Degassing N2 from the Direct Oxidation of Total Ammonia in Mariculture Using a Three-Dimensional Electrode System
Abstract
1. Introduction
2. Materials and Methods
2.1. Methods for Measuring Related Substances and Sources of Chemical Reagents
2.2. Synthesis of Granular Electrodes
2.3. Electrochemical Testing
2.4. Formulas for Relevant Parameters
3. Results and Discussion
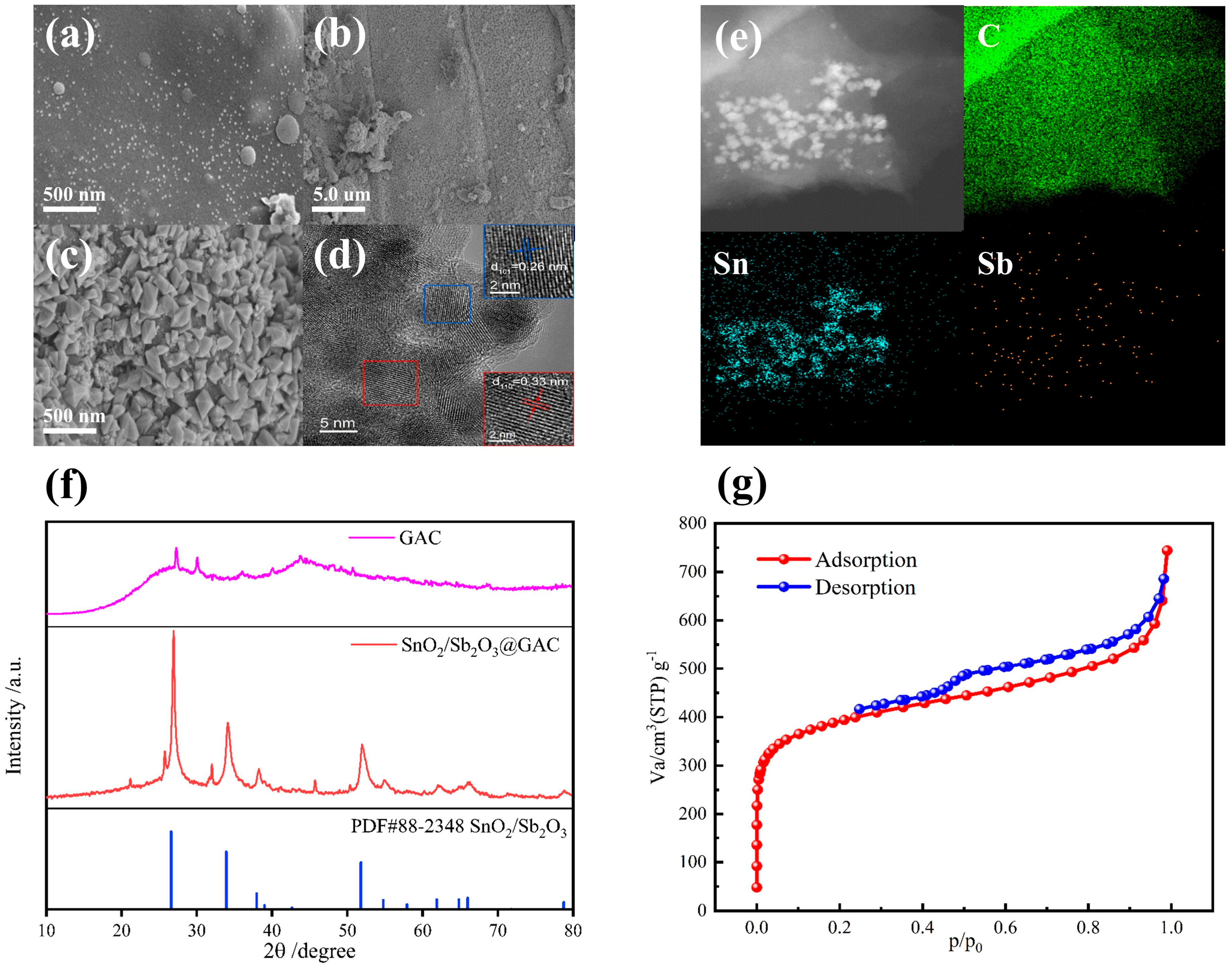
3.1. Characterization of Granular Electrode Materials
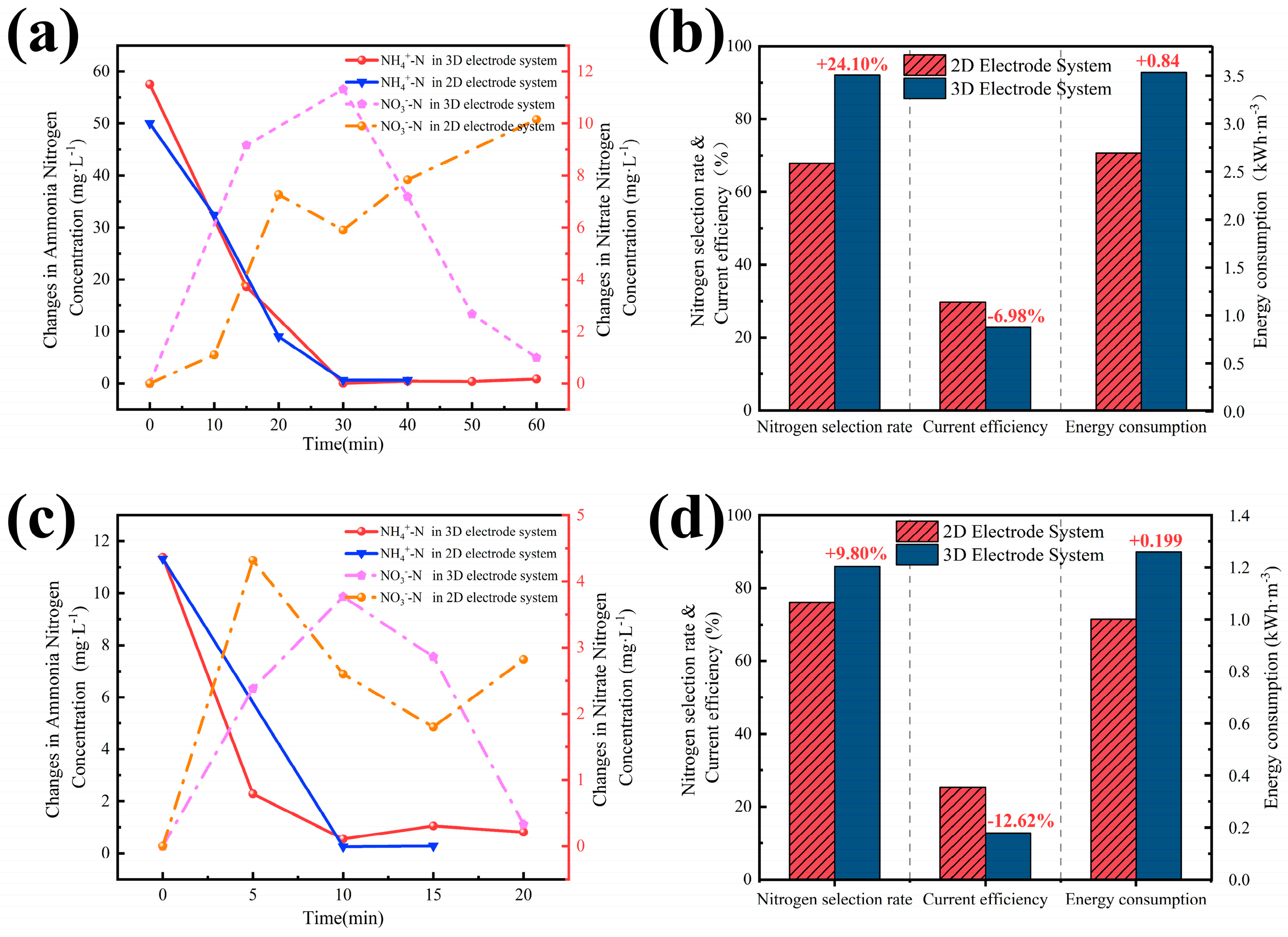
3.2. Mechanism and Effect of TAN Oxidation in Electrode Systems
3.3. Three-Dimensional Electrode System Treats Simulated Real Marine Aquaculture TAN Wastewater
4. Conclusions
Supplementary Materials
 , with TBA;
, with TBA;  , without TBA; Figure S3: (a,b) Variation curves of TAN and nitrate nitrogen concentrations at different Cl− concentrations in two-dimensional/three-dimensional electrode systems when the TAN concentration in the freshwater system is 10 mg·L−1;
, without TBA; Figure S3: (a,b) Variation curves of TAN and nitrate nitrogen concentrations at different Cl− concentrations in two-dimensional/three-dimensional electrode systems when the TAN concentration in the freshwater system is 10 mg·L−1;  , NH4+-N with 500 mg L−1 NaCl;
, NH4+-N with 500 mg L−1 NaCl;  , NH4+-N with 1250 mg L−1 NaCl;
, NH4+-N with 1250 mg L−1 NaCl;  , NO3−-N with 500 mg L−1 NaCl;
, NO3−-N with 500 mg L−1 NaCl;  , NO3−-N with 1250 mg L−1 NaCl; (c–e) Comparison of Cl− concentration, nitrogen selection rate, current efficiency, and energy consumption between two-dimensional and three-dimensional electrode systems;
, NO3−-N with 1250 mg L−1 NaCl; (c–e) Comparison of Cl− concentration, nitrogen selection rate, current efficiency, and energy consumption between two-dimensional and three-dimensional electrode systems;  , 2D Electrode System;
