Single Ventricle—A Comprehensive Review
Abstract
1. Introduction
2. Cardiac Defects with Functionally Single Ventricle
2.1. Hypoplastic Left Heart Syndrome
2.2. Tricuspid Atresia
2.3. Double-Inlet Left Ventricle
2.4. Unbalanced Atrioventricular Septal Defect
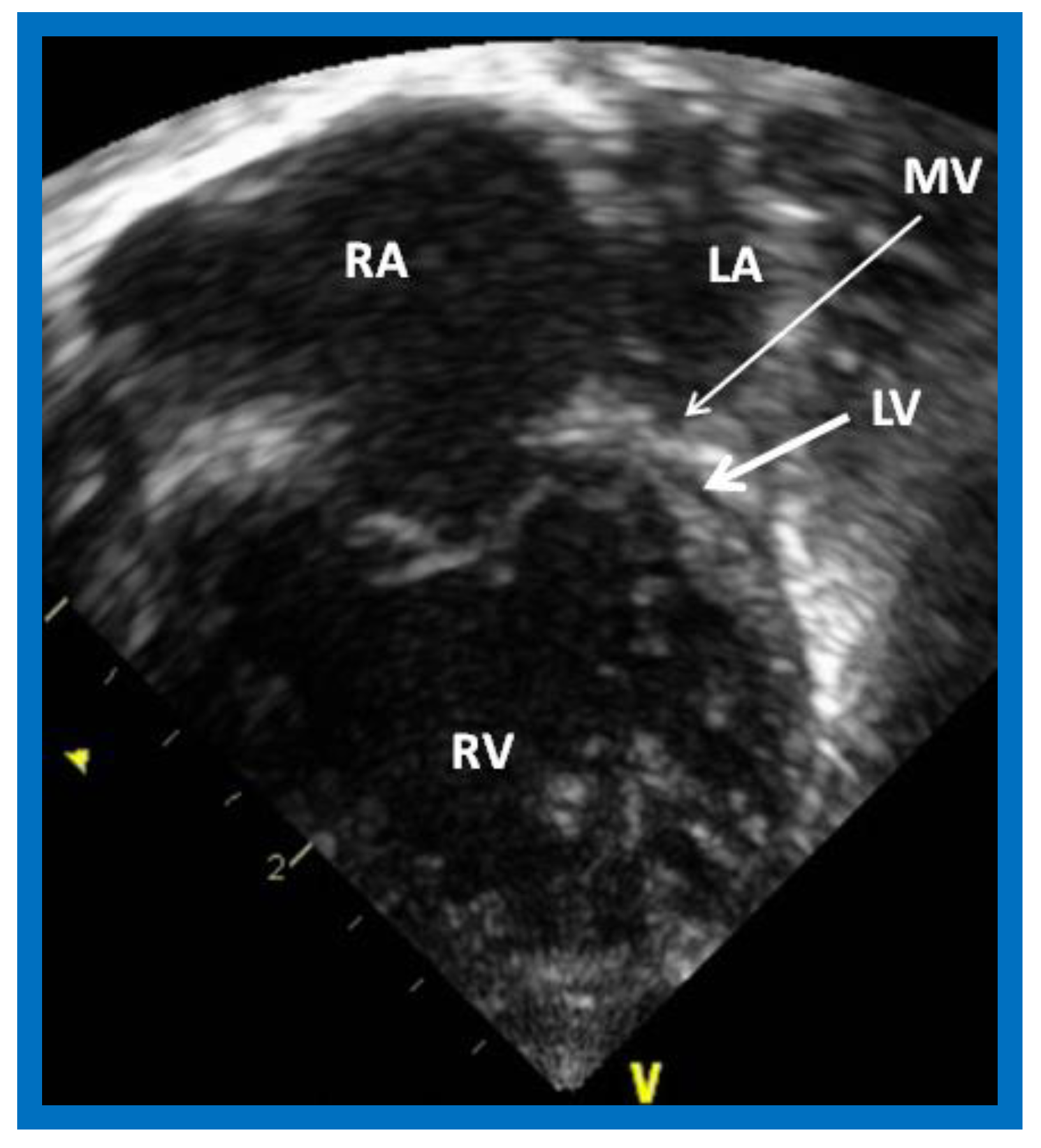
2.5. Mitral Atresia with Normal Aortic Root
2.6. Other Heart Defects with Single Ventricle Physiology
2.6.1. Pulmonary Atresia with Intact Ventricular Septum
2.6.2. Ebstein’s Anomaly of the Tricuspid Valve
