Comparison of Dynamic Controllability of Extractive Distillation and Pressure-Swing Distillation for the Separation of Dimethyl Carbonate/Methanol Azeotrope
Abstract
1. Introduction
2. Steady-State Design Statement
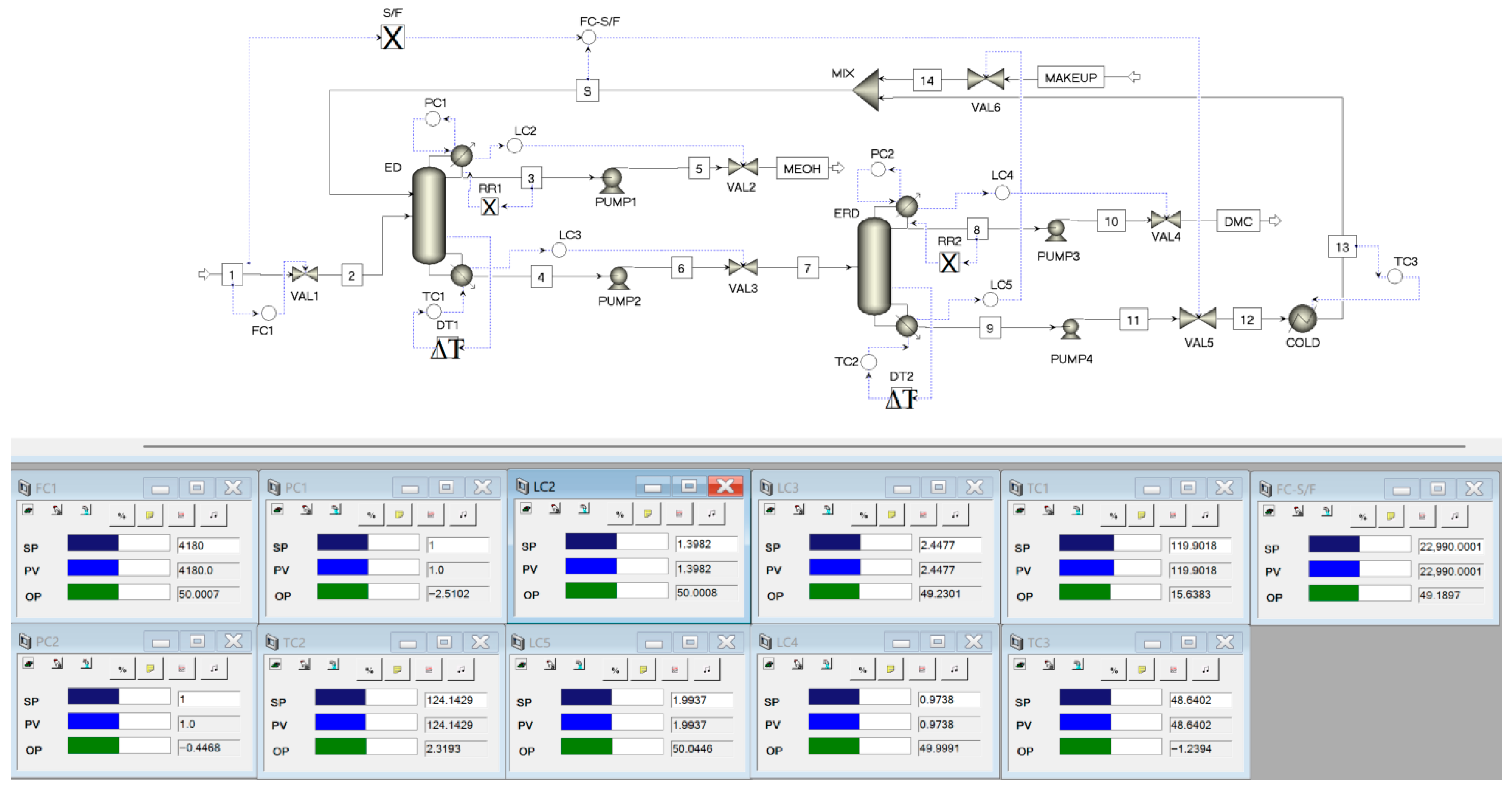
