Modeling and Simulation of Crystallization of Metal–Organic Frameworks
Abstract
1. Introduction
2. Methods
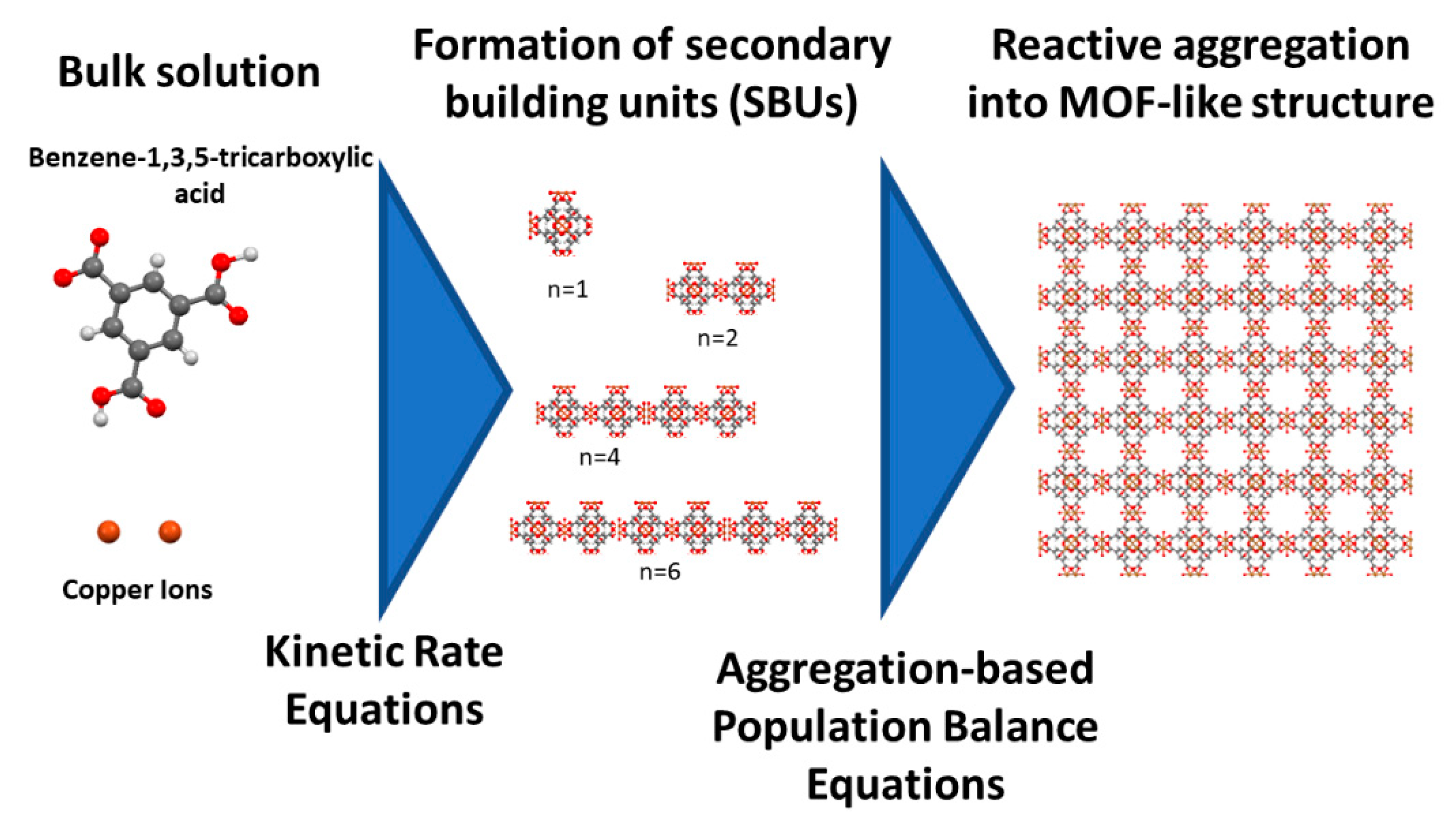
2.1. Formation of Secondary Building Units for MOF Production
2.2. Concurrent Crystallization of BTC
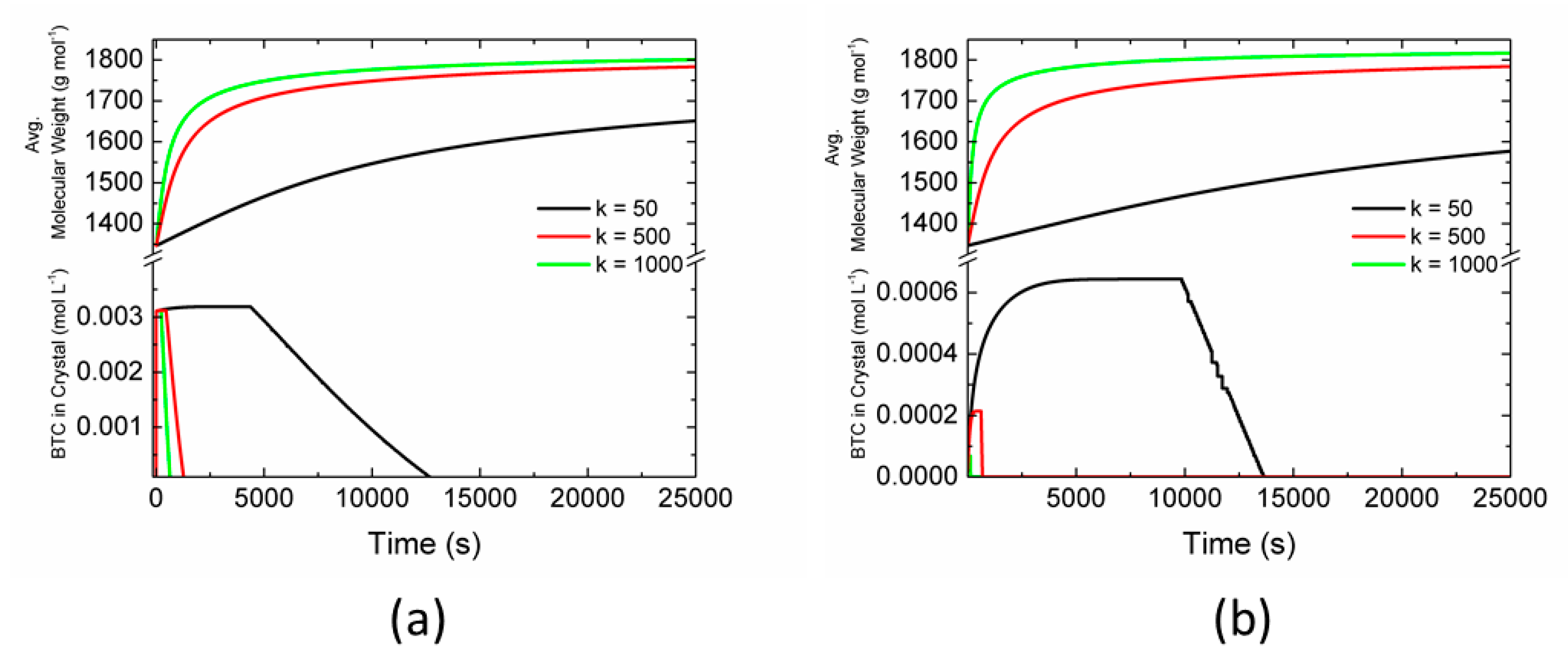
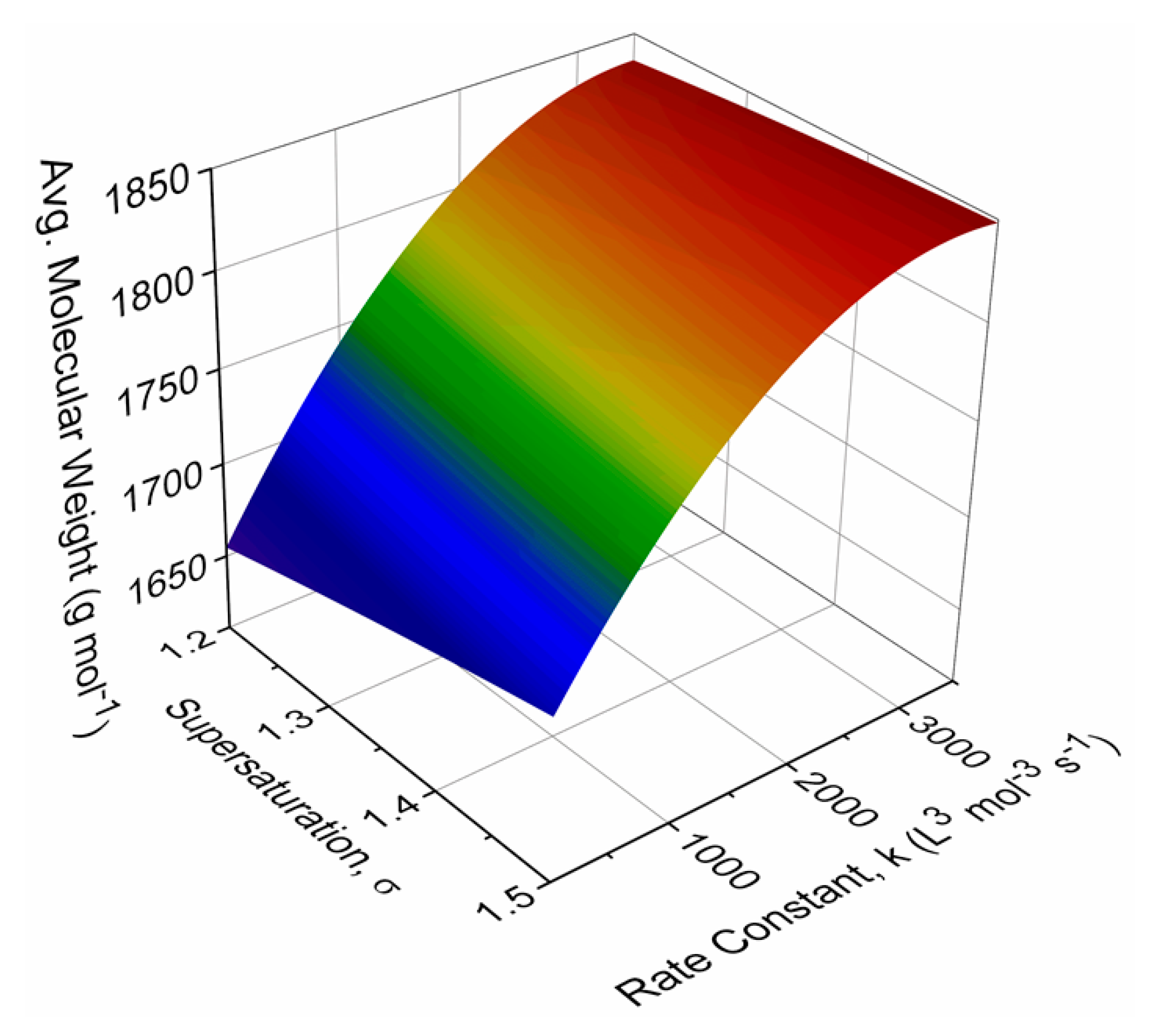
2.3. Effect of Parameters
2.4. Modeling of MOF Synthesis Using Aggregation-Based Population Balance Equation
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sumida, K.; Rogow, D.L.; Mason, J.A.; McDonald, T.M.; Bloch, E.D.; Herm, Z.R.; Bae, T.-H.; Long, J.R. Carbon Dioxide Capture in Metal–Organic Frameworks. Chem. Rev. 2012, 112, 724–781. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Sculley, J.; Zhou, H. Metal–Organic Frameworks for Separations. Chem. Rev. 2012, 112, 869–932. [Google Scholar] [CrossRef] [PubMed]
- Mason, J.A.; Veenstra, M.; Long, J.R. Evaluating metal–organic frameworks for natural gas storage. Chem. Sci. 2014, 5, 32–51. [Google Scholar] [CrossRef]
- Yaghi, O.M. Crystalline Metal-Organic Microporous Materials. U.S. Patent No. 5,648,508, 15 July 1997. [Google Scholar]
- Rubio-Martinez, M.; Avci-Camur, C.; Thornton, A.W.; Imaz, I.; Maspoch, D.; Hill, M.R. New synthetic routes towards MOF production at scale. Chem. Soc. Rev. 2017, 46, 3453–3480. [Google Scholar] [CrossRef] [PubMed]
- Kalmutzki, M.J.; Hanikel, N.; Yaghi, O.M. Secondary building units as the turning point in the development of the reticular chemistry of MOFs. Sci. Adv. 2018, 4, eaat9180. [Google Scholar] [CrossRef]
