The Pursuit of Energy Reduction in Generation of Stable Nanobubbles
Abstract
1. Introduction
2. Materials and Methods
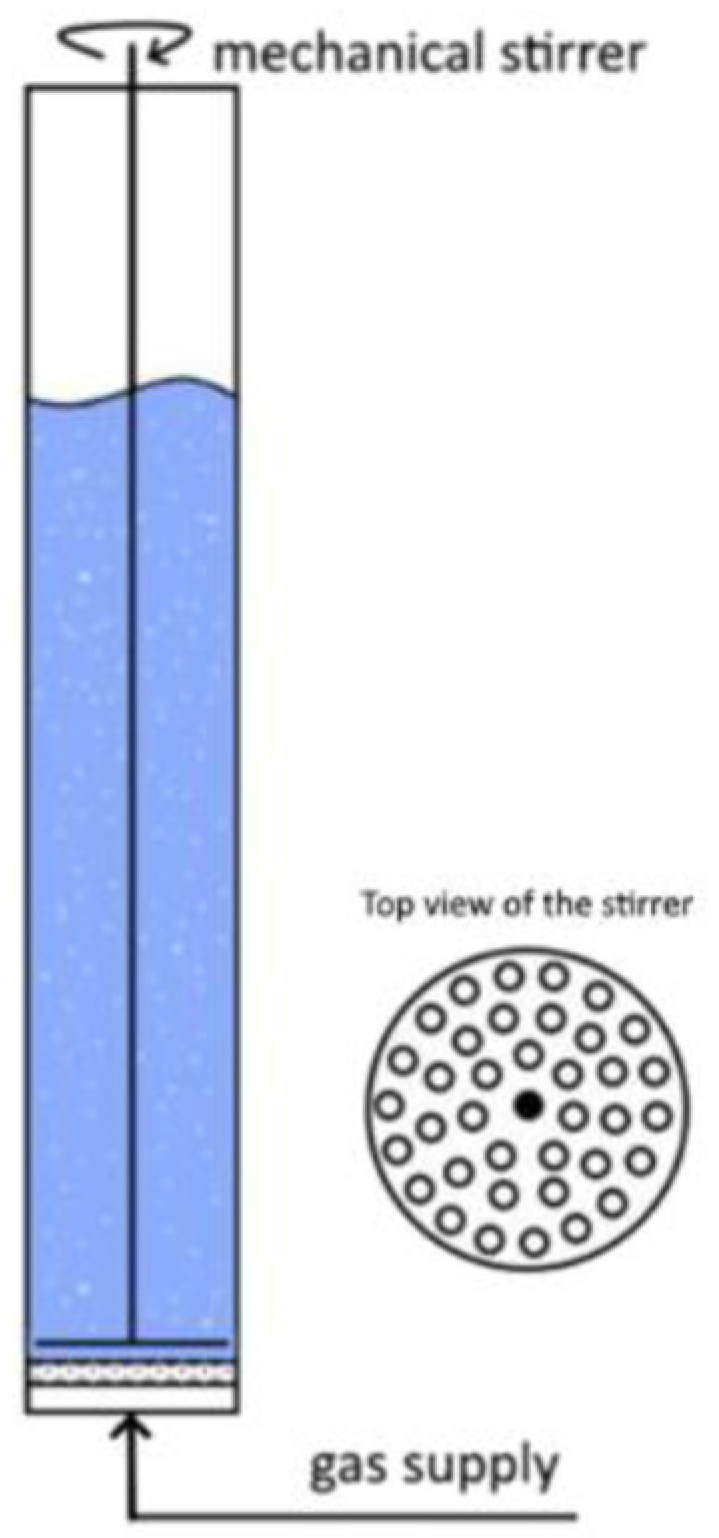
2.1. Generation Setup
2.2. Media Preparation
2.3. Experiment 1: Usability Study of Common Hydrodynamic Criteria for the Generation of Bubbles in Various Liquids
2.4. Experiment 2: Nanobubble Stability Assessment in Non-Ionic Surfactant Solutions