3. Dynamic Control of the Extractive Distillation Process
3.1. Selection of Temperature-Sensitive Trays
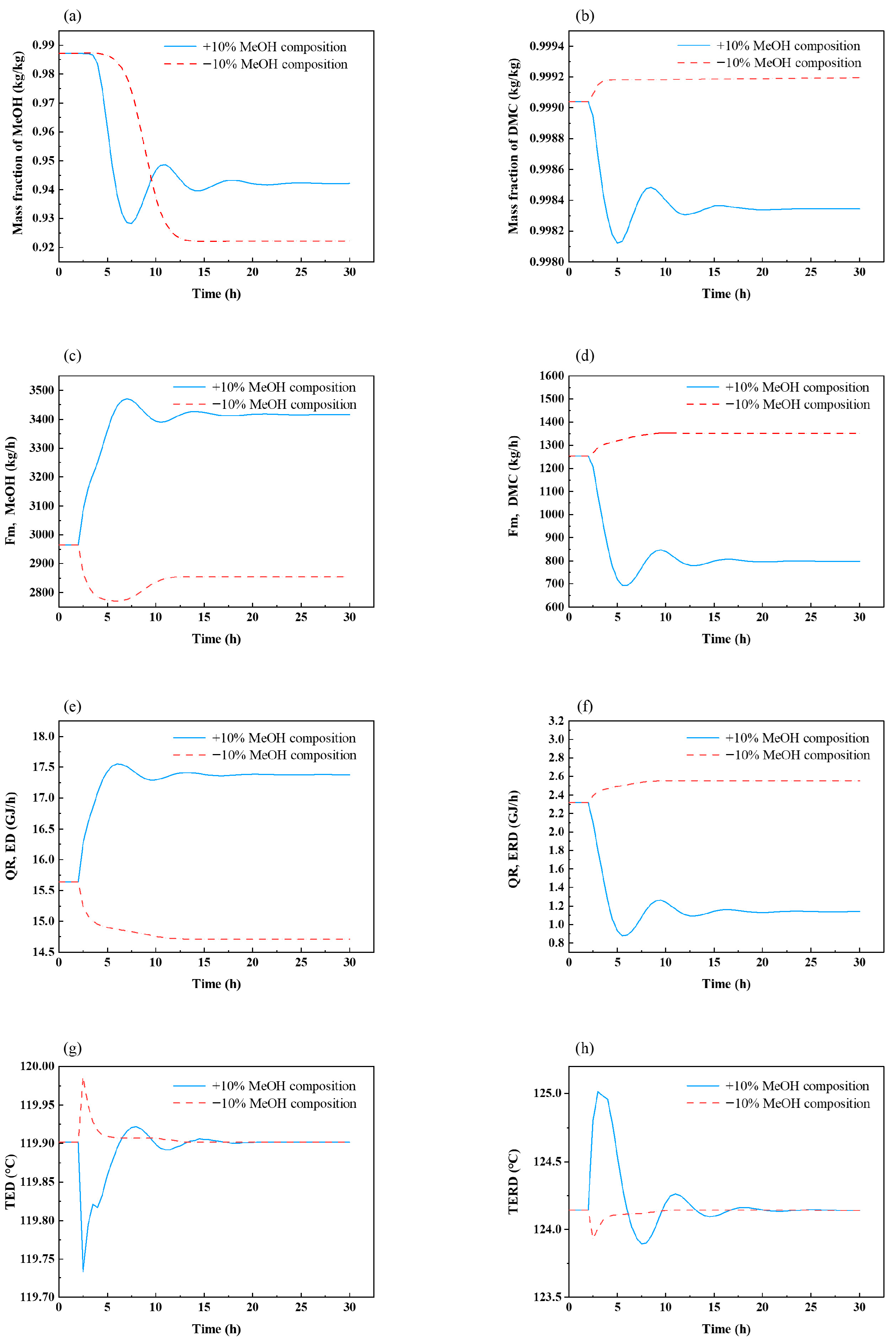
3.2. Fixed-Reflux-Ratio Control Structure for the ED Process
- (1)
- The feed flowrate FC1 was controlled by adjusting the opening of the feed valve.
