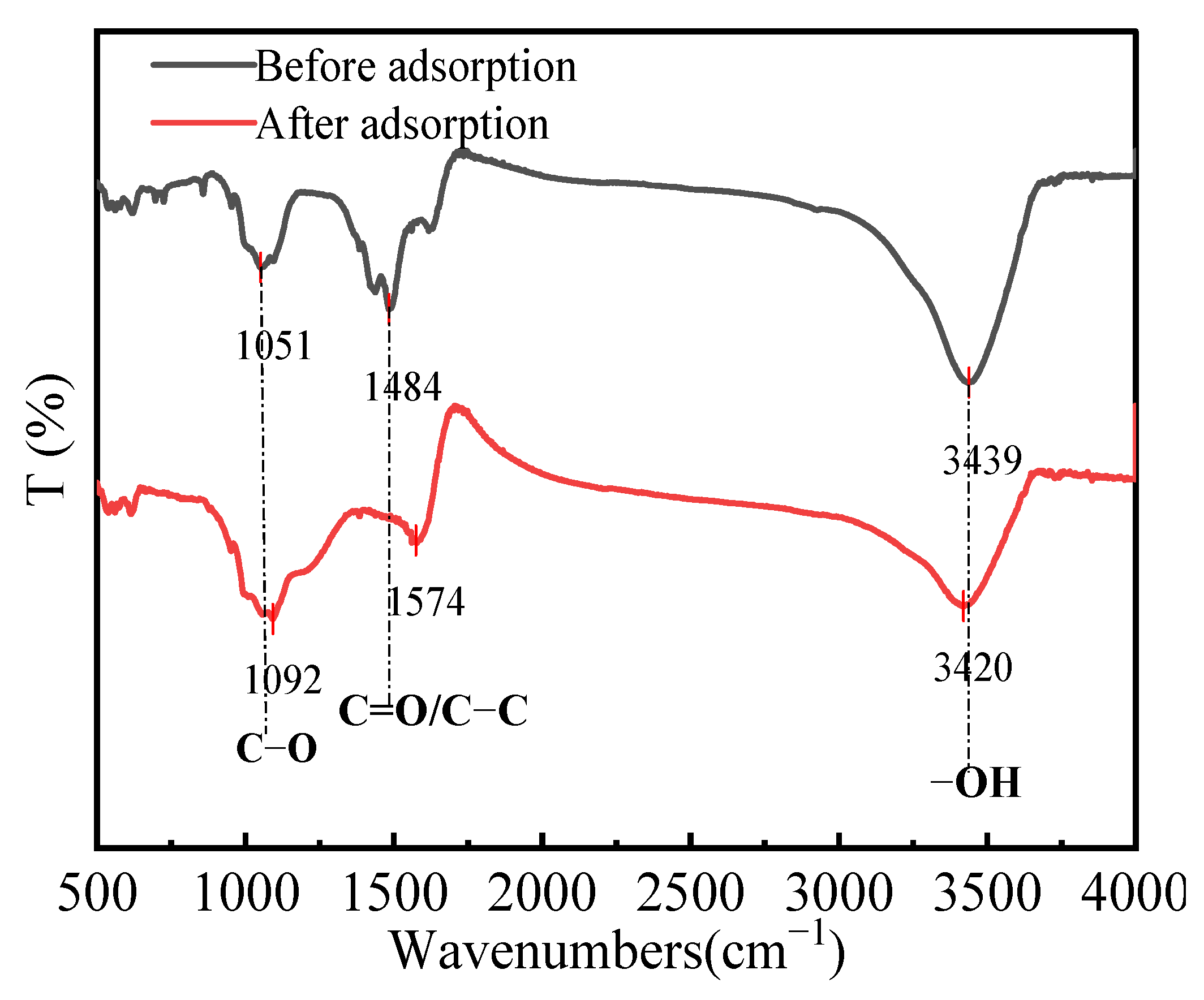
3.6.1. FT-IR Analysis
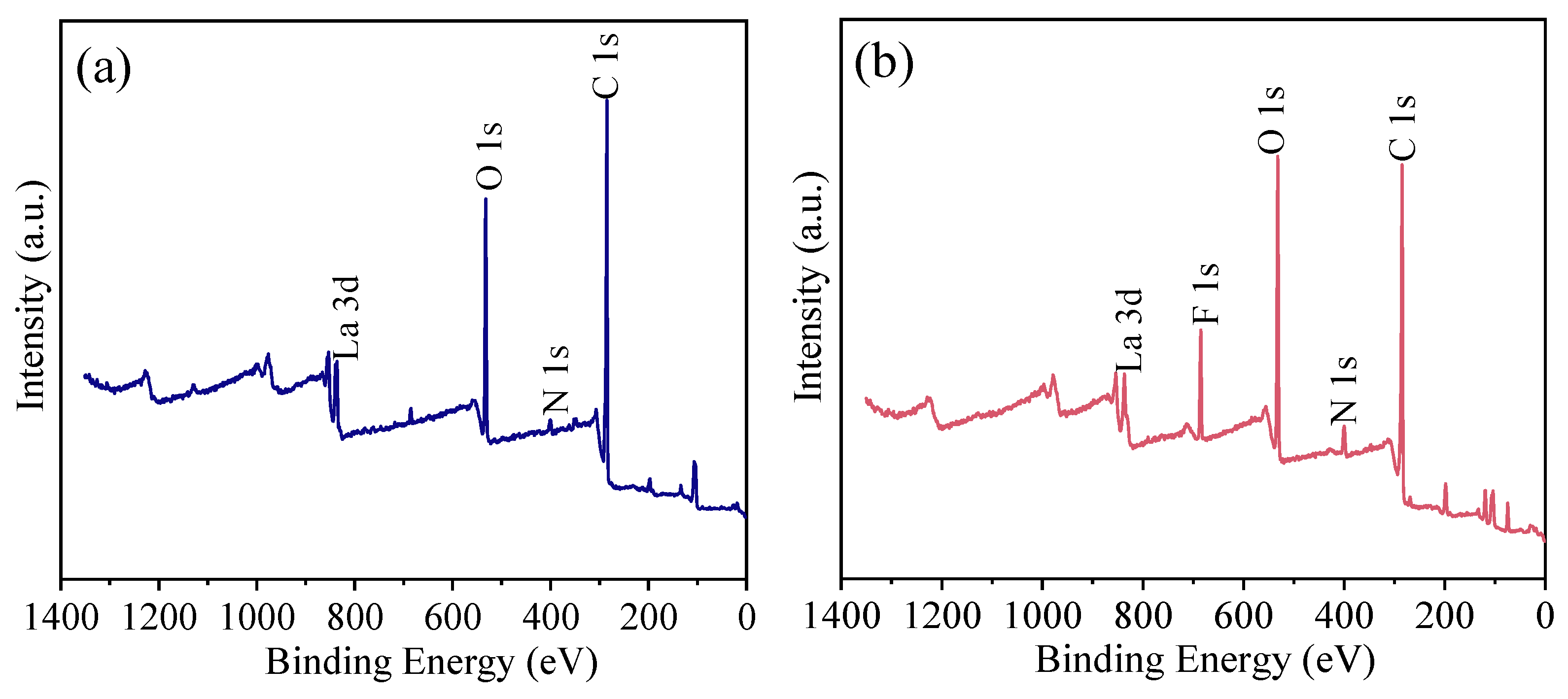
3.6.2. XPS Analysis
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| DETA | Diethylenetriamine |
| EDTA | Ethylene Diamine Tetraacetic Acid |
References
- Jha, S.K.; Singh, R.K.; Damodaran, T.; Mishra, V.K.; Sharma, D.K.; Rai, D. Fluoride in Groundwater: Toxicological Exposure and Remedies. J. Toxicol. Environ. Health Part B 2013, 16, 52–66. [Google Scholar] [CrossRef]
- Hug, S.J.; Winkel, L.H.E.; Voegelin, A.; Berg, M.; Johnson, A.C. Arsenic and Other Geogenic Contaminants in Groundwater—A Global Challenge. Chimia 2020, 74, 524–537. [Google Scholar] [CrossRef]
- Shaji, E.; Sarath, K.V.; Santosh, M.; Krishnaprasad, P.K.; Arya, B.K.; Babu, M.S. Fluoride contamination in groundwater: A global review of the status, processes, challenges, and remedial measures. Di Xue Qian Yuan 2024, 15, 101734. [Google Scholar] [CrossRef]
- Gan, Y.; Wang, X.; Zhang, L.; Wu, B.; Zhang, G.; Zhang, S. Coagulation removal of fluoride by zirconium tetrachloride: Performance evaluation and mechanism analysis. Chemosphere 2019, 218, 860–868. [Google Scholar] [CrossRef]
- Wang, Z.; Guo, M.; Zheng, S.; Yang, X.; Zheng, X. Self-inhibition mechanism of competitive ions for low-cost, highly selective ion removal in a standard ion-exchange resin-packed column process. Sep. Purif. Technol. 2025, 359, 130603. [Google Scholar] [CrossRef]
- Nunes-Pereira, J.; Lima, R.; Choudhary, G.; Sharma, P.R.; Ferdov, S.; Botelho, G.; Sharma, R.K.; Lanceros-Méndez, S. Highly efficient removal of fluoride from aqueous media through polymer composite membranes. Sep. Purif. Technol. 2018, 205, 1–10. [Google Scholar] [CrossRef]
- Collivignarelli, M.C.; Abbà, A.; Carnevale Miino, M.; Torretta, V.; Rada, E.C.; Caccamo, F.M.; Sorlini, S. Adsorption of Fluorides in Drinking Water by Palm Residues. Sustainability 2020, 12, 3786. [Google Scholar] [CrossRef]
- Du, H.; Fang, L.; Shi, X.; Li, T.; Sui, Z.; Zhong, J.; Li, K. Efficient defluoridation of high-fluoride water using rare earth-based adsorbents: Adsorption performance, mechanism, and kinetics. Sci. Rep. 2025, 15, 42709–42715. [Google Scholar] [CrossRef] [PubMed]
