Applications of Mesoporous Silica Nanoparticles in Oil & Gas and Biomedical Engineering
Abstract
1. Introduction
2. MSNs Synthesis, Structure, and Functionalization
2.1. Synthetic Strategies
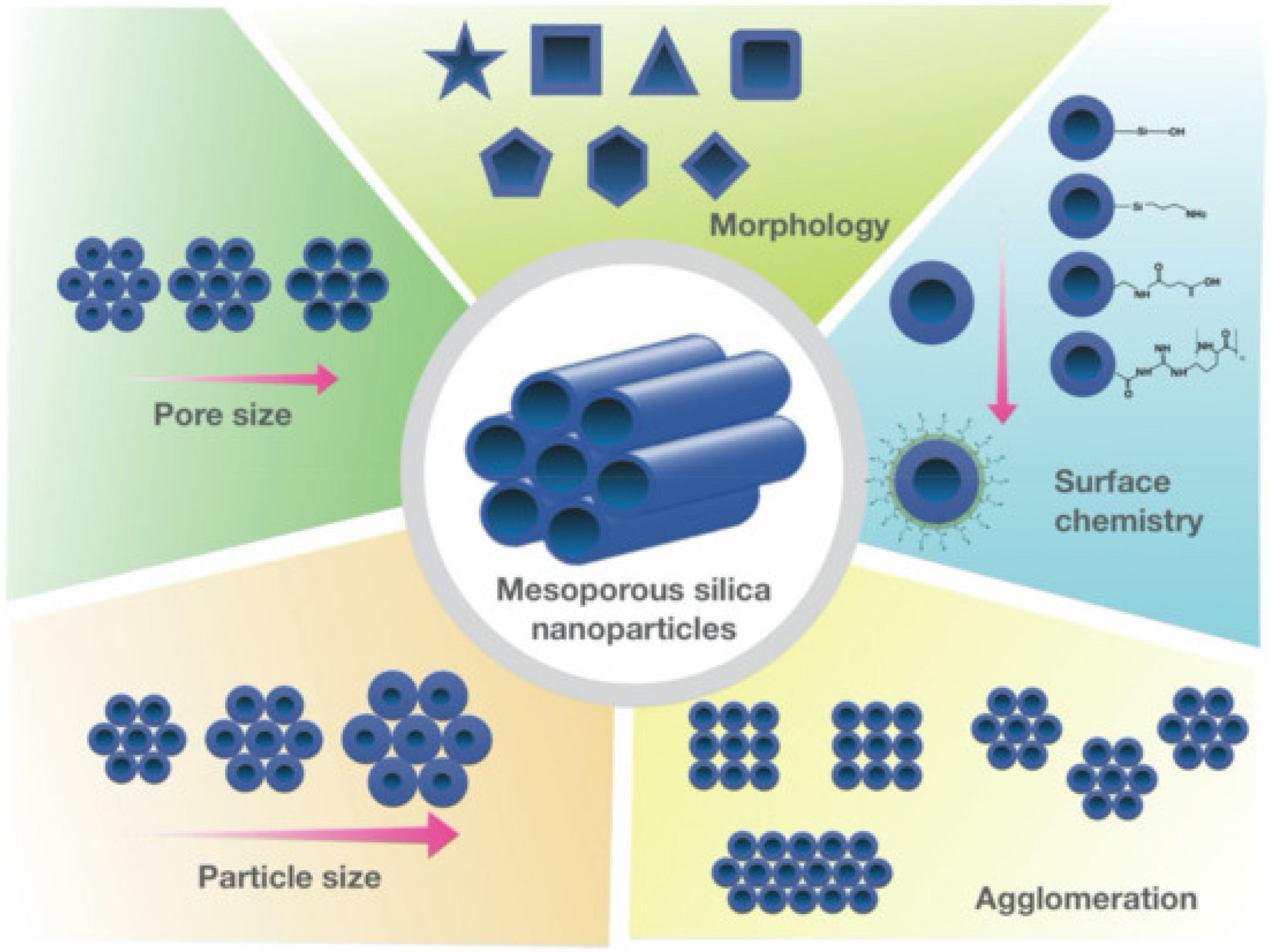
2.2. Structural and Physicochemical Tailoring
2.3. Surface Functionalization and Polymer Integration
2.4. Loading, Encapsulation, and Release Control
3. Intelligent and Stimuli-Responsive Systems
3.1. pH-Responsive Platforms
3.2. Redox- and Enzyme-Responsive Nanocarriers
3.3. Temperature and Light-Activated Systems
3.4. On-Command Gating and Smart Nanocontainers
4. Applications in Oil and Gas Engineering
4.1. Enhanced Oil Recovery (EOR)
4.2. Gas Capture, Separation, and Catalytic Conversion
4.3. Corrosion Protection and Inhibitor Delivery
4.4. Drilling Fluid and Wellbore Stability Additives
5. Biomedical Applications: Drug Delivery and Theranostics
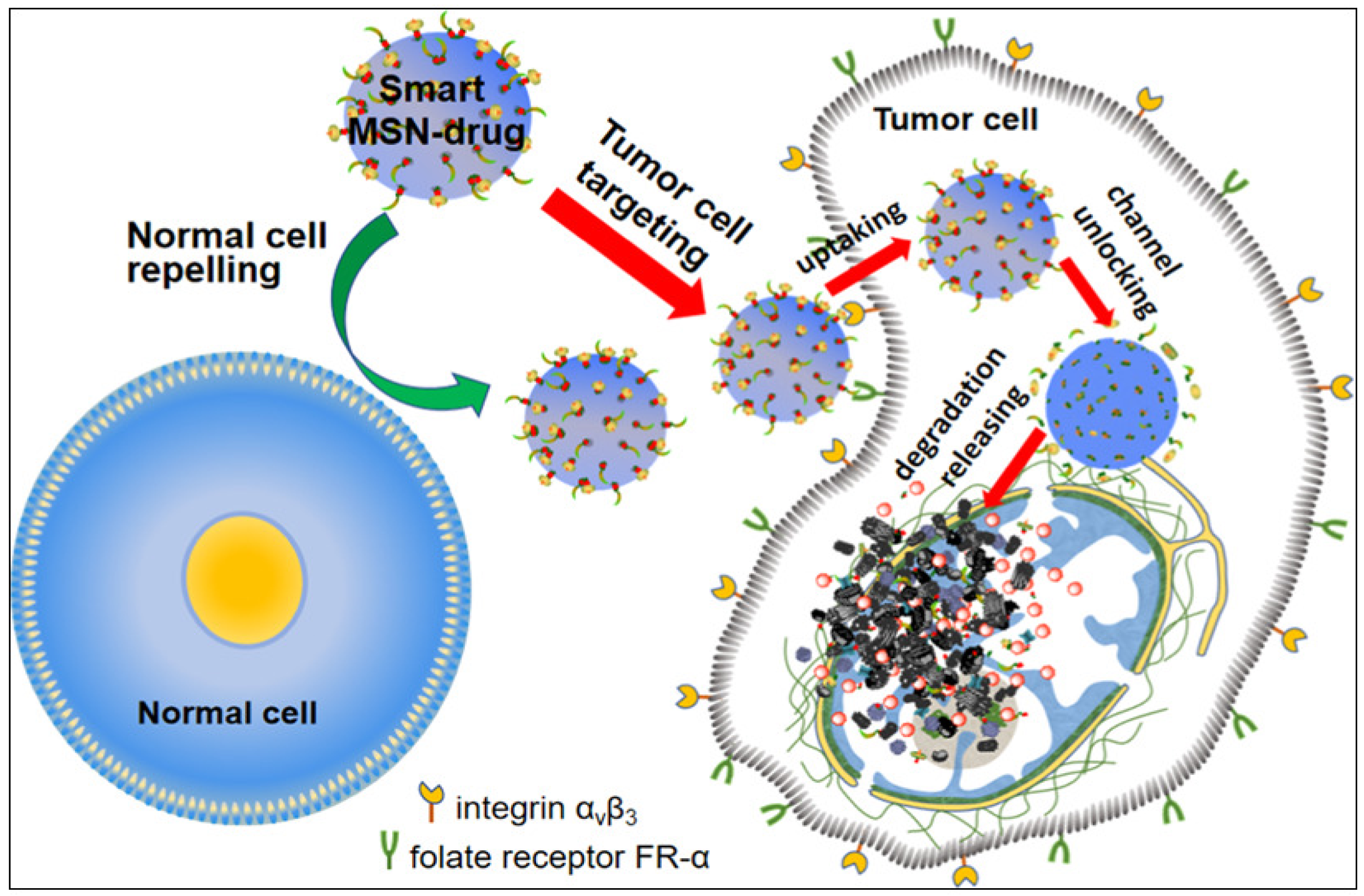
5.1. Targeted and Controlled Drug Delivery
5.2. Multifunctional Theranostic Platforms
5.3. Combination Therapy and Immune Modulation
5.4. MSN-Polymer Hybrid Delivery Systems
6. Tissue Engineering and Regenerative Medicine
6.1. Bone Regeneration and Mineralization Enhancement
6.2. Cartilage Repair and Growth Factor Release
6.3. Wound Healing and Soft-Tissue Repair
6.4. Stem Cell Preservation and Regeneration Support
7. Challenges, Safety Considerations, and Future Directions
7.1. Safety and Toxicology
7.2. Scale-Up and Manufacturing
7.3. Regulatory and Standardization Gaps
7.4. Cross-Domain Collaboration
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Hoffmann, F.; Cornelius, M.; Morell, J.; Fröba, M. Silica-Based mesoporous Organic–Inorganic hybrid materials. Angew. Chem. Int. Ed. 2006, 45, 3216–3251. [Google Scholar] [CrossRef]
- Wu, S.; Mou, C.; Lin, H. Synthesis of mesoporous silica nanoparticles. Chem. Soc. Rev. 2013, 42, 3862. [Google Scholar] [CrossRef]
- Narayan, R.; Nayak, U.Y.; Raichur, A.M.; Garg, S. Mesoporous silica nanoparticles: A Comprehensive review on synthesis and recent advances. Pharmaceutics 2018, 10, 118. [Google Scholar] [CrossRef] [PubMed]
- Rastegari, E.; Hsiao, Y.; Lai, W.; Lai, Y.; Yang, T.; Chen, S.; Huang, P.; Chiou, S.; Mou, C.; Chien, Y. An update on mesoporous silica nanoparticle applications in nanomedicine. Pharmaceutics 2021, 13, 1067. [Google Scholar] [CrossRef]
- Nair, A.H.R.C.; Day, C.M.; Garg, S.; Nayak, Y.; Shenoy, P.A.; Nayak, U.Y. Polymeric functionalization of mesoporous silica nanoparticles: Biomedical insights. Int. J. Pharm. 2024, 660, 124314. [Google Scholar] [CrossRef]
- Salve, R.; Kumar, P.; Ngamcherdtrakul, W.; Gajbhiye, V.; Yantasee, W. Stimuli-responsive mesoporous silica nanoparticles: A custom-tailored next generation approach in cargo delivery. Mater. Sci. Eng. C 2021, 124, 112084. [Google Scholar] [CrossRef]
- Olivieri, F.; Castaldo, R.; Cocca, M.; Gentile, G.; Lavorgna, M. Mesoporous silica nanoparticles as carriers of active agents for smart anticorrosive organic coatings: A critical review. Nanoscale 2021, 13, 9091–9111. [Google Scholar] [CrossRef]
