Traditional Herbs in Anatolian Medicine for Rosacea: A Basis for Non-Steroidal Magistral Therapy
Abstract
1. Introduction
2. Methodology
3. The Plants Effective in Rosacea Therapy
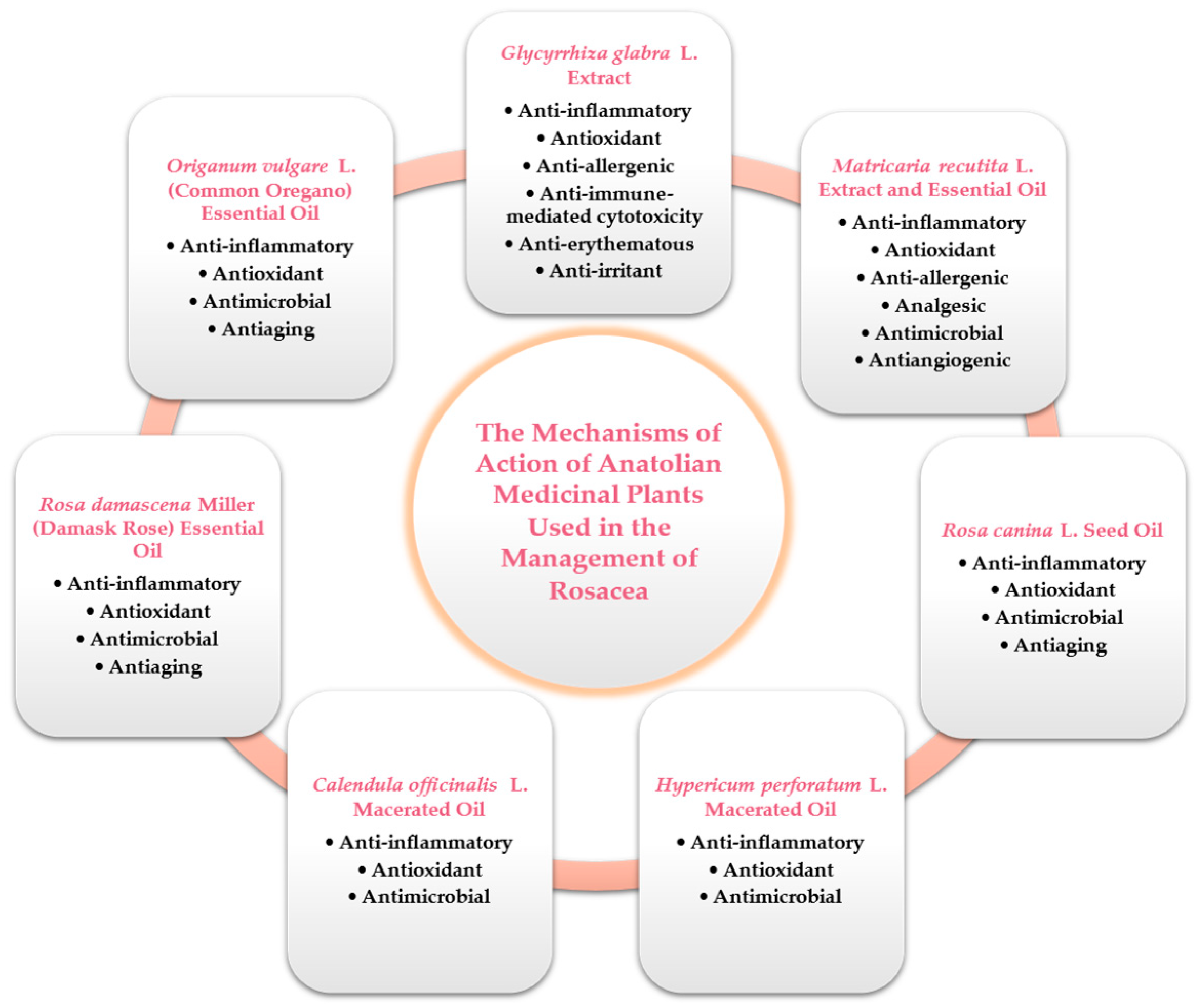
3.1. Glycyrrhiza glabra L. (Licorice) Extract
3.2. Matricaria recutita L. (Chamomile) Extract and Essential Oil
3.3. Rosa canina L. (Rosehip, Dog Rose) Seed Oil
3.4. Hypericum perforatum L. (St. John’s Wort) Macerated Oil
3.5. Calendula officinalis L. (Marigold) Macerated Oil
3.6. Rosa damascena Miller (Damask Rose) Essential Oil
3.7. Origanum vulgare L. (Common Oregano) Essential Oil
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- van Zuuren, E.J.; Arents, B.W.; van der Linden, M.M.; Vermeulen, S.; Fedorowicz, Z.; Tan, J. Rosacea: New concepts in classification and treatment. Am. J. Clin. Dermatol. 2021, 22, 457–465. [Google Scholar] [CrossRef]
- Solomon, C.G.; van Zuuren, E.J. Rosacea. N. Engl. J. Med. 2017, 377, 1754–1764. [Google Scholar] [CrossRef]
- Geng, R.S.; Bourkas, A.N.; Mufti, A.; Sibbald, R.G. Rosacea: Pathogenesis and therapeutic correlates. J. Cutan. Med. Surg. 2024, 28, 178–189. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.S.; Huang, W.W. Rosacea pathogenesis. Dermatol. Clin. 2018, 36, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Buddenkotte, J.; Steinhoff, M. Recent advances in understanding and managing rosacea. F1000Research 2018, 7, F1000 Faculty Rev-1885. [Google Scholar] [CrossRef] [PubMed]
- Margalit, A.; Kowalczyk, M.J.; Żaba, R.; Kavanagh, K. The role of altered cutaneous immune responses in the induction and persistence of rosacea. J. Dermatol. Sci. 2016, 82, 3–8. [Google Scholar] [CrossRef]
- Muto, Y.; Wang, Z.; Vanderberghe, M.; Two, A.; Gallo, R.L.; Di Nardo, A. Mast cells are key mediators of cathelicidin initiated skin inflammation in rosacea. J. Investig. Dermatol. 2014, 134, 2728. [Google Scholar] [CrossRef]
- Fisk, W.A.; Lev-Tov, H.A.; Clark, A.K.; Sivamani, R.K. Phytochemical and botanical therapies for rosacea: A systematic review. Phytother. Res. 2015, 29, 1439–1451. [Google Scholar] [CrossRef] [PubMed]
- Semenescu, I.; Similie, D.; Diaconeasa, Z.; Danciu, C. Recent advances in the management of rosacea through natural compounds. Pharmaceuticals 2024, 17, 212. [Google Scholar] [CrossRef] [PubMed]
- Husein-ElAhmed, H.; Steinhoff, M. Laser and light-based therapies in the management of rosacea: An updated systematic review. Lasers Med. Sci. 2021, 36, 1151–1160. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, S.A.; Einstein, G.P.; Tulp, O.L.; Sainvil, F.; Branly, R. Introduction to traditional medicine and their role in prevention and treatment of emerging and re-emerging diseases. Biomolecules 2022, 12, 1442. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Ma, Q.; Ye, L.; Piao, G. The traditional medicine and modern medicine from natural products. Molecules 2016, 21, 559. [Google Scholar] [CrossRef] [PubMed]
- Flieger, J.; Flieger, W.; Baj, J.; Maciejewski, R. Antioxidants: Classification, natural sources, activity/capacity measurements, and usefulness for the synthesis of nanoparticles. Materials 2021, 14, 4135. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Supuran, C.T. Natural products in drug discovery: Advances and opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef] [PubMed]
- Makgobole, M.U.; Mpofana, N.; Ajao, A.A.N. Medicinal plants for dermatological diseases: Ethnopharmacological significance of botanicals from West Africa in skin care. Cosmetics 2023, 10, 167. [Google Scholar] [CrossRef]
- Tabassum, N.; Hamdani, M. Plants used to treat skin diseases. Pharmacogn. Rev. 2014, 8, 52. [Google Scholar] [CrossRef]
- Tsioutsiou, E.E.; Amountzias, V.; Vontzalidou, A.; Dina, E.; Stevanović, Z.D.; Cheilari, A.; Aligiannis, N. Medicinal plants used traditionally for skin related problems in the south Balkan and east Mediterranean region—A review. Front. Pharmacol. 2022, 13, 936047. [Google Scholar] [CrossRef] [PubMed]
- Özşahin, E.; Eroğlu, İ. Spatiotemporal Change of Anthropogenic Biomes (Anthromes) of Turkey; Krystev, V., Efe, R., Atasoy, E., Eds.; Kliment Ohridski University Press: Sofia, Bulgaria, 2019; pp. 241–252. [Google Scholar]
- Çolak, A.H.; Rotherham, I.D. A review of the forest vegetation of Turkey: Its status past and present and its future conservation. Biol. Environ. 2006, 106B, 343–354. [Google Scholar] [CrossRef]
- Tilkat, E.; Jahan, I.; Hoşer, A.; Kaplan, A.; Özdemir, O.; Onay, A. Anatolian medicinal plants as potential antiviral agents: Bridging traditional knowledge and modern science in the fight against COVID-19 and related viral infections. Turk. J. Biol. 2024, 48, 218–241. [Google Scholar] [CrossRef] [PubMed]
- Sever, M. Folk medicine, folk healing. Gazi Ak. Bak. 2015, 9, 181–192. [Google Scholar] [CrossRef][Green Version]
- Kesik, M. Selçuklular’da sağlık, sağlık kurumları ve tıp eğitimi. Turk. J. Hist. 2020, 71, 115–144. [Google Scholar] [CrossRef]
- Melnyk, N.; Vlasova, I.; Skowrońska, W.; Bazylko, A.; Piwowarski, J.P.; Granica, S. Current knowledge on interactions of plant materials traditionally used in skin diseases in Poland and Ukraine with human skin microbiota. Int. J. Mol. Sci. 2022, 23, 9644. [Google Scholar] [CrossRef] [PubMed]
- Xiaoying, W.; Han, Z.; Yu, W. Glycyrrhiza glabra (Licorice): Ethnobotany and health benefits. In Sustained Energy for Enhanced Human Functions and Activity; Elsevier: Amsterdam, The Netherlands, 2017; pp. 231–250. [Google Scholar]
- Saeedi, M.; Morteza-Semnani, K.; Ghoreishi, M.R. The treatment of atopic dermatitis with licorice gel. J. Dermatol. Treat. 2003, 14, 153–157. [Google Scholar] [CrossRef] [PubMed]
- Isbrucker, R.; Burdock, G. Risk and safety assessment on the consumption of Licorice root (Glycyrrhiza sp.), its extract and powder as a food ingredient, with emphasis on the pharmacology and toxicology of glycyrrhizin. Regul. Toxicol. Pharmacol. 2006, 46, 167–192. [Google Scholar] [CrossRef] [PubMed]
- Fiore, C.; Eisenhut, M.; Ragazzi, E.; Zanchin, G.; Armanini, D. A history of the therapeutic use of liquorice in Europe. J. Ethnopharmacol. 2005, 99, 317–324. [Google Scholar] [CrossRef] [PubMed]
- Cerulli, A.; Masullo, M.; Montoro, P.; Piacente, S. Licorice (Glycyrrhiza glabra, G. uralensis, and G. inflata) and their constituents as active cosmeceutical ingredients. Cosmetics 2022, 9, 7. [Google Scholar] [CrossRef]
- Noreen, S.; Mubarik, F.; Farooq, F.; Khan, M.; Khan, A.U.; Pane, Y.S. Medicinal uses of licorice (Glycyrrhiza glabra L.): A comprehensive review. Open Access Maced. J. Med. Sci. 2021, 9, 668–675. [Google Scholar] [CrossRef]
- Quintana, S.E.; Cueva, C.; Villanueva-Bermejo, D.; Moreno-Arribas, M.V.; Fornari, T.; García-Risco, M.R. Antioxidant and antimicrobial assessment of licorice supercritical extracts. Ind. Crops Prod. 2019, 139, 111496. [Google Scholar] [CrossRef]
- Weber, T.; Ceilley, R.; Buerger, A.; Kolbe, L.; Trookman, N.; Rizer, R.; Schoelermann, A. Skin tolerance, efficacy, and quality of life of patients with red facial skin using a skin care regimen containing Licochalcone A. J. Cosmet. Dermatol. 2006, 5, 227–232. [Google Scholar] [CrossRef] [PubMed]
- Kolbe, L.; Immeyer, J.; Batzer, J.; Wensorra, U.; Dieck, K.T.; Mundt, C.; Wolber, R.; Stäb, F.; Schönrock, U.; Ceilley, R.I. Anti-inflammatory efficacy of Licochalcone A: Correlation of clinical potency and in vitro effects. Arch. Dermatol. Res. 2006, 298, 23–30. [Google Scholar] [CrossRef] [PubMed]
- Jovanovic, Z.; Angabini, N.; Ehlen, S.; Mokos, Z.B.; Subotic, M.; Neufang, G. Efficacy and Tolerability of a Cosmetic Skin Care Product With Trans-4-t-butylcyclohexanol and Licochalcone A in Subjects With Sensitive Skin Prone to Redness and Rosacea. J. Drugs Dermatol. 2017, 16, 605–610. [Google Scholar] [PubMed]
- Schoelermann, A.; Weber, T.; Arrowitz, C.; Rizer, R.; Qian, K.; Babcock, M. Skin compatibility and efficacy of a cosmetic skin care regimen with licochalcone A and 4-t-butylcyclohexanol in patients with rosacea subtype I. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 21–27. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Li, H.; Li, Y.; Li, M.; Chen, G. Effect of isoliquiritigenin for the treatment of atopic dermatitis-like skin lesions in mice. Arch. Dermatol. Res. 2017, 309, 805–813. [Google Scholar] [CrossRef] [PubMed]
- Yokota, T.; Nishio, H.; Kubota, Y.; Mizoguchi, M. The inhibitory effect of glabridin from licorice extracts on melanogenesis and inflammation. Pigment. Cell Res. 1998, 11, 355–361. [Google Scholar] [CrossRef]
- Xie, Y.; Huang, J.; Liu, J.; Zhang, Q. Efficacy of diammonium glycyrrhizinate in the treatment of rosacea with papules and pustules: A randomized, double-blind, placebo-controlled study. Dermatol. Ther. 2022, 35, e15905. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.M.; Hirota, S.; Jippo-Kanemoto, T.; Kim, H.R.; Shin, T.Y.; Yeom, Y.; Lee, K.K.; Kitamura, Y.; Nomura, S.; Kim, H.M. Inhibition of histamine synthesis by glycyrrhetinic acid in mast cells cocultured with Swiss 3T3 fibroblasts. Int. Arch. Allergy Immunol. 1996, 110, 272–277. [Google Scholar] [CrossRef] [PubMed]
