Kinetics and Thermodynamics of Ultrasound-Assisted Extraction of Taxanes from Taxus chinensis by Natural Deep Eutectic Solvents
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Extraction Procedures
2.3. HPLC Analysis of Taxanes
2.4. Establish the Standard Curve of Paclitaxel
2.5. Kinetic Model
2.6. Activation Energy and Thermodynamic Parameters
3. Results and Discussion
3.1. HPLC Analysis
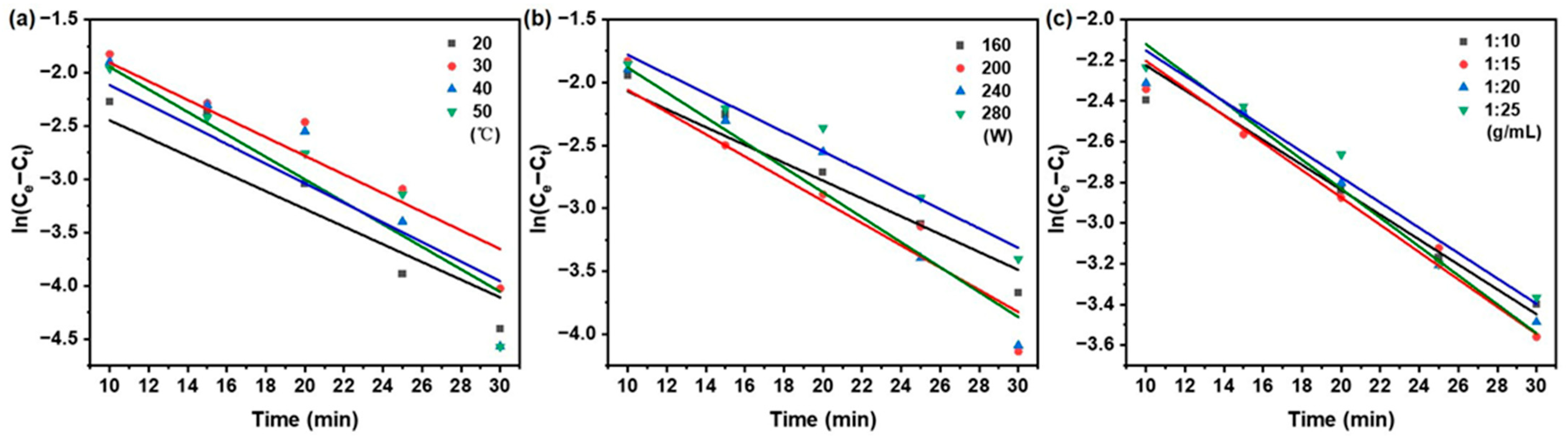
3.2. Analysis of Extraction Kinetics
3.3. Determination of Rate-Limiting Step
3.4. Determining the Effective Diffusion and Mass Transfer Coefficient
3.5. Analysis of Extraction Thermodynamics
3.6. Comparison of the Fitting Effects of the Peleg Model and the Second-Order Kinetic Model in the Extraction of Paclitaxel
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Christensen, S.B. Drugs that changed society: Microtubule-targeting agents belonging to taxanoids, macrolides and non-ribosomal peptides. Molecules 2022, 27, 5648. [Google Scholar] [CrossRef]
- Yang, Y.H.; Mao, J.W.; Tan, X.L. Research progress on the source, production, and anti-cancer mechanisms of paclitaxel. Chin. J. Nat. Med. 2020, 18, 890–897. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.; Hamid, I.A.A.; Kee, C.M.; Quan, J.X. RSM-rCCD Optimizing for Paclitaxel Extraction from Taxus chinensis byNatural Deep Eutectic Solvents and Studying Antioxidant Activity. Curr. Anal. Chem. 2025, 21, e15734110367450. [Google Scholar] [CrossRef]
- Šavikin, K.; Janković, T.; Ćujić, N.; Menković, N. Optimization of ultrasound-assisted extraction of polyphenolic compounds from pomegranate peel using response surface methodology. Sep. Purif. Technol. 2018, 194, 40–47. [Google Scholar] [CrossRef]
- Park, J.M.; Kim, J.H. Ultrasound-assisted micellar extraction for paclitaxel purification from Taxus chinensis. Korean Chem. Eng. Res. 2021, 59, 106–111. [Google Scholar] [CrossRef]
- Zhao, Z.R.; Zhang, Y.J.; Li, W.L.; Tang, Y.H.; Meng, H.W.; Wang, S.J. Improving the extraction yield of taxanes from Taxus cuspidata needles using cold plasma. J. Appl. Res. Med. Aromat. Plants 2023, 34, 100457. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Z.; Meng, H.; Li, W.; Wang, S. Ultrasonic extraction and separation of taxanes from Taxus cuspidata optimized by response surface methodology. Separations 2022, 9, 193. [Google Scholar] [CrossRef]
- Li, L.; Chen, Y.; Ma, Y.; Wang, Z.; Wang, T.; Xie, Y. Optimization of taxol extraction process using response surface methodology and investigation of temporal and spatial distribution of Taxol in Taxus mairei. Molecules 2021, 26, 5485. [Google Scholar] [CrossRef]
- Milićević, N.; Kojić, P.; Sakač, M.; Mišan, A.; Kojić, J.; Perussello, C.; Tiwari, B. Kinetic modeling of ultrasound-assisted extraction of phenolics from cereal brans. Ultrason. Sonochem. 2021, 79, 105761. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Shen, H.T.; Shen, C.; Li, Y.N.; Ying, Z.F.; Duan, Y.F. Kinetics and Mechanism Study of Mercury Adsorption by Activated Carbon in Wet Oxy-Fuel Conditions. Energy Fuels 2019, 33, 1344–1353. [Google Scholar] [CrossRef]
- Nie, J.G.; Chen, D.T.; Ye, J. Optimization and kinetic modeling of ultrasonic-assisted extraction of fucoxanthin from edible brown algae Sargassum fusiforme using green solvents. Ultrason. Sonochem. 2021, 77, 105671. [Google Scholar] [CrossRef]