3. Results and Discussion
3.1. Experiment 1
3.2. Experiment 2
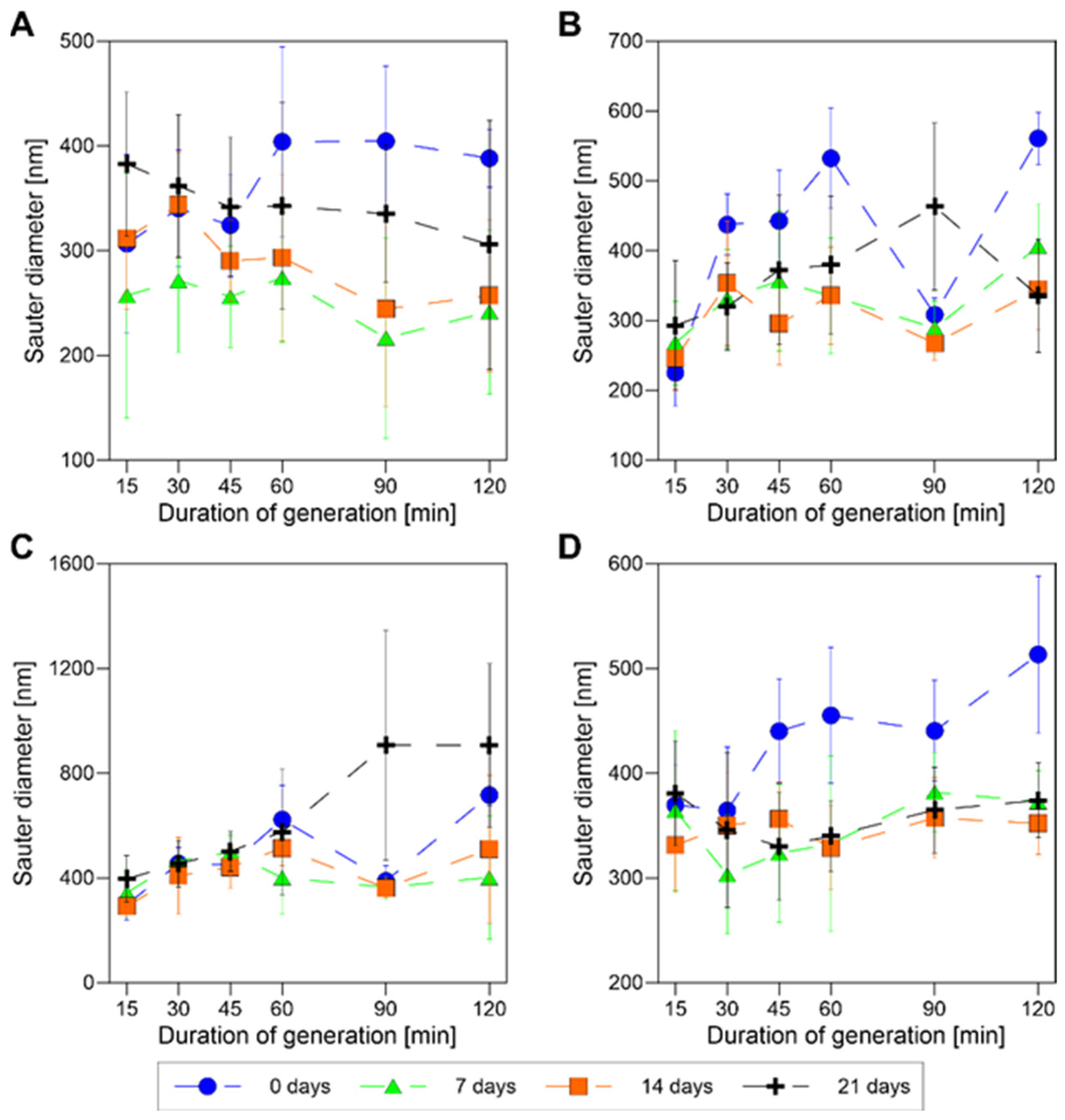
3.2.1. Does the Duration of the Generation of Bubbles in Our Generation Setup Affect the Stability of Nanobubbles in the Presence of Different Concentrations of P-123 Surfactant for the Constant Rotation Rate of the Impeller?