- Frattaruolo, L.; Carullo, G.; Brindisi, M.; Mazzotta, S.; Bellissimo, L.; Rago, V.; Curcio, R.; Dolce, V.; Aiello, F.; Cappello, A.R. Antioxidant and Anti-Inflammatory Activities of Flavanones from Glycyrrhiza glabra L. (licorice) Leaf Phytocomplexes: Identification of Licoflavanone as a Modulator of NF-kB/MAPK Pathway. Antioxidants 2019, 8, 186. [Google Scholar] [CrossRef] [PubMed]
- Samani, B.H.; Sharifi, A.; Jamshidi-Kia, F.; Ghaterehsamani, S.; Taki, K. Advanced extraction of Glycyrrhiza glabra root extract using a combined ultrasonic and cold plasma reactor. Sci. Rep. 2025, 15, 9994. [Google Scholar] [CrossRef] [PubMed]
- Alsaadi, D.H.M.; Raju, A.; Kusakari, K.; Karahan, F.; Sekeroglu, N.; Watanabe, T. Phytochemical Analysis and Habitat Suitability Mapping of Glycyrrhiza glabra L. Collected in the Hatay Region of Turkey. Molecules 2020, 25, 5529. [Google Scholar] [CrossRef] [PubMed]
- Semenescu, I.; Avram, S.; Similie, D.; Minda, D.; Diaconeasa, Z.; Muntean, D.; Lazar, A.E.; Gurgus, D.; Danciu, C. Phytochemical, Antioxidant, Antimicrobial and Safety Profile of Glycyrrhiza glabra L. Extract Obtained from Romania. Plants 2024, 13, 3265. [Google Scholar] [CrossRef] [PubMed]
- Selyutina, O.Y.; Polyakov, N.E. Glycyrrhizic acid as a multifunctional drug carrier—From physicochemical properties to biomedical applications: A modern insight on the ancient drug. Int. J. Pharm. 2019, 559, 271–279. [Google Scholar] [CrossRef] [PubMed]
- Wahab, S.; Annadurai, S.; Abullais, S.S.; Das, G.; Ahmad, W.; Ahmad, M.F.; Kandasamy, G.; Vasudevan, R.; Ali, M.S.; Amir, M. Glycyrrhiza glabra (Licorice): A Comprehensive Review on Its Phytochemistry, Biological Activities, Clinical Evidence and Toxicology. Plants 2021, 10, 2751. [Google Scholar] [CrossRef] [PubMed]
- Eghlima, G.; Tafreshi, Y.M.; Aghamir, F.; Ahadi, H.; Hatami, M. Regional environmental impacts on growth traits and phytochemical profiles of Glycyrrhiza glabra L. for enhanced medicinal and industrial use. BMC Plant Biol. 2025, 25, 116. [Google Scholar] [CrossRef] [PubMed]
- Krittanai, S.; Pichetpongtorn, P.; Sakamoto, S.; Putalun, W. Monoclonal antibody-based immunoassay for the specific quantification of licochalcone A: An active chalcone in licorice. Food Agric. Immunol. 2022, 33, 220–234. [Google Scholar] [CrossRef]
- Melnyk, N.; Nyczka, A.; Piwowarski, J.P.; Granica, S. Traditional Use of Chamomile Flowers (Matricariae flos) in Inflammatory-Associated Skin Disorders. Prospect. Pharm. Sci. 2024, 22, 59–73. [Google Scholar] [CrossRef]
- El Mihyaoui, A.; Esteves da Silva, J.C.G.; Charfi, S.; Candela Castillo, M.E.; Lamarti, A.; Arnao, M.B. Chamomile (Matricaria chamomilla L.): A Review of Ethnomedicinal Use, Phytochemistry and Pharmacological Uses. Life 2022, 12, 479. [Google Scholar] [CrossRef]
- Mehmood, M.H.; Munir, S.; Khalid, U.A.; Asrar, M.; Gilani, A.H. Antidiarrhoeal, antisecretory and antispasmodic activities of Matricaria chamomilla are mediated predominantly through K+-channels activation. BMC Complement. Altern. Med. 2015, 15, 75. [Google Scholar] [CrossRef] [PubMed]
- Sah, A.; Naseef, P.P.; Kuruniyan, M.S.; Jain, G.K.; Zakir, F.; Aggarwal, G. A Comprehensive Study of Therapeutic Applications of Chamomile. Pharmaceuticals 2022, 15, 1284. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency. Assessment Report on Matricaria recutita L., Flos; European Medicines Agency: Amsterdam, The Netherlands, 2015; p. 44. [Google Scholar]
- Tisserand, R.; Young, R. Essential Oil Safety: A Guide for Health Care Professionals; Elsevier Health Sciences: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Sharifi-Rad, M.; Nazaruk, J.; Polito, L.; Morais-Braga, M.F.B.; Rocha, J.E.; Coutinho, H.D.M.; Salehi, B.; Tabanelli, G.; Montanari, C.; Del Mar Contreras, M.; et al. Matricaria genus as a source of antimicrobial agents: From farm to pharmacy and food applications. Microbiol. Res. 2018, 215, 76–88. [Google Scholar] [CrossRef] [PubMed]
- Reszko, A.E.; Berson, D.; Lupo, M.P. Cosmeceuticals: Practical applications. Dermatol. Clin. 2009, 27, 401–416. [Google Scholar] [CrossRef] [PubMed]
- Singh, O.; Khanam, Z.; Misra, N.; Srivastava, M.K. Chamomile (Matricaria chamomilla L.): An overview. Pharmacogn. Rev. 2011, 5, 82–95. [Google Scholar] [CrossRef] [PubMed]
- Amellal, M.; Bronner, C.; Briancon, F.; Haag, M.; Anton, R.; Landry, Y. Inhibition of mast cell histamine release by flavonoids and biflavonoids. Planta Med. 1985, 51, 16–20. [Google Scholar] [CrossRef] [PubMed]
- Hirano, T.; Higa, S.; Arimitsu, J.; Naka, T.; Shima, Y.; Ohshima, S.; Fujimoto, M.; Yamadori, T.; Kawase, I.; Tanaka, T. Flavonoids such as luteolin, fisetin and apigenin are inhibitors of interleukin-4 and interleukin-13 production by activated human basophils. Int. Arch. Allergy Immunol. 2004, 134, 135–140. [Google Scholar] [CrossRef] [PubMed]
- Paiva-Santos, A.C.; Gonçalves, T.; Peixoto, D.; Pires, P.C.; Velsankar, K.; Jha, N.K.; Chavda, V.P.; Mohammad, I.S.; Cefali, L.C.; Mazzola, P.G.; et al. Rosacea Topical Treatment and Care: From Traditional to New Drug Delivery Systems. Mol. Pharm. 2023, 20, 3804–3828. [Google Scholar] [CrossRef] [PubMed]
- Emer, J.; Waldorf, H.; Berson, D. Botanicals and anti-inflammatories: Natural ingredients for rosacea. Semin. Cutan. Med. Surg. 2011, 30, 148–155. [Google Scholar] [CrossRef] [PubMed]
- Patzelt-Wenczler, R.; Ponce-Pöschl, E. Proof of efficacy of Kamillosan(R) cream in atopic eczema. Eur. J. Med. Res. 2000, 5, 171–175. [Google Scholar] [PubMed]
- Dos Santos, D.S.; Barreto, R.S.S.; Serafini, M.R.; Gouveia, D.N.; Marques, R.S.; Nascimento, L.C.; Nascimento, J.C.; Guimarães, A.G. Phytomedicines containing Matricaria species for the treatment of skin diseases: A biotechnological approach. Fitoterapia 2019, 138, 104267. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Bautista, R.J.; García-González, L.L.; Ocádiz-González, M.A.; Flores-Tochihuitl, J.; García-Villaseñor, A.; González-Hernández, M.; Muñoz-Hernández, L.; Ortiz-Figueroa, M.C.; Ramírez-Anaya, M.; Reyna-Téllez, S.; et al. Matricaria chamomilla (aqueous extract) improves atopic dermatitis-like lesions in a murine model. Rev. Med. Inst. Mex. Seguro Soc. 2017, 55, 587–593. [Google Scholar] [PubMed]
- Chen, G.; Lv, C.; Nie, Q.; Li, X.; Lv, Y.; Liao, G.; Liu, S.; Ge, W.; Chen, J.; Du, Y. Essential Oil of Matricaria chamomilla Alleviate Psoriatic-Like Skin Inflammation by Inhibiting PI3K/Akt/mTOR and p38MAPK Signaling Pathway. Clin. Cosmet. Investig. Dermatol. 2024, 17, 59–77. [Google Scholar] [CrossRef]
- Baylac, S.; Racine, P. Inhibition of 5-lipoxygenase by essential oils and other natural fragrant extracts. Int. J. Aromather. 2003, 13, 138–142. [Google Scholar] [CrossRef]
- Demirci, B.; Öztürk, G.; Demirci, F. Antibacterial Evaluation of Matricaria recutita L., Achillea millefolium L. Essential Oil and Tetracycline Combinations in Respect to in vivo Toxicity Data. Rec. Nat. Prod. 2025, 19, 278–285. [Google Scholar] [CrossRef]
- Tolekova, S.; Sharmanov, T.; Sınyavskıy, Y.; Berzhanova, R.; Mammadov, R.; Aksoy, Ö.K.; Yusifli, R. Antioxidant, pharmacological, medical properties and chemical content of Rosa L. extracts. Int. J. Second. Metab. 2020, 7, 200–212. [Google Scholar] [CrossRef]
- Ozay, C. Ethnopharmacological properties of rosehip (Rosa canina L.) and its importance of production in Turkey. In Current Research in Science and Mathematics; Gece Publishing: Ankara, Turkey, 2023; pp. 91–104. [Google Scholar]
- Stryjecka, M.; Kiełtyka-Dadasiewicz, A.; Michalak, M. Physico-Chemical Characteristics of Rosa canina L. Seeds and Determining Their Potential Use. Appl. Sci. 2025, 15, 168. [Google Scholar] [CrossRef]
- Vasić, D.; Trifunović, B.Š.; Pećinar, I.; Paunović, D.; Popović-Djordjević, J. Chemical Characterization of Rosa canina L. Rosehip Seed: Application of Raman Spectroscopy and Gas Chromatography. Biol. Life Sci. Forum 2021, 3, 50. [Google Scholar] [CrossRef]
- Popović-Djordjević, J.; Špirović-Trifunović, B.; Pećinar, I.; de Oliveira, L.F.C.; Krstić, Đ.; Mihajlović, D.; Akšić, M.F.; Simal-Gandara, J. Fatty acids in seed oil of wild and cultivated rosehip (Rosa canina L.) from different locations in Serbia. Ind. Crops Prod. 2023, 191, 115797. [Google Scholar] [CrossRef]
- Bakhtiar, Z.; Eghlima, G.; Hatami, M.; Mirjalili, M.H. Quantification of fatty acids in seed oil and important bioactive compounds in Iranian Rosa canina L. ecotypes for potential cosmetic and medicinal uses. Sci. Rep. 2023, 13, 22721. [Google Scholar] [CrossRef] [PubMed]
- Güney, M. Determination of fatty acid profile and antioxidant activity of Rosehip seeds from Turkey. Int. J. Agric. Environ. Food Sci. 2020, 4, 114–118. [Google Scholar] [CrossRef]
- Fromm, M.; Bayha, S.; Kammerer, D.R.; Carle, R. Identification and quantitation of carotenoids and tocopherols in seed oils recovered from different Rosaceae species. J. Agric. Food Chem. 2012, 60, 10733–10742. [Google Scholar] [CrossRef] [PubMed]
