Physicochemical Treatment of Electroplating Wastewater: Efficiency Evaluation and Process Optimization
Abstract
1. Introduction
2. Materials and Methods
2.1. Source and Characteristics of Electroplating Wastewater
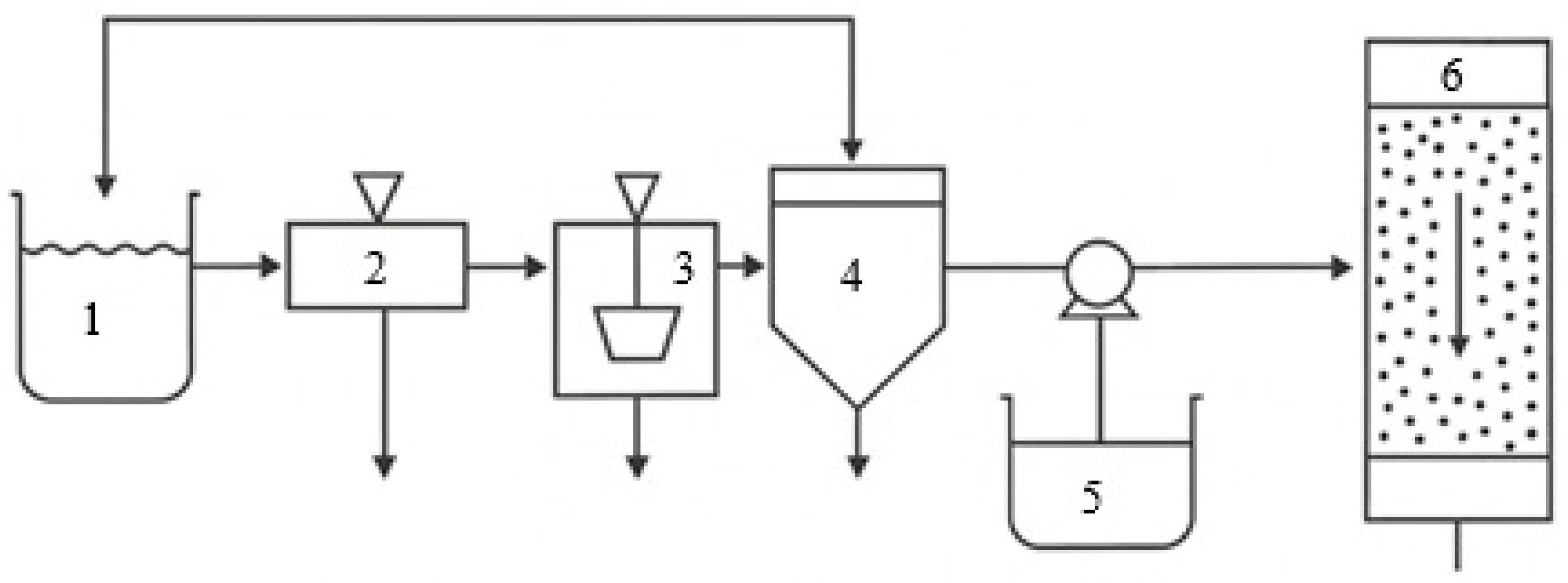
2.2. Treatment Processes
- -
- Removal efficiency of heavy metals: chromium (Cr), nickel (Ni), copper (Cu), and zinc (Zn).
- -
- Overall reagent balance and operating conditions, including pH correction and chemical dosages.
- -
- Operational stability of the GAC filter, expressed as the time to breakthrough.