- (2)
- The pressures of the ED and ERD columns were controlled by manipulating the heat removal in the overhead condensers.
- (3)
- The liquid levels in the reflux drums of the ED and ERD columns were controlled by adjusting the distillate flowrates.
- (4)
- The bottom level of the ED column was controlled by adjusting the bottoms flowrate.
- (5)
- The bottom level of the ERD column was controlled by adjusting the entrainer (OX) makeup flow.
- (6)
- The temperature of tray 67 in the ED column was controlled by manipulating the reboiler duty of the ED column.
- (7)
- The temperature of tray 29 in the ERD column was controlled by manipulating the reboiler duty of the ERD column.
- (8)
- The total entrainer flowrate (S) was kept proportional to the feed flowrate (F), with S/F = 5.5.
- (9)
- For both columns, the overhead reflux flowrate was kept at a fixed R.
4. Dynamic Control of the Pressure-Swing Distillation Process
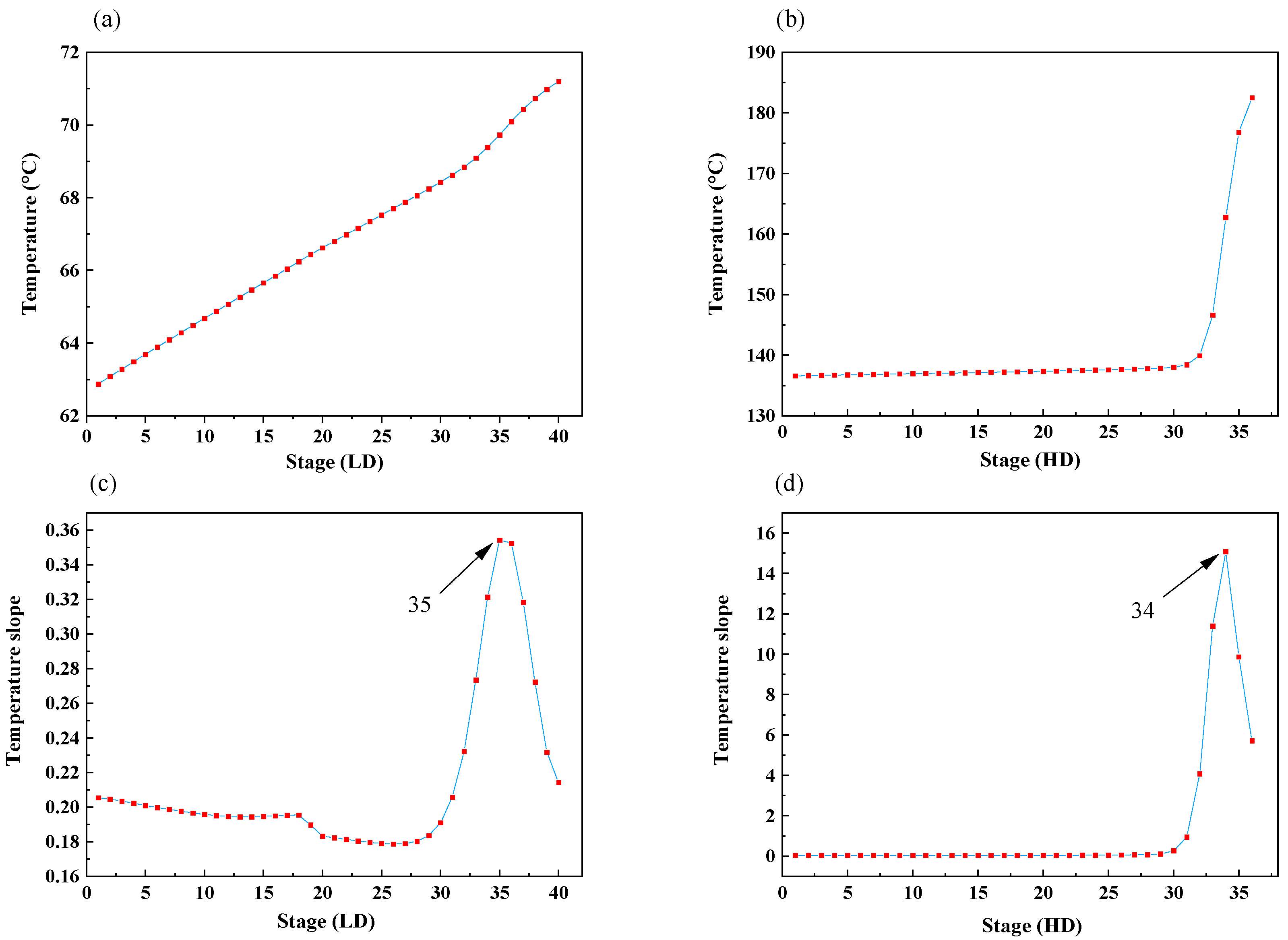
4.1. Selection of Temperature-Sensitive Trays
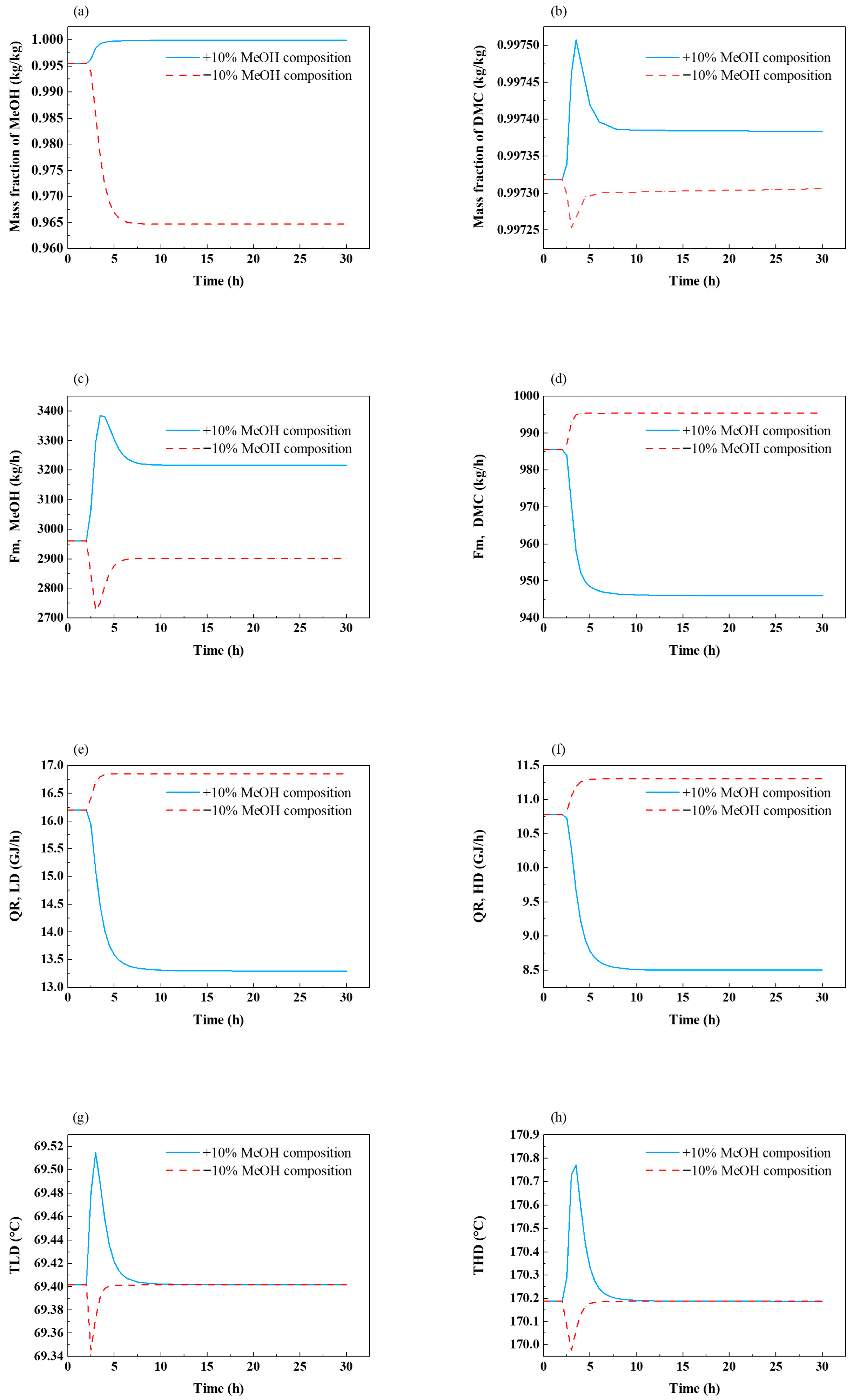
4.2. Fixed-Reflux-Ratio Control Structure for the PSD Process
- (1)
- The feed flowrate FC1 was controlled by adjusting the feed valve opening.
- (2)
- The pressures of the low-pressure and high-pressure columns were controlled via heat removal in their overhead condensers.
- (3)
- The liquid levels in the reflux drums of both columns were controlled by adjusting the distillate flowrates.
- (4)
- The bottom levels of both columns were controlled by manipulating the bottoms flowrates.
- (5)
- The temperature of tray 35 in the low-pressure column was controlled by adjusting the reboiler duty of that column.
- (6)
- The temperature of tray 34 in the high-pressure column was controlled by adjusting the reboiler duty of that column.
- (7)
- For both columns, the reflux flowrate was kept at a fixed R.

5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Nomenclature
| MeOH | Methanol |
| OX | O-xylene |
| ED | Extractive distillation |
| LPC | Low-pressure column |
| NFi | Feeding location for the feed to column |
| VLE | Vapor–liquid equilibrium |