- Van den Berg, H.; Faulks, R.; Granado, H.F.; Hirschberg, J.; Olmedilla, B.; Sandmann, G.; Southon, S.; Stahl, W. The potential for the improvement of carotenoid levels in foods and the likely systemic effects. J. Sci. Food Agric. 2000, 80, 880–912. [Google Scholar] [CrossRef]
- Oargă, D.P.; Cornea-Cipcigan, M.; Nemeș, S.A.; Cordea, M.I. The Effectiveness of a Topical Rosehip Oil Treatment on Facial Skin Characteristics: A Pilot Study on Wrinkles, UV Spots Reduction, Erythema Mitigation, and Age-Related Signs. Cosmetics 2025, 12, 125. [Google Scholar] [CrossRef]
- Grajzer, M.; Prescha, A.; Korzonek, K.; Wojakowska, A.; Dziadas, M.; Kulma, A.; Grajeta, H. Characteristics of rose hip (Rosa canina L.) cold-pressed oil and its oxidative stability studied by the differential scanning calorimetry method. Food Chem. 2015, 188, 459–466. [Google Scholar] [CrossRef]
- Winther, K.; Sophie Vinther Hansen, A.; Campbell-Tofte, J. Bioactive ingredients of rose hips (Rosa canina L) with special reference to antioxidative and anti-inflammatory properties: In vitro studies. Bot. Targets Ther. 2016, 6, 11–23. [Google Scholar] [CrossRef]
- Oargă Porumb, D.P.; Cornea-Cipcigan, M.; Cordea, M.I. Unveiling the mechanisms for the development of rosehip-based dermatological products: An updated review. Front. Pharmacol. 2024, 15, 1390419. [Google Scholar] [CrossRef] [PubMed]
- Ande, S.N.; Bakal, R.L. Potential herbal essential oils: Are they super natural skin protector. Innov. Pharm. Pharm. 2022, 10, 19–24. [Google Scholar]
- Pereira Oliveira, C.N.; Nani Leite, M.; de Paula, N.A.; Araújo Martins, Y.; Figueiredo, S.A.; Cipriani Frade, M.A.; Lopez, R.F.V. Nanoemulsions Based on Sunflower and Rosehip Oils: The Impact of Natural and Synthetic Stabilizers on Skin Penetration and an Ex Vivo Wound Healing Model. Pharmaceutics 2023, 15, 999. [Google Scholar] [CrossRef]
- Ahmad, N.; Anwar, F. Rose hip (Rosa canina L.) oils. In Essential Oils in Food Preservation, Flavor and Safety; Elsevier: Amsterdam, The Netherlands, 2016; pp. 667–675. [Google Scholar]
- Schwager, J.; Richard, N.; Schoop, R.; Wolfram, S. A Novel Rose Hip Preparation with Enhanced Anti-Inflammatory and Chondroprotective Effects. Mediators Inflamm. 2014, 2014, 105710. [Google Scholar] [CrossRef]
- Jovanović, A.A.; Ćujić, D.; Stojadinović, B.; Čutović, N.; Živković, J.; Šavikin, K. Liposomal bilayer as a carrier of Rosa canina L. seed oil: Physicochemical characterization, stability, and biological potential. Molecules 2022, 28, 276. [Google Scholar] [CrossRef] [PubMed]
- Ozyurt, D.; Demirata, B.; Apak, R.; Hamilton, J.F.; Lewis, A.C.; Ozel, M.Z. GC×GC-TOF/MS Chromatographic Analysis, Antioxidant Capacity and Phenolic Content of Rosa Canina L. at Different Maturities. Rec. Nat. Prod. 2016, 10, 407–425. [Google Scholar]
- Yücel, A.; Kan, Y.; Yesilada, E.; Akın, O. Effect of St. John’s wort (Hypericum perforatum) oily extract for the care and treatment of pressure sores; a case report. J. Ethnopharmacol. 2017, 196, 236–241. [Google Scholar] [CrossRef]
- Wölfle, U.; Seelinger, G.; Schempp, C.M. Topical application of St. John’s wort (Hypericum perforatum). Planta Med. 2014, 80, 109–120. [Google Scholar] [CrossRef]
- Klemow, K.M.; Bartlow, A.; Crawford, J.; Kocher, N.; Shah, J.; Ritsick, M. Medical Attributes of St. John’s Wort (Hypericum perforatum). In Herbal Medicine: Biomolecular and Clinical Aspects; Benzie, I.F.F., Wachtel-Galor, S., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2011. [Google Scholar]
- Arsić, I.; Zugić, A.; Tadić, V.; Tasić-Kostov, M.; Mišić, D.; Primorac, M.; Runjaić-Antić, D. Estimation of dermatological application of creams with St. John’s Wort oil extracts. Molecules 2011, 17, 275–294. [Google Scholar] [CrossRef] [PubMed]
- Lyles, J.T.; Kim, A.; Nelson, K.; Bullard-Roberts, A.L.; Hajdari, A.; Mustafa, B.; Quave, C.L. The Chemical and Antibacterial Evaluation of St. John’s Wort Oil Macerates Used in Kosovar Traditional Medicine. Front. Microbiol. 2017, 8, 1639. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E.; Kartal, M.; Gülpinar, A.R.; Cos, P.; Matheeussen, A.; Maes, L.; Tasdemir, D. Assessment of antimicrobial and antiprotozoal activity of the olive oil macerate samples of Hypericum perforatum and their LC-DAD-MS analyses. Food Chem. 2013, 138, 870–875. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.E.; Kartal, M.; Gülpinar, A.R.; Yetkin, G.; Orlikova, B.; Diederich, M.; Tasdemir, D. Inhibitory effect of St. John’s Wort oil macerates on TNFα-induced NF-κB activation and their fatty acid composition. J. Ethnopharmacol. 2014, 155, 1086–1092. [Google Scholar] [CrossRef] [PubMed]
- Isacchi, B.; Bergonzi, M.C.; Carnevali, F.; van der Esch, S.A.; Vincieri, F.F.; Bilia, A.R. Analysis and stability of the constituents of St. John’s wort oils prepared with different methods. J. Pharm. Biomed. Anal. 2007, 45, 756–761. [Google Scholar] [CrossRef] [PubMed]
- Yuksekdag, S. The efficacy of St John’s wort oil macerates on intractable skin lesions of patients with idiopathic granulomatous mastitis: Preliminary results. J. Wound Care 2022, 31, 1006–1010. [Google Scholar] [CrossRef] [PubMed]
- Nayak, S.B.; Isik, K.; Marshall, J.R. Wound-Healing Potential of Oil of Hypercium perforatum in Excision Wounds of Male Sprague Dawley Rats. Adv. Wound Care 2017, 6, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Schempp, C.M.; Windeck, T.; Hezel, S.; Simon, J.C. Topical treatment of atopic dermatitis with St. John’s wort cream—A randomized, placebo controlled, double blind half-side comparison. Phytomedicine 2003, 10, 31–37. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, E.M.; Baumann, L.S. The foundation for the use of olive oil in skin care and botanical cosmeceuticals. In Olives and Olive Oil in Health and Disease Prevention; Elsevier: Amsterdam, The Netherlands, 2021; pp. 425–434. [Google Scholar]
- Sun, P.; Kang, T.; Xing, H.; Zhang, Z.; Yang, D.; Zhang, J.; Li, M. Phytochemical changes in aerial parts of Hypericum perforatum at different harvest stages. Rec. Nat. Prod. 2019, 13, 1–9. [Google Scholar] [CrossRef]
- Ejiohuo, O.; Folami, S.; Maigoro, A.Y. Calendula in modern medicine: Advancements in wound healing and drug delivery applications. Eur. J. Med. Chem. Rep. 2024, 12, 100199. [Google Scholar] [CrossRef]
- Silva, D.; Ferreira, M.S.; Sousa-Lobo, J.M.; Cruz, M.T.; Almeida, I.F. Anti-Inflammatory Activity of Calendula officinalis L. Flower Extract. Cosmetics 2021, 8, 31. [Google Scholar] [CrossRef]
- Deniz, L.; Serteser, A.; Kargıoğlu, M. Uşak Üniversitesi ve yakın çevresindeki bazı bitkilerin mahalli adları ve etnobotanik özellikleri. Afyon Kocatepe Üniv. Fen. Müh. Bil. Derg. 2010, 10, 57–72. [Google Scholar]
- Ugulu, I. Fidelity level and knowledge of medicinal plants used to make therapeutic Turkish baths. Stud. Ethno-Med. 2012, 6, 1–9. [Google Scholar] [CrossRef]
- Güven, U.M.; Arslan, S.; Çıracı, M.B.; Kayıran, S.D. Calendula officinalis L. bitkisinin morfolojik özellikleri, ekstre içeren topikal ilaç formülasyonu geliştirilmesi ve in vitro değerlendirilmesi. Mersin Üniv. Tıp Fak. Lokman Hekim. Tıp Tarihi Folklorik Tıp Derg. 2022, 12, 105–115. [Google Scholar] [CrossRef]
- Burnett, C.L.; Bergfeld, W.F.; Belsito, D.V.; Hill, R.A.; Klaassen, C.D.; Liebler, D.; Marks, J.G., Jr.; Shank, R.C.; Slaga, T.J.; Snyder, P.W.; et al. Final report of the Cosmetic Ingredient Review Expert Panel on the safety assessment of cocamidopropyl betaine (CAPB). Int. J. Toxicol. 2012, 31, 77s–111s. [Google Scholar] [CrossRef] [PubMed]
- Della Loggia, R.; Tubaro, A.; Sosa, S.; Becker, H.; Saar, S.; Isaac, O. The role of triterpenoids in the topical anti-inflammatory activity of Calendula officinalis flowers. Planta Med. 1994, 60, 516–520. [Google Scholar] [CrossRef] [PubMed]
- Lima, M.D.R.; Lopes, A.P.; Martins, C.; Brito, G.A.C.; Carneiro, V.C.; Goes, P. The Effect of Calendula officinalis on Oxidative Stress and Bone Loss in Experimental Periodontitis. Front. Physiol. 2017, 8, 440. [Google Scholar] [CrossRef]
- Shahane, K.; Kshirsagar, M.; Tambe, S.; Jain, D.; Rout, S.; Ferreira, M.K.M.; Mali, S.; Amin, P.; Srivastav, P.P.; Cruz, J.; et al. An Updated Review on the Multifaceted Therapeutic Potential of Calendula officinalis L. Pharmaceuticals 2023, 16, 611. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Vásquez, A.; Visconti-Lopez, F.J.; Cabanillas-Ramirez, C.; Díaz-Seijas, D.; Meléndez-Escalante, J.; Comandé, D.; Santero, M. Efficacy and Safety of Topical Application of Olive Oil for Preventing Pressure Ulcers: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Int. J. Environ. Res. Public Health 2022, 19, 14921. [Google Scholar] [CrossRef] [PubMed]
- Aghaei, M.; Nilfoushzadeh, M.; Aghaei, S. Two cases of Rosacea treated with topical ozonate olive oil. Dermatol. Clin. Res. 2017, 3, 151–154. [Google Scholar]
