The Removal of Per- and Poly-Fluoroalkyl Substances from Water: A Review on Destructive and Non-Destructive Methods
Abstract
1. Introduction
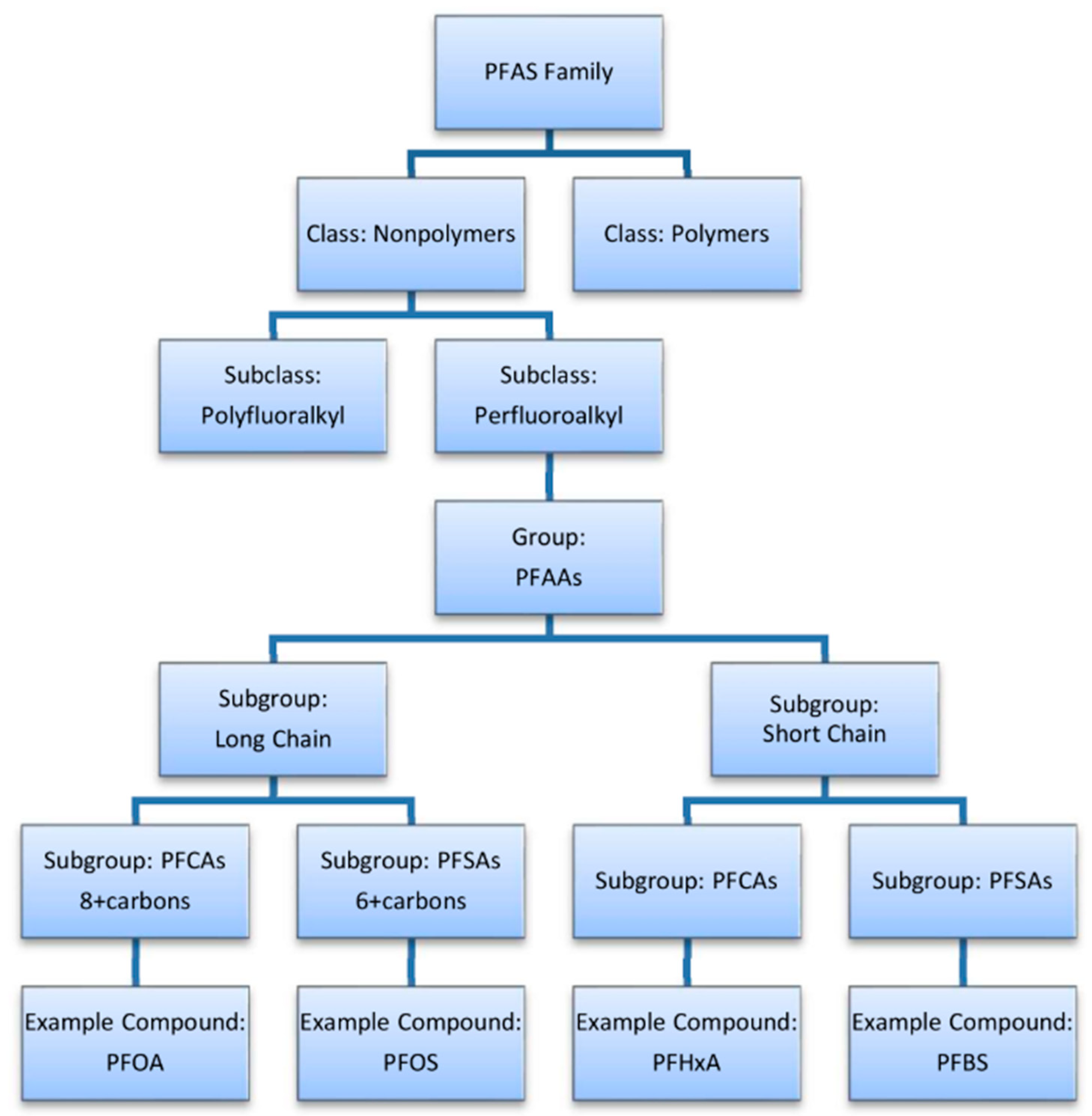
2. Classification of PFAS
2.1. Polymeric and Non-Polymeric PFAS
2.2. PFAS Chain Length
2.3. Legacy and Emerging PFAS