, 2D Electrode System;  , 3D Electrode System; Figure S4: Standard curves for (a) ammonia nitrogen, (b) nitrate nitrogen, (c) nitrite nitrogen, and (d) total chlorine. Table S1: Main Instruments and Equipment for the Experiment; Table S2: Chemicals; Table S3: Main components of ecological sea salt and their concentrations; Table S4: Determination methods of conventional water quality indicators; method for TAN, ref. [25]; free chlorine, ref. [28]; combined chlorine, total chlorine minus free chlorine; total chlorine, ref. [28]; nitrate nitrogen, ref. [36]; nitrite nitrogen, ref. [27]; pH, with pH meter; Table S5: Water quality standards for freshwater aquaculture water; standard for TAN, ref. [50]; total chlorine, ref. [51]; nitrate nitrogen, ref. [50]; nitrite nitrogen, ref. [36]; pH, ref. [52]; Table S6: Water quality standards for mariculture water; standard for TAN, ref. [50]; total chlorine, ref. [53]; nitrate nitrogen, ref. [50]; nitrite nitrogen, ref. [26]; pH, ref. [54]; Table S7: Nitrogen adsorption and desorption instrument parameters; Table S8: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 500 mg·L−1; Table S9: Values for each parameter; Table S10: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 1250 mg·L−1; Table S11: Values for each parameter; Table S12: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 1875 mg·L−1; Table S13: Values for each parameter; Table S14: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 500 mg·L−1; Table S15: Values for each parameter; Table S16: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 1250 mg·L−1; Table S17: Values for each parameter; Table S18: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 1875 mg·L−1; Table