Feasibility of Basalt Fiber Felt as Biocarrier for Rural Domestic Wastewater Treatment: Performance and Microbial Community Analysis
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Biofilter Reactor Design
2.3. Reactor Startup and Operation
2.4. Chemical Analyses
2.5. Microbial Analyses
3. Results and Discussion
3.1. Continuous-Flow Performance
3.1.1. COD Removal Performance Under Continuous-Flow Conditions
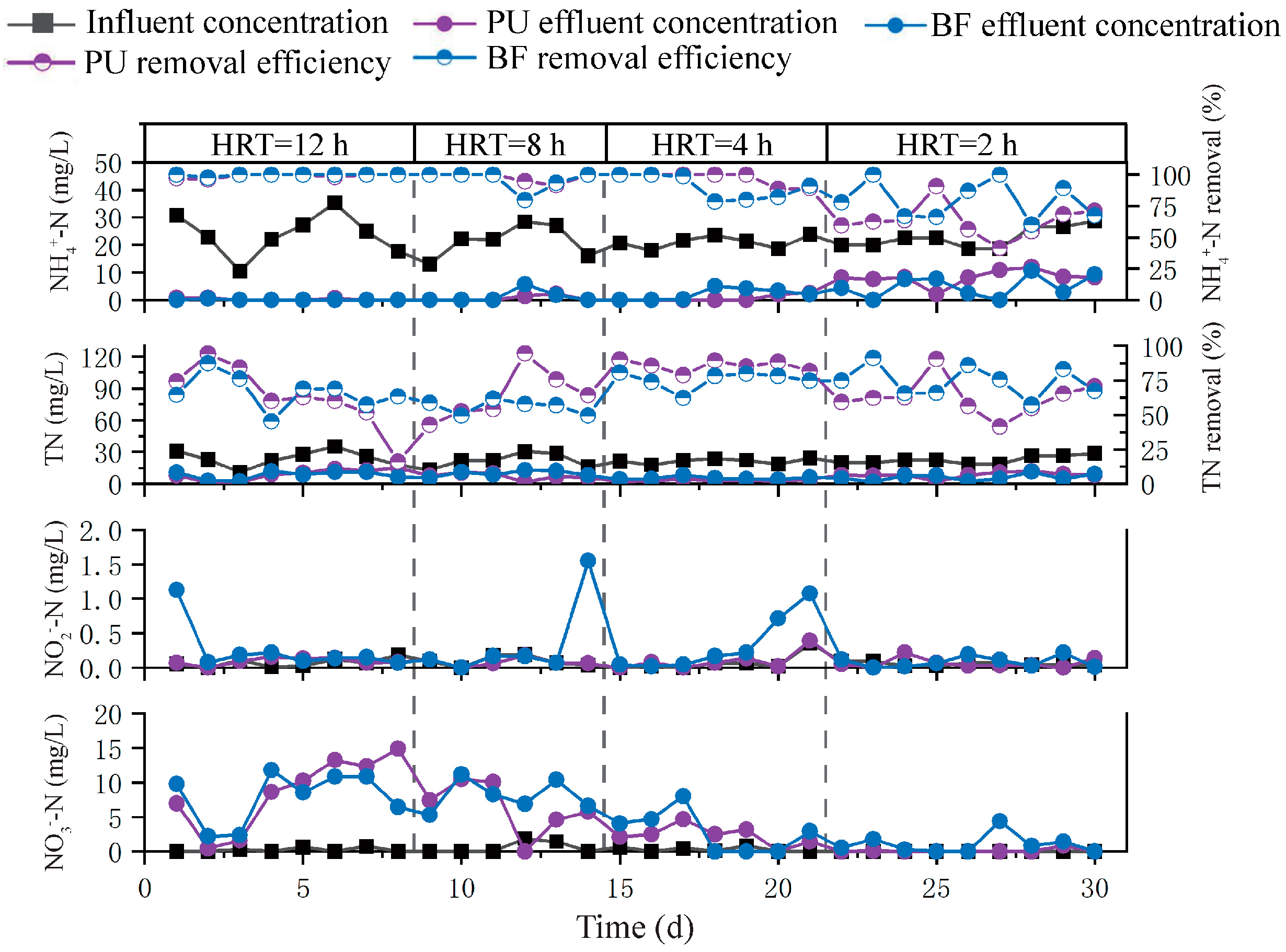
3.1.2. Nitrogen Removal Performance Under Continuous-Flow Conditions
3.2. Intermittent-Flow Performance
3.2.1. COD Removal Performance Under Intermittent-Flow Conditions
3.2.2. Nitrogen Removal Performance Under Intermittent-Flow Conditions
3.3. Microbial Community Succession
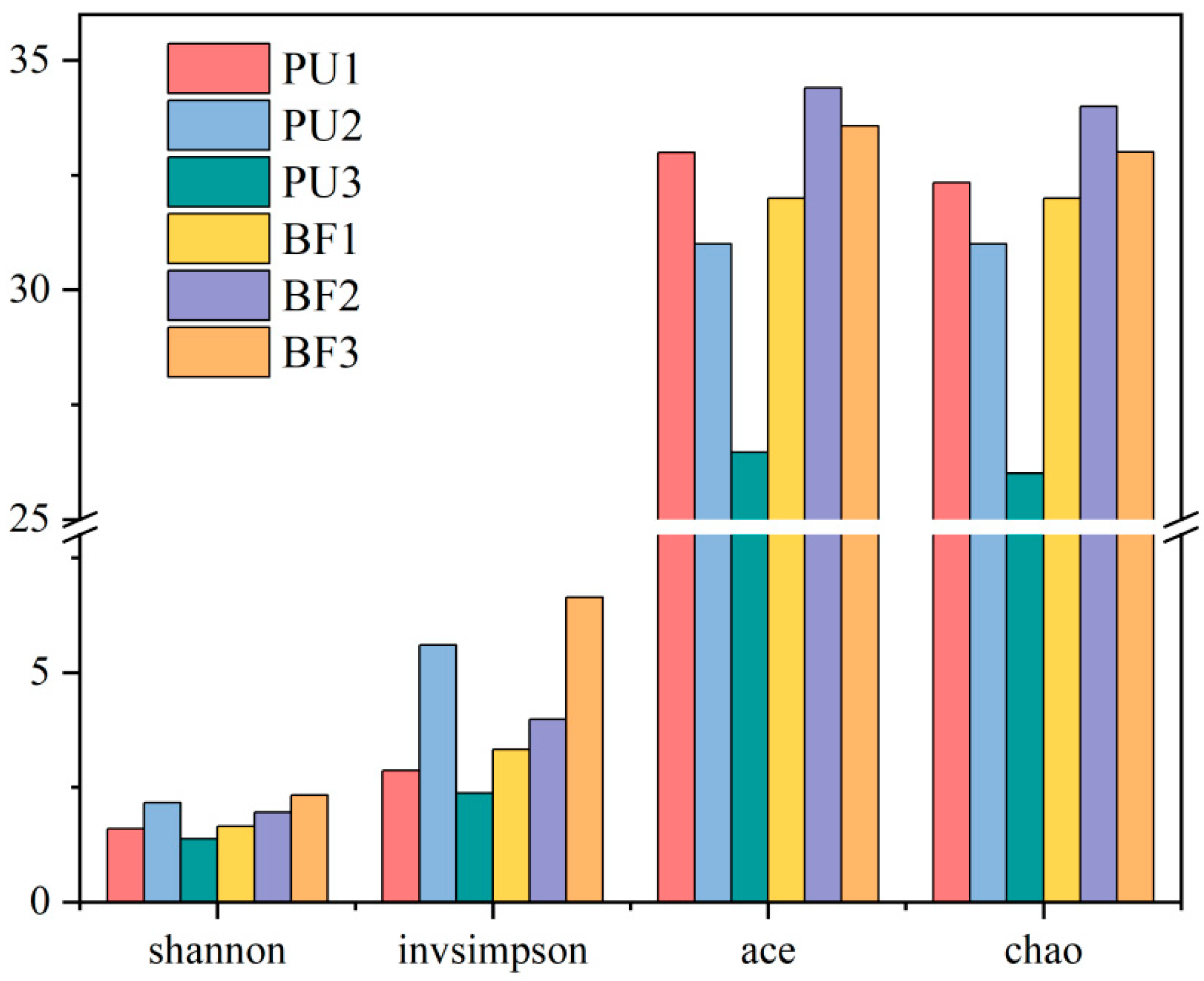
3.3.1. Microbial Community Diversity
3.3.2. Microbial Community Composition
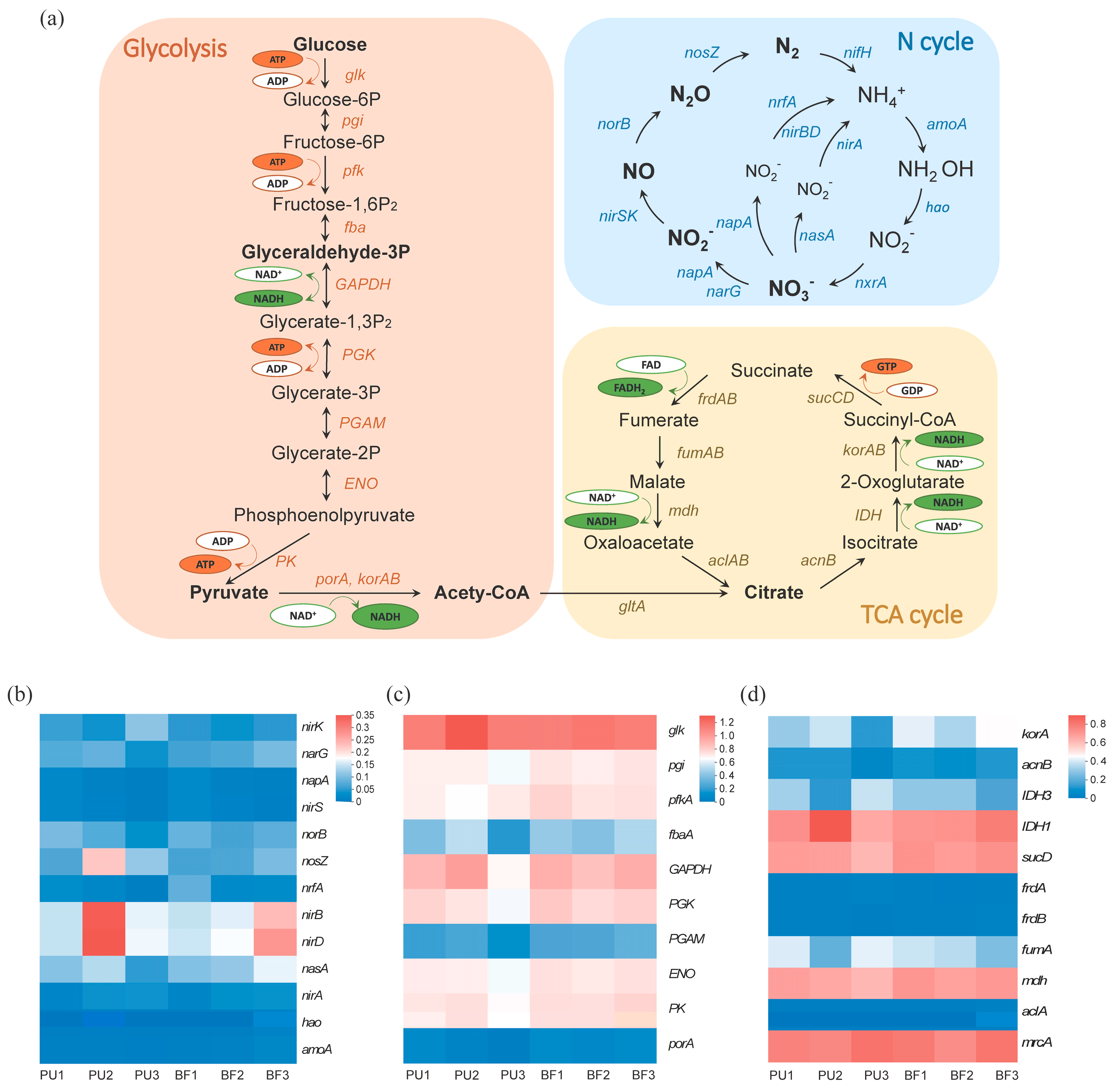
3.4. Functional Gene Abundance
3.4.1. N Cycling Functional Genes
3.4.2. C Cycling Functional Genes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Liu, J.G.; Zhou, Z.Y.; Li, P.Y.; Wang, Z.X.; Yan, Y.; Yu, X.Z.; Li, W.K.; Zheng, T.L.; Cao, Y.N.; Wu, W.J.; et al. Characteristics of rural domestic sewage discharge and their driving mechanisms: Evidence from the Northern Region, China. Front. Environ. Sci. Eng. 2024, 18, 83. [Google Scholar] [CrossRef]
- Wang, C.; Feng, B.B.; Wang, P.F.; Guo, W.Z.; Li, X.J.; Gao, H.; Zhang, B.; Chen, J. Revealing factors influencing spatial variation in the quantity and quality of rural domestic sewage discharge across China. Process Saf. Environ. Prot. 2022, 162, 200–210. [Google Scholar] [CrossRef]