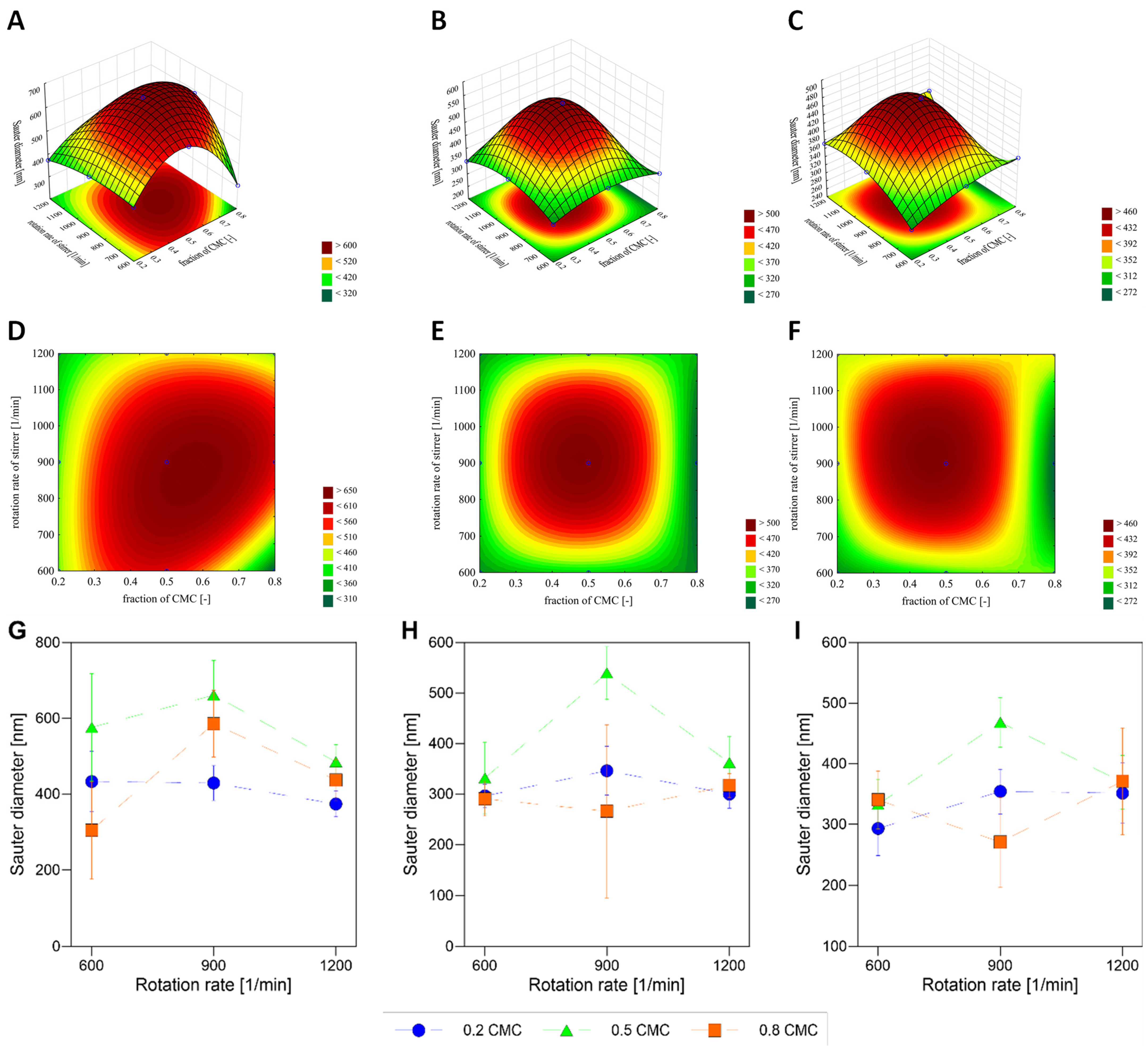
3.2.2. How Does the Change in the Rotation Rate of the Impeller Affect the Stability of Nanobubbles for Constant Generation Time for Pluronic P-123?
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Ca | Capillary number, (-) |
| CMC | Critical micellar concentration, (g/dm3) |
| CXTAB | Alkyl trimethylammonium bromide |
| d | Diameter of a impeller, (m) |
| d32 | Sauter diameter of nanobubbles, (nm) |
| dpore | Diameter of the membrane pore, (m) |
| DLS | Dynamic Light Scattering |
| Eo | Eötvös number, (-) |
| g | Gravitational acceleration, (m/s2) |
| n | Rotation rate of an impeller, (1/s) |
| nmin | Minimal rotation rate of the impeller needed for needed for nanobubble generation, (1/s) |
| Mo | Morton number, (-) |
| NTA | Nanoparticle Tracking Analysis |
| P-123 | Pluronic P-123 |
| PEG | Poly(ethylene glycol) |
| PPG | Poly(propylene glycol) |
| R2 | Regression coefficient, [-] |
| Re | Reynolds number, [-] |
| RSSE | Residual sum of squares error, unit varies |
| s | Solubility of gas in liquid, (ggas/100 gsolvent) |
| SDS | Sodium dodecyl sulphate |
| SLS | Static Light Scattering |
| Surface-averaged velocity of the impeller, (m/s) | |
| Greek letters: | |
| Δ+ | Overestimation error, (-) |
| Δ− | Underestimation error, (-) |
| μ | Dynamic viscosity of a liquid, (Pa∙s) |
| ρ | Density of a liquid, (kg/m3) |
| σ | Surface tension between liquid and air, (N/m) |
| Ratio of Sauter diameter and membrane pore diameter, (m/m) | |
| Subscripts: | |
| G | Corresponding to gas phase |
| L | Corresponding to liquid phase |
References
- Bunkin, N.F.; Kochergin, A.V.; Lobeyev, A.V.; Ninham, B.W.; Vinogradova, O.I. Existence of charged submicrobubble clusters in polar liquids as revealed by correlation between optical cavitation and electrical conductivity. Colloids Surf. A Physicochem. Eng. Asp. 1996, 110, 207–212. [Google Scholar] [CrossRef]
- Ljunggren, S.; Eriksson, J.C. The lifetime of a colloid-sized gas bubble in water and the cause of the hydrophobic attraction. Colloids Surf. A Physicochem. Eng. Asp. 1997, 129–130, 151–155. [Google Scholar] [CrossRef]
- Brenner, M.P.; Lohse, D. Dynamic equilibrium mechanism for surface nanobubble stabilization. Phys. Rev. Lett. 2008, 101, 214505. [Google Scholar] [CrossRef] [PubMed]
- Bunkin, N.F.; Suyazov, N.V.; Shkirin, V.; Ignat’ev, P.S.; Indukaev, K.V. Determination of the microstructure of gas bubbles in highly purified water by measuring the elements of the laser radiation scattering matrix. Quantum Electron. 2009, 39, 367–381. [Google Scholar] [CrossRef]
- Fan, M.; Tao, D.; Honaker, R.; Luo, Z. Nanobubble generation and its application in froth flotation (part I): Nanobubble generation and its effects on properties of microbubble and millimeter scale bubble solutions. Min. Sci. Technol. 2010, 20, 1–19. [Google Scholar] [CrossRef]