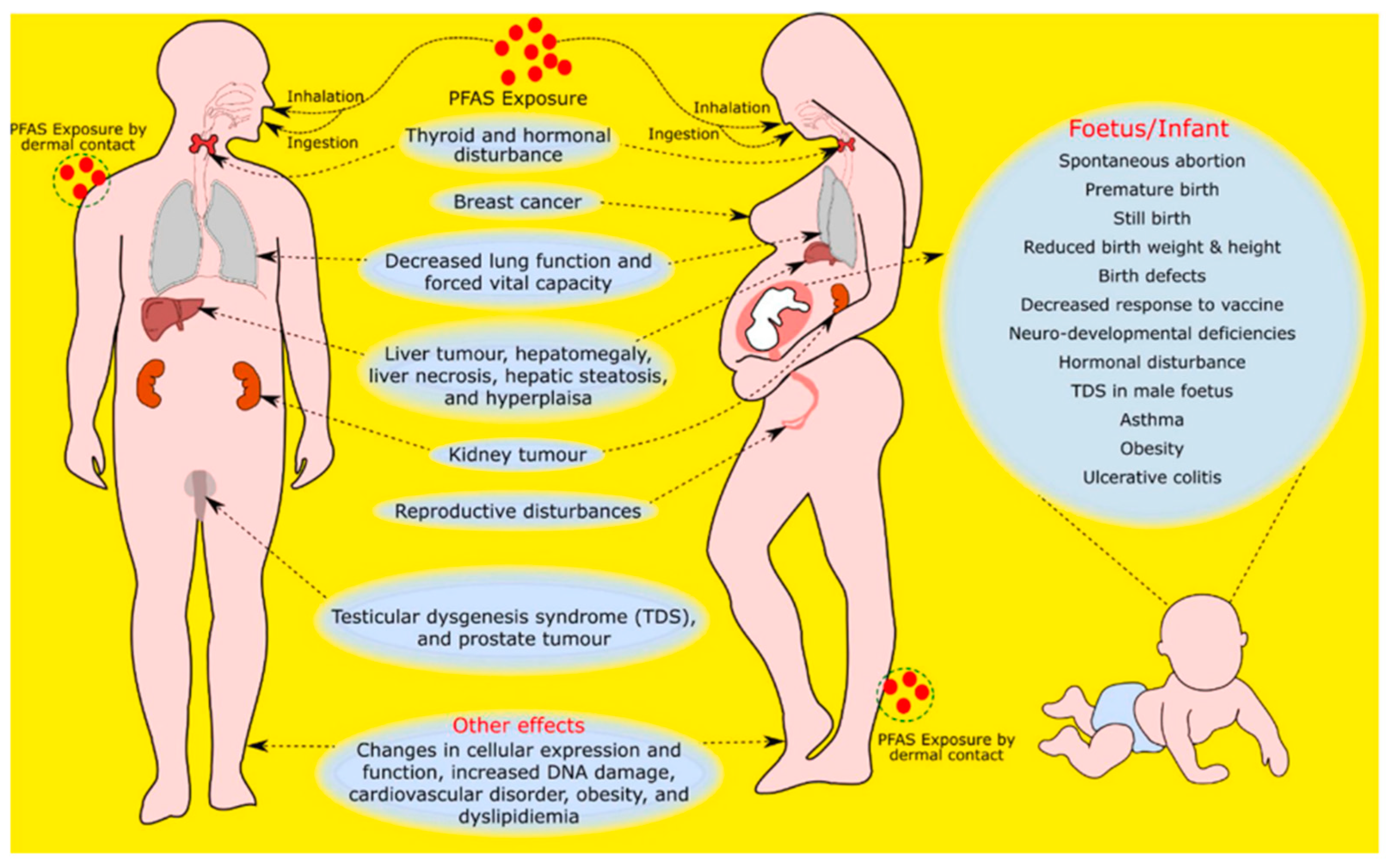
3. Effects on Human Health
4. Legislation
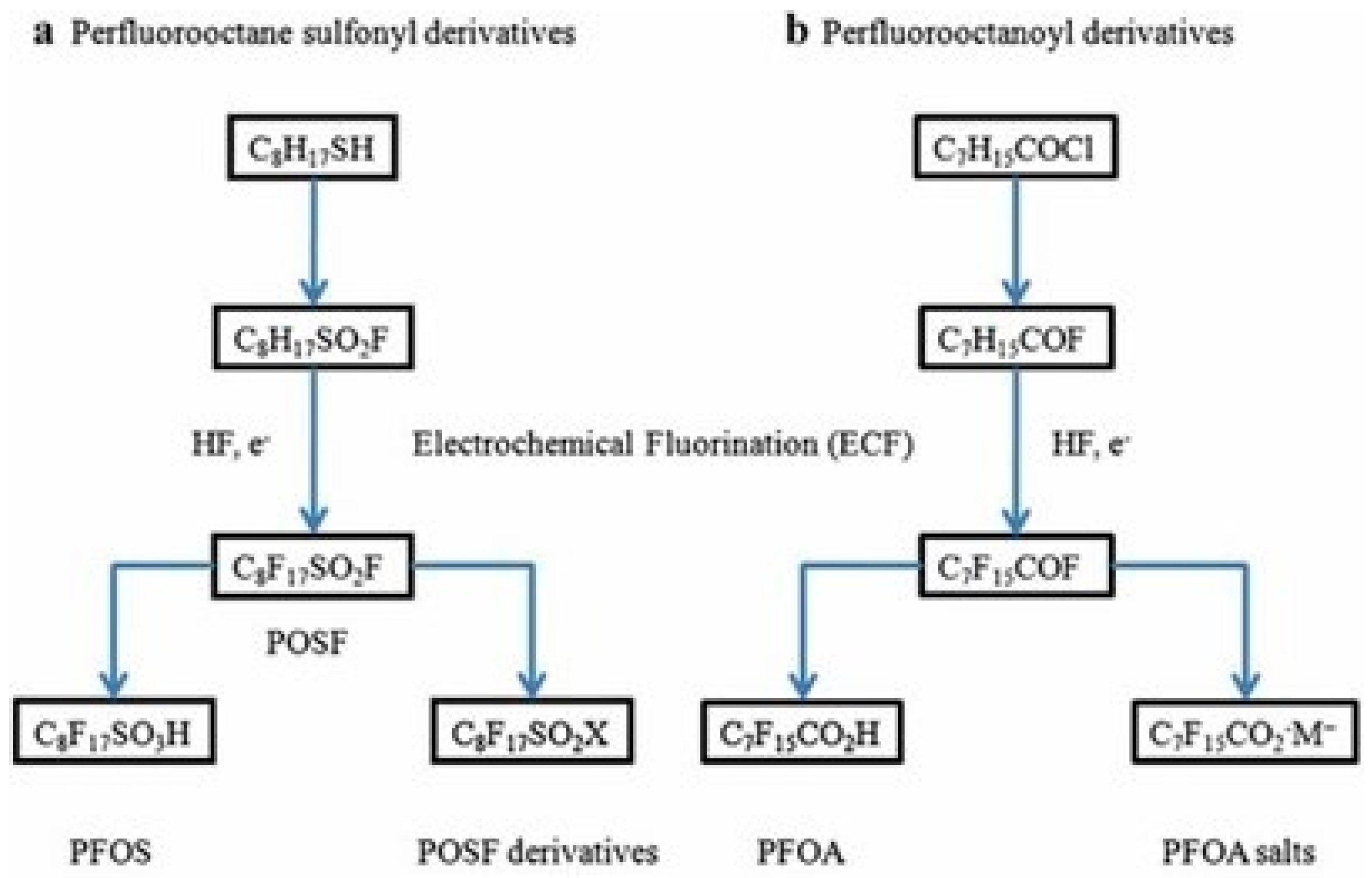
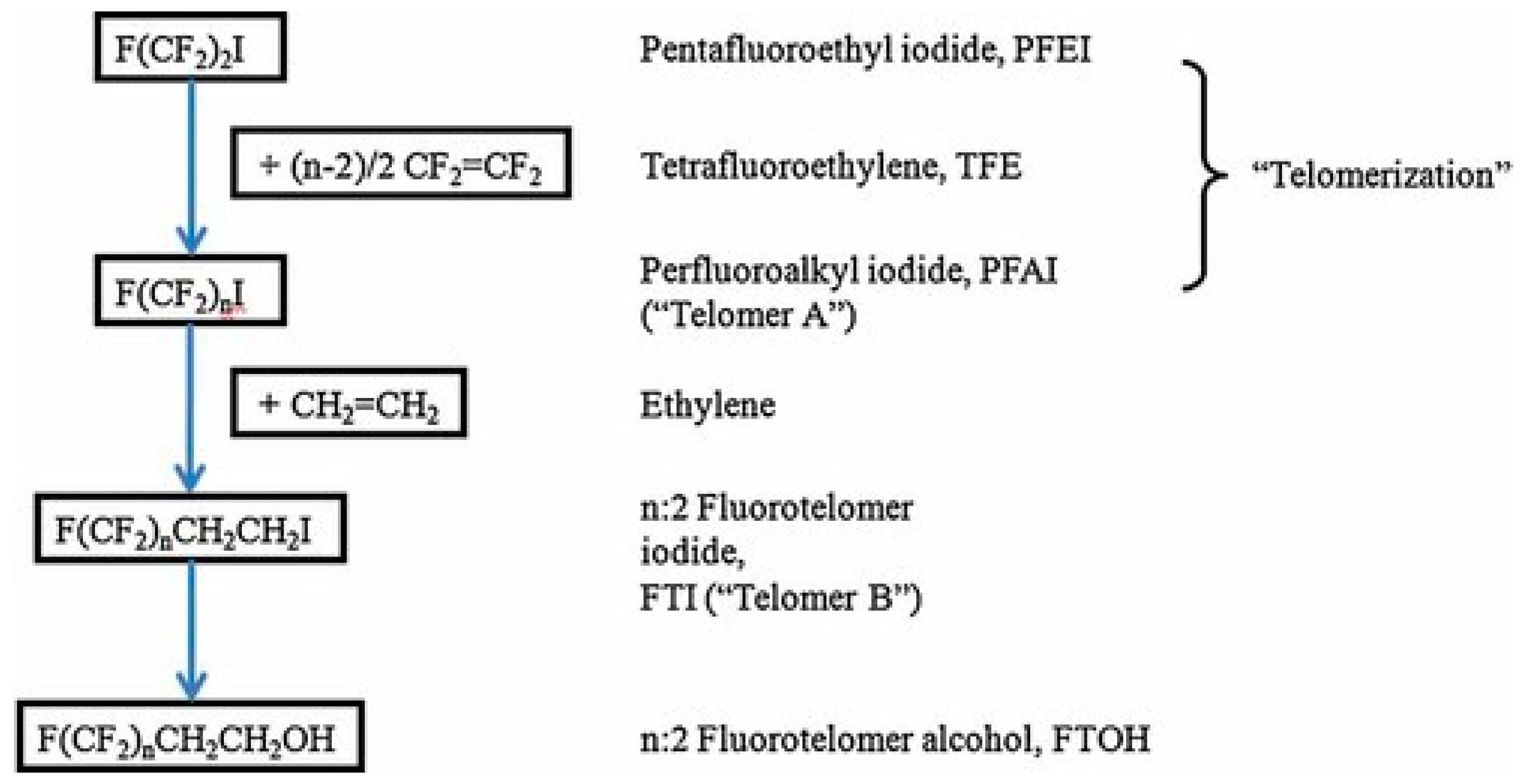
5. Manufacturing Processes
5.1. Electrochemical Fluorination (EFC)
5.2. Telomerization
6. Treatment Technologies
6.1. Adsorption
6.1.1. Carbonaceous Adsorbents