2.2.1. Coagulation Process
2.2.2. Filtration Process
2.3. Analytical Methods
3. Results and Discussion
3.1. Influent Quality (Raw Wastewater)
3.2. Effect of Coagulant Dose and pH
3.3. Combined Coagulation–GAC Filtration
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Fryczkowska, B.; Przywara, L.; Turek, T. Application of PAN-PANI composite membranes in purification of industrial wastewater generated during processing of metals. Inżynieria Ekol. 2017, 18, 21–29. [Google Scholar] [CrossRef]
- GracePavithra, K.; Jaikumar, V.; Kumar, P.S.; SundarRajan, P. A review on cleaner strategies for chromium industrial wastewater: Present research and future perspective. J. Clean. Prod. 2019, 228, 580–593. [Google Scholar] [CrossRef]
- Prasetyaningrum, A.; Jos, B.; Dharmawan, Y.; Praptyana, I.R. Effect of pH and Current Density on Electrocoagulation Process for Degradation of Chromium (VI) in Plating Industrial Wastewater. J. Phys. Conf. Ser. 2019, 1295, 012064. [Google Scholar] [CrossRef]
- Genchi, G.; Carocci, A.; Lauria, G.; Sinicropi, M.S.; Catalano, A. Nickel: Human Health and Environmental Toxicology. Int. J. Environ. Res. Public Health 2020, 17, 679. [Google Scholar] [CrossRef]
- Vaiopoulou, E.; Gikas, P. Regulations for chromium emissions to the aquatic environment in Europe and elsewhere. Chemosphere 2020, 254, 126876. [Google Scholar] [CrossRef]
- Fu, F.; Wang, Q. Removal of heavy metal ions from wastewaters: A review. J. Environ. Manag. 2011, 92, 407–418. [Google Scholar] [CrossRef]
- Qasem, N.A.A.; Mohammed, R.H.; Lawal, D.U. Removal of heavy metal ions from wastewater: A comprehensive and critical review. Npj Clean Water 2021, 4, 36. [Google Scholar] [CrossRef]
- Yang, X.; Wan, Y.; Zheng, Y.; He, F.; Yu, Z.; Huang, J.; Wang, H.; Ok, Y.S.; Jiang, Y.; Gao, B. Surface functional groups of carbon-based adsorbents and their roles in the removal of heavy metals from aqueous solutions: A critical review. Chem. Eng. J. 2019, 366, 608–621. [Google Scholar] [CrossRef] [PubMed]
- Costa, J.M.; da Costa, J.G.D.R.; de Almeida Neto, A.F. Techniques of nickel(II) removal from electroplating industry wastewater: Overview and trends. J. Water Process Eng. 2022, 46, 102593. [Google Scholar] [CrossRef]
- Callegari, A.; Boguniewicz-Zablocka, J.; Capodaglio, A.G. Energy recovery and efficiency improvement for an activated sludge, agro-food WWTP upgrade. Water Pract. Technol. 2018, 13, 909–921. [Google Scholar] [CrossRef]
- Boguniewicz-Zablocka, J.; Klosok-Bazan, I.; Callegari, A.; Capodaglio, A.G. Snack-food industry effluent pre-treatment for annatto dye and yeast removal: Process improvement for effectiveness and sustainability. J. Clean. Prod. 2020, 277, 124117. [Google Scholar] [CrossRef]
- Su, Q.; Zhang, J.; Wang, X.; Li, Y.; Lin, S.; Han, J. Adsorption removal of copper (II) and chromium (VI) from wastewater by Fe3O4-loaded granular activated carbon. Water Pract. Technol. 2024, 19, 99–112. [Google Scholar] [CrossRef]
- Yu, Z.; Han, H.; Feng, P.; Zhao, S.; Zhou, T.; Kakade, A.; Kulshrestha, S.; Majeed, S.; Li, X. Recent Advances in the Recovery of Metals from Waste through Biological Processes. Bioresour. Technol. 2020, 297, 122416. [Google Scholar] [CrossRef]
- Rout, D.R.; Jena, H.M.; Baigenzhenov, O.; Hosseini-Bandegharaei, A. Graphene-Based Materials for Effective Adsorption of Organic and Inorganic Pollutants: A Critical and Comprehensive Review. Sci. Total Environ. 2023, 863, 160871. [Google Scholar] [CrossRef]
