The Effects of Using Shortwave Infrared Lamp-Drying and Alkali Pretreatment on the Color, Texture, and Volatile Compounds of Gongliao Gelidium amansii Seaweed and Its Jelly Qualities
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Dried Gelidium Seaweed Sample Preparation
2.3. Moisture Content of Dried Seaweeds and Gelidium Jelly Preparation
2.4. Dried Seaweed Ash Content and Color Analysis
2.5. Texture Analysis of Gelidium Jelly
2.6. Agar Solution Rheological Measurement
2.7. Volatiles of Dried Gelidium Seaweed
2.8. Sensory Evaluation
2.9. Statistical Analysis
3. Results and Discussion
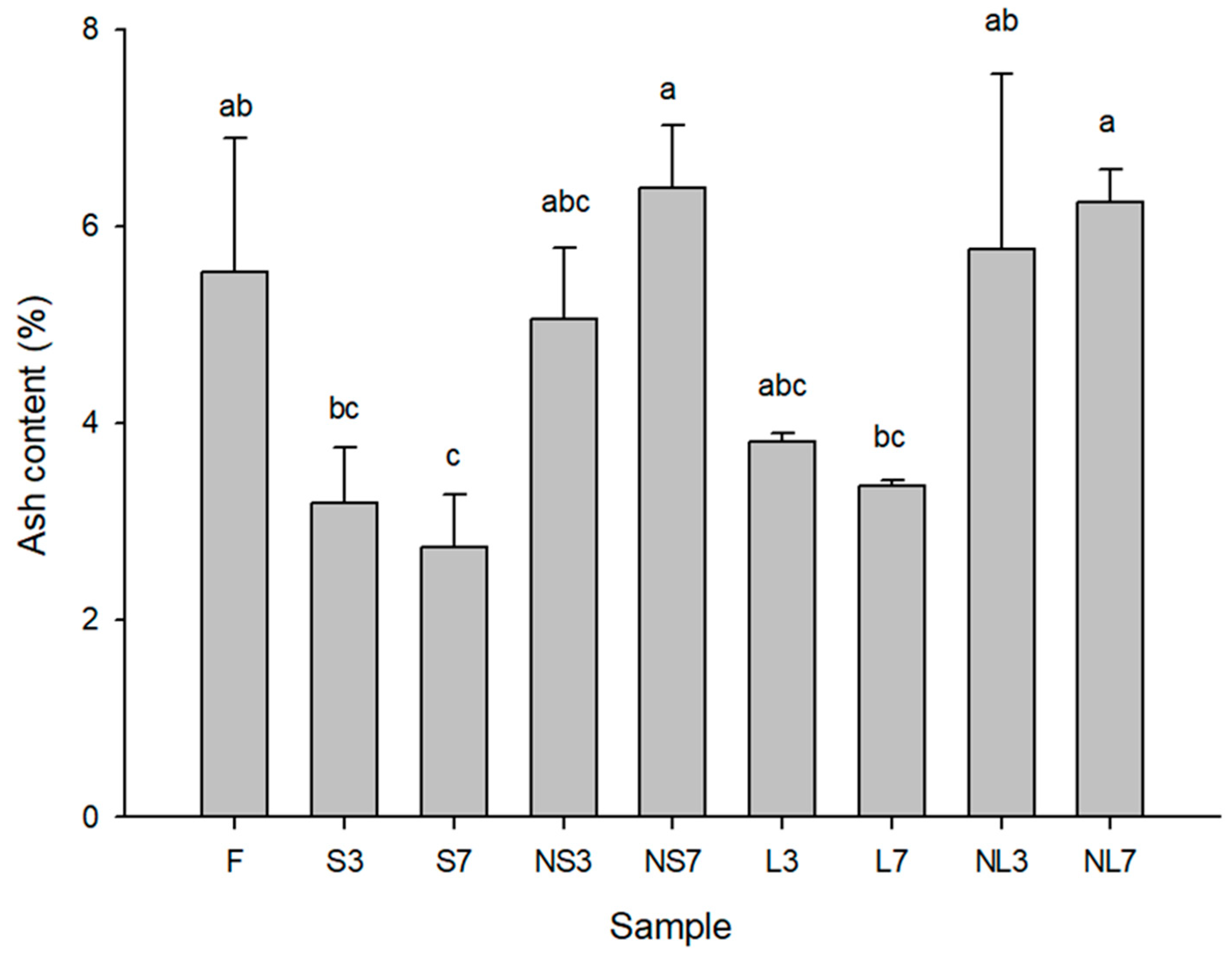
3.1. Results of Sun-Drying, Alkaline Pretreatment, Infrared-Drying, and Washing Cycles on the Weight, Moisture, and Ash Content of Gelidium Seaweeds
3.2. Color of Dried Gelidium Seaweeds with/Without Alkaline Pretreatment and Various Drying Methods and Washing Cycles
3.3. Volatile Compounds of Dried Gelidium Seaweeds
| Concentrations (mg/L) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Compound | Fresh | S3 | S7 | NS3 | NS7 | L3 | L7 | NL3 | NL7 | Odor (Odor Reference) |
| Hexanal | 609.33 | 357.58 | 541.00 | 78.47 | 58.33 | 27.80 | 22.88 | - | - | grass, tallow, fat (a) |
| 1-Octen-3-one | 123.87 | 246.41 | 190.62 | - | - | - | - | - | mushroom, metal (a) | |
| 2-Heptenal, (Z)- | 101.19 | 475.06 | 785.20 | 80.54 | - | 14.49 | - | - | - | fatty (c) |
| 3-Octanone, 2-methyl- | 105.49 | 29.88 | 46.42 | - | - | 27.28 | - | - | - | unknown |
| 5-Hepten-2-one, 6-methyl- | 100.46 | 40.45 | 31.28 | 31.33 | - | 21.18 | - | - | - | unknown |
| Nonanal | 238.78 | 211.26 | 355.73 | 136.38 | 79.48 | 24.70 | 39.70 | 76.82 | 57.22 | fat, citrus, green (a) |
| 2-Octenal, (E)- | 533.22 | 599.27 | 541.66 | 93.80 | 67.44 | 64.45 | 41.29 | 40.95 | 36.15 | green, nut, fat (a) |
| 1-Octen-3-ol | 338.03 | 676.58 | 384.87 | 220.38 | 26.18 | 73.25 | 49.46 | 119.30 | 98.32 | mushroom (a) |
| 1-Hexanol, 2-ethyl- | 96.75 | - | - | - | 23.21 | - | - | - | - | unknown |
| 2-Nonenal, (E)- | 398.18 | 454.99 | 377.07 | 79.62 | 31.69 | 60.67 | 41.54 | 38.73 | 39.98 | orris, fat, cucumber (a) |