- Li, X.Y.; Zhang, X.; Zhao, M.; Zheng, X.Y.; Wang, Z.Q.; Fan, C.Z. Application of Decentralized Wastewater Treatment Technology in Rural Domestic Wastewater Treatment. Sustainability 2024, 16, 8635. [Google Scholar] [CrossRef]
- Xie, Y.D.; Zhang, Q.H.; Wu, Q.; Zhang, J.Y.; Dzakpasu, M.; Wang, X.C. Novel adaptive activated sludge process leverages flow fluctuations for simultaneous nitrification and denitrification in rural sewage treatment. Water Res. 2024, 255, 121535. [Google Scholar] [CrossRef]
- Xu, B.; Wang, H.; Zhang, H.; Pu, M.; Zhao, M.; Tadayuki, F.; Zheng, X. Optimization of MBBR performance for rural domestic wastewater: Effects of carrier type, operating conditions, and zooplankton regulation. Process Saf. Environ. Prot. 2025, 202, 107847. [Google Scholar] [CrossRef]
- Zhao, Y.X.; Liu, D.; Huang, W.L.; Yang, Y.; Ji, M.; Nghiem, L.D.; Trinh, Q.T.; Tran, N.H. Insights into biofilm carriers for biological wastewater treatment processes: Current state-of-the-art, challenges, and opportunities. Bioresour. Technol. 2019, 288, 121619. [Google Scholar] [CrossRef] [PubMed]
- Morgan-Sagastume, F. Biofilm development, activity and the modification of carrier material surface properties in moving-bed biofilm reactors (MBBRs) for wastewater treatment. Crit. Rev. Environ. Sci. Technol. 2018, 48, 439–470. [Google Scholar] [CrossRef]
