Recovering Scandium from Scandium Rough Concentrate Using Roasting-Hydrolysis-Leaching Process
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Main Chemical Reagent and Equipment
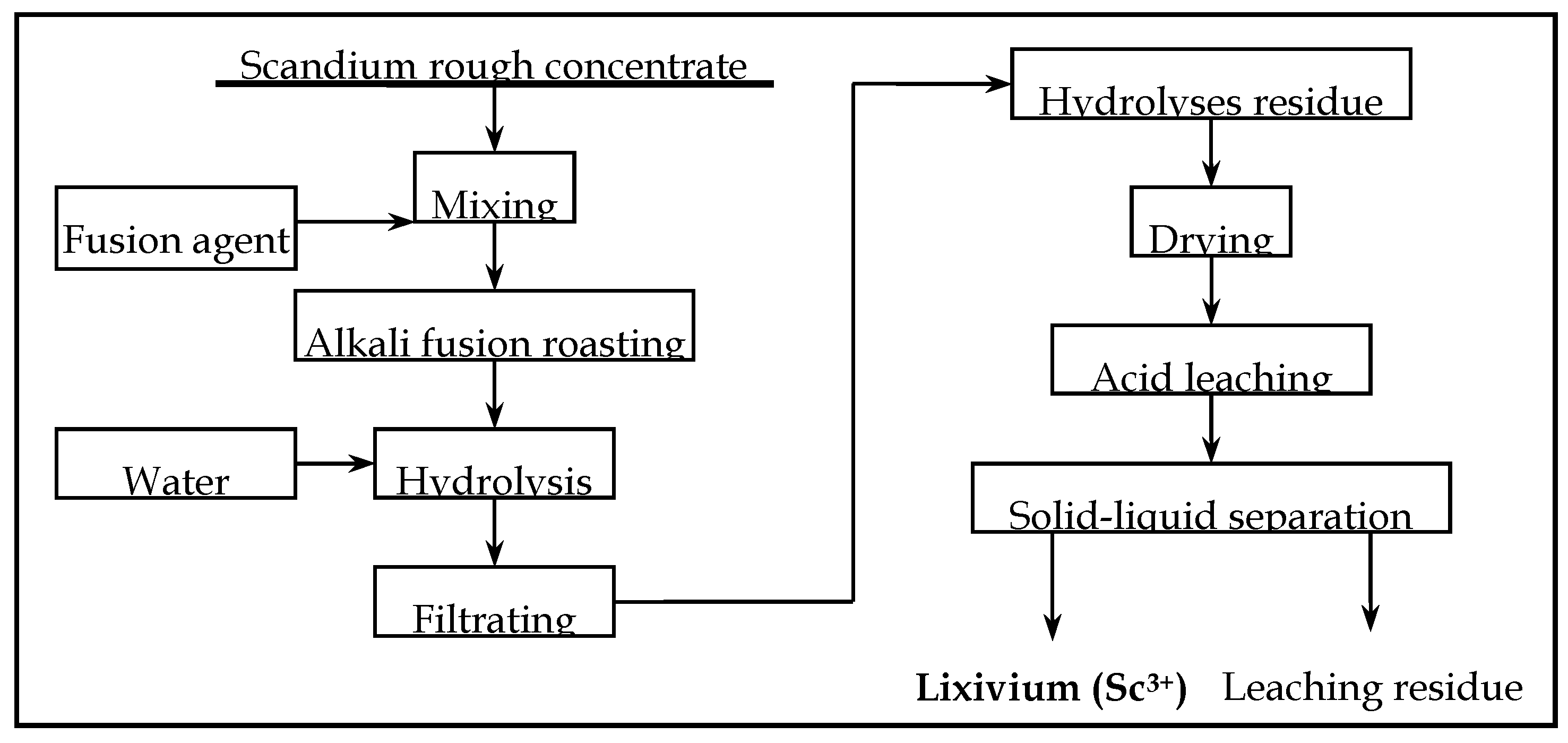
2.3. Procedure
2.4. Analysis and Characterization
3. Results and Discussion
3.1. Removal of Silica and Alumina by Roasting—Hydrolysis
3.1.1. Effect of Different Roasting Agent
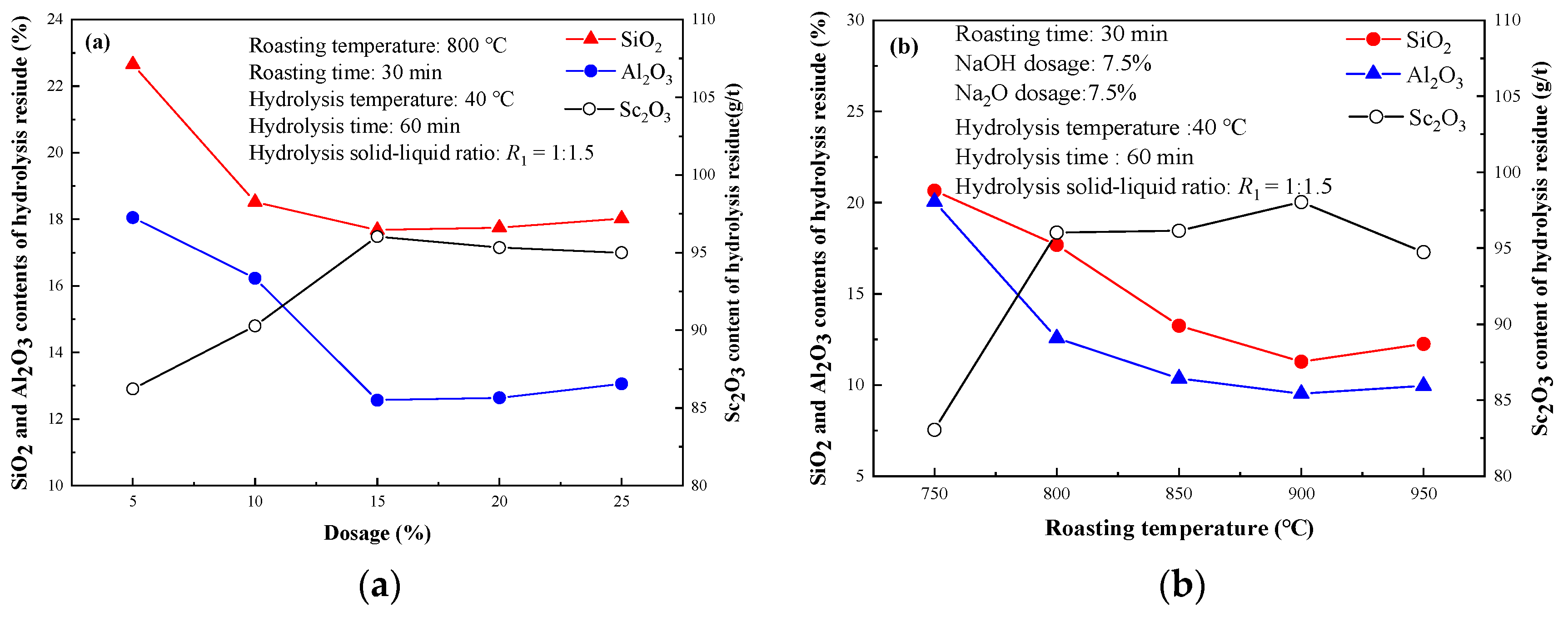
3.1.2. Effect of Roasting Agent Dosage
3.1.3. Effect of Roasting Temperature
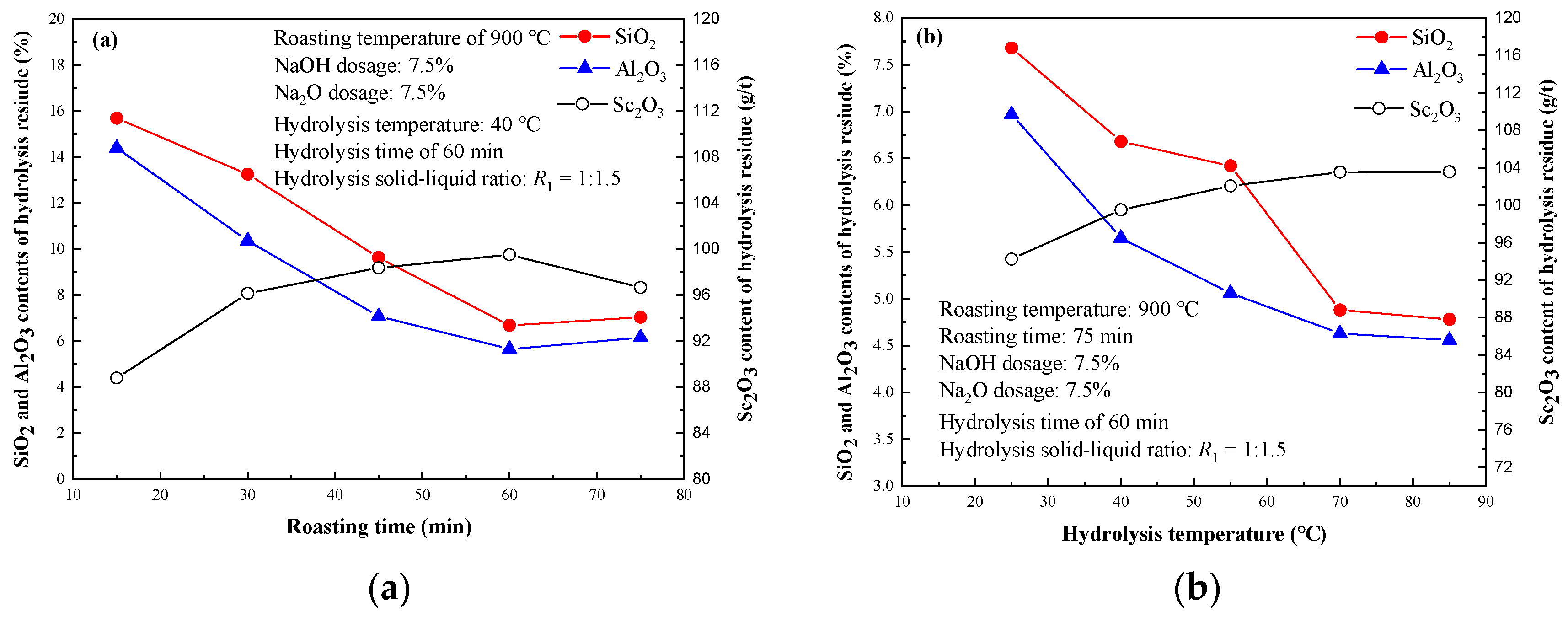
3.1.4. Effect of Roasting Time
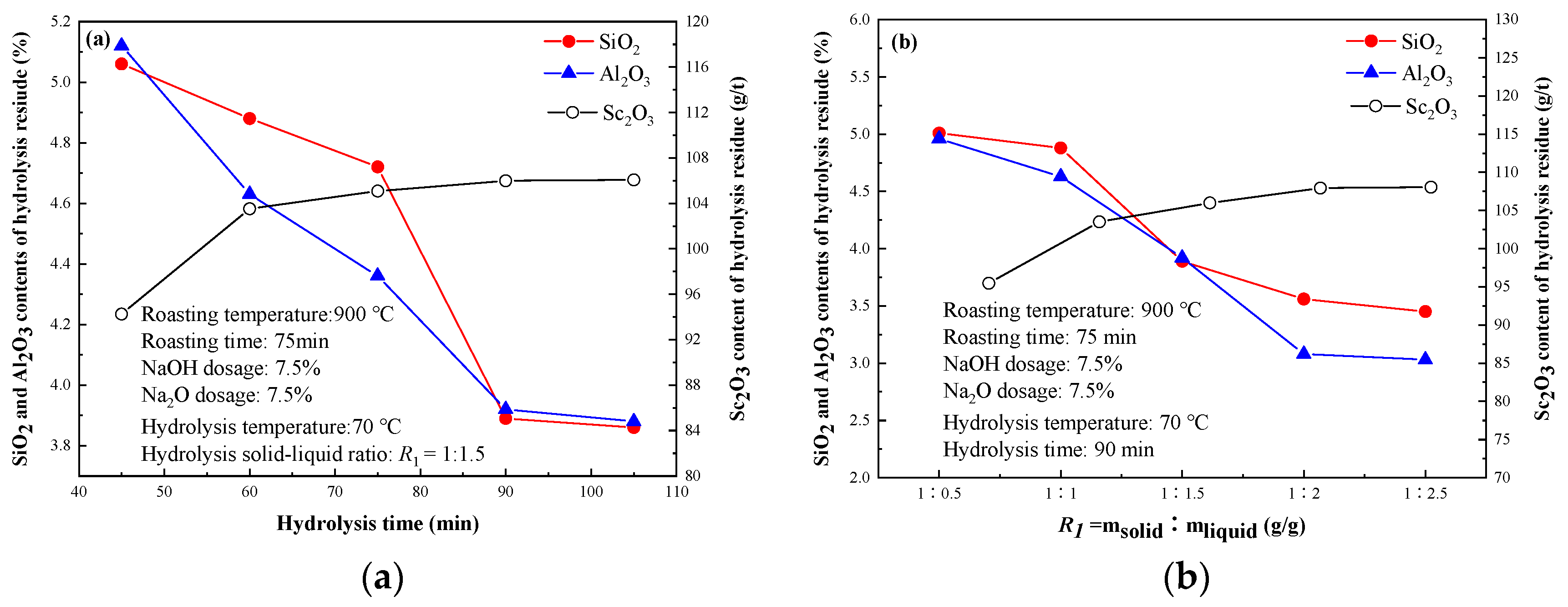
3.1.5. Effect of Hydrolysis Condition
3.1.6. Scale-Up Test of Roasting-Hydrolysis
3.2. Scandium Extraction of Hydrolysis Residue by Acid Leaching
3.2.1. Effect of Different Acid Leaching Agent
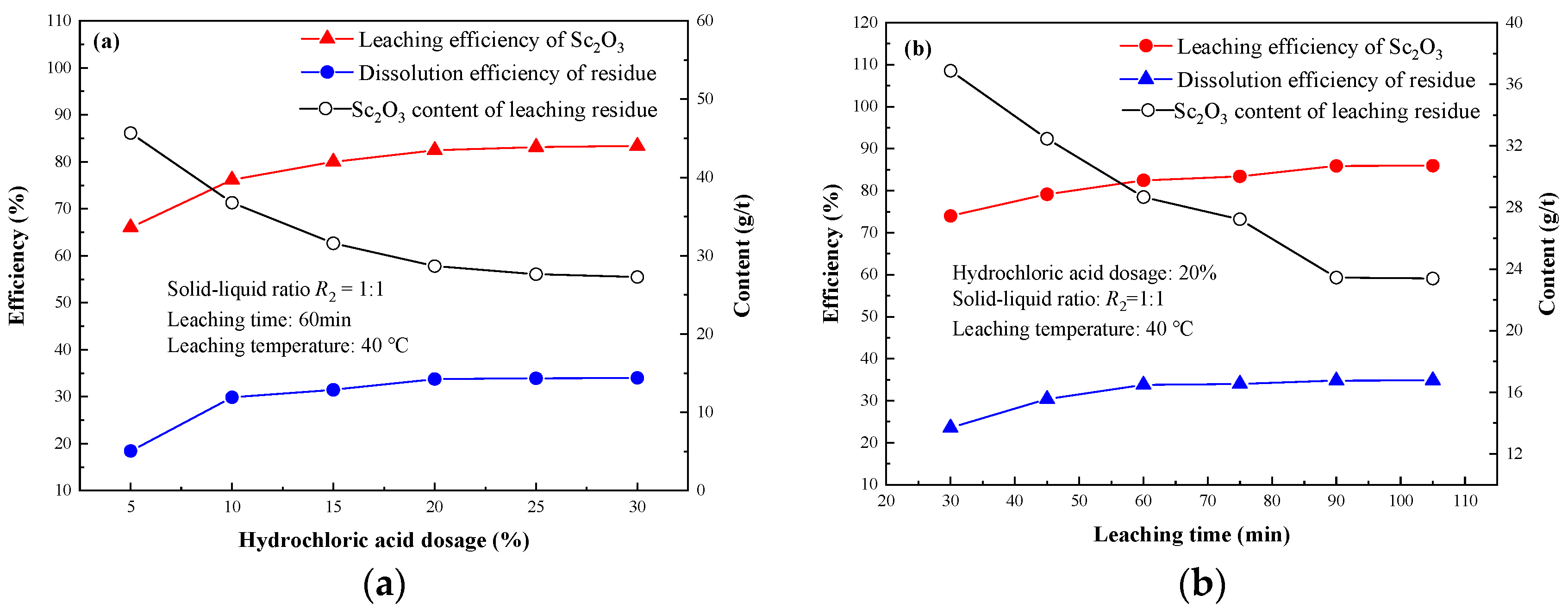
3.2.2. Effect of Hydrochloric Acid Dosage
3.2.3. Effect of Leaching Time
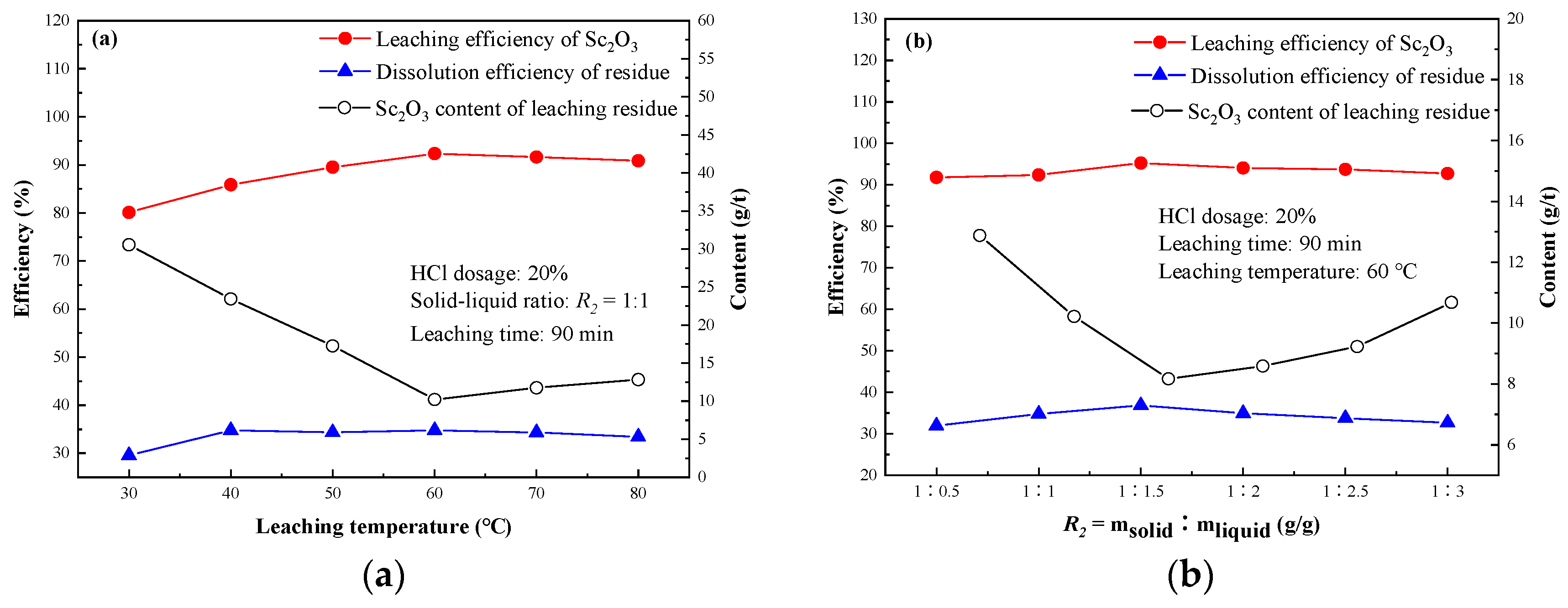
3.2.4. Effect of Leaching Temperature
3.2.5. Effect of Leaching Solid to Liquid Ratio
3.2.6. Scale-Up Acid Leaching Test of Scandium Extraction
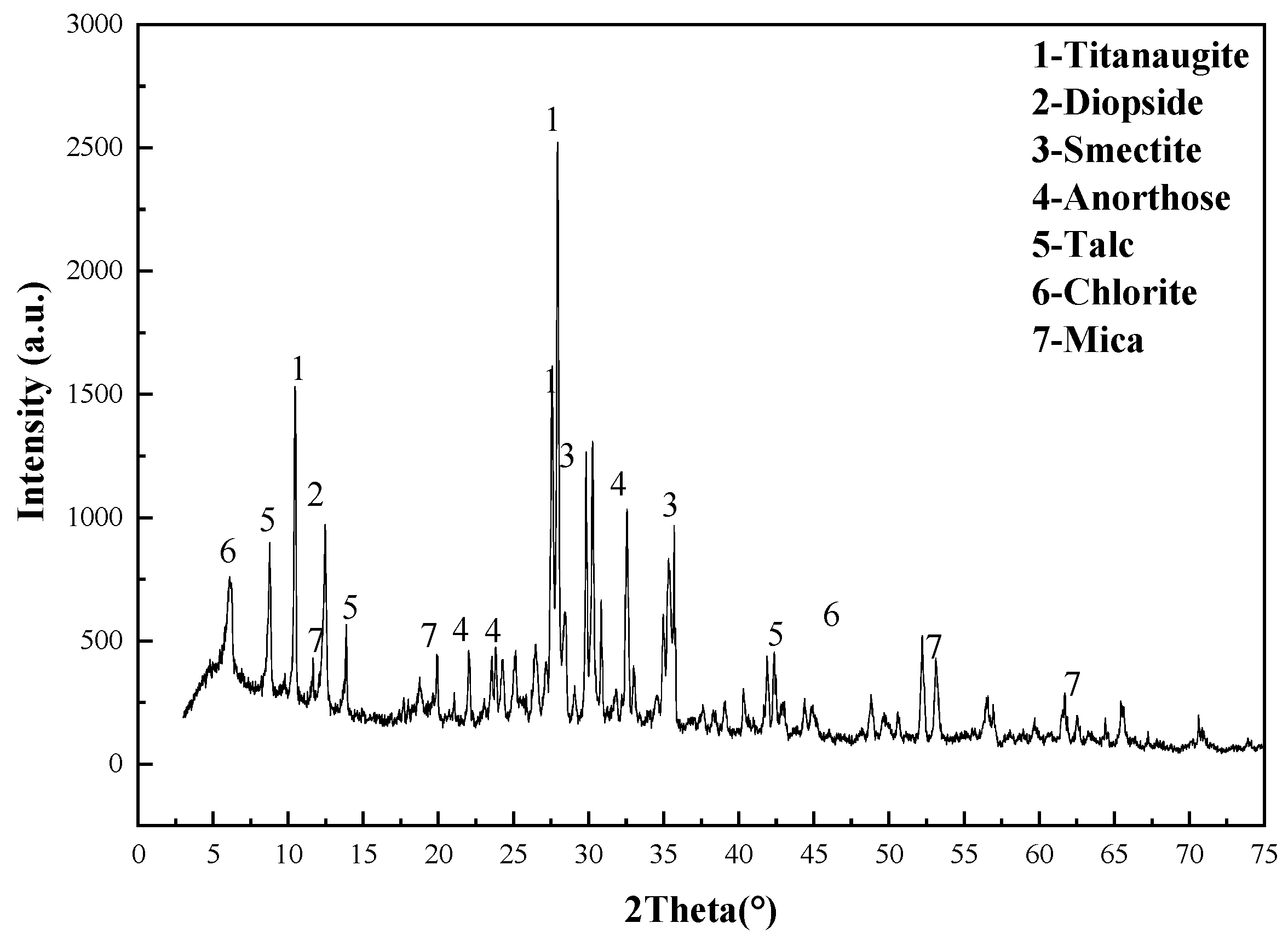
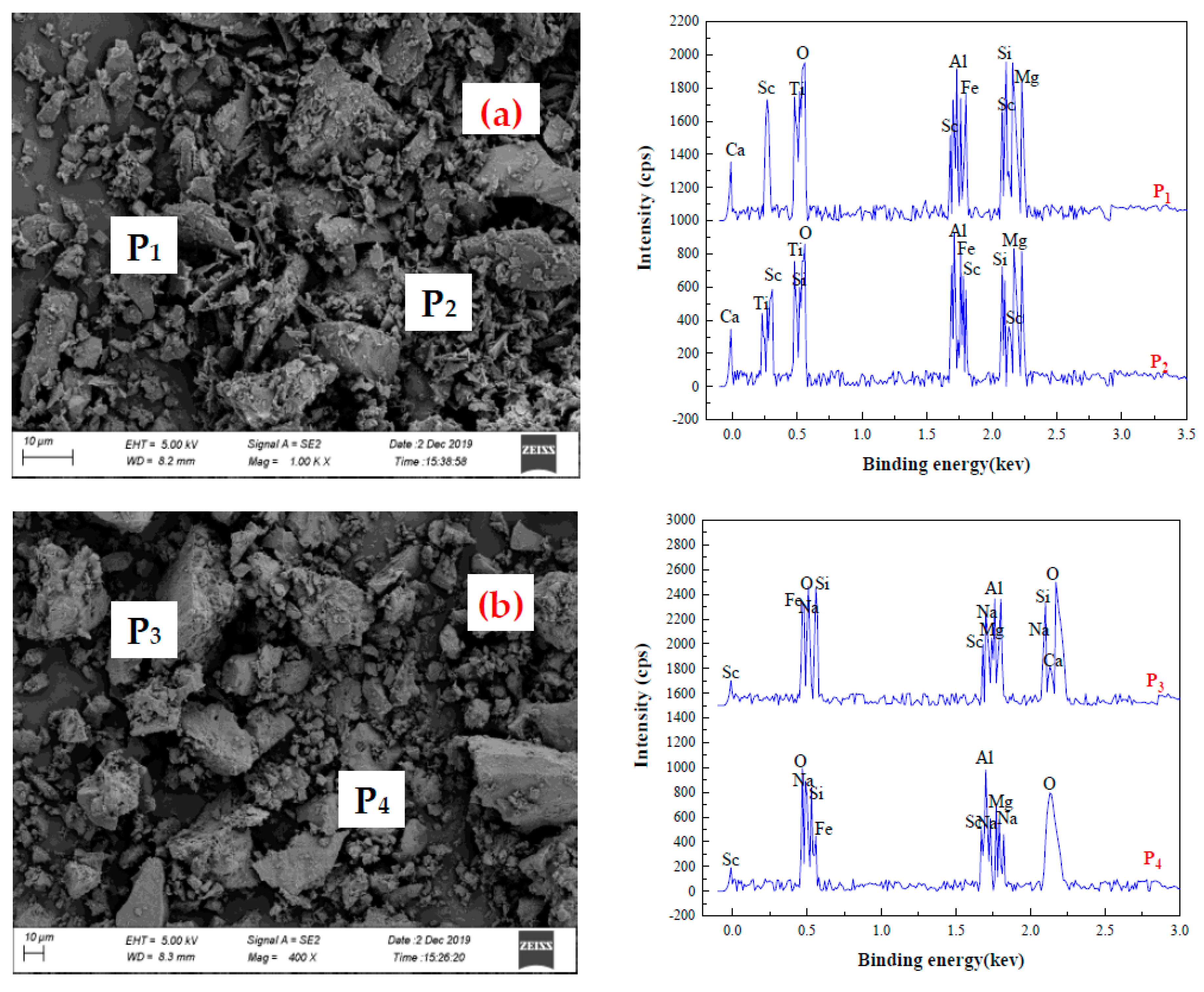
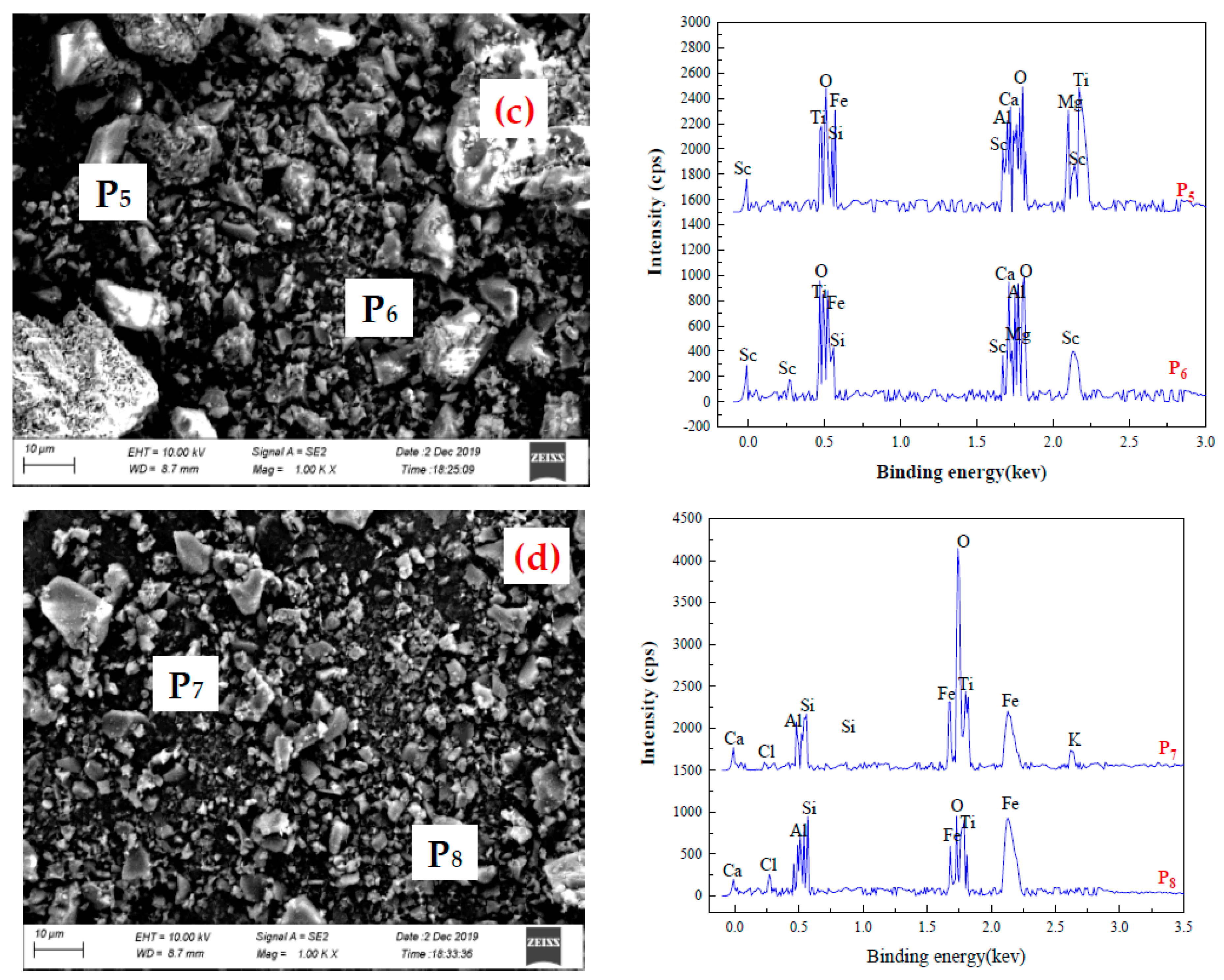
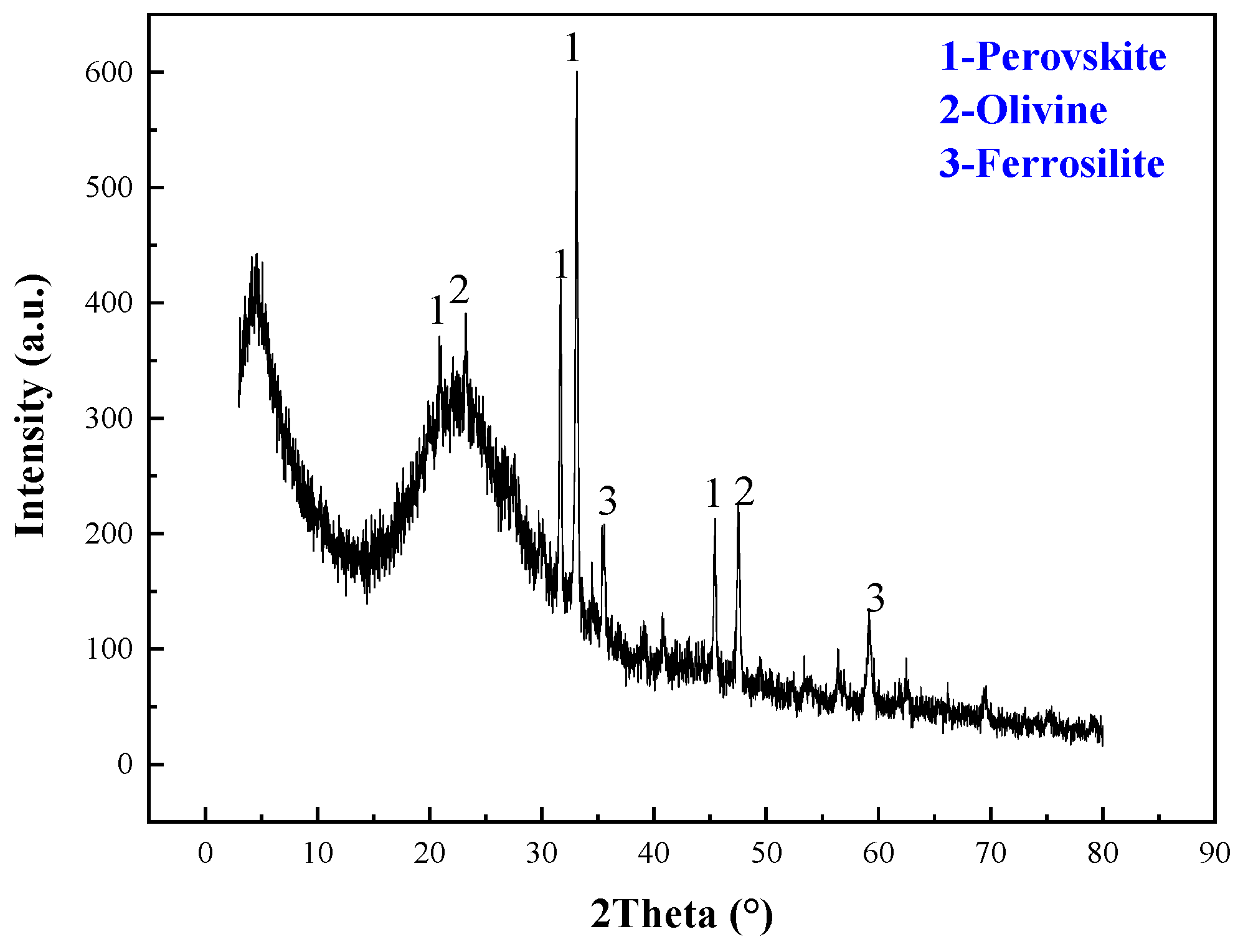
3.3. Analysis and Characterization of Scandium Rough Concentrate, Roasting Ores, Hydrolysis Residue, and Leaching Residue
