Comprehensive Profiling of Terpenes and Terpenoids in Different Cannabis Strains Using GC × GC-TOFMS
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cannabis Strains and Chemical Standards
2.2. Preparation of Extracts and Standard Solutions
2.3. Analytical Instrument
2.4. Data Analysis
3. Results and Discussion
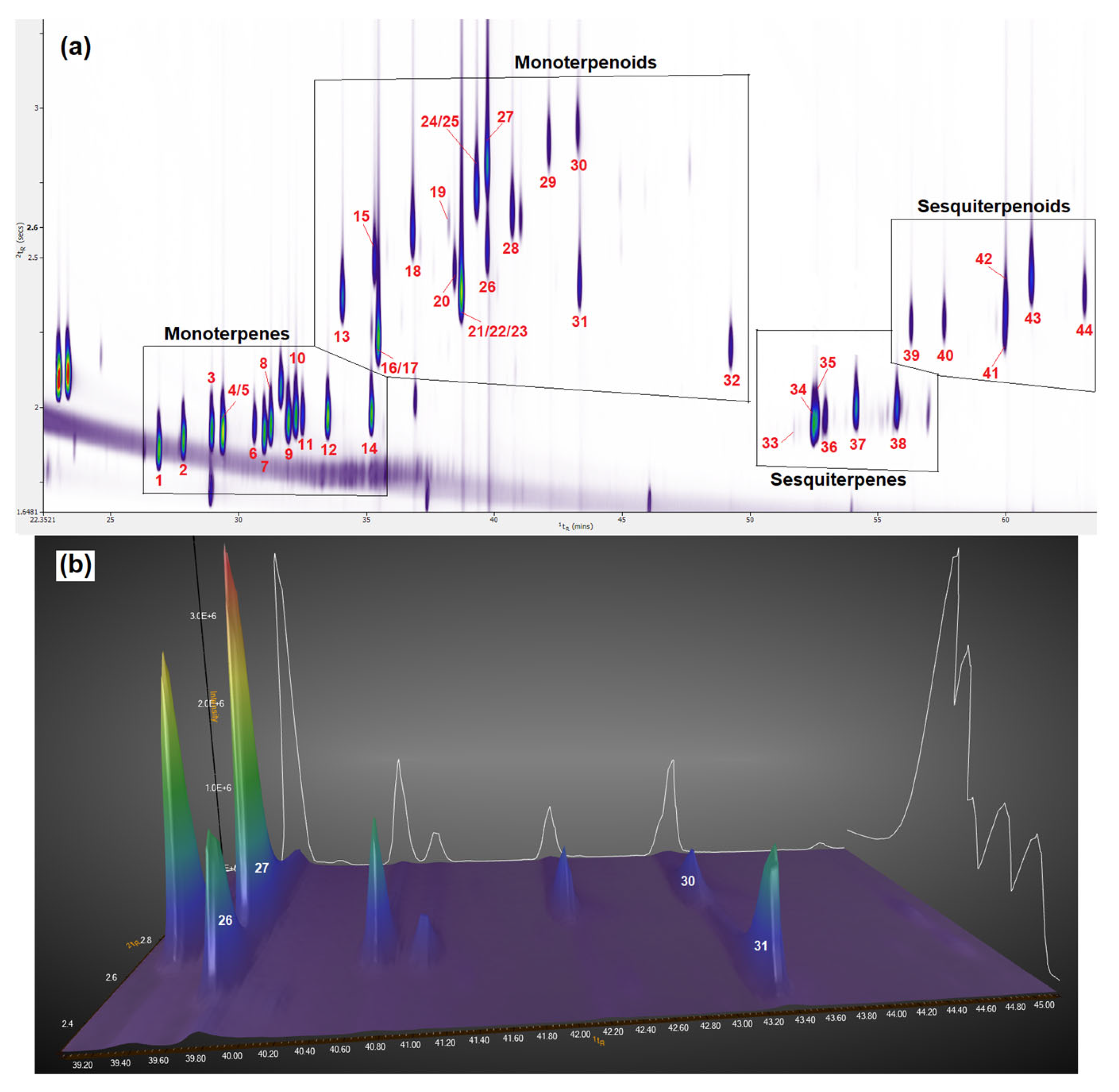
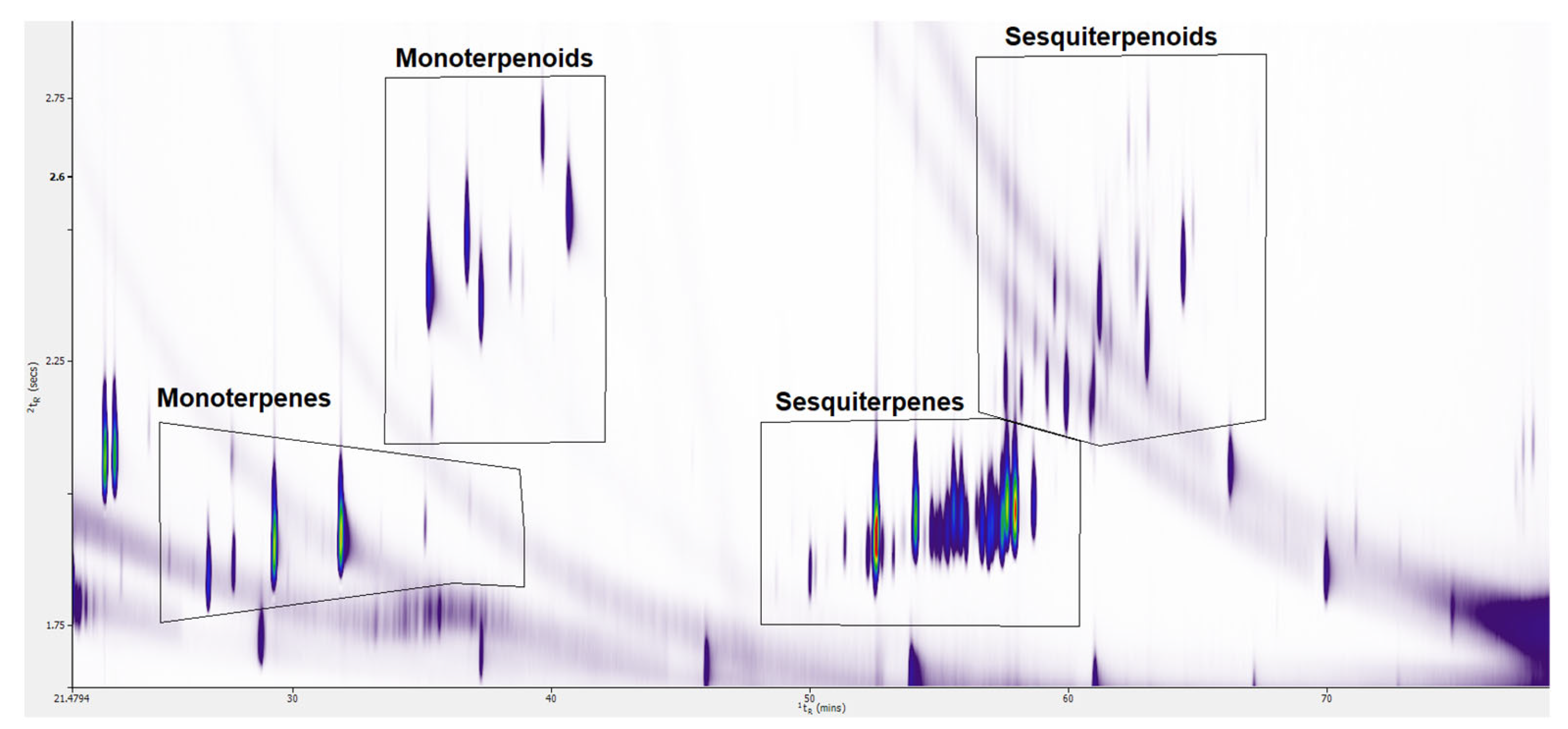
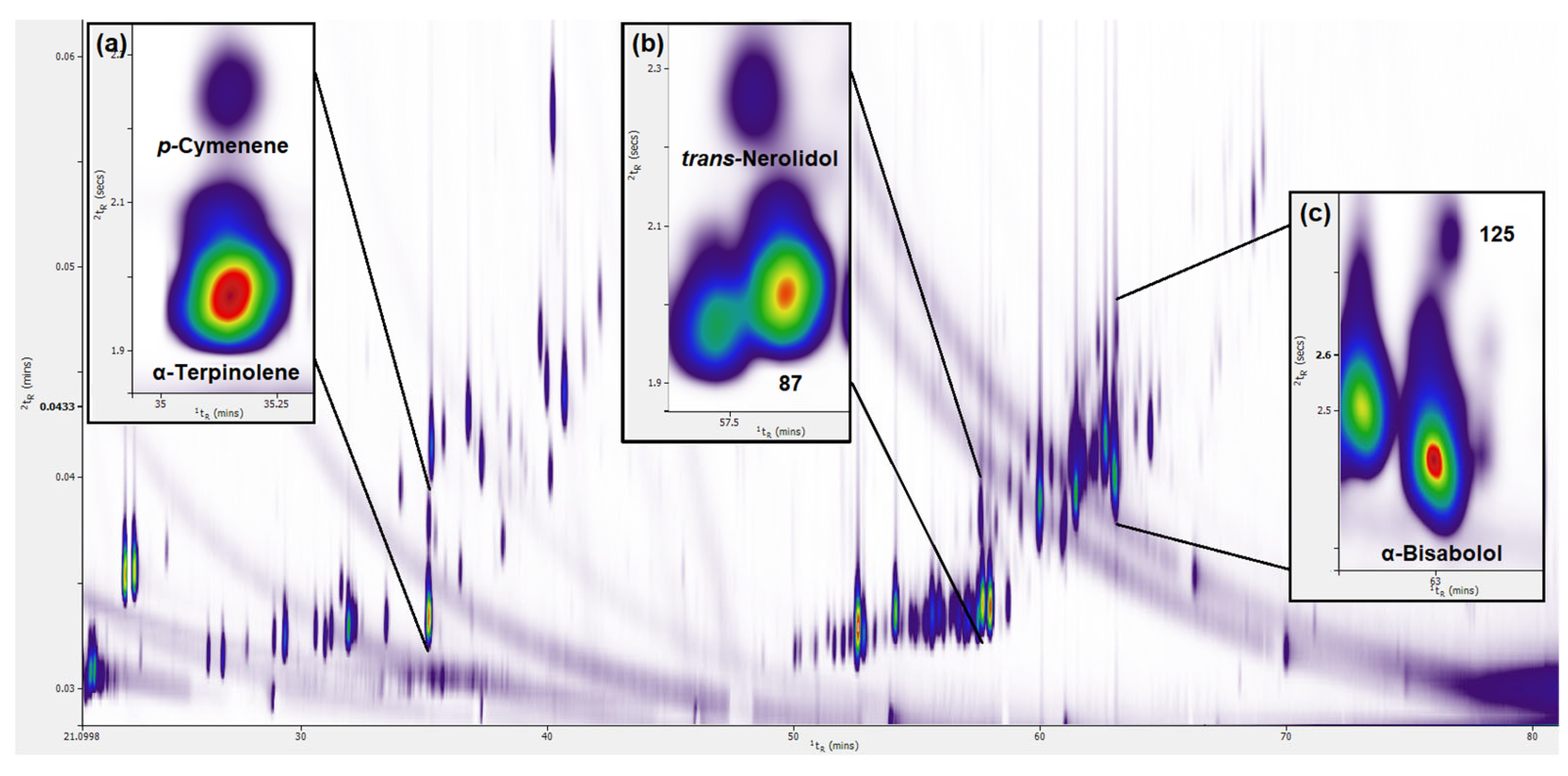
3.1. Enhanced Separation of Terpenes and Terpenoids in the Second Dimension Using GC × GC-TOFMS
3.1.1. Enhanced Separation of Terpenes and Terpenoids in the Standard Mixture
3.1.2. Enhanced Separation of Terpenes and Terpenoids in Six Cannabis Strains
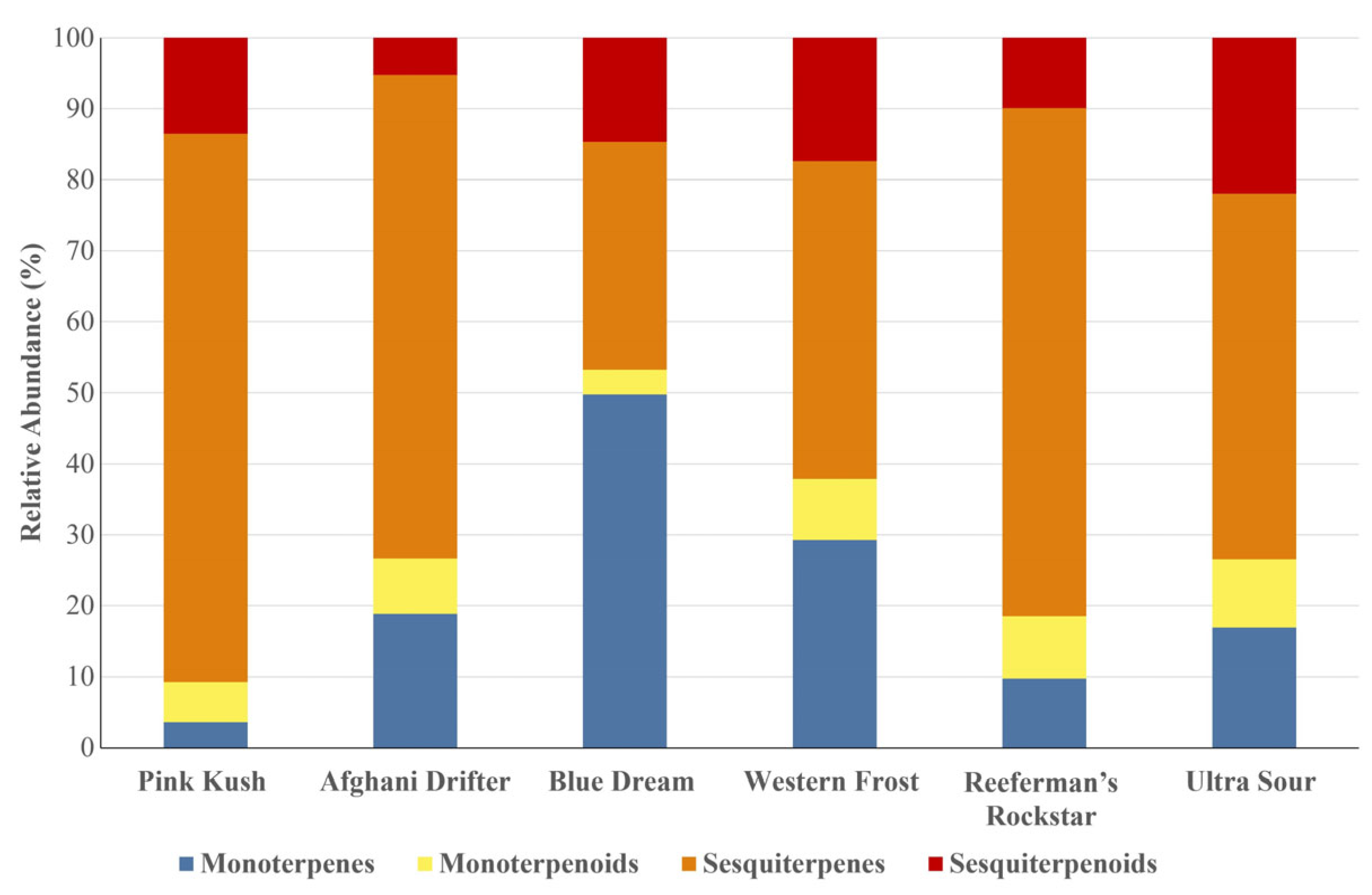
3.2. Identification of Terpenes and Terpenoids in Six Cannabis Strains
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bennett, C. Early/ancient history. In The Pot Book: A Complete Guide to Cannabis; Holland, J., Ed.; Park Street Press: Rochester, NY, USA, 2010; ISBN 1594773688. [Google Scholar]
- Manthey, J.; Freeman, T.P.; Kilian, C.; Lopez-Pelayo, H.; Rehm, J. Public health monitoring of cannabis use in Europe: Prevalence of use, cannabis potency, and treatment rates. Lancet. Reg. Health Eur. 2021, 10, 100227. [Google Scholar] [CrossRef] [PubMed]
- Radwan, M.M.; Chandra, S.; Gul, S.; ElSohly, M.A. Cannabinoids, Phenolics, Terpenes and Alkaloids of Cannabis. Molecules 2021, 26, 2774. [Google Scholar] [CrossRef] [PubMed]
- Elsohly, M.A.; Slade, D. Chemical constituents of marijuana: The complex mixture of natural cannabinoids. Life Sci. 2005, 78, 539–548. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.A.; Ross, S.A.; Slade, D.; Radwan, M.M.; Khan, I.A.; Elsohly, M.A. Minor oxygenated cannabinoids from high potency Cannabis sativa L. Phytochemistry 2015, 117, 194–199. [Google Scholar] [CrossRef] [PubMed]
- Jagannathan, R. Identification of Psychoactive Metabolites from Cannabis sativa, Its Smoke, and Other Phytocannabinoids Using Machine Learning and Multivariate Methods. ACS Omega 2020, 5, 281–295. [Google Scholar] [CrossRef]
- Dos Santos, R.G.; Hallak, J.E.C.; Crippa, J.A.S. Neuropharmacological Effects of the Main Phytocannabinoids: A Narrative Review. In Advances in Experimental Medicine and Biology; Murillo-Rodriguez, E., Pandi-Perumal, S.R., Monti, J.M., Eds.; Springer: Cham, Switzerland; New York City, NY, USA, 2020; Volume 1264, pp. 29–45. ISBN 978-3-030-57368-3. [Google Scholar]
- Mabou, F.D.; Yossa, I.B.N. TERPENES: Structural classification and biological activities. IOSR J. Pharm. Biol. Sci. 2021, 16, 25–40. [Google Scholar] [CrossRef]
- Masyita, A.; Sar, R.M.; Astuti, A.D.; Yasir, B.; Rumata, N.R.; Emran, T.B.; Nainu, F.; Simal-Gandara, J. Terpenes and terpenoids as main bioactive compounds of essential oils, their roles in human health and potential application as natural food preservatives. Food Chem. X 2022, 13, 100217. [Google Scholar] [CrossRef]
- Lowe, H.; Steele, B.; Bryant, J.; Toyang, N.; Ngwa, W. Non-Cannabinoid Metabolites of Cannabis sativa L. with Therapeutic Potential. Plants 2021, 10, 400. [Google Scholar] [CrossRef]
- Buchbauer, G.; Jirovetz, L.; Jager, W. Aromatherapy: Evidence for Sedative Effects of the Essential Oil of Lavender after Inhalation. Z. Naturforsch. C. J. Biosci. 1991, 46, 1067–1072. [Google Scholar] [CrossRef]
- Surendran, S.; Qassadi, F.; Surendran, G.; Lilley, D.; Heinrich, M. Myrcene-What Are the Potential Health Benefits of This Flavouring and Aroma Agent? Front. Nutr. 2021, 8, 699666. [Google Scholar] [CrossRef]
- Ferber, S.G.; Namdar, D.; Hen-Shoval, D.; Eger, G.; Koltai, H.; Shoval, G.; Shbiro, L.; Weller, A. The “Entourage Effect”: Terpenes Coupled with Cannabinoids for the Treatment of Mood Disorders and Anxiety Disorders. Curr. Pharmacol. 2020, 18, 87–96. [Google Scholar] [CrossRef] [PubMed]
- Chacon, F.T.; Raup-Konsavage, W.M.; Vrana, K.E.; Kellogg, J.J. Secondary Terpenes in Cannabis sativa L.: Synthesis and Synergy. Biomedicines 2022, 10, 3142. [Google Scholar] [CrossRef] [PubMed]
- Katiki, L.M.; Barbieri, A.M.E.; Araujo, R.C.; Verissimo, C.J.; Louvandini, H.; Ferreira, J.F.S. Synergistic interaction of ten essential oils against Haemonchus contortus in vitro. Vet. Parasitol. 2017, 243, 47–51. [Google Scholar] [CrossRef] [PubMed]
- Pavithra, P.S.; Mehta, A.; Verma, R.S. Synergistic interaction of β-caryophyllene with aromadendrene oxide 2 and phytol induces apoptosis on skin epidermoid cancer cells. Phytomedicine 2018, 47, 121–134. [Google Scholar] [CrossRef] [PubMed]