2.6.3. Heterotaxy Syndromes
3. Single Ventricle Physiology
- Group 1: Unobstructed Systemic Blood Flow
- Group 2: Obstructed Systemic Blood Flow
4. Management Strategies to Address the Single Ventricle Defects
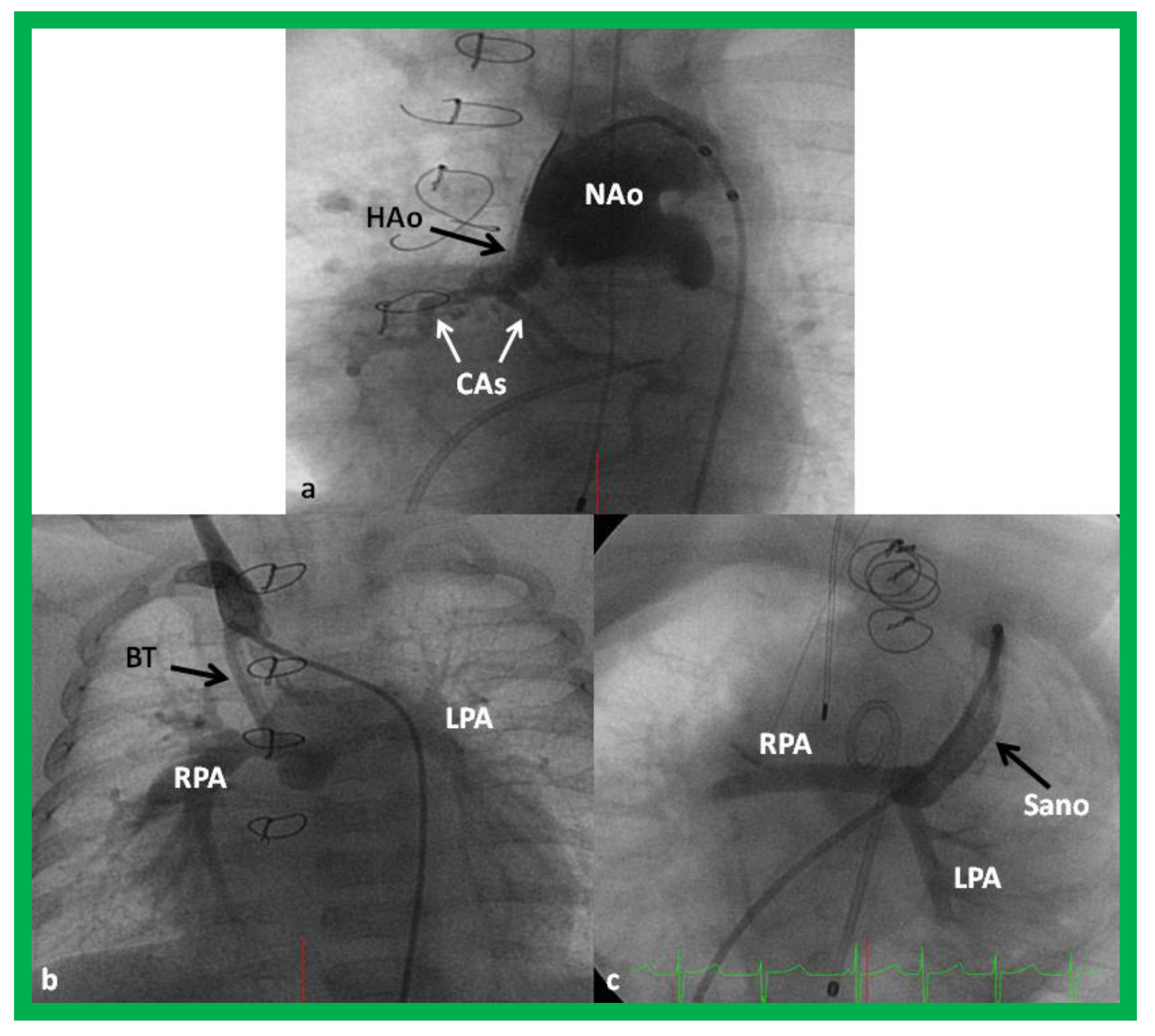