- Pommier, P.; Gomez, F.; Sunyach, M.P.; D’Hombres, A.; Carrie, C.; Montbarbon, X. Phase III randomized trial of Calendula officinalis compared with trolamine for the prevention of acute dermatitis during irradiation for breast cancer. J. Clin. Oncol. 2004, 22, 1447–1453. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Heris, Z.; Farahani, L.A.; Haghani, H.; Abdoli-Oskouee, S.; Hasanpoor-Azghady, S.B. Comparison the effects of topical application of olive and calendula ointments on Children’s diaper dermatitis: A triple-blind randomized clinical trial. Dermatol. Ther. 2018, 31, e12731. [Google Scholar] [CrossRef] [PubMed]
- Okuma, C.H.; Andrade, T.A.; Caetano, G.F.; Finci, L.I.; Maciel, N.R.; Topan, J.F.; Cefali, L.C.; Polizello, A.C.; Carlo, T.; Rogerio, A.P.; et al. Development of lamellar gel phase emulsion containing marigold oil (Calendula officinalis) as a potential modern wound dressing. Eur. J. Pharm. Sci. 2015, 71, 62–72. [Google Scholar] [CrossRef] [PubMed]
- Aslan, K.; Kızıltaş, H.; Güven, L.; Karagecili, H.; Arslan, D.; Gülçin, İ. Enzyme inhibition properties of Calendula officinalis, Matricaria chamomilla, and Anthemis pseudocotula: Kinetics and molecular docking studies. Rec. Nat. Prod. 2025, 19, 247–262. [Google Scholar] [CrossRef]
- Mahboubi, M. Rosa damascena as holy ancient herb with novel applications. J. Tradit. Complement. Med. 2016, 6, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Nayebi, N.; Khalili, N.; Kamalinejad, M.; Emtiazy, M. A systematic review of the efficacy and safety of Rosa damascena Mill. with an overview on its phytopharmacological properties. Complement. Ther. Med. 2017, 34, 129–140. [Google Scholar] [CrossRef] [PubMed]
- Widrlechner, M.P. History and utilization of Rosa damascena. Econ. Bot. 1981, 35, 42–58. [Google Scholar] [CrossRef]
- Timor, A.N. World production oil rose and rose oil. Nature Sci. 2011, 6, 93–110. [Google Scholar]
- Bıtrak, O.O.; Hatırlı, S.A. Dünyada Yağ Gülü Piyasası ve Türkiye’nin Rolü. Selçuk Üniv. Akş. Meslek Yüksekok. Sosyal Bilim. Dergisi 2022, 13, 85–94. [Google Scholar]
- Mohebitabar, S.; Shirazi, M.; Bioos, S.; Rahimi, R.; Malekshahi, F.; Nejatbakhsh, F. Therapeutic efficacy of rose oil: A comprehensive review of clinical evidence. Avicenna J. Phytomed 2017, 7, 206–213. [Google Scholar] [PubMed]
- Güler, A.E.; Karataş, S. Tarihin Önemli Sırrı: Gül, Gül Yağı ve Gülsuyu. In Proceedings of the XI. National Conference on the History of Turkish Pharmacy, Mersin, Turkey, 25–28 May 2014; p. 66. [Google Scholar]
- Almasirad, A.; Amanzadeh, Y.; Taheri, A.; Iranshahi, M. Composition of a historical rose oil sample (Rosa damascena Mill., Rosaceae). J. Essent. Oil Res. 2007, 19, 110–112. [Google Scholar] [CrossRef]
- Mileva, M.; Ilieva, Y.; Jovtchev, G.; Gateva, S.; Zaharieva, M.M.; Georgieva, A.; Dimitrova, L.; Dobreva, A.; Angelova, T.; Vilhelmova-Ilieva, N.; et al. Rose Flowers-A Delicate Perfume or a Natural Healer? Biomolecules 2021, 11, 127. [Google Scholar] [CrossRef] [PubMed]
- Boskabady, M.H.; Shafei, M.N.; Saberi, Z.; Amini, S. Pharmacological effects of rosa damascena. Iran. J. Basic. Med. Sci. 2011, 14, 295–307. [Google Scholar] [PubMed]
- Hadipour, E.; Rezazadeh Kafash, M.; Emami, S.A.; Asili, J.; Boghrati, Z.; Tayarani-Najaran, Z. Evaluation of anti-oxidant and antimelanogenic effects of the essential oil and extracts of Rosa × damascena in B16F10 murine melanoma cell line. Iran. J. Basic. Med. Sci. 2023, 26, 1076–1082. [Google Scholar] [CrossRef] [PubMed]
- Abdallah, H.M.; Koshak, A.E.; Farag, M.A.; El Sayed, N.S.; Badr-Eldin, S.M.; Ahmed, O.A.A.; Algandaby, M.M.; Abdel-Naim, A.B.; Ibrahim, S.R.M.; Mohamed, G.A.; et al. Taif Rose Oil Ameliorates UVB-Induced Oxidative Damage and Skin Photoaging in Rats via Modulation of MAPK and MMP Signaling Pathways. ACS Omega 2023, 8, 33943–33954. [Google Scholar] [CrossRef]
- Mohsen, E.; Younis, I.Y.; Farag, M.A. Metabolites profiling of Egyptian Rosa damascena Mill. flowers as analyzed via ultra-high-performance liquid chromatography-mass spectrometry and solid-phase microextraction gas chromatography-mass spectrometry in relation to its anti-collagenase skin effect. Ind. Crops Prod. 2020, 155, 112818. [Google Scholar] [CrossRef]
- Fahimi, S.; Abdollahi, M.; Mortazavi, S.A.; Hajimehdipoor, H.; Abdolghaffari, A.H.; Rezvanfar, M.A. Wound Healing Activity of a Traditionally Used Poly Herbal Product in a Burn Wound Model in Rats. Iran. Red. Crescent Med. J. 2015, 17, e19960. [Google Scholar] [CrossRef] [PubMed]
- Nurzyńska-Wierdak, R.; Walasek-Janusz, M. Chemical Composition, Biological Activity, and Potential Uses of Oregano (Origanum vulgare L.) and Oregano Essential Oil. Pharmaceuticals 2025, 18, 267. [Google Scholar] [CrossRef] [PubMed]
- Bora, L.; Avram, S.; Pavel, I.Z.; Muntean, D.; Liga, S.; Buda, V.; Gurgus, D.; Danciu, C. An Up-To-Date Review Regarding Cutaneous Benefits of Origanum vulgare L. Essential Oil. Antibiotics 2022, 11, 549. [Google Scholar] [CrossRef] [PubMed]
- Baytop, T. Türkiye’de Bitkiler ile Tedavi: Geçmişte ve Bugün; Nobel Tıp Kitabevleri: Istanbul, Turkey, 1999. [Google Scholar]
- Ertuğ, F. Etnobotanik kaynakları. Resim. Türkiye Florası 2014, 1, 381–420. [Google Scholar]
- Baser, K.; Özek, T.; Kürkçüoglu, M.; Tümen, G. The essential oil of Origanum vulgare subsp. hirtum of Turkish origin. J. Essent. Oil Res. 1994, 6, 31–36. [Google Scholar] [CrossRef]
- Karik, Ü.; Tınmaz, A.B.; Kürkçüoğlu, M.; Başer, K.H.C.; Tümen, G. İstanbul kekiği (Origanum vulgare L. Subsp. hirtum) populasyonlarinda farkli biçim zamanlarinin verim ve kaliteye etkileri. Bahçe 2007, 36, 37–48. [Google Scholar]
- Özcan, M.M.; Pedro, L.G.; Al-Juhaimi, F.; Endes, Z.; Erol, A.S.; Duman, E.; Er, F. Constituents of the Essential oil of Origanum vulgare subsp. hirtum Growing Wild in Turkey. J. Essent. Oil Bear. Plants 2012, 15, 572–576. [Google Scholar] [CrossRef]
- Esen, G.; Azaz, A.D.; Kurkcuoglu, M.; Baser, K.H.C.; Tinmaz, A. Essential oil and antimicrobial activity of wild and cultivated Origanum vulgare L. subsp. hirtum (Link) letswaart from the Marmara region, Turkey. Flavour. Fragr. J. 2007, 22, 371–376. [Google Scholar] [CrossRef]
- Leyva-López, N.; Gutiérrez-Grijalva, E.P.; Vazquez-Olivo, G.; Heredia, J.B. Essential Oils of Oregano: Biological Activity beyond Their Antimicrobial Properties. Molecules 2017, 22, 989. [Google Scholar] [CrossRef] [PubMed]
- Laothaweerungsawat, N.; Sirithunyalug, J.; Chaiyana, W. Chemical Compositions and Anti-Skin-Ageing Activities of Origanum vulgare L. Essential Oil from Tropical and Mediterranean Region. Molecules 2020, 25, 1101. [Google Scholar] [CrossRef] [PubMed]
- Chaftar, N.; Girardot, M.; Labanowski, J.; Ghrairi, T.; Hani, K.; Frère, J.; Imbert, C. Comparative evaluation of the antimicrobial activity of 19 essential oils. In Advances in Microbiology, Infectious Disease and Public Health; Advances in Experimental Medicine and Biology; Springer: Berlin/Heidelberg, Germany, 2016; Volume 901, pp. 1–15. [Google Scholar] [CrossRef]
- Avola, R.; Granata, G.; Geraci, C.; Napoli, E.; Graziano, A.C.E.; Cardile, V. Oregano (Origanum vulgare L.) essential oil provides anti-inflammatory activity and facilitates wound healing in a human keratinocytes cell model. Food Chem. Toxicol. 2020, 144. [Google Scholar] [CrossRef]
- Han, X.; Parker, T.L. Anti-inflammatory, tissue remodeling, immunomodulatory, and anticancer activities of oregano (Origanum vulgare) essential oil in a human skin disease model. Biochim. Open 2017, 4, 73–77. [Google Scholar] [CrossRef] [PubMed]
- Gören, H.Y. Dermatolojik Hastaliklarin Tedavisi İçin Fitokimyasal İlaç ve Endikasyonlari. TP-2020/17641, 11 March 2020. [Google Scholar]
- Kültür, Ş. Medicinal plants used in Kırklareli province (Turkey). J. Ethnopharmacol. 2007, 111, 341–364. [Google Scholar] [CrossRef] [PubMed]
- Ali, S.I.; Gopalakrishnan, B.; Venkatesalu, V. Pharmacognosy, phytochemistry and pharmacological properties of Achillea millefolium L.: A review. Phytother. Res. 2017, 31, 1140–1161. [Google Scholar] [CrossRef] [PubMed]
- Martins, C.A.F.; Campos, M.L.; Irioda, A.C.; Stremel, D.P.; Trindade, A.C.L.B.; Pontarolo, R. Anti-inflammatory effect of Malva sylvestris, Sida cordifolia, and Pelargonium graveolens is related to inhibition of prostanoid production. Molecules 2017, 22, 1883. [Google Scholar] [CrossRef] [PubMed]
- Kmail, A.; Said, O.; Saad, B. How thymoquinone from nigella sativa accelerates wound healing through multiple mechanisms and targets. Curr. Issues Mol. Biol. 2023, 45, 9039–9059. [Google Scholar] [CrossRef] [PubMed]
- Tanrıkulu, N. Bazı tıbbi bitkilerin kadim tıp konulu eserler, halk tıbbı ve bilimsel araştırmalardaki bulgularının karşılaştırılması. Biol. Divers. Conserv. 2024, 17, 267–280. [Google Scholar] [CrossRef]
- Pulaj, B.; Mustafa, B.; Hajdari, A. Differentiation of Achillea millefolium, A. crithmifolia, and A. nobilis through Analysis of Volatile Constituents using HS-SPME-GC/MS and Chemometric Techniques. Rec. Nat. Prod. 2024, 18, 597–609. [Google Scholar] [CrossRef]
- Hanoglu, D.; Hanoglu, A.; Yusufoglu, H.; Demirci, B.; Baser, K.; Çaliş, İ.; Yavuz, D. Phytochemical investigation of endemic Sideritis cypria post. Rec. Nat. Prod. 2020, 14, 105–115. [Google Scholar] [CrossRef]