- Lago, A.; Rocha, V.; Barros, O.; Silva, B.; Tavares, T. Bacterial biofilm attachment to sustainable carriers as a clean-up strategy for wastewater treatment: A review. J. Water Process Eng. 2024, 63, 105368. [Google Scholar] [CrossRef]
- Zhang, X.Y.; Zhou, X.T.; Xie, Y.J.; Rong, X.S.; Liu, Z.G.; Xiao, X.; Liang, Z.S.; Jiang, S.Y.; Wei, J.; Wu, Z.R. A sustainable bio-carrier medium for wastewater treatment: Modified basalt fiber. J. Clean. Prod. 2019, 225, 472–480. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, X.C.; Arslan, M.; Zhou, X.T.; Wu, Z.R.; Liu, J.X.; Li, X.Y.; Din, M.G. Impact of truncated modified basalt fibers on aerobic granular sludge stability and pollutant removal in piggery wastewater treatment. Chem. Eng. J. 2024, 495, 153475. [Google Scholar] [CrossRef]
- Ni, H.C.; Wang, C.C.; Arslan, M.; Qian, J.C.; Liang, Z.S.; Luo, Z.J.; Cai, R.Q.; El-Din, M.G.; Wu, Z.R. Enhanced wastewater treatment by modified basalt fiber bio-carriers: Effect of etching and surface functionalization. J. Clean. Prod. 2022, 343, 130927. [Google Scholar] [CrossRef]
- Bhat, T.; Fortomaris, D.; Kandare, E.; Mouritz, A.P. Properties of thermally recycled basalt fibres and basalt fibre composites. J. Mater. Sci. 2018, 53, 1933–1944. [Google Scholar] [CrossRef]
- Ni, H.C.; Zhou, X.T.; Zhang, X.Y.; Xiao, X.; Liu, J.F.; Huan, H.; Luo, Z.J.; Wu, Z.R. Feasibility of using basalt fiber as biofilm Carrier to construct bio-nest for wastewater treatment. Chemosphere 2018, 212, 768–776. [Google Scholar] [CrossRef]
