Unraveling Co-Pyrolysis Mechanisms for Municipal Sludge and Microplastics: Thermodynamic, Kinetic, and Product Insights
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Preparation and Characterization
2.2. TG Experiments and Parameter Estimation
2.2.1. TG Experiments
2.2.2. Comprehensive Pyrolysis Index
2.2.3. Interaction Analysis
2.2.4. Principal Component Analysis
2.2.5. Kinetic and Thermodynamic Analyses
2.2.6. Kinetic Analysis
2.3. Char Characterization via Tube Furnace Simulation
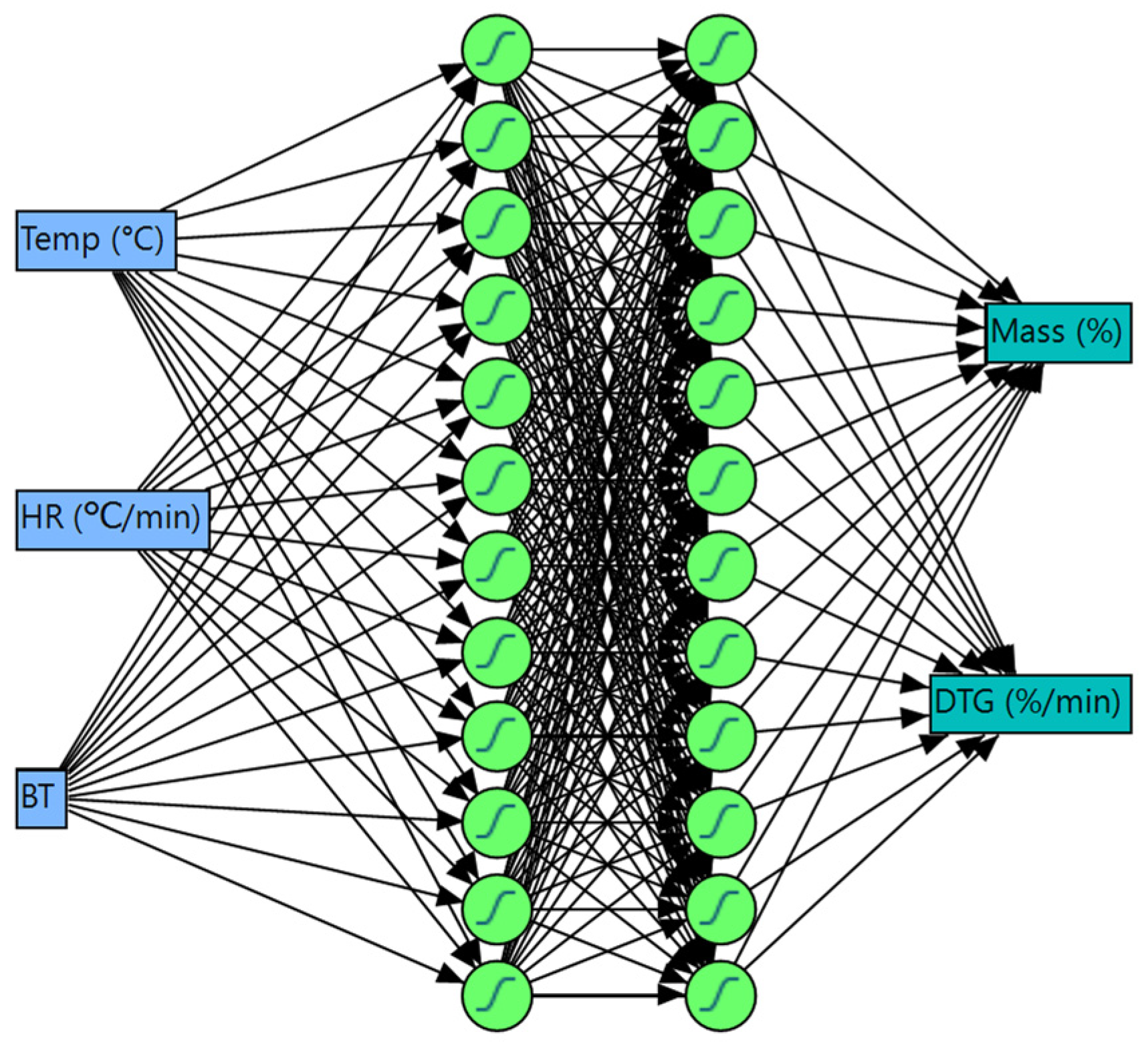
2.4. Artificial Neural Network-Based Joint Optimization
3. Results and Discussion
3.1. Physicochemical Drivers and Patterns
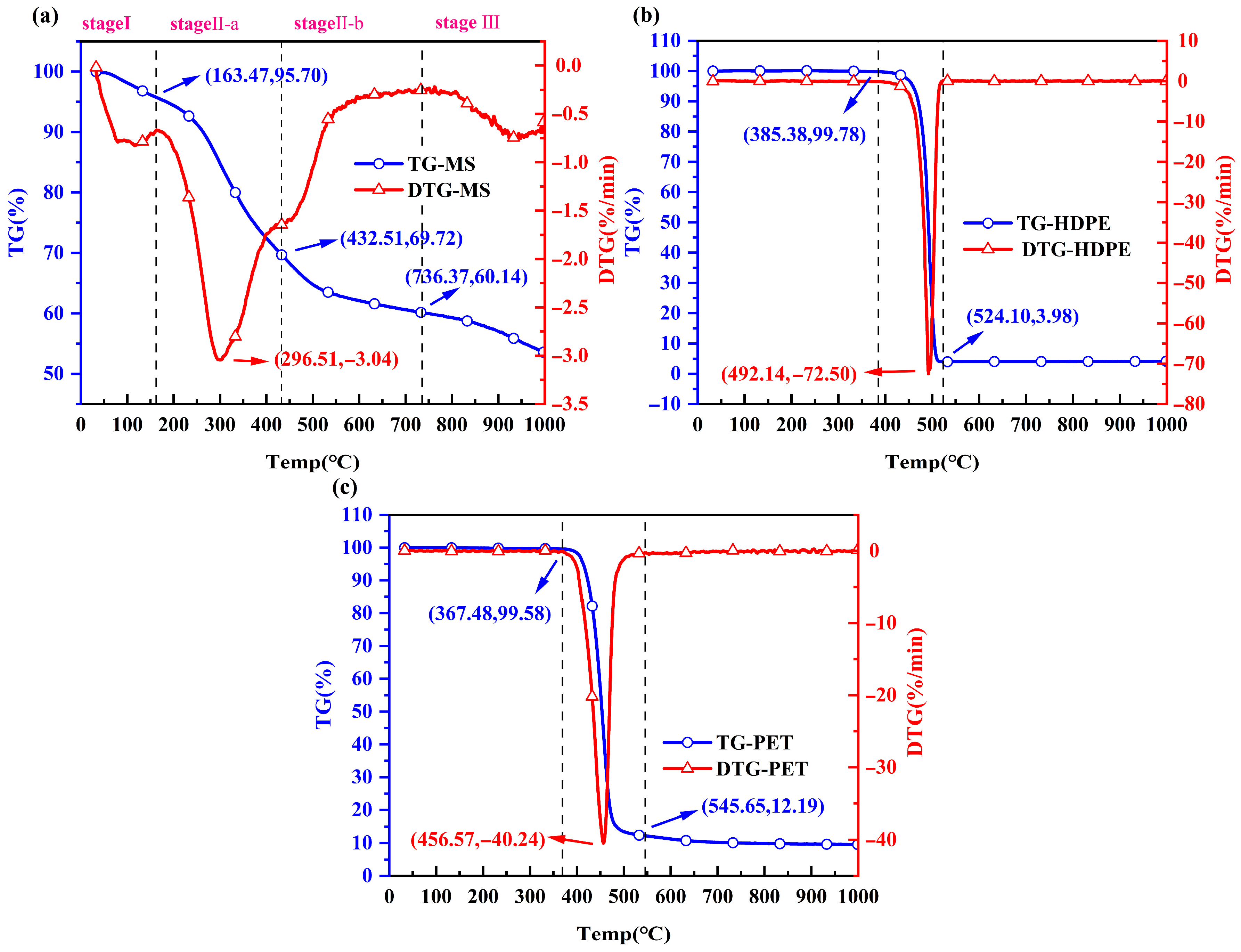
3.2. Thermal Decomposition Amount and Rate of Mono-Pyrolysis
3.3. (Co-)Pyrolysis Performances as a Function of Temperature
3.4. (Co-)Pyrolysis Performances as a Function of Blend Ratio
3.5. Interaction Effects
3.6. Relative Contributions of the Co-Pyrolysis Components
3.7. Mono-Pyrolytic Kinetics of HDPE and PET
3.8. Mono-Pyrolytic Thermodynamics of HDPE and PET
3.9. Mono-Pyrolysis Mechanisms of HDPE and PET
3.10. (Co-)Pyrolysis Kinetics
3.11. (Co-)Pyrolytic Chars
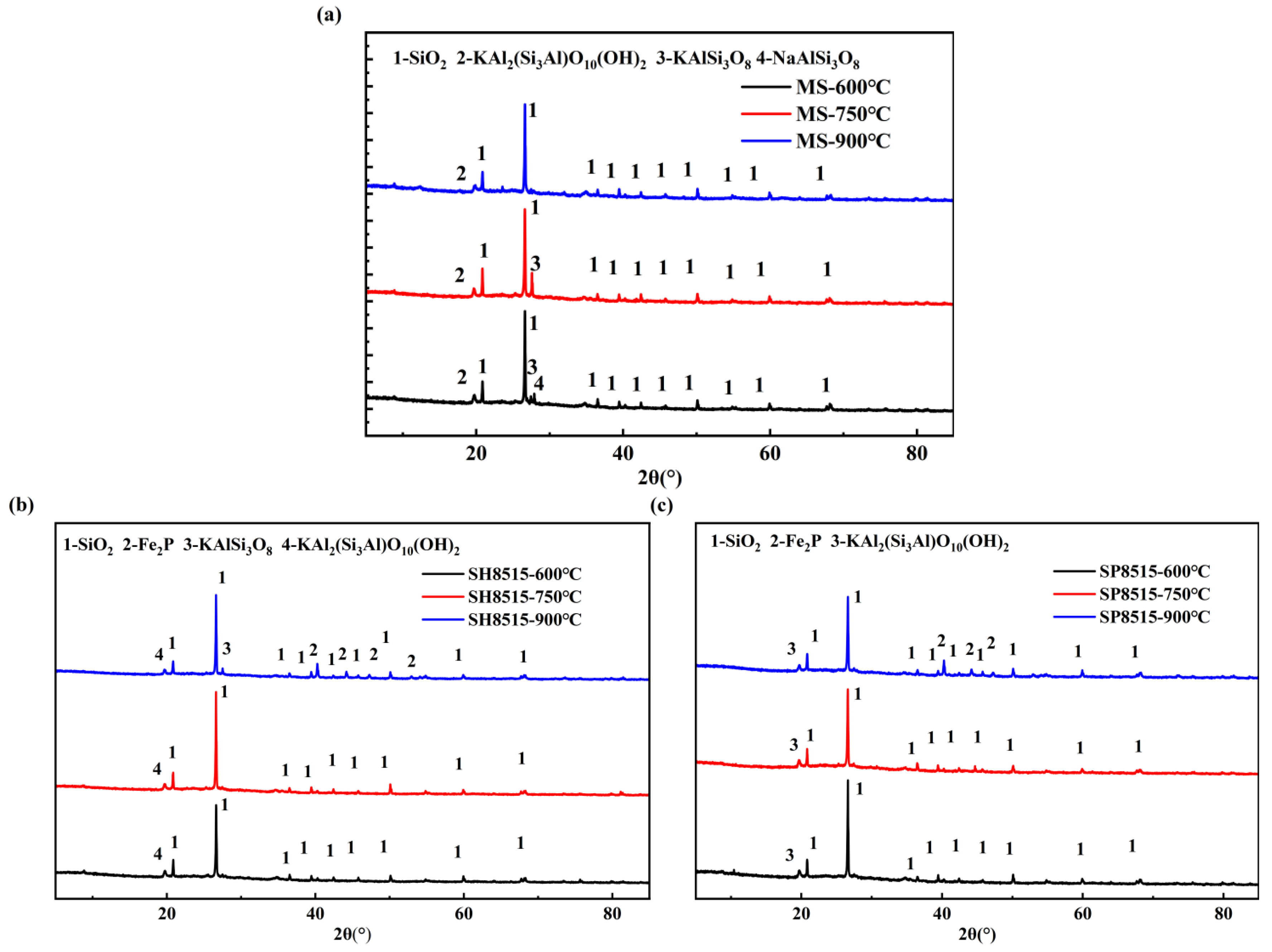
3.11.1. XRF Analysis
3.11.2. Crystal Phases of (Co-)Pyrolytic Chars
3.11.3. Micromorphology of (Co-)Pyrolytic Chars
3.11.4. Functional Groups of (Co-)Pyrolytic Chars
3.12. Operationally Optimal Settings
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Ding, Z.; Chen, Z.; Liu, J.; Evrendilek, F.; He, Y.; Xie, W. Co-combustion, life-cycle circularity, and artificial intelligence-based multi-objective optimization of two plastics and textile dyeing sludge. J. Hazard. Mater. 2022, 426, 128069. [Google Scholar] [CrossRef]
- Gatidou, G.; Arvaniti, O.S.; Stasinakis, A.S. Review on the occurrence and fate of microplastics in Sewage Treatment Plants. J. Hazard. Mater. 2019, 367, 504–512. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Chen, Y. Effects of microplastics on wastewater and sewage sludge treatment and their removal: A review. Chem. Eng. J. 2020, 382, 122955. [Google Scholar] [CrossRef]
- Eerkes-Medrano, D.; Thompson, R.C.; Aldridge, D.C. Microplastics in freshwater systems: A review of the emerging threats, identification of knowledge gaps and prioritisation of research needs. Water Res. 2015, 75, 63–82. [Google Scholar] [CrossRef]
- Ma, J.; Gong, Z.; Wang, Z.; Liu, H.; Chen, G.; Guo, G. Elucidating degradation properties, microbial community, and mechanism of microplastics in sewage sludge under different terminal electron acceptors conditions. Bioresour. Technol. 2022, 346, 126624. [Google Scholar] [CrossRef] [PubMed]
- Halsband, C.; Herzke, D. Plastic litter in the European Arctic: What do we know? Emerg. Contam. 2019, 5, 308–318. [Google Scholar] [CrossRef]
- Li, W.; Lo, H.-S.; Wong, H.-M.; Zhou, M.; Wong, C.-Y.; Tam, N.F.-Y.; Cheung, S.-G. Heavy metals contamination of sedimentary microplastics in Hong Kong. Mar. Pollut. Bull. 2020, 153, 110977. [Google Scholar] [CrossRef]
- Dai, L.; Zhou, N.; Lv, Y.; Cheng, Y.; Wang, Y.; Liu, Y.; Cobb, K.; Chen, P.; Lei, H.; Ruan, R. Pyrolysis technology for plastic waste recycling: A state-of-the-art review. Prog. Energy Combust. Sci. 2022, 93, 101021. [Google Scholar] [CrossRef]
- Milato, J.V.; França, R.J.; Calderari, M.R.M. Marques Calderari, Co-pyrolysis of oil sludge with polyolefins: Evaluation of different Y zeolites to obtain paraffinic products. J. Environ. Chem. Eng. 2020, 8, 103805. [Google Scholar] [CrossRef]