| FCi | Feed flowrate controller |
| F | Feed flowrate |
| PSD | Pressure-swing distillation |
| VLE | Vapor-liquid equilibrium |
| HPC | High-pressure column |
| DMC | Dimethyl carbonate |
| R | Reflux ratio |
| ERD | Entrainer recovery Column |
| S | Entrainer |
| PI | Proportional–integral |
| PID | Proportional–integral–derivative |
| S/F | Entrainer-to-feed ratio |
| TCi | Temperature controller |
| LCi | Liquid controller |
| NT | Total number of trays |
References
- Pacheco, M.A.; Marshall, C.L. Review of dimethyl carbonate (DMC) manufacture and its characteristics as a fuel additive. Energy Fuels 1997, 11, 2–29. [Google Scholar] [CrossRef]
- Liu, Z.; Lin, W.; Li, Q.; Rong, Q.; Zu, H.; Sang, M. Separation of dimethyl carbonate/methanol azeotropic mixture by pervaporation with dealcoholized room temperature-vulcanized silicone rubber/nanosilica hybrid active layer. Sep. Purif. Technol. 2020, 248, 116926. [Google Scholar] [CrossRef]
- Pyo, S.-H.; Park, J.H.; Chang, T.-S.; Hatti-Kaul, R. Dimethyl carbonate as a green chemical. Curr. Opin. Green Sustain. Chem. 2017, 5, 61–66. [Google Scholar] [CrossRef]
- Ono, Y. Catalysis in the production and reactions of dimethyl carbonate, an environmentally benign building block. Appl. Catal. A Gen. 1997, 155, 133–166. [Google Scholar] [CrossRef]
- Wang, S.-J.; Yu, C.-C.; Huang, H.-P. Plant-wide design and control of DMC synthesis process via reactive distillation and thermally coupled extractive distillation. Comput. Chem. Eng. 2010, 34, 361–373. [Google Scholar] [CrossRef]
- Matsuta, S.; Kato, Y.; Ota, T.; Kurokawa, H.; Yoshimura, S.; Fujitani, S. Electron-spin-resonance study of the reaction of electrolytic solutions on the positive electrode for lithium-ion secondary batteries. J. Electrochem. Soc. 2001, 148, A7–A10. [Google Scholar] [CrossRef]
- Tundo, P.; Selva, M. The chemistry of dimethyl carbonate. Acc. Chem. Res. 2002, 35, 706–716. [Google Scholar] [CrossRef]
- Aurbach, D.; Markovsky, B.; Shechter, A.; Ein-Eli, Y.; Cohen, H. A comparative study of synthetic graphite and Li electrodes in electrolyte solutions based on ethylene carbonate-dimethyl carbonate mixtures. J. Electrochem. Soc. 1996, 143, 3809. [Google Scholar] [CrossRef]