- Georgin, J.; Franco, D.S.P.; Manzar, M.S.; Meili, L.; El Messaoudi, N. A critical and comprehensive review of the current status of 17β-estradiol hormone remediation through adsorption technology. Environ. Sci. Pollut. Res. 2024, 31, 24679–24712. [Google Scholar] [CrossRef]
- El Messaoudi, N.; Franco, D.S.P.; Gubernat, S.; Georgin, J.; Şenol, Z.M.; Ciğeroğlu, Z.; Allouss, D.; El Hajam, M. Advances and future perspectives of water defluoridation by adsorption technology: A review. Environ. Res. 2024, 252, 118857. [Google Scholar] [CrossRef]
- Dehmani, Y.; Ba Mohammed, B.; Oukhrib, R.; Dehbi, A.; Lamhasni, T.; Brahmi, Y.; El-Kordy, A.; Franco, D.S.P.; Georgin, J.; Lima, E.C.; et al. Adsorption of various inorganic and organic pollutants by natural and synthetic zeolites: A critical review. Arab. J. Chem. 2024, 17, 105474. [Google Scholar] [CrossRef]
- Şenol, Z.M.; El Messaoudi, N.; Ciğeroglu, Z.; Miyah, Y.; Arslanoğlu, H.; Bağlam, N.; Kazan-Kaya, E.S.; Kaur, P.; Georgin, J. Removal of food dyes using biological materials via adsorption: A review. Food Chem. 2024, 450, 139398. [Google Scholar] [CrossRef]
- Ghosh, S.; Malloum, A.; Igwegbe, C.A.; Ighalo, J.O.; Ahmadi, S.; Dehghani, M.H.; Othmani, A.; Gökkuş, Ö.; Mubarak, N.M. New generation adsorbents for the removal of fluoride from water and wastewater: A review. J. Mol. Liq. 2022, 346, 118257. [Google Scholar] [CrossRef]
- Wan, S.; Lin, J.; Tao, W.; Yang, Y.; Li, Y.; He, F. Enhanced Fluoride Removal from Water by Nanoporous Biochar-Supported Magnesium Oxide. Ind. Eng. Chem. Res 2019, 58, 9988–9996. [Google Scholar] [CrossRef]
- Zhang, X.; Qi, Y.; Chen, Z.; Song, N.; Li, X.; Ren, D.; Zhang, S. Evaluation of fluoride and cadmium adsorption modification of corn stalk by aluminum trichloride. Appl. Surf. Sci. 2021, 543, 148727. [Google Scholar] [CrossRef]
- Aworn, A.; Thiravetyan, P.; Nakbanpote, W. Preparation and characteristics of agricultural waste activated carbon by physical activation having micro- and mesopores. J. Anal. Appl. Pyrolysis 2008, 82, 279–285. [Google Scholar] [CrossRef]
- Yadav, A.K.; Abbassi, R.; Gupta, A.; Dadashzadeh, M. Removal of fluoride from aqueous solution and groundwater by wheat straw, sawdust and activated bagasse carbon of sugarcane. Ecol. Eng. 2013, 52, 211–218. [Google Scholar] [CrossRef]
- Siddique, A.; Nayak, A.K.; Singh, J. Synthesis of FeCl3-activated carbon derived from waste Citrus limetta peels for removal of fluoride: An eco-friendly approach for the treatment of groundwater and bio-waste collectively. Groundw. Sustain. Dev. 2020, 10, 100339. [Google Scholar] [CrossRef]