- LaVigne, J.E.; Hecksel, R.; Keresztes, A.; Streicher, J.M. Cannabis sativa terpenes are cannabimimetic and selectively enhance cannabinoid activity. Sci. Rep. 2021, 11, 8232. [Google Scholar] [CrossRef] [PubMed]
- Krill, C.; Rochfort, S.; Spangenberg, G. A High-Throughput Method for the Comprehensive Analysis of Terpenes and Terpenoids in Medicinal Cannabis Biomass. Metabolites 2020, 10, 276. [Google Scholar] [CrossRef] [PubMed]
- Bakain, R.Z.A.; Al-Degs, Y.S.; Cizdziel, J.V.; ElSohly, M.A. Comprehensive chromatographic profiling of cannabis from 23 USA States marketed for medical purposes. Acta Chromatogr. 2021, 33, 78–90. [Google Scholar] [CrossRef]
- Jin, D.; Dai, K.; Xie, Z.; Chen, J. Secondary Metabolites Profiled in Cannabis Inflorescences, Leaves, Stem Barks, and Roots for Medicinal Purposes. Sci. Rep. 2020, 10, 3309. [Google Scholar] [CrossRef]
- Arnoldi, S.; Roda, G.; Casagni, E.; Acqua, L.D.; Cas, M.D.; Fare, F.; Rusconi, C.; Visconti, L.; Gambaro, V. Characterization of the Volatile Components of Cannabis Preparations by Solid-Phase Microextraction Coupled to Headspace-Gas Chromatography with Mass Detector (SPME-HSGC/MS). J. Chromatogr. Sep. Tech. 2017, 8, 1000350. [Google Scholar] [CrossRef]
- Boswell, H.; Carrillo, K.T.; Gorecki, T. Evaluation of the Performance of Cryogen-Free Thermal Modulation-Based Comprehensive Two-Dimensional Gas Chromatography-Time-of-Flight Mass Spectrometry (GC × GC-TOFMS) for the Qualitative Analysis of a Complex Bitumen Sample. Separations 2020, 7, 13. [Google Scholar] [CrossRef]
- Dubois, L.M.; Aczon, S.; Focant, J.; Perrault, K.A. Translation of a One-Dimensional to a Comprehensive Two-Dimensional Gas Chromatography Method with Dual-Channel Detection for Volatile Organic Compound Measurement in Forensic Applications. Anal. Chem. 2020, 92, 10091–10098. [Google Scholar] [CrossRef] [PubMed]
- Rees, C.A.; Franchina, F.A.; Nordick, K.V.; Kim, P.J.; Hill, J.E. Expanding the Klebsiella pneumoniae volatile metabolome using advanced analytical instrumentation for the detection of novel metabolites. J. Appl. Microbiol. 2016, 122, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Almstetter, M.F.; Oefner, P.J.; Dettmer, K. Comprehensive two-dimensional gas chromatography in metabolomics. Anal. Bioanal. Chem. 2012, 402, 1993–2013. [Google Scholar] [CrossRef] [PubMed]
- Marchini, M.; Charvoz, C.; Dujourdy, L.; Baldovini, N.; Filippi, J. Multidimensional analysis of cannabis volatile constituents: Identification of 5,5-dimethyl-1-vinylbicyclo[2.1.1]hexane as a volatile marker of hashish, the resin of Cannabis sativa L. J. Chromatogr. A 2014, 1370, 200–215. [Google Scholar] [CrossRef] [PubMed]
- Franchina, F.A.; Dubois, L.M.; Focant, J.F. In-Depth Cannabis Multiclass Metabolite Profiling Using Sorptive Extraction and Multidimensional Gas Chromatography with Low- and High-Resolution Mass Spectrometry. Anal. Chem. 2020, 92, 10512–10520. [Google Scholar] [CrossRef] [PubMed]
- Sumner, L.W.; Amberg, A.; Barrett, D.; Beale, M.H.; Beger, R.; Daykin, C.A.; Fan, T.M.; Fiehn, O.; Goodacre, R.; Griffin, J.L.; et al. Proposed minimum reporting standards for chemical analysis Chemical Analysis Working Group (CAWG) Metabolomics Standards Initiative (MSI). Metabolomics 2007, 3, 211–221. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.P. Identification of Essential Oil Components by Gas Chromatography/Mass Spectrometry, 4th ed.; Allured Publishing: Carol Stream, IL, USA, 2007; pp. 9–788. [Google Scholar]
- Fischedick, J.T.; Hazekamp, A.; Erkelens, T.; Choi, Y.H.; Verpoorte, R. Metabolic fingerprinting of Cannabis sativa L., cannabinoids and terpenoids for chemotaxonomic and drug standardization purposes. Phytochemistry 2010, 71, 2058–2073. [Google Scholar] [CrossRef]
- Jin, D.; Henry, P.; Shan, J.; Chen, J. Identification of Chemotypic Markers in Three Chemotype Categories of Cannabis Using Secondary Metabolites Profiled in Inflorescences, Leaves, Stem Bark, and Roots. Front. Plant Sci. 2021, 12, 699530. [Google Scholar] [CrossRef]
- Li, D.; Ilnytskyy, Y.; Gojani, E.G.; Kovalchuk, O.; Kovalchuk, I. Analysis of Anti-Cancer and Anti-Inflammatory Properties of 25 High-THC Cannabis Extracts. Molecules 2022, 27, 6057. [Google Scholar] [CrossRef]
- Nigam, M.C.; Handa, K.L.; Nigam, I.C.; Levi, L. Essential oils and their constituents. XXIX. The essential oil of marihuana: Composition of genuine indian Cannabis sativa L. Can. J. Chem. 1965, 43, 3372–3376. [Google Scholar] [CrossRef]
- Turner, C.E.; Elsohly, M.A.; Boeren, E.G. Constituents of Cannabis sativa L. XVII. A review of the natural constituents. J. Nat. Prod. 1980, 43, 169–234. [Google Scholar] [CrossRef] [PubMed]