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Moëlo, Y.; Lulzac, Y.; Rouer, O.; Palvadeau, P.; Gloaguen, É.; Léone, P. Scandium mineralogy: Pretulite with scandian zircon and xenotime-(Y) within an apatite-rich oolitic ironstone from Saint-Aubin-Des-Châteaux, Armorican Massif, France. Can. Mineral. 2002, 40, 1657–1673. [Google Scholar] [CrossRef]
- Chi, R.A.; Wang, D.Z. Rare Earth Dressing and Extraction Technology; Science Press: Beijing, China, 1996; p. 293. [Google Scholar]
- Xiao, J.H.; Wang, J.M.; Wang, Z. Study on Occurrence State of Scandium of Scandium-Containing Rare Earth Ore in Western of Sichuan Province. Chin. Rare Earths 2018, 39, 40–47. (In Chinese) [Google Scholar]
- Anawati, J.; Azimi, G. Recovery of scandium from Canadian bauxite residue utilizing acid baking followed by water leaching. Waste Manag. 2019, 95, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Borra, C.R.; Mermans, J.; Blanpain, B.; Pontikes, Y.; Binnemans, K.; Van Gerven, T. Selective recovery of rare earths from bauxite residue by combination of sulfation, roasting and leaching. Miner. Eng. 2016, 92, 151–159. [Google Scholar] [CrossRef]
- Le, W.H.; Kuang, S.T.; Zhang, Z.F.; Wu, G.L.; Li, Y.L.; Liao, C.F.; Liao, W.P. Selective extraction and recovery of Scandium from sulfate medium by Cextrant 230. Hydrometallurgy 2018, 178, 54–59. [Google Scholar] [CrossRef]