| cis-4-Decenal | 128.21 | 126.58 | 100.40 | 39.09 | - | 17.45 | - | - | - | green, must (a) |
| 1-Octanol | 107.66 | 123.45 | 178.69 | 80.41 | - | 13.31 | - | 35.88 | 28.80 | moss, nut, mushroom (a) |
| 2,6-Nonadienal, (E,Z)- | 119.29 | 115.36 | 65.64 | 21.71 | - | 22.10 | 15.93 | - | - | cucumber, wax, green (a) |
| β-Cyclocitral | 244.50 | 338.19 | 189.05 | 144.13 | - | 42.97 | 22.64 | 87.12 | 70.65 | mint (a) |
| 1-Menthol | 47.71 | - | - | - | - | - | - | - | - | peppermint (a) |
| Dodecanal | 35.94 | 48.37 | 56.97 | 19.57 | - | - | - | - | - | fatty, green (b) |
| 2,4-Decadienal | 62.39 | 203.77 | 114.75 | 22.47 | 27.00 | 12.58 | 10.06 | - | - | seaweed (a) |
| 2,6-Nonadien-1-ol | 39.43 | - | - | - | - | - | - | - | - | unknown |
| 2,4-Decadienal, (E,E)- | 119.51 | 329.40 | 279.71 | 62.93 | - | 15.48 | 11.06 | - | - | seaweed (a) |
| Tridecanal | 236.57 | 297.17 | 124.29 | 96.49 | 8.95 | 23.72 | 12.17 | - | - | flower, sweet, must (a) |
| α-Ionone | 276.36 | 243.02 | 57.69 | 145.83 | - | 41.74 | 23.47 | 148.38 | 121.47 | wood, violet (a) |
| Tetradecanal | 24.94 | 27.99 | 28.59 | - | - | - | - | - | - | fatty, green (b) |
| β-Ionone | 130.19 | 53.98 | - | 23.56 | - | 27.62 | 14.33 | 113.17 | 92.83 | seaweed, violet, flower, raspberry (a) |
| Pentadecanal | 192.70 | 73.83 | 32.52 | 25.97 | 16.06 | 35.91 | 23.27 | - | - | fresh (a) |
| cis-9-Hexadecenal | 22.00 | - | - | - | - | - | - | - | - | unknown |
| 2,4-Di-tert-butylphenol | 11.46 | 11.79 | 10.24 | 9.74 | 32.31 | - | 5.45 | - | - | unknown |
| 3-Octen-2-one, (E)- | - | 230.39 | 196.17 | 76.68 | - | - | - | - | - | unknown |
| 2,4-Heptadienal | - | 50.13 | 27.38 | - | - | - | - | - | - | fried (a) |
| 2,4-Heptadienal, (E,E)- | - | 77.59 | 72.94 | - | - | - | - | - | - | nut, fat (a) |
| 2,4-Nonadienal, (E,E)- | - | 110.38 | 219.93 | 35.27 | - | - | - | - | - | geranium, pungent (a) |
| 2,4-Undecadienal, (E,E)- | - | 28.46 | 19.75 | - | - | - | - | - | - | unknown |
| 1H-Pyrrole-2,5-dione, 3-ethyl-4-methyl- | - | 23.51 | - | - | - | - | - | - | - | unknown |
| Furan, 2-pentyl- | - | - | 119.33 | - | - | - | - | - | - | green bean, butter (a) |
| Octanal | - | - | 264.08 | - | - | - | - | - | - | fat, soap, lemon, green (a) |