- Ross, S.A.; ElSohly, M.A. The volatile oil composition of fresh and air-dried buds of Cannabis sativa. J. Nat. Prod. 1996, 59, 49–51. [Google Scholar] [CrossRef] [PubMed]
- Hood, L.V.; Dames, M.E.; Barry, G.T. Headspace volatiles of marijuana. Nature 1973, 242, 402–403. [Google Scholar] [CrossRef] [PubMed]
- Calvi, L.; Pavlovic, R.; Panseri, S.; Giupponi, L.; Leoni, V.; Giorgi, A. Quality Traits of Medical Cannabis sativa L. Inflorescences and Derived Products Based on Comprehensive Mass-Spectrometry Analytical Investigation. In Recent Advances in Cannabinoid Research; Costain, J.W., Laprairie, R.B., Eds.; IntechOpen: London, UK, 2019; ISBN 978-1-83880-152-6. [Google Scholar]
- Serafimovska, T.; Serafimovska, M.D.; Mitevska, M.; Stefanovski, S.; Keskovski, Z.; Stefkov, G.; Balkanov, T.; Ribarska, J.T. Determination of Terpenoid Profile in Dry Cannabis Flowers and Extracts Obtained from Different Cannabis Varieties. J. Pharm. Res. Int. 2021, 35, 214–228. [Google Scholar] [CrossRef]
- Graves, B.M.; Johnson, T.J.; Nishida, R.T.; Dias, R.P.; Savareear, B.; Harynuk, J.J.; Kazemimanesh, M.; Olfert, J.S.; Boies, A.M. Comprehensive characterization of mainstream marijuana and tobacco smoke. Sci. Rep. 2020, 10, 7160. [Google Scholar] [CrossRef] [PubMed]
- Satyal, P.; Setzer, W.N. Chemotyping and Determination of Antimicrobial, Insecticidal, and Cytotoxic Properties of Wild Grown Cannabis sativa from Nepal. J. Med. Act. Plants 2014, 3, 9–16. [Google Scholar] [CrossRef]
- Ovidi, E.; Masci, V.L.; Taddei, A.R.; Torresi, J.; Tomassi, W.; Iannone, M.; Tiezzi, A.; Maggi, F.; Garzoli, S. Hemp (Cannabis sativa L., Kompolti cv.) and Hop (Humulus lupulus L., Chinook cv.) Essential Oil and Hydrolate: HS-GC-MS Chemical Investigation and Apoptotic Activity Evaluation. Pharmaceuticals 2022, 15, 976. [Google Scholar] [CrossRef]
- Tomko, A.M.; Whynot, E.G.; Ellis, L.D.; Dupre, D.J. Anti-Cancer Potential of Cannabinoids, Terpenes, and Flavonoids Present in Cannabis. Cancers 2020, 12, 1985. [Google Scholar] [CrossRef]
- Aizpurua-Olaizola, O.; Soydaner, U.; Ozturk, E.; Schibano, D.; Simsir, Y.; Navarro, P.; Etxebarria, N.; Usobiaga, A. Evolution of the Cannabinoid and Terpene Content during the Growth of Cannabis sativa Plants from Different Chemotypes. J. Nat. Prod. 2016, 79, 324–331. [Google Scholar] [CrossRef]
- Pieracci, Y.; Ascrizzi, R.; Terreni, V.; Pistelli, L.; Flamini, G.; Bassolino, L.; Fulvio, F.; Montanari, M.; Paris, R. Essential Oil of Cannabis sativa L.: Comparison of Yield and Chemical Composition of 11 Hemp Genotypes. Molecules 2021, 26, 4080. [Google Scholar] [CrossRef]
- Jerushalmi, S.; Maymon, M.; Dombrovsky, A.; Regev, R.; Schmilovitch, Z.; Namdar, D.; Shalev, N.; Koltai, H.; Freeman, S. Effects of steam sterilization on reduction of fungal colony forming units, cannabinoids and terpene levels in medical cannabis inflorescences. Sci. Rep. 2021, 11, 13973. [Google Scholar] [CrossRef] [PubMed]
- Nagy, D.U.; Cianfaglione, K.; Maggi, F.; Sut, S.; DallAcqua, S. Chemical Characterization of Leaves, Male and Female Flowers from Spontaneous Cannabis (Cannabis sativa L.) Growing in Hungary. Chem. Biodivers. 2019, 16, e1800562. [Google Scholar] [CrossRef] [PubMed]
- Pegoraro, C.N.; Nutter, D.; Thevenon, M.; Ramirez, C.L. Chemical profiles of Cannabis sativa medicinal oil using different extraction and concentration methods. Nat. Prod. Res. 2021, 35, 2249–2252. [Google Scholar] [CrossRef] [PubMed]
- Corporation, L. Determining Terpene Profiles of Cannabis Strains Using GC and GCxGC with High Performance TOFMS; LECO Corporation: St. Joseph, MI, USA, 2019. [Google Scholar]
- Gallily, R.; Yekhtin, Z.; Hanuš, L.O. The Anti-Inflammatory Properties of Terpenoids from Cannabis. Cannabis Cannabinoid Res. 2018, 3, 282–290. [Google Scholar] [CrossRef] [PubMed]
- Alipour, Z.; Taheri, P.; Samadi, N. Chemical composition and antibacterial activity of the essential oils from flower, leaf and stem of Ferula cupularis growing wild in Iran. Pharm. Biol. 2015, 53, 483–487. [Google Scholar] [CrossRef]
- Tavakoli, S.; Vatandoost, H.; Zeidabadinezhad, R.; Hajiaghaee, R.; Hadjiakhoondi, A.; Abai, M.R.; Yassa, N. Gas Chromatography, GC/Mass Analysis and Bioactivity of Essential Oil from Aerial Parts of Ferulago trifida: Antimicrobial, Antioxidant, AChE Inhibitory, General Toxicity, MTT Assay and Larvicidal Activities. J. Arthropod Borne Dis. 2017, 11, 414–426. Available online: https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5758637/ (accessed on 13 August 2023). [PubMed]
