Functional Characterization of Scaptotrigona mexicana Honey: Physicochemical Properties, Antioxidant Capacity, and α-Amylase Inhibition for Food Process Applications
Abstract
1. Introduction
2. Materials and Methods
2.1. Honey Samples
2.2. Reagents
2.3. Physicochemical Analyses
2.3.1. Moisture and Ash
2.3.2. Diastase Activity
2.3.3. Hydroxymethylfurfural
2.3.4. Reducing Sugars
2.3.5. Free Acidity, Lactone, and Total Acidity
2.3.6. Brix
2.3.7. H2O Activity and pH
2.3.8. Protein
2.3.9. Color
2.4. Bioactive Compounds
2.4.1. Total Phenolic Compounds
2.4.2. Total Flavonoids
2.5. Antioxidant Assays
2.5.1. DPPH (1,1-Diphenyl-2-Picrylhydrazyl) Free Radical-Scavenging Assay
2.5.2. ABTS (2,2-Azino-Bis(3-Ethylbenzthiazoline-6-Sulfonic Acid)) Free Radical Scavenging Assay
2.6. α-Amylase Inhibition In Vitro Assay
Kinetics of α-Amylase Inhibition
2.7. Data Analysis
3. Results and Discussion
3.1. Physicochemical Analysis
3.1.1. Moisture
3.1.2. Ash
3.1.3. Diastase Activity
3.1.4. Hydroxymethylfurfural
3.1.5. Reducing Sugars
3.1.6. Free Acidity, Lactone, and Total Acidity
3.1.7. Brix
3.1.8. Water Activity (Aw)
3.1.9. pH
3.1.10. Protein
3.1.11. Color
3.2. Bioactive Compounds
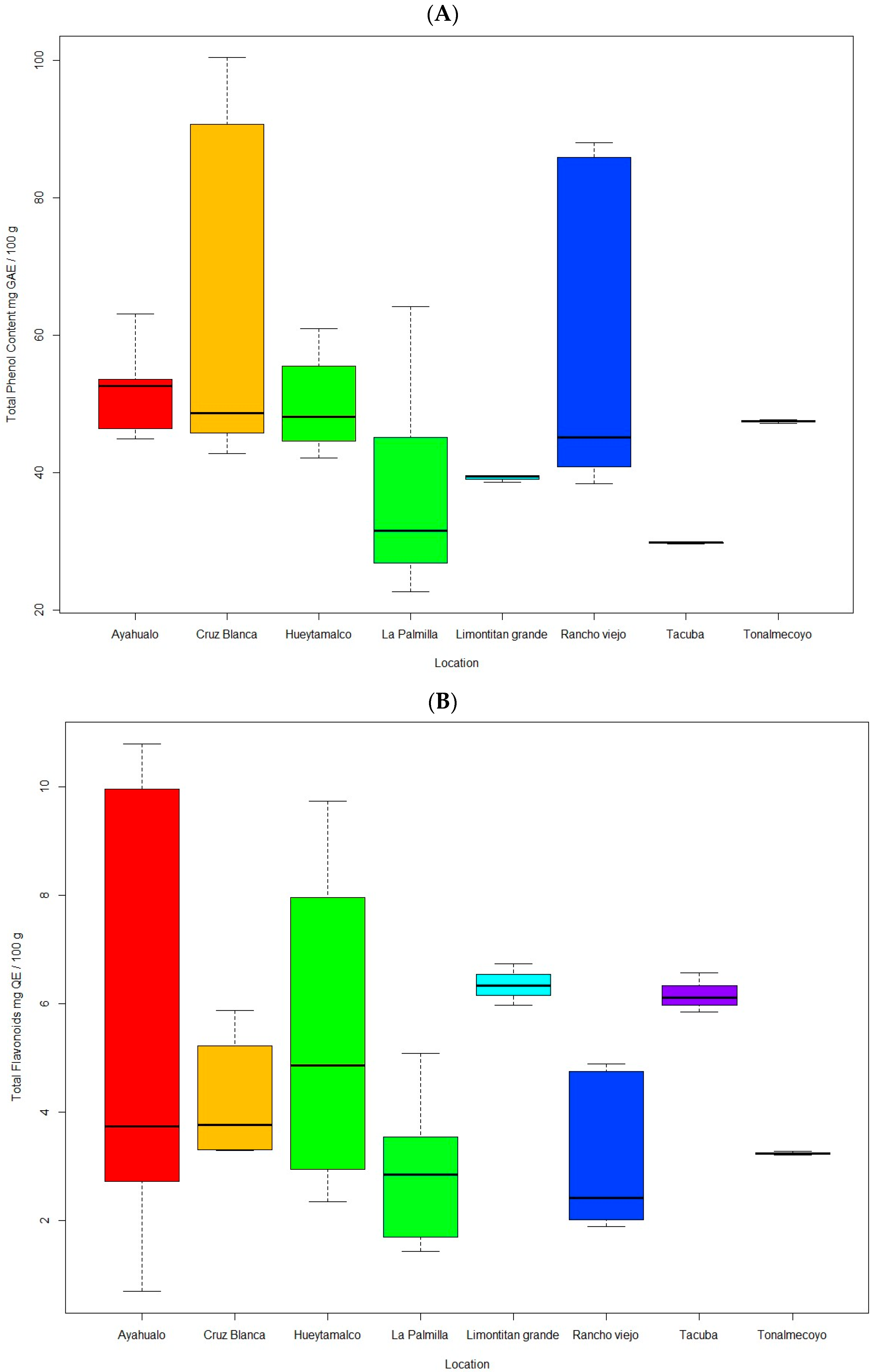
3.2.1. Phenols
3.2.2. Flavonoids
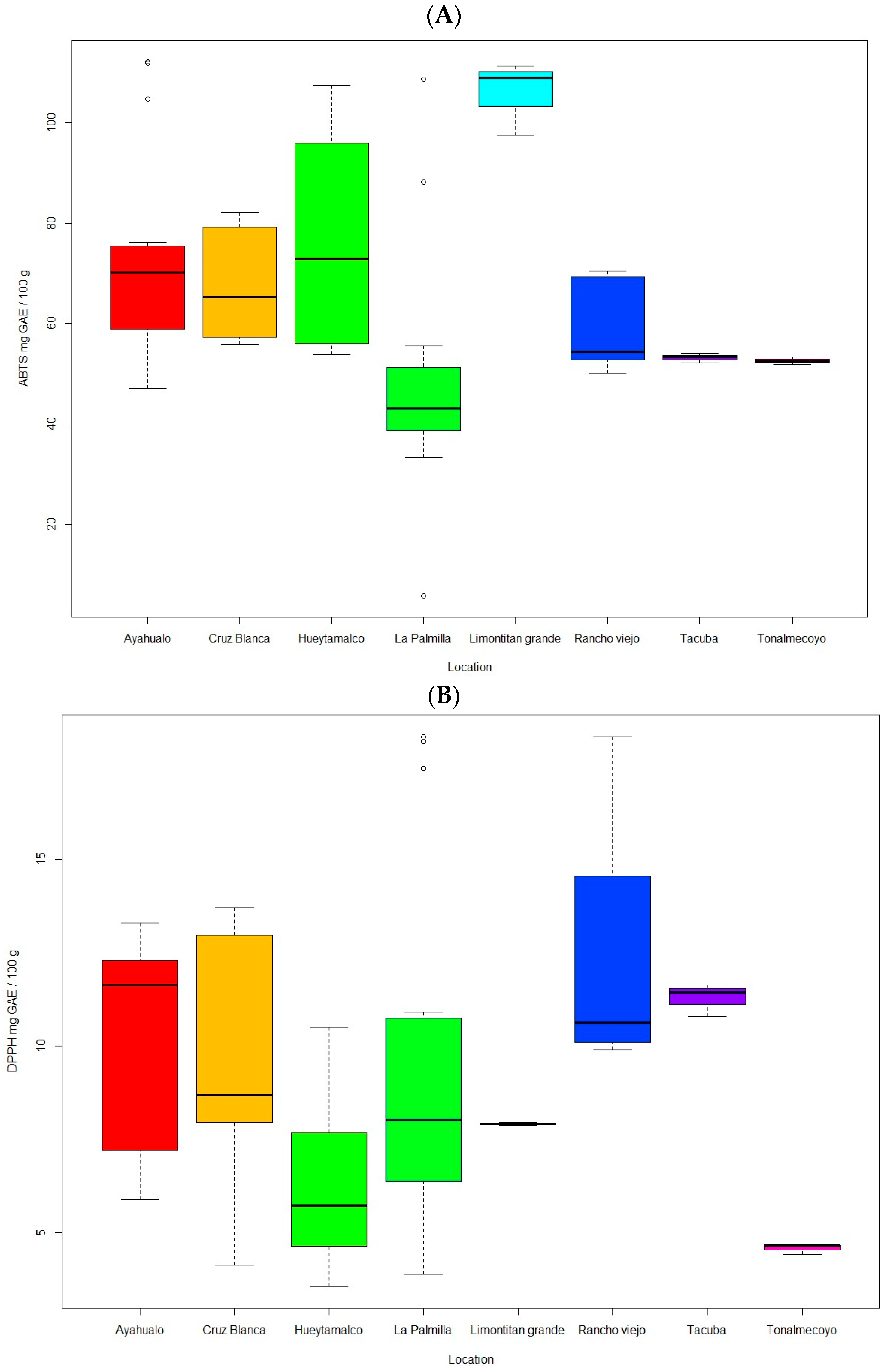
3.2.3. Antioxidants (DPPH and ABTS)
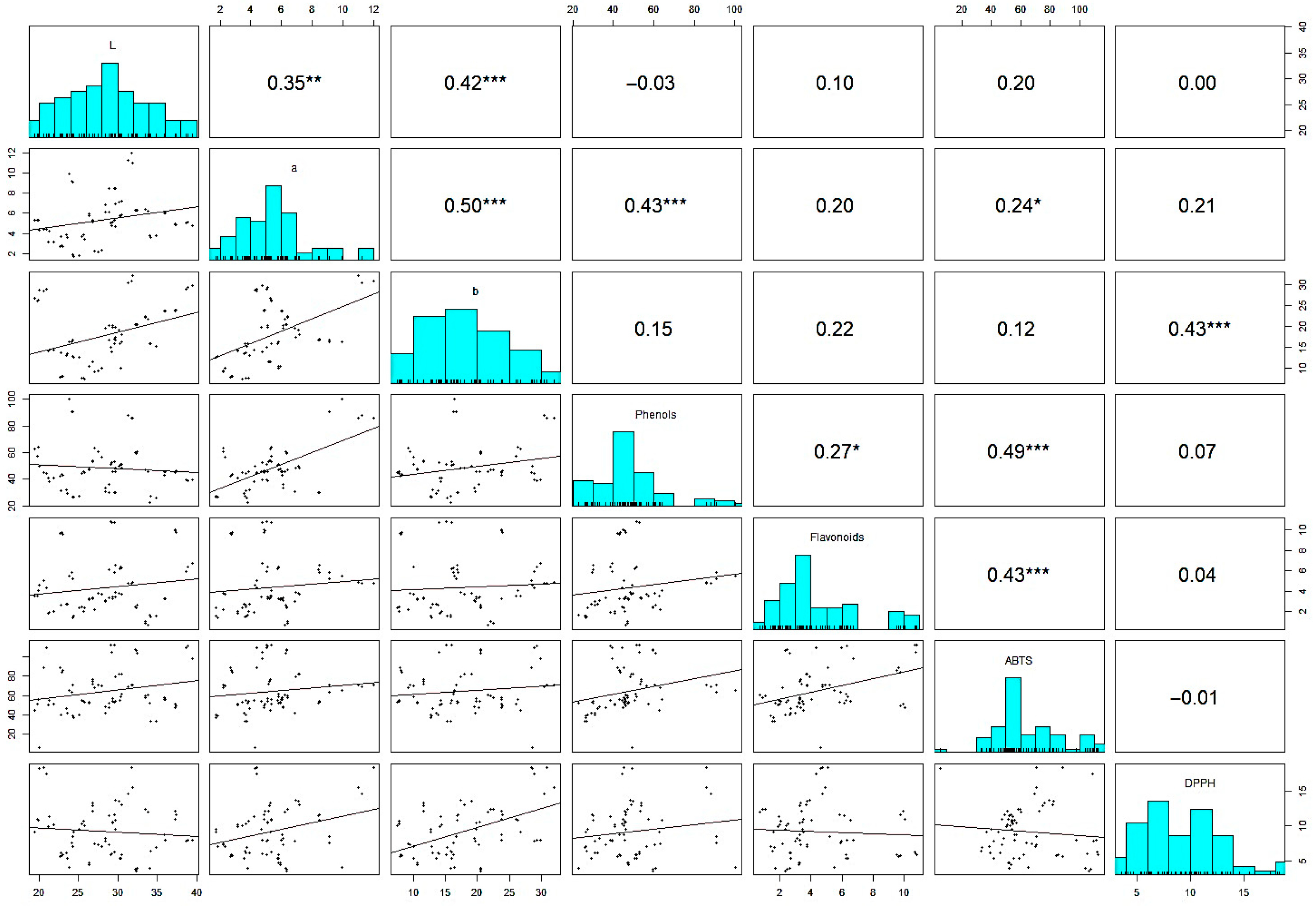
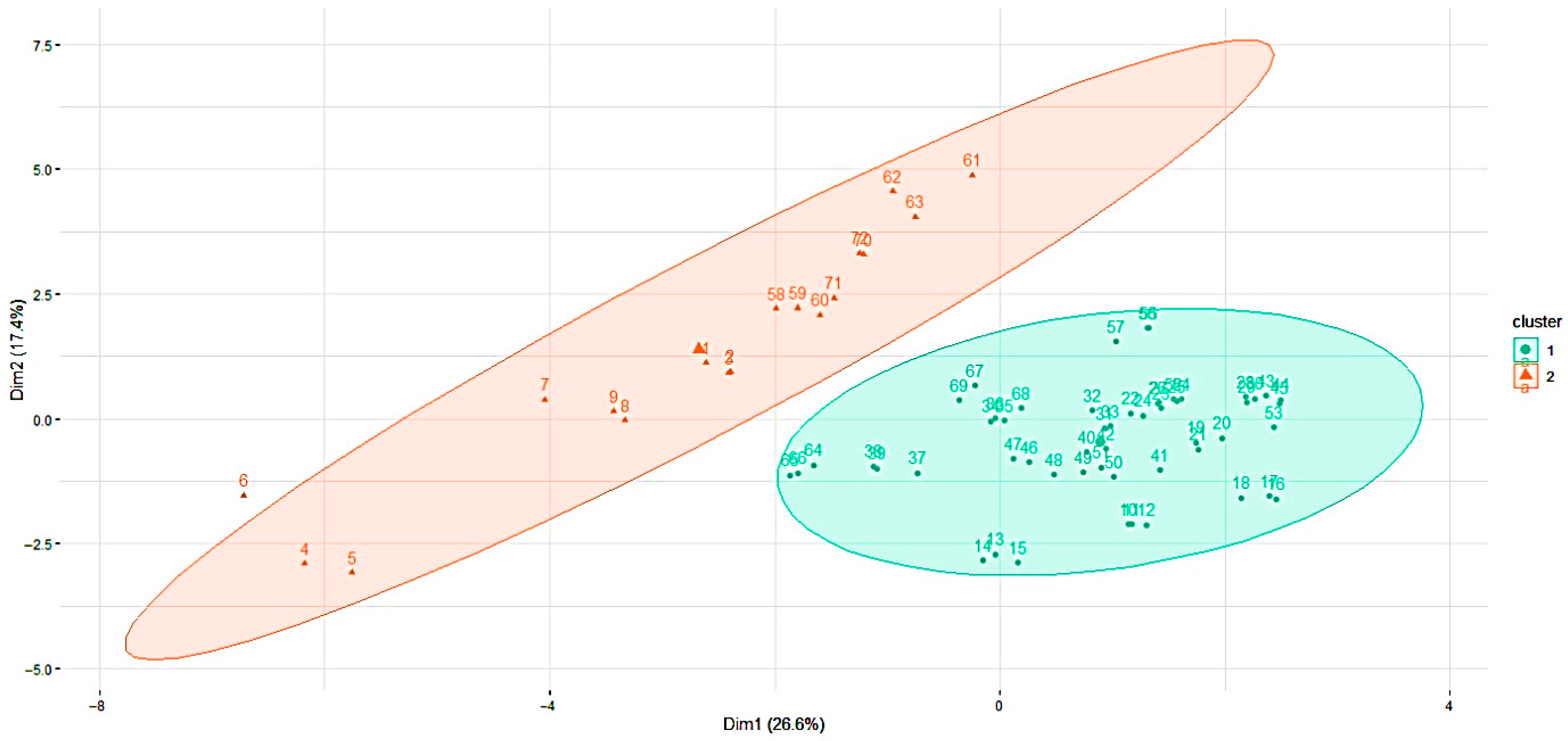
3.2.4. Scatter Plot Matrix and Spearman’s Correlation
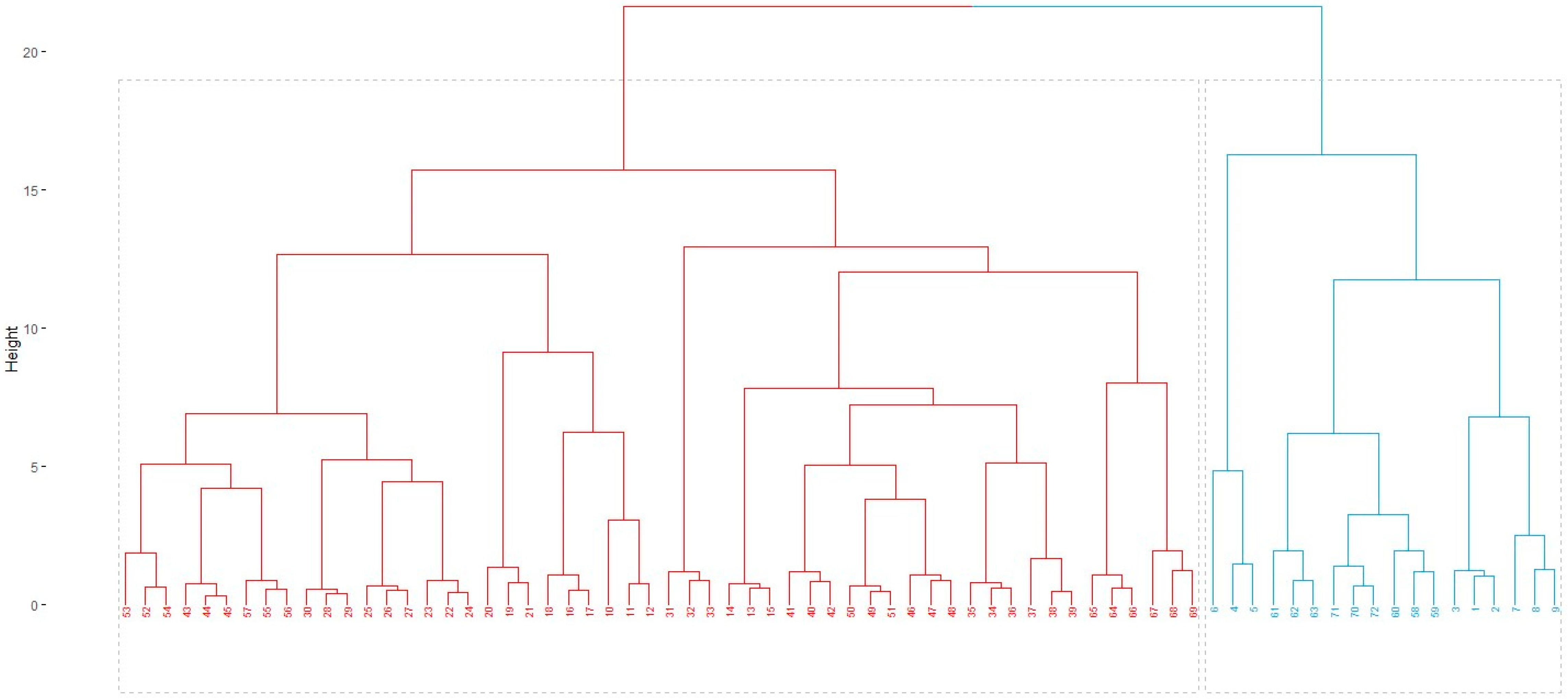
3.2.5. Hierarchical Cluster Analysis
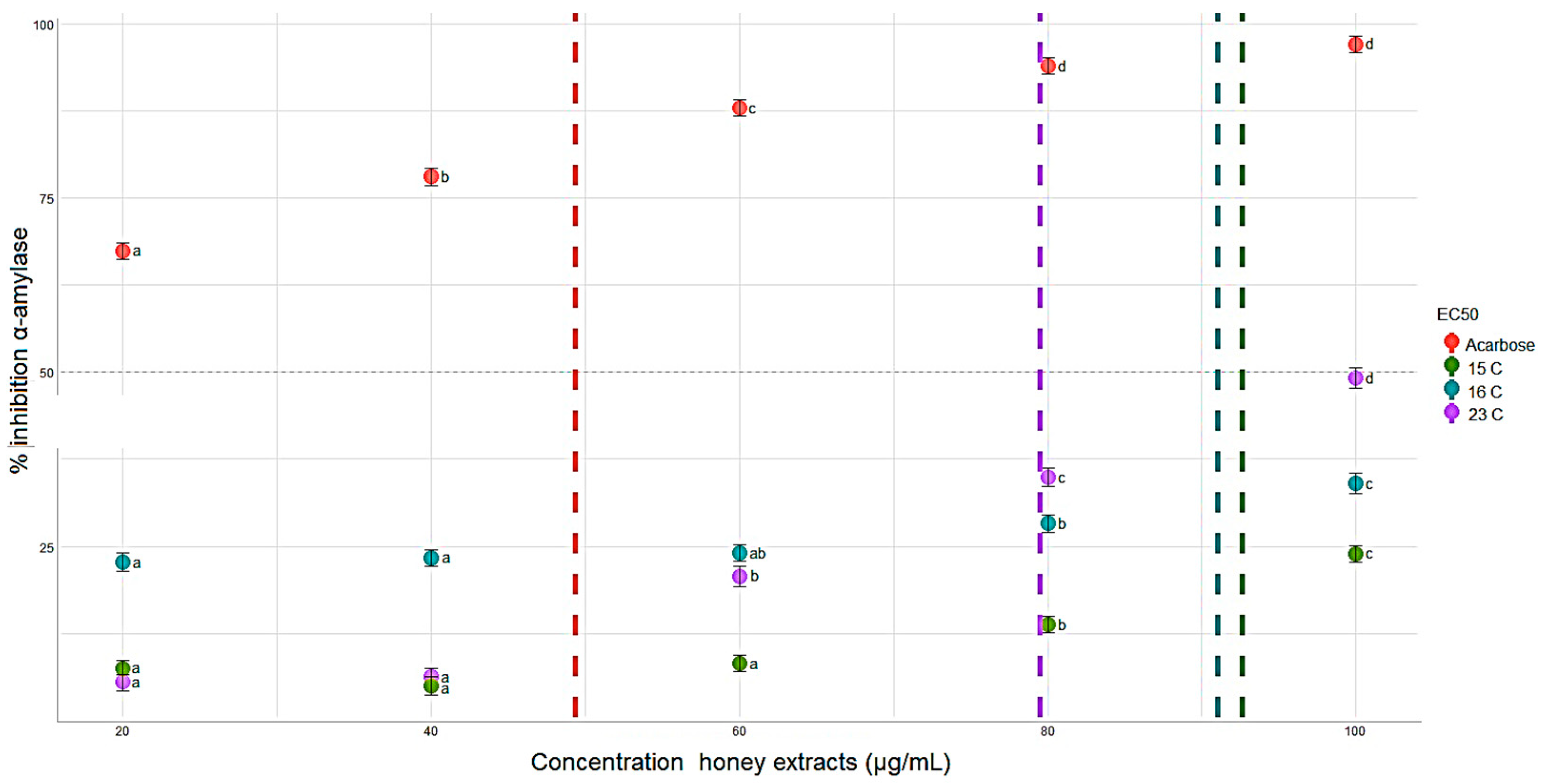
3.2.6. α-Amylase Inhibition In Vitro Assay
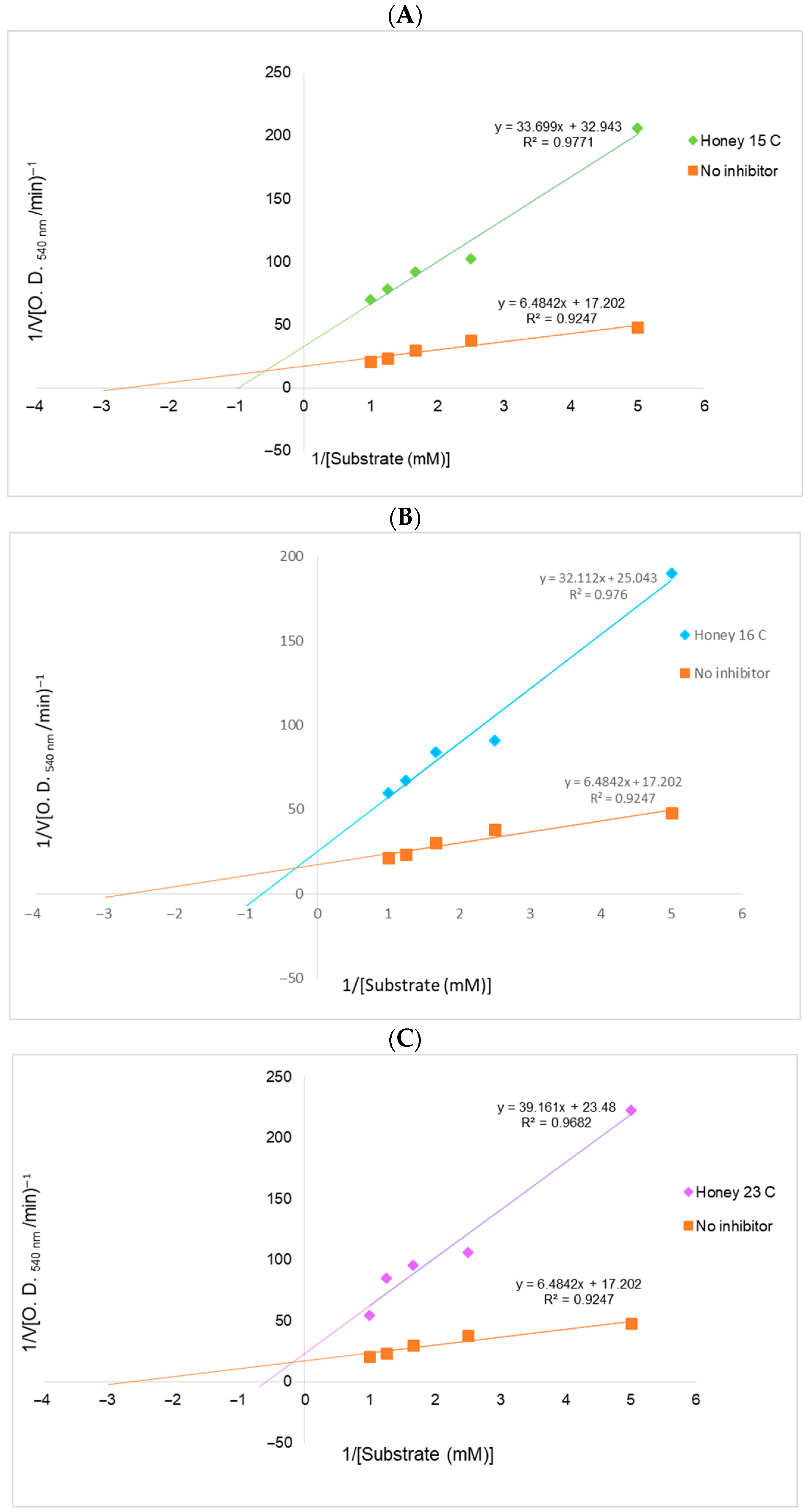
3.2.7. Kinetics of α-Amylase Inhibition
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Erejuwa, O.O.; Sulaiman, S.A.; Ab Wahab, M.S. Honey: A novel antioxidant. Molecules 2012, 17, 4400–4423. [Google Scholar] [CrossRef] [PubMed]
- Rozman, A.S.; Hashim, N.; Maringgal, B.; Abdan, K. A Comprehensive Review of Stingless Bee Products: Phytochemical Composition and Beneficial Properties of Honey, Propolis, and Pollen. Appl. Sci. 2022, 12, 6370. [Google Scholar] [CrossRef]
- Cheng, M.Z.S.Z.; Amin, F.A.Z.; Zawawi, N.; Chan, K.W.; Ismail, N.; Ishak, N.A.; Esa, N.M. Stingless Bee (Heterotrigona Itama) Honey and Its Phenolic-Rich Extract Ameliorate Oxidant–Antioxidant Balance via KEAP1-NRF2 Signalling Pathway. Nutrients 2023, 15, 2835. [Google Scholar] [CrossRef] [PubMed]