- Fakoya, M.F.; Shah, S.N. Emergence of nanotechnology in the oil and gas industry: Emphasis on the application of silica nanoparticles. Petroleum 2017, 3, 391–405. [Google Scholar] [CrossRef]
- Xu, B.; Li, S.; Shi, R.; Liu, H. Multifunctional mesoporous silica nanoparticles for biomedical applications. Signal Transduct. Target. Ther. 2023, 8, 435. [Google Scholar] [CrossRef] [PubMed]
- Shan, Y.; Cao, L.; Xu, C.; Zhao, P.; Cao, C.; Li, F.; Xu, B.; Huang, Q. Sulfonate-Functionalized Mesoporous Silica Nanoparticles as Carriers for Controlled Herbicide Diquat Dibromide Release through Electrostatic Interaction. Int. J. Mol. Sci. 2019, 20, 1330. [Google Scholar] [CrossRef]
- Khalil, M.; Amanda, A.; Yunarti, R.T.; Jan, B.M.; Irawan, S. Synthesis and application of mesoporous silica nanoparticles as gas migration control additive in oil and gas cement. J. Pet. Sci. Eng. 2020, 195, 107660. [Google Scholar] [CrossRef]
- Li, X.; Wang, X.; Qian, G.; Ito, A. Synergistical chemotherapy and cancer immunotherapy using dual drug-delivering and immunopotentiating mesoporous silica. Appl. Mater. Today 2019, 16, 102–111. [Google Scholar] [CrossRef]
- Lee, J.H.; Park, J.; Eltohamy, M.; Perez, R.; Lee, E.; Kim, H. Collagen gel combined with mesoporous nanoparticles loading nerve growth factor as a feasible therapeutic three-dimensional depot for neural tissue engineering. RSC Adv. 2013, 3, 24202. [Google Scholar] [CrossRef]
- Shadjou, N.; Hasanzadeh, M. Bone tissue engineering using silica-based mesoporous nanobiomaterials: Recent progress. Mater. Sci. Eng. C 2015, 55, 401–409. [Google Scholar] [CrossRef] [PubMed]
- Gisbert-Garzarán, M.; Manzano, M.; Vallet-Regí, M. Mesoporous silica nanoparticles for the treatment of complex bone diseases: Bone cancer, bone infection and osteoporosis. Pharmaceutics 2020, 12, 83. [Google Scholar] [CrossRef]
- Santhamoorthy, M.; Asaithambi, P.; Ramkumar, V.; Elangovan, N.; Perumal, I.; Kim, S.C. A review on the recent advancements of Polymer-Modified mesoporous silica nanoparticles for drug delivery under Stimuli-Trigger. Polymers 2025, 17, 1640. [Google Scholar] [CrossRef]
- Cristoforetti, A.; Rossi, S.; Deflorian, F.; Fedel, M. On the Limits of the EIS Low-Frequency Impedance Modulus as a Tool to Describe the Protection Properties of Organic Coatings Exposed to Accelerated Aging Tests. Coatings 2023, 13, 598. [Google Scholar] [CrossRef]
- Deng, Y.; Zhang, X.; Yang, X.; Huang, Z.; Wei, X.; Yang, X.; Liao, W. Subacute toxicity of mesoporous silica nanoparticles to the intestinal tract and the underlying mechanism. J. Hazard. Mater. 2020, 409, 124502. [Google Scholar] [CrossRef]
- Porrang, S.; Davaran, S.; Rahemi, N.; Allahyari, S.; Mostafavi, E. How Advancing are Mesoporous Silica Nanoparticles? A Comprehensive Review of the Literature. Int. J. Nanomed. 2022, 17, 1803–1827. [Google Scholar] [CrossRef]
- Zhe, Z.; Yuxiu, A. Nanotechnology for the oil and gas industry—An overview of recent progress. Nanotechnol. Rev. 2018, 7, 341–353. [Google Scholar] [CrossRef]