- Ni, H.C.; Arslan, M.; Qian, J.; Wang, Y.; Liu, Z.; Luo, Z.; Cai, R.; EI-Din, M.G.; Wu, Z. Application of basalt fibers in a biological contact oxidation reactor for the treatment of landfill leachate. J. Clean. Prod. 2021, 297, 126648. [Google Scholar] [CrossRef]
- Zhou, X.T.; Arslan, M.; Liu, Z.G.; Li, D.; Xi, H.P.; Feng, Y.J.; Li, S.W.; Wei, J.; Rong, X.S.; Liang, Z.S.; et al. Low carbon-to-nitrogen ratio digestate from high-rate anaerobic baffled reactor facilitates heterotrophic/autotrophic nitrifiers involved in nitrogen removal. Bioresour. Technol. 2022, 359, 127346. [Google Scholar] [CrossRef] [PubMed]
- Douglas, G.M.; Maffei, V.J.; Zaneveld, J.R.; Yurgel, S.N.; Brown, J.R.; Taylor, C.M.; Huttenhower, C.; Langille, M.G.I. PICRUSt2 for prediction of metagenome functions. Nat. Biotechnol. 2020, 38, 685–688. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.; Li, W.; Gong, Z.; Wen, C.; Zhang, C.; Lu, X. Treatment Performance and Characteristics of Biofilm Carriers in an Aerobic Waterwheel-Driven Rotating Biological Contactor. Water 2025, 17, 356. [Google Scholar] [CrossRef]
- Pang, Y.; Wang, J. Various electron donors for biological nitrate removal: A review. Sci. Total Environ. 2021, 794, 148699. [Google Scholar] [CrossRef]
- Loupasaki, E.; Diamadopoulos, E. Attached growth systems for wastewater treatment in small and rural communities: A review. J. Chem. Technol. Biotechnol. 2013, 88, 190–204. [Google Scholar] [CrossRef]