- Gerginova, D.; Popova, M.; Chimshirova, R.; Trusheva, B.; Shanahan, M.; Guzmán, M.; Bankova, V. The chemical composition of Scaptotrigona mexicana honey and propolis collected in two locations: Similarities and differences. Foods 2023, 12, 3317. [Google Scholar] [CrossRef]
- Kek, S.P.; Chin, N.L.; Yusof, Y.A.; Tan, S.W.; Chua, L.S. Total phenolic contents and colour intensity of Malaysian honeys from the Apis spp. and Trigona spp. bees. Agric. Agric. Sci. Procedia 2014, 2, 150–155. [Google Scholar] [CrossRef]
- Ali, H.; Abu Bakar, M.F.; Majid, M.; Muhammad, N.; Lim, S.Y. In vitro anti-diabetic activity of stingless bee honey from different botanical origins. Food Res. 2020, 4, 1421–1426. [Google Scholar] [CrossRef]
- Abd Jalil, M.A.; Kasmuri, A.R.; Hadi, H. Stingless bee honey, the natural wound healer: A review. Ski. Pharmacol. Physiol. 2017, 30, 66–75. [Google Scholar] [CrossRef]
- Xolalpa-Aroche, A.; Hernández-Mena, D.I.; Moguel-Chin, W.I.; Contreras-Peruyero, H.; Rivero-Cruz, B.E.; Ortiz-Vázquez, E.; Delgado-Suárez, E.J. Propiedades fisicoquímicas de dos mieles de abejas sin aguijón mexicanas para fortalecer su valor biocultural. Rev. Bras. Farmacog. 2024, 34, 1129–1139. [Google Scholar] [CrossRef]
- Jimenez, M.; Beristain, C.I.; Azuara, E.; Mendoza, M.R.; Pascual, L.A. Physicochemical and antioxidant properties of honey from Scaptotrigona mexicana bee. J. Apic. Res. 2016, 55, 151–160. [Google Scholar] [CrossRef]
- Nordin, A.; Sainik, N.Q.A.V.; Chowdhury, S.R.; Saim, A.B.; Idrus, R.B.H. Physicochemical properties of stingless bee honey from around the globe: A comprehensive review. J. Food Compos. Anal. 2018, 73, 91–102. [Google Scholar] [CrossRef]
- Ahmed, M.; Djebli, N.; Aissat, S.; Khiati, B.; Meslem, A.; Bacha, S. In vitro activity of natural honey alone and in combination with curcuma starch against Rhodotorula mucilaginosa in correlation with bioactive compounds and diastase activity. Asian Pac. J. Trop. Biomed. 2013, 3, 816–821. [Google Scholar] [CrossRef]
- Mokaya, H.O.; Nkoba, K.; Ndunda, R.M.; Vereecken, N.J. Characterization of honeys produced by sympatric species of Afrotropical stingless bees (Hymenoptera, Meliponini). Food Chem. 2022, 366, 130597. [Google Scholar] [CrossRef]