- Fatima, R.; Katiyar, P.; Kushwaha, K. Recent advances in mesoporous silica nanoparticle: Synthesis, drug loading, release mechanisms, and diverse applications. Front. Nanotechnol. 2025, 7, 1564188. [Google Scholar] [CrossRef]
- Aziz, M.; Jalil, A.; Triwahyono, S.; Sidik, S. Methanation of carbon dioxide on metal-promoted mesostructured silica nanoparticles. Appl. Catal. A Gen. 2014, 486, 115–122. [Google Scholar] [CrossRef]
- Wang, J.; Wang, G.; Zhang, Z.; Ouyang, G.; Hao, Z. Effects of mesoporous silica particle size and pore structure on the performance of polymer-mesoporous silica mixed matrix membranes. RSC Adv. 2021, 11, 36577–36586. [Google Scholar] [CrossRef]
- Kovtareva, S.; Kusepova, L.; Tazhkenova, G.; Mashan, T.; Bazarbaeva, K.; Kopishev, E. Surface modification of mesoporous silica nanoparticles for application in targeted delivery systems of antitumour drugs. Polymers 2024, 16, 1105. [Google Scholar] [CrossRef]
- Ma, Y.; Huang, H.; Zhou, H.; Graham, M.; Smith, J.; Sheng, X.; Chen, Y.; Zhang, L.; Zhang, X.; Shchukina, E.; et al. Superior anti-corrosion and self-healing bi-functional polymer composite coatings with polydopamine modified mesoporous silica/graphene oxide. J. Mater. Sci. Technol. 2021, 95, 95–104. [Google Scholar] [CrossRef]
- Dumontel, B.; Conejo-Rodríguez, V.; Vallet-Regí, M.; Manzano, M. Natural biopolymers as smart coating materials of mesoporous silica nanoparticles for drug delivery. Pharmaceutics 2023, 15, 447. [Google Scholar] [CrossRef]
- Sun, J.; Chang, X.; Zhang, F.; Bai, Y.; Lv, K.; Wang, J.; Zhou, X.; Wang, B. Salt-Responsive zwitterionic polymer brush based on modified silica nanoparticles as a Fluid-Loss additive in Water-Based drilling fluids. Energy Fuels 2020, 34, 1669–1679. [Google Scholar] [CrossRef]
- Mohan, A.; Suresh, R.; Ashwini, M.; Periyasami, G.; Guganathan, L.; Lin, M.; Kumarasamy, K.; Kim, S.; Santhamoorthy, M. Alginate functionalized magnetic-silica composites for pH-responsive drug delivery and magnetic hyperthermia applications. Mater. Lett. 2024, 361, 136088. [Google Scholar] [CrossRef]
- Wang, G.; Dong, J.; Yuan, T.; Zhang, J.; Wang, L.; Wang, H. Visible light and pH responsive Polymer-Coated mesoporous silica nanohybrids for controlled release. Macromol. Biosci. 2016, 16, 990–994. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Lü, S.; Gao, C.; Wang, X.; Bai, X.; Duan, H.; Gao, N.; Feng, C.; Liu, M. Polymeric micelle-coated mesoporous silica nanoparticle for enhanced fluorescent imaging and pH-responsive drug delivery. Chem. Eng. J. 2015, 279, 851–860. [Google Scholar] [CrossRef]
- Hanafi-Bojd, M.Y.; Jaafari, M.R.; Ramezanian, N.; Xue, M.; Amin, M.; Shahtahmassebi, N.; Malaekeh-Nikouei, B. Surface functionalized mesoporous silica nanoparticles as an effective carrier for epirubicin delivery to cancer cells. Eur. J. Pharm. Biopharm. 2014, 89, 248–258. [Google Scholar] [CrossRef]