- Shoaee, M.; Anderson, M.W.; Attfield, M.P. Crystal Growth of the Nanoporous Metal-Organic Framework HKUST-1 Revealed by In Situ Atomic Force Microscopy. Angew. Chem. Int. Ed. 2008, 47, 8525–8528. [Google Scholar] [CrossRef]
- Laybourn, A.; López-Fernández, A.M.; Thomas-Hillman, I.; Katrib, J.; Lewis, W.; Dodds, C.; Harvey, A.P.; Kingman, S.W. Combining continuous flow oscillatory baffled reactors and microwave heating: Process intensification and accelerated synthesis of metal-organic frameworks. Chem. Eng. J. 2019, 356, 170–177. [Google Scholar] [CrossRef]
- Shekhah, O.; Wang, H.; Zacher, D.; Fischer, R.A.; Wöll, C. Growth Mechanism of Metal-Organic Frameworks: Insights into the Nucleation by Employing a Step-by-Step Route. Angew. Chemie Int. Ed. 2009, 48, 5038–5041. [Google Scholar] [CrossRef]
- Millange, F.; El Osta, R.; Medina, M.E.; Walton, R.I. A time-resolved diffraction study of a window of stability in the synthesis of a copper carboxylate metal–organic framework. CrystEngComm 2011, 13, 103–108. [Google Scholar] [CrossRef]
- Colón, Y.J.; Snurr, R.Q. High-throughput computational screening of metal–organic frameworks. Chem. Soc. Rev. 2014, 43, 5735–5749. [Google Scholar] [CrossRef]
- Bernales, V.; Ortuño, M.A.; Truhlar, D.G.; Cramer, C.J.; Gagliardi, L. Computational Design of Functionalized Metal–Organic Framework Nodes for Catalysis. ACS Cent. Sci. 2018, 4, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Haldoupis, E.; Nair, S.; Sholl, D.S. Finding MOFs for Highly Selective CO2/N2 Adsorption Using Materials Screening Based on Efficient Assignment of Atomic Point Charges. J. Am. Chem. Soc. 2012, 134, 4313–4323. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.; Xie, L.-H.H.; Li, J.-R.R.; Ma, Y.; Seminario, J.M.; Balbuena, P.B. CO2 Capture and Separations Using MOFs: Computational and Experimental Studies. Chem. Rev. 2017, 117, 9674–9754. [Google Scholar] [CrossRef] [PubMed]
- Schäfer, P.; Kapteijn, F.; van der Veen, M.A.; Domke, K.F. Understanding the Inhibiting Effect of BTC on CuBTC Growth through Experiment and Modeling. Cryst. Growth Des. 2017, 17, 5603–5607. [Google Scholar] [CrossRef]
- Ray, W.H.; Laurence, R.L. Polymerization reaction engineering. In Chemical Reactor Theory; Prentice-Hall: Englewood Cliffs, NJ, USA, 1977; pp. 532–582. [Google Scholar]
- Kumar, S.; Ramkrishna, D. On the solution of population balance equations by discretization—I. A fixed pivot technique. Chem. Eng. Sci. 1996, 51, 1311–1332. [Google Scholar] [CrossRef]
- Van Assche, T.R.C.; Desmet, G.; Ameloot, R.; De Vos, D.E.; Terryn, H.; Denayer, J.F.M. Electrochemical synthesis of thin HKUST-1 layers on copper mesh. Microporous Mesoporous Mater. 2012, 158, 209–213. [Google Scholar] [CrossRef]
- Apelblat, A.; Manzurola, E.; Abo Balal, N. The solubilities of benzene polycarboxylic acids in water. J. Chem. Thermodyn. 2006, 38, 565–571. [Google Scholar] [CrossRef]