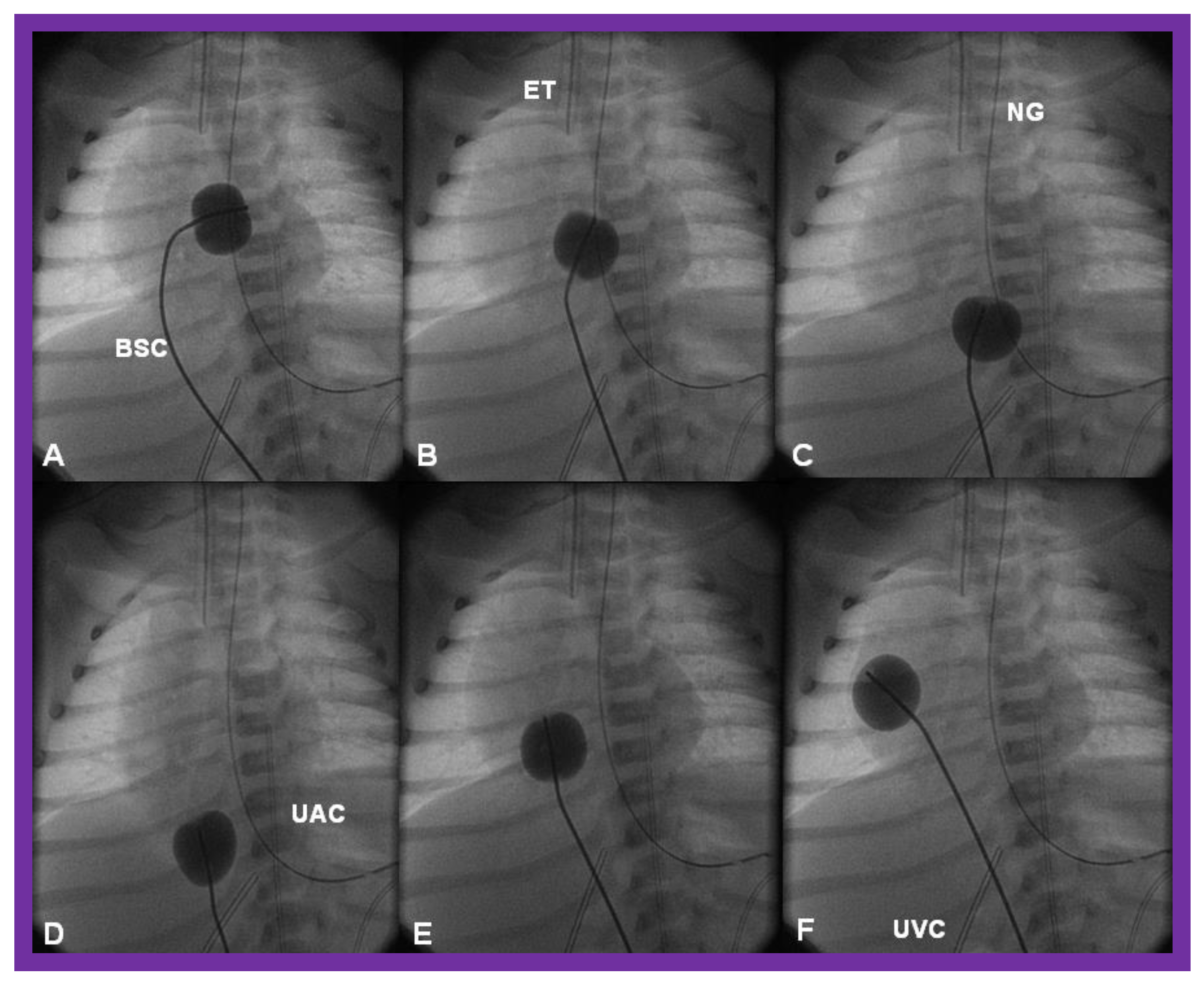
4.1. Stage I—Management at the Time of Initial Presentation
4.1.1. Status of Pulmonary Blood Flow
Reduced Pulmonary Blood Flow (Group 1C above)
Increased Pulmonary Blood Flow (Group 1A above)