S19: Values for each parameter; Table S20: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 10 mg·L−1 and NaCl concentration of 500 mg·L−1; Table S21: Values for each parameter; Table S22: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 10 mg·L−1 and NaCl concentration of 1250 mg·L−1; Table S23: Values for each parameter; Table S24: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 10 mg·L−1 and NaCl concentration of 500 mg·L−1; Table S25: Values for each parameter; Table S26: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 10 mg·L−1 and NaCl concentration of 1250 mg·L−1; Table S27: Values for each parameter; Table S28: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine concentration in two-dimensional electrode system of seawater system at initial ammonia nitrogen concentration of 50 mg·L−1; Table S29: Values for each parameter; Table S30: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine concentration in three-dimensional electrode system of seawater system at initial ammonia nitrogen concentration of 50 mg·L−1; Table S31: Values for each parameter; Table S32: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine concentration in two-dimensional electrode system of seawater system at initial ammonia nitrogen concentration of 10 mg·L−1; Table S33: Values for each parameter; Table S34: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine concentration in three-dimensional electrode system of seawater system at initial ammonia nitrogen concentration of 10 mg·L−1; Table S35: Values for each parameter; Table S36: Changes in ammonia nitrogen, nitrate nitrogen, and nitrite nitrogen concentrations in simulated real mariculture wastewater treated with three-dimensional electrode system; Table S37: Values for each parameter.