- Deng, W.; Shi, L.; Yao, J.; Zhang, Z. A review on transesterification of propylene carbonate and methanol for dimethyl carbonate synthesis. Carbon Resour. Convers. 2019, 2, 198–212. [Google Scholar] [CrossRef]
- Kumar, P.; Srivastava, V.C.; Mishra, I.M. Dimethyl carbonate synthesis by transesterification of propylene carbonate with methanol: Comparative assessment of Ce-M (M = Co, Fe, Cu and Zn) catalysts. Renew. Energy 2016, 88, 457–464. [Google Scholar] [CrossRef]
- Liu, Y.; Zhang, Y.-G.; Shi, H.; You, C.-X.; Tang, J.; Cui, M.; Qiao, X.; Xia, M. Energy-saving heterogeneous azeotropic distillation process for the high-purity separation of dimethyl carbonate/methanol azeotrope with cyclohexane as a light entrainer. Sep. Purif. Technol. 2025, 382, 135789. [Google Scholar] [CrossRef]
- Gerbaud, V.; Rodriguez-Donis, I.; Hegely, L.; Lang, P.; Denes, F.; You, X. Review of extractive distillation. Process design, operation, optimization and control. Chem. Eng. Res. Des. 2019, 141, 229–271. [Google Scholar] [CrossRef]
- Lei, Z.; Li, C.; Chen, B. Extractive distillation: A review. Sep. Purif. Rev. 2003, 32, 121–213. [Google Scholar] [CrossRef]
- Muñoz, R.; Montón, J.; Burguet, M.; De la Torre, J. Separation of isobutyl alcohol and isobutyl acetate by extractive distillation and pressure-swing distillation: Simulation and optimization. Sep. Purif. Technol. 2006, 50, 175–183. [Google Scholar] [CrossRef]
- Luyben, W.L. Comparison of extractive distillation and pressure-swing distillation for acetone–methanol separation. Ind. Eng. Chem. Res. 2008, 47, 2696–2707. [Google Scholar] [CrossRef]
- Luyben, W.L. Comparison of pressure-swing and extractive-distillation methods for methanol-recovery systems in the TAME reactive-distillation process. Ind. Eng. Chem. Res. 2005, 44, 5715–5725. [Google Scholar] [CrossRef]
- Liang, S.; Cao, Y.; Liu, X.; Li, X.; Zhao, Y.; Wang, Y.; Wang, Y. Insight into pressure-swing distillation from azeotropic phenomenon to dynamic control. Chem. Eng. Res. Des. 2017, 117, 318–335. [Google Scholar] [CrossRef]