6.1.2. Polysaccharide-Based Adsorbents
6.1.3. Mineral Adsorbents
6.1.4. Regeneration of Adsorbents
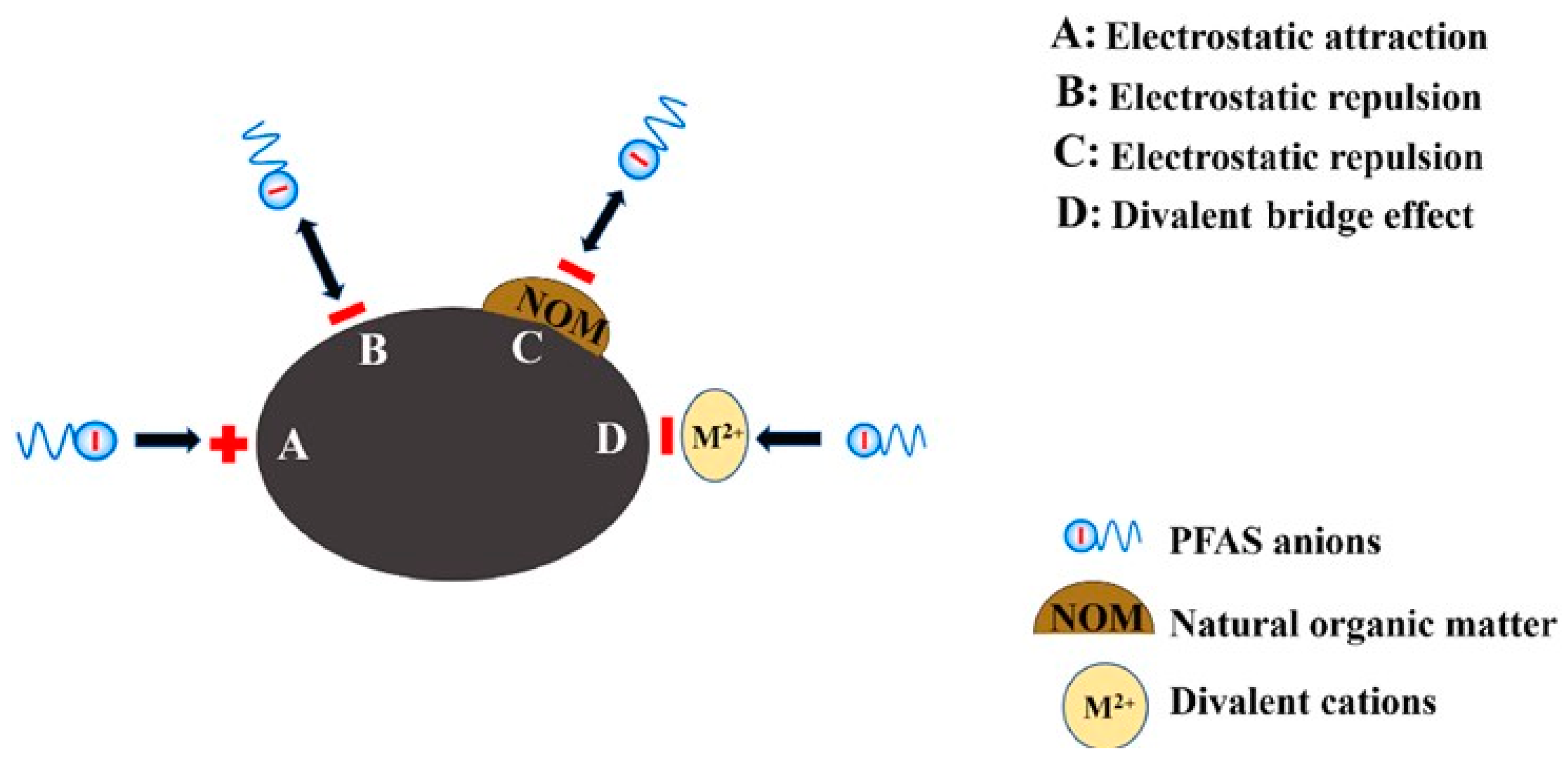
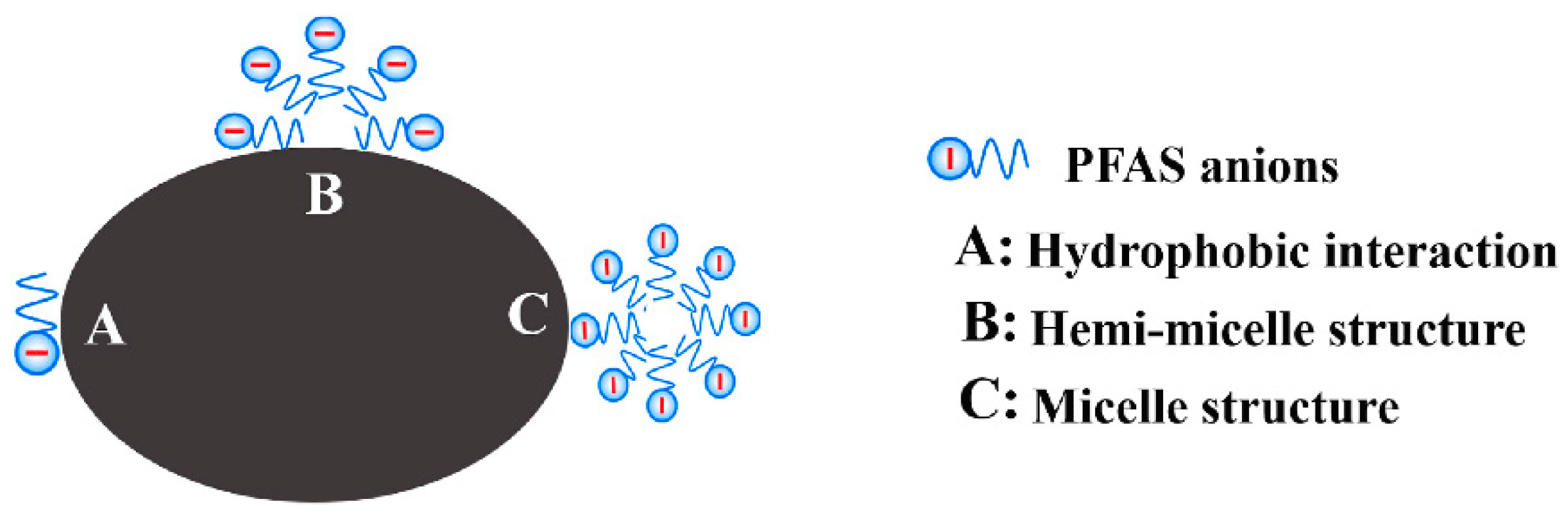
6.1.5. Adsorption Mechanism
6.1.6. Factors Affecting Adsorption
- Particle size
- 2.
- pH
- 3.
- Co-existing ions
- 4.