- Pang, Y.M.; Zhai, Y.X.; Fu, Y.X.; Lin, H.; Wang, J.L. A review of antibiotic effects on wastewater denitrification: From process inhibition to microbial resistance mechanism. Process Saf. Environ. Prot. 2025, 201, 107590. [Google Scholar] [CrossRef]
- Allison, S.D.; Martiny, J.B.H. Resistance, resilience, and redundancy in microbial communities. Proc. Natl. Acad. Sci. USA 2008, 105, 11512–11519. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Zhou, Q.; Sun, H.; Jia, L.; Zhao, L.; Wu, W. Metagenomic analyses of microbial structure and metabolic pathway in solid-phase denitrification systems for advanced nitrogen removal of wastewater treatment plant effluent: A pilot-scale study. Water Res. 2021, 196, 117067. [Google Scholar] [CrossRef] [PubMed]
- Brunings, A.M.; Gabriel, D.W. Xanthomonas citri: Breaking the surface. Mol. Plant Pathol. 2003, 4, 141–157. [Google Scholar] [CrossRef]
- Pang, Y.M.; Xia, L.; Li, S.J.; Wang, J.L.; Lin, H. Metagenomic insights into a solid phase heterotrophic-autotrophic denitrification reactor driven by biodegradable polymers and pyrite. J. Clean. Prod. 2025, 523, 146411. [Google Scholar] [CrossRef]
- Tan, H.; Wang, L.; Chen, Y.W.; Li, X.; Zhou, H.Z.; Tan, Z.L. Revealing the differences in metabolisms and nitrogen removal mechanisms of the full and shortcut nitrification-denitrification processes for the treatment of livestock wastewater. J. Environ. Chem. Eng. 2024, 12, 112381. [Google Scholar] [CrossRef]
- Kim, J.K.; Park, K.J.; Cho, K.S.; Nam, S.W.; Park, T.J.; Bajpai, R. Aerobic nitrification-denitrification by heterotrophic Bacillus strains. Bioresour. Technol. 2005, 96, 1897–1906. [Google Scholar] [CrossRef]