- Zhou, P.; Li, X.Z.; Zhou, J.; Yang, Y.H.; Zhi, J.; Shen, L.Q. Mass transfer mechanism of the multivariate consecutive extraction process of pectin and hesperidin from Citrus aurantium L.: Kinetics, thermodynamics, diffusion and mass transfer coefficients. Sep. Purif. Technol. 2023, 311, 123339. [Google Scholar] [CrossRef]
- Alrugaibah, M.; Yagiz, Y.; Gu, L.W. Use natural deep eutectic solvents as efficient green reagents to extract procyanidins and anthocyanins from cranberry pomace and predictive modeling by RSM and artificial neural networking. Sep. Purif. Technol. 2021, 255, 117720. [Google Scholar] [CrossRef]
- Loganathan, D.; Yi, R.; Patel, B.; Zhang, J.; Kong, N. A sensitive HPLC-MS/MS method for the detection, resolution and quantitation of cathinone enantiomers in horse blood plasma and urine. Anal. Bioanal. Chem. 2021, 413, 1618–1632. [Google Scholar] [CrossRef]
- Theodoridis, G.; Haasnoot, W.; Cazemier, G.; Schilt, R.; Jaziri, M.; Diallo, B.; Papadoyannis, I.N.; de Jong, G.J. Immunoaffinity chromatography for the sample pretreatment of Taxus plant and cell extracts prior to analysis of taxanes by high-performance liquid chromatography. J. Chromatogr. A 2002, 948, 177–185. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhao, Z.; Li, W.; Tang, Y.; Meng, H.; Wang, S. Purification of Two Taxanes from Taxus cuspidata by Preparative High-Performance Liquid Chromatography. Separations 2022, 9, 446. [Google Scholar] [CrossRef]
- Quan, J.X.; Nie, G.R.; Xue, H.; Luo, L.; Zhang, R.P.; Li, H.B. Macroscopic Chiral Recognition by Calix [4] arene-Based Host-Guest Interactions. Chem. A Eur. J. 2018, 24, 15502–15506. [Google Scholar] [CrossRef]
- Quan, J.X.; Yan, H.W.; Periyasami, G.; Li, H.B. A Visible-Light Regulated ATP Transport in Retinal-modified Pillar[6] arene Layer-by-Layer Self-assembled Sub-nanochannel. Chem.-A Eur. J. 2024, 30, e202401045. [Google Scholar] [CrossRef]
- Li, W.; Li, G.; Xu, W.; Li, Z.; Qu, H.; Ma, C.; Zhang, H.; Cai, M.; Noruzi, E.B.; Quan, J. Visible Light-Gating Responsive Nanochannel for Controlled Release of the Fungicide. Small 2024, 20, 2401503–2401508. [Google Scholar] [CrossRef] [PubMed]
- Zahari, N.A.; Chong, G.H.; Abdullah, L.C.; Chua, B.L. Ultrasonic-assisted extraction (UAE) process on thymol concentration from Plectranthus amboinicus leaves: Kinetic modeling and optimization. Processes 2020, 8, 322. [Google Scholar] [CrossRef]
- Thakur, R.; Gupta, V.; Dhar, P.; Deka, S.C.; Das, A.B. Ultrasound-assisted extraction of anthocyanin from black rice bran using natural deep eutectic solvents: Optimization, diffusivity, and stability. J. Food Process. Preserv. 2022, 46, e16309. [Google Scholar] [CrossRef]
- Giri, S.; Dash, K.K.; Raj, G.B.; Kovács, B.; Mukarram, S.A. Ultrasound assisted phytochemical extraction of persimmon fruit peel: Integrating ANN modeling and genetic algorithm optimization. Ultrason. Sonochem. 2024, 102, 106759. [Google Scholar] [CrossRef] [PubMed]
- Min, H.S.; Kim, J.H. Study of the extraction kinetics and calculation of effective diffusivity and mass transfer coefficient in negative pressure cavitation extraction of paclitaxel from Taxus chinensis. Biotechnol. Bioprocess Eng. 2022, 27, 111–118. [Google Scholar] [CrossRef]