- Ni, B.-J.; Zhu, Z.-R.; Li, W.-H.; Yan, X.; Wei, W.; Xu, Q.; Xia, Z.; Dai, X.; Sun, J. Microplastics Mitigation in Sewage Sludge through Pyrolysis: The Role of Pyrolysis Temperature. Environ. Sci. Technol. Lett. 2020, 7, 961–967. [Google Scholar] [CrossRef]
- Yuan, Z.; Huang, Q.; Wang, Z.; Wang, H.; Luo, J.; Zhu, N.; Cao, X.; Lou, Z. Medium-Low Temperature Conditions Induce the Formation of Environmentally Persistent Free Radicals in Microplastics with Conjugated Aromatic-Ring Structures during Sewage Sludge Pyrolysis. Environ. Sci. Technol. 2022, 56, 2c04453. [Google Scholar] [CrossRef]
- Li, W.; Meng, J.; Zhang, Y.; Haider, G.; Ge, T.; Zhang, H.; Li, Z.; Yu, Y.; Shan, S. Co-pyrolysis of sewage sludge and metal-free/metal-loaded polyvinyl chloride (PVC) microplastics improved biochar properties and reduced environmental risk of heavy metals. Environ. Pollut. 2022, 302, 119092. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Chen, X.; He, Y.; Evrendilek, F.; Chen, Z.; Liu, J. Co-pyrolytic performances, mechanisms, gases, oils, and chars of textile dyeing sludge and waste shared bike tires under varying conditions. Chem. Eng. J. 2022, 428, 131053. [Google Scholar] [CrossRef]
- Nguyen, Q.V.; Choi, Y.S.; Choi, S.K.; Jeong, Y.W.; Han, S.Y. Co-pyrolysis of coffee-grounds and waste polystyrene foam: Synergistic effect and product characteristics analysis. Fuel 2021, 292, 120375. [Google Scholar] [CrossRef]
- Ma, M.-Y.; Xu, D.-H.; Guo, Y.; Wang, S.-Z.; He, Y.-L. Impacts of microplastics in municipal sludge pyrolysis-gasification: Melt-induced microaggregation and increased deposition risk. Renew. Sustain. Energy Rev. 2025, 214, 115542. [Google Scholar] [CrossRef]
- Xiong, Q.; Li, Y.; Hou, C.; Yang, J.; Zhou, X.; Ma, X.; Zuo, X.; Wu, X. How microplastics affect sludge pyrolysis behavior: Thermogravimetry-mass spectrum analysis and biochar characteristics. Waste Manag. 2023, 172, 108–116. [Google Scholar] [CrossRef]
- Wang, Z.; Burra, K.G.; Lei, T.; Gupta, A.K. Co-pyrolysis of waste plastic and solid biomass for synergistic production of biofuels and chemicals-A review. Prog. Energy Combust. Sci. 2021, 84, 100899. [Google Scholar] [CrossRef]
- Kong, Y.; Wang, R.; Zhou, Q.; Li, J.; Fan, Y.; Chen, Q. Recent progresses and perspectives of polyethylene biodegradation by bacteria and fungi: A review. J. Contam. Hydrol. 2025, 269, 104499. [Google Scholar] [CrossRef]
- Lee, W.-B.; Jae, J.; Kim, J.; Kwon, J.; Kim, Y.-M. Co-feeding effect of municipal sludge on the pyrolysis of polyethylene terephthalate. Korean J. Chem. Eng. 2023, 40, 2701–2707. [Google Scholar] [CrossRef]
- Fardi, Z.; Shahbeik, H.; Nosrati, M.; Motamedian, E.; Tabatabaei, M.; Aghbashlo, M. Waste-to-energy: Co-pyrolysis of potato peel and macroalgae for biofuels and biochemicals. Environ. Res. 2023, 242, 117614. [Google Scholar] [CrossRef] [PubMed]
- Shafizadeh, A.; Shahbeik, H.; Nadian, M.H.; Gupta, V.K.; Nizami, A.-S.; Lam, S.S.; Peng, W.; Pan, J.; Tabatabaei, M.; Aghbashlo, M. Turning hazardous volatile matter compounds into fuel by catalytic steam reforming: An evolutionary machine learning approach. J. Clean. Prod. 2023, 413, 137329. [Google Scholar] [CrossRef]
- Chen, X.; Shafizadeh, A.; Shahbeik, H.; Rafiee, S.; Golvirdizadeh, M.; Moradi, A.; Peng, W.; Tabatabaei, M.; Aghbashlo, M. Machine learning-based optimization of catalytic hydrodeoxygenation of biomass pyrolysis oil. J. Clean. Prod. 2024, 437, 140738. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, J.; Tao, L.; Jia, D.; Ke, G.; Evrendilek, F.; Apul, O.G.; Zhuang, P.; He, Y.; Li, W.; et al. Interaction effects of feedstock and temperature on biogas production during torrefaction-coupled catalytic stepwise pyrolysis of phytoremediation biomass. Renew. Energy 2025, 260, 125131. [Google Scholar] [CrossRef]