- Onghena, B.; Borra, C.R.; Van Gerven, T.; Binnemans, K. Recovery of Scandium from sulfation-roasted leachates of bauxite residue by solvent extraction with the ionic liquid betainium bis (trifluoromethylsulfonyl) imide. Sep. Purif. Technol. 2017, 176, 208–219. [Google Scholar] [CrossRef]
- Li, G.H.; Ye, Q.; Deng, B.N.; Luo, J.; Rao, M.J.; Peng, Z.W.; Jiang, T. Extraction of scandium from scandium-rich material derived from bauxite ore residues. Hydrometallurgy 2018, 176, 62–68. [Google Scholar] [CrossRef]
- Li, S.C.; Kim, S.C.; Kang, C.G. Recovery of Scandium from KOH sub-molten salt leaching cake of fergusonite. Miner. Eng. 2019, 137, 200–206. [Google Scholar] [CrossRef]
- Biswal, J.; Goswami, S.; Pant, H.J.; Bamankar, Y.R.; Rao, T.V.R.V.; Upadhay, R.K.; Dash, A. Synthesis, Characterization, Neutron Activation, and Application of Scandium Oxide Microsphere in Radioactive Particle Tracking Experiments. Ind. Eng. Chem. Res. 2016, 55, 3–12. [Google Scholar] [CrossRef]
- Gao, L.K.; Rao, B.; Dai, H.X.; Hong, Z.; Xie, H.Y. Separation and Extraction of Scandium and Titanium from a Refractory Anatase Lixivium by Solvent Extraction with D2EHPA and Primary Amine N1923. J. Chem. Eng. Jpn. 2019, 52, 822–828. [Google Scholar] [CrossRef]
- Hu, J.S.; Zou, D.; Chen, J.; Li, D.Q. A novel synergistic extraction system for the recovery of Scandium (III) by Cyanex 272 and Cyanex 923 in sulfuric acid medium. Sep. Purif. Technol. 2019, 23, 115977. [Google Scholar]