- Young, B.; Delatolla, R.; Abujamel, T.; Kennedy, K.; Laflamme, E.; Stintzi, A. Rapid start-up of nitrifying MBBRs at low temperatures: Nitrification, biofilm response and microbiome analysis. Bioprocess Biosyst. Eng. 2017, 40, 731–739. [Google Scholar] [CrossRef]
- Zhao, Y.; Li, W.G.; Chen, L.; Zhou, Y.J. Characterization of heterotrophic nitrification by a thermotolerant Brevibacillus Agri N2 isolated from sewage sludge composting. Environ. Res. 2022, 214, 113903. [Google Scholar] [CrossRef]
- Pang, Y.; Wang, J. Effect of ferric iron (Fe(III)) on heterotrophic solid-phase denitrification: Denitrification performance and metabolic pathway. Bioresour. Technol. 2023, 369, 128401. [Google Scholar] [CrossRef]
- Ivanov, V.; Stabnikov, V.; Zhuang, W.Q.; Tay, J.H.; Tay, S.T.L. Phosphate removal from the returned liquor of municipal wastewater treatment plant using iron-reducing bacteria. J. Appl. Microbiol. 2005, 98, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.F.; Xu, M.Y.; Deng, T.C.; Hu, W.Z.; He, Z.L.; Yang, X.A.; Wang, B.; Song, D.; Chen, L.T.; Huang, Y.D.; et al. Synergistic interactions of Desulfovibrio and Petrimonas for sulfate-reduction coupling polycyclic aromatic hydrocarbon degradation. J. Hazard. Mater. 2021, 407, 124385. [Google Scholar] [CrossRef] [PubMed]
- Al-Amshawee, S.; Yunus, M.Y.B.M.; Vo, D.V.N.; Tran, N.H. Biocarriers for biofilm immobilization in wastewater treatments: A review. Environ. Chem. Lett. 2020, 18, 1925–1945. [Google Scholar] [CrossRef]