- Yang, Y.C.; Wang, C.S.; Wei, M.C. A green approach for the extraction and characterization of oridonin and ursolic and oleanolic acids from Rabdosia rubescens and its kinetic behavior. Food Chem. 2020, 319, 126582. [Google Scholar] [CrossRef]
- Krishnan, R.Y.; Rajan, K.S. Microwave assisted extraction of flavonoids from Terminalia bellerica: Study of kinetics and thermodynamics. Sep. Purif. Technol. 2016, 157, 169–178. [Google Scholar] [CrossRef]
- Liao, J.; Guo, Z.; Yu, G. Process intensification and kinetic studies of ultrasound-assisted extraction of flavonoids from peanut shells. Ultrason. Sonochem. 2021, 76, 105661. [Google Scholar] [CrossRef] [PubMed]
- Leveneur, S. Kinetic modelling: Regression and validation stages, a compulsory tandem for kinetic model assessment. Can J Chem Eng. 2023, 101, 7078–7101. [Google Scholar] [CrossRef]
- Xiao, P.; Yu, Z.X.; Chen, T.X.; Yu, L.; He, Y.; Jin, W.F. Systematic evaluation and platform construction of kinetics modeling for herbal extraction in phytomedicine. Ultrason. Sonochem. 2025, 123, 107646. [Google Scholar] [CrossRef] [PubMed]
- Raj, G.B.; Dash, K.K. Ultrasound-assisted extraction of phytocompounds from dragon fruit peel: Optimization, kinetics and thermodynamic studies. Ultrason. Sonochem. 2020, 68, 105180. [Google Scholar] [CrossRef] [PubMed]
- Lima, E.C.; Gomes, A.A.; Tran, H.N. Comparison of the nonlinear and linear forms of the van’t Hoff equation for calculation of adsorption thermodynamic parameters (∆S and ∆H). J. Mol. Liq. 2020, 311, 113315. [Google Scholar] [CrossRef]
- Bullerjahn, J.T.; Hanson, S.M. Extracting thermodynamic properties from van’t Hoff plots with emphasis on temperature-sensing ion channels. Temperature 2024, 11, 60–71. [Google Scholar] [CrossRef]
- Krishnan, R.Y.; Chandran, M.N.; Vadivel, V.; Rajan, K.S. Insights on the influence of microwave irradiation on the extraction of flavonoids from Terminalia chebula. Sep. Purif. Technol. 2016, 170, 224–233. [Google Scholar] [CrossRef]
- Sahin, S.; Samlı, R. Optimization of olive leaf extract obtained by ultrasound-assisted extraction with response surface methodology. Ultrason. Sonochem. 2013, 20, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Z.; Zhang, Y.; Li, W.; Tang, Y.; Wang, S. Parameter Optimization of Ultrasonic–Microwave Synergistic Extraction of Taxanes from Taxus cuspidata Needles. Molecules 2023, 28, 7746. [Google Scholar] [CrossRef]
- Liu, H.; Zhong, D.; Zhang, R.; Wang, G.; Zhang, Y. Extraction and purification of polyphenols and determination of antioxidant activity. Trans CSAE 2020, 36, 324–332. [Google Scholar] [CrossRef]
- Sun, Q.; Du, B.; Wang, C.; Xu, W.; Zhang, H. Ultrasound-Assisted Ionic Liquid Solid-Liquid Extraction Coupled with Aqueous Two-Phase Extraction of Naphthoquinone Pigments in Arnebia euchroma (Royle) Johnst. Chromatographia 2019, 82, 1777–1789. [Google Scholar] [CrossRef]
- Cai, Z.; Han, M.; Zhang, X.; Gao, X.; Wang, F.; Pang, M. Extraction of Anthraquinone Compounds from Chinese Chestnut by Using Ultrasonic-assisted Technology. J. Plant Sci. 2019, 7, 43–47. [Google Scholar] [CrossRef]
- Wang, Y.G.; Wang, C.L. Comparative analysis of three kinds of extraction kinetic models of crude polysaccharides from Codonopsis pilosula and evaluate the characteristics of crude polysaccharides. Biomass Convers. Biorefinery 2023, 13, 12917–12933. [Google Scholar] [CrossRef]