- Ma, D.; Yao, Q.; Wang, J.; Hao, Q.; Chen, H.; Ma, L.; Sun, M.; Ma, X. Simple descriptor based machine learning model development for synergy prediction of different metal loadings and solvent swellings on coal pyrolysis. Chem. Eng. Sci. 2022, 252, 117538. [Google Scholar] [CrossRef]
- Derringer, G.; Suich, R. Simultaneous optimization of several response variables. J. Qual. Technol. 1980, 12, 214–219. [Google Scholar] [CrossRef]
- Chang, X.; Wu, P.; Chu, Y.; Zhou, Y.; Tang, Y. Pyrolysis-induced migration and transformation of heavy metals in sewage sludge containing microplastics. Waste Manag. 2024, 189, 401–409. [Google Scholar] [CrossRef]
- Zhao, X.; Wan, C.; Pan, Y.; Fan, Y.; Liu, X. Pyrolysis behavior of sewage sludge coexisted with microplastics: Kinetics, mechanism, and product characteristics. J. Environ. Manag. 2024, 370, 123030. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Barta-Rajnai, E.; Skreiberg, Ø.; Khalil, R.; Czégény, Z.; Jakab, E.; Barta, Z.; Grønli, M. Effect of torrefaction on physiochemical characteristics and grindability of stem wood, stump and bark. Appl. Energy 2018, 227, 137–148. [Google Scholar] [CrossRef]
- Ding, K.; Zhong, Z.; Zhang, B.; Song, Z.; Qian, X. Pyrolysis Characteristics of Waste Tire in an Analytical Pyrolyzer Coupled with Gas Chromatography/Mass Spectrometry. Energy Fuels 2015, 29, 3181–3187. [Google Scholar] [CrossRef]
- Chen, J.; Mu, L.; Jiang, B.; Yin, H.; Song, X.; Li, A. TG/DSC-FTIR and Py-GC investigation on pyrolysis characteristics of petrochemical wastewater sludge. Bioresour. Technol. 2015, 192, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, J.; Li, L.; Chen, X.; Lin, Z.; Yang, C.; Evrendilek, F.; Li, W.; Huang, W.; He, Y.; et al. Optimizing pyrolysis of herbal tea and Salvia miltiorrhiza residues for sustainable energy and product recovery. Chem. Eng. J. 2025, 513, 162694. [Google Scholar] [CrossRef]
- Barta-Rajnai, E.; Jakab, E.; Sebestyén, Z.; May, Z.; Barta, Z.; Wang, L.; Skreiberg, Ø.; Grønli, M.; Bozi, J.; Czégény, Z. Comprehensive Compositional Study of Torrefied Wood and Herbaceous Materials by Chemical Analysis and Thermoanalytical Methods. Energy Fuels 2016, 30, 8019–8030. [Google Scholar] [CrossRef]
- Naqvi, S.R.; Tariq, R.; Hameed, Z.; Ali, I.; Naqvi, M.; Chen, W.-H.; Ceylan, S.; Rashid, H.; Ahmad, J.; Taqvi, S.A.; et al. Pyrolysis of high ash sewage sludge: Kinetics and thermodynamic analysis using Coats-Redfern method. Renew. Energy 2019, 131, 854–860. [Google Scholar] [CrossRef]
- Ilo, O.P.; Simatele, M.D. Water hyacinth biorefinery: Improved biofuel production using Trichoderma atroviride pretreatment. Biofuels Bioprod. Biorefin. 2024, 19, 68–84. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, Q.; Li, Y.; Zhang, H. Modeling and optimization of photo-fermentation biohydrogen production from co-substrates basing on response surface methodology and artificial neural network integrated genetic algorithm. Bioresour. Technol. 2023, 374, 128789. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Liu, B.; Xue, Y.; Wang, W.; Chen, S. Effect of textile waste on incineration behavior of dyeing sludge: Combustion characteristics, gas emissions, kinetics. J. Clean. Prod. 2024, 435, 140619. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, G.; Liu, J.; Evrendilek, F.; Buyukada, M. Co-combustion of textile dyeing sludge with cattle manure: Assessment of thermal behavior, gaseous products, and ash characteristics. J. Clean. Prod. 2020, 253, 119950. [Google Scholar] [CrossRef]