| 5-Ethylcyclopent-1-enecarboxaldehyde | - | - | 71.90 | - | - | - | - | - | - | unknown |
| Decanal | - | - | 67.49 | 19.57 | - | - | - | - | - | soap, orange peel, tallow (a) |
| Undecanal | - | - | 29.42 | 28.80 | - | - | - | - | - | citrusy, pungent, cilantro (b) |
| 2,4-Nonadienal | - | - | 50.97 | - | - | - | - | - | - | watermelon (a) |
| 2-Undecanone, 6,10-dimethyl- | - | - | 26.14 | - | - | - | - | - | - | unknown |
| 2-Undecenal | - | - | 20.65 | - | - | - | - | - | - | sweet (a) |
| 1-Dodecanol | - | - | 18.77 | - | - | - | 8.81 | - | - | oily, citrusy (b) |
| 1-Tetradecanol | - | - | 11.34 | - | - | 3.86 | - | - | - | coconut |
| 2-Decanone | - | - | - | 68.39 | - | - | - | 45.34 | 42.48 | orange, peach, floral, fatty (d) |
| Levomenthol | - | - | - | 19.93 | - | - | - | 14.66 | 18.54 | menthol (a) |
| Heptadecane | - | - | - | - | 42.35 | 29.08 | 34.18 | - | - | alkane (a) |
3.4. Principal Component Analysis of Gelidium Seaweed Volatiles
3.5. Effects of Alkaline Pretreating, Washing, Sun-Drying, and Infrared-Drying Cycles on the Gelidium Jelly’s Appearance
3.6. Texture Analysis of Alkaline-Pretreated, Washed, Sun-Dried, and Infrared-Dried Gelidium Seaweed Jellies
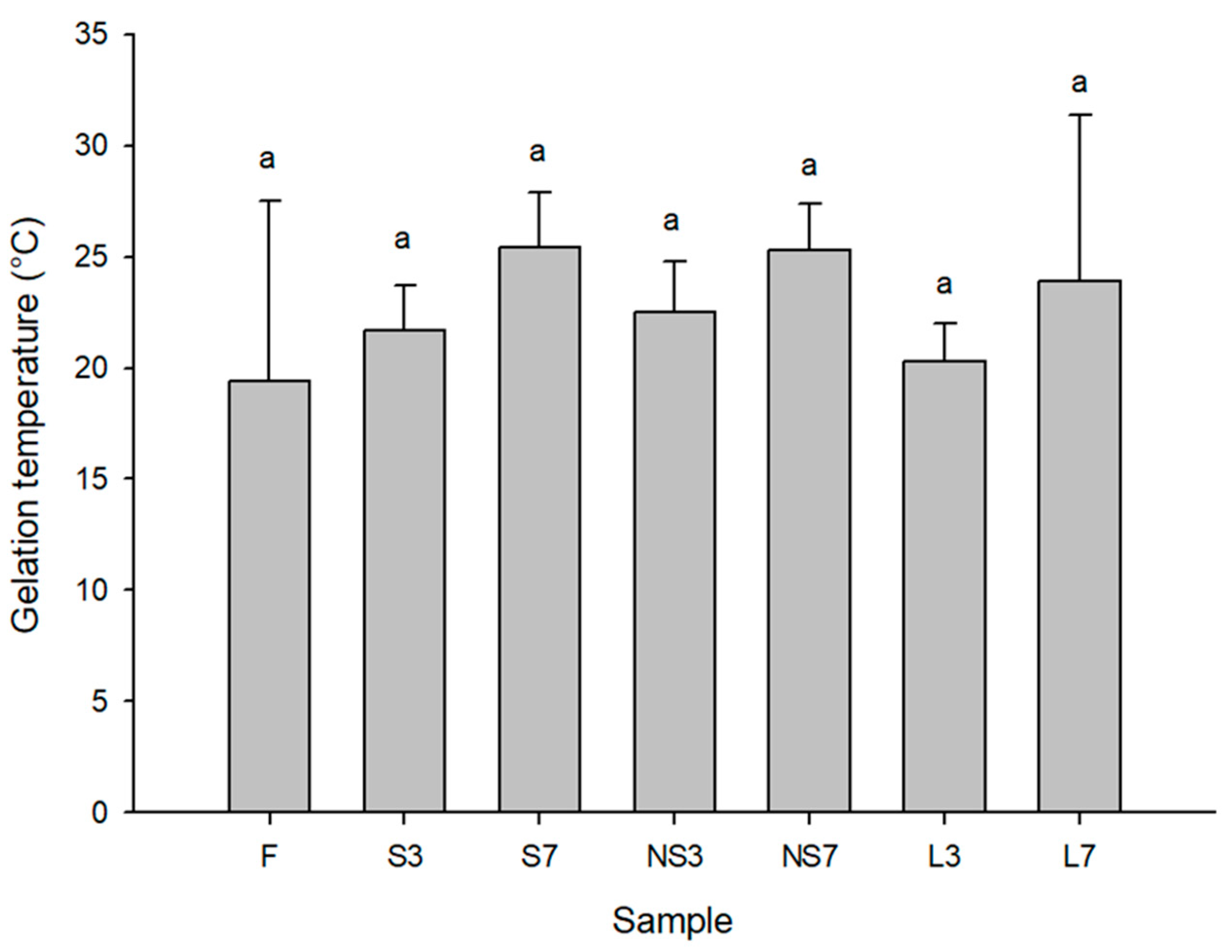
3.7. Rheological Analysis of Gelidium Boiling Extraction Solutions
3.8. Sensory Evaluation of Gelidium Jellies
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Deepika, C.; Ravishankar, G.A.; Rao, A.R. Potential products from macroalgae. In An Overview. Sustainable Global Resources of Seaweeds Volume 1: Bioresources, Cultivation, Trade and Multifarious Applications; Springer: Cham, Switzerland, 2022; pp. 17–44. [Google Scholar]
- Robertson, R.C.; Mateo, M.R.G.; O’Grady, M.N.; Guihéneuf, F.; Stengel, D.B.; Ross, R.P.; Fitzgerald, G.F.; Kerry, J.P.; Stanton, C. An assessment of the techno-functional and sensory properties of yoghurt fortified with a lipid extract from the microalga Pavlova lutheri. Innov. Food Sci. Emerg. Technol. 2016, 37, 237–246. [Google Scholar] [CrossRef]
- Leandro, A.; Pereira, L.; Gonçalves, A.M. Diverse applications of marine macroalgae. Mar. Drugs 2019, 18, 17. [Google Scholar] [CrossRef]