- Seljak, K.B.; Kocbek, P.; Gašperlin, M. Mesoporous silica nanoparticles as delivery carriers: An overview of drug loading techniques. J. Drug Deliv. Sci. Technol. 2020, 59, 101906. [Google Scholar] [CrossRef]
- Qu, Q.; Ma, X.; Zhao, Y. Targeted delivery of doxorubicin to mitochondria using mesoporous silica nanoparticle nanocarriers. Nanoscale 2015, 7, 16677–16686. [Google Scholar] [CrossRef] [PubMed]
- Racles, C.; Zaltariov, M.; Peptanariu, D.; Vasiliu, T.; Cazacu, M. Functionalized mesoporous silica as doxorubicin carriers and cytotoxicity boosters. Nanomaterials 2022, 12, 1823. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Su, H.; Peng, Q.; Cui, N.; Li, F.; Qu, W.; Cao, Y.; Feng, J.; Li, X.; Wang, Z.; et al. Cross-linked chitosan encapsulated hollow mesoporous silica nanoparticle: A dual-functional smart nanocapsule design for targeted corrosion inhibition and controlled emulsification. Carbohydr. Polym. 2024, 351, 123092. [Google Scholar] [CrossRef]
- Borisova, D.; Möhwald, H.; Shchukin, D.G. Mesoporous silica nanoparticles for active corrosion protection. ACS Nano 2011, 5, 1939–1946. [Google Scholar] [CrossRef]
- Chen, Q.; Chen, Y.; Zhang, W.; Huang, Q.; Hu, M.; Peng, D.; Peng, C.; Wang, L.; Chen, W. Acidity and glutathione Dual-Responsive Polydopamine-Coated Organic-Inorganic hybrid hollow mesoporous silica nanoparticles for controlled drug delivery. ChemMedChem 2020, 15, 1940–1946. [Google Scholar] [CrossRef] [PubMed]
- Dong, J.; Ma, Y.; Li, R.; Zhang, W.; Zhang, M.; Meng, F.; Ding, K.; Jiang, H.; Gong, Y. Smart MSN-Drug-Delivery system for tumor cell targeting and tumor microenvironment release. ACS Appl. Mater. Interfaces 2021, 13, 42522–42532. [Google Scholar] [CrossRef]
- Ahmadi, F.; Sodagar-Taleghani, A.; Ebrahimnejad, P.; Moghaddam, S.P.H.; Ebrahimnejad, F.; Asare-Addo, K.; Nokhodchi, A. A review on the latest developments of mesoporous silica nanoparticles as a promising platform for diagnosis and treatment of cancer. Int. J. Pharm. 2022, 625, 122099. [Google Scholar] [CrossRef]
- Aznar, E.; Oroval, M.; Pascual, L.; Murguía, J.R.; Martínez-Máñez, R.; Sancenón, F. Gated materials for On-Command release of guest molecules. Chem. Rev. 2016, 116, 561–718. [Google Scholar] [CrossRef]
- Yang, K.; Zhang, C.; Wang, W.; Wang, P.C.; Zhou, J.; Liang, X. pH-Responsive Mesoporous Silica Nanoparticles Employed in Controlled Drug Delivery Systems for Cancer Treatment. 2014. Available online: https://pmc.ncbi.nlm.nih.gov/articles/PMC3969802 (accessed on 10 January 2026).
- Lu, H.; Chen, A.; Zhang, X.; Wei, Z.; Cao, R.; Zhu, Y.; Lu, J.; Wang, Z.; Tian, L. A pH-responsive T1-T2 dual-modal MRI contrast agent for cancer imaging. Nat. Commun. 2022, 13, 7948. [Google Scholar] [CrossRef] [PubMed]
- De La Torre, C.; Coll, C.; Ultimo, A.; Sancenón, F.; Martínez-Máñez, R.; Ruiz-Hernández, E. In Situ-Forming Gels Loaded with Stimuli-Responsive Gated Mesoporous Silica Nanoparticles for Local Sustained Drug Delivery. Pharmaceutics 2023, 15, 1071. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Qiu, X.; Xia, T.; Li, Q.; Wen, Z.; Huang, B.; Li, Y. Mesoporous materials make hydrogels more powerful in biomedicine. Gels 2023, 9, 207. [Google Scholar] [CrossRef]