- Xiao, J.H.; Zhang, Y.S. Recovering Cobalt and Sulfur in Low Grade Cobalt-Bearing V–Ti Magnetite Tailings Using Flotation Process. Processes 2019, 7, 536. [Google Scholar] [CrossRef]
- Jankovsky, O.; Sedmidubsky, D.; Simek, P.; Klimova, K.; Bousa, D.; Boothroyd, C.; Mackova, A.; Sofer, Z. Separation of thorium ions from wolframite and Scandium concentrates using graphene oxide. Phys. Chem. Chem. Phys. 2015, 17, 25272–25277. [Google Scholar] [CrossRef] [PubMed]
- Kostikova, G.V.; Mal’tseva, I.E.; Zhilov, V.I. Extraction Recovery of Scandium and Concomitant Elements with Isoamyldialkylphosphine Oxide from Different Media. Russ. J. Inorg. Chem. 2019, 64, 277–282. [Google Scholar] [CrossRef]
- Purcell, W.; Potgieter, H.; Nete, M.; Mnculwane, H. Possible methodology for niobium, tantalum and Scandium extraction in ferrocolumbite. Miner. Eng. 2018, 119, 57–66. [Google Scholar] [CrossRef]
- Rychkov, V.N.; Semenishchev, V.S.; Mashkovtsev, M.A.; Kirillov, E.V.; Kirillov, S.V.; Bunkov, G.M.; Botalov, M.S. Deactivation of the Scandium concentrate recovered from uranium leach liquors. J. Radioanal. Nucl. Chem. 2016, 310, 1247–1253. [Google Scholar] [CrossRef]
- Ding, W.; Xiao, J.H.; Peng, Y.; Shen, S.Y.; Chen, T. Iron Extraction from Red Mud using Roasting with Sodium Salt. Miner. Process. Extr. Metall. Rev. 2019. [Google Scholar] [CrossRef]
