H2/CH4 Competitive Adsorption of LTA Zeolite: Effects of Cations, Si/Al Ratio, Adsorption Temperature, and Pressure
Abstract
1. Introduction
2. Modeling Method
2.1. Construction of LTA Framework
2.2. GCMC Simulation Details
| Fluid/Functional Group | Site | , Å | , K | , e | References |
|---|---|---|---|---|---|
| H2 | H | 2.96 | 36.7 | 0.468 | [28] |
| CH4 | C | 3.40 | 55.05 | −0.612 | [29] |
| H | 2.65 | 7.9 | 0.153 | ||
| Atoms of the LTA zeolite framework | Si | 2.30 | 22.0 | 1.5 | [30] |
| O | 3.30 | 53.0 | −0.75 | ||
| Al | 2.30 | 22.0 | 1.4 | ||
| H2-LTA | H-Si | 1.95 | 27.9 | 0.702 | [30,31] |
| H-Al | 1.29 | 27.9 | 0.655 | ||
| H-O | 1.593 | 50.1 | −0.351 | ||
| CH4-LTA | C-Si | 3.675 | 57.7 | +1.5 | [30,31] |
| C-O | 3.345 | 109.1 | −0.8 | ||
| C-Al | 3.015 | 57.7 | 1.4 | ||
| H-Si | 3.103 | 42.2 | 1.5 | ||
| H-O | 2.768 | 82.0 | −0.8 | ||
| H-Al | 2.438 | 42.2 | 1.4 |
3. Results and Discussion
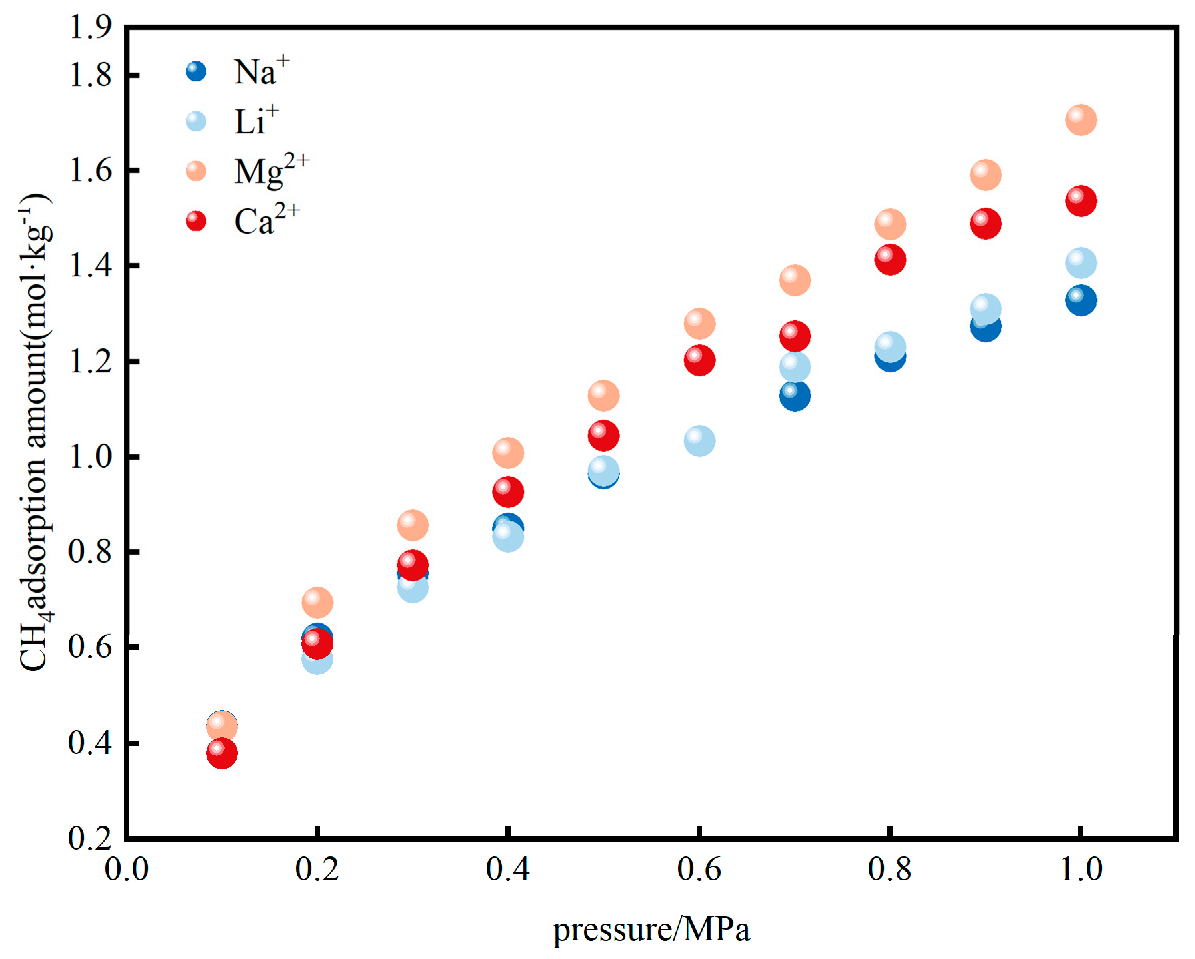
3.1. Synergistic Effect of Cation Charge Density and Radius
3.1.1. CH4 Adsorption Evolution with Different Cations
3.1.2. H2 Adsorption Evolution with Different Cations
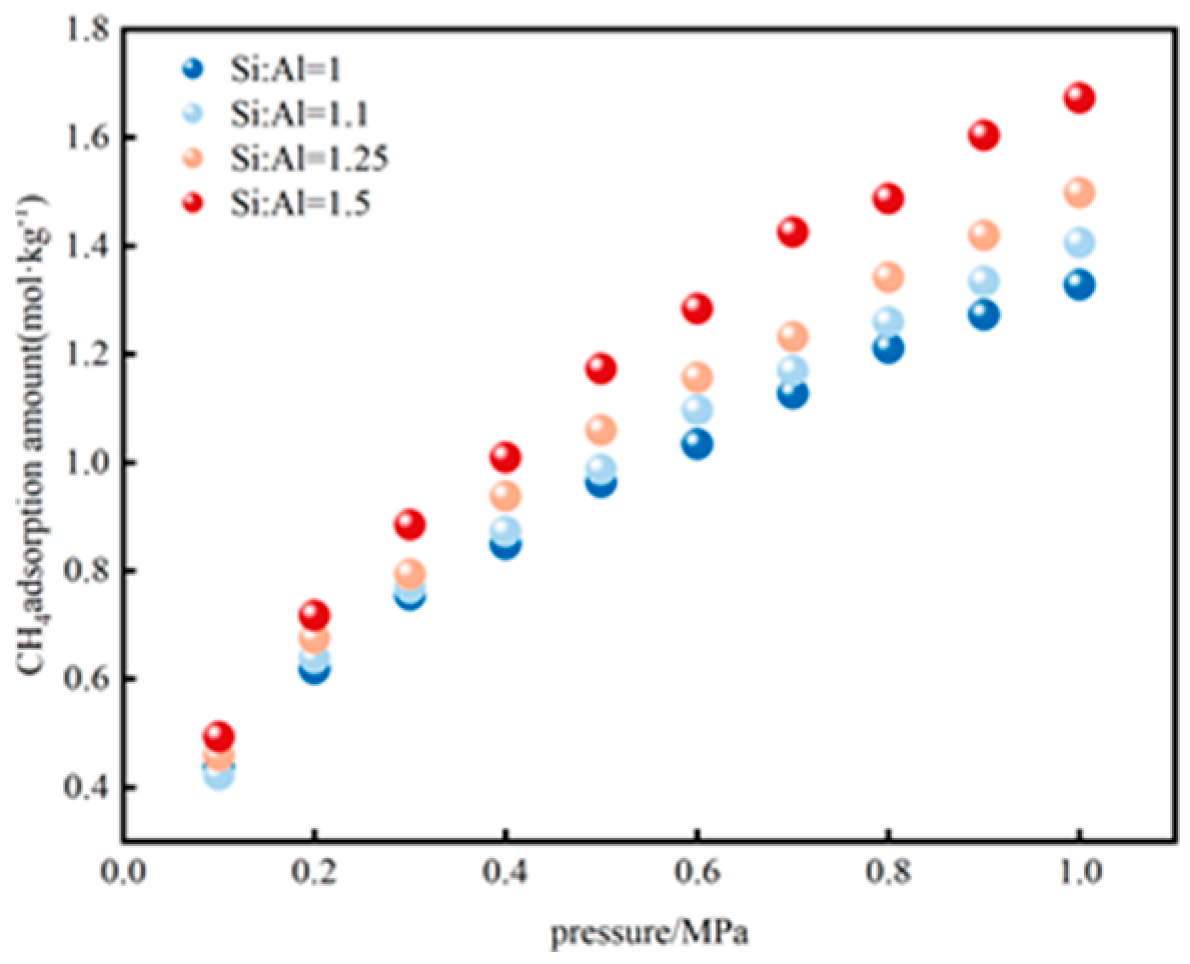
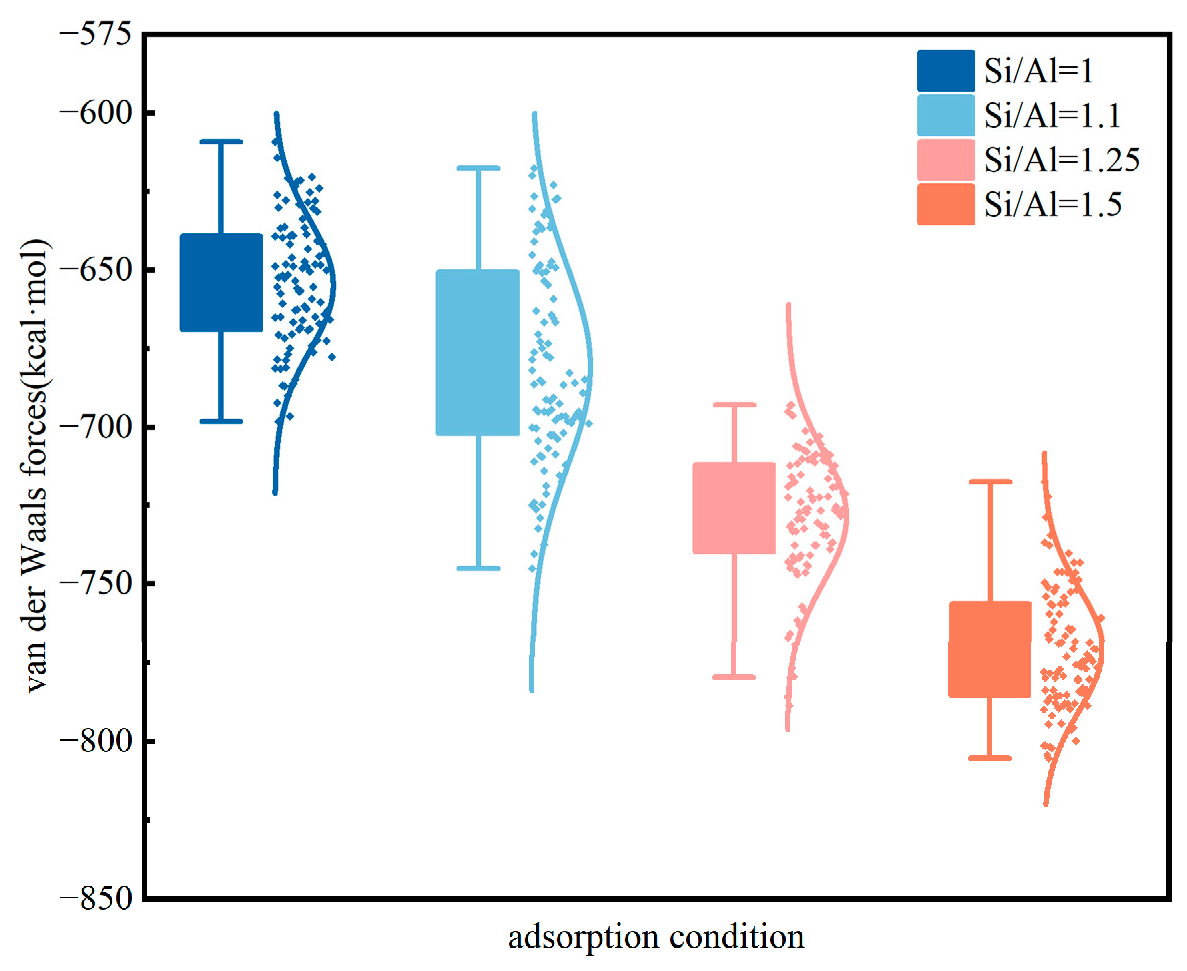
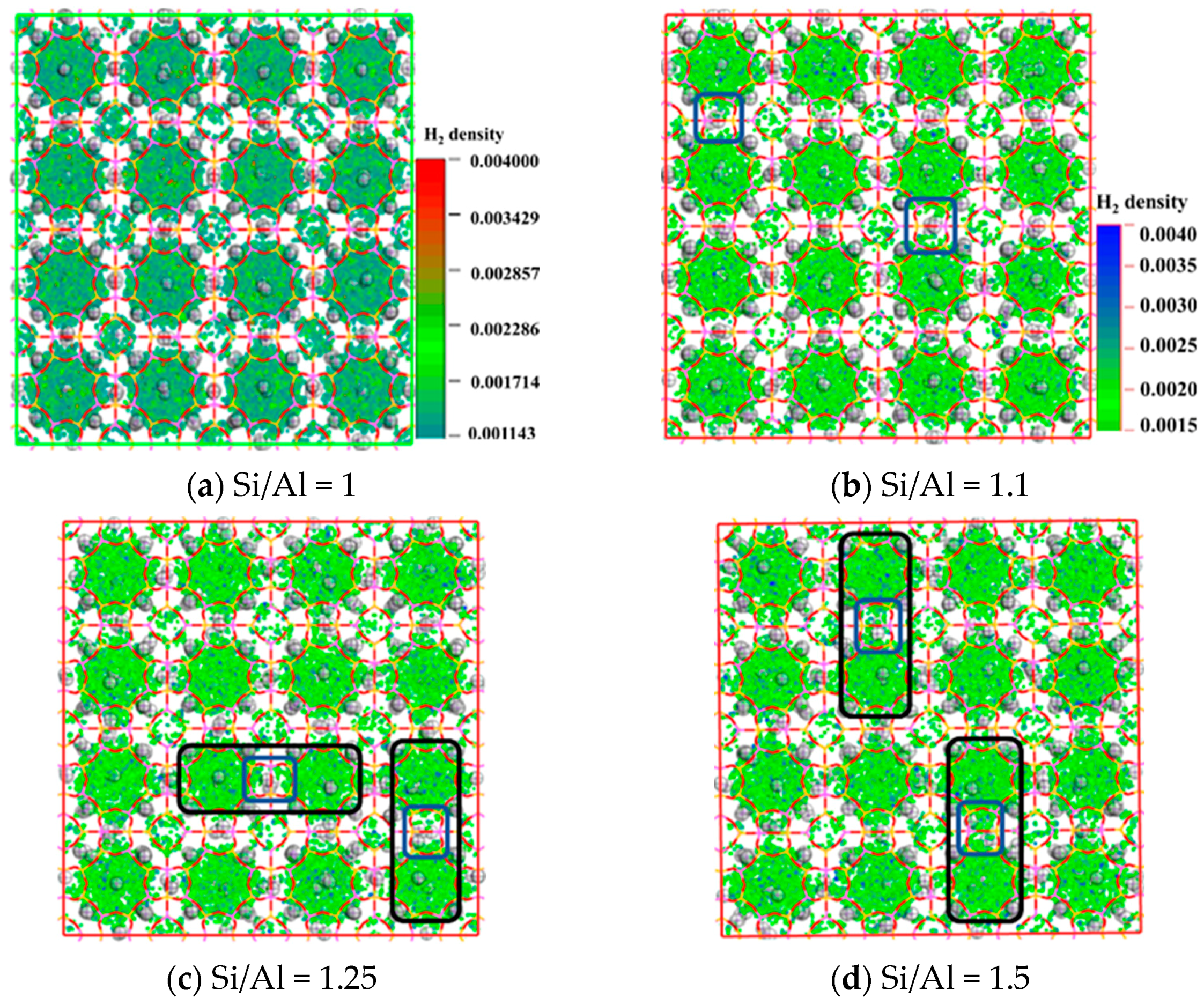
3.2. Effect of Si/Al Ratio on Adsorption Sites
3.2.1. CH4 Adsorption Evolution with Si/Al Ratios
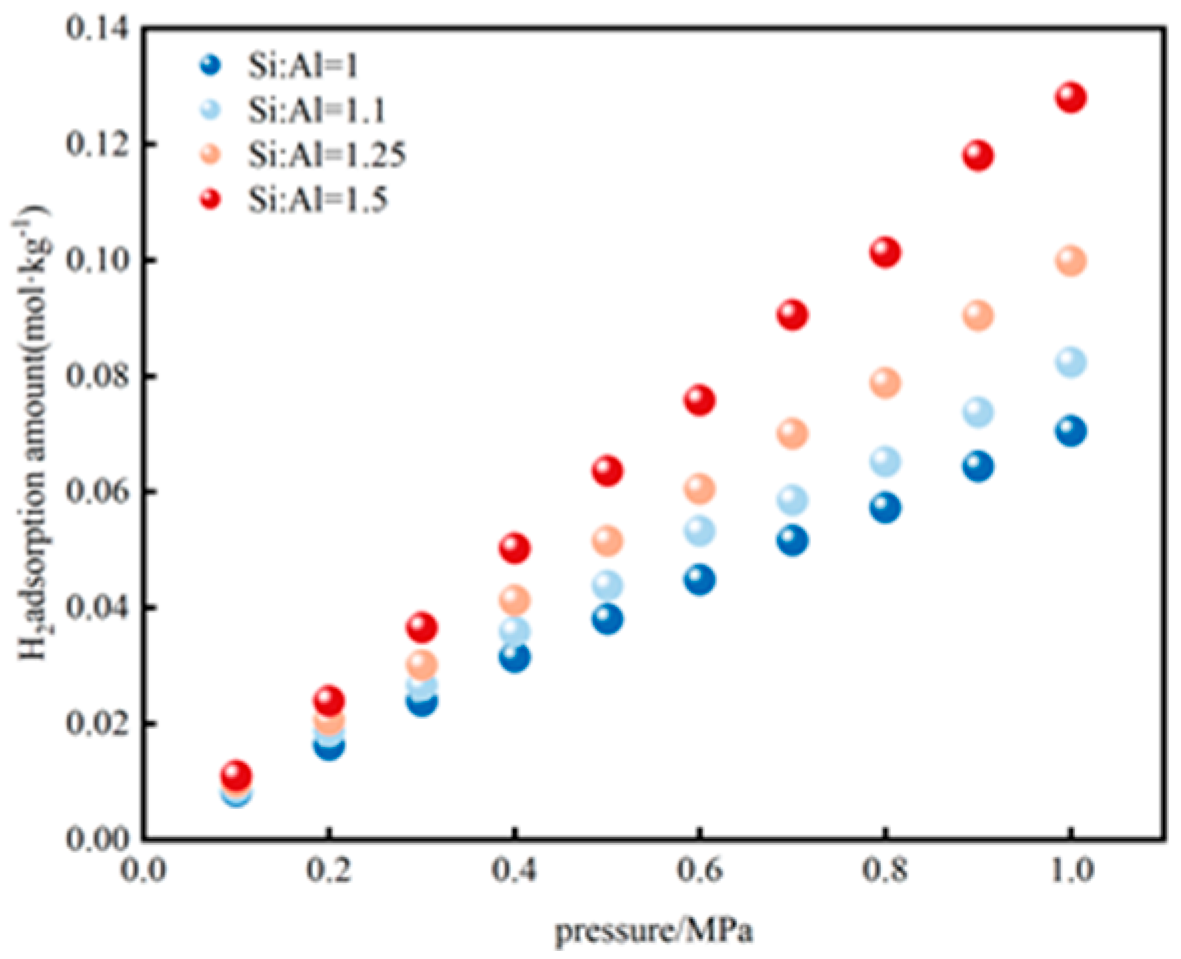
3.2.2. H2 Adsorption Evolution with Si/Al Ratios
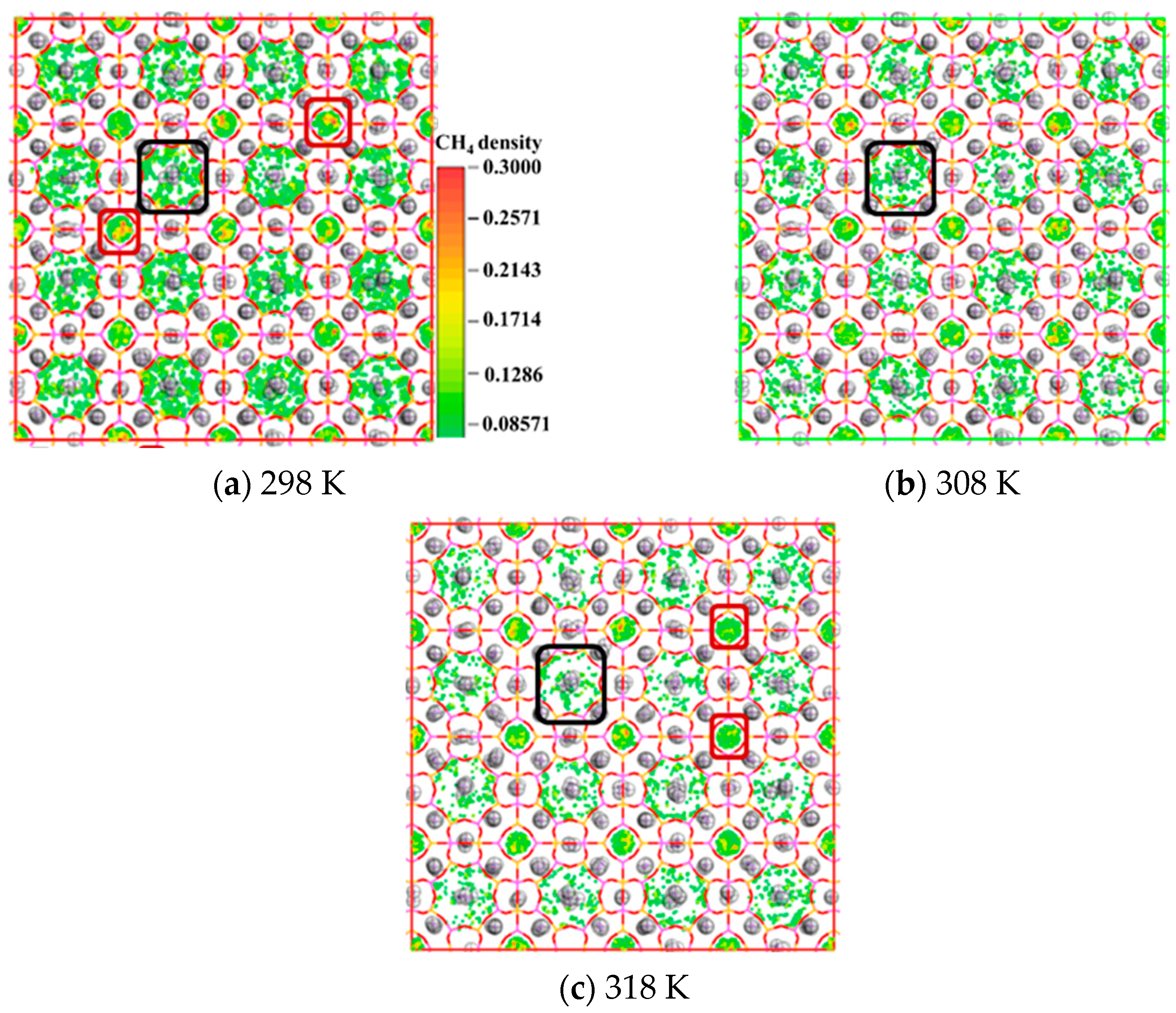
3.3. Attenuating Effect of Temperature Elevation on CH4 Adsorption
3.3.1. CH4 Adsorption Evolution with Temperature
3.3.2. H2 Adsorption Evolution with Temperature
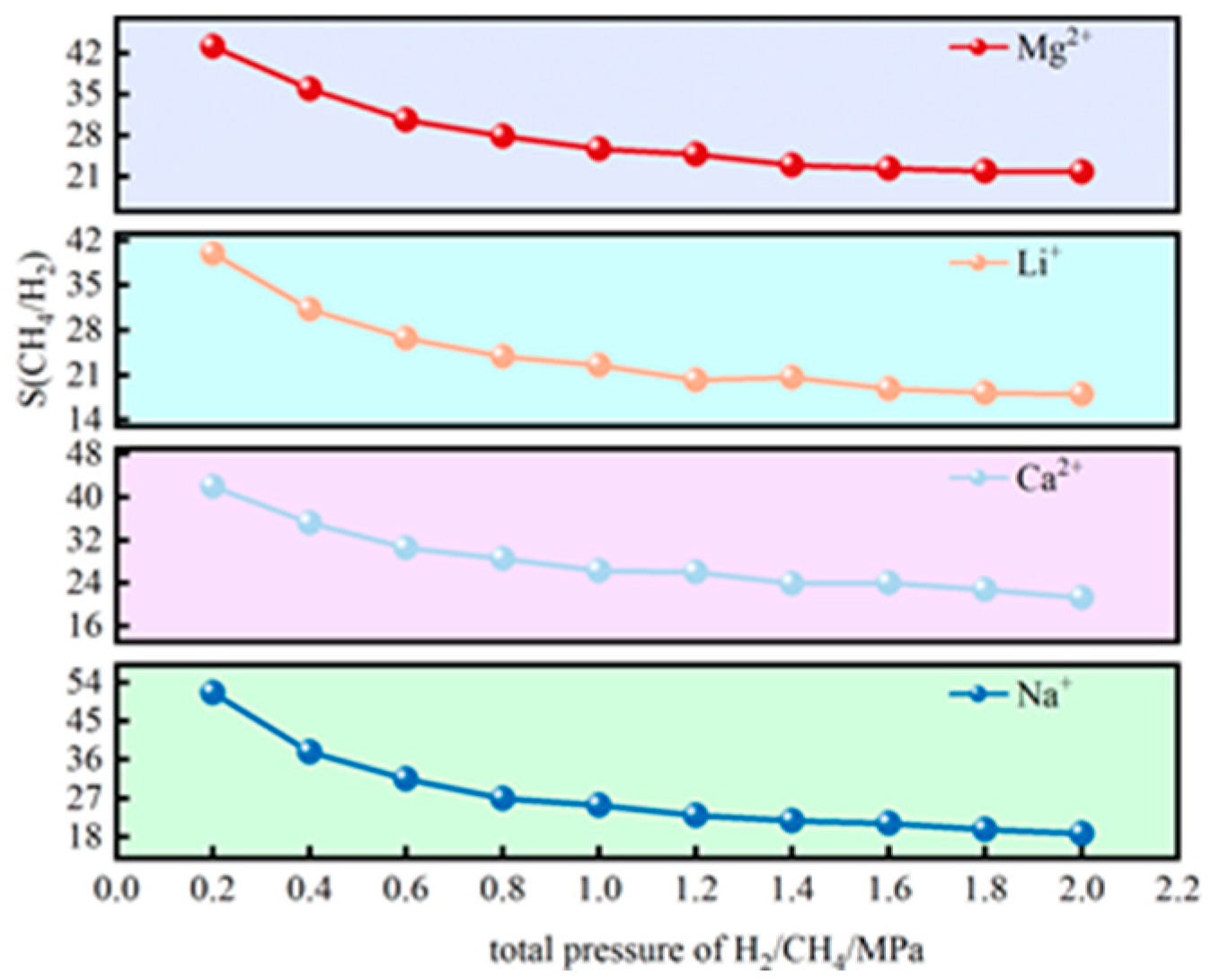
3.4. Microspore Filling of Hydrogen Under Pressure
3.4.1. CH4 Adsorption Evolution with Pressure
3.4.2. H2 Adsorption Evolution with Pressure
4. Conclusions
- (1)
- CH4 adsorbs preferentially in β-cage windows and eight-membered rings, forming high-density zones near cations and oxygen sites, whereas H2 distributes uniformly with no distinct adsorption sites.
- (2)