- Huggins, M.L. Principles of Polymer Chemistry. J. Am. Chem. Soc. 1954, 76, 2854. [Google Scholar] [CrossRef]
- Varga, J.; Stoll, K.; Menyhárd, A.; Horváth, Z. Crystallization of isotactic polypropylene in the presence of a β-nucleating agent based on a trisamide of trimesic acid. J. Appl. Polym. Sci. 2011, 121, 1469–1480. [Google Scholar] [CrossRef]
- Singh, M.R.; Nere, N.; Tung, H.-H.; Mukherjee, S.; Bordawekar, S.; Ramkrishna, D. Measurement of Polar Plots of Crystal Dissolution Rates Using Hot-Stage Microscopy. Some Further Insights into Dissolution Morphologies. Cryst. Growth Des. 2014, 14, 5647–5661. [Google Scholar] [CrossRef]
- Ramkrishna, D.; Singh, M.R. Population Balance Modeling: Current Status and Future Prospects. Annu. Rev. Chem. Biomol. Eng. 2014, 5, 123–146. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.R.; Ramkrishna, D. A Comprehensive Approach to Predicting Crystal Morphology Distributions with Population Balances. Cryst. Growth Des. 2013, 13, 1397–1411. [Google Scholar] [CrossRef]
- Singh, M.R.; Ramkrishna, D. Dispersions in crystal nucleation and growth rates: Implications of fluctuation in supersaturation. Chem. Eng. Sci. 2014, 107, 102–113. [Google Scholar] [CrossRef]
- Singh, M.R.; Verma, P.; Tung, H.-H.; Bordawekar, S.; Ramkrishna, D. Screening Crystal Morphologies from Crystal Structure. Cryst. Growth Des. 2013, 13, 1390–1396. [Google Scholar] [CrossRef]
- Singh, M.R.; Chakraborty, J.; Nere, N.; Tung, H.-H.; Bordawekar, S.; Ramkrishna, D. Image-Analysis-Based Method for 3D Crystal Morphology Measurement and Polymorph Identification Using Confocal Microscopy. Cryst. Growth Des. 2012, 12, 3735–3748. [Google Scholar] [CrossRef]