- Meinita, M.D.N.; Harwanto, D.; Hannan, M.A.; Jeong, G.T.; Moon, I.S.; Choi, J.S. A concise review of the potential utilization based on bioactivity and pharmacological properties of the genus Gelidium (Gelidiales, Rhodophyta). J. Appl. Phycol. 2023, 35, 1499–1523. [Google Scholar] [CrossRef]
- Hurler, J.; Engesland, A.; Kermany, B.P.; Škalko-Basnet, N. Improved texture analysis for hydrogel characterization: Gel cohesiveness, adhesiveness, and hardness. J. Appl. Polym. Sci. 2012, 125, 180–188. [Google Scholar] [CrossRef]
- Sung, W.C.; Lin, H.T.V.; Liao, W.C.; Fang, M. Effects of Halogen Lamp and Traditional Sun Drying on the Volatile Compounds, Color Parameters and Gel Texture of Gongliao Gelidium Seaweed. Foods 2023, 12, 4508. [Google Scholar] [CrossRef] [PubMed]
- Meena, R.; Prasad, K.; Siddhanta, A.K. Preparation of superior quality products from two Indian agarophytes. J. Appl. Phycol. 2011, 23, 183–189. [Google Scholar] [CrossRef]
- Lemus, A.; Bird, K.; Kapraun, D.F.; Koehn, F. Agar yield, quality and standing crop biomass of Gelidium serrulatum, Gelidium floridanum and Pterocladia capillacea in Venezuela. Food Hydrocoll. 1991, 5, 469–479. [Google Scholar] [CrossRef]
- Yarnpakdee, S.; Benjakul, S.; Kingwascharapong, P. Physico-chemical and gel properties of agar from Gracilaria tenuistipitata from the lake of Songkhla, Thailand. Food Hydrocoll. 2015, 51, 217–226. [Google Scholar] [CrossRef]
- Ahmed, N.; Singh, J.; Chauhan, H.; Anjum, P.G.A.; Kour, H. Different drying methods: Their applications and recent advances. Int. J. Food Nutr. Saf. 2013, 4, 34–42. [Google Scholar]
- Chen, Q.; Bi, J.; Wu, X.; Yi, J.; Zhou, L.; Zhou, Y. Drying kinetics and quality attributes of jujube (Zizyphus jujuba Miller) slices dried by hot-air and short-and medium-wave infrared radiation. LWT-Food Sci. Technol. 2015, 64, 759–766. [Google Scholar] [CrossRef]
- Huang, D.; Yang, P.; Tang, X.; Luo, L.; Sunden, B. Application of infrared radiation in the drying of food products. Trends Food Sci. Technol. 2021, 110, 765–777. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists (AOAC). Official Methods of Analysis, 21st ed.; AOAC International: Gaithersburg, MD, USA, 2019. [Google Scholar]