- Thirupathi, K.; Santhamoorthy, M.; Radhakrishnan, S.; Ulagesan, S.; Nam, T.; Phan, T.T.V.; Kim, S. Thermosensitive Polymer-Modified mesoporous silica for pH and Temperature-Responsive drug delivery. Pharmaceutics 2023, 15, 795. [Google Scholar] [CrossRef] [PubMed]
- Ren, S.; Zhou, Y.; Fan, R.; Peng, W.; Xu, X.; Li, L.; Xu, Y. Constructing biocompatible MSN@Ce@PEG nanoplatform for enhancing regenerative capability of stem cell via ROS-scavenging in periodontitis. Chem. Eng. J. 2021, 423, 130207. [Google Scholar] [CrossRef]
- Gerstenberg, M.; Stürzel, C.M.; Weil, T.; Kirchhoff, F.; Lindén, M. Modular Hydrogel−Mesoporous silica nanoparticle constructs for therapy and diagnostics. Adv. NanoBiomed Res. 2021, 2, 2100125. [Google Scholar] [CrossRef]
- Liu, Z.; Zhang, B.; Yu, H.; Zhang, Z.; Jiang, W.; Ma, Z. A smart anticorrosive epoxy coating based on graphene Oxide/Functional mesoporous silica nanoparticles for controlled release of corrosion inhibitors. Coatings 2022, 12, 1749. [Google Scholar] [CrossRef]
- Zhang, W.; Yang, Z.; Zhang, M.; He, J.; Li, S.; Sun, X.; Ni, P. A hybrid hydrogel constructed using drug loaded mesoporous silica and multiple response copolymer as an intelligent dressing for wound healing of diabetic foot ulcers. J. Mater. Chem. B 2023, 11, 4922–4933. [Google Scholar] [CrossRef]
- Mao, H.; Qiu, Z.; Shen, Z.; Huang, W. Hydrophobic associated polymer-based silica nanoparticles composite with core–shell structure as a filtrate reducer for drilling fluid at utra-high temperature. J. Pet. Sci. Eng. 2015, 129, 1–14. [Google Scholar] [CrossRef]
- Zeynali, M.E. Mechanical and physico-chemical aspects of wellbore stability during drilling operations. J. Pet. Sci. Eng. 2012, 82–83, 120–124. [Google Scholar] [CrossRef]
- Lamprakou, Z.; Bi, H.; Weinell, C.E.; Tortajada, S.; Dam-Johansen, K. Smart epoxy coating with mesoporous silica nanoparticles loaded with calcium phosphate for corrosion protection. Prog. Org. Coat. 2022, 165, 106740. [Google Scholar] [CrossRef]
- Joshi, D.; Maurya, N.K.; Kumar, N.; Mandal, A. Experimental investigation of silica nanoparticle assisted Surfactant and polymer systems for enhanced oil recovery. J. Pet. Sci. Eng. 2022, 216, 110791. [Google Scholar] [CrossRef]
- Kandiel, Y.E.; Attia, G.M.; Metwalli, F.I.; Khalaf, R.E.; Mahmoud, O. Nanoparticles in enhanced oil recovery: State-of-the-art review. J. Pet. Explor. Prod. Technol. 2025, 15, 66. [Google Scholar] [CrossRef]
- El-Masry, J.F.; Bou-Hamdan, K.F.; Abbas, A.H.; Martyushev, D.A. A comprehensive review on utilizing nanomaterials in enhanced oil recovery applications. Energies 2023, 16, 691. [Google Scholar] [CrossRef]
- Enhanced Oil Recovery. Available online: https://www.energy.gov/fecm/enhanced-oil-recovery (accessed on 10 January 2026).
- Deng, X.; Tariq, Z.; Murtaza, M.; Patil, S.; Mahmoud, M.; Kamal, M.S. Relative contribution of wettability Alteration and interfacial tension reduction in EOR: A critical review. J. Mol. Liq. 2020, 325, 115175. [Google Scholar] [CrossRef]