Adequate Pulmonary Blood Flow (Group 1B above)
4.1.2. Obstructed Systemic Blood Flow (Group 2 above)
4.1.3. Other Cardiac Abnormalities
Intra-Cardiac Obstruction
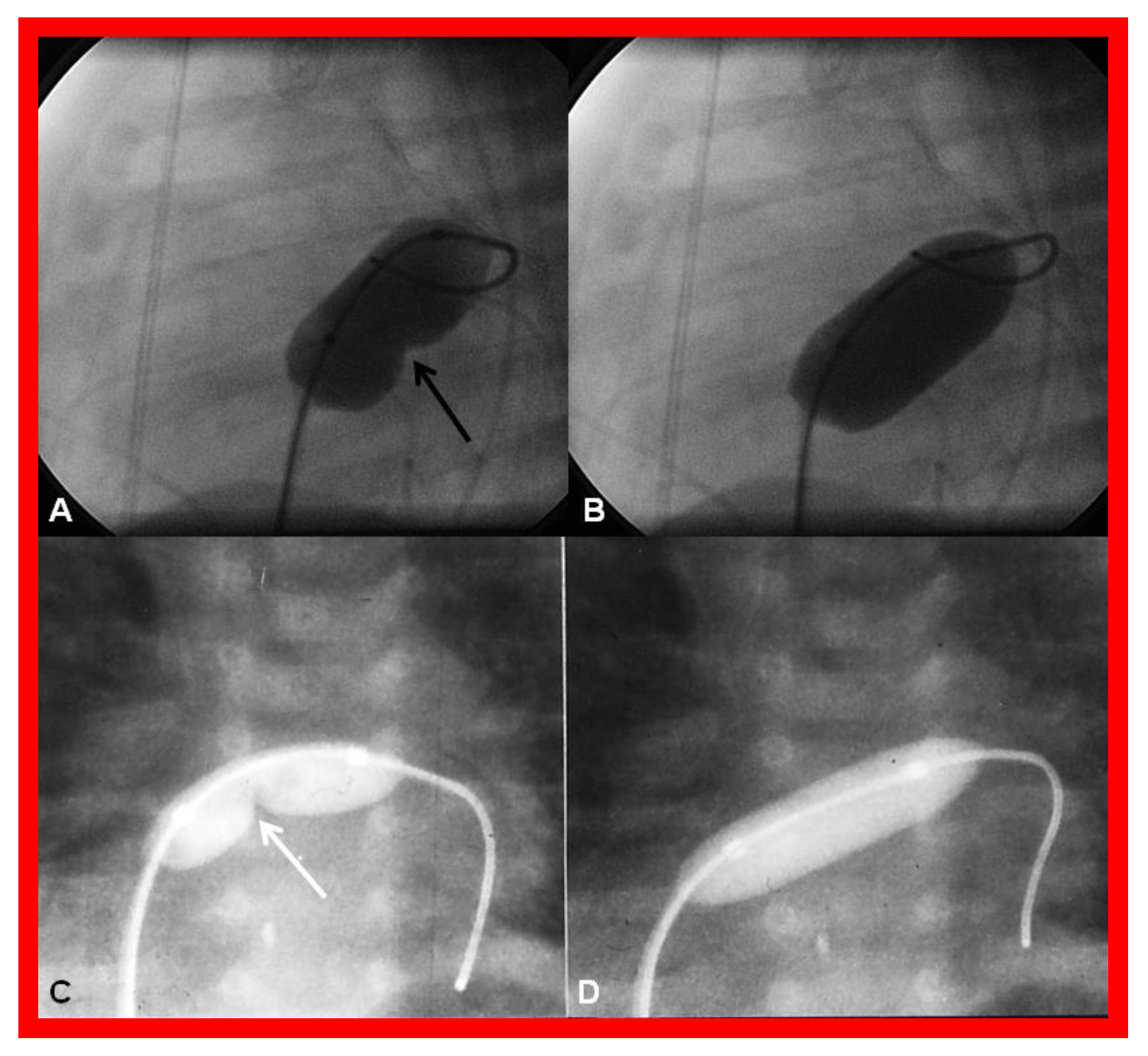
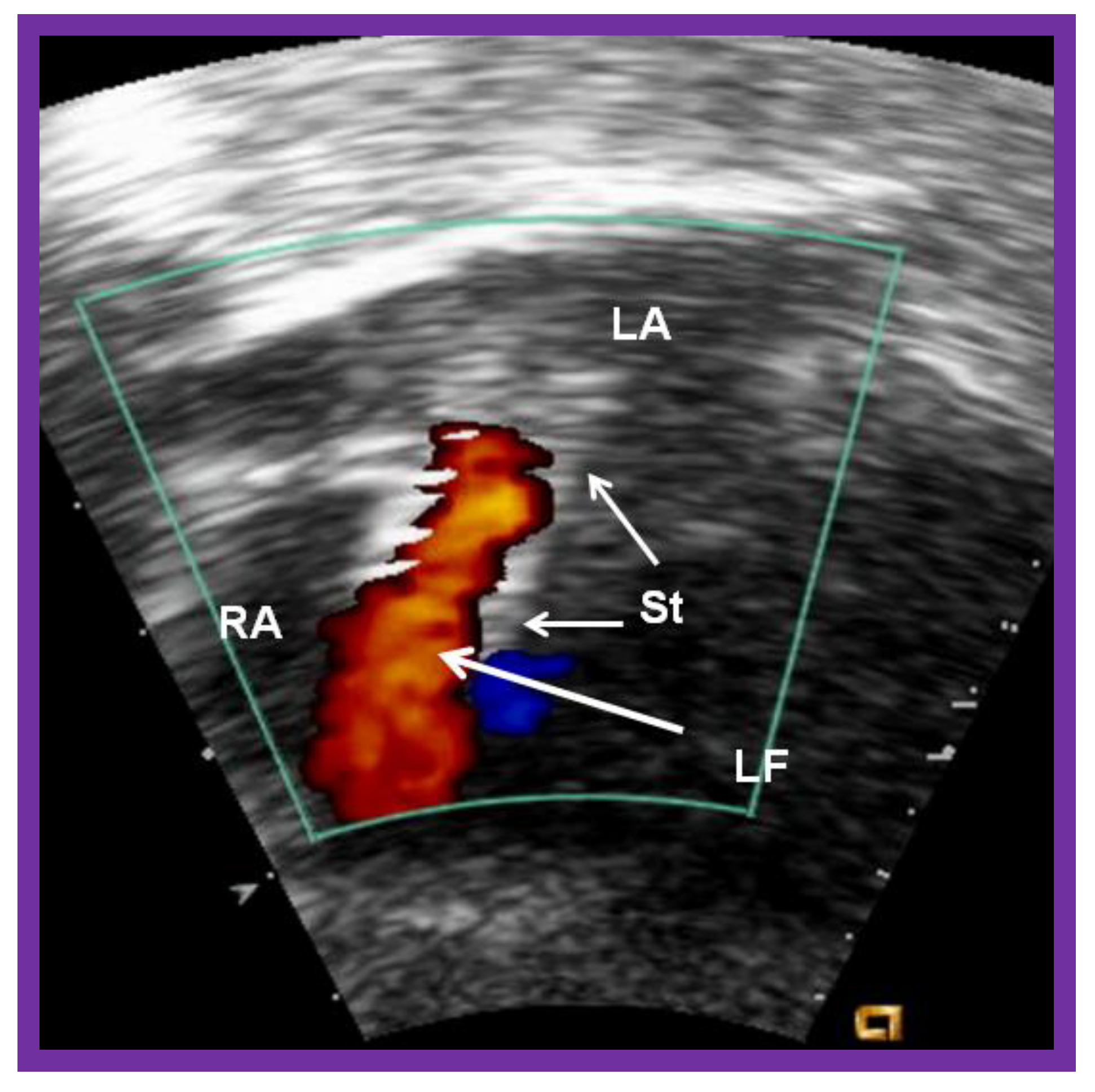
Inter-Atrial Obstruction