- Matheus, J.; Alegria, M.J.; Nunes, M.C.; Raymundo, A. Algae-boosted chickpea hummus: Improving nutrition and texture with seaweeds and microalgae. Foods 2024, 13, 2178. [Google Scholar] [CrossRef]
- Kim, D.S.; Iida, F. Texture characteristics of sea buckthorn (Hippophae rhamnoides) jelly for the elderly based on the gelling agent. Foods 2022, 11, 1892. [Google Scholar] [CrossRef]
- Garrido, J.I.; Lozano, J.E.; Genovese, D.B. Effect of formulation variables on rheology, texture, colour, and acceptability of apple jelly: Modelling and optimization. LWT-Food Sci. Technol. 2015, 62, 325–332. [Google Scholar] [CrossRef]
- Yamamoto, M.; Baldermann, S.; Yoshikawa, K.; Fujita, A.; Mase, N.; Watanabe, N. Determination of volatile compounds in four commercial samples of Japanese green algae using solid phase microextraction gas chromatography mass spectrometry. Sci. World 2014, 2014, 289780. [Google Scholar] [CrossRef]
- Kwon, Y.; Ryu, J.; Ju, S. Sensory attributes of buckwheat jelly (Memilmuk) with mung bean starch added to improve texture and taste. Foods 2021, 10, 2860. [Google Scholar] [CrossRef] [PubMed]
- Paiva, L.; Lima, E.; Neto, A.I.; Marcone, M.; Baptista, J. Nutritional and functional bioactivity value of selected Azorean macroalgae: Ulva compressa, Ulva rigida, Gelidium microdon, and Pterocladiella capillacea. J. Food Sci. 2017, 82, 1757–1764. [Google Scholar] [CrossRef]
- Setyaningsih, D.; Hidayat, A.; Aryanti, E.Y.V.; Muna, N. Alkaline pre-treatment of Gelidium latifolium and Caulerpa racemosa for bioethanol production. IOP Conf. Ser. Earth Environ. Sci. 2019, 309, 12005. [Google Scholar] [CrossRef]
- Xiao, Q.; Wang, X.; Zhang, J.; Zhang, Y.; Chen, J.; Chen, F.; Xiao, A. Pretreatment techniques and green extraction technologies for agar from Gracilaria lemaneiformis. Mar. Drugs 2021, 19, 617. [Google Scholar] [CrossRef]
- Li, H.; Yu, X.; Jin, Y.; Zhang, W.; Liu, Y. Development of an eco-friendly agar extraction technique from the red seaweed Gracilaria lemaneiformis. Bioresour. Technol. 2008, 99, 3301–3305. [Google Scholar] [CrossRef] [PubMed]