- Co-existing organic matter
6.2. Anion Exchange Resins
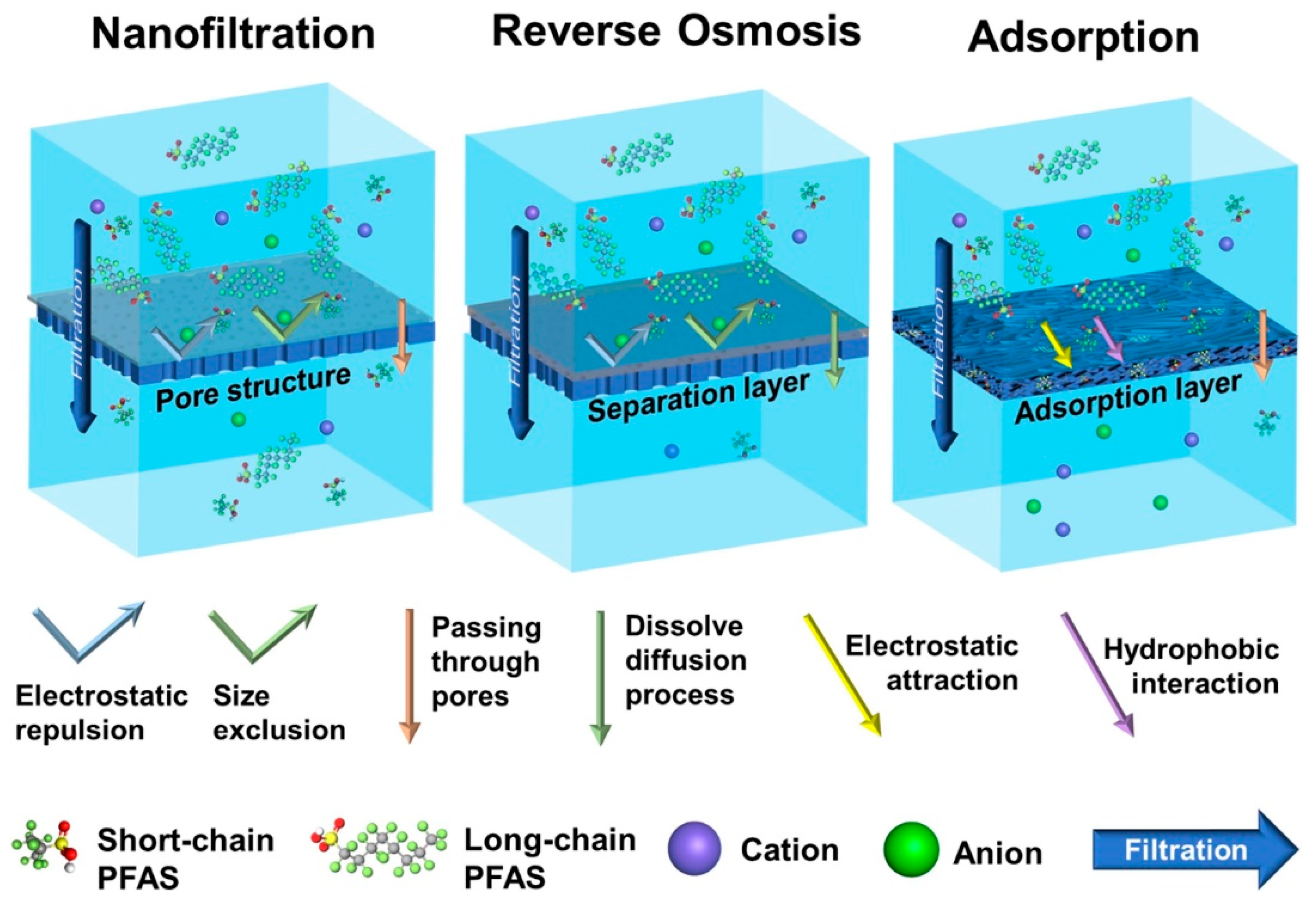
6.3. High-Pressure Membranes
6.4. Electrocoagulation
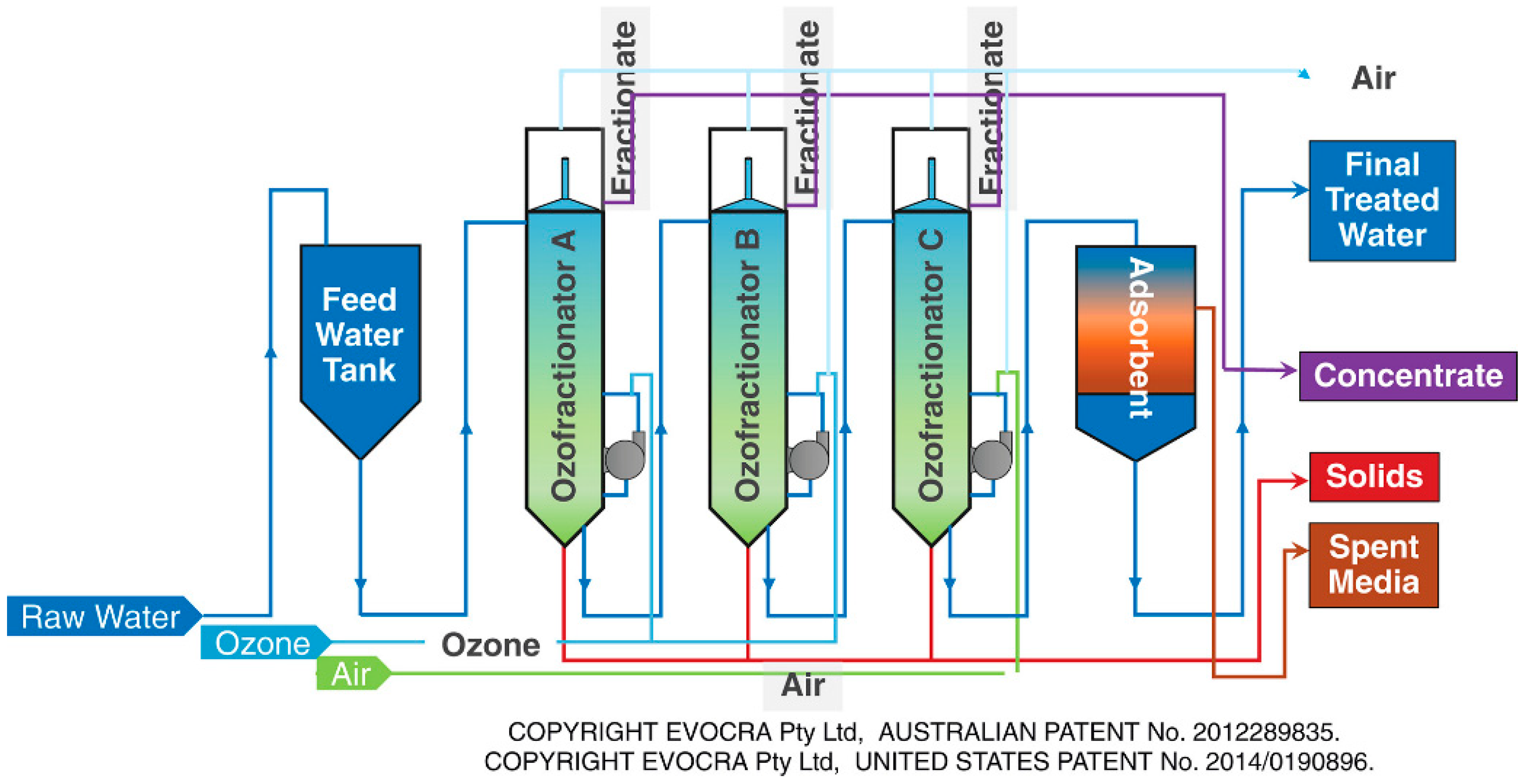
6.5. Foam/Ozon Fractionation
6.6. Biodegradation
6.7. AOPs and ARPs
6.8. Sonolysis-Ultrasonication
6.9. Electrochemical Oxidation
6.10. Non-Thermal Plasma
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- OECD. Reconciling Terminology of the Universe of Per- and Polyfluoroalkyl Substances: Recommendations and Practical Guidance; OECD Series on Risk Management—No. 61; OECD: Paris, France, 2021. [Google Scholar]
- Meegoda, J.N.; Kewalramani, J.A.; Li, B.; Marsh, R.W. A Review of the Applications, Environmental Release, and Remediation Technologies of Per- and Polyfluoroalkyl Substances. Int. J. Environ. Res. Public Health 2020, 17, 8117. [Google Scholar] [CrossRef]
- Teymourian, T.; Teymoorian, T.; Kowsari, E.; Ramakrishna, S. A review of emerging PFAS contaminants: Sources, fate, health risks, and a comprehensive assortment of recent sorbents for PFAS treatment by evaluating their mechanism. Res. Chem. Intermed. 2021, 47, 4879–4914. [Google Scholar] [CrossRef]
- Ehsan, M.N.; Riza, M.; Pervez, N.; Khyum, M.M.O.; Liang, Y.; Naddeo, V. Environmental and health impacts of PFAS: Sources, distribution and sustainable management in North Carolina (USA). Sci. Total Environ. 2023, 878, 163123. [Google Scholar] [CrossRef]
- Mueller, R.; Yingling, V. Per- and Polyfluoroalkyl Substances (PFAS) Fact Sheets; Interstate Technology Regulatory Council Sheets: Washington, DC, USA, 2017. [Google Scholar]
- Brennan, N.M.; Evans, A.T.; Fritz, M.K.; Peak, S.A.; von Holst, H.E. Trends in the Regulation of Per- and Polyfluoroalkyl Substances (PFAS): A Scoping Review. Int. J. Environ. Res. Public Health 2021, 18, 10900. [Google Scholar] [CrossRef]
- Wang, Z.; Buser, A.M.; Cousins, I.T.; Demattio, S.; Drost, W.; Johansson, O.; Ohno, K.; Patlewicz, G.; Richard, A.M.; Walker, G.W.; et al. A New OECD Definition for Per- and Polyfluoroalkyl Substances. Environ. Sci. Technol. 2021, 55, 15575–15578. [Google Scholar] [CrossRef]
- Buck, R.C.; Franklin, J.; Berger, U.; Conder, J.M.; Cousins, I.T.; De Voogt, P.; Jensen, A.A.; Kannan, K.; Mabury, S.A.; Van Leeuwen, S.P.J. Perfluoroalkyl and polyfluoroalkyl substances in the environment: Terminology, classification, and origins. Integr. Environ. Assess. Manag. 2011, 7, 513–541. [Google Scholar] [CrossRef]
- Li, H.; Dong, Q.; Zhang, M.; Gong, T.; Zan, R.; Wang, W. Transport behavior difference and transport model of long- and short-chain per- and polyfluoroalkyl substances in underground environmental media: A review. Environ. Pollut. 2023, 327, 121579. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Zhang, W.; Liang, Y. Adsorption of perfluoroalkyl and polyfluoroalkyl substances (PFASs) from aqueous solution—A review. Sci. Total Environ. 2019, 694, 133606. [Google Scholar] [CrossRef] [PubMed]
- Kjølholt, J.; Jensen, A.A.; Warmning, M. Short-Chain Polyfluoroalkyl Substances (PFAS). A Literature Review on Human Health Effects and Environmental Fate and Effect Aspects of Short-Chain PFAS; Environmental Project No. 1707; Danish Environmental Protection Agency: Odense, Denmark, 2015. [Google Scholar]
- Fenton, S.E.; Ducatman, A.; Boobis, A.; DeWitt, J.C.; Lau, C.; Ng, C.; Smith, J.S.; Roberts, S.M. Per- and Polyfluoroalkyl Substance Toxicity and Human Health Review: Current State of Knowledge and Strategies for Informing Future Research. Environ. Toxicol. Chem. 2020, 40, 606–630. [Google Scholar] [CrossRef]
- Calore, F.; Badetti, E.; Bonetto, A.; Pozzobon, A.; Marcomini, A. Non-conventional sorption materials for the removal of legacy and emerging PFAS from water: A review. Emerg. Contam. 2024, 10, 100303. [Google Scholar] [CrossRef]
- Mahoney, H.; Xie, Y.; Brinkmann, M.; Giesy, J.P. Next generation per- and poly-fluoroalkyl substances: Status and trends, aquatic toxicity, and risk assessment. Eco-Environ. Health 2022, 1, 117–131. [Google Scholar] [CrossRef] [PubMed]
- Dixit, F.; Dutta, R.; Barbeau, B.; Berube, P.; Mohseni, M. PFAS removal by ion exchange resins: A review. Chemosphere 2021, 272, 129777. [Google Scholar] [CrossRef]
- Beale, D.J.; Sinclair, G.M.; Shah, R.; Paten, A.M.; Kumar, A.; Long, S.M.; Vardy, S.; Jones, O.A. A review of omics-based PFAS exposure studies reveals common biochemical response pathways. Sci. Total Environ. 2022, 845, 157255. [Google Scholar] [CrossRef] [PubMed]
- Dvorakova, D.; Jurikova, M.; Svobodova, V.; Parizek, O.; Kozisek, F.; Kotal, F.; Jeligova, H.; Mayerova, L.; Pulkrabova, J. Complex monitoring of perfluoroalkyl substances (PFAS) from tap drinking water in the Czech Republic. Water Res. 2023, 247, 120764. [Google Scholar] [CrossRef]
- Garg, S.; Kumar, P.; Mishra, V.; Guijt, R.; Singh, P.; Dumée, L.F.; Sharma, R.S. A review on the sources, occurrence and health risks of per-/poly-fluoroalkyl substances (PFAS) arising from the manufacture and disposal of electric and electronic products. J. Water Process. Eng. 2020, 38, 101683. [Google Scholar] [CrossRef]
- Post, G.B.; Gleason, J.A.; Cooper, K.R. Key scientific issues in developing drinking water guidelines for perfluoroalkyl acids: Contaminants of emerging concern. PLoS Biol. 2017, 15, e2002855. [Google Scholar] [CrossRef] [PubMed]