- El-taweel, R.M.; Mohamed, N.; Alrefaey, K.A.; Husien, S.; Abdel-Aziz, A.B.; Salim, A.I.; Mostafa, N.G.; Said, L.A.; Fahim, I.S.; Radwan, A.G. A review of coagulation explaining its definition, mechanism, coagulant types, and optimization models; RSM, and ANN. Curr. Res. Green Sustain. Chem. 2023, 6, 100358. [Google Scholar] [CrossRef]
- Liu, J.; Hou, Y.; Zhou, X.; Xu, X.; Peng, W.; Fan, J.; Zhao, Z. Combination of TiCl3 reduction/coagulation and ceramic membrane filtration for heavy metal complex removal. Sep. Purif. Technol. 2025, 353, 128410. [Google Scholar] [CrossRef]
- Zhang, L.; Qin, L.; Ma, L.; Shen, Z.; Jin, Y.; Chen, S. Treatment of electroplating wastewater using electrocoagulation and integrated membrane. Water Sci. Technol. 2024, 89, 2538–2557. [Google Scholar] [CrossRef]
- Jaradat, A.Q.; Telfah, D.B.; Ismail, R. Heavy metals removal from landfill leachate by coagulation/flocculation process combined with continuous adsorption using eggshell waste materials. Water Sci. Technol. 2021, 84, 3817–3832. [Google Scholar] [CrossRef]
- Twizerimana, P.; Wu, Y. Overview of integrated electrocoagulation-adsorption strategies for the removal of heavy metal pollutants from wastewater. Discov. Chem. Eng. 2024, 4, 14. [Google Scholar] [CrossRef]
- Rosińska, A.; Dąbrowska, L. Influence of type and dose of coagulants on effectiveness of PAH removal in coagulation water treatment. Water Sci. Eng. 2021, 14, 193–200. [Google Scholar] [CrossRef]
- Water Law Act. Act of 20 July 2017—Water Law. Journal of Laws of the Republic of Poland 2017, Item 1566, as Amended. Available online: https://unece.org/sites/default/files/2025-01/frPartyC146_06.06.2022_annex1.pdf (accessed on 14 November 2024).
- Regulation of the Minister of Construction the Conditions to be Met When Discharging Wastewater into Sewerage Systems. Journal of Laws of Poland; 2006; No. 136, Item 964. Available online: https://www.mgsi.gov.rs/en/odsek/law-planning-and-construction (accessed on 14 November 2024).
- PN-EN ISO 10523:2012; Water Quality—Determination of pH. PKN: Warsaw, Poland, 2012.
- PN-EN 27888:1999; Water Quality—Determination of Electrical Conductivity. PKN: Warsaw, Poland, 1999.
- PN-EN ISO 6060:2006; Water Quality—Determination of the Chemical Oxygen Demand (COD). PKN: Warsaw, Poland, 2006.
- ISO 6060:1989; Water Quality—Determination of the Chemical Oxygen Demand. ISO: Geneva, Switzerland, 1989.
- PN-EN ISO 11885:2009; Water Quality—Determination of Selected Elements by Inductively Coupled Plasma Optical Emission Spectrometry (ICP-OES). PKN: Warsaw, Poland, 2009.
- Yan, M.; Wang, D.; Yu, J.; Ni, J.; Edwards, M.; Qu, J. Enhanced Coagulation with Polyaluminum Chlorides: Role of pH/Alkalinity and Speciation. Chemosphere 2008, 71, 1665–1673. [Google Scholar] [CrossRef]
- El Ouadrhiri, F.; Saleh, E.A.M.; Lahkimi, A. From Mineral Salts to Smart Hybrids: Coagulation–Flocculation at the Nexus of Water, Energy, and Resources—A Critical Review. Processes 2025, 13, 3405. [Google Scholar] [CrossRef]
- Naceradska, J.; Pivokonska, L.; Pivokonsky, M. On the Importance of pH Value in Coagulation. J. Water Supply Res. Technol.-Aqua 2019, 68, 222–230. [Google Scholar] [CrossRef]
- Nowacka, A.; Włodarczyk-Makuła, M.; Macherzyński, B. Comparison of Effectiveness of Coagulation with Aluminum Sulfate and Pre-Hydrolyzed Aluminum Coagulants. Desalination Water Treat. 2014, 52, 3843–3851. [Google Scholar] [CrossRef]