- Cheung, Y.C.; Wu, J.Y. Kinetic models and process parameters for ultrasound-assisted extraction of water-soluble components and polysaccharides from a medicinal fungus. Biochem. Eng. J. 2013, 79, 214–220. [Google Scholar] [CrossRef]
- McKay, G.; Ho, Y.S.; Ng, J.C.Y. Biosorption of copper from wastewater: A review. Sep. Purif. Methods 1999, 28, 87–125. [Google Scholar] [CrossRef]
- Singh, N.; Kumar, S.; Patle, D.S. Intensification of extraction of bioactive compounds from pomegranate peel using an ultrasound-microwave assisted extraction approach: Parametric optimization, kinetics and thermodynamics. Sep. Purif. Technol. 2025, 359, 130681. [Google Scholar] [CrossRef]
- Zou, Y.; Yang, H. Effect of ultrasound power on extraction kinetic model, and physicochemical and structural characteristics of collagen from chicken lung. Food Prod. Process. Nutr. 2020, 2, 3. [Google Scholar] [CrossRef]
- Alara, O.R.; Abdurahman, N.H. Microwave-assisted extraction of phenolics from Hibiscus sabdariffa calyces:Kinetic modelling and process intensification. Ind. Crops Prod. 2019, 137, 528–535. [Google Scholar] [CrossRef]
- Weber, J.; Morris, J.C. Kinetics of adsorption on carbon from solution. J. Sanit. Eng. Div. 1963, 89, 31–59. [Google Scholar] [CrossRef]
- Amadi, O.K.; Odih, C.; Nwachukwu, O.U. Kinetics and mechanism modelling of pb (ii) and ni (ii) ions sorption from aqueous medium: Evaluating the performance of curcuma longa rhizome in the treatment of waste water. J. Chem. Soc. Niger. 2024, 49, 1. [Google Scholar] [CrossRef]
- Lach, J.; Okoniewska, E. Equilibrium, kinetic, and diffusion mechanism of lead (II) and cadmium (II) adsorption onto commercial activated carbons. Molecules 2024, 29, 2418. [Google Scholar] [CrossRef] [PubMed]
- Kavitha, D.; Namasivayam, C. Experimental and kinetic studies on methylene blue adsorption by coir pith carbon. Bioresour. Technol. 2007, 98, 14–21. [Google Scholar] [CrossRef]
- Asheghmoalla, M.; Mehrvar, M. Adsorption of a Multicomponent Pharmaceutical Wastewater on Charcoal-Based Activated Carbon: Equilibrium and Kinetics. Water 2024, 16, 2086. [Google Scholar] [CrossRef]
- Dulo, B.; De Somer, T.; Moyo, M.; Nakyese, E.; Githaiga, J.; Raes, K.; De Meester, S. Kinetic modeling of phenolic compounds extraction from nutshells: Influence of particle size, temperature and solvent ratio. Biomass Convers. Biorefinery 2024, 14, 23565–23579. [Google Scholar] [CrossRef]
- Yoo, K.W.; Kim, J.H. Kinetics and mechanism of ultrasound-assisted extraction of paclitaxel from Taxus chinensis. Biotechnol. Bioprocess Eng. 2018, 23, 532–540. [Google Scholar] [CrossRef]
- Hou, F.; Song, S.; Yang, S.; Wang, Y.; Jia, F.; Wang, W. Study on the Optimization, Extraction Kinetics and Thermodynamics of the Ultrasound-Assisted Enzymatic Extraction of Tremella fuciformis Polysaccharides. Foods 2024, 13, 1408. [Google Scholar] [CrossRef]
- Huang, C.; Wang, Y.; Zhou, C.; Sun, Q.; Zhang, L.; Niu, Y.; Ma, H. Ultrasonic vacuum synergistic assisted ethanol extraction of steviol glycosides and kinetic studies. Ind. Crops Prod. 2024, 221, 119385. [Google Scholar] [CrossRef]