- Grandjean, P.; Andersen, E.W.; Budtz-Jørgensen, E.; Nielsen, F.; Mølbak, K.; Weihe, P.; Heilmann, C. Serum Vaccine Antibody Concentrations in Children Exposed to Perfluorinated Compounds. JAMA 2012, 307, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Gallo, V.; Leonardi, G.; Genser, B.; Lopez-Espinosa, M.-J.; Frisbee, S.J.; Karlsson, L.; Ducatman, A.M.; Fletcher, T. Serum Perfluorooctanoate (PFOA) and Perfluorooctane Sulfonate (PFOS) Concentrations and Liver Function Biomarkers in a Population with Elevated PFOA Exposure. Environ. Health Perspect. 2012, 120, 655–660. [Google Scholar] [CrossRef] [PubMed]
- Zeeshan, M.; Yang, Y.; Zhou, Y.; Huang, W.; Wang, Z.; Liu, R.-Q.; Yang, B.-Y.; Hu, L.-W.; Zeng, X.-W.; Sun, X.; et al. Incidence of ocular conditions associated with perfluoroalkyl substances exposure: Isomers of C8 Health Project in China. Environ. Int. 2020, 137, 105555. [Google Scholar] [CrossRef]
- US EPA. Fifth Unregulated Contaminant Monitoring Rule; Environmental Protection Agency: Washington, DC, USA, 2022. [Google Scholar]
- EU Parliament. Directive (EU) 2020/2184 of the European Parliament and of the Council of 16 December 2020 on the Quality of Water Intended for Human Consumption; The European Parliament and of The Council: Brussels, Belgium, 2020. [Google Scholar]
- Barisci, S.; Suri, R. Occurrence and removal of poly/perfluoroalkyl substances (PFAS) in municipal and industrial wastewater treatment plants. Water Sci. Technol. 2021, 84, 3442–3468. [Google Scholar] [CrossRef]
- Lenka, S.P.; Kah, M.; Padhye, L.P. A review of the occurrence, transformation, and removal of poly- and perfluoroalkyl substances (PFAS) in wastewater treatment plants. Water Res. 2021, 199, 117187. [Google Scholar] [CrossRef] [PubMed]
- Zhou, T.; Li, X.; Liu, H.; Dong, S.; Zhang, Z.; Wang, Z.; Li, J.; Nghiem, L.D.; Khan, S.J.; Wang, Q. Occurrence, fate, and remediation for per-and polyfluoroalkyl substances (PFAS) in sewage sludge: A comprehensive review. J. Hazard. Mater. 2024, 466, 133637. [Google Scholar] [CrossRef]
- Kunacheva, C.; Tanaka, S.; Fujii, S.; Boontanon, S.K.; Musirat, C.; Wongwattana, T.; Shivakoti, B.R. Mass flows of perfluorinated compounds (PFCs) in central wastewater treatment plants of industrial zones in Thailand. Chemosphere 2011, 83, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Bossi, R.; Strand, J.; Sortkjær, O.; Larsen, M. Perfluoroalkyl compounds in Danish wastewater treatment plants and aquatic environments. Environ. Int. 2008, 34, 443–450. [Google Scholar] [CrossRef]
- Shigei, M.; Ahrens, L.; Hazaymeh, A.; Dalahmeh, S.S. Per- and polyfluoroalkyl substances in water and soil in wastewater-irrigated farmland in Jordan. Sci. Total Environ. 2020, 716, 137057. [Google Scholar] [CrossRef]
- Chen, H.; Peng, H.; Yang, M.; Hu, J.; Zhang, Y. Detection, Occurrence, and Fate of Fluorotelomer Alcohols in Municipal Wastewater Treatment Plants. Environ. Sci. Technol. 2017, 51, 8953–8961. [Google Scholar] [CrossRef]
- Zhou, Y.; Meng, J.; Zhang, M.; Chen, S.; He, B.; Zhao, H.; Li, Q.; Zhang, S.; Wang, T. Which type of pollutants need to be controlled with priority in wastewater treatment plants: Traditional or emerging pollutants? Environ. Int. 2019, 131, 104982. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, X.; Wang, L.; Zhang, T.; Li, F.; He, N.; Alder, A.C. Perfluoroalkyl compounds in municipal WWTPs in Tianjin, China—Concentrations, distribution and mass flow. Environ. Sci. Pollut. Res. 2012, 19, 1405–1415. [Google Scholar] [CrossRef]
- Lu, J.; Lu, H.; Liang, D.; Feng, S.; Li, Y.; Li, J. A review of the occurrence, monitoring, and removal technologies for the remediation of per- and polyfluoroalkyl substances (PFAS) from landfill leachate. Chemosphere 2023, 332, 138824. [Google Scholar] [CrossRef] [PubMed]
- Wanninayake, D.M. Comparison of currently available PFAS remediation technologies in water: A review. J. Environ. Manag. 2021, 283, 111977. [Google Scholar] [CrossRef]
- Gagliano, E.; Sgroi, M.; Falciglia, P.P.; Vagliasindi, F.G.A.; Roccaro, P. Removal of poly- and perfluoroalkyl substances (PFAS) from water by adsorption: Role of PFAS chain length, effect of organic matter and challenges in adsorbent regeneration. Water Res. 2020, 171, 115381. [Google Scholar] [CrossRef] [PubMed]
- Ross, I.; McDonough, J.; Miles, J.; Storch, P.; Kochunarayanan, P.T.; Kalve, E.; Hurst, J.; Dasgupta, S.S.; Burdick, J. A review of emerging technologies for remediation of PFASs. Remediat. J. 2018, 28, 101–126. [Google Scholar] [CrossRef]
- Belkouteb, N.; Franke, V.; McCleaf, P.; Köhler, S.; Ahrens, L. Removal of per- and polyfluoroalkyl substances (PFASs) in a full-scale drinking water treatment plant: Long-term performance of granular activated carbon (GAC) and influence of flow-rate. Water Res. 2020, 182, 115913. [Google Scholar] [CrossRef] [PubMed]