- Chuttong, B.; Chanbang, Y.; Sringarm, K.; Burgett, M. Physicochemical profiles of stingless bee (Apidae: Meliponini) honey from South east Asia (Thailand). Food Chem. 2016, 192, 149–155. [Google Scholar] [CrossRef]
- Tuksitha, L.; Chen, Y.L.S.; Chen, Y.L.; Wong, K.Y.; Peng, C.C. Antioxidant and antibacterial capacity of stingless bee honey from Borneo (Sarawak). J. Asia-Pac. Entomol. 2018, 21, 563–570. [Google Scholar] [CrossRef]
- Ruiz-Ruiz, J.C.; Matus-Basto, A.J.; Acereto-Escoffié, P.; Segura-Campos, M.R. Antioxidant and anti-inflammatory activities of phenolic compounds isolated from Melipona beecheii honey. Food Agric. Immunol. 2017, 28, 1424–1437. [Google Scholar] [CrossRef]
- Sousa, J.M.; De Souza, E.L.; Marques, G.; Meireles, B.; de Magalhães Cordeiro, Â.T.; Gullón, B.; Magnani, M. Polyphenolic profile and antioxidant and antibacterial activities of monofloral honeys produced by Meliponini in the Brazilian semiarid region. Food Res. Int. 2016, 84, 61–68. [Google Scholar] [CrossRef]
- Zaldivar-Ortega, A.K.; Cenobio-Galindo, A.D.J.; Morfin, N.; Aguirre-Álvarez, G.; Campos-Montiel, R.G.; Esturau-Escofet, N.; Angeles-Hernandez, J.C. The physicochemical parameters, phenolic content, and antioxidant activity of honey from stingless bees and Apis mellifera: A systematic review and meta-analysis. Antioxidants 2024, 13, 1539. [Google Scholar] [CrossRef] [PubMed]
- Ja’afar, N.L.; Ab Aziz, N.A.; Zakaria, Z.; Mustapha, M.; Mohamed, M.; Mustafa, M.Z.; Hashim, S.A. Narrative review on the physicochemical profiles, bioactive compounds, and therapeutic potentials of stingless bee honey. Discov. Food 2025, 5, 150. [Google Scholar] [CrossRef]
- Thummajitsakul, S.; Boonburapong, B.; Silprasit, K. Antioxidant and Antidiabetic Effects of Garcinia schomburgkiana Extracts and Fermented Juices. Pertanika J. Trop. Agric. Sci. 2019, 45. [Google Scholar]
- Aziz, M.S.A.; Giribabu, N.; Rao, P.V.; Salleh, N. Pancreatoprotective effects of Geniotrigona thoracica stingless bee honey in streptozotocin-nicotinamide-induced male diabetic rats. Biomed. Pharmacother. 2017, 89, 135–145. [Google Scholar] [CrossRef]