- Kudryavskii, Y. Technology of deep removal of impurities from Scandium oxide with the acquisition of high-purity Scandium oxide. Russ. J. Non-Ferr. Met. 2011, 52, 420–422. [Google Scholar] [CrossRef]
- Xiao, J.H.; Zhang, Y.S. Extraction of Cobalt and Iron from Refractory Co-Bearing Sulfur Concentrate. Processes 2020, 8, 200. [Google Scholar] [CrossRef]
- Kuzmin, V.I.; Kuzmina, V.N.; Gudkova, N.V.; Kuzmin, D.V. Extraction of metal bromides from chloride brines with mixtures of molecular iodine and tributyl phosphate. Hydrometallurgy 2018, 180, 221–228. [Google Scholar] [CrossRef]
- Sharaf, M.; Yoshida, W.; Kubota, F.; Goto, M. Selective Extraction of Scandium by a Long Alkyl Chain Carboxylic Acid/Organophosphonic Ester Binary Extractant. Solvent Extr. Ion Exch. 2018, 36, 647–657. [Google Scholar] [CrossRef]
- Xiao, J.H.; Zhou, L.L. Increasing Iron and Reducing Phosphorus Grades of Magnetic-Roasted High-Phosphorus Oolitic Iron Ore by Low-Intensity Magnetic Separation–Reverse Flotation. Processes 2019, 7, 388. [Google Scholar] [CrossRef]
- Smirnov, P.R.; Grechin, O.V. Structure of concentrated aqueous solutions of Scandium chloride. Russ. J. Phys. Chem. A 2017, 91, 517–520. [Google Scholar] [CrossRef]