- Yamamoto, T.; Ohnishi, S. Molecular dynamics study on helium nanobubbles in water. Phys. Chem. Chem. Phys. 2011, 13, 16142. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Nesset, K.; Masliyah, J.; Xu, Z. Generation and characterization of submicron size bubbles. Adv. Colloid Interface Sci. 2012, 179–182, 123–132. [Google Scholar] [CrossRef]
- Nakatake, Y.; Kisu, S.; Shigyo, K.; Eguchi, T.; Watanabe, T. Effect of nano air-bubbles mixed into gas oil on common-rail diesel engine. Energy 2013, 59, 233–239. [Google Scholar] [CrossRef]
- Calgaroto, S.; Wilberg, K.Q.; Rubio, J. On the nanobubbles interfacial properties and future applications in flotation. Miner. Eng. 2014, 60, 33–40. [Google Scholar] [CrossRef]
- Zhao, W.; Hu, X.; Duan, J.; Liu, T.; Liu, M.; Dong, Y. Oxygen release from nanobubbles adsorbed on hydrophobic particles. Chem. Phys. Lett. 2014, 608, 224–228. [Google Scholar] [CrossRef]
- Wang, X.; Yuan, T.; Lei, Z.; Kobayashi, M.; Adachi, Y.; Shimizu, K.; Lee, D.J.; Zhang, Z. Supplementation of O2-containing gas nanobubble water to enhance methane production from anaerobic digestion of cellulose. Chem. Eng. J. 2020, 398, 125652. [Google Scholar] [CrossRef]
- Kim, J.Y.; Song, M.G.; Kim, J.D. Zeta potential of nanobubbles generated by ultrasonication in aqueous alkyl polyglycoside solutions. J. Colloid Interface Sci. 2000, 223, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Kikuchi, K.; Takeda, H.; Rabolt, B.; Okaya, T.; Ogumi, Z.; Saihara, Y.; Noguchi, H. Hydrogen particles and supersaturation in alkaline water from an Alkali-Ion-Water electrolyzer. J. Electroanal. Chem. 2001, 506, 22–27. [Google Scholar] [CrossRef]
- Rodrigues, R.T.; Rubio, J. New basis for measuring the size distribution of bubbles. Miner. Eng. 2003, 16, 757–765. [Google Scholar] [CrossRef]
- Cho, S.H.; Kim, J.Y.; Chun, J.H.; Kim, J.D. Ultrasonic formation of nanobubbles and their zeta-potentials in aqueous electrolyte and surfactant solutions. Colloids Surf. A Physicochem. Eng. Asp. 2005, 269, 28–34. [Google Scholar] [CrossRef]
- Kikuchi, K.; Tanaka, Y.; Saihara, Y.; Maeda, M.; Kawamura, M.; Ogumi, Z. Concentration of hydrogen nanobubbles in electrolyzed water. J. Colloid Interface Sci. 2006, 298, 914–919. [Google Scholar] [CrossRef]
- Alheshibri, M.; Qian, J.; Jehannin, M.; Craig, V.S.J. A History of Nanobubbles. Langmuir 2016, 32, 11086–11100. [Google Scholar] [CrossRef] [PubMed]
- Tsai, J.C.; Kumar, M.; Chen, S.Y.; Lin, J.G. Nano-bubble flotation technology with coagulation process for the cost-effective treatment of chemical mechanical polishing wastewater. Sep. Purif. Technol. 2007, 58, 61–67. [Google Scholar] [CrossRef]