- Yang, Y.; Li, L.; Wang, X.; Fu, Y.; He, X.; Zhang, S.; Guo, J. A surfactant for enhanced heavy oil recovery in carbonate reservoirs in High-Salinity and High-Temperature conditions. Energies 2020, 13, 4525. [Google Scholar] [CrossRef]
- Isaac, O.T.; Pu, H.; Oni, B.A.; Samson, F.A. Surfactants employed in conventional and unconventional reservoirs for enhanced oil recovery—A review. Energy Rep. 2022, 8, 2806–2830. [Google Scholar] [CrossRef]
- Li, D.; Wang, Y.; Bai, B.; Xu, N.; Shi, W.; Zhang, Y.; Ding, W.; Ma, P.; Gao, Z. Reinforcing Polymer Flooding System with Dendritic Mesoporous Silica Nanoparticles for Improved Oil Recovery. Energy Fuels 2025, 39, 1889–1902. [Google Scholar] [CrossRef]
- Bae, J.Y.; Jang, S.G.; Cho, J.; Kang, M. Amine-Functionalized mesoporous silica for efficient CO2 capture: Stability, performance, and industrial feasibility. Int. J. Mol. Sci. 2025, 26, 4313. [Google Scholar] [CrossRef]
- Chen, L.; Ting, V.P.; Zhang, Y.; Deng, S.; Li, S.; Yin, Z.; Wang, F.; Wang, X. Modeling adsorption-based hydrogen storage in nanoporous activated carbon beds at moderate temperature and pressure. Int. J. Hydrog. Energy 2025, 122, 159–179. [Google Scholar] [CrossRef]
- Zhou, R.; Pan, Y.; Xing, W.; Xu, N. Advanced microporous membranes for H2/CH4 separation: Challenges and perspectives. Adv. Membr. 2021, 1, 100011. [Google Scholar] [CrossRef]
- Shen, T.; Mao, S.; Ding, F.; Han, T.; Gao, M. Selective adsorption of cationic/anionic tritoluene dyes on functionalized amorphous silica: A mechanistic correlation between the precursor, modifier and adsorbate. Colloids Surf. A Physicochem. Eng. Asp. 2021, 618, 126435. [Google Scholar] [CrossRef]
- Al-Shargabi, M.; Davoodi, S.; Wood, D.A.; Al-Musai, A.; Rukavishnikov, V.S.; Minaev, K.M. Nanoparticle applications as beneficial oil and gas drilling fluid additives: A review. J. Mol. Liq. 2022, 352, 118725. [Google Scholar] [CrossRef]
- Asad, M.S.; Jaafar, M.T.; Rashid, F.L.; Togun, H.; Rasheed, M.K.; Al-Obaidi, M.A.; Al-Amir, Q.R.; Mohammed, H.I.; Sarris, I.E. Sustainable Drilling Fluids: A Review of Nano-Additives for Improved Performance and Reduced Environmental impact. Processes 2024, 12, 2180. [Google Scholar] [CrossRef]
- Gokapai, V.; Pothana, P.; Ling, K. Nanoparticles in Drilling Fluids: A review of types, mechanisms, applications, and future prospects. Eng.—Adv. Eng. 2024, 5, 2462–2495. [Google Scholar] [CrossRef]
- Zarei, V.; Yavari, H.; Nasiri, A.; Mirzaasadi, M.; Davarpanah, A. Implementation of Amorphous Mesoporous Silica Nanoparticles to formulate a novel water-based drilling fluid. Arab. J. Chem. 2023, 16, 104818. [Google Scholar] [CrossRef]
- Liu, H.; Cui, S.; Meng, Y.; Li, Z.; Yu, X.; Sun, H.; Zhou, Y.; Luo, Y. Rock mechanics and wellbore stability of deep shale during drilling and completion processes. J. Pet. Sci. Eng. 2021, 205, 108882. [Google Scholar] [CrossRef]