, 3D Electrode System; Figure S4: Standard curves for (a) ammonia nitrogen, (b) nitrate nitrogen, (c) nitrite nitrogen, and (d) total chlorine. Table S1: Main Instruments and Equipment for the Experiment; Table S2: Chemicals; Table S3: Main components of ecological sea salt and their concentrations; Table S4: Determination methods of conventional water quality indicators; method for TAN, ref. [25]; free chlorine, ref. [28]; combined chlorine, total chlorine minus free chlorine; total chlorine, ref. [28]; nitrate nitrogen, ref. [36]; nitrite nitrogen, ref. [27]; pH, with pH meter; Table S5: Water quality standards for freshwater aquaculture water; standard for TAN, ref. [50]; total chlorine, ref. [51]; nitrate nitrogen, ref. [50]; nitrite nitrogen, ref. [36]; pH, ref. [52]; Table S6: Water quality standards for mariculture water; standard for TAN, ref. [50]; total chlorine, ref. [53]; nitrate nitrogen, ref. [50]; nitrite nitrogen, ref. [26]; pH, ref. [54]; Table S7: Nitrogen adsorption and desorption instrument parameters; Table S8: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 500 mg·L−1; Table S9: Values for each parameter; Table S10: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 1250 mg·L−1; Table S11: Values for each parameter; Table S12: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 1875 mg·L−1; Table S13: Values for each parameter; Table S14: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 500 mg·L−1; Table S15: Values for each parameter; Table S16: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 1250 mg·L−1; Table S17: Values for each parameter; Table S18: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 50 mg·L−1 and NaCl concentration of 1875 mg·L−1; Table S19: Values for each parameter; Table S20: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 10 mg·L−1 and NaCl concentration of 500 mg·L−1; Table S21: Values for each parameter; Table S22: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in two-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 10 mg·L−1 and NaCl concentration of 1250 mg·L−1; Table S23: Values for each parameter; Table S24: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 10 mg·L−1 and NaCl concentration of 500 mg·L−1; Table S25: Values for each parameter; Table S26: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine chloride concentration in three-dimensional electrode system of freshwater system at initial ammonia nitrogen concentration of 10 mg·L−1 and NaCl concentration of 1250 mg·L−1; Table S27: Values for each parameter; Table S28: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine concentration in two-dimensional electrode system of seawater system at initial ammonia nitrogen concentration of 50 mg·L−1; Table S29: Values for each parameter; Table S30: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine concentration in three-dimensional electrode system of seawater system at initial ammonia nitrogen concentration of 50 mg·L−1; Table S31: Values for each parameter; Table S32: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine concentration in two-dimensional electrode system of seawater system at initial ammonia nitrogen concentration of 10 mg·L−1; Table S33: Values for each parameter; Table S34: Changes of ammonia nitrogen, nitrate nitrogen, and active chlorine concentration in three-dimensional electrode system of seawater system at initial ammonia nitrogen concentration of 10 mg·L−1; Table S35: Values for each parameter; Table S36: Changes in ammonia nitrogen, nitrate nitrogen, and nitrite nitrogen concentrations in simulated real mariculture wastewater treated with three-dimensional electrode system; Table S37: Values for each parameter.Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| TAN | Total Ammonia Nitrogen |