- Wang, C.; Bi, H.; Lin, Q.; Jiang, X.; Jiang, C. Co-pyrolysis of sewage sludge and rice husk by TG-FTIR-MS: Pyrolysis behavior, kinetics, and condensable/non-condensable gases characteristics. Renew Energy 2020, 160, 1048–1066. [Google Scholar] [CrossRef]
- Singh, R.; Ruj, B.; Sadhukhan, A.; Gupta, P. A TG-FTIR investigation on the co-pyrolysis of the waste HDPE, PP, PS and PET under high heating conditions. J. Energy Inst. 2020, 93, 1020–1035. [Google Scholar] [CrossRef]
- Singh, R.K.; Ruj, B.; Sadhukhan, A.; Gupta, P. Impact of fast and slow pyrolysis on the degradation of mixed plastic waste: Product yield analysis and their characterization. J. Energy Inst. 2019, 92, 1647–1657. [Google Scholar] [CrossRef]
- Dimitrov, N.; Krehula, L.K.; Siročić, A.P.; Hrnjak-Murgić, Z. Analysis of recycled PET bottles products by pyrolysis-gas chromatography. Polym. Degrad. Stab. 2013, 98, 972–979. [Google Scholar] [CrossRef]
- Vamvuka, D.; Salpigidou, N.; Kastanaki, E.; Sfakiotakis, S. Possibility of using paper sludge in co-firing applications. Fuel 2009, 88, 637–643. [Google Scholar] [CrossRef]
- Kan, T.; Strezov, V.; Evans, T. Catalytic Pyrolysis of Coffee Grounds Using NiCu-Impregnated Catalysts. Energy Fuels 2013, 28, 228–235. [Google Scholar] [CrossRef]
- Kan, T.; Grierson, S.; de Nys, R.; Strezov, V. Comparative Assessment of the Thermochemical Conversion of Freshwater and Marine Micro- and Macroalgae. Energy Fuels 2013, 28, 104–114. [Google Scholar] [CrossRef]
- Yao, D.; Li, H.; Dai, Y.; Wang, C.-H. Impact of temperature on the activity of Fe-Ni catalysts for pyrolysis and decomposition processing of plastic waste. Chem. Eng. J. 2020, 408, 127268. [Google Scholar] [CrossRef]
- Bi, H.; Wang, C.; Lin, Q.; Jiang, X.; Jiang, C.; Bao, L. Pyrolysis characteristics, artificial neural network modeling and environmental impact of coal gangue and biomass by TG-FTIR. Sci. Total. Environ. 2021, 751, 142293. [Google Scholar] [CrossRef] [PubMed]
- Brems, A.; Baeyens, J.; Vandecasteele, C.; Dewil, R. Polymeric Cracking of Waste Polyethylene Terephthalate to Chemicals and Energy. J. Air Waste Manag. Assoc. 2011, 61, 721–731. [Google Scholar] [CrossRef]
- Xiang, Z.; Liang, J.; Morgan, H.M.; Liu, Y.; Mao, H.; Bu, Q. Thermal behavior and kinetic study for co-pyrolysis of lignocellulosic biomass with polyethylene over Cobalt modified ZSM-5 catalyst by thermogravimetric analysis. Bioresour. Technol. 2018, 247, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Burra, K.G.; Wang, Z.; Li, J.; Che, D.; Gupta, A.K. On deconvolution for understanding synergistic effects in co-pyrolysis of pinewood and polypropylene. Appl. Energy 2020, 279, 115811. [Google Scholar] [CrossRef]
- Zhou, L.; Wang, Y.; Huang, Q.; Cai, J. Thermogravimetric characteristics and kinetic of plastic and biomass blends co-pyrolysis. Fuel Process. Technol. 2006, 87, 963–969. [Google Scholar] [CrossRef]
- Ko, K.-H.; Rawal, A.; Sahajwalla, V. Analysis of thermal degradation kinetics and carbon structure changes of co-pyrolysis between macadamia nut shell and PET using thermogravimetric analysis and 13C solid state nuclear magnetic resonance. Energy Convers. Manag. 2014, 86, 154–164. [Google Scholar] [CrossRef]
- Ko, K.-H.; Sahajwalla, V.; Rawal, A. Specific molecular structure changes and radical evolution during biomass-polyethylene terephthalate co-pyrolysis detected by 13C and 1H solid-state NMR. Bioresour. Technol. 2014, 170, 248–255. [Google Scholar] [CrossRef]