- González-Burgos, E.; Carretero, M.; Gómez-Serranillos, M. Sideritis spp.: Uses, chemical composition and pharmacological activities—A review. J. Ethnopharmacol. 2011, 135, 209–225. [Google Scholar] [CrossRef] [PubMed]
- Tatli, I.I.; Akdemir, Z.F. Traditional uses and biological activities of Verbascum species. FABAD J. Pharm. Sci. 2006, 31, 85. [Google Scholar]
- Panchal, M.A.; Murti, K.; Lambole, V. Pharmacological properties of Verbascum thapsus—A review. Int. J. Pharm. Sci. Rev. Res. 2010, 5, 73–77. [Google Scholar]
- Prudente, A.S.; Loddi, A.M.; Duarte, M.R.; Santos, A.R.; Pochapski, M.T.; Pizzolatti, M.G.; Hayashi, S.S.; Campos, F.R.; Pontarolo, R.; Santos, F.A. Pre-clinical anti-inflammatory aspects of a cuisine and medicinal millennial herb: Malva sylvestris L. Food Chem. Toxicol. 2013, 58, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Tadić, V.; Arsić, I.; Zvezdanović, J.; Zugić, A.; Cvetković, D.; Pavkov, S. The estimation of the traditionally used yarrow (Achillea millefolium L. Asteraceae) oil extracts with anti-inflamatory potential in topical application. J. Ethnopharmacol. 2017, 199, 138–148. [Google Scholar] [CrossRef]
- Krgović, N.; Jovanović, M.; Aradski, A.A.; Janković, T.; Stević, T.; Zdunić, G.; Laušević, S.D.; Šavikin, K. Bioassay-guided skin-beneficial effects of fractionated Sideritis raeseri subsp. raeseri extract. Plants 2022, 11, 2677. [Google Scholar] [CrossRef] [PubMed]
- Maghalian, M.; Alizadeh, A.; Raphi, F.; Islambulchilar, Z.; Khodaie, L.; Nabighadim, M.; Taghavi, S.; Mirghafourvand, M. The effect of Nigella Sativa emulgel on episiotomy wound healing and pain intensity in primiparous women: A triple-blind randomized controlled trial. PLoS ONE 2025, 20, e0325112. [Google Scholar] [CrossRef] [PubMed]
- Tsai, M.-L.; Lin, C.-C.; Lin, W.-C.; Yang, C.-H. Antimicrobial, antioxidant, and anti-inflammatory activities of essential oils from five selected herbs. Biosci. Biotechnol. Biochem. 2011, 75, 1977–1983. [Google Scholar] [CrossRef] [PubMed]
- Radi, F.Z.; Bouhrim, M.; Mechchate, H.; Al-Zahrani, M.; Qurtam, A.A.; Aleissa, A.M.; Drioiche, A.; Handaq, N.; Zair, T. Phytochemical analysis, antimicrobial and antioxidant properties of Thymus zygis L. and Thymus willdenowii Boiss. essential oils. Plants 2021, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.-S.; Choi, W.J.; Lee, S.; Kirn, W.J.; Lee, D.C.; Sohn, U.D.; Shin, H.-S.; Kim, W. Anti-inflammatory, antioxidant and antimicrobial effects of artemisinin extracts from Artemisia annua L. Korean J. Physiol. Pharmacol. 2015, 19, 21–27. [Google Scholar] [CrossRef]
- Erel, Ş.B.; Reznicek, G.; Şenol, S.G.; Yavaşoğlu, N.Ü.K.; Konyalioğlu, S.; Zeybek, A.U. Antimicrobial and antioxidant properties of Artemisia L. species from western Anatolia. Turk. J. Biol. 2012, 36, 75–84. [Google Scholar] [CrossRef]
- Mirković, S.; Martinović, M.; Tadić, V.M.; Nešić, I.; Jovanović, A.S.; Žugić, A. Antimicrobial and Antioxidant Activity of Essential Oils from Selected Pinus Species from Bosnia and Herzegovina. Antibiotics 2025, 14, 677. [Google Scholar] [CrossRef] [PubMed]
- Tümen, İ.; Akkol, E.K.; Taştan, H.; Süntar, I.; Kurtca, M. Research on the antioxidant, wound healing, and anti-inflammatory activities and the phytochemical composition of maritime pine (Pinus pinaster Ait). J. Ethnopharmacol. 2018, 211, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.-Y.; Choi, W.S.; Kwon, O.S.; Lee, J.S.; Son, S.Y.; Lee, C.H.; Lee, S.; Song, J.Y.; Lee, Y.J.; Lee, J.-Y. Extract of Pinus densiflora needles suppresses acute inflammation by regulating inflammatory mediators in RAW264. 7 macrophages and mice. Pharm. Biol. 2022, 60, 1148–1159. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, A.S.; Rolo, J.; Gaspar, C.; Ramos, L.; Cavaleiro, C.; Salgueiro, L.; Palmeira-de-Oliveira, R.; Teixeira, J.P.; Martinez-de-Oliveira, J.; Palmeira-de-Oliveira, A. Thymus mastichina (L.) L. and Cistus ladanifer L. for skin application: Chemical characterization and in vitro bioactivity assessment. J. Ethnopharmacol. 2023, 302, 115830. [Google Scholar] [CrossRef] [PubMed]
| Plant | Active Compounds | Activity/Mechanism | Reference |
|---|---|---|---|
| Glycyrrhiza glabra L. Extract | Saponins (glycyrrhizic acid–glycyrrhizin, glycyrrhetinic acid), Chalcones (isoliquiritigenin, licochalcone A, isoliquiritin), Flavanones (liquiritigenin, liquiritigenin apiosyl glucoside, liquiritin, licoflavone A, kaempferol, glabrin A, glabrin B, apigenin rutinoside), and Isoflavones (isoangustone A, glabridin, formononetin) | Anti-inflammatory, Antioxidant, Anti-allergenic, Anti-immune-mediated cytotoxicity, Anti-erythematous, Anti-irritant | [24,25,26,27,28,29,30,31,32,33,34,35,36,37,38,39,40,41,42,43,44,45,46] |
| Matricaria recutita L. Extract and Essential Oil | Phenolic acids (caffeic acid, chlorogenic acid, p-coumaric acid), Flavanones (apigenin, luteolin, penduletin, rutin, quercetin, and their derivatives), Terpenes (α-bisabolol, bisabolol oxide A, bisabolol oxide B, β-farnesene, chamazulene, and matricin), and Coumarins (umbelliferone, herniarin, and 7-methoxy-coumarin) | Anti-inflammatory, Antioxidant, Anti-allergenic, Analgesic, Antimicrobial, Antiangiogenic | [47,48,49,50,51,52,53,54,55,56,57,58,59,60,61,62,63,64,65] |
| Rosa canina L. Seed Oil | Unsaturated Fatty Acids (α-linolenic, linoleic, and oleic acids), Phytosterols (β-sitosterol, campesterol, stigmasterol, ∆5 avenasterol, and ∆7 avenasterol), Tocopherols (α-, γ-, and δ-tocopherol), Carotenoids (β-carotene, lycopene, zeaxanthin, and lutein), and Polyphenolic compounds (methyl esters of p-coumaric acid, vanillic acid, and 4-hydroxybenzoic acid) | Anti-inflammatory, Antioxidant, Antimicrobial, Anti-aging | [66,67,68,69,70,71,72,73,74,75,76,77,78,79,80,81,82,83,84] |
| Hypericum perforatum L. Macerated Oil | Naphthodianthrone derivatives (hypericin and pseudohypericin), Acylated phloroglucinols (hyperforin and adhyperforin), and Flavonoids (quercetin, quercitrin, hyperoside, rutin, kaempferol, biapigenin, and amentoflavone) | Anti-inflammatory, Antioxidant, Antimicrobial | [85,86,87,88,89,90,91,92,93,94,95,96,97] |
| Calendula officinalis L. Macerated Oil | Triterpenoids, along with their esters (faradiol, taraxasterol, and lupeol; faradiol-3-palmitate and faradiol-3-myristate), Carotenoids (lutein, β-carotene, flavoxanthin, and rubixanthin), and Flavonoids (quercetin, isorhamnetin, kaempferol, and their derivatives) | Anti-inflammatory, Antioxidant, Antimicrobial | [98,99,100,101,102,103,104,105,106,107,108,109,110,111,112] |
| Rosa damascena Miller (Damask Rose) Essential Oil | Terpenes (citronellol, geraniol, nerol) and Aliphatic hydrocarbons (nonadecane, heptadecane, and heneicosane) | Anti-inflammatory, Antioxidant, Antimicrobial, Anti-aging | [113,114,115,116,117,118,119,120,121,122,123,124,125,126] |
| Origanum vulgare L. (Common Oregano) Essential Oil | Terpenes (carvacrol, thymol, linalool, γ-terpinene, and p-cymene) | Anti-inflammatory, Antioxidant, Antimicrobial, Anti-aging | [127,128,129,130,131,132,133,134,135,136,137,138,139] |
| Type of Active Extract Tested | Type of Experiment Used to Prove Activity | Active Compounds Responsible | Active Dose/Regimen | Most Probable Molecular Mechanism | Improvement Related to Rosacea | Reference |
|---|---|---|---|---|---|---|
| Licorice supercritical extract obtained using supercritical CO2 with ethanol as co-solvent | In vitro antioxidant (TPC, radical scavenging assays) | Liquiritin, liquiritigenin, glycyrrhizin, isoliquiritigenin, glabridin | Concentration-dependent effects reported; antioxidant capacity correlated with total phenolic content. Highest values were 556 and 760 μmol/g for the ABTS and DPPH assay | Antioxidant activity mainly attributed to high phenolic content; free radical scavenging and redox modulation | Indirect benefit: reduction in oxidative stress, a key factor in rosacea pathophysiology | [30] |
| Topical formulation containing licorice-derived flavonoid | Open-label clinical study in rosacea patients (n = 62); concomitant use with topical metronidazole evaluated in a subset of participants | Licochalcone A | Once-daily topical application for 8 weeks | Anti-inflammatory and antioxidant effects; modulation of erythema-associated inflammatory pathways | Significant reduction in facial erythema; improvement in skin appearance and quality of life | [31] |
| Oil-in-water topical formulation containing licorice aqueous extract | Controlled clinical study in healthy volunteers; shaving- and UV-induced erythema model; complementary in vitro assays | Licochalcone A | 0.025% and 0.05% (w/w) topical application, twice daily for 3 days | Potent anti-inflammatory activity via inhibition of oxidative burst in granulocytes and suppression of pro-inflammatory mediators (PGE2, LTB4, IL-6, TNF-α) | Significant reduction in erythema induced by UV exposure and shaving; relevant to erythema-dominant rosacea | [32] |