- Ekiert, H.; Pajor, J.; Klin, P.; Rzepiela, A.; Slesak, H.; Szopa, A. Significance of Artemisia Vulgaris L. (Common Mugwort) in the History of Medicine and Its Possible Contemporary Applications Substantiated by Phytochemical and Pharmacological Studies. Molecules 2020, 25, 4415. [Google Scholar] [CrossRef]
- Pandian, R.S.; Noora, A.T. GC-MS Analysis of Phytochemical Compounds Present in the Leaves of Citrus medica. L. Res. J. Pharm. Technol. 2019, 12, 1823–1826. [Google Scholar] [CrossRef]
- Abd-ElGawad, A.M.; El-Amier, Y.A.; Bonanomi, G.; El Gendy, A.E.G.; Elgorban, A.M.; Alamery, S.F.; Elshamy, A.I. Chemical Composition of Kickxia aegyptiaca Essential Oil and Its Potential Antioxidant and Antimicrobial Activities. Plants 2022, 11, 594. [Google Scholar] [CrossRef]
- Ilic, Z.; Stanojevic, L.; Milenkovic, L.; Sunic, L.; Milenkovic, A.; Stanojevic, J.; Cvetkovic, D. The Yield, Chemical Composition, and Antioxidant Activities of Essential Oils from Different Plant Parts of the Wild and Cultivated Oregano (Origanum vulgare L.). Horticulturae 2022, 8, 1042. [Google Scholar] [CrossRef]
- Myers, C.; Herrington, J.S.; Hamrah, P.; Anderson, K. Accelerated Solvent Extraction of Terpenes in Cannabis Coupled with Various Injection Techniques for GC-MS Analysis. Front. Chem. 2021, 9, 619770. [Google Scholar] [CrossRef] [PubMed]
- Stenerson, K.K. Headspace SPME-GC/MS Analysis of Terpenes in Hops and Cannabis. In Food & Beverage; MilliporeSigma: Darmstadt, Germany, 2018. [Google Scholar]
- Cicaloni, V.; Salvini, L.; Vitalini, S.; Garzoli, S. Chemical Profiling and Characterization of Different Cultivars of Cannabis sativa L. Inflorescences by SPME-GC-MS and UPLC-MS. Separations 2022, 9, 90. [Google Scholar] [CrossRef]
- Kamal, B.S.; Kamal, F.; Lantela, D.E. Cannabis and the Anxiety of Fragmentation—A Systems Approach for Finding an Anxiolytic Cannabis Chemotype. Front. Neurosci. 2018, 12, 730. [Google Scholar] [CrossRef] [PubMed]
- Sabogal-Guaqueta, A.M.; Hobbie, F.; Keerthi, A.; Oun, A.; Kortholt, A.; Boddeke, E.; Dolga, A. Linalool attenuates oxidative stress and mitochondrial dysfunction mediated by glutamate and NMDA toxicity. Biomed. Pharmacother. 2019, 118, 109295. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lv, O.; Zhou, F.; Li, Q.; Wu, Z.; Zheng, Y. Linalool inhibits LPS-induced inflammation in BV2 microglia cells by activating Nrf2. Neurochem. Res. 2015, 40, 1520–1525. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.K.; Jung, A.N.; Jung, Y. Linalool ameliorates memory loss and behavioral impairment induced by REM-sleep deprivation through the serotonergic pathway. Biomol. Ther. 2018, 26, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Kasuya, H.; Okada, N.; Kubohara, M.; Satou, T.; Masuo, Y.; Koike, K. Expression of BDNF and TH mRNA in the brain following inhaled administration of α-pinene. Phytother. Res. 2014, 29, 43–47. [Google Scholar] [CrossRef]
- Lee, G.; Lee, C.; Park, G.H.; Jang, J. Amelioration of scopolamine-induced learning and memory impairment by α-pinene in C57BL/6 mice. Evid. Based Complement. Altern. Med. 2017, 2017, 4926815. [Google Scholar] [CrossRef]
- Sun, J.; Li, H.; Sun, J.; Liu, H.; Chen, J.; Wang, C. Chemical composition and antimigraine activity of essential oil of Angelicae dahuricae radix. J. Med. Food 2017, 20, 1–7. [Google Scholar] [CrossRef]
- Wang, H.; Chang, J.; Fang, L.; Hsu, H.; Lee, L.; Yang, J.; Liang, M.; Hsiao, P.; Wang, C.; Wang, S.; et al. Bulnesia sarmientoi supercritical fluid extract exhibits necroptotic effects and anti-metastatic activity on lung cancer cells. Molecules 2018, 23, 3304. [Google Scholar] [CrossRef]
- Li, Y.; Xuan, H.; Shou, Q.; Zhan, Z.; Lu, X.; Hu, F. Therapeutic effects of propolis essential oil on anxiety of restraint-stressed mice. Hum. Exp. Toxicol. 2011, 31, 157–165. [Google Scholar] [CrossRef]
- Jang, H.; Rhee, K.; Eom, Y. Antibacterial and antibiofilm effects of α-humulene against Bacteroides fragilis. Can. J. Microbiol. 2020, 66, 389–399. [Google Scholar] [CrossRef]
- Legault, J.; Pichette, A. Potentiating effect of β-caryophyllene on antiancer activity of α-humulene, isocaryophyllene and paclitaxel. Pharm. Pharmacol. 2010, 59, 1643–1647. [Google Scholar] [CrossRef]
- Yeo, D.; Hwang, S.; Song, Y.; Lee, H. Humulene inhibits acute gastric mucosal injury by enhancing mucosal integrity. Antioxidants 2021, 10, 761. [Google Scholar] [CrossRef]
| S. No. | Compound Name 1 | Chemical Formula | RI (Exp) 4 | RI (Lib) | 1tR (min) 5 | 2tR (s) 6 | Pink Kush (%) 5 | Afghani Drifter (%) 5 | Blue Dream (%) 5 | Western Frost (%) 5 | Reeferman’s Rockstar (%) 5 | Ultra Sour (%) 5 | Previously Reported in Cannabis |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | α-Thujene | C10H16 | 934 | 934 | 26.19 | 1.87 ± 0.01 | - | - | - | - | - | 0.27 | [18,26,27] |