- Chattopadhyay, J.; Pathak, T.; Srivastava, R.; Singh, A. Catalytic co-pyrolysis of paper biomass and plastic mixtures (HDPE (high density polyethylene), PP (polypropylene) and PET (polyethylene terephthalate)) and product analysis. Energy 2016, 103, 513–521. [Google Scholar] [CrossRef]
- Xue, Y.; Kelkar, A.; Bai, X. Catalytic co-pyrolysis of biomass and polyethylene in a tandem micropyrolyzer. Fuel 2016, 166, 227–236. [Google Scholar] [CrossRef]
- Zhang, X.; Lei, H.; Zhu, L.; Qian, M.; Zhu, X.; Wu, J.; Chen, S. Enhancement of jet fuel range alkanes from co-feeding of lignocellulosic biomass with plastics via tandem catalytic conversions. Appl. Energy 2016, 173, 418–430. [Google Scholar] [CrossRef]
- Gómez, C.; Mészáros, E.; Jakab, E.; Velo, E.; Puigjaner, L. Thermogravimetry/mass spectrometry study of woody residues and an herbaceous biomass crop using PICA techniques. J. Anal. Appl. Pyrolysis 2007, 80, 416–426. [Google Scholar] [CrossRef]
- Zhang, J.; Zou, H.; Liu, J.; Evrendilek, F.; Xie, W.; He, Y.; Buyukada, M. Comparative (co-)pyrolytic performances and by-products of textile dyeing sludge and cattle manure: Deeper insights from Py-GC/MS, TG-FTIR, 2D-COS and PCA analyses. J. Hazard. Mater. 2021, 401, 123276. [Google Scholar] [CrossRef]
- Mishra, R.K.; Sahoo, A.; Mohanty, K. Pyrolysis kinetics and synergistic effect in co-pyrolysis of Samanea saman seeds and polyethylene terephthalate using thermogravimetric analyser. Bioresour. Technol. 2019, 289, 121608. [Google Scholar] [CrossRef] [PubMed]
- Osman, A.I.; Farrell, C.; Al-Muhtaseb, A.H.; Al-Fatesh, A.S.; Harrison, J.; Rooney, D.W. Pyrolysis kinetic modelling of abundant plastic waste (PET) and in-situ emission monitoring. Environ. Sci. Eur. 2020, 32, 112. [Google Scholar] [CrossRef]
- Fu, J.; Wu, X.; Liu, J.; Evrendilek, F.; Chen, T.; Xie, W.; Xu, W.; He, Y. Co-circularity of spent coffee grounds and polyethylene via co-pyrolysis: Characteristics, kinetics, and products. Fuel 2022, 337, 127061. [Google Scholar] [CrossRef]
- Hussein, Z.A.; Shakor, Z.M.; Alzuhairi, M.; Al-Sheikh, F. Kinetic and Thermodynamic Study of the Pyrolysis of Plastic Waste. Environ. Process. Int. J. 2023, 10, 27. [Google Scholar] [CrossRef]
- Hu, M.; Chen, Z.; Wang, S.; Guo, D.; Ma, C.; Zhou, Y.; Chen, J.; Laghari, M.; Fazal, S.; Xiao, B.; et al. Thermogravimetric kinetics of lignocellulosic biomass slow pyrolysis using distributed activation energy model, Fraser–Suzuki deconvolution, and iso-conversional method. Energy Convers. Manag. 2016, 118, 1–11. [Google Scholar] [CrossRef]
- Habibi, R.; Kopyscinski, J.; Masnadi, M.S.; Lam, J.; Grace, J.R.; Mims, C.A.; Hill, J.M. Co-gasification of Biomass and Non-biomass Feedstocks: Synergistic and Inhibition Effects of Switchgrass Mixed with Sub-bituminous Coal and Fluid Coke During CO2 Gasification. Energy Fuels 2012, 27, 494–500. [Google Scholar] [CrossRef]
- Kim, J.; Heo, E.; Kim, S.-J.; Kim, J.-Y. Investigation of the mineral components of porcelain raw material and their phase evolution during a firing process by using a Rietveld quantitative analysis. J. Korean Phys. Soc. 2016, 68, 126–130. [Google Scholar] [CrossRef]
- Ding, Z.; Liu, J.; Chen, H.; Huang, S.; Evrendilek, F.; He, Y.; Zheng, L. Co-pyrolysis performances, synergistic mechanisms, and products of textile dyeing sludge and medical plastic wastes. Sci. Total Environ. 2021, 799, 149397. [Google Scholar] [CrossRef]
- Liu, Y.; Ran, C.; Siyal, A.A.; Song, Y.; Jiang, Z.; Dai, J.; Chtaeva, P.; Fu, J.; Ao, W.; Deng, Z.; et al. Comparative study for fluidized bed pyrolysis of textile dyeing sludge and municipal sewage sludge. J. Hazard. Mater. 2020, 396, 122619. [Google Scholar] [CrossRef]