- High-valence, small-radius cations (e.g., Mg2+, Ca2+) enhance CH4 adsorption and diffusion by generating stronger local electric fields and reducing pore blockage compared to monovalent cations. H2 is less sensitive to cation charge but benefits from smaller cation radii, which increase accessible pore volume and thus H2 uptake.
- (3)
- Increasing the Si/Al ratio improves CH4 selectivity by reducing cation content and exposing more framework oxygen sites, especially in Si–O–Si environments, which strengthens CH4–framework interactions.
- (4)
- Temperature elevation weakens CH4 adsorption but promotes H2 diffusion into deeper pores. Higher pressure raises both uptakes, but H2 adsorption increases more, reducing CH4/H2 selectivity.
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Dehdari, L.; Burgers, I.; Xiao, P.; Li, K.G.; Singh, R.; Webley, P.A. Purification of hydrogen from natural gas/hydrogen pipeline mixtures. Sep. Purif. Technol. 2022, 282, 120094. [Google Scholar] [CrossRef]
- Zhang, N.; Bénard, P.; Chahine, R.; Yang, T.; Xiao, J. Optimization of pressure swing adsorption for hydrogen purification based on Box-Behnken design method. Int. J. Hydrogen Energy 2021, 46, 5403–5417. [Google Scholar] [CrossRef]
- Brea, P.; Delgado, J.A.; Agueda, V.I.; Gutierrez, P.; Uguina, M.A. Multicomponent adsorption of H2, CH4, CO and CO2 in zeolites NaX, CaX and MgX. Evaluation of performance in PSA cycles for hydrogen purification. Microporous Mesoporous Mater. 2019, 286, 187–198. [Google Scholar] [CrossRef]
- Epiepang, F.E.; Li, J.; Liu, Y.; Yang, R.T. Low-pressure performance evaluation of CO2, H2O and CH4 on Li-LSX as a superior adsorbent for air prepurification. Chem. Eng. Sci. 2016, 147, 100–108. [Google Scholar] [CrossRef]
- Yanez, M.; Relvas, F.; Ortiz, A.; Gorri, D.; Mendes, A.; Ortiz, I. PSA purification of waste hydrogen from ammonia plants to fuel cell grade. Sep. Purif. Technol. 2020, 240, 116334. [Google Scholar] [CrossRef]
- Lopes, F.V.S.; Grande, C.A.; Rodrigues, A.E. Activated carbon for hydrogen purification by pressure swing adsorption: Multicomponent breakthrough curves and PSA performance. Chem. Eng. Sci. 2011, 66, 303–317. [Google Scholar] [CrossRef]