- Jiao, R.; Fabris, R.; Chow, C.W.K.; Drikas, M.; van Leeuwen, J.; Wang, D.; Xu, Z. Influence of Coagulation Mechanisms and Floc Formation on Filterability. J. Environ. Sci. 2017, 57, 338–345. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, L.; Elzinga, E.J. A Comparison of the Solubility Products of Layered Me(II)–Al(III) Hydroxides Based on Sorption Studies with Ni(II), Zn(II), Co(II), Fe(II), and Mn(II). Soil Syst. 2018, 2, 20. [Google Scholar] [CrossRef]
- Altmann, J.; Rehfeld, D.; Träder, K.; Sperlich, A.; Jekel, M. Combination of Granular Activated Carbon Adsorption and Deep-Bed Filtration as a Single Advanced Wastewater Treatment Step for Organic Micropollutant and Phosphorus Removal. Water Res. 2016, 92, 131–139. [Google Scholar] [CrossRef]
- Castro Jiménez, C.C.; Grueso-Dominguez, M.C.; Correa, M.; García, E. A Coagulation Process Combined with a Multi-Stage Filtration System for Drinking Water Treatment: An Alternative for Small Communities. Water 2022, 14, 3256. [Google Scholar] [CrossRef]
- Shofia, S.I.; Vickram, A.S.; Saravanan, A.; Deivayanai, V.C.; Yaashikaa, P.R. Sustainable Separation Technologies for Heavy Metal Removal from Wastewater: An Upgraded Review of Physicochemical Methods and Its Advancements. Sustain. Chem. Environ. 2025, 10, 100264. [Google Scholar] [CrossRef]
- Zahmatkesh, S.; Karimian, M.; Chen, Z.; Ni, B.J. Combination of Coagulation and Adsorption Technologies for Advanced Wastewater Treatment for Potable Water Reuse: By ANN, NSGA-II, and RSM. J. Environ. Manag. 2024, 349, 119429. [Google Scholar] [CrossRef]
- Abdel-Shafy, H.I.; Morsy, R.M.; Hewehy, M.A.; Razek, T.M.; Hamid, M.M. Treatment of industrial electroplating wastewater for metals removal via electrocoagulation continous flow reactors. Water Pract. Technol. 2022, 17, 555–566. [Google Scholar] [CrossRef]
- Mariana, M.; Khalil, H.P.S.A.; Mistar, E.M.; Yahya, E.B.; Alfatah, T.; Danish, M.; Amayreh, M. Recent advances in activated carbon modification techniques for enhanced heavy metal adsorption. J. Water Process Eng. 2021, 43, 102221. [Google Scholar] [CrossRef]
- Joshi, A.A.; Ragupathy, G. Performance and Mechanisms of Waste-Based Carbon Adsorbents in Heavy Metal Removal–An Experimental and Theoretical Approach. RSC Adv. 2025, 15, 34609–34634. [Google Scholar] [CrossRef]
- Chakraborty, R.; Asthana, A.; Singh, A.K.; Jain, B.; Susan, A.B.H. Adsorption of heavy metal ions by various low-cost adsorbents: A review. Int. J. Environ. Anal. Chem. 2020, 102, 342–379. [Google Scholar] [CrossRef]
- Pet, I.; Sanad, M.N.; Farouz, M.; ElFaham, M.M.; El-Hussein, A.; Abd El-Sadek, M.S.; Althobiti, R.A.; Ioanid, A. Recent developments in the implementation of activated carbon as heavy metal removal management. Water Conserv. Sci. Eng. 2024, 9, 63. [Google Scholar] [CrossRef]
- Tang, X.; Zheng, H.; Teng, H.; Sun, Y.; Guo, J.; Xie, W.; Yang, Q.; Chen, W. Chemical coagulation process for the removal of heavy metals from water: A review. Desalination Water Treat. 2016, 57, 1733–1748. [Google Scholar] [CrossRef]
- Li, Z.; Du, C. Current status and research trends of electroplating wastewater treatment: Systematic review and bibliometric analysis. Desalination 2025, 613, 119004. [Google Scholar] [CrossRef]
- Xiao, X.; Sun, Y.; Liu, J.; Zheng, H. Flocculation of heavy metal by functionalized starch-based bioflocculants: Characterization and process evaluation. Sep. Purif. Technol. 2021, 267, 118628. [Google Scholar] [CrossRef]