- Kikuchi, K.; Ioka, A.; Oku, T.; Tanaka, Y.; Saihara, Y.; Ogumi, Z. Concentration determination of oxygen nanobubbles in electrolyzed water. J. Colloid Interface Sci. 2009, 329, 306–309. [Google Scholar] [CrossRef]
- Ebina, K.; Shi, K.; Hirao, M.; Hashimoto, J.; Kawato, Y.; Kaneshiro, S.; Morimoto, T.; Koizumi, K.; Yoshikawa, H. Oxygen and Air Nanobubble Water Solution Promote the Growth of Plants, Fishes, and Mice. PLoS ONE 2013, 8, e65339. [Google Scholar] [CrossRef]
- Hamamoto, S.; Takemura, T.; Suzuki, K.; Nishimura, T. Effects of pH on nano-bubble stability and transport in saturated porous media. J. Contam. Hydrol. 2017, 208, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Temesgen, T.; Bui, T.T.; Han, M.; Kim, T.I.; Park, H. Micro and nanobubble technologies as a new horizon for water-treatment techniques: A review. Adv. Colloid Interface Sci. 2017, 246, 40–51. [Google Scholar] [CrossRef]
- Ulatowski, K.; Sobieszuk, P.; Mróz, A.; Ciach, T. Stability of nanobubbles generated in water using porous membrane system. Chem. Eng. Process. Process Intensif. 2019, 136, 62–71. [Google Scholar] [CrossRef]
- Alheshibri, M.; Craig, V.S.J. Armoured nanobubbles; ultrasound contrast agents under pressure. J. Colloid Interface Sci. 2019, 537, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Nirmalkar, N.; Pacek, A.W.; Barigou, M. On the existence and stability of bulk nanobubbles. Langmuir 2018, 34, 10964–10973. [Google Scholar] [CrossRef] [PubMed]
- Mo, C.R.; Wang, J.; Fang, Z.; Zhou, L.M.; Zhang, L.J.; Hu, J. Formation and stability of ultrasonic generated bulk nanobubbles. Chin. Phys. B 2018, 27, 118104. [Google Scholar] [CrossRef]
- Ulatowski, K.; Fiuk, J.; Sobieszuk, P. Sterilisation of nanobubble dispersions. Chem. Process Eng. 2020, 41, 69–76. [Google Scholar] [CrossRef]
- Michailidi, E.D.; Bomis, G.; Varoutoglou, A.; Kyzas, G.Z.; Mitrikas, G.; Mitropoulos, A.C.; Efthimiadou, E.K.; Favvas, E.P. Bulk nanobubbles: Production and investigation of their formation/stability mechanism. J. Colloid Interface Sci. 2020, 564, 371–380. [Google Scholar] [CrossRef]
- Phan, K.K.T.; Truong, T.; Wang, Y.; Bhandari, B. Nanobubbles: Fundamental characteristics and applications in food processing. Trends Food Sci. Technol. 2020, 95, 118–130. [Google Scholar] [CrossRef]
- Xiao, W.; Xu, G. Mass transfer of nanobubble aeration and its effect on biofilm growth: Microbial activity and structural properties. Sci. Total Environ. 2020, 703, 134976. [Google Scholar] [CrossRef]
- Sayed Ahmed, A.S. Cavitation Nanobubble Enhanced Flotation Process for More Efficient Coal Recovery. 2013. Theses and Dissertations--Mining Engineering. 8. Available online: https://uknowledge.uky.edu/mng_etds/8 (accessed on 20 August 2023).