- Lopes, F.V.S.; Grande, C.A.; Ribeiro, A.M.; Loureiro, J.M.; Evaggelos, O.; Nikolakis, V.; Rodrigues, A.E. Adsorption of H2; CO2; CH4, N2 and H2O in activated carbon and zeolite for hydrogen production. Sep. Sci. Technol. 2009, 44, 1045–1073. [Google Scholar] [CrossRef]
- Pérez-Botella, E.; Valencia, S.; Rey, F. Zeolites in Adsorption Processes: State of the Art and Future Prospects. Chem. Rev. 2022, 122, 17647–17695. [Google Scholar] [CrossRef]
- Li, C.; Luo, H.; Yuan, Y.; Tong, L.; Chen, B.; Yang, T.; Yuan, C.; Chahine, R.; Xiao, J. Equilibrium and dynamic adsorption characteristics of zeolite 5A, LiX, 13X and MOF UTSA-16 adsorbents for hydrogen purification. Int. J. Hydrogen Energy 2025, 140, 889–899. [Google Scholar] [CrossRef]
- Shi, W.; Yang, H.; Shen, Y.; Fu, Q.; Zhang, D.; Fu, B. Two-stage PSA/VSA to produce H2 with CO2 capture via steam methane reforming (SMR). Int. J. Hydrogen Energy 2018, 43, 19057–19074. [Google Scholar] [CrossRef]
- Delgado, J.A.; Agueda, V.I.; Uguina, M.A.; Sotelo, J.L.; Brea, P.; Grande, C.A. Adsorption and diffusion of H2/CO, CH4, and CO2 in BPL activated carbon and 13X zeolite: Evaluation of performance in pressure swing adsorption hydrogen purification by simulation. Ind. Eng. Chem. Res. 2014, 53, 15414–15426. [Google Scholar] [CrossRef]
- Gao, S.; Peng, H.; Song, B.; Zhang, J.X.; Wu, W.X.; Vaughan, J.; Zardo, P.; Vogrin, J.; Tulloch, S.; Zhu, Z.H. Synthesis of zeolites from low-cost feeds and its sustainable environmental applications. J. Environ. Chem. Eng. 2023, 11, 108995. [Google Scholar] [CrossRef]
- Jeong, S.R.; Kim, K.-M.; Lee, C.-H. Adsorption equilibria and kinetics of CO2, CO, CH4, N2, and H2 on zeolite 5A up to 1.0 MPa. Chem. Eng. J. 2025, 525, 170014. [Google Scholar] [CrossRef]
- Liu, B.; Yu, X.; Shi, W.; Shen, Y.; Zhang, D.; Tang, Z. Two-stage VSA/PSA for capturing carbon dioxide (CO2) and producing hydrogen (H-2) from steam-methane reforming gas. Int. J. Hydrogen Energy 2020, 45, 24870–24882. [Google Scholar] [CrossRef]
- Yuan, J.-P.; Liu, X.-Y.; Li, X.-D.; Yu, J.-X. Molecular simulation for adsorption and separation of CH4/H2 in zeolites. Acta Phys. Sin. 2021, 70, 156801. [Google Scholar] [CrossRef]