| 3D | Three-Dimensional |
| 2D | Two-Dimensional |
| EO | Electrochemical Oxidation |
| GAC | Granular Activated Carbon |
| SEM | Scanning Electron Microscopy |
| TEM | Transmission Electron Microscopy |
| XRD | X-ray Diffraction |
| CV | Cyclic Voltammograms |
| CER | Chlorine-Evolution Reaction |
| OER | Oxygen Evolution Reaction |
| DPD | N, N-diethyl-1,4-phenylenediamine |
| TBA | Tert-Butanol |
| MABR | Membrane Aerated Biofilm Reactor |
| AOB | Ammonia-Oxidizing Bacteria |
| UV | Ultraviolet |
| DC | Direct Current |
| PMMA | Polymethyl Methacrylate |
| RHE | Reversible Hydrogen Electrode |
| N-DBPs | Nitrogenous Disinfection Byproducts |
| HAAs | Haloacetic Acids |
| HNMs | Halo-N-methylamines |
| NAs | Nitrosamines |
| LEV | Levofloxacin |
| SMX | Sulfamethoxazole |
References
- Shao, Q.; Zhang, Y.; Liu, Z.; Long, L.; Liu, Z.; Chen, Y.; Hu, X.-M.; Lu, M.; Huang, L.-Z. Phosphorus and nitrogen recovery from wastewater by ceramsite: Adsorption mechanism, plant cultivation and sustainability analysis. Sci. Total Environ. 2022, 805, 150288. [Google Scholar] [CrossRef]
- Han, F.; Zhou, W. Nitrogen recovery from wastewater by microbial assimilation—A review. Bioresour. Technol. 2022, 363, 127933. [Google Scholar] [CrossRef] [PubMed]
- Zangeneh, A.; Sabzalipour, S.; Takdatsan, A.; Yengejeh, R.J.; Khafaie, M.A. Ammonia removal form municipal wastewater by air stripping process: An experimental study. S. Afr. J. Chem. Eng. 2021, 36, 134–141. [Google Scholar] [CrossRef]
- Pourbavarsad, M.S.; Jalalieh, B.J.; Harkins, C.; Sevanthi, R.; Jackson, W.A. Nitrogen oxidation and carbon removal from high strength nitrogen habitation wastewater with nitrification in membrane aerated biological reactors. J. Environ. Chem. Eng. 2021, 9, 106271. [Google Scholar] [CrossRef]
- Guida, S.; Conzelmann, L.; Remy, C.; Vale, P.; Jefferson, B.; Soares, A. Resilience and life cycle assessment of ion exchange process for ammonium removal from municipal wastewater. Sci. Total Environ. 2021, 783, 146834. [Google Scholar] [CrossRef] [PubMed]
- Randall, D.; Tsui, T. Ammonia toxicity in fish. Mar. Pollut. Bull. 2002, 45, 17–23. [Google Scholar] [CrossRef]
- Zhong, L.; Liu, S.; Zuo, F.; Geng, Y.; Ouyang, P.; Chen, D.; Yang, S.; Zheng, W.; Xiong, Y.; Cai, W.; et al. The IL17 signaling pathway: A potential signaling pathway mediating gill hyperplasia and inflammation under ammonia nitrogen stress was identified by multi-omics analysis. Sci. Total Environ. 2023, 867, 161581. [Google Scholar] [CrossRef]
- Xu, Z.; Cao, J.; Qin, X.; Qiu, W.; Mei, J.; Xie, J. Toxic Effects on Bioaccumulation, Hematological Parameters, Oxidative Stress, Immune Responses and Tissue Structure in Fish Exposed to Ammonia Nitrogen: A Review. Animals 2021, 11, 3304. [Google Scholar] [CrossRef]
- Shen, C.; Cao, S.; Mohsen, M.; Li, X.; Wang, L.; Lu, K.; Zhang, C.; Song, K. Effects of chronic nitrite exposure on hematological parameters, oxidative stress and apoptosis in spotted seabass (Lateolabrax maculatus) reared at high temperature. Aquac. Rep. 2024, 35, 102022. [Google Scholar] [CrossRef]
- Camargo, J.A.; Alonso, A.; Salamanca, A. Nitrate toxicity to aquatic animals: A review with new data for freshwater invertebrates. Chemosphere 2005, 58, 1255–1267. [Google Scholar] [CrossRef]
- Cheng, N.; Wang, B.; Feng, Q.; Zhang, X.; Chen, M. Co-adsorption performance and mechanism of nitrogen and phosphorus onto eupatorium adenophorum biochar in water. Bioresour. Technol. 2021, 340, 11. [Google Scholar] [CrossRef]