- Sobieszuk, P.; Strzyżewska, A.; Ulatowski, K. Investigation of the possibility of culturing aerobic yeast with oxygen nanobubble addition and evaluation of the results of batch and semi-batch cultures of Saccharomyces cerevisiae. Chem. Eng. Process. Process Intensif. 2021, 159, 108247. [Google Scholar] [CrossRef]
- Ulatowski, K.; Sobieszuk, P. Gas nanobubble dispersions as the important agent in environmental processes—Generation methods review. Water Environ. J. 2020, 34, 772–790. [Google Scholar] [CrossRef]
- Jin, J.; Wang, R.; Tang, J.; Yang, L.; Feng, Z.; Xu, C.; Yang, F.; Gu, N. Dynamic tracking of bulk nanobubbles from microbubbles shrinkage to collapse. Colloids Surf. A Physicochem. Eng. Asp. 2020, 589, 124430. [Google Scholar] [CrossRef]
- Tan, B.H.; An, H.; Ohl, C.D. Stability of surface and bulk nanobubbles. Curr. Opin. Colloid Interface Sci. 2021, 53, 101428. [Google Scholar] [CrossRef]
- Yadav, G.; Fabiano, L.A.; Soh, L.; Zimmerman, J.; Sen, R.; Seider, W.D. CO2 process intensification of algae oil extraction to biodiesel. AIChE J. 2021, 67, e16992. [Google Scholar] [CrossRef]
- Kim, H.; Han, M. A Study on the Development of Sub-Micron Bubble Generator and Characterization of Sub-Micron Bubble. Ph.D. Thesis, Seoul National University, Seoul, Republic of Korea, 2014. [Google Scholar]
- Li, H.; Hu, L.; Song, D.; Lin, F. Characteristics of Micro-Nano Bubbles and Potential Application in Groundwater Bioremediation. Water Environ. Res. 2014, 86, 844–851. [Google Scholar] [CrossRef]
- An, H.; Liu, G.; Craig, V.S.J. Wetting of nanophases: Nanobubbles, nanodroplets and micropancakes on hydrophobic surfaces. Adv. Colloid Interface Sci. 2015, 222, 9–17. [Google Scholar] [CrossRef]
- Yasui, K.; Tuziuti, T.; Kanematsu, W.; Kato, K. Dynamic Equilibrium Model for a Bulk Nanobubble and a Microbubble Partly Covered with Hydrophobic Material. Langmuir 2016, 32, 11101–11110. [Google Scholar] [CrossRef]
- Park, J.B.; Shin, D.; Kang, S.; Cho, S.P.; Hong, B.H. Distortion in Two-Dimensional Shapes of Merging Nanobubbles: Evidence for Anisotropic Gas Flow Mechanism. Langmuir 2016, 32, 11303–11308. [Google Scholar] [CrossRef]
- Leroy, V.; Norisuye, T. Investigating the Existence of Bulk Nanobubbles with Ultrasound. ChemPhysChem 2016, 17, 2787–2790. [Google Scholar] [CrossRef]
- Hu, L.; Xia, Z. Application of ozone micro-nano-bubbles to groundwater remediation. J. Hazard. Mater. 2018, 342, 446–453. [Google Scholar] [CrossRef] [PubMed]
- Ushikubo, F.Y.; Furukawa, T.; Nakagawa, R.; Enari, M.; Makino, Y.; Kawagoe, Y.; Shiina, T.; Oshita, S. Evidence of the existence and the stability of nano-bubbles in water. Colloids Surf. A Physicochem. Eng. Asp. 2010, 361, 31–37. [Google Scholar] [CrossRef]
- Uchida, T.; Liu, S.; Enari, M.; Oshita, S.; Yamazaki, K.; Gohara, K. Effect of NaCl on the lifetime of micro- and nanobubbles. Nanomaterials 2016, 6, 31. [Google Scholar] [CrossRef] [PubMed]
- Zinjenab, Z.T.; Azimi, E.; Shadman, M.; Hosseini, M.R.; Abbaszadeh, M.; Namgar, S.M. Nano-microbubbles and feed size interaction in lead and zinc sulfide minerals flotation. Chem. Eng. Process. Process Intensif. 2023, 189, 109401. [Google Scholar] [CrossRef]
- Ohgaki, K.; Khanh, N.Q.; Joden, Y.; Tsuji, A.; Nakagawa, T. Physicochemical approach to nanobubble solutions. Chem. Eng. Sci. 2010, 65, 1296–1300. [Google Scholar] [CrossRef]
- Terasaka, K.; Hirabayashi, A.; Nishino, T.; Fujioka, S.; Kobayashi, D. Development of microbubble aerator for waste water treatment using aerobic activated sludge. Chem. Eng. Sci. 2011, 66, 3172–3179. [Google Scholar] [CrossRef]
- Ulatowski, K.; Sobieszuk, P. Influence of liquid flowrate on size of nanobubbles generated by porous-membrane modules. Chem. Process Eng. 2018, 39, 335–345. [Google Scholar] [CrossRef]