- Kleinhans, U.; Wieland, C.; Frandsen, F.J.; Spliethoff, H. Ash formation and deposition in coal and biomass fired combustion systems: Progress and challenges in the field of ash particle sticking and rebound behavior. Prog. Energy Combust. Sci. 2018, 68, 65–168. [Google Scholar] [CrossRef]
- Gu, L.; Dong, G.; Yu, H.; Zhang, K.; Lu, X.; Wen, H.; Zou, T. Preparation of porous biochar by urine assisted pyrolysis of sewage sludge and their application for Eriochrome Black T adsorption. J. Anal. Appl. Pyrolysis 2021, 153, 104975. [Google Scholar] [CrossRef]
- Chen, L.; Wang, S.; Meng, H.; Wu, Z.; Zhao, J. Synergistic effect on thermal behavior and char morphology analysis during co-pyrolysis of paulownia wood blended with different plastics waste. Appl. Therm. Eng. 2017, 111, 834–846. [Google Scholar] [CrossRef]
- Zhang, D.; Cao, C.-Y.; Lu, S.; Cheng, Y.; Zhang, H.-P. Experimental insight into catalytic mechanism of transition metal oxide nanoparticles on combustion of 5-Amino-1H-Tetrazole energetic propellant by multi kinetics methods and TG-FTIR-MS analysis. Fuel 2019, 245, 78–88. [Google Scholar] [CrossRef]
- Das, P.; Tiwari, P. Valorization of packaging plastic waste by slow pyrolysis. Resour. Conserv. Recycl. 2018, 128, 69–77. [Google Scholar] [CrossRef]














| Reaction Mechanism | Code | f(α) | G(α) |
|---|---|---|---|
| First-order (n = 1) | F1 | 1 − α | −ln(1 − α) |
| Three-halves order (n = 1.5) | F1.5 | (1 − α)3/2 | 2[(1 − α)−1/2 − 1] |
| Second-order (n = 2) | F2 | (1 − α)2 | (1 − α)−1 − 1 |
| Third-order (n = 3) | F3 | (1 − α)3 | [(1 − α)−2 − 1]/2 |
| Multi-order reaction | Fn | (1 − α)n | [(1 − α)1−n−1]/(n−1) |
| One-dimensional diffusion | D1 | 1/2α | α2 |
| Two-dimensional diffusion | D2 | [−ln(1 − α)]−1 | (1 − α) ln(1 − α) + α |
| Three-dimensional diffusion | D3 | (3/2) (1 − α)2/3[1 − (1 − α)1/3]−1 | [1 − (1 − α)1/3]2 |
| Four-dimensional diffusion | D4 | (3/2) [(1 − α)−1/3 − 1]−1 | (1 − 2α/3) − (1 − α)2/3 |
| Power law | P2 | 2α1/2 | α1/2 |
| Power law | P3 | 3α2/3 | α1/3 |
| Index law | P4 | 4α3/4 | α1/4 |
| One dimensional | R1 | 1 | α |
| Two dimensional | R2 | 2(1 − α)1/2 | 1 − (1 − α)1/2 |
| Three dimensional | R3 | 3(1 − α)2/3 | 1 − (1 − α)1/3 |
| Nucleation growth (n = 1.5) | A1.5 | 3/2(1 − α) [−ln(1 − α)]1/3 | [−ln(1 − α)]2/3 |
| Nucleation growth (n = 2) | A2 | 2(1 − α) [−ln(1 − α)]1/2 | [−ln(1 − α)]1/2 |
| Nucleation growth (n = 3) | A3 | 3(1 − α) [−ln(1 − α)]2/3 | [−ln(1 − α)]1/3 |
| Multi-dimensions | An | n(1 − α) [−ln(1 − α)](n−1)/n | [−ln(1 − α)]1/n |
| Sample | Proximate Analysis (wt.%) | Ultimate Analysis (wt.%) | LHV (MJ/kg) | HHV (MJ/kg) | Reference | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| M | A | V | FC | C | H | N | O | S | ||||
| MS | 4.37 | 55.21 | 37.87 | 2.55 | 20.62 | 3.78 | 3.53 | 12.06 | 0.43 | 7.73 | 8.69 | -- |
| HDPE | 0.05 | 0.06 | 99.89 | 0 | 86.14 | 14.18 | nd | 0.32 | nd | 43.14 | 46.33 | -- |
| PET | 0.23 | 0.05 | 95.68 | 4.04 | 62.63 | 4.01 | nd | 33.36 | nd | 21.98 | 22.89 | -- |
| PP | 0.11 | 17.56 | 75.43 | 6.9 | 66.35 | 10.48 | 0 | 5.5 | 0.09 | 30.70 | 33.06 | [1] |
| TDS | 5.02 | 60.26 | 34.72 | 0 | 16.23 | 2.51 | 0.96 | 11.82 | 3.21 | 3.42 | 4.11 | [37] |
| Sample | Stage I | Stage II | Stage III | Residue (%) | |||
|---|---|---|---|---|---|---|---|
| Temperature Range (°C) | Weight Loss (%) | Temperature Range (°C) | Weight Loss (%) | Temperature Range (°C) | Weight Loss (%) | ||
| MS | 33.00–163.47 | 4.30 | 163.47–736.37 | 35.56 | 736.37–1000.00 | 6.58 | 53.56 |
| HDPE | 33.00–385.38 | 0.22 | 385.38–524.10 | 95.80 | 524.10–1000.00 | 0.01 | 3.97 |