- Saleh, T.A.; Nur, M.M.; Satria, M.; Al-Arfaj, A.A. Synthesis of novel hydrophobic nanocomposite-modified silica as efficient shale inhibitor in fuel industry. Surf. Interfaces 2023, 38, 102837. [Google Scholar] [CrossRef]
- Kelkar, S.S.; Reineke, T.M. Theranostics: Combining imaging and therapy. Bioconjugate Chem. 2011, 22, 1879–1903. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Hong, H.; Zhang, Y.; Valdovinos, H.F.; Shi, S.; Kwon, G.S.; Theuer, C.P.; Barnhart, T.E.; Cai, W. In Vivo Tumor Targeting and Image-Guided Drug Delivery with Antibody-Conjugated, Radiolabeled Mesoporous Silica Nanoparticles. ACS Nano 2013, 7, 9027–9039. [Google Scholar] [CrossRef]
- Fang, Z.; Li, X.; Xu, Z.; Du, F.; Wang, W.; Shi, R.; Gao, D. Hyaluronic acid-modified mesoporous silica-coated superparamagnetic Fe3O4 nanoparticles for targeted drug delivery. Int. J. Nanomed. 2019, 14, 5785–5797. [Google Scholar] [CrossRef]
- Dadashpour, M.; Mahmoudi, H.; Rahimi, Z.; Poodeh, R.J.; Mousazadeh, H.; Firouzi-Amandi, A.; Yazdani, Y.; Asl, A.N.; Akbarzadeh, A. Sustained in vitro delivery of metformin-loaded mesoporous silica nanoparticles for delayed senescence and stemness preservation of adipose-derived stem cells. J. Drug Deliv. Sci. Technol. 2023, 87, 104769. [Google Scholar] [CrossRef]
- Mashayekhi, S.; Rasoulpoor, S.; Shabani, S.; Esmaeilizadeh, N.; Serati-Nouri, H.; Sheervalilou, R.; Pilehvar-Soltanahmadi, Y. Curcumin-loaded mesoporous silica nanoparticles/nanofiber composites for supporting long-term proliferation and stemness preservation of adipose-derived stem cells. Int. J. Pharm. 2020, 587, 119656. [Google Scholar] [CrossRef]
- Lukin, I.; Erezuma, I.; Desimone, M.F.; Zhang, Y.S.; Dolatshahi-Pirouz, A.; Orive, G. Nanomaterial-based drug delivery of immunomodulatory factors for bone and cartilage tissue engineering. Biomater. Adv. 2023, 154, 213637. [Google Scholar] [CrossRef]
- Sun, L.; Liu, H.; Ye, Y.; Lei, Y.; Islam, R.; Tan, S.; Tong, R.; Miao, Y.B. Smart nanoparticles for cancer therapy. Sig. Transduct. Target. Ther. 2023, 8, 418. [Google Scholar] [CrossRef]
- Pablos, J.L.; Lozano, D.; Manzano, M.; Vallet-Regí, M. Regenerative medicine: Hydrogels and mesoporous silica nanoparticles. Mater. Today Bio 2024, 29, 101342. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zhou, X.; He, C. Mesoporous silica nanoparticles for tissue-engineering applications. Wiley Interdiscip. Rev. Nanomed. Nanobiotech. 2019, 11, e1573. [Google Scholar] [CrossRef] [PubMed]
- Heidari, R.; Assadollahi, V.; Manesh, M.H.S.; Mirzaei, S.A.; Elahian, F. Recent advances in mesoporous silica nanoparticles formulations and drug delivery for wound healing. Int. J. Pharm. 2024, 665, 124654. [Google Scholar] [CrossRef]
- Ghosh, S.; Webster, T.J. Mesoporous silica based nanostructures for bone tissue regeneration. Front. Mater. 2021, 8, 692309. [Google Scholar] [CrossRef]
- Eivazzadeh-Keihan, R.; Chenab, K.K.; Taheri-Ledari, R.; Mosafer, J.; Hashemi, S.M.; Mokhtarzadeh, A.; Maleki, A.; Hamblin, M.R. Recent advances in the application of mesoporous silica-based nanomaterials for bone tissue engineering. Mater. Sci. Eng. C 2019, 107, 110267. [Google Scholar] [CrossRef]