- You, C.-X.; Zhou, C.; Shi, H.; Tang, J.; Cui, M.; Qiao, X.; Xia, M. Energy-saving extractive distillation system using o-xylene as an entrainer for the high-purity separation of dimethyl carbonate/methanol azeotrope. Sep. Purif. Technol. 2024, 350, 127893. [Google Scholar] [CrossRef]
- Ma, K.; Pan, X.; Zhao, T.; Yang, J.; Zhu, Z.; Xu, D.; Wang, Y. Dynamic control of hybrid processes with liquid–liquid extraction for propylene glycol methyl ether dehydration. Ind. Eng. Chem. Res. 2018, 57, 13811–13820. [Google Scholar] [CrossRef]
- Cao, Y.; Hu, J.; Jia, H.; Bu, G.; Zhu, Z.; Wang, Y. Comparison of pressure-swing distillation and extractive distillation with varied-diameter column in economics and dynamic control. J. Process Control 2017, 49, 9–25. [Google Scholar] [CrossRef]
- Wang, Q.; Yu, B.; Xu, C. Design and control of distillation system for methylal/methanol separation. Part 1: Extractive distillation using DMF as an entrainer. Ind. Eng. Chem. Res. 2012, 51, 1281–1292. [Google Scholar] [CrossRef]
- Wang, X.; Xie, L.; Tian, P.; Tian, G. Design and control of extractive dividing wall column and pressure-swing distillation for separating azeotropic mixture of acetonitrile/N-propanol. Chem. Eng. Process. Process Intensif. 2016, 110, 172–187. [Google Scholar] [CrossRef]
- Peng, J.-Y. Process Optimization and Control Strategy for Special Distillation Separation of the Dimethyl Carbonate–Methanol Azeotrope. Master’s Thesis, Qingdao University of Science and Technology, Qingdao, China, 2017. (In Chinese) [Google Scholar]
- Zhang, H.; Ye, Q.; Qin, J.; Xu, H.; Li, N. Design and control of extractive dividing-wall column for separating ethyl acetate–isopropyl alcohol mixture. Ind. Eng. Chem. Res. 2014, 53, 1189–1205. [Google Scholar] [CrossRef]
- Sun, L.; Wang, Q.; Li, L.; Zhai, J.; Liu, Y. Design and control of extractive dividing wall column for separating benzene/cyclohexane mixtures. Ind. Eng. Chem. Res. 2014, 53, 8120–8131. [Google Scholar] [CrossRef]