- Hobbi, P.; Okoro, O.V.; Delporte, C.; Alimoradi, H.; Podstawczyk, D.; Nie, L.; Shavandi, A. Kinetic modelling of the solid–liquid extraction process of polyphenolic compounds from apple pomace: Influence of solvent composition and temperature. Bioresour. Bioprocess. 2021, 8, 114. [Google Scholar] [CrossRef]
- Thaisamak, P.; Jaturonglumlert, S.; Varith, J.; Taip, F.S.; Nitatwichit, C. Kinetic model of ultrasonic-assisted extraction with controlled temperature of c-phycocyaninfrom s. platensis. Int. J. GEOMATE 2019, 16, 176–183. [Google Scholar] [CrossRef]
- Wang, Y.G.; Xinjian Wang, X.J.; Kang Zhang, K.; Xuan Zhang, X.; Li, S.W.; Li, Y.L.; Fan, W.G.; Leng, F.F.; Yang, M.J.; Chen, J.X. Extraction kinetics, thermodynamics, rheological properties and anti-BVDV activity of the hot water assisted extraction of Glycyrrhiza polysaccharide. Food Funct. 2020, 11, 4067–4080. [Google Scholar] [CrossRef] [PubMed]
- Ji, J.B.; Lu, X.H.; Cai, M.Q.; Xu, Z.C. Improvement of leaching process of Geniposide with ultrasound. Ultrason. Sonochem. 2006, 13, 455–462. [Google Scholar] [CrossRef]
- Charpe, T.W.; Rathod, V.K. Kinetics of ultrasound assisted extraction of wedelolactone from Eclipta alba. Braz. J. Chem. Eng. 2016, 33, 1003–1010. [Google Scholar] [CrossRef]
- Patil, S.S.; Pathak, A.; Rathod, V.K. Optimization and kinetic study of ultrasound assisted deep eutectic solvent based extraction: A greener route for extraction of curcuminoids from Curcuma longa. Ultrason. Sonochem. 2021, 70, 105267. [Google Scholar] [CrossRef]
- Lasunon, P.; Sengkhamparn, N. Effect of Ultrasound-Assisted, Microwave-Assisted and Ultrasound-Microwave-Assisted Extraction on Pectin Extraction from Industrial Tomato Waste. Molecules 2022, 27, 1157. [Google Scholar] [CrossRef]
- Merouani, S.; Dehane, A.; Hamdaoui, O. A review on the integration of ultrasonication in hybrid systems for enhanced hydrogen yield. Ultrason. Sonochem. 2025, 121, 107552. [Google Scholar] [CrossRef]
- Singla, M.; Singh, A.; Sit, N. Effect of microwave and enzymatic pretreatment and type of solvent on kinetics of ultrasound assisted extraction of bioactive compounds from ripe papaya peel. J. Food Process Eng. 2023, 46, e14119. [Google Scholar] [CrossRef]
- Albarri, R.; Şahin, S. Kinetics, thermodynamics, and mass transfer mechanism of the ultrasound-assisted extraction of bioactive molecules from Moringa oleifera leaves. Biomass Convers. Biorefinery 2023, 13, 7919–7926. [Google Scholar] [CrossRef]
- Jo, Y.J.; Kim, J.H. Effective diffusivity and mass transfer coefficient during the extraction of paclitaxel from Taxus chinensis using methanol. Biotechnol. Bioprocess Eng. 2019, 24, 818–823. [Google Scholar] [CrossRef]
- Reche, C.; Rosselló, C.; Umaña, M.M.; Eim, V.; Simal, S. Mathematical modelling of ultrasound-assisted extraction kinetics of bioactive compounds from artichoke by-products. Foods 2021, 10, 931. [Google Scholar] [CrossRef]
- González-Centeno, M.R.; Comas-Serra, F.; Femenia, A.; Rosselló, C.; Simal, S. Effect of power ultrasound application on aqueous extraction of phenolic compounds and antioxidant capacity from grape pomace (Vitis vinifera L.): Experimental kinetics and modeling. Ultrason. Sonochem. 2015, 22, 506–514. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Kim, J.H. Kinetic and thermodynamic characteristics of microwave-assisted extraction for the recovery of paclitaxel from Taxus chinensis. Process Biochem. 2019, 76, 187–193. [Google Scholar] [CrossRef]