| 2 | α-Pinene 2 | C10H16 | 945 | 940 | 26.75 ± 0.02 | 1.86 ± 0.02 | 0.29 | 1.14 | 20.38 | 5.53 | 0.86 | 0.64 | [19,26,27,30,31] |
| 3 | Camphene 2 | C10H16 | 962 | 951 | 27.72 ± 0.02 | 1.89 ± 0.02 | 0.06 | 0.31 | 0.39 | 0.33 | 0.22 | 0.07 | [18,20,26] |
| 4 | Sabinene 2 | C10H16 | 983 | 977 | 28.85 ± 0.02 | 1.92 ± 0.01 | - | - | 0.03 | 0.01 | - | 0.21 | [19,20,27,31] |
| 5 | β-Myrcene/β-Pinene * | C10H16 | 991 | 990 | 29.29 ± 0.01 | 1.92 ± 0.02 | 1.35 | 7.90 | 25.50 | 17.01 | 2.12 | 1.58 | [19,26,27,30] |
| 6 | Monoterpene—unidentified | - | 1002 | - | 29.85 ± 0.01 | 1.87 ± 0.01 | - | - | - | - | - | 0.02 | - |
| 7 | α-Phellandrene 2 | C10H16 | 1015 | 1005 | 30.56 | 1.95 ± 0.01 | - | - | - | - | - | 0.33 | [26,27,30,31] |
| 8 | 3-Carene 2 | C10H16 | 1022 | 1010 | 30.95 | 1.92 ± 0.01 | - | - | - | - | - | 0.36 | [18,26,30,31] |
| 9 | α-Terpinene 2 | C10H16 | 1027 | 1018 | 31.19 ± 0.01 | 1.95 ± 0.01 | - | - | - | - | - | 0.45 | [18,26,30] |
| 10 | o-/p-Cymene | C10H14 | 1034 | 1026 | 31.59 ± 0.01 | 2.07 ± 0.01 | - | - | - | - | - | 0.18 | [26,32,33] |
| 11 | Limonene 2 | C10H16 | 1039 | 1030 | 31.87 ± 0.01 | 1.95 ± 0.02 | 1.84 | 9.30 | 2.10 | 5.75 | 6.48 | 2.84 | [18,26,30] |
| 12 | β-Phellandrene | C10H16 | 1042 | 1045 | 32.04 | 1.96 ± 0.01 | - | - | 0.34 | 0.36 | - | 0.49 | [5,18,30] |
| 13 | Eucalyptol 2 | C10H18O | 1044 | 1046 | 32.15 ± 0.03 | 1.97 | - | - | 0.30 | - | - | 0.06 | [18,26,27] |
| 14 | β-Ocimene 2 | C10H16 | 1050 | 1050 | 32.44 ± 0.01 | 1.96 ± 0.02 | - | - | 0.90 | 0.14 | - | 0.04 | [18,26,30] |
| 15 | γ-Terpinene 2 | C10H16 | 1068 | 1064 | 33.43 ± 0.01 | 1.96 ± 0.01 | - | - | 0.06 | 0.04 | 0.02 | 0.42 | [18,26,27,30] |
| 16 | cis-Sabinene hydrate 2 | C10H18O | 1078 | 1074 | 34.01 | 2.32 ± 0.03 | - | 0.03 | 0.13 | 0.08 | 0.03 | 0.20 | [20,26,31] |
| 17 | α-Terpinolene ** | C10H16 | 1099 | 1093 | 35.13 ± 0.02 | 1.97 ± 0.02 | 0.03 | 0.13 | 0.02 | 0.08 | 0.07 | 8.76 | [18,26,30] |
| 18 | p-Cymenene (Benzene, 1-methyl-4-(1-methylethenyl)-) ** | C10H12 | 1100 | 1095 | 35.16 ± 0.01 | 2.26 ± 0.01 | - | - | - | - | - | 0.32 | [20,26,34] |
| 19 | Linalool 2 | C10H18O | 1102 | 1104 | 35.26 ± 0.01 | 2.44 ± 0.03 | 2.27 | 2.45 | 1.38 | 1.91 | 2.98 | 2.07 | [18,20,26,30] |
| 20 | Fenchone 2 | C10H16O | 1104 | 1096 | 35.39 ± 0.02 | 2.19 ± 0.02 | 0.07 | 0.14 | 0.06 | - | 0.16 | 0.08 | [18,20,26,27] |
| 21 | trans-Sabinene hydrate | C10H18O | 1111 | 1099 | 35.75 ± 0.01 | 2.49 ± 0.03 | - | - | 0.09 | - | - | 0.23 | [5,26,34] |
| 22 | Monoterpenoid—unidentified | - | 1124 | - | 36.44 ± 0.02 | 2.12 ± 0.01 | - | - | - | - | - | 0.17 | - |
| 23 | Fenchol 2 | C10H18O | 1130 | 1123 | 36.74 ± 0.02 | 2.53 ± 0.03 | 0.94 | 1.68 | 0.44 | 2.15 | 1.64 | 0.90 | [18,19,20,26] |
| 24 | trans-p-Menth-2-en-1-ol | C10H18O | 1135 | 1137 | 37.03 ± 0.01 | 2.48 ± 0.01 | - | - | - | - | - | 0.04 | - |
| 25 | trans-2-Pinanol | C10H18O | 1140 | 1135 | 37.29 ± 0.01 | 2.40 ± 0.03 | 0.66 | 1.24 | 0.34 | 1.58 | 1.29 | 0.65 | [18] |
| 26 | Ipsdienol | C10H16O | 1151 | 1150 | 37.84 ± 0.02 | 2.69 ± 0.05 | 0.05 | - | - | - | - | - | [5,26,35] |
| 27 | Monoterpenoid—unidentified | - | 1153 | - | 37.96 ± 0.04 | 2.61 | - | - | - | - | - | 0.02 | - |
| 28 | Monoterpenoid—unidentified | - | 1155 | - | 38.05 ± 0.01 | 2.36 ± 0.01 | - | - | - | - | - | 0.03 | - |
| 29 | Monoterpenoid—unidentified | - | 1157 | - | 38.16 ± 0.02 | 2.20 ± 0.01 | - | - | - | - | - | 0.21 | - |
| 30 | Monoterpenoid—unidentified | - | 1162 | - | 38.42 ± 0.01 | 2.48 ± 0.03 | 0.05 | 0.19 | 0.08 | 0.23 | 0.17 | 0.10 | - |
| 31 | trans-β-Terpineol 2 | C10H18O | 1171 | 1161 | 38.90 ± 0.02 | 2.44 ± 0.03 | 0.05 | 0.08 | - | 0.12 | 0.10 | 0.04 | [5] |
| 32 | Monoterpenoid—unidentified | - | 1175 | - | 39.08 ± 0.02 | 2.41 ± 0.01 | - | - | - | - | - | 0.04 | - |
| 33 | (−)-Borneol 2 | C10H18O | 1187 | 1171 | 39.68 ± 0.01 | 2.73 ± 0.04 | 0.46 | 0.45 | 0.24 | 0.84 | 0.56 | 0.39 | [18,20,26,36] |
| 34 | trans-Carveol | C10H16O | 1192 | 1201 | 39.95 ± 0.02 | 2.66 ± 0.01 | - | - | - | - | - | 0.57 | [26] |
| 35 | Terpinen-4-ol | C10H18O | 1194 | 1206 | 40.08 | 2.36 ± 0.03 | 0.03 | 0.03 | - | 0.05 | 0.04 | 0.29 | [20,26,27,31] |
| 36 | p-Cymen-8-ol | C10H14O | 1197 | 1193 | 40.19 | 3.42 ± 0.01 | - | - | - | - | - | 1.00 | [26,27,34] |
| 37 | α-Terpineol 2 | C10H18O | 1206 | 1192 | 40.67 | 2.58 ± 0.03 | 1.11 | 1.48 | 0.45 | 1.53 | 1.54 | 1.82 | [20,26,34] |
| 38 | trans-Piperitol | C10H18O | 1222 | 1208 | 41.46 ± 0.02 | 2.74 ± 0.02 | - | - | - | - | - | 0.10 | [37] |