- Sabiu, S.; O’neill, F.H.; Ashafa, A.O.T. Kinetics of α-amylase and α-glucosidase inhibitory potential of Zea mays Linnaeus (Poaceae), Stigma maydis aqueous extract: An in vitro assessment. J. Ethnopharmacol. 2016, 183, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Kotowaroo, M.I.; Mahomoodally, M.F.; Gurib-Fakim, A.; Subratty, A.H. Screening of traditional antidiabetic medicinal plants of mauritius for possible α-amylase inhibitory effects in vitro. Phytother. Res. Int. J. Pharmacol. Toxicol. Nat. Prod. Deriv. 2006, 20, 228–231. [Google Scholar] [CrossRef] [PubMed]
- Noh, Y.H.; Lee, D.B.; Lee, Y.W.; Pyo, Y.H. In vitro inhibitory effects of organic acids identified in commercial vinegars on α-amylase and α-glucosidase. PNF 2020, 25, 319. [Google Scholar] [CrossRef]
- Sun, H.; Saeedi, P.; Karuranga, S.; Pinkepank, M.; Ogurtsova, K.; Duncan, B.B.; Magliano, D.J. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Prac. 2022, 183, 109119. [Google Scholar] [CrossRef]
- Lim, D.C.C.; Abu Bakar, M.F.; Majid, M. Nutritional composition of stingless bee honey from different botanical origins. IOP Conf. Ser. Earth Environ. Sci. 2019, 269, 012025. [Google Scholar] [CrossRef]
- de Almeida-Muradian, L.B.; Stramm, K.M.; Horita, A.; Barth, O.M.; da Silva de Freitas, A.; Estevinho, L.M. Comparative study of the physicochemical and palynological characteristics of honey from Melipona subnitida and Apis mellifera. Int. J. Food Sci. Technol. 2013, 48, 1698–1706. [Google Scholar] [CrossRef]
- Sebaugh, J.L. Guidelines for accurate EC50/IC50 estimation. Pharm. Stat. 2011, 10, 128–134. [Google Scholar] [CrossRef]
- Gilbert-Kawai, E.T.; Wittenberg, M.D. Michaelis–Menten equation. In Essential Equations for Anaesthesia: Key Clinical Concepts for the FRCA and EDA; Cambridge University Press: Cambridge, UK, 2014; pp. 75–76. [Google Scholar]
- Slater, E.C. Leonor Michaelis in Japan. IUBMB Life 2006, 58, 376–377. [Google Scholar] [CrossRef]
- Association of Official Analytical Chemists (AOAC). Determination of moisture, ash, protein and fat. In Official Method of Analysis, 18th ed.; AOAC International: Gaithersburg, MD, USA, 2005. [Google Scholar]
- Secretaría de Agricultura y Desarrollo Rural. NOM-004-SAG/GAN-2018: Producción de Miel y Especificaciones. Diario Oficial de la Federación. 2018. Available online: https://www.dof.gob.mx (accessed on 28 June 2025).