- Umeh, A.C.; Hassan, M.; Egbuatu, M.; Zeng, Z.; Amin, A.; Samarasinghe, C.; Naidu, R. Multicomponent PFAS sorption and desorption in common commercial adsorbents: Kinetics, isotherm, adsorbent dose, pH, and index ion and ionic strength effects. Sci. Total Environ. 2023, 904, 166568. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Ulrich, B.A.; Chen, B.; Higgins, C.P. Sorption of Poly- and Perfluoroalkyl Substances (PFASs) Relevant to Aqueous Film-Forming Foam (AFFF)-Impacted Groundwater by Biochars and Activated Carbon. Environ. Sci. Technol. 2017, 51, 6342–6351. [Google Scholar] [CrossRef] [PubMed]
- Beesley, L.; Moreno-Jiménez, E.; Gomez-Eyles, J.L.; Harris, E.; Robinson, B.; Sizmur, T. A review of biochars’ potential role in the remediation, revegetation and restoration of contaminated soils. Environ. Pollut. 2011, 159, 3269–3282. [Google Scholar] [CrossRef]
- Bei, Y.; Deng, S.; Du, Z.; Wang, B.; Huang, J.; Yu, G. Adsorption of perfluorooctane sulfonate on carbon nanotubes: Influence of pH and competitive ions. Water Sci. Technol. 2014, 69, 1489–1495. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Li, D.; Li, C.; Ji, R.; Tian, X. Metal nanoparticles by doping carbon nanotubes improved the sorption of perfluorooctanoic acid. J. Hazard. Mater. 2018, 351, 206–214. [Google Scholar] [CrossRef] [PubMed]
- Oyetade, O.A.; Varadwaj, G.B.B.; Nyamori, V.O.; Jonnalagadda, S.B.; Martincigh, B.S. A critical review of the occurrence of perfluoroalkyl acids in aqueous environments and their removal by adsorption onto carbon nanotubes. Rev. Environ. Sci. Biotechnol. 2018, 17, 603–635. [Google Scholar] [CrossRef]
- Ateia, M.; Attia, M.F.; Maroli, A.S.; Tharayil, N.; Alexis, F.; Whitehead, D.C.; Karanfil, T. Rapid Removal of Poly- and Perfluorinated Alkyl Substances by Poly(ethylenimine)-Functionalized Cellulose Microcrystals at Environmentally Relevant Conditions. Environ. Sci. Technol. Lett. 2018, 5, 764–769. [Google Scholar] [CrossRef]
- Yang, Y.; Yang, M.; Zheng, Z.; Zhang, X. Highly effective adsorption removal of perfluorooctanoic acid (PFOA) from aqueous solution using calcined layer-like Mg-Al hydrotalcites nanosheets. Environ. Sci. Pollut. Res. 2020, 27, 13396–13408. [Google Scholar] [CrossRef] [PubMed]
- Hellsing, M.S.; Josefsson, S.; Hughes, A.V.; Ahrens, L. Sorption of perfluoroalkyl substances to two types of minerals. Chemosphere 2016, 159, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.-H.; Jiang, W.-T.; Li, Z. Removal of perfluorooctanoic acid from water using calcined hydrotalcite—A mechanistic study. J. Hazard. Mater. 2019, 368, 487–495. [Google Scholar] [CrossRef] [PubMed]
- Chang, P.-H.; Li, Z.; Jiang, W.-T. Calcination of hydrotalcite to enhance the removal of perfluorooctane sulfonate from water. Appl. Clay Sci. 2020, 190, 105563. [Google Scholar] [CrossRef]
- Lei, X.; Lian, Q.; Zhang, X.; Karsili, T.K.; Holmes, W.; Chen, Y.; Zappi, M.E.; Gang, D.D. A review of PFAS adsorption from aqueous solutions: Current approaches, engineering applications, challenges, and opportunities. Environ. Pollut. 2023, 321, 121138. [Google Scholar] [CrossRef] [PubMed]
- Pauletto, P.S.; Bandosz, T.J. Activated carbon versus metal-organic frameworks: A review of their PFAS adsorption performance. J. Hazard. Mater. 2022, 425, 127810. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Cao, H.; Pan, W.; Wang, C.; Liang, Y. The role of dissolved organic matter during Per- and Polyfluorinated Substance (PFAS) adsorption, degradation, and plant uptake: A review. J. Hazard. Mater. 2022, 436, 129139. [Google Scholar] [CrossRef]
- Falciglia, P.P.; Malarbì, D.; Maddalena, R.; Greco, V.; Vagliasindi, F.G. Remediation of Hg-contaminated marine sediments by simultaneous application of enhancing agents and microwave heating (MWH). Chem. Eng. J. 2017, 321, 1–10. [Google Scholar] [CrossRef]
- Wang, W.; Mi, X.; Zhou, Z.; Zhou, S.; Li, C.; Hu, X.; Qi, D.; Deng, S. Novel insights into the competitive adsorption behavior and mechanism of per- and polyfluoroalkyl substances on the anion-exchange resin. J. Colloid Interface Sci. 2019, 557, 655–663. [Google Scholar] [CrossRef]
- Park, M.; Daniels, K.D.; Wu, S.; Ziska, A.D.; Snyder, S.A. Magnetic ion-exchange (MIEX) resin for perfluorinated alkylsubstance (PFAS) removal in groundwater: Roles of atomic charges for adsorption. Water Res. 2020, 181, 115897. [Google Scholar] [CrossRef]
- Crone, B.C.; Speth, T.F.; Wahman, D.G.; Smith, S.J.; Abulikemu, G.; Kleiner, E.J.; Pressman, J.G. Occurrence of per- and polyfluoroalkyl substances (PFAS) in source water and their treatment in drinking water. Crit. Rev. Environ. Sci. Technol. 2019, 49, 2359–2396. [Google Scholar] [CrossRef]
- Jin, T.; Peydayesh, M.; Mezzenga, R. Membrane-based technologies for per- and poly-fluoroalkyl substances (PFASs) removal from water: Removal mechanisms, applications, challenges and perspectives. Environ. Int. 2021, 157, 106876. [Google Scholar] [CrossRef]
- Appleman, T.D.; Dickenson, E.R.; Bellona, C.; Higgins, C.P. Nanofiltration and granular activated carbon treatment of perfluoroalkyl acids. J. Hazard. Mater. 2013, 260, 740–746. [Google Scholar] [CrossRef]
- Steinle-Darling, E.; Reinhard, M. Nanofiltration for trace organic contaminant removal: Structure, solution, and membrane fouling effects on the rejection of perfluorochemicals. Environ. Sci. Technol. 2008, 42, 5292–5297. [Google Scholar] [CrossRef]