| PET | 33.00–367.48 | 0.42 | 367.48–545.65 | 87.39 | 545.65–1000.00 | 2.64 | 9.55 |
| β | Ti | Tmax | Rmax | Rmean | mf | ΔT1/2 | CPI | |
|---|---|---|---|---|---|---|---|---|
| °C/min | (°C) | (°C) | (%/min) | (%/min) | (%) | (°C) | [10−6·%3/(min2·°C3)] | |
| MS | 10 | 243.90 | 288.36 | −1.60 | −0.49 | 52.59 | 232.50 | 2.27 |
| 20 | 248.40 | 296.51 | −3.04 | −0.96 | 53.56 | 239.63 | 7.68 | |
| 30 | 282.10 | 303.28 | −4.44 | −1.33 | 56.96 | 246.32 | 12.06 | |
| SH7030 | 10 | 245.50 | 480.31 | −10.00 | −0.65 | 36.80 | 460.09 | 7.57 |
| 20 | 254.40 | 491.54 | −17.09 | −1.21 | 41.77 | 472.93 | 20.37 | |
| 30 | 259.30 | 499.51 | −25.06 | −1.72 | 44.72 | 480.29 | 38.30 |
| Sample | Ti | Tmax | Rmax | Rmean | mf | ΔT1/2 | CPI |
|---|---|---|---|---|---|---|---|
| (°C) | (°C) | (%/min) | (%/min) | (%) | (°C) | [10−6·%3/(min2·°C3)] | |
| MS | 248.40 | 296.51 | −3.04 | −0.96 | 53.56 | 239.63 | 7.68 |
| SH8515 | 252.80 | 488.62 | −9.98 | −1.07 | 48.14 | 466.75 | 9.61 |
| SH7030 | 254.40 | 491.54 | −17.09 | −1.21 | 41.77 | 472.93 | 20.37 |
| HDPE | 484.70 | 491.98 | −72.50 | −2.00 | 3.97 | 482.55 | 121.01 |
| SP8515 | 248.20 | 407.46 | −4.43 | −1.02 | 50.60 | 274.64 | 8.03 |
| SP7030 | 254.90 | 411.57 | −8.13 | −1.14 | 45.03 | 381.15 | 12.74 |
| PET | 428.00 | 456.49 | −40.46 | −1.87 | 9.55 | 432.62 | 80.97 |
| Sample | α | FWO | KAS | Starink | |||
|---|---|---|---|---|---|---|---|
| Eα | R2 | Eα | R2 | Eα | R2 | ||
| PET | 0.10 | 187.79 | 0.9994 | 186.06 | 0.9993 | 185.04 | 0.9993 |
| 0.15 | 192.37 | 0.9991 | 190.78 | 0.9989 | 189.72 | 0.9990 | |
| 0.20 | 193.76 | 0.9987 | 192.17 | 0.9985 | 191.11 | 0.9985 | |
| 0.25 | 194.09 | 0.9980 | 192.45 | 0.9978 | 191.39 | 0.9978 | |
| 0.30 | 195.27 | 0.9979 | 193.64 | 0.9976 | 192.57 | 0.9976 | |
| 0.35 | 197.06 | 0.9975 | 195.47 | 0.9973 | 194.39 | 0.9973 | |
| 0.40 | 197.87 | 0.9973 | 196.28 | 0.9970 | 195.20 | 0.9971 | |
| 0.45 | 201.23 | 0.9960 | 199.78 | 0.9955 | 198.66 | 0.9955 | |
| 0.50 | 202.69 | 0.9944 | 201.28 | 0.9938 | 200.16 | 0.9938 | |
| 0.55 | 203.79 | 0.9933 | 202.40 | 0.9925 | 201.27 | 0.9925 | |
| 0.60 | 204.02 | 0.9932 | 202.61 | 0.9924 | 201.48 | 0.9924 | |
| 0.65 | 203.35 | 0.9932 | 201.87 | 0.9924 | 200.75 | 0.9924 | |
| 0.70 | 203.91 | 0.9939 | 202.42 | 0.9932 | 201.29 | 0.9932 | |
| 0.75 | 204.25 | 0.9940 | 202.75 | 0.9933 | 201.62 | 0.9933 | |
| 0.80 | 204.41 | 0.9942 | 202.87 | 0.9936 | 201.74 | 0.9936 | |
| 0.85 | 205.08 | 0.9948 | 203.53 | 0.9942 | 202.40 | 0.9942 | |
| 0.90 | 206.84 | 0.9960 | 205.33 | 0.9955 | 204.18 | 0.9955 | |
| Mean | 199.87 | 198.34 | 197.23 | ||||
| HDPE | 0.10 | 217.96 | 0.9941 | 216.96 | 0.9934 | 217.28 | 0.9934 |
| 0.15 | 227.41 | 0.9956 | 226.82 | 0.9951 | 227.13 | 0.9951 | |
| 0.20 | 233.32 | 0.9967 | 232.97 | 0.9964 | 233.29 | 0.9964 | |
| 0.25 | 247.66 | 0.9995 | 248.02 | 0.9994 | 248.32 | 0.9994 | |
| 0.30 | 252.22 | 0.9998 | 252.78 | 0.9998 | 253.08 | 0.9998 | |
| 0.35 | 257.03 | 1.0000 | 257.80 | 1.0000 | 258.10 | 1.0000 | |
| 0.40 | 263.46 | 0.9998 | 264.55 | 0.9998 | 264.84 | 0.9998 | |
| 0.45 | 267.61 | 0.9999 | 268.89 | 0.9999 | 269.18 | 0.9999 | |
| 0.50 | 267.86 | 0.9999 | 269.12 | 0.9999 | 269.41 | 0.9999 | |
| 0.55 | 269.59 | 1.0000 | 270.92 | 1.0000 | 271.21 | 1.0000 | |
| 0.60 | 273.87 | 1.0000 | 275.40 | 1.0000 | 275.69 | 1.0000 | |
| 0.65 | 271.94 | 0.9990 | 273.34 | 0.9989 | 273.63 | 0.9989 | |
| 0.70 | 271.11 | 0.9993 | 272.45 | 0.9992 | 272.74 | 0.9992 | |
| 0.75 | 270.90 | 0.9994 | 272.21 | 0.9993 | 272.50 | 0.9993 | |
| 0.80 | 269.40 | 0.9994 | 270.60 | 0.9993 | 270.90 | 0.9994 | |