- Bakhta, S.; Sadaoui, Z.; Lassi, U.; Romar, H.; Kupila, R.; Vieillard, J. Performances of metals modified activated carbons for fluoride removal from aqueous solutions. Chem. Phys. Lett. 2020, 754, 137705. [Google Scholar] [CrossRef]
- Hernández-Montoya, V.; Ramírez-Montoya, L.A.; Bonilla-Petriciolet, A.; Montes-Morán, M.A. Optimizing the removal of fluoride from water using new carbons obtained by modification of nut shell with a calcium solution from egg shell. Biochem. Eng. J. 2012, 62, 1–7. [Google Scholar] [CrossRef]
- Jeyaseelan, A.; Viswanathan, N.; Naushad, M.; Albadarin, A.B. Eco-friendly design of functionalized graphene oxide incorporated alginate beads for selective fluoride retention. Diam. Relat. Mater. 2022, 121, 108747. [Google Scholar] [CrossRef]
- Kanrar, S.; Debnath, S.; De, P.; Parashar, K.; Pillay, K.; Sasikumar, P.; Ghosh, U.C. Preparation, characterization and evaluation of fluoride adsorption efficiency from water of iron-aluminium oxide-graphene oxide composite material. Chem. Eng. J. 2016, 306, 269–279. [Google Scholar] [CrossRef]
- Ruan, Z.; Tian, Y.; Ruan, J.; Cui, G.; Iqbal, K.; Iqbal, A.; Ye, H.; Yang, Z.; Yan, S. Synthesis of hydroxyapatite/multi-walled carbon nanotubes for the removal of fluoride ions from solution. Appl. Surf. Sci. 2017, 412, 578–590. [Google Scholar] [CrossRef]
- Affonso, L.N.; Marques, J.L.; Lima, V.V.C.; Gonçalves, J.O.; Barbosa, S.C.; Primel, E.G.; Burgo, T.A.L.; Dotto, G.L.; Pinto, L.A.A.; Cadaval, T.R.S. Removal of fluoride from fertilizer industry effluent using carbon nanotubes stabilized in chitosan sponge. J. Hazard. Mater. 2020, 388, 122042. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, A.; Kumar, E.; Sillanpää, M. Fluoride removal from water by adsorption—A review. Chem. Eng. J. 2011, 171, 811–840. [Google Scholar] [CrossRef]
- Han, M.; Zhang, J.; Hu, Y.; Han, R. Preparation of Novel Magnetic Microspheres with the La and Ce-Bimetal Oxide Shell for Excellent Adsorption of Fluoride and Phosphate from Solution. J. Chem. Eng. Data 2019, 64, 3641–3651. [Google Scholar] [CrossRef]
- Liang, Z.; Fan, J.; Sun, Y.; Liu, Y.; Zheng, W. Enhanced fluoride removal by nanosized lanthanum hydroxide loaded on magnetic resin. Colloids Surf. A Physicochem. Eng. Asp. 2025, 725, 137715. [Google Scholar] [CrossRef]