- Munier, M.; Jubeau, S.; Wijaya, A.; Morancais, M.; Dumay, J.; Marchal, L.; Jaouen, P.; Fleurence, J. Physicochemical factors affecting the stability of two pigments: R-phycoerythrin of Grateloupia turuturu and B-phycoerythrin of Porphyridium cruentum. Food Chem. 2014, 150, 400–407. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-García, F.; Mirzayeva, A.; Roldán, A.; Castro, R.; Palacios, V.; García-Barroso, C.; Durán-Guerrero, E. Evolution of volatile compounds and sensory characteristics of edible green seaweed (Ulva rigida) during storage at different temperatures. J. Sci. Food Agric. 2019, 99, 5475–5482. [Google Scholar] [CrossRef]
- Le Pape, M.A.; Grua-Priol, J.; Prost, C.; Demaimay, M. Optimization of dynamic headspace extraction of the edible red algae Palmaria palmata and identification of the volatile components. J. Agric. Food Chem. 2004, 52, 550–556. [Google Scholar] [CrossRef]
- Giri, A.; Osako, K.; Ohshima, T. Identification and characterisation of headspace volatiles of fish miso, a Japanese fish meat based fermented paste, with special emphasis on effect of fish species and meat washing. Food Chem. 2010, 120, 621–631. [Google Scholar] [CrossRef]
- Urlass, S.; Wu, Y.; Nguyen, T.T.; Winberg, P.; Turner, M.S.; Smyth, H. Unravelling the aroma and flavour of algae for future food applications. Trends Food Sci. Technol. 2023, 138, 370–381. [Google Scholar] [CrossRef]
- Reineccius, G. Flavor Chemistry and Technology, 2nd ed.; CRC Press: Boca Raton, FL, USA, 2005. [Google Scholar]
- Gu, S.Q.; Wang, X.C.; Tao, N.P.; Wu, N. Characterization of volatile compounds in different edible parts of steamed Chinese mitten crab (Eriocheir sinensis). Food Res. Int. 2013, 54, 81–92. [Google Scholar] [CrossRef]
- Quynh, C.T.T. Study on the aroma model of Vietnamese coriander leaves (Polygonum odoratum). Vietnam. J. Sci. Technol. 2016, 54, 73. [Google Scholar] [CrossRef]
- Xu, L.; Wang, J.; Tian, A.; Wang, S.; Zhao, K.; Zhang, R.; Wu, X.; Liu, Y.; Liu, X.; Chen, K.; et al. Characteristic volatiles fingerprints in olive vegetable stored at different conditions by HS-GC-IMS. Food Chem. X 2023, 18, 100707. [Google Scholar] [CrossRef]