| Topical cosmetic formulation containing licorice-derived flavonoids (licochalcone A and trans-4-t-butylcyclohexanol) | Open-label, international, multicenter clinical study in subjects with sensitive, redness-prone, and rosacea-affected skin (n = 1221) | Licochalcone A; trans-4-t-butylcyclohexanol | Twice-daily topical application for 4 weeks | TRPV1 inhibition leading to reduced calcium influx in keratinocytes; anti-inflammatory and barrier-protective effects | Marked reduction in facial redness and erythema; improvement of clinical symptoms in rosacea-prone skin | [33] |
| Topical cosmetic skin care regimen (cleanser, day care SPF 25, night care) containing licorice-derived flavonoids (licochalcone A and trans-4-t-butylcyclohexanol) | Open-label clinical study in patients with mild to moderate rosacea subtype I (n = 32) | Licochalcone A; 4-t-butylcyclohexanol (SymSitive®) | Twice-daily topical application for 8 weeks | Anti-inflammatory effects combined with TRPV1 inhibition, leading to reduced neurogenic inflammation and improved skin barrier function | Significant reduction in erythema and skin irritation; improved skin compatibility in rosacea subtype I | [34] |
| Isolated chalcone compound licorice-derived | In vivo, DNCB-induced AD-like murine model (BALB/c mice, n = 6/group); in vitro THP-1 monocyte model | Isoliquiritigenin | 1% (topical), once daily for 13 days | Suppression of Th2 cytokines (IL-4, IL-13) and IgE; inhibition of TNF-α, IL-6; blockade of p38α and ERK MAPK signaling | Significant reduction in erythema-associated dermatitis severity, scratching behavior, and inflammatory markers | [35] |
| Glabridin isolated from the hydrophobic (ethyl acetate-type) fraction of licorice extract | In vivo UVB-induced pigmentation and erythema model in guinea pigs; complementary in vitro assays | Glabridin | 0.5% topical glabridin applied to UVB-irradiated skin | Inhibition of inflammatory mediators via suppression of superoxide anion production and cyclooxygenase (COX) activity | Significant inhibition of UVB-induced erythema, suggesting a potential role in redness and inflammation control | [36] |
| Diammonium glycyrrhizinate, a glycyrrhizin salt extracted from licorice | Randomized, double-blind, placebo-controlled study in 117 rosacea patients (mainly papules and pustules) | Glycyrrhizin (as diammonium salt) | 150 mg DG orally, three times daily (450 mg/day) | Anti-inflammatory and immunomodulatory: inhibits NF-κB, decreases TNF-α, IL-1, IL-6, IL-17, reduces ROS and nitric oxide synthase, increases SOD and catalase activity, protects liver cells | Enhanced reduction in papules and pustules; earlier symptom improvement, higher total effective rates (93–94% vs. 68% in standard therapy alone); allowed halving of isotretinoin and clarithromycin doses; lower incidence of adverse events (dryness, liver enzyme elevation, GI discomfort) | [37] |
| Topical licorice extract gel (standardized based on glycyrrhizinic acid, 20% in extract, 19.6% in gel) | Double-blind clinical trial in patients with atopic dermatitis (30 patients/group, 2-week study); compared 1% and 2% gels with base gel | Glycyrrhizinic acid | 1% and 2% topical gel, applied twice daily for 2 weeks | Anti-inflammatory and antioxidant; reduces cytokine-mediated inflammation in skin; reduces erythema, edema, and pruritus | 2% gel more effective than 1% in reducing erythema, edema, and itching; implies potential to reduce inflammation and erythema in rosacea | [25] |
| Glycyrrhetinic acid (licorice-derived metabolite) | In vitro, mast cells co-cultured with Swiss 3T3 fibroblasts | Glycyrrhetinic acid | Glycyrrhetinic acid at 50 µM strongly inhibited histidine decarboxylase activity (~80% inhibition) in mast cells | Direct inhibition of histidine decarboxylase (HDC) activity → decreased histamine synthesis; reduced maturation of mast cells; downregulation of nPKCδ mRNA expression, suggesting signaling modulation | Potential to reduce mast cell–mediated inflammation and histamine-related vasodilation/erythema in rosacea by suppressing histamine synthesis and inflammatory mediator release | [38] |
| Licorice leaf ethanol 96% extract | In vitro, LPS-stimulated RAW 264.7 macrophages; anti-inflammatory and antioxidant assays; NF-kB/MAPK pathway analysis | Pinocembrin, licoflavone, and glabranin | 12.5–50 µg/mL (dried leaf ethanol extract, UAE); Licoflavanone: 10–200 µM, IC50 = 37.68 µM | Inhibition of NF-kB nuclear translocation; suppression of MAPK phosphorylation (ERK1/2, JNK, p38); reduction in iNOS, COX2, TNF-α, IL1-β, and IL-6 expression; antioxidant activity | Expected reduction in inflammation, oxidative stress, and erythema in rosacea due to NF-kB/MAPK modulation and cytokine suppression | [39] |
| Type of Active Extract Tested | Type of Experiment Used to Prove Activity | Active Compounds Responsible | Active Dose/Regimen | Most Probable Molecular Mechanism | Improvement Related to Rosacea | Reference |
|---|---|---|---|---|---|---|
| Kamillosan® cream (topical cream containing 2% ethanolic extract of chamomile flowers) | Randomized, partially double-blind, half-side comparison clinical trial in patients with moderate atopic eczema: Kamillosan® vs. 0.5% hydrocortisone cream vs. vehicle cream | Chamomile phytoactives (likely bisabolol, chamazulene, apigenin, and other flavonoids/sesquiterpenes)—principal anti-inflammatory constituents of chamomile extract | Topical application twice daily for 2 weeks; cream with 2% chamomile extract compared mainly with 0.5% hydrocortisone cream | Anti-inflammatory and anti-pruritic effects via modulation of inflammatory mediators; reduction in local inflammatory response in skin; inhibition of pro-inflammatory enzymes and cytokine pathways | Mild superiority over 0.5% hydrocortisone in improving pruritus, erythema, and desquamation, symptoms common to rosacea inflammatory presentations | [60] |
| Chamomile extract granules/capsules and ointment | Clinical trial (rosacea patients, n = 700) + preclinical study in mice (n = 210) | Bisabolol, chamazulene, apigenin | Oral: 10 g, 3×/day; topical ointment: 10 g, 1×/day, concentration 10–100 mg/mL | Improvement of microcirculation, skin nutrition, anti-inflammatory, and soothing effects | Reduced erythema, improved skin appearance, enhanced skin nutrition and microcirculation | [61] |
| Lyophilized aqueous extract of Matricaria chamomilla (7%) in an emollient vehicle | In vivo murine model of atopic dermatitis-like lesions (BALB/c mice, n = 12) | Apigenin (major), caffeic acid (minor) identified by HPLC | Topical application: 7% extract in petrolatum, 1×/day, 6 days/week for 4 weeks | Anti-inflammatory, antioxidant, immunomodulatory effects; reduction in epidermal hyperplasia and leukocyte infiltration | Histopathological improvement of lesions (50% of mice showed lesion normalization), reduction in epidermal inflammation, potential correlation with rosacea-associated erythema and inflammation | [62] |
| Chamomile essential oil | In vitro: HaCaT keratinocytes stimulated with IL-22/TNF-α/LPS; in vivo: IMQ-induced psoriatic-like skin inflammation in mice | Azulene (~88.9%), Isocaryophillene (13.82%), Cedrene (10.37%), Bisabolol, other sesquiterpenes, and monoterpenes | In vitro: 30–60 μg/mL; In vivo: 16 mg/time/mice, topical twice daily for 7 days | Downregulation of PI3K/Akt/mTOR and p38MAPK pathways; inhibition of inflammatory cytokines (IL-1β, IL-6, TNF-α, IL-10, TGF-β) | Reduced erythema, scaling, and epidermal thickening; anti-inflammatory effects that could improve rosacea-related skin inflammation | [63] |
| Chamomile essential oil (α-bisabolol-rich) | In vitro 5-LOX inhibition | α-Bisabolol | IC50 ≈ 10–30 ppm | 5-lipoxygenase inhibition → anti-inflammatory | Potential, based on anti-inflammatory activity | [64] |
| Type of Active Extract Tested | Type of Experiment Used to Prove Activity | Active Compounds Responsible | Active Dose/Regimen | Most Probable Molecular Mechanism | Improvement Related to Rosacea | Reference |
|---|---|---|---|---|---|---|
| Rosehip seed oil (topical) | In vivo study on 20 female volunteers; 8-week treatment; facial skin imaging analysis and participant feedback | Unsaturated fatty acids (linoleic acid, alpha-linolenic acid) and phenolic compounds | Topical application once daily for 8 weeks | Antioxidant activity, anti-inflammatory effects, support of skin regeneration and barrier protection | Reduced erythema, improved barrier function; potential benefit for rosacea due to anti-inflammatory and redness-mitigating effects | [75] |
| Nanoemulsions containing rosehip oil (3% w/w) + sunflower oil (15% w/w) with synthetic emulsifiers (Nano-1 and Nano-2) | In vitro: NIH-3T3 fibroblasts, HaCaT keratinocytes; Ex vivo: human skin explants (hOSEC); Confocal microscopy penetration studies; Cytokine release assay | Rosehip oil bioactives (unsaturated fatty acids, phenolics, carotenoids) | Nano-1: 6.5 × 1013–3.2 × 1011 particles/mL; Nano-2: 3.2 × 1011 particles/mL; daily application for 7–14 days (ex vivo wound healing) | Improved delivery of bioactive compounds through skin layers; modulation of keratinocyte and fibroblast uptake; no induction of pro-inflammatory cytokines (IL-6, TNF-α) | Enhanced skin barrier penetration, improved wound closure, non-inflammatory profile suggesting potential for rosacea management by reducing irritant-induced inflammation | [80] |