- Simonova, T.N.; Fedotov, A.N.; Alemasova, N.V. Extraction of Scandium Nitrate and Thiocyanate Complexes Using Two-Phase Aqueous Systems and the Determination of Scandium. J. Anal. Chem. 2018, 73, 18–22. [Google Scholar] [CrossRef]
- Peng, Y.; Xiao, J.H.; Deng, B.; Wang, Z.; Liu Ny Yang, D.G.; Ding, W.; Chen, T.; Wu, Q. Study on separation of fine-particle ilmenite and mechanism using flocculation flotation with sodium oleate and polyacrylamide. Physicochem. Probl. Miner. Process. 2020, 56, 161–172. [Google Scholar]
- Zhang, W.; Yu, S.Q.; Zhang, S.C.; Zhou, J.; Ning, S.Y.; Wang, X.P.; Wei, Y.Z. Separation of Scandium from the other rare earth elements with a novel macro-porous silica-polymer based adsorbent HDEHP/SiO2-P. Hydrometallurgy 2019, 185, 117–124. [Google Scholar] [CrossRef]
- Souza, A.G.O.; Aliprandini, P.; Espinosa, D.C.R.; Tenorio, J.A.S. Scandium Extraction from Nickel Processing Waste Using Cyanex 923 in Sulfuric Medium. JOM 2019, 71, 2003–2009. [Google Scholar] [CrossRef]
- Xiao, J.H.; Ding, W.; Peng, Y.; Wu, Q.; Chen, Z.Q.; Wang, Z.; Wang, J.M.; Peng, T.F. Upgrading iron and removing phosphorus of high phosphorus Oolitic iron ore by segregation roasting with Calcium chloride and Calcium hypochlorite. J. Min. Metall. Sect. B Metall. 2019, 55, 305–314. [Google Scholar] [CrossRef]