- Sommer, S.; Lang, L.M.; Drummond, L.; Buchhaupt, M.; Fraatz, M.A.; Zorn, H. Odor characteristics of novel non-canonical terpenes. Molecules 2022, 27, 3827. [Google Scholar] [CrossRef] [PubMed]
- Hollósy, F. Effects of ultraviolet radiation on plant cells. Micron 2002, 33, 179–197. [Google Scholar] [CrossRef]




| L* | a* | b* | ΔE# | |
|---|---|---|---|---|
| F | 16.79 ± 1.90 d | 3.89 ± 0.77 ab | 5.79 ± 1.30 b | - |
| S3 | 29.88 ± 2.30 c | 5.93 ± 1.22 a | 11.42 ± 1.36 b | 14.46 ± 1.92 b |
| S7 | 50.15 ± 3.90 b | 6.33 ± 0.89 a | 26.32 ± 2.11 a | 39.28 ± 5.10 a |
| NS3 | 47.86 ± 4.29 b | −0.39 ± 1.06 d | 26.55 ± 2.87 a | 37.64 ± 5.66 a |
| NS7 | 61.84 ± 3.47 a | 0.25 ± 0.53 cd | 31.25 ± 2.03 a | 51.89 ± 5.92 a |
| L3 | 21.60 ± 0.64 cd | 2.55 ± 0.54 bc | 5.63 ± 1.34 b | 5.61 ± 2.72 b |
| L7 | 20.92 ± 4.26 cd | 2.76 ± 0.21 bc | 5.49 ± 2.52 b | 6.59 ± 4.09 b |
| NL3 | 21.06 ± 3.27 cd | −3.39 ± 1.14 e | 5.57 ± 1.15 b | 9.17 ± 3.07 b |
| NL7 | 24.07 ± 2.46 cd | −4.34 ± 0.34 e | 6.70 ± 0.83 b | 11.30 ± 3.31 b |
| Hardness (N) | Springiness | Gumminess (N) | |
|---|---|---|---|
| F | 0.37 ± 0.12 cd | 0.42 ± 0.10 b | 0.16 ± 0.09 c |
| S3 | 0.61 ± 0.11 abc | 0.69 ± 0.05 ab | 0.40 ± 0.10 abc |
| S7 | 1.08 ± 0.22 a | 0.84 ± 0.07 a | 0.82 ± 0.21 a |
| NS3 | 0.56 ± 0.28 abc | 0.57 ± 0.19 ab | 0.33 ± 0.24 bc |
| NS7 | 0.95 ± 0.21 ab | 0.77 ± 0.06 a | 0.66 ± 0.18 ab |
| L3 | 0.59 ± 0.01 abc | 0.64 ± 0.02 ab | 0.36 ± 0.01 abc |
| L7 | 0.49 ± 0.30 bcd | 0.54 ± 0.20 ab | 0.30 ± 0.24 bc |
| NL3 | 0.00 ± 0.00 d | 0.00 ± 0.00 c | 0.00 ± 0.00 c |
| NL7 | 0.00 ± 0.00 d | 0.00 ± 0.00 c | 0.00 ± 0.00 c |
| Transparency | Algal Odor | Hardness | Springiness | Appearance | Aroma | Texture | Overall Acceptability | |
|---|---|---|---|---|---|---|---|---|
| F | 3.57 ± 1.51 a | 3.14 ± 1.35 a | 1.43 ± 1.13 cd | 1.57 ± 1.51 a | 4.29 ± 1.25 ab | 2.71 ± 1.11 b | 2.14 ± 1.07 c | 3.00 ± 0.82 ab |
| S3 | 3.57 ± 0.98 a | 2.57 ± 1.13 a | 2.71 ± 1.11 bc | 2.29 ± 1.25 a | 4.29 ± 1.60 ab | 4.14 ± 1.77 ab | 3.86 ± 1.57 abc | 3.57 ± 0.79 ab |
| S7 | 4.29 ± 1.11 a | 2.00 ± 1.29 a | 4.57 ± 0.53 a | 3.29 ± 1.50 a | 5.86 ± 0.90 a | 5.43 ± 1.27 a | 5.14 ± 1.21 a | 5.14 ± 1.21 a |