- Liu, Y.; Xu, J.; Jin, L.; Fang, Y.; Hu, H. Synthesis and modification of zeolite NaA adsorbents for separation of hydrogen and methane. Asia Pacific J. Chem. Eng. 2009, 4, 666–671. [Google Scholar] [CrossRef]
- Liu, C. The Study on Process Technology of Hydrogen Purification in a PSA Plant. Master’s Thesis, 2012. [Google Scholar]
- Karki, S.; Chakraborty, S.N. Hydrogen adsorption in Si-LTA and LTA-4A zeolites: A Gibbs Ensemble Monte Carlo simulation study. Mater. Chem. Phys. 2024, 313, 128722. [Google Scholar] [CrossRef]
- Lao, K.; Rabideau, B.D. Effect of Pre-Adsorbed Species on High-Pressure Adsorption of Methane in Zeolite 5A Using Grand Canonical Monte Carlo (GCMC) Simulations; University of South Alabama: Mobile, AL, USA, 2025; Available online: https://jagworks.southalabama.edu/southalabama-shgrf-posters/21 (accessed on 11 January 2026).
- Tao, Z.; Tian, Y.; Hanif, A.; Chan, V.; Gu, Q.; Shang, J. Metal cation-exchanged LTA zeolites for CO2/N2 and CO2/CH4 separation: The roles of gas-framework and gas-cation interactions. Carbon Capture Sci. Technol. 2023, 8, 100126. [Google Scholar] [CrossRef]
- Xu, Z.; Stebbins, J. Oxygen sites in the zeolite stilbite: A comparison of static, MAS, VAS, DAS and triple quantum MAS NMR techniques. Solid State Nucl. Magn. Reson. 1998, 11, 243–251. [Google Scholar] [CrossRef]
- Kumar, K.V.; Müller, E.A.; Rodríguez-Reinoso, F. Effect of Pore Morphology on the Adsorption of Methane/Hydrogen Mixtures on Carbon Micropores. J. Phys. Chem. C 2012, 116, 11820–11829. [Google Scholar] [CrossRef]
- Gopalsamy, K.; Subramanian, V.J.N.J.O.C. Carbon flakes based metal organic frameworks for H2, CH4 and CO2 gas storage: A GCMC simulation study. New J. Chem. 2018, 42, 4240–4250. [Google Scholar] [CrossRef]
- Dasgupta, T.; Punnathanam, S.N.; Ayappa, K. Effect of functional groups on separating carbon dioxide from CO2/N2 gas mixtures using edge functionalized graphene nanoribbons. Chem. Eng. Sci. 2015, 121, 279–291. [Google Scholar] [CrossRef]
- Herrera, L.; Fan, C.; Do, D.; Nicholson, D. Novel method to determine accessible volume, area, and pore size distribution of activated carbon. Ind. Eng. Chem. Res. 2011, 50, 4150–4160. [Google Scholar] [CrossRef]