- Yousefiasl, S.; Manoochehri, H.; Makvandi, P.; Afshar, S.; Salahinejad, E.; Khosraviyan, P.; Saidijam, M.; Asl, S.S.; Sharifi, E. Chitosan/alginate bionanocomposites adorned with mesoporous silica nanoparticles for bone tissue engineering. J. Nanostruct. Chem. 2022, 13, 389–403. [Google Scholar] [CrossRef]
- Wang, Y.; Cui, W.; Zhao, X.; Wen, S.; Sun, Y.; Han, J.; Zhang, H. Bone remodeling-inspired dual delivery electrospun nanofibers for promoting bone regeneration. Nanoscale 2018, 11, 60–71. [Google Scholar] [CrossRef]
- Barati, D.; Gegg, C.; Yang, F. Nanoparticle-Mediated TGF-β Release from Microribbon-Based Hydrogels Accelerates Stem Cell-Based Cartilage Formation In Vivo. Ann. Biomed. Eng. 2020, 48, 1971–1981. [Google Scholar] [CrossRef] [PubMed]
- Airuddin, S.S.; Halim, A.S.; Sulaiman, W.A.W.; Kadir, R.; Nasir, N.A.M. Adipose-Derived Stem Cell: “Treat or Trick”. Biomedicines 2021, 9, 1624. [Google Scholar] [CrossRef]
- Niroumand, U.; Firouzabadi, N.; Goshtasbi, G.; Hassani, B.; Ghasemiyeh, P.; Mohammadi-Samani, S. The effect of size, morphology and surface properties of mesoporous silica nanoparticles on pharmacokinetic aspects and potential toxicity concerns. Front. Mater. 2023, 10, 1189463. [Google Scholar] [CrossRef]
- Li, J.; Sun, R.; Xu, H.; Wang, G. Integrative metabolomics, Proteomics and transcriptomics analysis reveals liver toxicity of mesoporous silica nanoparticles. Front. Pharmacol. 2022, 13, 835359. [Google Scholar] [CrossRef]
- Lérida-Viso, A.; Estepa-Fernández, A.; García-Fernández, A.; Martí-Centelles, V.; Martínez-Máñez, R. Biosafety of mesoporous silica nanoparticles; towards clinical translation. Adv. Drug Deliv. Rev. 2023, 201, 115049. [Google Scholar] [CrossRef]
- Bukara, K.; Schueller, L.; Rosier, J.; Martens, M.A.; Daems, T.; Verheyden, L.; Eelen, S.; Van Speybroeck, M.; Libanati, C.; Martens, J.A.; et al. Ordered mesoporous silica to enhance the bioavailability of poorly water-soluble drugs: Proof of concept in man. Eur. J. Pharm. Biopharm. 2016, 108, 220–225. [Google Scholar] [CrossRef] [PubMed]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Shah, R.; Lotwin, M.; Nitodas, S. Applications of Mesoporous Silica Nanoparticles in Oil & Gas and Biomedical Engineering. Processes 2026, 14, 592. https://doi.org/10.3390/pr14040592
Shah R, Lotwin M, Nitodas S. Applications of Mesoporous Silica Nanoparticles in Oil & Gas and Biomedical Engineering. Processes. 2026; 14(4):592. https://doi.org/10.3390/pr14040592
Chicago/Turabian StyleShah, Raj, Michael Lotwin, and Stefanos (Steve) Nitodas. 2026. "Applications of Mesoporous Silica Nanoparticles in Oil & Gas and Biomedical Engineering" Processes 14, no. 4: 592. https://doi.org/10.3390/pr14040592
APA StyleShah, R., Lotwin, M., & Nitodas, S. (2026). Applications of Mesoporous Silica Nanoparticles in Oil & Gas and Biomedical Engineering. Processes, 14(4), 592. https://doi.org/10.3390/pr14040592