| NS3 | 4.00 ± 1.15 a | 2.43 ± 0.98 a | 2.71 ± 0.95 bc | 3.14 ± 1.35 a | 4.29 ± 0.49 ab | 4.14 ± 0.90 ab | 4.57 ± 1.13 ab | 4.29 ± 1.98 ab |
| NS7 | 4.43 ± 1.51 a | 2.00 ± 1.00 a | 3.14 ± 1.35 ab | 3.71 ± 1.38 a | 5.14 ± 1.57 ab | 3.57 ± 1.72 ab | 5.14 ± 1.77 a | 4.71 ± 1.80 ab |
| L3 | 3.71 ± 1.25 a | 2.86 ± 1.21 a | 1.43 ± 0.53 cd | 1.86 ± 0.69 a | 3.43 ± 1.13 ab | 3.86 ± 1.77 ab | 3.14 ± 1.35 abc | 3.00 ± 1.29 ab |
| L7 | 3.14 ± 0.69 a | 2.43 ± 1.13 a | 1.43 ± 1.13 cd | 1.71 ± 1.50 a | 3.43 ± 1.62 ab | 3.29 ± 1.25 ab | 2.29 ± 1.11 c | 2.86 ± 1.35 ab |
| NL3 | 3.57 ± 1.27 a | 3.43 ± 1.40 a | 1.00 ± 0.00 d | 1.86 ± 1.57 a | 3.57 ± 2.07 ab | 3.00 ± 1.63 ab | 2.43 ± 1.27 bc | 2.57 ± 1.13 b |
| NL7 | 3.57 ± 1.62 a | 2.71 ± 1.25 a | 1.00 ± 0.00 d | 1.57 ± 1.51 a | 3.14 ± 1.68 b | 3.43 ± 1.90 ab | 2.29 ± 1.25 c | 2.71 ± 1.25 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Lin, H.-T.V.; Wang, S.-T.; Chen, L.-A.; Sung, W.-C. The Effects of Using Shortwave Infrared Lamp-Drying and Alkali Pretreatment on the Color, Texture, and Volatile Compounds of Gongliao Gelidium amansii Seaweed and Its Jelly Qualities. Processes 2026, 14, 345. https://doi.org/10.3390/pr14020345
Lin H-TV, Wang S-T, Chen L-A, Sung W-C. The Effects of Using Shortwave Infrared Lamp-Drying and Alkali Pretreatment on the Color, Texture, and Volatile Compounds of Gongliao Gelidium amansii Seaweed and Its Jelly Qualities. Processes. 2026; 14(2):345. https://doi.org/10.3390/pr14020345
Chicago/Turabian StyleLin, Hong-Ting Victor, Shang-Ta Wang, Ling-An Chen, and Wen-Chieh Sung. 2026. "The Effects of Using Shortwave Infrared Lamp-Drying and Alkali Pretreatment on the Color, Texture, and Volatile Compounds of Gongliao Gelidium amansii Seaweed and Its Jelly Qualities" Processes 14, no. 2: 345. https://doi.org/10.3390/pr14020345
APA StyleLin, H.-T. V., Wang, S.-T., Chen, L.-A., & Sung, W.-C. (2026). The Effects of Using Shortwave Infrared Lamp-Drying and Alkali Pretreatment on the Color, Texture, and Volatile Compounds of Gongliao Gelidium amansii Seaweed and Its Jelly Qualities. Processes, 14(2), 345. https://doi.org/10.3390/pr14020345