- Yang, R.T. Adsorbents: Fundamentals and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2003. [Google Scholar]
- Bari, A.; Hayder, A.; Shua, A.K.; Yunus, R.B.M. Equilibrium Adsorption of Hydrogen and Methane on 5A Molecular Sieve. Am. J. Eng. Appl. Sci. 2008, 1, 157–160. [Google Scholar] [CrossRef]
- Mert, H.; Deniz, C.U.; Baykasoglu, C. Adsorptive separation of CH4, H2, CO2, and N2 using fullerene pillared graphene nanocomposites: Insights from molecular simulations. J. Mol. Model. 2023, 29, 1–12. [Google Scholar] [CrossRef]
- Wang, S.; Lu, L.; Wu, D.; Lu, X.; Cao, W.; Yang, T.; Zhu, Y. Molecular Simulation Study of the Adsorption and Diffusion of a Mixture of CO2/CH4 in Activated Carbon: Effect of Textural Properties and Surface Chemistry. J. Chem. Eng. Data 2016, 61, 4139–4147. [Google Scholar] [CrossRef]
- Rappe, A.K.; Casewit, C.J.; Colwell, K.S.; Goddard, W.A.; Skid, W.M. UFF, a Full Periodic Table Force Field for Molecular Mechanics and Molecular Dynamics Simulations. J. Am. Chem. Soc. 1992, 114, 10024–10035. [Google Scholar] [CrossRef]
- Martin, M.G.; Siepmann, J.I. Transferable potentials for phase equilibria. 1. United-atom description of n-alkanes. J. Phys. Chem. B 1998, 102, 2569–2577. [Google Scholar] [CrossRef]
- Travis, K.P.; Gubbins, K.E. Combined diffusive and viscous transport of methane in a carbon slit pore. Mol. Simul. 2000, 25, 209–227. [Google Scholar] [CrossRef]
- Zhou, J.; Wang, W. Adsorption and diffusion of supercritical carbon dioxide in slit pores. Langmuir 2000, 16, 8063–8070. [Google Scholar] [CrossRef]
- Golipour, H.; Mokhtarani, B.; Mafi, M.; Khadivi, M.; Godini, H.R. Systematic measurements of CH4 and CO2 adsorption isotherms on cation-exchanged zeolites 13X. J. Chem. Eng. Data 2019, 64, 4412–4423. [Google Scholar] [CrossRef]
- García-Pérez, E.; Dubbeldam, D.; Maesen, T.L.M.; Calero, S. Influence of Cation Na/Ca Ratio on Adsorption in LTA 5A: A Systematic Molecular Simulation Study of Alkane Chain Length. J. Phys. Chem. B 2006, 110, 23968–23976. [Google Scholar] [CrossRef] [PubMed]
- Shannon, R.D. Revised effective ionic radii and systematic studies of interatomic distances in halides and chalcogenides. Acta Cryst. 1976, A32, 751–766. [Google Scholar] [CrossRef]