| 39 | Citronellol | C10H20O | 1231 | 1232 | 41.89 ± 0.01 | 2.67 ± 0.02 | - | - | 0.02 | 0.08 | 0.02 | - | [5,34,38] |
| 40 | trans-Chrysanthenyl acetate | C12H18O2 | 1235 | 1233 | 42.10 ± 0.01 | 2.90 ± 0.01 | - | - | - | - | - | 0.19 | [39] |
| 41 | trans-Verbenyl acetate | C12H18O2 | 1301 | 1293 | 45.33 ± 0.01 | 3.16 ± 0.01 | - | - | - | - | - | 0.18 | - |
| 42 | Bornyl acetate | C12H20O2 | 1303 | 1287 | 45.46 ± 0.02 | 2.10 ± 0.02 | 0.01 | 0.02 | - | 0.03 | 0.07 | 0.02 | [5,26] |
| 43 | Monoterpenoid—unidentified | - | 1328 | - | 46.59 ± 0.01 | 2.81 ± 0.01 | - | - | - | - | - | 0.02 | - |
| 44 | Monoterpenoid—unidentified | - | 1334 | - | 46.91 ± 0.03 | 3.47 ± 0.01 | - | - | - | - | - | 0.08 | - |
| 45 | Monoterpenoid—unidentified | - | 1354 | - | 47.71 ± 0.01 | 3.26 | - | - | - | - | - | 0.03 | - |
| 46 | Monoterpenoid—unidentified | - | 1356 | - | 47.96 ± 0.01 | 2.28 ± 0.03 | - | - | - | - | 0.05 | - | - |
| 47 | Verbenone 3 | C10H14O | 1367 | - | 48.44 ± 0.02 | 2.80 ± 0.05 | 0.01 | - | - | - | 0.05 | 0.01 | [26] |
| 48 | α-Cubebene | C15H24 | 1372 | 1374 | 48.69 ± 0.02 | 1.86 ± 0.02 | 0.02 | 0.02 | - | - | 0.04 | 0.01 | [5,26,34] |
| 49 | α-Ylangene | C15H24 | 1400 | 1406 | 50.01 ± 0.02 | 1.87 ± 0.02 | 0.29 | 0.30 | 0.06 | 0.11 | 0.22 | 0.15 | [5,26,27] |
| 50 | α-Copaene | C15H24 | 1404 | 1416 | 50.22 ± 0.02 | 1.88 ± 0.02 | 0.12 | 0.07 | - | 0.04 | 0.10 | 0.09 | [26,27,34] |
| 51 | β-Copaene | C15H24 | 1414 | 1414 | 50.66 ± 0.02 | 1.89 ± 0.02 | 0.03 | 0.05 | - | - | 0.04 | 0.02 | [40] |
| 52 | cis-α-Bergamotene | C15H24 | 1419 | 1415 | 50.87 ± 0.02 | 1.89 ± 0.01 | - | - | - | - | - | 0.12 | [5,18,26] |
| 53 | α-Guaiene | C15H24 | 1430 | 1438 | 51.36 ± 0.02 | 1.93 ± 0.02 | 0.24 | 0.20 | 0.05 | 0.14 | 0.31 | 0.12 | [5,18,30] |
| 54 | trans-α-Bergamotene | C15H24 | 1436 | 1436 | 51.61 ± 0.02 | 1.90 | 0.03 | - | - | - | 0.03 | 0.19 | [5,18,26,34] |
| 55 | Sesquiterpene—unidentified | - | 1441 | - | 51.82 ± 0.02 | 1.94 | 0.05 | - | - | - | 0.01 | - | - |
| 56 | α-Himachalene | C15H24 | 1444 | 1444 | 51.98 ± 0.03 | 1.90 ± 0.02 | - | 0.03 | - | - | 0.01 | 0.27 | [41] |
| 57 | β-Gurjunene | C15H24 | 1450 | 1450 | 52.25 ± 0.02 | 1.91 ± 0.02 | 0.15 | 0.43 | 0.17 | 0.12 | 0.20 | 0.17 | [21] |
| 58 | trans-β-Caryophyllene 2 | C15H24 | 1457 | 1455 | 52.56 ± 0.02 | 1.94 ± 0.02 | 15.80 | 16.79 | 14.82 | 10.40 | 12.38 | 12.79 | [20,26,30] |
| 59 | cis-β-Farnesene | C15H24 | 1463 | 1457 | 52.81 ± 0.01 | 1.92 ± 0.02 | 0.19 | 0.20 | 0.10 | 0.14 | 0.09 | 1.64 | [18,26,34] |
| 60 | Sesquiterpene—unidentified | - | 1467 | - | 52.97 ± 0.01 | 1.91 ± 0.01 | - | - | 0.05 | 0.02 | - | 0.05 | - |
| 61 | Sesquiterpene—unidentified | - | 1473 | - | 53.23 ± 0.03 | 1.92 ± 0.02 | 0.12 | 0.19 | 0.06 | 0.04 | 0.10 | 0.17 | - |
| 62 | β-Selinene | C15H24 | 1482 | 1485 | 53.65 ± 0.03 | 1.94 ± 0.02 | 0.14 | 0.12 | - | 0.04 | 0.08 | 0.10 | [18,20,26,34] |
| 63 | Humulene 2 | C15H24 | 1492 | 1495 | 54.09 ± 0.02 | 1.98 ± 0.02 | 6.38 | 6.63 | 3.60 | 2.61 | 4.56 | 5.43 | [19,26,30,34] |
| 64 | Sesquiterpene—unidentified | - | 1499 | - | 54.42 ± 0.01 | 1.94 ± 0.02 | 0.08 | 0.12 | - | 0.02 | 0.07 | 0.05 | - |
| 65 | Cuparene | C15H22 | 1500 | 1505 | 54.45 ± 0.03 | 2.04 ± 0.01 | - | - | - | - | - | 0.03 | [20,31] |
| 66 | Valencene 2 | C15H24 | 1506 | 1491 | 54.71 ± 0.02 | 1.96 ± 0.02 | 0.83 | 0.63 | 0.19 | 0.27 | 0.68 | 0.30 | [42] |
| 67 | Eremophila-1(10),11-diene | C15H24 | 1511 | 1503 | 54.89 ± 0.02 | 1.95 ± 0.02 | 0.42 | 0.28 | 0.09 | - | 0.32 | 0.51 | [26] |
| 68 | α-Farnesene | C15H24 | 1514 | 1507 | 55.03 ± 0.01 | 1.94 ± 0.02 | - | 0.38 | 0.24 | 0.57 | - | - | [18,20,34] |
| 69 | Sesquiterpene—unidentified | - | 1517 | - | 55.16 ± 0.04 | 1.96 | 0.26 | - | - | - | 0.16 | 0.07 | - |
| 70 | β-Cadinene | C15H24 | 1521 | 1526 | 55.32 ± 0.02 | 1.96 ± 0.02 | 1.02 | 1.18 | 0.38 | 0.43 | 0.8 | 0.55 | [18] |
| 71 | α-Selinene | C15H24 | 1526 | 1526 | 55.55 ± 0.01 | 1.99 ± 0.02 | 3.35 | 3.23 | 0.82 | 1.33 | 3.41 | 0.78 | [18,20,26,34] |
| 72 | β-Bisabolene | C15H24 | 1528 | 1517 | 55.62 ± 0.02 | 1.98 ± 0.01 | - | - | - | - | - | 0.94 | [5,26,34] |
| 73 | Sesquiterpene—unidentified | - | 1534 | - | 55.85 ± 0.02 | 1.99 ± 0.02 | 2.86 | 2.04 | 0.57 | 0.93 | 2.74 | 1.03 | - |
| 74 | γ-Selinene | C15H24 | 1538 | 1532 | 56.05 ± 0.02 | 1.95 ± 0.02 | 0.64 | 0.72 | 0.31 | 0.26 | 0.35 | 0.45 | [43] |
| 75 | Sesquiterpene—unidentified | - | 1546 | - | 56.37 ± 0.02 | 1.99 ± 0.01 | - | - | - | - | - | 0.24 | - |
| 76 | δ-Cadinene | C15H24 | 1547 | 1530 | 56.43 ± 0.04 | 1.99 ± 0.02 | 0.32 | 0.31 | - | - | 0.25 | 0.25 | [5,26,34] |
| 77 | Sesquiterpene—unidentified | - | 1545 | - | 56.35 ± 0.06 | 1.96 ± 0.01 | - | - | - | 0.06 | - | - | - |