- Habib, H.M.; Al Meqbali, F.T.; Kamal, H.; Souka, U.D.; Ibrahim, W.H. Physicochemical and biochemical properties of honeys from arid regions. Food Chem. 2014, 153, 35–43. [Google Scholar] [CrossRef]
- Folin, O.; Ciocalteu, V. On tyrosine and tryptophane determinations in proteins. J. Biol. Chem. 1927, 73, 627–650. [Google Scholar] [CrossRef]
- Chang, C.C.; Yang, M.H.; Wen, H.M.; Chern, J.C. Estimation of total flavonoid content in propolis by two complementary colorimetric methods. J. Food Drug Anal. 2002, 10, 178–182. [Google Scholar]
- Brand-Williams, W.; Cuvelier, M.E.; Berset, C.L.W.T. Use of a free radical method to evaluate antioxidant activity. LWT-Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef] [PubMed]
- Abirami, A.; Nagarani, G.; Siddhuraju, P. In vitro antioxidant, anti-diabetic, cholinesterase and tyrosinase inhibitory potential of fresh juice from Citrus hystrix and C. maxima fruits. Food Sci. Hum. Well. 2014, 3, 16–25. [Google Scholar] [CrossRef]
- Thummajitsakul, S.; Suppasat, T.; Silprasit, K. Glucosidase inhibition and compound identification of stingless bee honey and preserved fruits of Citrus japonica. Heliyon 2024, 10, e29740. [Google Scholar] [CrossRef]
- Gela, A.; Hora, Z.A.; Kebebe, D.; Gebresilassie, A. Physico-chemical characteristics of honey produced by stingless bees (Meliponula beccarii) from West Showa zone of Oromia Region, Ethiopia. Heliyon 2021, 7, e05875. [Google Scholar] [CrossRef]
- Codex Alimentarius Committee on Sugars. Codex standard 12, Revised Codex Standard for Honey. Stand. Stand. Methods 2001, 11, 1–7. [Google Scholar]
- Sousa, J.M.B.; Aquino, I.S.; Magnani, M.; de Albuquerque, J.R.; Dos Santos, G.G.; De Souza, E.L. Physicochemical aspects and sensory profile of stingless bee honeys from Serido ’ region, State of Rio Grande do Norte, Brazil. Semina Cienc. Agrar. 2013, 34, 1765–1774. [Google Scholar] [CrossRef][Green Version]
- Mohammed, M.E.A. Factors affecting the physicochemical properties and chemical composition of bee’s honey. Food Rev. Int. 2022, 38, 1330–1341. [Google Scholar] [CrossRef]
- Agus, A.; Agussalim, A.; Sahlan, M.; Sabir, A. Honey sugars profile stingless bee Tetragonula laeviceps (Hymenoptera: Meliponinae). BJBD 2021, 22, 5205–5210. [Google Scholar] [CrossRef]
- Saxena, S.; Gautam, S.; Sharma, A. Physical, biochemical and antioxidant properties of some Indian honeys. Food Chem. 2010, 118, 391–397. [Google Scholar] [CrossRef]
- Alves, A.; Ramos, A.; Gonçalves, M.M.; Bernardo, M.; Mendes, B. Antioxidant activity, quality parameters and mineral content of Portuguese monofloral honeys. J. Food Compos. Anal. 2013, 30, 130–138. [Google Scholar] [CrossRef]
- Moo-Huchin, V.M.; González-Aguilar, G.A.; Lira-Maas, J.D.; Pérez-Pacheco, E.; Estrada-León, R.; Moo-Huchin, M.I.; Sauri-Duch, E. Physicochemical properties of Melipona beecheii honey of the Yucatan Peninsula. J. Food Res. 2015, 4, 25. [Google Scholar] [CrossRef][Green Version]
- Marcolin, L.C.; Lima, L.R.; de Oliveira Arias, J.L.; Berrio, A.C.B.; Kupski, L.; Barbosa, S.C.; Primel, E.G. Meliponinae and Apis mellifera honey in southern Brazil: Physicochemical characterization and determination of pesticides. Food Chem. 2021, 363, 130175. [Google Scholar] [CrossRef] [PubMed]
- Alvarez-Suarez, J.M.; Giampieri, F.; Brenciani, A.; Mazzoni, L.; Gasparrini, M.; González-Paramás, A.M.; Battino, M. Apis mellifera vs Melipona beecheii Cuban polifloral honeys: A comparison based on their physicochemical parameters, chemical composition and biological properties. LWT 2018, 87, 272–279. [Google Scholar] [CrossRef]
- Souza, B.; Roubik, D.; Barth, O.; Heard, T.; EnrÍquez, E.; Carvalho, C.; Vit, P. Composition of stingless bee honey: Setting quality standards. Interciencia 2006, 31, 867–875. [Google Scholar]
- Madejczyk, M.; Baralkiewicz, D. Characterization of Polish rape and honeydew honey according to their mineral contents using ICP-MS and F-AAS/AES. Anal. Chim. Acta 2008, 617, 11–17. [Google Scholar] [CrossRef]
- Alqarni, A.S.; Owayss, A.A.; Mahmoud, A.A.; Hannan, M.A. Mineral content and physical properties of local and imported honeys in Saudi Arabia. J. Saudi Chem. Soc. 2014, 18, 618–625. [Google Scholar] [CrossRef]
- da Silva, P.M.; Gauche, C.; Gonzaga, L.V.; Costa, A.C.O.; Fett, R. Honey: Chemical composition, stability and authenticity. Food Chem. 2016, 196, 309–323. [Google Scholar] [CrossRef]
- Alaerjani, W.M.A.; Mohammed, M.E.A. Impact of floral and geographical origins on honey quality parameters in Saudi Arabian regions. Sci. Rep. 2024, 14, 8720. [Google Scholar] [CrossRef]
- Biluca, F.C.; Braghini, F.; Gonzaga, L.V.; Costa, A.C.O.; Fett, R. Physicochemical profiles, minerals and bioactive compounds of stingless bee honey (Meliponinae). J. Food Compos. Anal. 2016, 50, 61–69. [Google Scholar] [CrossRef]
- Eshete, Y. Investigation of The Quality of Domestically Produced Honey, and the Sources, Trends and Quality of the Imported Honey: With Special Reference to Addis Ababa and Surrounding Markets. EC Nut. 2021, 16, 33–40. [Google Scholar] [CrossRef]
- Baroyi, S.A.H.M.; Yusof, Y.A.; Chin, N.L.; Othman, S.H.; Ghazali, N.S.M. A comparative study of high-pressure processing and microwave pasteurisation on the formation of hydroxymethylfurfural in stingless bee (Heterotrigona itama) honey. LCM 2022, 5, 1–14. [Google Scholar] [CrossRef]
- Biluca, F.C.; Della Betta, F.; de Oliveira, G.P.; Pereira, L.M.; Gonzaga, L.V.; Costa, A.C.O.; Fett, R. 5-HMF and carbohydrates content in stingless bee honey by CE before and after thermal treatment. Food Chem. 2014, 159, 244–249. [Google Scholar] [CrossRef] [PubMed]
- Fatima, I.J.; AB, M.H.; Salwani, I.; Lavaniya, M. Physicochemical characteristics of Malaysian stingless bee honey from Trigona species. IIUM Med. J. Malays. 2018, 17, 187–191. [Google Scholar] [CrossRef]
- Sant’ana, R.D.S.; de Carvalho, C.A.L.; Oda-Souza, M.; Souza, B.D.A.; Dias, F.D.S. Characterization of honey of stingless bees from the Brazilian semi-arid region. Food Chem. 2020, 327, 127041. [Google Scholar] [CrossRef] [PubMed]
- Zawawi, N.; Zhang, J.; Hungerford, N.L.; Yates, H.S.; Webber, D.C.; Farrell, M.; Fletcher, M.T. Unique physicochemical properties and rare reducing sugar trehalulose mandate new international regulation for stingless bee honey. Food Chem. 2022, 373, 131566. [Google Scholar] [CrossRef]
- Silva, A.D.S.; Alves, C.N.; Fernandes, K.D.G.; Müller, R. Classification of honeys from Pará State (Amazon region, Brazil) produced by three different species of bees. J. Braz. Chem. Soc. 2013, 24, 1135–1145. [Google Scholar] [CrossRef]
- Fletcher, M.T.; Hungerford, N.L.; Webber, D.; Carpinelli de Jesus, M.; Zhang, J.; Stone, I.S.; Zawawi, N. Stingless bee honey, a novel source of trehalulose: A biologically active disaccharide with health benefits. Sci. Rep. 2020, 10, 12128. [Google Scholar] [CrossRef]
- Instituto Nacional de Estadística y Geografía. (s.f.). Clima en Puebla; INEGI. Available online: https://cuentame.inegi.org.mx/monografias/informacion/pue/territorio/clima.aspx?tema=me&e=21 (accessed on 6 October 2024).