- Wei, M.; Zhao, R.; Peng, X.; Feng, C.; Gu, H.; Yang, L. Ultrasound-assisted extraction of taxifolin, diosmin, and quercetin from abies nephrolepis (trautv.) maxim: Kinetic and thermodynamic characteristics. Molecules 2020, 25, 1401. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.; Patle, D.S.; Sushil Kumar, S. Microwave- and Ultrasonication-Based Intensified and Synergetic Approaches for Extraction of Bioactive Compounds from Pomegranate Peels: Parametric and Kinetic Studies. Ind. Eng. Chem. Res. 2024, 63, 9214–9224. [Google Scholar] [CrossRef]
- Guo, X.; Liu, S.; Wang, Z.; Zhang, G. Ultrasonic-assisted extraction of polysaccharide from Dendrobium officinale: Kinetics, thermodynamics and optimization. Biochem. Eng. J. 2022, 177, 108227. [Google Scholar] [CrossRef]
- Arimalala, A.F.; Hervé, R.P.; Rafihavanana, R. Modeling and kinetics study of Avocado oil extraction from Madagascar using different mathematical models. S. Afr. J. Chem. Eng. 2022, 41, 93–97. [Google Scholar] [CrossRef]
- Satao, N.T.; Mahindrakar, K.V.; Rathod, V.K. Extraction of phenolics from Spilanthes Acmella plant: Kinetic studies using Peleg’s model. J. Indian Chem. Soc. 2025, 102, 101634. [Google Scholar] [CrossRef]







| Model | Equation | Parameter | Refs. |
|---|---|---|---|
| Pseudo-first-order model | Ct (mg/mL): the instantaneous taxane concentration in the suspension Ce (mg/mL): concentration of extracted taxanes at equilibrium k1 (1/min): first-order rate constant | Equation (2) | |
| Pseudo-second-order model | k2 (mL/mg·min): second-order rate constant h (mg/mL·min): initial extraction rate | Equation (3) | |
| Intraparticle diffusion model | qt: yield of taxanes extracted in the suspension at any time kP (mg/g·min1/2): intraparticle diffusivity | Equation (4) | |
| Fick’s first law | Ct (mg/mL): concentration of taxanes in the suspension at any time Ce (mg/mL): concentration of extracted taxanes at equilibrium A (m2): total surface area of the particles mplant (kg): cell weight introduced in extractor ρ: cell wet density (1150 kg/m3) rp: cell size (average radius: 8.9 × 10−5 m) Vs (m3): volume of solution KT (m/s): mass transfer coefficient | Equation (5) | |
| Fick’s second law | Ct (mg/mL): concentration of taxanes in the suspension at any time Ce (mg/mL): concentration of extracted taxanes at equilibrium De (m2/s): effective diffusion coefficient R: cell radius (average radius: 8.9 × 10−5 m) | Equation (6) | |
| Bi: Biot number | Equation (7) | ||
| Root Mean Square Deviation | n: the number of experimental runs | Equation (8) | |
| Arrhenius equation | k2 (mL/mg min): second-order extraction rate constant Ea (kJ/mol): activation energy k0: pre-exponential factor T (K): temperature R (8.314 J/mol·K): universal gas constant | Equation (9) | |
| Van’t Hoff equation | ys (mg/mL): amount of taxanes after extraction at an optimum time (t) ymax (mg/mL): the maximum extractable taxane content in the solvent mixture after exhaustive extraction Ke: the taxane yield to residual mass ratio | Equation (10) | |
| Peleg’s model | Ct (mg/mL): concentration of taxanes in the suspension at any time K1: Peleg’s the rate constant (min g/mg) K2: Peleg’s capacity constant (g/mg) | Equation (11) | |