- Thompson, J.; Eaglesham, G.; Reungoat, J.; Poussade, Y.; Bartkow, M.; Lawrence, M.; Mueller, J.F. Removal of PFOS, PFOA and other perfluoroalkyl acids at water reclamation plants in South East Queensland Australia. Chemosphere 2011, 82, 9–17. [Google Scholar] [CrossRef]
- Leung, S.C.E.; Shukla, P.; Chen, D.; Eftekhari, E.; An, H.; Zare, F.; Ghasemi, N.; Zhang, D.; Nguyen, N.-T.; Li, Q. Emerging technologies for PFOS/PFOA degradation and removal: A review. Sci. Total Environ. 2022, 827, 153669. [Google Scholar] [CrossRef]
- Lin, H.; Wang, Y.; Niu, J.; Yue, Z.; Huang, Q. Efficient Sorption and Removal of Perfluoroalkyl Acids (PFAAs) from Aqueous Solution by Metal Hydroxides Generated in Situ by Electrocoagulation. Environ. Sci. Technol. 2015, 49, 10562–10569. [Google Scholar] [CrossRef]
- Hubert, M.; Meyn, T.; Hansen, M.C.; Hale, S.E.; Arp, H.P.H. Per- and polyfluoroalkyl substance (PFAS) removal from soil washing water by coagulation and flocculation. Water Res. 2024, 249, 120888. [Google Scholar] [CrossRef]
- Wang, Y.; Lin, H.; Jin, F.; Niu, J.; Zhao, J.; Bi, Y.; Li, Y. Electrocoagulation mechanism of perfluorooctanoate (PFOA) on a zinc anode: Influence of cathodes and anions. Sci. Total Environ. 2016, 557–558, 542–550. [Google Scholar] [CrossRef]
- Yang, B.; Han, Y.; Yu, G.; Zhuo, Q.; Deng, S.; Wu, J.; Zhang, P. Efficient removal of perfluoroalkyl acids (PFAAs) from aqueous solution by electrocoagulation using iron electrode. Chem. Eng. J. 2016, 303, 384–390. [Google Scholar] [CrossRef]
- Baudequin, C.; Couallier, E.; Rakib, M.; Deguerry, I.; Severac, R.; Pabon, M. Purification of firefighting water containing a fluorinated surfactant by reverse osmosis coupled to electrocoagulation–filtration. Sep. Purif. Technol. 2011, 76, 275–282. [Google Scholar] [CrossRef]
- Burns, D.J.; Stevenson, P.; Murphy, P.J.C. PFAS removal from groundwaters using Surface-Active Foam Fractionation. Remediat. J. 2021, 31, 19–33. [Google Scholar] [CrossRef]
- Dai, X.; Xie, Z.; Dorian, B.; Gray, S.; Zhang, J. Comparative study of PFAS treatment by UV, UV/ozone, and fractionations with air and ozonated air. Environ. Sci. 2019, 5, 1897–1907. [Google Scholar] [CrossRef]
- Horst, J.; McDonough, J.; Ross, I.; Dickson, M.; Miles, J.; Hurst, J.; Storch, P. Water Treatment Technologies for PFAS: The Next Generation. Groundw. Monit. Remediat. 2018, 38, 13–23. [Google Scholar] [CrossRef]
- Wang, Y.; Munir, U.; Huang, Q. Occurrence of per- and polyfluoroalkyl substances (PFAS) in soil: Sources, fate, and remediation. Soil Environ. Health 2023, 1, 100004. [Google Scholar] [CrossRef]
- Vo, H.N.P.; Ngo, H.H.; Guo, W.; Nguyen, T.M.H.; Li, J.; Liang, H.; Deng, L.; Chen, Z.; Nguyen, T.A.H. Poly-and perfluoroalkyl substances in water and wastewater: A comprehensive review from sources to remediation. J. Water Process. Eng. 2020, 36, 101393. [Google Scholar] [CrossRef]
- Beškoski, V.P.; Yamamoto, A.; Nakano, T.; Yamamoto, K.; Matsumura, C.; Motegi, M.; Beškoski, L.S.; Inui, H. Defluorination of perfluoroalkyl acids is followed by production of monofluorinated fatty acids. Sci. Total Environ. 2018, 636, 355–359. [Google Scholar] [CrossRef]
- Huang, S.; Jaffé, P.R. Defluorination of Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS) by Acidimicrobium sp. Strain A6. Environ. Sci. Technol. 2019, 53, 11410–11419. [Google Scholar] [CrossRef]
- Capodaglio, A.G. Critical Perspective on Advanced Treatment Processes for Water and Wastewater: AOPs, ARPs, and AORPs. Appl. Sci. 2020, 10, 4549. [Google Scholar] [CrossRef]
- Vatankhah, H.; Anderson, R.H.; Ghosh, R.; Willey, J.; Leeson, A. A review of innovative approaches for onsite management of PFAS-impacted investigation derived waste. Water Res. 2023, 247, 120769. [Google Scholar] [CrossRef]
- Park, S.; Lee, L.S.; Medina, V.F.; Zull, A.; Waisner, S. Heat-activated persulfate oxidation of PFOA, 6:2 fluorotelomer sulfonate, and PFOS under conditions suitable for in-situ groundwater remediation. Chemosphere 2016, 145, 376–383. [Google Scholar] [CrossRef]
- Jin, L.; Zhang, P.; Shao, T.; Zhao, S. Ferric ion mediated photodecomposition of aqueous perfluorooctane sulfonate (PFOS) under UV irradiation and its mechanism. J. Hazard. Mater. 2014, 271, 9–15. [Google Scholar] [CrossRef]
- Nzeribe, B.N.; Crimi, M.; Thagard, S.M.; Holsen, T.M. Physico-Chemical Processes for the Treatment of Per- And Polyfluoroalkyl Substances (PFAS): A review. Crit. Rev. Environ. Sci. Technol. 2019, 49, 866–915. [Google Scholar] [CrossRef]
- Park, H.; Vecitis, C.D.; Cheng, J.; Dalleska, N.F.; Mader, B.T.; Hoffmann, M.R. Reductive degradation of perfluoroalkyl compounds with aquated electrons generated from iodide photolysis at 254 nm. Photochem. Photobiol. Sci. 2011, 10, 1945–1953. [Google Scholar] [CrossRef]
- Qu, Y.; Zhang, C.; Li, F.; Chen, J.; Zhou, Q. Photo-reductive defluorination of perfluorooctanoic acid in water. Water Res. 2010, 44, 2939–2947. [Google Scholar] [CrossRef]
- Zhang, C.; Qu, Y.; Zhao, X.; Zhou, Q. Photoinduced Reductive Decomposition of Perflurooctanoic Acid in Water: Effect of Temperature and Ionic Strength. Clean 2015, 43, 223–228. [Google Scholar] [CrossRef]
- Ambaye, T.G.; Vaccari, M.; Prasad, S.; Rtimi, S. Recent progress and challenges on the removal of per- and poly-fluoroalkyl substances (PFAS) from contaminated soil and water. Environ. Sci. Pollut. Res. 2022, 29, 58405–58428. [Google Scholar] [CrossRef]