- Clearwater, M.; Revell, M.; Noe, S.; Manley-Harris, M. Influence of genotype, floral stage, and water stress on floral nectar yield and composition of mānuka (Leptospermum scoparium). Ann. Bot. 2018, 121, 501–512. [Google Scholar] [CrossRef]
- Cavia, M.M.; Fernández-Muino, M.A.; Alonso-Torre, S.R.; Huidobro, J.F.; Sancho, M.T. Evolution of acidity of honeys from continental climates: Influence of induced granulation. Food Chem. 2007, 100, 1728–1733. [Google Scholar] [CrossRef]
- Escuredo, O.; Míguez, M.; Fernández-González, M.; Seijo, M.C. Nutritional value and antioxidant activity of honeys produced in a European Atlantic area. Food Chem. 2013, 138, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, V.H.; Oyen, K.; Vitale, N.; Ospina, R. Neotropical stingless bees display a strong response in cold tolerance with changes in elevation. Conserv. Physiol. 2022, 10, coac073. [Google Scholar] [CrossRef] [PubMed]
- López-Garay, L.A.; Trejo-Téllez, L.I.; Gómez-Merino, F.C.; Contreras-Oliva, A.; Pérez-Sato, J.A.; Salinas-Ruiz, J. Physicochemical properties of Scaptotrigona mexicana honey from the Highlands of Veracruz, Mexico. Ecosistemas Recur. Agropecu. 2023, 10, e3380. [Google Scholar] [CrossRef]
- Lage, L.G.; Coelho, L.L.; Resende, H.C.; Tavares, M.G.; Campos, L.A.; Fernandes-Salomão, T.M. Honey physicochemical properties of three species of the brazilian Melipona. An. Acad. Bras. Ciênc. 2012, 84, 605–608. [Google Scholar] [CrossRef]
- Munro, J. A Study of the Factors Influencing the Yield of Honey during the Main Nectar Flow. J. Econ. Entomol. 1929, 22, 518–521. [Google Scholar] [CrossRef]
- Kek, S.P.; Chin, N.L.; Yusof, Y.A.; Tan, S.W.; Chua, L.S. Classification of entomological origin of honey based on its physicochemical and antioxidant properties. Int. J. Food Prop. 2017, 20, S2723–S2738. [Google Scholar] [CrossRef]
- Buba, F.; Gidado, A.; Shugaba, A. Analysis of biochemical composition of honey samples from North-East Nigeria. Biochem Anal Biochem. 2013, 2, 139. [Google Scholar] [CrossRef]
- Chua, L.S.; Abdul-Rahaman, N.L.; Sarmidi, M.R.; Aziz, R. Multi-elemental composition and physical properties of honey samples from Malaysia. Food Chem. 2012, 135, 880–887. [Google Scholar] [CrossRef]
- Shafiee, S.; Minaei, S.; Moghaddam-Charkari, N.; Ghasemi-Varnamkhasti, M.; Barzegar, M. Potential application of machine vision to honey characterization. Trends Food Sci. Technol. 2013, 30, 174–177. [Google Scholar] [CrossRef]
- Bertoncelj, J.; Doberšek, U.; Jamnik, M.; Golob, T. Evaluation of the phenolic content, antioxidant activity and colour of Slovenian honey. Food Chem. 2007, 105, 822–828. [Google Scholar] [CrossRef]
- Becerril-Sánchez, A.L.; Quintero-Salazar, B.; Dublán-García, O.; Escalona-Buendía, H.B. Phenolic compounds in honey and their relationship with antioxidant activity, botanical origin, and color. Antioxidants 2021, 10, 1700. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.W.F.; dos Santos Vasconcelos, M.R.; de Menezes, A.P.D.; da Silva, S.C.; Oda-souza, M.; López, A.M.Q. Composition and antioxidant activity of honey from Africanized and stingless bees in Alagoas (Brazil): A multivariate analysis. J. Apic. Res. 2012, 51, 23–35. [Google Scholar] [CrossRef]
- Makawi, S.Z.A.; Gadkariem, E.A.; Ayoub, S.M.H. Determination of antioxidant flavonoids in Sudanese honey samples by solid phase extraction and high performance liquid chromatography. J. Chem. 2009, 6, S429–S437. [Google Scholar] [CrossRef]
- Pimentel, T.C.; Rosset, M.; de Sousa, J.M.B.; de Oliveira, L.I.G.; Mafaldo, I.M.; Pintado, M.M.E.; Magnani, M. Stingless bee honey: An overview of health benefits and main market challenges. J. Food Biochem. 2022, 46, e13883. [Google Scholar] [CrossRef] [PubMed]
- Cavazza, A.; Corradini, C.; Musci, M.; Salvadeo, P. High-performance liquid chromatographic phenolic compound fingerprint for authenticity assessment of honey. J. Sci. Food Agric. 2013, 93, 1169–1175. [Google Scholar] [CrossRef]
- Das, A.; Datta, S.; Mukherjee, S.; Bose, S.; Ghosh, S.; Dhar, P. Evaluation of antioxidative, antibacterial and probiotic growth stimulatory activities of Sesamum indicum honey containing phenolic compounds and lignans. LWT-Food Sci. Technol. 2015, 61, 244–250. [Google Scholar] [CrossRef]
- Shamsudin, S.; Selamat, J.; Abdul Shomad, M.; Ab Aziz, M.F.; Haque Akanda, M.J. Antioxidant properties and characterization of Heterotrigona itama honey from various botanical origins according to their polyphenol compounds. J. Food Qual. 2022, 2022, 2893401. [Google Scholar] [CrossRef]
- Moreira, F.I.N.; de Medeiros, L.L.; de Carvalho, L.M.; Olegario, L.S.; de Sousa Galvão, M.; da Franca, S.A.M.; Madruga, M.S. Quality of Brazilian stingless bee honeys: Cephalotrigona capitata/mombucão and Melipona scutellaris Latrelle/uruçu. Food Chem. 2023, 404, 134306. [Google Scholar] [CrossRef]
- Suleiman, M.H.; ALaerjani, W.M.A.; Mohammed, M.E.A. Influence of altitudinal variation on the total phenolic and flavonoid content of Acacia and Ziziphus honey. Int. J. Food Prop. 2020, 23, 2077–2086. [Google Scholar] [CrossRef]
- Turkmen, N.; Sari, F.; Poyrazoglu, E.S.; Velioglu, Y.S. Effects of prolonged heating on antioxidant activity and colour of honey. Food Chem. 2006, 95, 653–657. [Google Scholar] [CrossRef]
- de Abreu, I.N.; Mazzafera, P. Effect of water and temperature stress on the content of active constituents of Hypericum brasiliense Choisy. Plant Physiol. Biochem. 2005, 43, 241–248. [Google Scholar] [CrossRef] [PubMed]
- Cirak, C.; Radusiene, J. Factors affecting the variation of bioactive compounds in Hypericum species. Biol. Futura. 2019, 70, 198–209. [Google Scholar] [CrossRef] [PubMed]
- Shakoori, Z.; Salaseh, E.; Mehrabian, A.R.; Tehrani, D.M.; Dardashti, N.F.; Salmanpour, F. The amount of antioxidants in honey has a strong relationship with the plants selected by honey bees. Sci. Rep. 2024, 14, 351. [Google Scholar] [CrossRef]
- Zehiroglu, C.; Ozturk Sarikaya, S.B. The importance of antioxidants and place in today’s scientific and technological studies. J. Food Sci. Technol. 2019, 56, 4757–4774. [Google Scholar] [CrossRef]
- Martinello, M.; Mutinelli, F. Antioxidant Activity in Bee Products: A Review. Antioxidants 2021, 10, 71. [Google Scholar] [CrossRef]
- Mahmood, R.; Asif, J.A.; Shahidan, W.N.S. Stingless-bee (Trigona itama) honey adversely impacts the growth of oral squamous cell carcinoma cell lines (HSC-2). Eur. J. Integr. Med. 2020, 37, 101162. [Google Scholar] [CrossRef]
- Seraglio, S.K.T.; Schulz, M.; Brugnerotto, P.; Silva, B.; Gonzaga, L.V.; Fett, R.; Costa, A.C.O. Quality, composition and health-protective properties of citrus honey: A review. Food Res. Int. 2021, 143, 110268. [Google Scholar] [CrossRef]