- Asare, J.A.; Alhassan, S.I.; Anaman, R.; Amanze, C.; Gang, H.; Wei, D.; Wang, H.; Huang, L. Synergistic influence of lanthanum and cellulose on fly ash synthesized zeolite for enhanced fluoride removal from wastewater: Performance and mechanism. Sep. Purif. Technol. 2025, 379, 134810. [Google Scholar] [CrossRef]
- Ratthika, S.A.; Ramkumar, K.; Elanchezhiyan, S.S.; Kumar, R.; Meenakshi, S. Synthesis and characterization of lanthanum-assisted aluminium metal-organic frameworks for the removal of fluoride ions from an aqueous environment. J. Environ. Chem. Eng. 2025, 13, 118181. [Google Scholar] [CrossRef]
- Qin, J.-C.; Ning, S.; Xu, J.-N.; Guo, F.; Li, Z.; Wei, Y.-Z.; Dodbiba, G.; Fujita, T. Study on the adsorption behavior of tin from waste liquid crystal display using a novel macroporous silica-based adsorbent in one-step separation. Sep. Purif. Technol. 2022, 292, 121006. [Google Scholar] [CrossRef]
- Ho, Y.-S. Review of second-order models for adsorption systems. J. Hazard. Mater. 2006, 136, 681–689. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Ning, S.; Zhang, S.; Wang, X.; Chen, L.; Fujita, T.; Wei, Y. Preparation of a mesoporous ion-exchange resin for efficient separation of palladium from simulated electroplating wastewater. J. Environ. Chem. Eng. 2022, 10, 106966. [Google Scholar] [CrossRef]
- Chen, Y.; Yin, X.; Zheng, N.; Lin, Z.; Fujita, T.; Ning, S.; Chen, Y.; Wang, X. Flexible self-supporting Na3MnTi(PO4)3@C fibers for uranium extraction from seawater by electro sorption. J. Hazard. Mater. 2024, 461, 132664. [Google Scholar] [CrossRef]
- Wang, S.-Q.; Bon, V.; Darwish, S.; Wang, S.-M.; Yang, Q.-Y.; Xu, Z.; Kaskel, S.; Zaworotko, M.J. Insight into the Gas-Induced Phase Transformations in a 2D Switching Coordination Network via Coincident Gas Sorption and In Situ PXRD. ACS Mater. Lett. 2024, 6, 666–673. [Google Scholar] [CrossRef]
- Mechnou, I.; Oukkass, S.; El kartouti, A.; Hlaibi, M.; Saleh, N.i.; Alanazi, A.K. High adsorption capacity of diclofenac and paracetamol using OMWW based W-carboxylate doped activated carbon. Sep. Purif. Technol. 2025, 378, 134719. [Google Scholar] [CrossRef]