- Xiao, J.H.; Liang, G.J.; Huang, W.X.; Ding, W.; Peng, Y.; Wu, Q. Research on Separating Iron and Scandium of Scandium-contained Red Mud Using Sodium Chloride Segregation Roasting-Low Intensity Magnetic Separation-Hydrochloric Acid Leaching. Adv. Eng. Sci. 2019, 51, 199–209. (In Chinese) [Google Scholar]
- Zhou, G.T.; Li, Q.G.; Sun, P.; Guan, W.J.; Zhang, G.Q.; Cao, Z.Y.; Zeng, L. Removal of impurities from Scandium chloride solution using 732-type resin. J. Rare Earths 2018, 36, 311–316. [Google Scholar] [CrossRef]
| MgO | Fe2O3 | SiO2 | Al2O3 | S | P2O5 | Sc2O3 | TiO2 | K2O | Na2O | CaO |
|---|---|---|---|---|---|---|---|---|---|---|
| 5.92 | 18.74 | 27.08 | 21.67 | 0.008 | 0.25 | 0.007698 | 6.93 | 1.63 | 1.72 | 5.91 |
| Diopside | Titanaugite | Smectite | Talc | Anorthose | Chlorite | Mica |
|---|---|---|---|---|---|---|
| 0.008256 | 0.008158 | 0.006822 | 0.004512 | 0.00522 | 0.003818 | 0.006623 |
| Fusion Agent | Content of Hydrolysis Residue | ||
|---|---|---|---|
| Sc2O3 (g/t) | SiO2 (wt %) | Al2O3 (wt %) | |
| NaHCO3 | 83.98 | 18.65 | 19.63 |
| NaOH | 95.91 | 12.86 | 18.23 |
| Na2O2 | 85.41 | 21.63 | 18.29 |
| NaHCO3 | 86.25 | 20.88 | 19.25 |
| Na2CO3 | 94.98 | 22.63 | 13.25 |
| CaO | MgO | Fe2O3 | SiO2 | Al2O3 | S | P2O5 | Sc2O3 | TiO2 | K2O | Na2O |
|---|---|---|---|---|---|---|---|---|---|---|
| 10.75 | 7.37 | 25.19 | 11.24 | 10.78 | 0.002 | 0.25 | 0.010824 | 14.93 | 0.83 | 1.19 |
| Leaching Agent | Sc2O3 Content of Leaching Residue (g/t) | Leaching Efficiency of Sc2O3 (wt %) | Dissolution Efficiency of Residue (wt %) |