- Daouli, A.; Rey, J.; Lahrar, E.H.; Valtchev, V.; Badawi, M.; Guillet-Nicolas, R. Ab Initio screening of divalent cations for CH4, CO2, H2, and N2 separations in chabazite zeolite. Langmuir 2023, 39, 15962–15973. [Google Scholar] [CrossRef] [PubMed]
- Paoli, H.; Corradi-Marchesi, E.; Rebours, B.; Ducreux, O.; Jobic, H. Long-chain paraffins adsorption over 5A zeolites. Stud. Surf. Sci. Catal. Today 2004, 154, 1950–1956. [Google Scholar] [CrossRef]
- Ferrari, A.M.; Neyman, K.M.; Huber, S.; Knözinger, H.; Rösch, N. Density functional study of methane interaction with alkali and alkaline-earth metal cations in zeolites. Langmuir 1998, 14, 5559–5567. [Google Scholar] [CrossRef]
- Ahn, S.H.; Wang, Q.; Wang, Y.; Chu, Y.; Deng, F.; Hong, S.B. Identifying Crystallographically Different Si−OH−Al Brønsted Acid Sites in LTA Zeolites. Angew. Chem. Int. Ed. 2022, 61, e202203603. [Google Scholar] [CrossRef]
- Barrer, R.M. Zeolites and Clay Minerals as Sorbents and Molecular Sieves; Academic Press: Cambridge, MA, USA, 1978. [Google Scholar]
- Radhakrishnan, S.; Lejaegere, C.; Duerinckx, K.; Lo, W.-S.; Morais, A.F.; Dom, D.; Chandran, C.V.; Hermans, I.; Martens, J.A.; Breynaert, E. Hydrogen bonding to oxygen in siloxane bonds drives liquid phase adsorption of primary alcohols in high-silica zeolites. Mater. Horiz. 2023, 10, 3702–3711. [Google Scholar] [CrossRef]
- Liu, H.; Yuan, D.; Liu, G.; Xing, J.; Liu, Z.; Xu, Y. Oxygen-selective adsorption on high-silica LTA zeolite. Chem. Commun. 2020, 56, 11130–11133. [Google Scholar] [CrossRef]
- Cavenati, S.; Grande, C.A.; Rodrigues, A.E. Adsorption equilibrium of methane, carbon dioxide, and nitrogen on zeolite 13X at high pressures. J. Chem. Eng. Data 2004, 49, 1095–1101. [Google Scholar] [CrossRef]
- Choi, B.U.; Choi, D.K.; Lee, Y.W.; Lee, B.K.; Kim, S.H. Adsorption equlibria of methane, ethane, ethylene, nitrogen, and hydrogen onto cctivated carbon. J. Chem. Eng. Data 2003, 48, 603–607. [Google Scholar] [CrossRef]
- Price, L.A.; Jones, Z.; Nearchou, A.; Stenning, G.; Nye, D.; Sartbaeva, A. The Effect of Cation Exchange on the Pore Geometry of Zeolite L. AppliedChem 2022, 2, 149–159. [Google Scholar] [CrossRef]