| 78 | Sesquiterpene—unidentified | - | 1549 | - | 56.49 ± 0.03 | 1.99 ± 0.02 | - | - | 0.09 | 0.21 | - | - | - |
| 79 | Sesquiterpenoid—unidentified | - | 1550 | - | 56.54 | 2.20 | 0.04 | - | - | - | 0.04 | - | - |
| 80 | Sesquiterpene—unidentified | - | 1553 | - | 56.66 ± 0.02 | 1.96 ± 0.02 | 1.49 | 1.72 | 0.41 | 0.70 | 1.36 | 0.70 | - |
| 81 | α-Bisabolene 2 | C15H24 | 1559 | 1545 | 56.92 ± 0.02 | 1.95 ± 0.02 | 4.11 | 1.45 | 1.25 | 1.42 | 1.98 | 0.76 | [18,26] |
| 82 | Sesquiterpene—unidentified | - | 1562 | - | 57.05 ± 0.02 | 1.97 ± 0.02 | 2.13 | 2.06 | 0.42 | 1.03 | 2.86 | 1.26 | - |
| 83 | trans-Sesquisabinene hydrate | C15H26O | 1564 | 1559 | 57.14 ± 0.02 | 2.21 ± 0.02 | 0.11 | 0.03 | - | 0.04 | 0.07 | 0.06 | [38,44] |
| 84 | Sesquiterpene—unidentified | - | 1566 | - | 57.21 ± 0.01 | 1.98 ± 0.02 | 0.90 | 0.62 | 0.16 | 0.32 | 0.7 | 0.32 | - |
| 85 | Sesquiterpene—unidentified | - | 1571 | - | 57.43 ± 0.02 | 1.97 ± 0.02 | 3.43 | 4.33 | 1.14 | 1.72 | 3.38 | 2.17 | - |
| 86 | trans-Nerolidol 2,** | C15H26O | 1574 | 1567 | 57.56 ± 0.02 | 2.25 ± 0.04 | - | 0.38 | 0.10 | - | - | 0.52 | [20,26,27] |
| 87 | Sesquiterpene—unidentified ** | - | 1576 | - | 57.62 ± 0.01 | 2.01 ± 0.02 | 15.48 | 9.75 | 2.78 | 8.46 | 14.92 | 8.61 | - |
| 88 | α-Calacorene | C15H20 | 1578 | 1562 | 57.74 ± 0.01 | 2.17 ± 0.02 | 0.31 | 0.06 | 0.02 | 0.03 | 0.17 | 0.06 | [26] |
| 89 | Selina-3,7(11)-diene 3 | C15H24 | 1583 | - | 57.94 ± 0.02 | 1.99 ± 0.02 | 15.06 | 12.96 | 4.05 | 12.89 | 18.50 | 10.76 | [18,26,34,43] |
| 90 | Sesquiterpenoid—unidentified | - | 1589 | - | 58.18 ± 0.01 | 2.23 ± 0.02 | 0.17 | 0.23 | 0.06 | 0.11 | 0.26 | 0.14 | - |
| 91 | Sesquiterpene—unidentified | - | 1592 | - | 58.30 ± 0.07 | 1.98 ± 0.02 | 0.05 | 0.03 | - | - | - | 0.02 | - |
| 92 | Sesquiterpenoid—unidentified | - | 1597 | - | 58.51 ± 0.01 | 2.75 ± 0.02 | 0.06 | - | - | - | 0.29 | - | - |
| 93 | Sesquiterpene—unidentified | - | 1600 | - | 58.66 ± 0.01 | 2.01 ± 0.02 | 0.88 | 1.23 | 0.24 | 0.42 | 0.54 | 0.35 | - |
| 94 | Sesquiterpenoid—unidentified | - | 1613 | - | 59.16 ± 0.02 | 2.24 ± 0.02 | 0.11 | 0.28 | 0.10 | 0.11 | 0.21 | 0.14 | - |
| 95 | trans-Longipinocarveol ** | C15H24O | 1620 | 1618 | 59.46 ± 0.02 | 2.41 ± 0.03 | 0.24 | 0.17 | - | - | 0.12 | 0.14 | [45] |
| 96 | Sesquiterpene—unidentified ** | - | 1621 | - | 59.49 | 2.05 ± 0.02 | 0.02 | 0.01 | - | - | 0.24 | - | - |
| 97 | Caryophyllene oxide 2 | C15H24O | 1632 | 1617 | 59.92 ± 0.03 | 2.24 ± 0.05 | 0.73 | 0.77 | 2.61 | 4.04 | 0.52 | 0.62 | [19,20,26,27] |
| 98 | Sesquiterpenoid—unidentified | - | 1642 | - | 60.32 ± 0.04 | 2.21 ± 0.07 | - | - | - | - | 0.02 | 0.10 | - |
| 99 | Sesquiterpenoid—unidentified | - | 1647 | - | 60.56 ± 0.24 | 2.34 ± 0.13 | - | - | - | - | - | 0.23 | - |
| 100 | Sesquiterpenoid—unidentified | - | 1654 | - | 60.82 ± 0.03 | 2.23 ± 0.06 | 0.21 | - | - | - | - | 0.29 | - |
| 101 | Sesquiterpenoid—unidentified | - | 1657 | - | 60.96 ± 0.02 | 2.23 ± 0.04 | - | 0.21 | - | - | 0.18 | 0.38 | - |
| 102 | Aromadendrene oxide | C15H24O | 1661 | 1672 | 61.07 | 2.68 ± 0.01 | - | - | - | - | - | 0.04 | - |
| 103 | Eudesm-7(11)-en-4-ol | C15H26O | 1663 | 1675 | 61.18 ± 0.02 | 2.40 ± 0.03 | 0.58 | 0.65 | 0.35 | 0.28 | 0.50 | 0.26 | [46] |
| 104 | Cedren-13-ol, 8- | C15H24O | 1666 | 1688 | 61.31 ± 0.02 | 2.44 ± 0.03 | 0.34 | 0.04 | - | - | - | - | [44] |
| 105 | Sesquiterpenoid—unidentified | - | 1666 | - | 61.31 ± 0.01 | 2.48 | - | - | - | - | 0.25 | 0.25 | - |
| 106 | Sesquiterpenoid—unidentified | - | 1669 | - | 61.42 | 2.32 ± 0.02 | - | - | 3.41 | 5.01 | - | 4.39 | - |
| 107 | Sesquiterpenoid—unidentified | - | 1669 | - | 61.44 ± 0.01 | 2.54 | 0.21 | - | - | - | 0.11 | - | - |
| 108 | Sesquiterpenoid—unidentified | - | 1670 | - | 61.48 ± 0.02 | 2.45 ± 0.05 | 0.15 | - | - | - | - | - | - |
| 109 | Bulnesol ** | C15H26O | 1673 | 1672 | 61.61 | 2.38 ± 0.03 | 0.05 | 0.09 | 0.54 | 0.72 | 0.14 | 0.79 | [26,30,47] |
| 110 | α-Santalol ** | C15H24O | 1675 | 1675 | 61.66 ± 0.01 | 2.55 ± 0.04 | 0.05 | - | - | - | - | - | [48] |
| 111 | Sesquiterpenoid—unidentified ** | - | 1677 | - | 61.75 ± 0.01 | 2.42 ± 0.02 | - | - | 0.24 | 0.41 | - | 0.50 | - |
| 112 | Sesquiterpenoid—unidentified ** | - | 1679 | - | 61.77 ± 0.02 | 2.65 ± 0.01 | - | - | - | - | - | 0.02 | - |
| 113 | Sesquiterpenoid—unidentified | - | 1679 | - | 61.84 ± 0.01 | 2.41 | - | - | - | - | - | 0.08 | - |
| 114 | Sesquiterpenoid—unidentified ** | - | 1682 | - | 61.93 ± 0.07 | 2.64 ± 0.04 | 0.04 | - | - | - | 0.05 | 0.04 | - |
| 115 | Sesquiterpenoid—unidentified ** | - | 1682 | - | 61.96 | 2.42 | - | - | 0.05 | - | - | 0.13 | - |