- Khan, M.S.; Hwang, J.; Lee, K.; Choi, Y.; Kim, K.; Koo, H.J.; Hong, J.W.; Choi, J. Oxygen-carrying micro/nanobubbles: Composition, synthesis techniques and potential prospects in photo-triggered theranostics. Molecules 2018, 23, 2210. [Google Scholar] [CrossRef]
- Wang, Q.; Zhao, H.; Qi, N.; Qin, Y.; Zhang, X.; Li, Y. Generation and Stability of Size-Adjustable Bulk Nanobubbles Based on Periodic Pressure Change. Sci. Rep. 2019, 9, 1118. [Google Scholar] [CrossRef]
- Ashokkumar, M. The characterization of acoustic cavitation bubbles—An overview. Ultrason. Sonochem. 2011, 18, 864–872. [Google Scholar] [CrossRef]
- German, S.R.; Edwards, M.A.; Chen, Q.; Liu, Y.; Luo, L.; White, H.S. Electrochemistry of single nanobubbles. Estimating the critical size of bubble-forming nuclei for gas-evolving electrode reactions. Faraday Discuss. 2016, 193, 223–240. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Li, M.; Xu, X.; Sun, C. On the role of surface charge and surface tension tuned by surfactant in stabilizing bulk nanobubbles. Appl. Surf. Sci. 2023, 608, 155232. [Google Scholar] [CrossRef]
- Hayashi, H.; Yamada, S.; Kumada, Y.; Matsuo, H.; Toriyama, T.; Kawahara, H. Immersing Feet in Carbon Dioxide-enriched Water Prevents Expansion and Formation of Ischemic Ulcers after Surgical Revascularization in Diabetic Patients with Critical Limb Ischemia. Ann. Vasc. Dis. 2008, 1, 111–117. [Google Scholar] [CrossRef] [PubMed]
- Shalan, N.; Al-Bazzaz, A.; Al-Ani, I.; Najem, F.; Al-Masri, M. Effect of Carbon Dioxide Therapy on Diabetic Foot Ulcer. J. Diabetes Mellit. 2015, 5, 284–289. [Google Scholar] [CrossRef]
- Abdulhamza, G.R.; Al-Omary, H.L. Physiological Effects of Carbon Dioxide Treatment on Diabetic Foot Ulcer Patients. IOSR J. Pharm. Biol. Sci. (IOSR-JPBS) 2018, 13, 1–7. [Google Scholar] [CrossRef]
- Guan, Y.; Huang, J.; Zuo, L.; Xu, J.; Si, L.; Qiu, J.; Li, G. Effect of pluronic P123 and F127 block copolymer on P-glycoprotein transport and CYP3A metabolism. Arch. Pharm. Res. 2011, 34, 1719–1728. [Google Scholar] [CrossRef]
- Yang, T.F.; Chen, C.N.; Chen, M.C.; Lai, C.H.; Liang, H.F.; Sung, H.W. Shell-crosslinked Pluronic L121 micelles as a drug delivery vehicle. Biomaterials 2007, 28, 725–734. [Google Scholar] [CrossRef]
- Dluska, E.; Markowska-Radomska, A.; Metera, A.; Rudniak, L.; Kosicki, K. Mass transfer of anti-cancer drug delivery to brain tumors by a multiple emulsion-based implant. AIChE J. 2022, 68, e17501. [Google Scholar] [CrossRef]
- Odziomek, M.; Ulatowski, K.; Dobrowolska, K.; Górniak, I.; Sobieszuk, P.; Sosnowski, T.R. Aqueous dispersions of oxygen nanobubbles for potential application in inhalation therapy. Sci. Rep. 2022, 12, 12455. [Google Scholar] [CrossRef]
- Ulatowski, K.; Wierzchowski, K.; Fiuk, J.; Sobieszuk, P. Effect of Nanobubble Presence on Murine Fibroblasts and Human Leukemia Cell Cultures. Langmuir 2022, 38, 8575–8584. [Google Scholar] [CrossRef]
- Bhandari, P.N.; Cui, Y.; Elzey, B.D.; Goergen, C.J.; Long, C.M.; Irudayaraj, J. Oxygen nanobubbles revert hypoxia by methylation programming. Sci. Rep. 2017, 7, 9268. [Google Scholar] [CrossRef] [PubMed]
- Gao, Y.; Hernandez, C.; Yuan, H.X.; Lilly, J.; Kota, P.; Zhou, H.; Wu, H.; Exner, A.A. Ultrasound molecular imaging of ovarian cancer with CA-125 targeted nanobubble contrast agents, Nanomedicine Nanotechnology. Biol. Med. 2017, 13, 2159–2168. [Google Scholar] [CrossRef]
- Canavese, G.; Ancona, A.; Racca, L.; Canta, M.; Dumontel, B.; Barbaresco, F.; Limongi, T.; Cauda, V. Nanoparticle-assisted ultrasound: A special focus on sonodynamic therapy against cancer. Chem. Eng. J. 2018, 340, 155–172. [Google Scholar] [CrossRef] [PubMed]
- Etchepare, R.; Oliveira, H.; Nicknig, M.; Azevedo, A.; Rubio, J. Nanobubbles: Generation using a multiphase pump, properties and features in flotation. Miner. Eng. 2017, 112, 19–26. [Google Scholar] [CrossRef]