- Otmani, A.; Amessis-Ouchemoukh, N.; Birinci, C.; Yahiaoui, S.; Kolaylı, S.; Rodríguez-Flores, M.; Escuredo, O.; Seijo, M.; Ouchemoukh, S. Phenolic compounds and antioxidant and antibacterial activities of Algerian honeys. Food Biosci. 2021, 42, 101070. [Google Scholar] [CrossRef]
- Zakaria, N.N.A.; Jaafar, N.M.; Mohamad, A.Z. Antioxidant, antibacterial and anti-diabetic activities of stingless bee honey from selected areas in Peninsular Malaysia. IOP Conf. Ser. Earth Environ. Sci. 2020, 596, 012093. [Google Scholar] [CrossRef]
- Al-Kafaween, M.A.; Alwahsh, M.; Mohd Hilmi, A.B.; Abulebdah, D.H. Physicochemical characteristics and bioactive compounds of different types of honey and their biological and therapeutic properties: A comprehensive review. Antibiotics 2023, 12, 337. [Google Scholar] [CrossRef]
- Prokisch, J.; El-Ramady, H.; Daróczi, L.; Nagy, É.; Badgar, K.; Kiss, A.; Oláh, C. Functional yogurt fortified with honey produced by feeding bees natural plant extracts for controlling human blood sugar level. Plants 2022, 11, 1391. [Google Scholar] [CrossRef]
- Bogdanov, S. Honey as nutrient and functional food. Proteins 2012, 1100, 1400–2700. [Google Scholar]
| Location | Samples | Latitude, Longitude | Altitude (masl) | Temperature (°C) | Climate | Precipitation (mm) |
|---|---|---|---|---|---|---|
| La Palmilla | 6 | 20.018477, 97.145660 | 147 | 23–32 | Warm-humid | 1500 |
| Ayahualo | 5 | 19.971886, 97.325348 | 603 | 17.5–28.5 | Warm-humid | 1270 |
| Hueytamalco | 4 | 19.940156, 97.288513 | 862 | 17.5–28.5 | Warm-humid | 1270 |
| Rancho viejo | 3 | 19.445556, 96.781389 | 753 | 17.5–28.5 | temperate subhumid | 1270 |
| Cruz Blanca | 3 | 19.893000, 97.288383 | 1238 | 17.5–28.5 | temperate subhumid | 1270 |
| Limontitan grande | 1 | 19.991056, 97.275853 | 543 | 17.5–28.5 | temperate subhumid | 1270 |
| Tonalmecoyo | 1 | 19.954604, 97.290276 | 737 | 17.5–28.5 | temperate subhumid | 1270 |
| Tacuba | 1 | 19.976806, 97.313148 | 533 | 17.5–28.5 | temperate subhumid | 1270 |
| Sample Identification | Moisture (%) | Ash | Diastase Activity (Göthe Units) | HMF (mg/kg) | Reducing Sugars | Free Acidity (meq/kg) |
|---|---|---|---|---|---|---|
| 1 P | 27.07 ± 0.05 abc | 0.54 ± 0.02 abc | 1.66 ± 0.01 ab | 1.61 ± 0.14 ab | 50.42 ± 1.91 a | 13.67 ± 0.58 abc |
| 2 P | 24.76 ± 0.08 a | 0.36 ± 0.01 abc | 28.33 ± 2.89 a | 1.58 ± 0.01 ab | 58.33 ± 0.72 abc | 3.67 ± 0.58 c |
| 3 P | 26.4 ± 0.08 abc | 0.33 ± 0.01 abc | 1.42 ± 0.01 ab | 1.57 ± 0.01 ab | 55 ± 5.73 abc | 20.33 ± 1.53 abc |
| 4 H | 27.29 ± 0.02 abc | 0.85 ± 0.05 b | 1.02 ± 0.02 ab | 1.95 ± 0.5 ab | 68.75 ± 1.25 c | 34 ± 1 abc |
| 5 H | 25.2 ± 0.04 ac | 0.55 ± 0.05 abc | 6.44 ± 0.38 ab | 1.53 ± 0.06 ab | 62.08 ± 0.72 abc | 38.5 ± 0.5 abc |
| 6 A | 28.28 ± 0.07 abc | 0.4 ± 0.01 abc | 1.07 ± 0.02 ab | 1.57 ± 0.01 ab | 62.92 ±0.72 abc | 44 ± 1 abc |
| 7 L | 30.55 ± 0.17 bc | 0.55 ± 0.05 abc | 3.40 ± 0.11 ab | 1.61 ± 0.14 ab | 58.58 ± 0.29 abc | 67.67 ± 0.58 abc |
| 8 H | 28.57 ± 0.05 abc | 0.46 ± 0.05 abc | 1.61 ± 0.05 ab | 1.78 ± 0.05 b | 58.29 ± 0.59 abc | 44.33 ± 1.53 abc |
| 9 T | 28.59 ± 0.1 abc | 0.38 ± 0.03 abc | 1.39 ± 0.04 ab | 1.64 ± 0.01 ab | 63.75 ± 1.25 abc | 50.33 ± 0.58 abc |
| 10 A | 29.05 ± 0.06 abc | 0.19 ± 0.01 abc | 1.01 ± 0.01 ab | 1.72 + 0.04 ab | 58.33 ± 0.72 abc | 66.67 ± 0.58 abc |
| 11 T | 28.46 ± 0.08 abc | 0.6 ± 0.1 abc | 1.54 ± 0.04 ab | 0.05 + 0.01 a | 67.08 ± 1.91 bc | 36.67 ± 1.53 abc |
| 12 H | 28.39 ± 0.04 abc | 0.55 ± 0.05 abc | 7.22 ± 0.48 ab | 1.57 ± 0.06 ab | 51.67 ± 0.72 ab | 47.33 ± 0.58 abc |
| 13 A | 26.04 ± 0.11 abc | 0.65 ± 0.05 abc | 1.08 ± 0.02 ab | 1.64 ± 0.04 ab | 53.58 ± 3.61 abc | 29.83 ± 0.76 abc |
| 14 A | 27.61 ± 0.09 abc | 0.29 ± 0.01 abc | 1.05 ± 0.02 ab | 1.62 ± 0.01 ab | 63.33 ± 1.44 abc | 55.67 ± 1.33 abc |
| 15 C | 28.82± 0.03 abc | 0.16 ± 0.01 abc | 3.22 ± 0.1 ab | 1.68 ± 0.01 ab | 57.92 ± 1.44 abc | 76 ± 1 a |
| 16 C | 27.33 ± 0.02 abc | 0.37 ± 0.05 abc | 1.22 ± 0.01 ab | 1.58 ± 0.01 ab | 59.58 ± 1.44 abc | 33.67 ± 0.58 abc |
| 17 A | 27.11 ± 0.07 abc | 0.42 ± 0.01 abc | 1.62 ± 0.04 ab | 1.56 ± 0.01 ab | 60.83 ± 1.44 abc | 49.33 ± 1.53 abc |
| 18 R | 29.34 ± 0.01 abc | 0.5 ± 0.01 abc | 9.05 ± 0.82 a | 1.75 ± 0.05 ab | 57.50 ± 1.25 abc | 69 ± 2.65 ab |
| 19 R | 29.54 ± 0.19 abc | 0.12± 0.01 c | ND | 1.69 ± 0.01 ab | 57.92 ± 0.72 abc | 49.33 ± 1.15 abc |
| 20 P | 28.09 ± 0.06 abc | 0.19 ± 0.01 abc | 1 ± 0.01 ab | 1.68 ± 0.01 ab | 55 ± 3.3 abc | 15 ± 1 abc |
| 21 P | 32.64 ± 0.87 b | 0.32 ± 0.01 abc | 1 ± 0.01 ab | 1.62 ± 0.01 ab | 57.08 ± 1.44 abc | 9 ± 1 bc |
| 22 R | 26.34 ± 0.11 abc | 0.19 ± 0.01 abc | ND | 1.55 ± 0.01 ab | 54.58 ± 0.72 abc | 40 ± 1 abc |
| 23 C | 29.60 ± 0.07 abc | 0.15 ± 0.01 ac | 1.43 ± 0.01 ab | 1.77 ± 0.08 ab | 55.83 ± 3.15 abc | 48.67 ± 1.53 abc |
| 24 P | 30.10 ± 0.81 abc | 0.17 ± 0.01 abc | 1.25 ± 0.01 ab | 1.67 ± 0.05 ab | 57.08 ± 0.72 abc | 13.67 ± 0.58 abc |
| Overall mean | 28.13 ± 1.75 | 0.39 ± 0.19 | 3.25 ± 5.74 | 1.58 ± 0.35 | 58.60 ± 4.64 | 39.85 ± 19.74 |
| A. mellifera Standards * | <20% | 0.60 g/100g | >3 Schade Units | 40–80 mg/kg | >60 g/100g | 50 meq/kg |
| Min–max | 24.76–32.64 | 0.12–0.85 | 1–28.33 | 0.05–1.95 | 50.42–68.75 | 3.67–76 |
| Kurtosis | 3.58 | 2.7 | 16.5 | 16.8 | 3.03 | 2.21 |
| Asymmetry | 0.34 | 0.49 | 3.66 | −3.27 | 0.21 | −0.08 |
| Variation coefficient | 6.25 | 48.42 | 176.55 | 21.97 | 7.92 | 49.54 |
| Sample | Lactone (meq/kg) | Total Acidity (meq/kg) | Brix | Activity Water (Aw) | pH | Protein (g/100 g) |
|---|---|---|---|---|---|---|
| 1 P | 22.33 ± 1.53 a | 36 ± 2 ab | 72.92 ± 0.06 abc | 0.88 ± 0.006 ab | 3.56 ± 0.02 a | 0.34 ± 0.01 abc |
| 2 P | 17.33 ± 1.04 a | 21 ± 1.5 b | 75.23 ± 0.09 a | 0.89 ± 0.005 b | 4.63 ± 0.06 b | 0.47 ± 0.05 cde |
| 3 P | 8.67 ± 1.53 a | 29 ± 2.65 ab | 73.59 ± 0.08 abc | 0.88 ± 0.01 ab | 3.74 ± 0.01 ab | 0.49 ± 0.05 def |
| 4 H | 29.17 ± 1.04 a | 63.16 ± 1.60 ab | 72.71 ± 0.02 abc | 0.79 ± 0.01 ab | 3.57 ± 0.06 ab | 0.25 ± 0.01 a |
| 5 H | 28.33 ± 1.53 a | 66.83 ± 1.44 ab | 74.83 ± 0.04 ac | 0.77 ± 0.00 ab | 3.48 ± 0.01 ab | 0.61 ± 0.01 fg |
| 6 A | 31 ± 2.65 a | 75 ± 1.73 ab | 71.72 ± 0.07 abc | 0.78 ± 0.01 ab | 3.16 ± 0.01 ab | 0.32 ± 0.02 ab |
| 7 L | 27 ± 2 a | 94.67 ± 2.52 ab | 69.45 ± 0.17 bc | 0.89 ± 0.01 ab | 3.49 ± 0.01 ab | 0.56 ± 0.05 ef |
| 8 H | 27.33 ± 1.53 a | 71.67 ± 3.06 ab | 71.43 ± 0.05 abc | 0.78 ± 0.0 ab | 3.19 ± 0.01 ab | 0.34 ± 0.02 abc |
| 9 T | 25.83 ± 0.7 a | 76.17 ± 0.29 ab | 71.10 ± 0.1 abc | 0.76 ± 0.0 ab | 3.16 ± 0.01 ab | 0.25 ± 0.01 a |