- Groen, J.C.; Bach, T.; Ziese, U.; Donk, A.M.P.-V.; De Jong, K.P.; Moulijn, J.A.; Pérez-Ramírez, J. Creation of hollow zeolite architectures by controlled desilication of Al-zoned ZSM-5 crystals. J. Am. Chem. Soc. 2005, 127, 10792–10793. [Google Scholar] [CrossRef]
- Yamada, T.; Johkan, K.I.; Okuhara, T. Micropore size distribution by argon porosimetry for cesium hydrogen salts of 12-tungstophosphoric acid. Microporous Mesoporous Mater. 1998, 26, 109–115. [Google Scholar] [CrossRef]





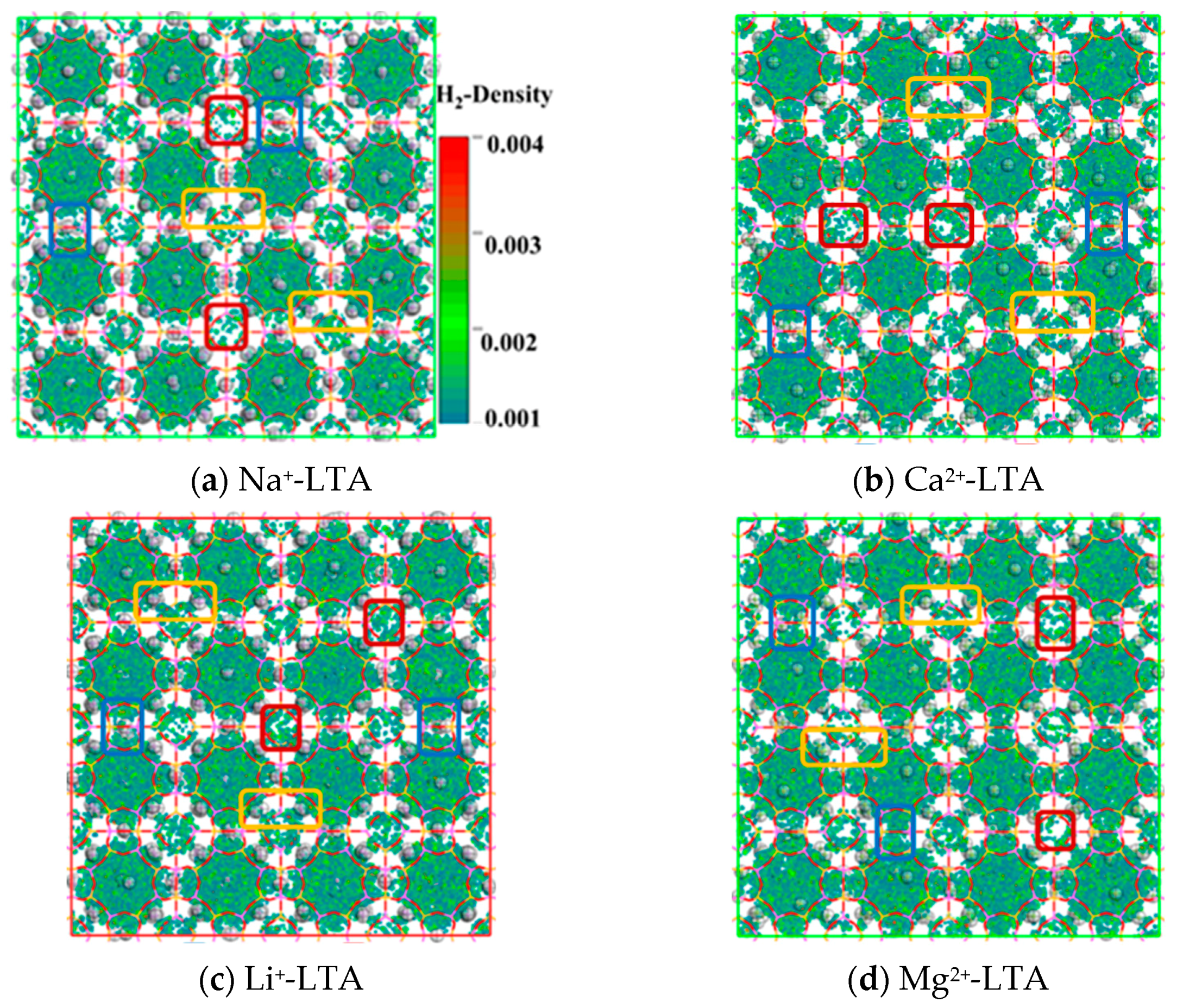







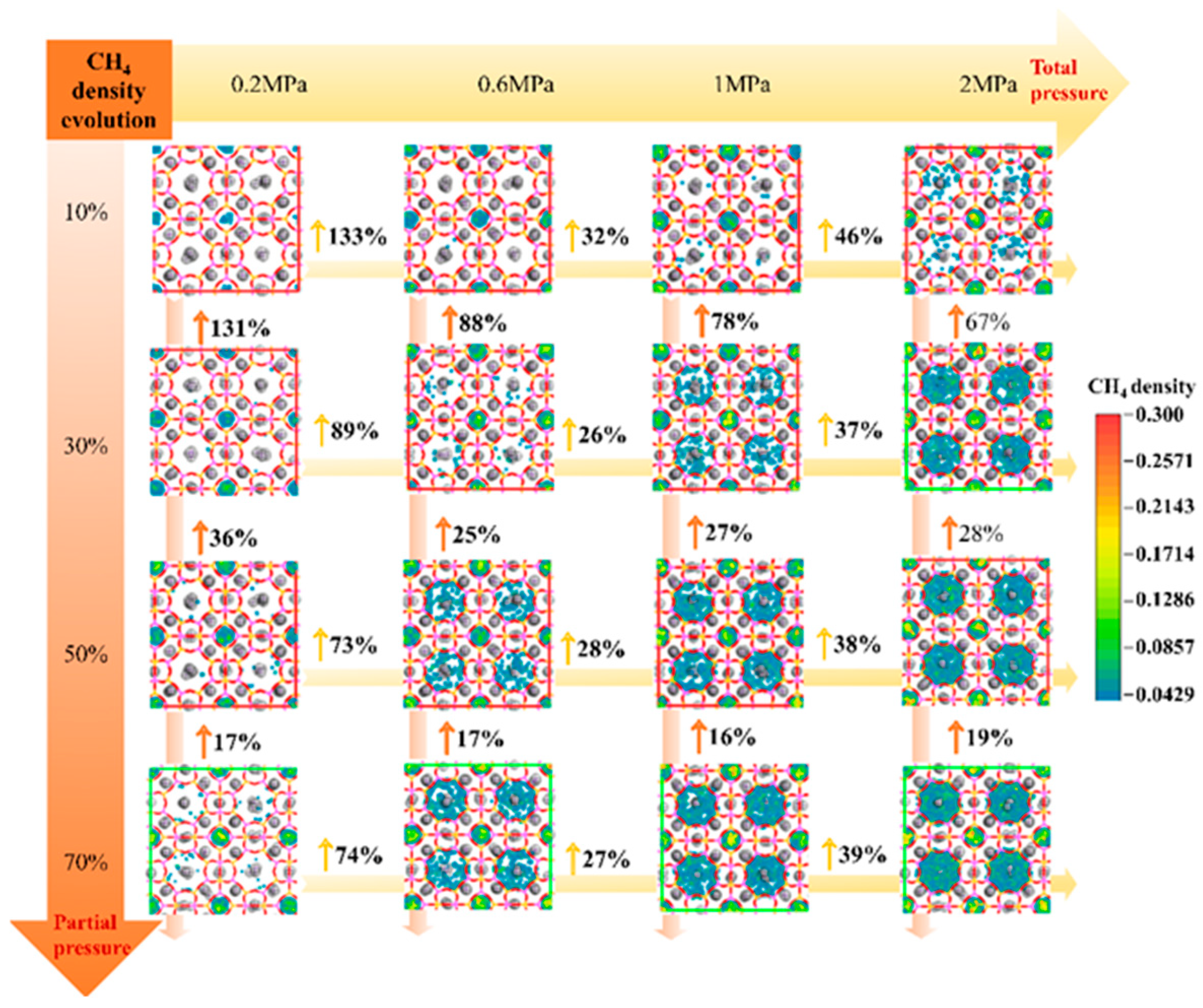
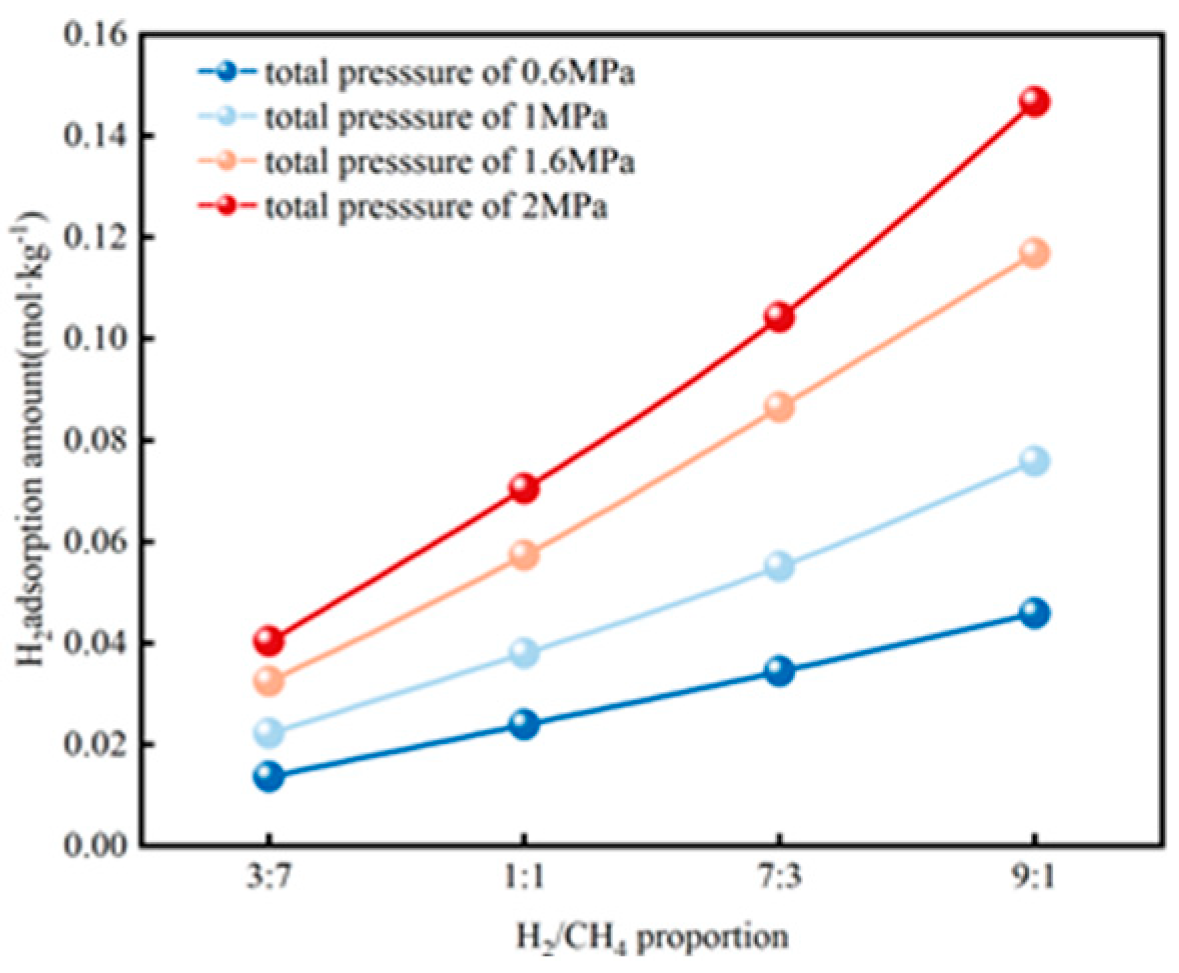
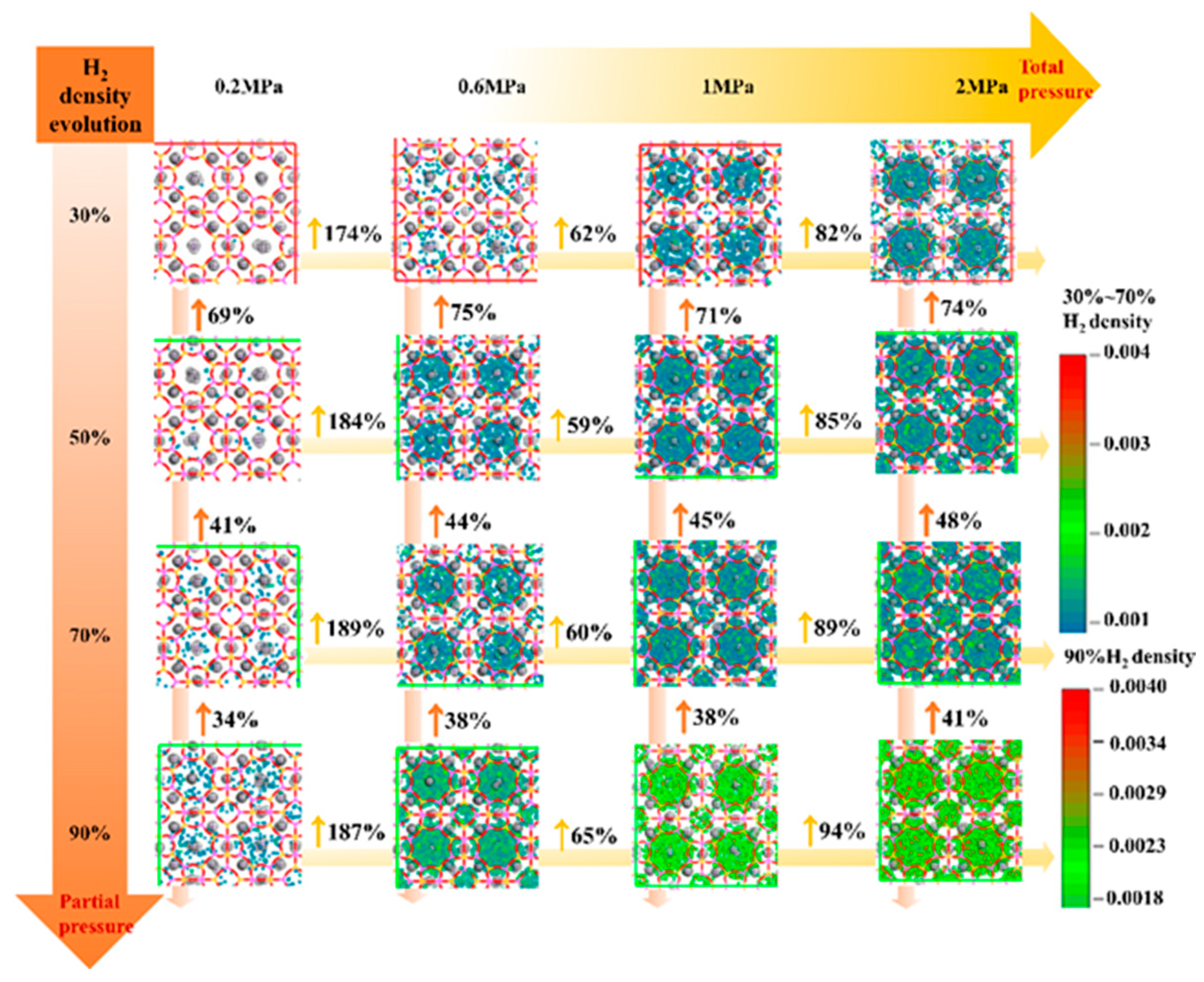

| Adsorption Density of CH4 (Molecules/Å3) | Diffusion Coefficient of CH4 (cm2/s) | ||
|---|---|---|---|
| Maximum | Average | ||
| Na+-LTA | (6.0681 ± 0.0012) × 10−1 | (1.1895 ± 0.0011) × 10−3 | (1.5661 ± 0.0004) × 10−6 |
| Li+-LTA | (6.7756 ± 0.0008) × 10−1 | (1.2594 ± 0.0005) × 10−3 | (1.6938 ± 0.0006) × 10−6 |
| Ca2+-LTA | (9.2500 ± 0.0010) × 10−1 | (1.5276 ± 0.0009) × 10−3 | (2.6228 ± 0.0007) × 10−6 |
| Mg2+-LTA | 1.2516 ± 0.0007 | (1.3750 ± 0.0004) × 10−3 | (3.6474 ± 0.0005) × 10−6 |
| Average Adsorption Density (Molecules/Å3) | Diffusion Coefficient (cm2/s) | |
|---|---|---|
| Na+-LTA | (6.3100 ± 0.0005) × 10−5 | (4.7939 ± 0.0006) × 10−5 |
| Li+-LTA | (6.9750 ± 0.0013) × 10−5 | (4.2885 ± 0.0005) × 10−4 |
| Ca2+-LTA | (6.4500 ± 0.0008) × 10−5 | (2.9366 ± 0.0007) × 10−4 |
| Mg2+-LTA | (6.9980 ± 0.0006) × 10−5 | (4.8540 ± 0.0004) × 10−4 |
| Adsorption Density of CH4 (Molecules/Å3) | Diffusion Coefficient of CH4 (cm2/s) | ||
|---|---|---|---|
| Maximum | Average | ||
| Si/Al = 1 | (6.0681 ± 0.0010) × 10−1 | (1.1895 ± 0.0005) × 10−3 | (1.5661 ± 0.0009) × 10−6 |
| Si/Al = 1.1 | (7.5072 ± 0.0008) × 10−1 | (1.2768 ± 0.0004) × 10−3 | (1.8647 ± 0.0004) × 10−6 |
| Si/Al = 1.25 | (8.1268 ± 0.0005) × 10−1 | (1.3467 ± 0.0003) × 10−3 | (2.2012 ± 0.0004) × 10−6 |
| Si/Al = 1.5 | (8.9389 ± 0.0007) × 10−1 | (1.4833 ± 0.0006) × 10−3 | (3.3467 ± 0.0010) × 10−6 |
| Diffusion Coefficient (cm2/s) | Average Adsorption Density (Molecules/Å3) | Maximum Adsorption Density (Molecules/Å3) | |