- Farghali, M.; Chen, Z.; Osman, A.I.; Ali, I.M.; Hassan, D.; Ihara, I.; Rooney, D.W.; Yap, P.-S. Strategies for ammonia recovery from wastewater: A review. Environ. Chem. Lett. 2024, 22, 2699–2751. [Google Scholar] [CrossRef]
- Terada, A.; Yamamoto, T.; Igarashi, R.; Tsuneda, S.; Hirata, A. Feasibility of a membrane-aerated biofilm reactor to achieve controllable nitrification. Biochem. Eng. J. 2006, 28, 123–130. [Google Scholar] [CrossRef]
- Zhang, X.; Li, W.; Blatchley, E.; Wang, X.; Ren, P. UV/chlorine process for ammonia removal and disinfection by-product reduction: Comparison with chlorination. Water Res. 2015, 68, 804–811. [Google Scholar] [CrossRef]
- Capodaglio, A.G.; Hlavínek, P.; Raboni, M. Physico-chemical technologies for nitrogen removal from wastewaters: A review. Rev. Ambient. Água 2015, 10, 481–498. [Google Scholar] [CrossRef]
- Zhang, Y.; Yin, S.; Li, H.; Liu, J.; Li, S.; Zhang, L. Treatment of ammonia-nitrogen wastewater by the ultrasonic strengthened break point chlorination method. J. Water Process Eng. 2022, 45, 102501. [Google Scholar] [CrossRef]
- Yao, J.; Mei, Y.; Yuan, T.; Chen, J.; Pan, H.; Wang, J. Electrochemical removal of nitrate from wastewater with a Ti cathode and Pt anode for high efficiency and N2 selectivity. J. Electroanal. Chem. 2021, 882, 7. [Google Scholar] [CrossRef]
- Díaz, V.; Ibáñez, R.; Gómez, P.; Urtiaga, A.; Ortiz, I. Kinetics of electro-oxidation of ammonia-N, nitrites and COD from a recirculating aquaculture saline water system using BDD anodes. Water Res. 2011, 45, 125–134. [Google Scholar] [CrossRef]
- Li, F.; Peng, X.; Liu, Y.; Mei, J.; Sun, L.; Shen, C.; Ma, C.; Huang, M.; Wang, Z.; Sand, W. A chloride-radical-mediated electrochemical filtration system for rapid and effective transformation of ammonia to nitrogen. Chemosphere 2019, 229, 383–391. [Google Scholar] [CrossRef]
- Baqer, A.R.; Beddai, A.A.; Farhan, M.M.; Badday, B.A.; Mejbel, M.K. Efficient coating of titanium composite electrodes with various metal oxides for electrochemical removal of ammonia. Results Eng. 2021, 9, 100199. [Google Scholar] [CrossRef]
- Luna-Trujillo, M.; Palma-Goyes, R.; Vazquez-Arenas, J.; Manzo-Robledo, A. Formation of active chlorine species involving the higher oxide MOx+1 on active Ti/RuO2-IrO2 anodes: A DEMS analysis. J. Electroanal. Chem. 2020, 878, 7. [Google Scholar] [CrossRef]
- Quan, F.; Zhan, G.; Zhou, B.; Ling, C.; Wang, X.; Shen, W.; Li, J.; Jia, F.; Zhang, L. Electrochemical removal of ammonium nitrogen in high efficiency and N2 selectivity using non-noble single-atomic iron catalyst. J. Environ. Sci. 2023, 125, 544–552. [Google Scholar] [CrossRef]
- Cao, F.; Tan, J.; Zhang, S.; Wang, H.; Yao, C.; Li, Y. Preparation and Recent Developments of Ti/SnO2-Sb Electrodes. J. Chem. 2021, 2021, 2107939. [Google Scholar] [CrossRef]
- Hoang, V.; Nguyen, D.; Tu, N.; Tran, V.; Lam, V.; Duong, T. One-Step Hydrothermal Synthesis and Characterization of Highly Dispersed Sb-Doped SnO2 Nanoparticles for Supercapacitor Applications. Electrochem 2025, 6, 22. [Google Scholar] [CrossRef]
- HJ 535-2009; Water Quality—Determination of Ammonia Nitrogen—Nessler’s Reagent Spectrophotometry. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2009.
- HJ/T 346-2007; Water Quality—Determination of Nitrate-Nitrogen-Ultraviolet Spectrophotometry. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2007.
- GB 7493-1987; Water Quality—Determination of Nitrogen (Nitrite)—Spectrophotometric Method. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 1987.
- HJ 586-2010; Water Quality—Determination of Free Chlorine and Total Chlorine-Spectrophotonetric Method Using N,N-Diethyl-1,4-Phenylenediamine. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2010.