- Coliaie, P.; Kelkar, M.S.; Nere, N.K.; Singh, M.R. Continuous-flow, well-mixed, microfluidic crystallization device for screening of polymorphs, morphology, and crystallization kinetics at controlled supersaturation. Lab Chip 2019, 19, 2373–2382. [Google Scholar] [CrossRef] [PubMed]
- Ramkrishna, D. Population Balances. Chem. Eng. 2000, 1, 355. [Google Scholar]


| Description | Equation | Parameters | Ranges | Units |
|---|---|---|---|---|
| Saturation (in terms of BTC concentration) | - | |||
| Nucleation | 1.8 | - | ||
| Growth | 2 | - | ||
| Oligomerization Reaction |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dighe, A.V.; Nemade, R.Y.; Singh, M.R. Modeling and Simulation of Crystallization of Metal–Organic Frameworks. Processes 2019, 7, 527. https://doi.org/10.3390/pr7080527
Dighe AV, Nemade RY, Singh MR. Modeling and Simulation of Crystallization of Metal–Organic Frameworks. Processes. 2019; 7(8):527. https://doi.org/10.3390/pr7080527
Chicago/Turabian StyleDighe, Anish V., Roshan Y. Nemade, and Meenesh R. Singh. 2019. "Modeling and Simulation of Crystallization of Metal–Organic Frameworks" Processes 7, no. 8: 527. https://doi.org/10.3390/pr7080527
APA StyleDighe, A. V., Nemade, R. Y., & Singh, M. R. (2019). Modeling and Simulation of Crystallization of Metal–Organic Frameworks. Processes, 7(8), 527. https://doi.org/10.3390/pr7080527