| Rosehip powder | In vitro assays on human peripheral blood leukocytes (LPS/IFN-γ stimulated) and primary chondrocytes (IL-1β stimulated) | Ursolic acid, betulinic acid, galactolipids (GLGPG), DHA, α-tocopherol | Tested at µg/mL concentrations in cell culture; effects seen from ~10–100 µg/mL depending on cytokine | Downregulation of pro-inflammatory chemokines/cytokines (CCL5/RANTES, CXCL10/IP-10, IL-6, IL-12), decreased MMPs (MMP 1,3,13) and ADAMTS-4 expression | Anti-inflammatory potential via reduced cytokine/chemokine release and gene expression; theoretical benefit for rosacea inflammation and erythema | [82] |
| Rosehip seed oil | In vitro antioxidant assays (ABTS and DPPH radical scavenging) | Polyphenols, tocopherols (vitamin E), carotenoids, unsaturated fatty acids | DPPH IC50 ≈ 0.150 mg/mL; ABTS antioxidant capacity ≈ 0.215–0.269 µmol/mL (pure oil) | Free radical scavenging; lipid peroxidation inhibition; antioxidant defense | May reduce oxidative stress and inflammation in rosacea-affected skin by neutralizing ROS and protecting cells from oxidative damage | [83] |
| Type of Active Extract Tested | Type of Experiment Used to Prove Activity | Active Compounds Responsible | Active Dose/Regimen | Most Probable Molecular Mechanism | Improvement Related to Rosacea | Reference |
|---|---|---|---|---|---|---|
| St. John’s wort oil macerate | In vitro biofilm inhibition assay, quorum-sensing inhibition assay against Staphylococcus aureus | Hyperforin (phloroglucinol derivative) | MBIC50 (biofilm inhibition): 0.004–0.016% v/v; QSIC50 (quorum-sensing inhibition): 0.064–0.512% v/v | Anti-biofilm, quorum-sensing inhibition; reduces bacterial attachment, virulence, and biofilm formation; hyperforin also has anti-inflammatory properties | Limits S. aureus colonization and biofilm-mediated inflammatory triggers on skin; may reduce erythema, inflammation, and irritation in rosacea-prone skin; supports overall skin barrier protection and microbial balance | [89] |
| St. John’s wort oil macerate (topical oil massage) | Clinical case series on 21 women with idiopathic granulomatous mastitis-associated persistent skin lesions (pre- vs. post-treatment assessment) | Hypericin, hyperforin, and fatty acids | Topical massage twice daily for 2 min for 6 weeks (after standard steroid/antibiotic therapy) | Anti-inflammatory and wound-healing effects via reduction in hyperaemia, scaling, induration, and ulceration (skin lesion regression); may involve modulation of inflammatory response and enhancement of tissue repair mechanisms | Very significant regression of persistent skin lesions (overall success rate ~94%; total clearance + decreased severity) suggests potential to reduce chronic inflammation and erythema—relevant to rosacea inflammation control | [93] |
| St. John’s wort oil macerate | In vivo—excision wound model in male Sprague Dawley rats | Hypericin, hyperforin, flavonoids, fatty acids | Topical application on wound site once daily | Anti-inflammatory, antioxidant, and tissue regeneration effects: reduces pro-inflammatory cytokines, enhances collagen deposition, promotes angiogenesis and epithelialization | Accelerated wound closure, reduced inflammation, improved tissue repair; suggests potential benefit in rosacea-like inflammation and erythema due to anti-inflammatory and healing properties | [94] |
| H. perforatum apolar extract obtained by supercritical CO2 extraction, formulated as a topical cream | Randomized, double-blind, placebo-controlled half-side (split-body) clinical trial; additional histological and immunohistochemical evaluation | Predominantly hyperforin (lipophilic phloroglucinol derivative enriched by CO2 extraction); other apolar constituents | 5% H. perforatum extract in cream; applied to one body side vs. placebo on contralateral side for 4 weeks; n = 20 patients with atopic dermatitis | Anti-inflammatory effect via significant reduction in TNF-α expression, decreased inflammatory cell infiltration, and reduced epidermal hyperplasia; modulation of cytokine-mediated skin inflammation | Reduction in erythema and inflammatory severity; suppression of pro-inflammatory cytokines relevant to rosacea pathophysiology (TNF-α–driven inflammation and vascular responses), suggesting potential benefit for rosacea-associated redness and inflammation | [95] |
| Type of Active Extract Tested | Type of Experiment Used to Prove Activity | Active Compounds Responsible | Active Dose/Regimen | Most Probable Molecular Mechanism | Improvement Related to Rosacea | Reference |
|---|---|---|---|---|---|---|
| Marigold extract (commercial extract in refined soybean oil, stabilized with tocopherol) | In vitro inflammation model: LPS-stimulated RAW 264.7 murine macrophages; NO production measured by Griess assay; cell viability by AlamarBlue® | Terpenoids (faradiol esters, bisabolol), flavonoids (quercetin, kaempferol, isorhamnetin), carotenoids, polyunsaturated fatty acids (calendic acid) | 16–147 μL/mL; dose-dependent activity; ~50% inhibition of NO production at 147 μL/mL without cytotoxicity after 24 h incubation | Inhibition of inducible nitric oxide synthase (iNOS)-mediated NO production in activated macrophages; attenuation of LPS/TLR4-driven inflammatory signaling; indirect reduction in downstream pro-inflammatory mediators | Reduction in NO-mediated vasodilation and inflammatory signaling may alleviate erythema, flushing, and inflammatory exacerbations characteristic of rosacea; supports barrier-friendly, soothing adjunctive skincare | [99] |
| Marigold lipophilic ointment (Pommade au Calendula par Digestion; petrolatum-based digestion, non-solvent extract) | Randomized, controlled clinical trial in humans (breast cancer patients receiving adjuvant radiotherapy; n = 254) | Triterpenoid esters (faradiol monoesters), terpenoids, flavonoids (quercetin, isorhamnetin derivatives), carotenoids | Topical application to irradiated skin after each radiotherapy session throughout treatment period | Suppression of inflammatory cascade via inhibition of pro-inflammatory mediators (TNF-α, COX-2–related pathways), reduction in oxidative stress, stabilization of skin barrier and vascular response | Reduced inflammation, erythema and pain; mechanisms overlap with rosacea pathophysiology (neurovascular dysregulation, cytokine-driven inflammation), supporting potential adjunctive benefit in rosacea | [109] |
| Marigold ointment (1.5%) formulated in a beeswax- and lanolin-based carrier system; topical semisolid preparation | Triple-blind randomized clinical trial conducted in infants with non-severe, non-infected diaper dermatitis | Triterpenoids, flavonoids, carotenoids | Topical application of 1.5% ointment after each diaper change, for a duration of 7 days | Anti-inflammatory and antioxidant activity; enhancement of skin barrier function; reduction in erythema through modulation of inflammatory mediators | Anti-inflammatory and barrier-protective effects may be relevant to inflammatory dermatoses, including rosacea | [110] |
| Marigold oil formulated as a lamellar gel phase (LGP) emulsion | In vitro cytotoxicity assays (L929 fibroblasts, Annexin V/PI flow cytometry) and in vivo full-thickness excisional wound healing model in Wistar rats with histological (H&E, Gomori trichrome) and morphometric analyses | Flavonoids, triterpenoid esters, saponins, tannins, coumarins, and terpenoids | LGP emulsion containing 15% (w/w) calendula oil, applied topically once daily under occlusive dressing; in vivo dose reported as 15 mg/mL formulation; non-cytotoxic in vitro at 50–1000 µg/mL | Modulation of the inflammatory phase of tissue repair: early controlled leukocyte recruitment, followed by reduction in inflammatory infiltrate, enhanced angiogenesis, accelerated re-epithelialization, regulation of collagen deposition, and maintenance of wound hydration via lamellar structures enabling sustained release | Anti-inflammatory modulation, improved barrier restoration, reduced prolonged inflammation, and enhanced tissue repair are mechanistically relevant to rosacea, a condition characterized by impaired barrier function, chronic inflammation, and erythema | [111] |
| Type of Active Extract Tested | Type of Experiment Used to Prove Activity | Active Compounds Responsible | Active Dose/Regimen | Most Probable Molecular Mechanism | Improvement Related to Rosacea | Reference |
|---|---|---|---|---|---|---|