- Razack, G.; Wang, J.; Zhao, X.; Noel, W.; Sun, H.; Pang, J.; Ding, J.; Wang, W.; Yang, X.; Cui, C.; et al. Current State of Research on the Three-Dimensional Particle Electrode System for Treating Organic Pollutants from Wastewater Streams: Particle Electrode, Degradation Mechanism, and Synergy Effects. Water 2025, 17, 2490. [Google Scholar] [CrossRef]
- Liu, S.; Lu, J.; Yu, X.; Pang, H.; Zhang, Q.; Park, H.S. Rational design of metal–organic framework-nanoparticle composite electrocatalysts for sustainable nitrogen electrochemistry. eScience 2025, 5, 100378. [Google Scholar] [CrossRef]
- Teng, X.; Si, D.; Chen, L.; Shi, J. Synergetic catalytic effects by strong metal–support interaction for efficient electrocatalysis. eScience 2024, 4, 100272. [Google Scholar] [CrossRef]
- Pu, Y.; Zhao, F.; Chen, Y.; Lin, X.; Yin, H.; Tang, X. Enhanced Electrocatalytic Oxidation of Phenol by SnO2-Sb2O3/GAC Particle Electrodes in a Three-Dimensional Electrochemical Oxidation System. Water 2023, 15, 1844. [Google Scholar] [CrossRef]
- Li, X.; Lu, S.; Zhang, G. Three-dimensional structured electrode for electrocatalytic organic wastewater purification: Design, mechanism and role. J. Hazard. Mater. 2023, 445, 130524. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Liang, J.; Wei, P.; Liu, Q.; Xie, L.; Luo, Y.; Sun, X. ITO@TiO2 nanoarray:An efficient and robust nitrite reduction reaction electrocatalyst toward NH3 production under ambient conditions. eScience 2022, 2, 382–388. [Google Scholar] [CrossRef]
- Huang, F.; Pan, L.; He, Z.; Zhang, M.; Zhang, M. Culturable heterotrophic nitrification-aerobic denitrification bacterial consortia with cooperative interactions for removing ammonia and nitrite nitrogen in mariculture effluents. Aquaculture 2020, 523, 735211. [Google Scholar] [CrossRef]
- Hu, H. Study on Ammonia Treatment for Aquaculture Wastewater Abstract. Ph.D. Thesis, Ocean University of China, Qingdao, China, 2007. [Google Scholar]
- Ashour, M.; Alprol, A.; Heneash, A.; Saleh, H.; Abualnaja, K.; Alhashmialameer, D.; Mansour, A. Ammonia Bioremediation from Aquaculture Wastewater Effluents Using Arthrospira platensis NIOF17/003: Impact of Biodiesel Residue and Potential of Ammonia-Loaded Biomass as Rotifer Feed. Materials 2021, 14, 5460. [Google Scholar] [CrossRef]
- Ding, Y.; Guo, Z.; Ma, B.; Wang, F.; You, H.; Mei, J.; Hou, X.; Liang, Z.; Li, Z.; Jin, C. The Influence of Different Operation Conditions on the Treatment of Mariculture Wastewater by the Combined System of Anoxic Filter and Membrane Bioreactor. Membranes 2021, 11, 729. [Google Scholar] [CrossRef]
- Yan, Z.; Dai, Z.; Zheng, W.; Lei, Z.; Qiu, J.; Kuang, W.; Huang, W.; Feng, C. Facile ammonium oxidation to nitrogen gas in acid wastewater by in situ photogenerated chlorine radicals. Water Res. 2021, 205, 117678. [Google Scholar] [CrossRef] [PubMed]
- Shin, Y.; Yoo, H.; Kim, S.; Chung, K.; Park, Y.; Hwang, K.; Hong, S.; Park, H.; Cho, K.; Lee, J. Sequential Combination of Electro-Fenton and Electrochemical Chlorination Processes for the Treatment of Anaerobically-Digested Food Wastewater. Environ. Sci. Technol. 2017, 51, 10700–10710. [Google Scholar] [CrossRef] [PubMed]
- Xiao, S.; Qu, J.; Zhao, X.; Liu, H.; Wan, D. Electrochemical process combined with UV light irradiation for synergistic degradation of ammonia in chloride-containing solutions. Water Res. 2009, 43, 1432–1440. [Google Scholar] [CrossRef]
- Xu, J.; Wang, L.; Mao, X.; Zou, H.; Liu, G. Enhanced electrochlorination for efficient ammonia oxidation facilitated by accelerating electron cycling on Co2+/Co3+. J. Environ. Chem. Eng. 2025, 13, 115415. [Google Scholar] [CrossRef]
- Rahardjo, S.; Shih, Y. Electrochemical characteristics of silver/nickel oxide (Ag/Ni) for direct ammonia oxidation and nitrogen selectivity in paired electrode system. Chem. Eng. J. 2023, 452, 139370. [Google Scholar] [CrossRef]