| 10 A | 24.83 ± 0.29 a | 91.50 ± 0.87 ab | 70.94 ± 0.06 abc | 0.77 ± 0.0 ab | 2.95 ± 0.04 ab | 0.33 ± 0.03 ab |
| 11 T | 24.33 ± 0.58 a | 61 ± 2 ab | 71.54 ± 0.08 abc | 0.77 ± 0.0 ab | 3.25 ± 0.01 ab | 0.57 ± 0.03 ef |
| 12 H | 26.67 ± 0.58 a | 74 ± 1 ab | 71.61 + 0.04 abc | 0.78 ± 0.00 ab | 3.41 ± 0.02 ab | 0.84 ± 0.04 h |
| 13 A | 28.67 ± 0.58 a | 58.5 ± 0.87 ab | 73.96 ± 0.11 abc | 0.75 ± 0.00 ab | 3.55 ± 0.01 ab | 0.71 ± 0.03 gh |
| 14 A | 26.50 ± 0.5 a | 82.17 ± 6.53 ab | 72.39 ± 0.09 abc | 0.77 ± 0.00 ab | 3.43 ± 0.03 ab | 0.73 ± 0.04 gh |
| 15 C | 29.33 ± 0.58 a | 105.33 ± 0.58 a | 71.18 ± 0.03 abc | 0.78 ± 0.00 ab | 2.95 ± 0.01 a | 0.46 ± 0.02 bcde |
| 16 C | 28 ± 1 a | 61.67 ± 1.15 ab | 72.67 ± 0.02 abc | 0.75 ± 0.00 a | 3.17 ± 0.02 ab | 0.73 ± 0.1 gh |
| 17 A | 27.50 ± 0.5 a | 76.83 ± 1.76 ab | 72.89 + 0.07 abc | 0.75 ± 0.003 ab | 3.16 ± 0.01 ab | 0.33 ± 0.02 ab |
| 18 R | 29.67 ± 4.73 a | 98.67 ± 6.43 a | 70.66 ± 0.01 abc | 0.77 ±0.001 ab | 3.14 ± 0.02 ab | 0.25 ± 0.01 a |
| 19 R | 25.67 ± 0.58 a | 75 ± 1 ab | 70.45 ± 0.2 abc | 0.75 ± 0.001 ab | 3.11 ± 0.11 ab | 0.39 ± 0.04 bcd |
| 20 P | 18.67 ± 3.79 a | 33.67 ± 4.62 ab | 71.18 ± 0.03 abc | 0.76 ± 0.002 ab | 3.81 ± 0.03 ab | 0.4 ± 0.09 bcd |
| 21 P | 20 ± 1 a | 29 ± 2 ab | 67.35 ± 0.88 b | 0.76 ± 0.001 ab | 3.83 ± 0.02 ab | 0.4 ± 0.04 bcd |
| 22 R | 17.67 ± 1.15 a | 57.67 ± 0.58 ab | 73.66 + 0.12 abc | 0.75 ± 0.001 ab | 3.87 ± 0.03 ab | 0.6 ± 0.01 fg |
| 23 C | 18.67 ± 2.08 a | 67.33 ± 3.21 ab | 70.39 ± 0.07 abc | 0.77 ± 0.002 ab | 3.77 ± 0.04 ab | 1.15 ± 0.06 i |
| 24 P | 18.57 ± 0.58 a | 32.33 ± 0.58 ab | 69.89 ± 0.81 abc | 0.75 ± 0.003 a | 3.92 ± 0.02 b | 0.33 ± 0.03 ab |
| Overall mean | 24.24 ± 5.49 | 64.09 ± 23.32 | 71.87 ± 1.76 | 0.79 ± 0.05 | 3.47 ± 0.38 | 0.08 ± 0.04 |
| Min–Max | 8.67–31 | 21–105.33 | 67.35–75.23 | 0.75–0.9 | 2.95–4.63 | 0.24–1.22 |
| Kurtosis | 3.85 | 2.23 | 3.58 | 3.9 | 4.39 | 4.37 |
| Asymmetry | −0.09 | −0.24 | −0.35 | 1.59 | 1.02 | 1.23 |
| Variation coefficient | 22.66 | 36.39 | 2.44 | 6.03 | 11.01 | 44.27 |
| Sample | Color | ||||
|---|---|---|---|---|---|
| L (Lightness) | a (Red+/Green − Axis) | b (Yellow+/Blue − Axis) | C (Chroma) | H (Hue) | |
| 1 P | 24.76 ± 0.6 ef | 1.8 ± 0.08 a | 12.67 ± 0.25 e | 24.83 ± 0.61 d | 8.12 ± 0.52 a |
| 2 P | 20.52 ± 0.45 b | 4.37 ± 0.06 de | 28.66 ± 0.13 o | 20.98 ± 0.45 ab | 8.68 ± 0.13 a |
| 3 P | 19.6 ± 0.23 a | 5.28 ± 0.06 fg | 26.21 ± 0.38 n | 20.3 ± 0.24 a | 11.38 ± 0.16 b |
| 4 H | 22.96 ± 0.17 d | 2.68 ± 0.06 b | 8.09 ± 0.11 ab | 23.12 ± 0.17 c | 18.36 ± 0.36 gh |
| 5 H | 32.28 ± 0.1 l | 6.25 ± 0.12 hi | 20.44 ± 0.24 k | 32.88 ± 0.11 i | 17.01 ± 0.12 fg |
| 6 A | 29.34 ± 0.21 hi | 5.02 ± 0.23 f | 15.2 ± 0.73 gh | 29.77 ± 0.23 g | 18.29 ± 0.51 gh |
| 7 L | 38.87 ± 0.37 q | 4.99 ± 0.15 f | 29.19 ± 0.43 o | 39.19 ± 0.35 n | 9.71 ± 0.41 ab |
| 8 H | 30.48 ± 0.18 k | 5.77 ± 0.03 gh | 16.18 ± 0.17 hi | 31.02 ± 0.18 h | 19.63 ± 0.19 h |
| 9 T | 29.55 ± 0.39 ij | 8.47 ± 0.03 k | 16.82 ± 0.08 i | 30.74 ± 0.38 h | 26.73 ± 0.14 k |
| 10 A | 37.63 ± 0.41 p | 4.88 ± 0.05 ef | 24.15 ± 0.5 m | 37.94 ± 0.4 m | 11.42 ± 0.27 b |
| 11 T | 26.29 ± 0.13 g | 5.8 ± 0.13 gh | 10.36 ± 0.29 c | 26.93 ± 0.15 f | 29.23 ± 0.31 l |
| 12 H | 29.45 ± 0.17 hi | 5.02 ± 0.2 f | 20.07 ± 0.25 k | 29.87 ± 0.14 g | 14.04 ± 0.5 cd |
| 13 A | 33.49 ± 0.25 m | 6.37 ± 0.11 i | 22.42 ± 0.54 l | 34.09 ± 0.23 jk | 15.87 ± 0.16 ef |
| 14 A | 27.09 ± 0.7 g | 2.21 ± 0.09 ab | 9.1 ± 0.75 b | 27.18 ± 0.71 f | 13.69 ± 0.78 c |
| 15 C | 35.93 ± 0.02 o | 6.03 ± 0.02 hi | 23.78 ± 0.02 m | 36.44 ± 0.22 l | 14.23 ± 0.05 cde |
| 16 C | 30.31 ± 0.4 jk | 7.11 ± 0.13 j | 18.10 ± 0.63 j | 31.13 ± 0.42 h | 21.47 ± 0.65 i |
| 17 A | 26.8 ± 0.03 g | 5.21 ± 0.05 f | 11.49 ± 0.07 d | 27.34 ± 0.03 f | 24.4 ± 0.17 j |
| 18 R | 25.43 ± 0.34 f | 3.91 ± 0.54 cd | 8.01 ± 0.59 a | 25.73 ± 0.27 e | 25.92 ± 1.42 jk |
| 19 R | 21.51 ± 0.4 c | 3.41 ± 0.48 c | 14.39 ± 0.31 fg | 21.78 ± 0.4 b | 13.32 ± 1.86 c |
| 20 P | 23.19 ± 0.41 d | 3.76 ± 0.14 c | 13.50 ± 0.32 ef | 23.49 ± 0.4 c | 15.59 ± 0.83 def |
| 21 P | 34.51 ± 0.41 n | 3.79 ± 0.15 c | 15.74 ± 0.3 h | 34.72 ± 0.41 k | 13.56 ± 0.48 c |
| 22 R | 31.54 ± 0.31 l | 11.32 ± 0.4 m | 31.24 ± 0.64 p | 33.52 ± 0.33 ij | 19.93 ± 0.88 hi |
| 23 C | 24.31 ± 0.39 e | 9.35 ± 0.34 l | 16.94 ± 0.67 i | 26.05 ± 0.31 e | 28.93 ± 1.43 l |
| 24 P | 28.64 ± 0.29 h | 6.53 ± 0.40 i | 19.53 ± 0.5 k | 29.37 ± 0.28 g | 18.5 ± 1.36 gh |
| Overall mean | 28.52 ± 5.2 | 5.39 ± 2.16 | 18.01 ± 6.63 | 29.1 ± 5.24 | 17.42 ± 6.11 |
| Min–Max | 19.33–39.44 | 1.74–11.99 | 7.5–32.05 | 20.02–29.73 | 7.72–31.2 |
| Sample | Equation | Vmax (O.D.540/min) | Km (mM) | Inhibition Type |
|---|---|---|---|---|
| Control | y = 6.4842x + 17.202 | 0.058 | 0.37 | No inhibition |
| Honey 15 C | y = 33.699x + 32.943 | 0.030 | 1.02 | Mixed non-competitive inhibition |
| Honey 16 C | y = 32.112x + 25.043 | 0.039 | 1.28 | Mixed non-competitive inhibition |
| Honey 23 C | y = 39.161x + 23.48 | 0.042 | 1.66 | Mixed non-competitive inhibition |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zaldivar-Ortega, A.K.; Morfin, N.; Angeles-Hernandez, J.C.; González-Montiel, L.; Vicente-Flores, M.; Aguirre-Álvarez, G.; Cenobio-Galindo, A.d.J. Functional Characterization of Scaptotrigona mexicana Honey: Physicochemical Properties, Antioxidant Capacity, and α-Amylase Inhibition for Food Process Applications. Processes 2025, 13, 2788. https://doi.org/10.3390/pr13092788
Zaldivar-Ortega AK, Morfin N, Angeles-Hernandez JC, González-Montiel L, Vicente-Flores M, Aguirre-Álvarez G, Cenobio-Galindo AdJ. Functional Characterization of Scaptotrigona mexicana Honey: Physicochemical Properties, Antioxidant Capacity, and α-Amylase Inhibition for Food Process Applications. Processes. 2025; 13(9):2788. https://doi.org/10.3390/pr13092788
Chicago/Turabian StyleZaldivar-Ortega, Ana Karen, Nuria Morfin, Juan Carlos Angeles-Hernandez, Lucio González-Montiel, Macario Vicente-Flores, Gabriel Aguirre-Álvarez, and Antonio de Jesús Cenobio-Galindo. 2025. "Functional Characterization of Scaptotrigona mexicana Honey: Physicochemical Properties, Antioxidant Capacity, and α-Amylase Inhibition for Food Process Applications" Processes 13, no. 9: 2788. https://doi.org/10.3390/pr13092788
APA StyleZaldivar-Ortega, A. K., Morfin, N., Angeles-Hernandez, J. C., González-Montiel, L., Vicente-Flores, M., Aguirre-Álvarez, G., & Cenobio-Galindo, A. d. J. (2025). Functional Characterization of Scaptotrigona mexicana Honey: Physicochemical Properties, Antioxidant Capacity, and α-Amylase Inhibition for Food Process Applications. Processes, 13(9), 2788. https://doi.org/10.3390/pr13092788