| Essential oil of R. damascena | In vitro cell culture assays on B16F10 murine melanoma cells: cell viability, melanin content, mushroom tyrosinase activity, ROS suppression, and tyrosinase protein level evaluation | Major volatile compounds: citronellol (~37.1%), geraniol (~12.7%), linalool, phenylethyl alcohol, and a total of 63 volatile components identified by GC–MS; oxygenated monoterpenes dominate (~56%). | Tested at 0.2–200 µg/mL in B16F10 cells; effects on melanin and ROS were significant at 2, 20, and 200 µg/mL concentrations. | Antioxidant and anti-melanogenic effects: suppression of intracellular ROS production; reduction in melanin content. | Demonstrates dose-dependent antioxidant and enzyme-modulating activities that are relevant to skin inflammation control; reduction of ROS and melanin synthesis may overlap with mechanisms beneficial in rosacea, particularly where oxidative stress and pigment-related inflammation are implicated | [123] |
| R. damascena essential oil formulated in an emulgel base (100 mg/g) and nano-emulgel (ROSE-NANO) at 50 mg/g and 100 mg/g for topical application | In vivo UVB-induced photoaging model in adult male Wistar rats, with topical pretreatment (0.5 g of emulgel per rat) 1 h before daily UVB exposure; evaluated via biochemical markers, histology, and gene/protein assays | Major volatile constituents: geraniol (29.2%), nerol (23.4%), citronellol (16.34%), phenylethyl alcohol (4.96%), linalool (3.24%), and others identified by GC–MS | 0.5 g per rat per day topically of: (a) emulgel with 100 mg/g ROSE, (b) nano-emulgel with 50 mg/g ROSE, (c) nano-emulgel with 100 mg/g ROSE; applied 1 h before UVB irradiation | Mitigation of UVB-induced oxidative damage through enhanced catalase and superoxide dismutase activities, reduction in pro-inflammatory cytokines (IL-6, TNF-α), downregulation of MAPK signaling (JNK, ERK1/2, p38), and decreased MMP-9 expression, resulting in protection against collagen degradation | The antioxidant, anti-inflammatory, and extracellular matrix protective effects are relevant to rosacea, a chronic inflammatory skin condition involving oxidative stress, MAPK/MMP dysregulation, and inflammation | [124] |
| Methanolic flower extracts ERF (Expanded Rose Flowers) and URF (Unexpanded Rose Flowers) and essential oil of R. damascena | In vitro anti-collagenase assay (methanolic extract); chemical profiling by UHPLC–MS/MS and HS-SPME–GC–MS (essential oil) | Methanolic extract: polyphenols and flavonoids (quercetin and kaempferol derivatives); essential oil: monoterpene alcohols (citronellol, geraniol, nerol, phenylethyl alcohol) | ERF: IC50 ≈ 25–30 µg/mL URF: IC50 ≈ 45–50 µg/mL | Inhibition of collagenase activity and antioxidant protection of dermal extracellular matrix (extract); potential anti-inflammatory and antioxidant effects inferred for the essential oil based on its terpene composition | May help limit matrix degradation, oxidative stress, and inflammatory skin damage associated with rosacea | [125] |
| Polyherbal cream (PHC): aqueous extracts of Malva sylvestris leaves + aqueous extracts of Solanum nigrum leaves + R. damascena essential oil | In vivo second-degree burn wound healing in rats; histopathological evaluation; antioxidant (DPPH) and antimicrobial (micro-dilution) assays | Phenolic compounds and tannins in aqueous extracts; volatile compounds in essential oil | Topical application of PHC (cream with 5% aqueous extracts each, 33% oily extract in a base of eucerin/petrolatum/beeswax) daily for 14 days | Accelerates wound closure via enhanced re-epithelialization, neovascularization, and collagen deposition; antioxidant radical scavenging | Suggests potential to mitigate inflammatory and oxidative processes in chronic skin conditions like rosacea by reducing inflammation and scavenging free radicals | [126] |
| Type of Active Extract Tested | Type of Experiment Used to Prove Activity | Active Compounds Responsible | Active Dose/Regimen | Most Probable Molecular Mechanism | Improvement Related to Rosacea | Reference |
|---|---|---|---|---|---|---|
| O. vulgare essential oil | In vitro enzymatic assays (collagenase, elastase, hyaluronidase inhibition); antioxidant assays (DPPH radical scavenging, FRAP, ferric thiocyanate lipid peroxidation assay) | Carvacrol (major constituent, 79.5%), thymol, p-cymene, and γ-terpinene | Collagenase inhibition: 92.0 ± 9.7% at 67 µg/mL (IC50 = 35.1 ± 0.9 µg/mL); elastase inhibition: 53.1 ± 13.3% at 25 µg/mL (IC50 = 24.3 ± 0.5 µg/mL); hyaluronidase inhibition: 16.7 ± 0.3% at 4 µg/mL; DPPH IC50: 1.8 ± 0.8 mg/mL | Antioxidant-mediated suppression of ROS; inhibition of matrix metalloproteinases (collagenase/MMPs and elastase), preventing extracellular matrix degradation; reduced lipid peroxidation limiting ROS-induced MMP-1 and MMP-3 upregulation | Potential attenuation of rosacea-associated erythema and inflammatory tissue damage through oxidative stress reduction; preservation of dermal collagen and elastin integrity may contribute to improved skin barrier function and reduced skin sensitivity; indirect mitigation of rosacea progression via anti-inflammatory and antioxidative pathways | [136] |
| O. vulgare essential oil | In vitro antimicrobial microdilution assays (96-well plates) against Gram-positive and Gram-negative bacteria and fungi (yeasts, molds, dermatophytes); MIC determination according to CLSI-adapted protocols | Carvacrol (66.89%), p-cymene (21.20%), and γ-terpinene | Bacteria: MIC ≈ 1.13 mg/mL (Gram-positive), MIC ≈ 0.34–1.13 mg/mL (Gram-negative); fungi: MIC ≈ 1.80 mg/mL (yeasts and molds), MIC ≈ 0.45 mg/mL (dermatophytes) | Disruption of microbial cytoplasmic membrane integrity via hydrophobic interaction of phenolic monoterpenes (carvacrol); increased membrane permeability, depolarization, and leakage of intracellular components | Potential reduction in rosacea-associated microbial burden (e.g., opportunistic skin bacteria and yeasts); indirect mitigation of inflammation and erythema by lowering microbe-induced immune activation; possible benefit in papulopustular rosacea through antimicrobial and barrier-supportive effects | [137] |
| O. vulgare essential oil | In vitro human keratinocyte (NCTC 2544) model; inflammatory stimulation with IFN-γ (200 U/mL) + histamine (10−4 M); scratch wound healing assay; ROS (H2DCFDA), RT-PCR, Western blot, immunofluorescence | Carvacrol (35.95%), thymol (25.02%), p-cymene (21.54%), linalool (4.26%), and γ-terpinene | Non-cytotoxic and effective concentration: 25 μg/mL (72 h); reference comparator: indomethacin 10 μM | Inhibition of pro-inflammatory signaling via downregulation of ICAM-1, iNOS, and COX-2; reduction in oxidative stress (ROS, 8-OHdG); modulation of ECM remodeling through suppression of MMP-1 and MMP-12; promotion of keratinocyte migration and controlled proliferation (PCNA modulation) | Potential attenuation of rosacea-associated inflammation and erythema by reducing oxidative stress and inflammatory mediators; support of epidermal barrier repair and tissue remodeling; possible benefit in papulopustular and erythematotelangiectatic rosacea through improved keratinocyte homeostasis and wound-healing capacity | [138] |
| O. vulgare essential oil | In vitro human dermal fibroblast disease model (BioMAP HDF3CGF) mimicking chronic skin inflammation and fibrosis; protein biomarker analysis (ELISA, multiplex immunoassays), antiproliferation assays, genome-wide gene expression (microarray, IPA pathway analysis) | Carvacrol (major constituent) | Non-cytotoxic, biologically active concentration: 0.0037% (v/v); treatment duration: 24–72 h depending on endpoint | Broad suppression of inflammatory and immune-related signaling (MCP-1, VCAM-1, ICAM-1, IP-10, I-TAC, MIG); inhibition of tissue remodeling and ECM degradation pathways (collagen I/III, MMP-1, PAI-1, TIMP-1/2, EGFR); immunomodulation via reduced M-CSF; global downregulation of inflammation-, fibrosis-, and cancer-related gene networks | Potential attenuation of rosacea-associated chronic inflammation and immune activation; reduction in dermal matrix degradation, contributing to erythema persistence and tissue damage; possible benefit in inflammatory and papulopustular rosacea phenotypes through modulation of fibroblast-driven inflammatory signaling and tissue remodeling | [139] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
İnal-Kılıçarslan, E.; Kartal, M.; Gören, H.Y.; Taşlıdere, N.; Küçük, Ö.S.; Akaslan, T.Ç.; Gören, A.C. Traditional Herbs in Anatolian Medicine for Rosacea: A Basis for Non-Steroidal Magistral Therapy. Processes 2026, 14, 424. https://doi.org/10.3390/pr14030424
İnal-Kılıçarslan E, Kartal M, Gören HY, Taşlıdere N, Küçük ÖS, Akaslan TÇ, Gören AC. Traditional Herbs in Anatolian Medicine for Rosacea: A Basis for Non-Steroidal Magistral Therapy. Processes. 2026; 14(3):424. https://doi.org/10.3390/pr14030424
Chicago/Turabian Styleİnal-Kılıçarslan, Ebrar, Murat Kartal, Hasan Yavuz Gören, Nazan Taşlıdere, Özlem Su Küçük, T. Çağdaş Akaslan, and Ahmet C. Gören. 2026. "Traditional Herbs in Anatolian Medicine for Rosacea: A Basis for Non-Steroidal Magistral Therapy" Processes 14, no. 3: 424. https://doi.org/10.3390/pr14030424
APA Styleİnal-Kılıçarslan, E., Kartal, M., Gören, H. Y., Taşlıdere, N., Küçük, Ö. S., Akaslan, T. Ç., & Gören, A. C. (2026). Traditional Herbs in Anatolian Medicine for Rosacea: A Basis for Non-Steroidal Magistral Therapy. Processes, 14(3), 424. https://doi.org/10.3390/pr14030424