| Residual Sum of Squares | Ct (mg/mL): the actual extraction value of paclitaxel at any time Ci (mg/mL): the predicted extraction value of paclitaxel at any time | Equation (12) | |
| Akaike Information Criterion | φ1: the number of experimental data points φ2: the number of model parameters | Equation (13) |
| Order | Compounds | Regression Equation | R2 | Linear Range (μg/mL) | Formula | M.W. (g/mol) |
|---|---|---|---|---|---|---|
| 1 | 10-DAB III | y = 18,530x – 21,993 | R2 = 0.9998 | 1.25–125 | C29H36O10 | 544.590 |
| 2 | Baccatin III | y = 11,274x + 101,571 | R2 = 0.9992 | 1.25–125 | C31H38O11 | 586.627 |
| 3 | 10-DAT | y = 7285x + 2697 | R2 = 0.9998 | 1.25–125 | C45H49NO13 | 811.870 |
| 4 | DXT | y = 16,326x + 27,972 | R2 = 0.9998 | 1.25–125 | C50H57NO17 | 943.984 |
| 5 | Cephalomannine | y = 15,990x + 4008 | R2 = 0.9996 | 1.25–125 | C45H53NO14 | 831.901 |
| 6 | Paclitaxel | y = 27,486x + 17,276 | R2 = 0.9998 | 1.25–125 | C47H51NO14 | 853.906 |
| The Pseudo-First-Order Model | The Pseudo-Second-Order Model | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| k1 (1/min) | R2 * | RMSD * | RSS | AIC | K2 (mg/g·min) | Ce (mg/mL) | H (mg/mL·min) | R2 * | RMSD * | RSS | AIC | |
| Temperature (°C) | ||||||||||||
| 20 | 0.083 | 0.871 | 0.018 | 0.00221 | −63.553 | 0.163 | 0.296 | 0.014 | 0.959 | 0.010 | 0.00085 | −67.227 |
| 30 | 0.088 | 0.908 | 0.013 | 0.00164 | −65.919 | 0.171 | 0.462 | 0.037 | 0.996 | 0.008 | 0.00049 | −71.659 |
| 40 | 0.106 | 0.886 | 0.015 | 0.00175 | −65.399 | 0.232 | 0.496 | 0.057 | 0.997 | 0.008 | 0.00056 | −70.500 |
| 50 | 0.092 | 0.841 | 0.015 | 0.00121 | −68.360 | 0.263 | 0.366 | 0.035 | 0.989 | 0.012 | 0.00108 | −65.251 |
| Power (W) | ||||||||||||
| 160 | 0.071 | 0.955 | 0.011 | 0.00144 | −66.987 | 0.129 | 0.375 | 0.018 | 0.986 | 0.007 | 0.00043 | −72.627 |
| 200 | 0.088 | 0.927 | 0.016 | 0.00247 | −62.677 | 0.167 | 0.417 | 0.029 | 0.983 | 0.012 | 0.00110 | −65.102 |
| 240 | 0.099 | 0.972 | 0.010 | 0.00118 | −68.542 | 0.230 | 0.496 | 0.057 | 0.998 | 0.007 | 0.00039 | −73.373 |
| 280 | 0.077 | 0.966 | 0.012 | 0.00184 | −65.029 | 0.182 | 0.463 | 0.039 | 0.997 | 0.006 | 0.00033 | −74.803 |
| Solid/liquid ratio (g/mL) | ||||||||||||
| 1:10 | 0.061 | 0.952 | 0.008 | 0.00089 | −70.790 | 0.207 | 0.271 | 0.015 | 0.983 | 0.007 | 0.00040 | −73.313 |
| 1:15 | 0.067 | 0.969 | 0.007 | 0.00086 | −71.149 | 0.222 | 0.289 | 0.019 | 0.994 | 0.005 | 0.00018 | −79.517 |
| 1:20 | 0.071 | 0.958 | 0.009 | 0.00093 | −70.500 | 0.244 | 0.312 | 0.024 | 0.994 | 0.006 | 0.00029 | −75.798 |
| 1:25 | 0.062 | 0.964 | 0.006 | 0.00057 | −74.369 | 0.216 | 0.282 | 0.017 | 0.981 | 0.008 | 0.00049 | −71.675 |
| Ultrasonic Power (W) | kp1 (mg/g·min1/2) | kp2 (mg/g·min1/2) |
|---|---|---|
| 160 | 0.054 | 0.033 |
| 200 | 0.108 | 0.042 |
| 240 | 0.063 | 0.053 |
| 280 | 0.087 | 0.046 |
| Conditions | Parameters | De (m2/s) × 10−9 | KT (m/s) × 10−3 | Bi |
|---|---|---|---|---|
| Temperature (°C) | 20 | 4.50 | 3.01 | 59.45 |
| 30 | 4.79 | 3.43 | 63.70 | |
| 40 | 6.21 | 4.14 | 59.37 | |
| 50 | 5.74 | 3.82 | 59.21 | |
| Power (W) | 160 | 3.85 | 2.77 | 63.94 |
| 200 | 4.31 | 3.29 | 67.89 | |
| 240 | 4.68 | 3.89 | 73.99 | |
| 280 | 3.55 | 3.09 | 77.30 | |