- Xia, M.; Xin, Y.; Luo, J.; Li, W.; Shi, L.; Min, Y.; Xu, C. Temperature control for extractive dividing-wall column with an adjustable vapor split: Methylal/methanol azeotrope separation. Ind. Eng. Chem. Res. 2013, 52, 17996–18013. [Google Scholar] [CrossRef]
- Hsu, K.-Y.; Hsiao, Y.-C.; Chien, I.-L. Design and control of dimethyl carbonate–methanol separation via extractive distillation in the dimethyl carbonate reactive-distillation process. Ind. Eng. Chem. Res. 2010, 49, 735–749. [Google Scholar] [CrossRef]
- Yang, A.; Zou, H.; Chien, I.L.; Wang, D.; Wei, S.A.; Ren, J.; Shen, W. Optimal Design and Effective Control of Triple-Column Extractive Distillation for Separating Ethyl Acetate/Ethanol/Water with Multiazeotrope. Ind. Eng. Chem. Res. 2019, 58, 7265–7283. [Google Scholar] [CrossRef]
- Yang, A.; Wei, R.; Sun, S.; Wei, S.A.; Shen, W.; Chien, I.L. Energy-Saving Optimal Design and Effective Control of Heat Integration-Extractive Dividing Wall Column for Separating Heterogeneous Mixture Methanol/Toluene/Water with Multiazeotropes. Ind. Eng. Chem. Res. 2018, 57, 8036–8056. [Google Scholar] [CrossRef]
- Shi, T.; Chun, W.; Yang, A.; Su, Y.; Jin, S.; Ren, J.; Shen, W. Optimization and control of energy saving side-stream extractive distillation with heat integration for separating ethyl acetate-ethanol azeotrope. Chem. Eng. Sci. 2020, 215, 115373. [Google Scholar] [CrossRef]
- Yang, A.; Shen, W.; Wei, S.A.; Dong, L.; Li, J.; Gerbaud, V. Design and control of pressure-swing distillation for separating ternary systems with three binary minimum azeotropes. AIChE J. 2019, 65, 1281–1293. [Google Scholar] [CrossRef]
- Zhu, Z.; Xu, D.; Liu, X.; Zhang, Z.; Wang, Y. Separation of acetonitrile/methanol/benzene ternary azeotrope via triple column pressure-swing distillation. Sep. Purif. Technol. 2016, 169, 66–77. [Google Scholar] [CrossRef]
- Wei, H.-M.; Wang, F.; Zhang, J.-L.; Liao, B.; Zhao, N.; Xiao, F.-k.; Wei, W.; Sun, Y.-H. Design and control of dimethyl carbonate–methanol separation via pressure-swing distillation. Ind. Eng. Chem. Res. 2013, 52, 11463–11478. [Google Scholar] [CrossRef]