- Pous, N.; Baneras, L.; Corvini, P.F.-X.; Liu, S.-J.; Puig, S. Direct ammonium oxidation to nitrogen gas (Dirammox) in Alcaligenes strain HO-1: The electrode role. Environ. Sci. Ecotechnol. 2023, 15, 100253. [Google Scholar] [CrossRef] [PubMed]
- Ji, Y.; Bai, J.; Li, J.; Luo, T.; Qiao, L.; Zeng, Q.; Zhou, B. Highly selective transformation of ammonia nitrogen to N2 based on a novel solar-driven photoelectrocatalytic-chlorine radical reactions system. Water Res. 2017, 125, 512–519. [Google Scholar] [CrossRef]
- Liu, S.; Wang, Z.; Li, J.; Zhao, C.; He, X.; Yang, G. Fabrication of slag particle three-dimensional electrode system for methylene blue degradation: Characterization, performance and mechanism study. Chemosphere 2018, 213, 377–383. [Google Scholar] [CrossRef] [PubMed]
- Lan, H.; Liu, X.; Liu, H.; Liu, R.; Hu, C.; Qu, J. Efficient Nitrate Reduction in a Fluidized Electrochemical Reactor Promoted by Pd-Sn/AC Particles. Catal. Lett. 2016, 146, 91–99. [Google Scholar] [CrossRef]
- Yao, J.; Mei, Y.; Jiang, J.; Xia, G.; Chen, J. Process Optimization of Electrochemical Treatment of COD and Total Nitrogen Containing Wastewater. Int. J. Environ. Res. Public Health 2022, 19, 850. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Chen, Z.; Mow, M.; Liao, X.; Wei, X.; Ma, G.; Wang, X.; Yu, H. Chloramine Disinfection of Levofloxacin and Sulfaphenazole: Unraveling Novel Disinfection Byproducts and Elucidating Formation Mechanisms for an Enhanced Understanding of Water Treatment. Molecules 2024, 29, 396. [Google Scholar] [CrossRef] [PubMed]
- GB 3838-2002; Environmental Quality Standards for Surface Water. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 2002.
- SC/T 9101-2007; Requirement for Water Discharge from Freshwater Aquaculture Pond. Ministry of Agriculture and Rural Affairs of the People’s Republic of China: Beijing, China, 2007.
- GB 11607-89; Water Quality Standard for Fisheries. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 1989.
- SC/T 9103—2007; Water Drainage Standard for Sea Water Mariculture. Ministry of Agriculture and Rural Affairs of the People’s Republic of China: Beijing, China, 2007.
- GB 3097-1997; Sea Water Quality Standard. Ministry of Ecology and Environment of the People’s Republic of China: Beijing, China, 1997.




| Methods | Craftsmanship | Initial TAN Concentration | TAN Removal Efficiency | Final Product |
|---|---|---|---|---|
| Physical Method [11] | EBC Adsorption | 50.0 mg·L−1 | 1.909 mg·g−1 | within EBC |
| Biological Method [13] | MABR (nitrification) | 100.0 mg·L−1 | Nearly 100% | NO3− |
| Breakpoint Chlorination [14] | 254 nm UV with HClO | 1.0 mg·L−1 | 50% | NO3−, NO2− |
| Electrochemical Oxidation [48] | Ti/PbO2 Electrodes | 60.0 mg·L−1 | Nearly 100% | N2 (87.00%), NO3−, NH2Cl |
| This work | SnO2-Sb2O3@GAC granular electrode | 50.0 mg·L−1 | Nearly 100% | N2 (92.60%), NO3− (1.96%), NH2Cl (5.44%) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, Y.; Pan, Z.; Lv, Y.; Ling, G.; Zhang, C. Degassing N2 from the Direct Oxidation of Total Ammonia in Mariculture Using a Three-Dimensional Electrode System. Processes 2025, 13, 3851. https://doi.org/10.3390/pr13123851
He Y, Pan Z, Lv Y, Ling G, Zhang C. Degassing N2 from the Direct Oxidation of Total Ammonia in Mariculture Using a Three-Dimensional Electrode System. Processes. 2025; 13(12):3851. https://doi.org/10.3390/pr13123851
Chicago/Turabian StyleHe, Yuxiang, Ziyi Pan, Ya’nan Lv, Guowei Ling, and Chen Zhang. 2025. "Degassing N2 from the Direct Oxidation of Total Ammonia in Mariculture Using a Three-Dimensional Electrode System" Processes 13, no. 12: 3851. https://doi.org/10.3390/pr13123851
APA StyleHe, Y., Pan, Z., Lv, Y., Ling, G., & Zhang, C. (2025). Degassing N2 from the Direct Oxidation of Total Ammonia in Mariculture Using a Three-Dimensional Electrode System. Processes, 13(12), 3851. https://doi.org/10.3390/pr13123851