- Grace, J.R.; Wairegi, T.; Nguyen, T.H. Shapes and velocities of single drops and bubbles moving freely through immiscible liquids. Trans. Inst. Chem. Eng. 1976, 54, 167–173. [Google Scholar]
- Azevedo, A.; Oliveira, H.; Rubio, J. Historical perspective Bulk nanobubbles in the mineral and environmental areas: Updating research and applications. Adv. Colloid Interface Sci. 2019, 271, 101992. [Google Scholar] [CrossRef]
- Zimmerman, W.B.; Tesař, V.; Bandulasena, H.C.H. Towards energy efficient nanobubble generation with fluidic oscillation. Curr. Opin. Colloid Interface Sci. 2011, 16, 350–356. [Google Scholar] [CrossRef]
- Tang, J.; Yu, S.; Sun, L.; Xie, G.; Li, X. Continuous formation of microbubbles during partial coalescence of bubbles from a submerged capillary nozzle. AIChE J. 2020, 66, e16233. [Google Scholar] [CrossRef]
- Ahmed, A.K.A.; Sun, C.; Hua, L.; Zhang, Z.; Zhang, Y.; Zhang, W.; Marhaba, T. Generation of nanobubbles by ceramic membrane filters: The dependence of bubble size and zeta potential on surface coating, pore size and injected gas pressure. Chemosphere 2018, 203, 327–335. [Google Scholar] [CrossRef]
- Khirani, S.; Kunwapanitchakul, P.; Augier, F.; Guigui, C.; Guiraud, P.; Hébrard, G. Microbubble generation through porous membrane under aqueous or organic liquid shear flow. Ind. Eng. Chem. Res. 2012, 51, 1997–2009. [Google Scholar] [CrossRef]
- Cerrón-Calle, G.A.; Magdaleno, A.L.; Graf, J.C.; Apul, O.G.; Garcia-Segura, S. Elucidating CO2 nanobubble interfacial reactivity and impacts on water chemistry. J. Colloid Interface Sci. 2022, 607, 720–728. [Google Scholar] [CrossRef] [PubMed]
- Sobieszuk, P.; Cygański, P.; Pohorecki, R. Volumetric liquid side mass transfer coefficient in a gas-liquid microreactor. Chem. Process Eng. 2008, 29, 651–661. [Google Scholar]
- Long, J.; Ding, Y.; Wang, G. Contact Problems at Micro/Nano Scale with Surface Tension. Procedia IUTAM 2017, 21, 40–47. [Google Scholar] [CrossRef]
- Cox, F.; Khalib, K.; Conlon, N. PEG That Reaction: A Case Series of Allergy to Polyethylene Glycol. J. Clin. Pharmacol. 2021, 61, 832–835. [Google Scholar] [CrossRef] [PubMed]
| Liquid | Gas | ||||
|---|---|---|---|---|---|
| ethanol | nitrogen | 789 | 0.02239 | 0.001040 | 0.035 |
| ethanol | carbon dioxide | 789 | 0.02239 | 0.001040 | 0.177 |
| water | nitrogen | 997 | 0.07199 | 0.001002 | 0.019 |
| water | carbon dioxide | 997 | 0.07199 | 0.001002 | 1.450 |
| Liquid | Gas | |||||
|---|---|---|---|---|---|---|
| ethanol | nitrogen | 350 | 108 ± 54 | 0.0412 | 4.03 × 10−9 | 0.54 |
| ethanol | carbon dioxide | 450 | 250 ± 89 | 0.0530 | 21.6 × 10−9 | 1.25 |
| water | nitrogen | 400 | 118 ± 42 | 0.0471 | 1.89 × 10−9 | 0.59 |
| water | carbon dioxide | 600 | 389 ± 40 | 0.0707 | 20.5 × 10−9 | 1.95 |
| Correlation (Equation No.) | R2 | RSSE | Δ− | Δ+ | |
|---|---|---|---|---|---|
| (7) | 0.999 | 1.58 × 10−3 [m/s] | 5.4% | 5.3% | |
| (8) | 0.590 | 9.78 × 10−9 [-] | 127% | 28.1% | |
| (9) | 0.804 | 5.41 × 10−1 [-] | 92.4% | 53.4% | |
| (10) | 0.999 | 2.27 × 10−10 [-] | 2.8% | 2.7% | |
| (11) | 0.999 | 2.24 × 10−2 [-] | 3.4% | 3.3% | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ulatowski, K.; Cecuga, A.; Sobieszuk, P. The Pursuit of Energy Reduction in Generation of Stable Nanobubbles. Processes 2023, 11, 2739. https://doi.org/10.3390/pr11092739
Ulatowski K, Cecuga A, Sobieszuk P. The Pursuit of Energy Reduction in Generation of Stable Nanobubbles. Processes. 2023; 11(9):2739. https://doi.org/10.3390/pr11092739
Chicago/Turabian StyleUlatowski, Karol, Andrzej Cecuga, and Paweł Sobieszuk. 2023. "The Pursuit of Energy Reduction in Generation of Stable Nanobubbles" Processes 11, no. 9: 2739. https://doi.org/10.3390/pr11092739
APA StyleUlatowski, K., Cecuga, A., & Sobieszuk, P. (2023). The Pursuit of Energy Reduction in Generation of Stable Nanobubbles. Processes, 11(9), 2739. https://doi.org/10.3390/pr11092739