- Mukherjee, S.; Kamila, B.; Paul, S.; Hazra, B.; Chowdhury, S.; Halder, G. Optimizing fluoride uptake influencing parameters of paper industry waste derived activated carbon. Microchem. J. 2021, 160, 105643. [Google Scholar] [CrossRef]
- Sahoo, S.K.; Hota, G. Surface functionalization of GO with MgO/MgFe2O4 binary oxides: A novel magnetic nanoadsorbent for removal of fluoride ions. J. Environ. Chem. Eng. 2018, 6, 2918–2931. [Google Scholar] [CrossRef]








| T (K) | C0 (mg/L) | Pseudo-First-Order Model | Pseudo-Second-Order Model | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Qe (mg/g) | k1 | R2 | Prob > F | χ2 | h1 | Qe (mg/g) | k2 | R2 | Prob > F | χ2 | h2 | ||
| 298 | 300 | 116.04 | 0.690 | 0.9592 | 9.16 × 10−5 | 18.15 | 80.07 | 120.40 | 0.109 | 0.9636 | 1.23 × 10−11 | −6.33 | 1580.08 |
| T (K) | pH | Langmuir Isotherm | Freundlich Isotherm | ||||
|---|---|---|---|---|---|---|---|
| KL (L/mg) | Qmax (cal) (mg/g) | R2 | n | Kf (mg1−n·Ln/g) | R2 | ||
| 298 | 2 | 0.00549 | 209.7 | 0.9976 | 0.613 | 4.2 | 0.8703 |
| Materials | pH | Qmax (mg/g) | References |
|---|---|---|---|
| HPAC-La (HPAC-La-750) | 2 | 126.7 | This work |
| AC-CLP500 | 6.6 | 9.709 | [18] |
| AC-Al5 | 5–11 | 13.03 | [19] |
| CMPNS-4 | 7 | 2.3 | [20] |
| GO-HAMO | 5 | 27.75 | [22] |
| HW-MWCNTs | 7 | 30.22 | [23] |
| Sawdust/wheat traw/bagasse | 6 | 1.73/1.93/1.15 | [17] |
| PWA | 4 | 2.93 | [36] |
| MgO-MgFe2O4/GO | 6 | 34 | [37] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Jiang, Z.; Zhou, G.; Bu, W.; Huang, Z.; He, C. Lanthanum Nitrate Modification of Soybean Protein Activated Carbon for Enhanced Fluoride Adsorption. Separations 2026, 13, 59. https://doi.org/10.3390/separations13020059
Jiang Z, Zhou G, Bu W, Huang Z, He C. Lanthanum Nitrate Modification of Soybean Protein Activated Carbon for Enhanced Fluoride Adsorption. Separations. 2026; 13(2):59. https://doi.org/10.3390/separations13020059
Chicago/Turabian StyleJiang, Zhengnan, Guanyu Zhou, Wenchao Bu, Zhenhai Huang, and Chunlin He. 2026. "Lanthanum Nitrate Modification of Soybean Protein Activated Carbon for Enhanced Fluoride Adsorption" Separations 13, no. 2: 59. https://doi.org/10.3390/separations13020059
APA StyleJiang, Z., Zhou, G., Bu, W., Huang, Z., & He, C. (2026). Lanthanum Nitrate Modification of Soybean Protein Activated Carbon for Enhanced Fluoride Adsorption. Separations, 13(2), 59. https://doi.org/10.3390/separations13020059