- Luyben, W.L.; Chien, I.-L. Design and Control of Distillation Systems for Separating Azeotropes; John Wiley & Sons: Hoboken, NJ, USA, 2011. [Google Scholar]
- Luyben, W.L. Distillation Design and Control Using Aspen Simulation; John Wiley & Sons: Hoboken, NJ, USA, 2013. [Google Scholar]





| Controller | Control Variable | Manipulated Variable | Gain (%/%) | Integral Time (min) |
|---|---|---|---|---|
| TC1 | 67th tray temperature | Reboiler duty | 2.61 | 6.60 |
| TC2 | 29th tray temperature | Reboiler duty | 5.54 | 15.84 |
| Controller | Control Variable | Manipulated Variable | Gain (%/%) | Integral Time (min) |
|---|---|---|---|---|
| TC1 | 35th tray temperature | Reboiler duty | 6.52 | 6.6 |
| TC2 | 34th tray temperature | Reboiler duty | 1.50 | 7.92 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sui, J.; Liu, Y.; Wang, Z.; Li, T.; Gao, K.-Y.; Chu, J.-K.; Zhang, Y.-G.; Shi, H.; Tang, J.; Xia, M. Comparison of Dynamic Controllability of Extractive Distillation and Pressure-Swing Distillation for the Separation of Dimethyl Carbonate/Methanol Azeotrope. Separations 2026, 13, 48. https://doi.org/10.3390/separations13020048
Sui J, Liu Y, Wang Z, Li T, Gao K-Y, Chu J-K, Zhang Y-G, Shi H, Tang J, Xia M. Comparison of Dynamic Controllability of Extractive Distillation and Pressure-Swing Distillation for the Separation of Dimethyl Carbonate/Methanol Azeotrope. Separations. 2026; 13(2):48. https://doi.org/10.3390/separations13020048
Chicago/Turabian StyleSui, Jiancai, Yang Liu, Zhenhua Wang, Tao Li, Kun-Yu Gao, Jin-Ke Chu, Yang-Guang Zhang, Hui Shi, Jihai Tang, and Ming Xia. 2026. "Comparison of Dynamic Controllability of Extractive Distillation and Pressure-Swing Distillation for the Separation of Dimethyl Carbonate/Methanol Azeotrope" Separations 13, no. 2: 48. https://doi.org/10.3390/separations13020048
APA StyleSui, J., Liu, Y., Wang, Z., Li, T., Gao, K.-Y., Chu, J.-K., Zhang, Y.-G., Shi, H., Tang, J., & Xia, M. (2026). Comparison of Dynamic Controllability of Extractive Distillation and Pressure-Swing Distillation for the Separation of Dimethyl Carbonate/Methanol Azeotrope. Separations, 13(2), 48. https://doi.org/10.3390/separations13020048