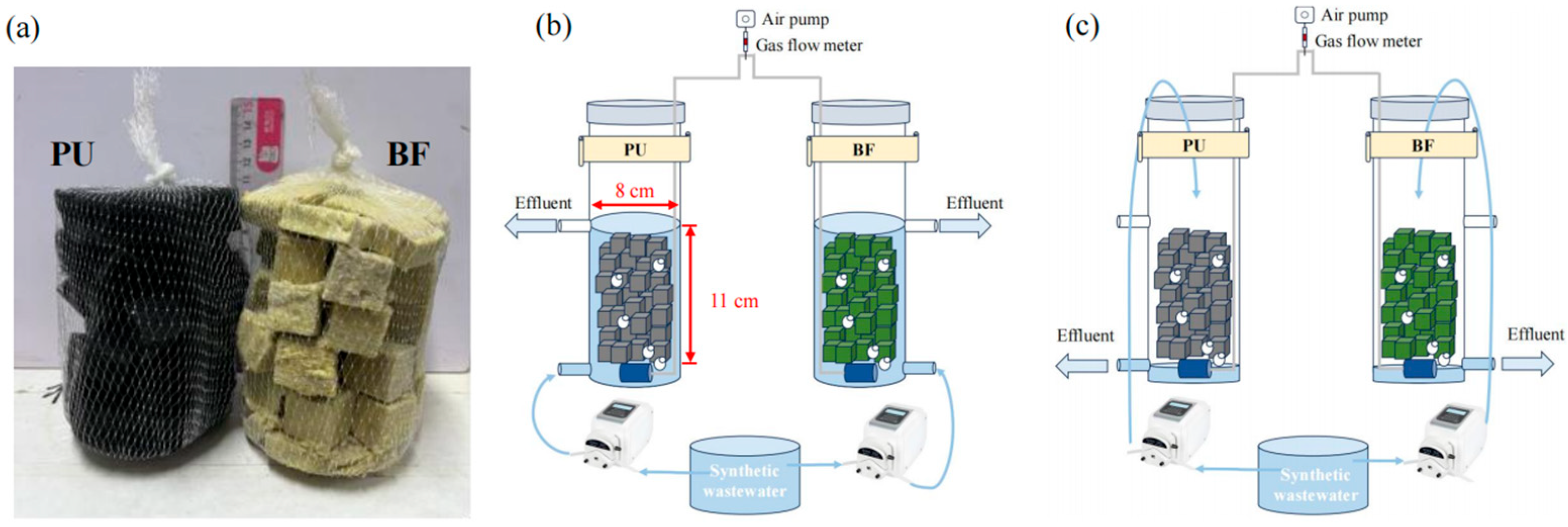




| Physical Properties | BF | PU |
|---|---|---|
| Total pore volume (mL/g) | 3.83 | 0.43 |
| Total pore surface area (m2/g) | 0.27 | 0.08 |
| Average pore diameter (nm) | 56,675.31 | 21,957.85 |
| Bulk density (g/mL) | 0.24 | 0.77 |
| Skeletal density (g/mL) | 2.71 | 1.16 |
| Porosity (%) | 91.22 | 33.34 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Xu, Q.; Zhai, Y.; Gao, J.; Lin, H.; Pang, Y. Feasibility of Basalt Fiber Felt as Biocarrier for Rural Domestic Wastewater Treatment: Performance and Microbial Community Analysis. Processes 2026, 14, 349. https://doi.org/10.3390/pr14020349
Xu Q, Zhai Y, Gao J, Lin H, Pang Y. Feasibility of Basalt Fiber Felt as Biocarrier for Rural Domestic Wastewater Treatment: Performance and Microbial Community Analysis. Processes. 2026; 14(2):349. https://doi.org/10.3390/pr14020349
Chicago/Turabian StyleXu, Qian, Yuxuan Zhai, Jilong Gao, Hai Lin, and Yunmeng Pang. 2026. "Feasibility of Basalt Fiber Felt as Biocarrier for Rural Domestic Wastewater Treatment: Performance and Microbial Community Analysis" Processes 14, no. 2: 349. https://doi.org/10.3390/pr14020349
APA StyleXu, Q., Zhai, Y., Gao, J., Lin, H., & Pang, Y. (2026). Feasibility of Basalt Fiber Felt as Biocarrier for Rural Domestic Wastewater Treatment: Performance and Microbial Community Analysis. Processes, 14(2), 349. https://doi.org/10.3390/pr14020349