- Rodriguez-Freire, L.; Balachandran, R.; Sierra-Alvarez, R.; Keswani, M. Effect of sound frequency and initial concentration on the sonochemical degradation of perfluorooctane sulfonate (PFOS). J. Hazard. Mater. 2015, 300, 662–669. [Google Scholar] [CrossRef]
- Lei, Y.-J.; Tian, Y.; Sobhani, Z.; Naidu, R.; Fang, C. Synergistic degradation of PFAS in water and soil by dual-frequency ultrasonic activated persulfate. Chem. Eng. J. 2020, 388, 124215. [Google Scholar] [CrossRef]
- Cao, H.; Zhang, W.; Wang, C.; Liang, Y. Sonochemical degradation of poly- and perfluoroalkyl substances—A review. Ultrason. Sonochem. 2020, 69, 105245. [Google Scholar] [CrossRef] [PubMed]
- Kucharzyk, K.H.; Darlington, R.; Benotti, M.; Deeb, R.; Hawley, E. Novel treatment technologies for PFAS compounds: A critical review. J. Environ. Manag. 2017, 204, 757–764. [Google Scholar] [CrossRef] [PubMed]
- Babu, S.G.; Ashokkumar, M.; Neppolian, B. The role of ultrasound on advanced oxidation processes. Top. Curr. Chem. 2016, 374, 75. [Google Scholar] [CrossRef] [PubMed]
- Schaefer, C.E.; Andaya, C.; Urtiaga, A.; McKenzie, E.R.; Higgins, C.P. Electrochemical treatment of perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS) in groundwater impacted by aqueous film forming foams (AFFFs). J. Hazard. Mater. 2015, 295, 170–175. [Google Scholar] [CrossRef] [PubMed]
- Trautmann, A.M.; Schell, H.; Schmidt, K.R.; Mangold, K.-M.; Tiehm, A. Electrochemical degradation of perfluoroalkyl and polyfluoroalkyl substances (PFASs) in groundwater. Water Sci. Technol. 2015, 71, 1569–1575. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.K.; Fernando, S.; Baygi, S.F.; Multari, N.; Thagard, S.M.; Holsen, T.M. Breakdown Products from Perfluorinated Alkyl Substances (PFAS) Degradation in a Plasma-Based Water Treatment Process. Environ. Sci. Technol. 2019, 53, 2731–2738. [Google Scholar] [CrossRef]
- Alam, D.; Lee, S.; Hong, J.; Fletcher, D.F.; McClure, D.; Cook, D.; Cullen, P.; Kavanagh, J.M. Experimental investigations of Per- and Poly-fluoroalkyl substances (PFAS) degradation by non-thermal plasma in aqueous solutions. J. Environ. Chem. Eng. 2023, 11, 111588. [Google Scholar] [CrossRef]


| Number of Carbons | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 |
| PFCAs | Short-chain PFCAs | Long-chain PFCAs | |||||||
| PFBS | PFPeA | PFHxA | PFHpA | PFOA | PFNA | PFDA | PFUnA | PFDoA | |
| PFSAs | PFBS | PFPeS | PFHxS | PFHpS | PFOS | PFNS | PFDS | PFUnS | PFDoS |
| Short-chain PFSAs | Long-chain PFSAs | ||||||||
| Legacy PFAS | |||
| PFOA |  | C8HF15O2 | PFOA |
| PFOS | 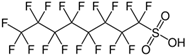 | C8HF17O3S | PFOS |
| Emerging new-generation PFAS | |||
| PFNA | 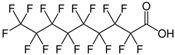 | C9HF17O2 | PFNA |
| HFPO-DA, GenX Chemicals | 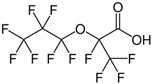 | C6HF11O3 | HFPO-DA, GenX Chemicals |
| PFHxS | 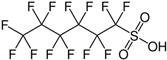 | C6HF13O3S | PFHxS |
| PFBS |  | C4HF9O3S | PFBS |
| Parameter | Parametric Value | Unit | Notes |
|---|---|---|---|
| PFAS Total | 0.5 | μg/L | “PFAS Total” indicates the sum of per- and poly-fluoroalkyl substances. This parametric value is applied when the necessary technical guidelines for monitoring this parameter are developed in accordance with Article 13 (7). EU member states can then adopt either one or both of the parameters ‘PFAS Total’ or “Sum of PFAS”. |
| Sum of PFAS | 0.1 | μg/L | “Sum of PFAS” indicates the sum of the most concerning per- and polyfluoroalkyl substances as to water intended for human consumption listed in point 3 of Part B of Annex III. This is a subsection of “PFAS Total” substances that contain a perfluor-oalkyl moiety with three or more carbons (i.e., –CnF2n–, n ≥ 3) or a perfluoroalkyl-lether moiety with two or more carbons (i.e. –CnF2nOCmF2m–, n and m ≥ 1). |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malouchi, N.; Chatzimichailidou, S.; Tolkou, A.K.; Kyzas, G.Z.; Calgaro, L.; Marcomini, A.; Katsoyiannis, I.A. The Removal of Per- and Poly-Fluoroalkyl Substances from Water: A Review on Destructive and Non-Destructive Methods. Separations 2024, 11, 122. https://doi.org/10.3390/separations11040122
Malouchi N, Chatzimichailidou S, Tolkou AK, Kyzas GZ, Calgaro L, Marcomini A, Katsoyiannis IA. The Removal of Per- and Poly-Fluoroalkyl Substances from Water: A Review on Destructive and Non-Destructive Methods. Separations. 2024; 11(4):122. https://doi.org/10.3390/separations11040122
Chicago/Turabian StyleMalouchi, Natalia, Stella Chatzimichailidou, Athanasia K. Tolkou, George Z. Kyzas, Loris Calgaro, Antonio Marcomini, and Ioannis A. Katsoyiannis. 2024. "The Removal of Per- and Poly-Fluoroalkyl Substances from Water: A Review on Destructive and Non-Destructive Methods" Separations 11, no. 4: 122. https://doi.org/10.3390/separations11040122
APA StyleMalouchi, N., Chatzimichailidou, S., Tolkou, A. K., Kyzas, G. Z., Calgaro, L., Marcomini, A., & Katsoyiannis, I. A. (2024). The Removal of Per- and Poly-Fluoroalkyl Substances from Water: A Review on Destructive and Non-Destructive Methods. Separations, 11(4), 122. https://doi.org/10.3390/separations11040122