- Sun, Y.; Zhou, S.; Sun, W.; Zhu, S.; Zheng, H. Flocculation activity and evaluation of chitosan-based flocculant CMCTS-g-P(AM CA) for heavy metal removal. Sep. Purif. Technol. 2020, 241, 116737. [Google Scholar] [CrossRef]
- Sun, Y.; Chen, A.; Pan, S.Y.; Sun, W.; Zhu, C.; Shah, K.J.; Zheng, H. Novel chitosan-based flocculants for chromium and nickel removal in wastewater via integrated chelation and flocculation. J. Environ. Manag. 2019, 248, 109241. [Google Scholar] [CrossRef] [PubMed]
- López-Maldonado, E.A.; Zavala García, O.G.; Escobedo, K.C.; Oropeza-Guzman, M.T. Evaluation of the chelating performance of biopolyelectrolyte green complexes (NIBPEGCs) for wastewater treatment from the metal finishing industry. J. Hazard. Mater. 2017, 335, 18–27. [Google Scholar] [CrossRef]
- Thomas, M.; Melichová, Z.; Šuránek, M.; Łochyński, P. Removal of Zinc from Concentrated Galvanic Wastewater by Sodium Trithiocarbonate: Process Optimization and Toxicity Assessment. Molecules 2023, 28, 546. [Google Scholar] [CrossRef] [PubMed]
- Kowalik-Klimczak, A. Removal of Heavy Metals from Galvanic Industry Wastewater: A Review of Different Possible Methods. Sustainability 2025, 17, 8562. [Google Scholar] [CrossRef]


| Parameter | Unit | S1 | S2 | S3 |
|---|---|---|---|---|
| pH | - | 4.1 ± 0.7 | 7.0 ± 0.4 | 6.7 ± 0.7 |
| Conductivity | mS/cm | 26.0 ± 6.4 | 39.0 ± 8.3 | 25.0 ± 7.2 |
| COD | mg/L | 440 ± 56 | 505 ± 78 | 494 ± 53 |
| Chromium | mg/L | 2.50 ± 0.17 | – | 2.00 ± 0.16 |
| Nickel | mg/L | 0.10 ± 0.02 | 0.90 ± 0.42 | 1.00 ± 0.35 |
| Copper | mg/L | – | 0.78 ± 0.36 | 0.80 ± 0.34 |
| Zinc | mg/L | – | 2.90 ± 0.21 | 2.90 ± 0.20 |
| WW Stream | pH | Cr [mg/L] | Cu [mg/L] | Ni [mg/L] | Zn [mg/L] |
|---|---|---|---|---|---|
| WWS 1 | 6.0 | 2.66 | 0.06 | 0.10 | 0.02 |
| WWS 2 | 8.7 | 0.02 | 0.80 | 0.98 | 2.90 |
| WWS 3 | 7.6 | 2.20 | 0.70 | 0.99 | 2.94 |
| Metal | Coagulation PIX/PAX | Coagulation PIX or PAX + GAC | Dominant Removal Mechanisms | Literature Data with Key References |
|---|---|---|---|---|
| Ni | 70/89% | 91% | Precipitation as Ni(OH)2; adsorption on GAC via surface complexation | 90% [7,28,29] |
| Zn | 83/89% | 92% | Zn(OH)2 precipitation; strong affinity to activated carbon; micropore uptake | 90% [7,30,31] |
| Cu | 80/92% | 93% | Hydroxo-complex adsorption; interaction with oxygen groups on GAC | 75% [16,27,30] |
| Cr | 94/80% | 95% | Precipitation of Cr(OH)3; sweep flocculation; minimal gain after GAC | 80–95% [18,26,30] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Boguniewicz-Zabłocka, J.; Corpuz, M.V.A.; Naddeo, V. Physicochemical Treatment of Electroplating Wastewater: Efficiency Evaluation and Process Optimization. Processes 2026, 14, 182. https://doi.org/10.3390/pr14020182
Boguniewicz-Zabłocka J, Corpuz MVA, Naddeo V. Physicochemical Treatment of Electroplating Wastewater: Efficiency Evaluation and Process Optimization. Processes. 2026; 14(2):182. https://doi.org/10.3390/pr14020182
Chicago/Turabian StyleBoguniewicz-Zabłocka, Joanna, Mary V. A. Corpuz, and Vincenzo Naddeo. 2026. "Physicochemical Treatment of Electroplating Wastewater: Efficiency Evaluation and Process Optimization" Processes 14, no. 2: 182. https://doi.org/10.3390/pr14020182
APA StyleBoguniewicz-Zabłocka, J., Corpuz, M. V. A., & Naddeo, V. (2026). Physicochemical Treatment of Electroplating Wastewater: Efficiency Evaluation and Process Optimization. Processes, 14(2), 182. https://doi.org/10.3390/pr14020182