| 0.85 | 270.15 | 0.9995 | 271.37 | 0.9995 | 271.66 | 0.9995 | |
| 0.90 | 269.76 | 0.9996 | 270.92 | 0.9996 | 271.22 | 0.9996 | |
| Mean | 258.90 | 259.71 | 260.01 |
| Sample | Stage | Temp Range (°C) | Weight Loss (%) | Model | f(α) | R2 | A (s−1) | Eα (kJ/mol) |
|---|---|---|---|---|---|---|---|---|
| SH7030 | I | 164.2–409.7 | 16.62 | F1 | 1 − α | 0.9991 | 5.2 × 104 | 43.85 |
| II | 409.7–733.8 | 35.22 | A2 | 2(1 − α) [−ln(1 − α)]1/2 | 0.9941 | 8.8 × 109 | 128.35 | |
| SH8515 | I | 174.6–411.3 | 19.53 | F1 | 1 − α | 0.9979 | 7.1 × 104 | 45.26 |
| II | 411.3–692.1 | 23.87 | A2 | 2(1 − α) [−ln(1 − α)]1/2 | 0.9771 | 3.2 × 107 | 94.46 | |
| MS | I | 162.6–431.5 | 27.27 | F1 | 1 − α | 0.9966 | 4.9 × 103 | 33.46 |
| II | 431.5–735.8 | 9.62 | F6 | [(1 − α)−5 − 1]/5 | 0.9944 | 2.3 × 1020 | 267.22 | |
| SP8515 | I | 162.3–352.7 | 15.69 | F1 | 1 − α | 0.9935 | 3.2 × 105 | 49.54 |
| II | 352.7–734.2 | 25.83 | F6 | [(1 − α)−5 − 1]/5 | 0.9970 | 7.8 × 1022 | 275.04 | |
| SP7030 | I | 177.4–352.5 | 12.66 | F1 | 1 − α | 0.9980 | 1.1 × 106 | 55.27 |
| II | 352.5–702.1 | 34.78 | F6 | [(1 − α)−5 − 1]/5 | 0.9988 | 1.0 × 1029 | 352.75 |
| Sample | MS | MS-Char | SH8515-Char | SH7030-Char | SP8515-Char | SP7030-Char |
|---|---|---|---|---|---|---|
| O | 44.93 | 37.569 | 38.039 | 38.713 | 36.532 | 41.550 |
| Si | 18.625 | 21.792 | 21.542 | 21.044 | 20.046 | 20.066 |
| Al | 12.649 | 15.221 | 15.223 | 15.250 | 14.400 | 13.983 |
| Fe | 9.503 | 10.329 | 10.289 | 9.971 | 13.739 | 10.604 |
| P | 5.562 | 6.165 | 6.139 | 6.127 | 5.950 | 5.558 |
| Ca | 2.576 | 2.758 | 2.717 | 2.719 | 3.080 | 2.733 |
| K | 2.008 | 2.282 | 2.209 | 2.163 | 2.409 | 2.139 |
| S | 1.743 | 1.046 | 1.052 | 1.004 | 0.840 | 0.791 |
| Mg | 0.748 | 0.909 | 0.910 | 0.882 | 0.844 | 0.844 |
| Ti | 0.609 | 0.692 | 0.690 | 0.646 | 0.722 | 0.655 |
| Cl | 0.239 | 0.175 | 0.163 | 0.153 | 0.116 | 0.099 |
| Na | 0.216 | 0.262 | 0.281 | 0.266 | 0.275 | 0.243 |
| Mn | 0.165 | 0.185 | 0.185 | 0.180 | 0.257 | 0.192 |
| Zn | 0.126 | 0.143 | 0.135 | 0.139 | 0.206 | 0.149 |
| F | nd | 0.117 | 0.107 | 0.114 | 0.097 | 0.085 |
| Training | 5-Fold Cross-Validation | |||
|---|---|---|---|---|
| Measures | Mass (%) | DTG (%/min) | Mass (%) | DTG (%/min) |
| R2 (%) | 99.99 | 94.77 | 99.99 | 94.70 |
| RMSE | 0.393 | 0.626 | 0.395 | 0.626 |
| MAD | 0.085 | 0.272 | 0.084 | 0.272 |
| N | 811,437 | 811,437 | 202,859 | 202,859 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Liu, J.; Chen, Z.; Liang, F.; Lin, Z.; Tao, L.; Evrendilek, F.; He, Y.; Xie, Y.; Li, W.; Yang, C. Unraveling Co-Pyrolysis Mechanisms for Municipal Sludge and Microplastics: Thermodynamic, Kinetic, and Product Insights. Processes 2026, 14, 591. https://doi.org/10.3390/pr14040591
Liu J, Chen Z, Liang F, Lin Z, Tao L, Evrendilek F, He Y, Xie Y, Li W, Yang C. Unraveling Co-Pyrolysis Mechanisms for Municipal Sludge and Microplastics: Thermodynamic, Kinetic, and Product Insights. Processes. 2026; 14(4):591. https://doi.org/10.3390/pr14040591
Chicago/Turabian StyleLiu, Jingyong, Zhibin Chen, Fanjing Liang, Ziting Lin, Leyao Tao, Fatih Evrendilek, Yao He, Yuan Xie, Weixin Li, and Chunxiao Yang. 2026. "Unraveling Co-Pyrolysis Mechanisms for Municipal Sludge and Microplastics: Thermodynamic, Kinetic, and Product Insights" Processes 14, no. 4: 591. https://doi.org/10.3390/pr14040591
APA StyleLiu, J., Chen, Z., Liang, F., Lin, Z., Tao, L., Evrendilek, F., He, Y., Xie, Y., Li, W., & Yang, C. (2026). Unraveling Co-Pyrolysis Mechanisms for Municipal Sludge and Microplastics: Thermodynamic, Kinetic, and Product Insights. Processes, 14(4), 591. https://doi.org/10.3390/pr14040591