|---|---|---|---|
| Sulfuric acid | 42.66 | 67.42 | 17.33 |
| hydrochloric acid | 33.87 | 76.17 | 23.86 |
| nitric acid | 38.71 | 73.79 | 26.74 |
| hydrofluoric acid | 56.89 | 52.37 | 9.37 |
| CaO | MgO | Fe2O3 | SiO2 | Al2O3 | S | P2O5 | Sc2O3 | TiO2 | K2O | Na2O |
|---|---|---|---|---|---|---|---|---|---|---|
| 8.89 | 6.93 | 15.65 | 35.89 | 1.44 | 0.001 | 0.05 | 0.000812 | 20.63 | 0.15 | 0.09 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiao, J.; Peng, Y.; Ding, W.; Chen, T.; Zou, K.; Wang, Z. Recovering Scandium from Scandium Rough Concentrate Using Roasting-Hydrolysis-Leaching Process. Processes 2020, 8, 365. https://doi.org/10.3390/pr8030365
Xiao J, Peng Y, Ding W, Chen T, Zou K, Wang Z. Recovering Scandium from Scandium Rough Concentrate Using Roasting-Hydrolysis-Leaching Process. Processes. 2020; 8(3):365. https://doi.org/10.3390/pr8030365
Chicago/Turabian StyleXiao, Junhui, Yang Peng, Wei Ding, Tao Chen, Kai Zou, and Zhen Wang. 2020. "Recovering Scandium from Scandium Rough Concentrate Using Roasting-Hydrolysis-Leaching Process" Processes 8, no. 3: 365. https://doi.org/10.3390/pr8030365
APA StyleXiao, J., Peng, Y., Ding, W., Chen, T., Zou, K., & Wang, Z. (2020). Recovering Scandium from Scandium Rough Concentrate Using Roasting-Hydrolysis-Leaching Process. Processes, 8(3), 365. https://doi.org/10.3390/pr8030365