| Solid/Liquid Ratio (g/mL) | 1:10 | 2.59 | 2.48 | 85.08 |
| 1:15 | 2.91 | 2.71 | 83.02 | |
| 1:20 | 3.05 | 2.84 | 83.05 | |
| 1:25 | 2.90 | 2.50 | 76.76 |
| Temperature (°C) | ||||
|---|---|---|---|---|
| 20 | 30 | 40 | 50 | |
| Ke | 1.80 | 3.29 | 4.86 | 4.77 |
| ΔG (KJ/mol) | −1.44 | −2.90 | −3.86 | −3.81 |
| Ea (KJ/mol) | 18.84 | |||
| ΔH (KJ/mol) | 26.33 | |||
| ΔS (KJ/mol·K) | 95.79 | |||
| The Peleg Model | The Pseudo-Second-Order Model | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| K1 (min·g/mg) | K2 (g/mg) | R2 | RMSD | RSS | AIC | k2 (mg/g·min) | Ce (mg/mL) | h (mg/mL·min) | R2 | RMSD | RSS | AIC | |
| Temperature (°C) | |||||||||||||
| 20 | 69.010 | 3.367 | 0.933 | 0.010 | 0.00083 | −69.359 | 0.163 | 0.296 | 0.014 | 0.959 | 0.010 | 0.00085 | −67.227 |
| 30 | 30.974 | 2.626 | 0.940 | 0.052 | 0.02131 | −43.423 | 0.171 | 0.462 | 0.037 | 0.996 | 0.008 | 0.00049 | −71.659 |
| 40 | 18.226 | 1.988 | 0.976 | 0.008 | 0.00053 | −72.959 | 0.232 | 0.496 | 0.057 | 0.997 | 0.008 | 0.00056 | −70.500 |
| 50 | 27.973 | 2.136 | 0.981 | 0.052 | 0.02182 | −43.236 | 0.263 | 0.366 | 0.035 | 0.989 | 0.012 | 0.00108 | −65.251 |
| Power (W) | |||||||||||||
| 160 | 55.249 | 2.643 | 0.976 | 0.007 | 0.00042 | −74.827 | 0.129 | 0.375 | 0.018 | 0.986 | 0.007 | 0.00043 | −72.627 |
| 200 | 34.735 | 2.365 | 0.952 | 0.012 | 0.00108 | −67.270 | 0.167 | 0.417 | 0.029 | 0.983 | 0.012 | 0.00110 | −65.102 |
| 240 | 18.256 | 1.993 | 0.983 | 0.007 | 0.00037 | −75.784 | 0.230 | 0.496 | 0.057 | 0.998 | 0.007 | 0.00039 | −73.373 |
| 280 | 26.144 | 2.143 | 0.986 | 0.006 | 0.00032 | −77.001 | 0.182 | 0.463 | 0.039 | 0.997 | 0.006 | 0.00033 | −74.803 |
| Solid/liquid ratio (g/mL) | |||||||||||||
| 1:10 | 66.361 | 3.662 | 0.956 | 0.007 | 0.00039 | −75.335 | 0.207 | 0.271 | 0.015 | 0.983 | 0.007 | 0.00040 | −73.313 |
| 1:15 | 54.124 | 3.453 | 0.981 | 0.005 | 0.00018 | −81.522 | 0.222 | 0.289 | 0.019 | 0.994 | 0.005 | 0.00018 | −79.517 |
| 1:20 | 42.195 | 3.207 | 0.972 | 0.006 | 0.00029 | −77.798 | 0.244 | 0.312 | 0.024 | 0.994 | 0.006 | 0.00029 | −75.798 |
| 1:25 | 60.032 | 3.468 | 0.955 | 0.008 | 0.00047 | −73.878 | 0.216 | 0.282 | 0.017 | 0.981 | 0.008 | 0.00049 | −71.675 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, Y.; Song, W.; Hu, L.; Liu, R.; Hamid, I.A.A.; Quan, J. Kinetics and Thermodynamics of Ultrasound-Assisted Extraction of Taxanes from Taxus chinensis by Natural Deep Eutectic Solvents. Processes 2025, 13, 4074. https://doi.org/10.3390/pr13124074
Guo Y, Song W, Hu L, Liu R, Hamid IAA, Quan J. Kinetics and Thermodynamics of Ultrasound-Assisted Extraction of Taxanes from Taxus chinensis by Natural Deep Eutectic Solvents. Processes. 2025; 13(12):4074. https://doi.org/10.3390/pr13124074
Chicago/Turabian StyleGuo, Ying, Wenna Song, Lingyu Hu, Runbo Liu, Izni Atikah Abd Hamid, and Jiaxin Quan. 2025. "Kinetics and Thermodynamics of Ultrasound-Assisted Extraction of Taxanes from Taxus chinensis by Natural Deep Eutectic Solvents" Processes 13, no. 12: 4074. https://doi.org/10.3390/pr13124074
APA StyleGuo, Y., Song, W., Hu, L., Liu, R., Hamid, I. A. A., & Quan, J. (2025). Kinetics and Thermodynamics of Ultrasound-Assisted Extraction of Taxanes from Taxus chinensis by Natural Deep Eutectic Solvents. Processes, 13(12), 4074. https://doi.org/10.3390/pr13124074