|---|---|---|---|
| Si/Al = 1 | (4.7939 ± 0.0011) × 10−5 | (6.3121 ± 0.0008) × 10−5 | (1.7189 ± 0.0004) × 10−2 |
| Si/Al = 1.1 | (1.8564 ± 0.0012) × 10−4 | (6.3835 ± 0.0005) × 10−5 | (1.8192 ± 0.0010) × 10−2 |
| Si/Al = 1.25 | (1.9634 ± 0.0013) × 10−4 | (6.4501 ± 0.0004) × 10−5 | (1.9791 ± 0.0006) × 10−2 |
| Si/Al = 1.5 | (4.3781 ± 0.0009) × 10−4 | (6.5543 ± 0.0009) × 10−5 | (2.1986 ± 0.0008) × 10−2 |
| Diffusion Coefficient (cm2/s) | Average Adsorption Density (Molecules/Å3) | Maximum Adsorption Density (Molecules/Å3) | |
|---|---|---|---|
| 298 K | (1.5804 ± 0.0011) × 10−6 | (1.1895 ± 0.0006) × 10−3 | (6.2123 ± 0.0015) × 10−1 |
| 308 K | (1.5692 ± 0.0009) × 10−6 | (1.1085 ± 0.0005) × 10−3 | (6.0681 ± 0.0008) × 10−1 |
| 318 K | (1.5661 ± 0.0012) × 10−6 | (1.0083 ± 0.0005) × 10−3 | (5.3326 ± 0.0006) × 10−1 |
| Diffusion Coefficient (cm2/s) | Average Adsorption Density (Molecules/Å3) | Maximum Adsorption Density (Molecules/Å3) | |
|---|---|---|---|
| 298 K | (6.7491 ± 0.0015) × 10−5 | (6.3114 ± 0.0004) × 10−5 | (1.7189 ± 0.0008) × 10−2 |
| 308 K | (5.2229 ± 0.0008) × 10−5 | (6.3461 ± 0.0006) × 10−5 | (1.7659 ± 0.0010) × 10−2 |
| 318 K | (4.7939 ± 0.0010) × 10−5 | (6.4173 ± 0.0003) × 10−5 | (1.8433 ± 0.0014) × 10−2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Zhang, X.; Tang, J.; Liu, H. H2/CH4 Competitive Adsorption of LTA Zeolite: Effects of Cations, Si/Al Ratio, Adsorption Temperature, and Pressure. Processes 2026, 14, 387. https://doi.org/10.3390/pr14020387
Zhang X, Tang J, Liu H. H2/CH4 Competitive Adsorption of LTA Zeolite: Effects of Cations, Si/Al Ratio, Adsorption Temperature, and Pressure. Processes. 2026; 14(2):387. https://doi.org/10.3390/pr14020387
Chicago/Turabian StyleZhang, Xue, Jianfeng Tang, and Hui Liu. 2026. "H2/CH4 Competitive Adsorption of LTA Zeolite: Effects of Cations, Si/Al Ratio, Adsorption Temperature, and Pressure" Processes 14, no. 2: 387. https://doi.org/10.3390/pr14020387
APA StyleZhang, X., Tang, J., & Liu, H. (2026). H2/CH4 Competitive Adsorption of LTA Zeolite: Effects of Cations, Si/Al Ratio, Adsorption Temperature, and Pressure. Processes, 14(2), 387. https://doi.org/10.3390/pr14020387