| 116 | Sesquiterpenoid—unidentified | - | 1686 | - | 62.11 | 2.42 ± 0.02 | - | - | 0.34 | 0.48 | - | 0.36 | - |
| 117 | Sesquiterpenoid—unidentified | - | 1689 | - | 62.23 | 2.43 ± 0.02 | - | - | 0.37 | 0.53 | - | 0.45 | - |
| 118 | Sesquiterpenoid—unidentified | - | 1692 | - | 62.31 ± 0.02 | 2.72 ± 0.05 | - | 0.11 | - | 0.13 | 0.03 | 0.21 | - |
| 119 | Sesquiterpenoid—unidentified | - | 1693 | - | 62.39 ± 0.02 | 2.42 ± 0.04 | 0.18 | 0.06 | - | - | 0.08 | - | - |
| 120 | Sesquiterpenoid—unidentified | - | 1693 | - | 62.41 ± 0.02 | 2.58 ± 0.01 | - | - | - | - | - | 0.10 | - |
| 121 | Sesquiterpenoid—unidentified | - | 1698 | - | 62.55 ± 0.02 | 2.65 ± 0.05 | 0.05 | - | - | - | - | - | - |
| 122 | Sesquiterpenoid—unidentified | - | 1699 | - | 62.62 ± 0.02 | 2.45 ± 0.03 | 0.19 | 0.08 | 3.20 | 3.85 | 0.20 | 4.27 | - |
| 123 | Sesquiterpenoid—unidentified | - | 1706 | - | 62.88 ± 0.02 | 2.57 ± 0.01 | 0.10 | - | - | - | 0.07 | - | - |
| 124 | α-Bisabolol ** | C15H26O | 1709 | 1700 | 63.01 ± 0.01 | 2.34 ± 0.04 | 6.95 | 0.76 | 2.72 | - | 4.11 | 5.88 | [19,20,26,30] |
| 125 | Sesquiterpenoid—unidentified ** | - | 1711 | - | 63.06 ± 0.01 | 2.74 ± 0.04 | 0.30 | 0.11 | 0.09 | - | 0.15 | 0.17 | - |
| 126 | Sesquiterpenoid—unidentified ** | - | 1714 | - | 63.22 ± 0.03 | 2.36 ± 0.03 | - | 0.05 | - | 0.08 | 0.22 | 0.07 | - |
| 127 | Sesquiterpenoid—unidentified ** | - | 1715 | - | 63.26 ± 0.03 | 2.52 ± 0.09 | 0.14 | 0.03 | - | - | 0.08 | 0.06 | - |
| 128 | Sesquiterpenoid—unidentified | - | 1722 | - | 63.50 ± 0.03 | 2.61 ± 0.02 | - | - | - | - | 0.03 | - | - |
| 129 | Sesquiterpenoid—unidentified | - | 1726 | - | 63.66 ± 0.03 | 2.33 ± 0.02 | - | - | - | - | 0.02 | - | - |
| 130 | Sesquiterpenoid—unidentified | - | 1729 | - | 63.76 | 2.59 ± 0.02 | - | - | - | - | 0.02 | - | - |
| 131 | Sesquiterpenoid—unidentified | - | 1731 | - | 63.85 ± 0.01 | 2.52 ± 0.02 | - | - | 0.05 | 0.15 | - | 0.17 | - |
| 132 | Sesquiterpenoid—unidentified | - | 1731 | - | 63.87 ± 0.02 | 2.38 ± 0.01 | 0.05 | - | - | - | 0.03 | - | - |
| 133 | Sesquiterpenoid—unidentified | - | 1736 | - | 64.02 ± 0.03 | 2.68 | - | - | - | - | - | 0.04 | - |
| 134 | Farnesol | C15H26O | 1734 | 1740 | 63.99 | 2.49 ± 0.01 | 0.030 | - | - | - | 0.06 | - | [5,18,26] |
| 135 | 5β, 7βH, 10α-Eudesm-11-en-1α-ol | C15H26O | 1746 | 1748 | 64.43 ± 0.02 | 2.48 ± 0.03 | 1.89 | 0.91 | 0.30 | 0.66 | 1.46 | 0.70 | - |
| 136 | Sesquiterpenoid—unidentified | - | 1756 | - | 64.80 ± 0.01 | 2.55 ± 0.03 | 0.19 | 0.09 | - | 0.14 | 0.20 | 0.07 | - |
| 137 | Sesquiterpenoid—unidentified | - | 1768 | - | 65.27 ± 0.05 | 2.37 ± 0.02 | - | - | - | - | 0.04 | - | - |
| 138 | Sesquiterpenoid—unidentified | - | 1799 | - | 66.48 | 2.42 ± 0.02 | - | - | - | - | 0.02 | - | - |
| 139 | Sesquiterpenoid—unidentified | - | 1812 | - | 66.93 ± 0.01 | 2.61 ± 0.02 | - | - | - | - | 0.02 | - | - |
| 140 | Sesquiterpenoid—unidentified | - | 1817 | - | 67.11 ± 0.01 | 2.73 ± 0.02 | - | - | - | - | 0.03 | - | - |
| 141 | Sesquiterpenoid—unidentified | - | 1821 | - | 67.25 ± 0.01 | 2.67 ± 0.02 | 0.05 | - | - | - | 0.09 | - | - |
| 142 | Sesquiterpenoid—unidentified | - | 1845 | - | 68.13 ± 0.02 | 3.01 ± 0.03 | 0.05 | - | - | - | 0.06 | - | - |
| 143 | Sesquiterpenoid—unidentified | - | 1859 | - | 68.63 | 3.11 ± 0.03 | - | - | 0.13 | 0.31 | - | - | - |
| 144 | Sesquiterpenoid—unidentified | - | 1869 | - | 69.03 | 3.25 ± 0.05 | - | - | - | 0.13 | - | 0.12 | - |
| 145 | Sesquiterpenoid—unidentified | - | 1875 | - | 69.25 | 2.91 ± 0.03 | - | - | - | - | 0.02 | - | - |
| 146 | Phytol 3 | C20H40O | 1994 | - | 77.95 ± 0.02 | 2.13 ± 0.02 | 0.19 | 0.12 | - | 0.21 | 0.11 | 0.13 | [5,27,34,38] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaur, J.; Sun, N.; Hill, J.E. Comprehensive Profiling of Terpenes and Terpenoids in Different Cannabis Strains Using GC × GC-TOFMS. Separations 2023, 10, 500. https://doi.org/10.3390/separations10090500
Kaur J, Sun N, Hill JE. Comprehensive Profiling of Terpenes and Terpenoids in Different Cannabis Strains Using GC × GC-TOFMS. Separations. 2023; 10(9):500. https://doi.org/10.3390/separations10090500
Chicago/Turabian StyleKaur, Jasmeen, Ning Sun, and Jane E. Hill. 2023. "Comprehensive Profiling of Terpenes and Terpenoids in Different Cannabis Strains Using GC × GC-TOFMS" Separations 10, no. 9: 500. https://doi.org/10.3390/separations10090500
APA StyleKaur, J., Sun, N., & Hill, J. E. (2023). Comprehensive Profiling of Terpenes and Terpenoids in Different Cannabis Strains Using GC × GC-TOFMS. Separations, 10(9), 500. https://doi.org/10.3390/separations10090500