Inter-Ventricular Obstruction
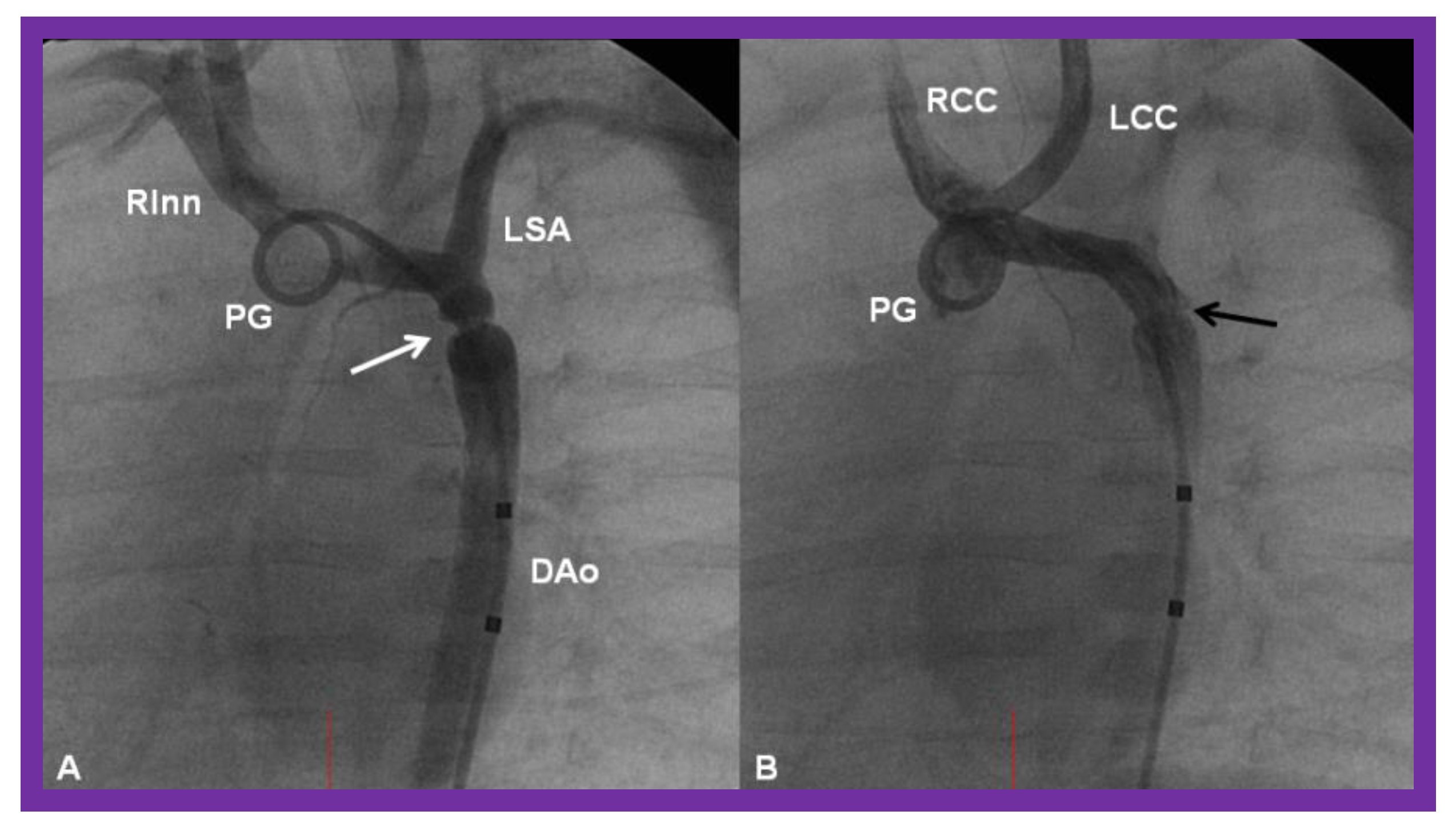
Aortic Obstruction
Aortic Coarctation
Interrupted Aortic Arch
Atrio-Ventricular Valve Insufficiency
4.2. Stage II—Bidirectional Glenn
4.3. Stage III—Fontan/Kruetzer Procedure
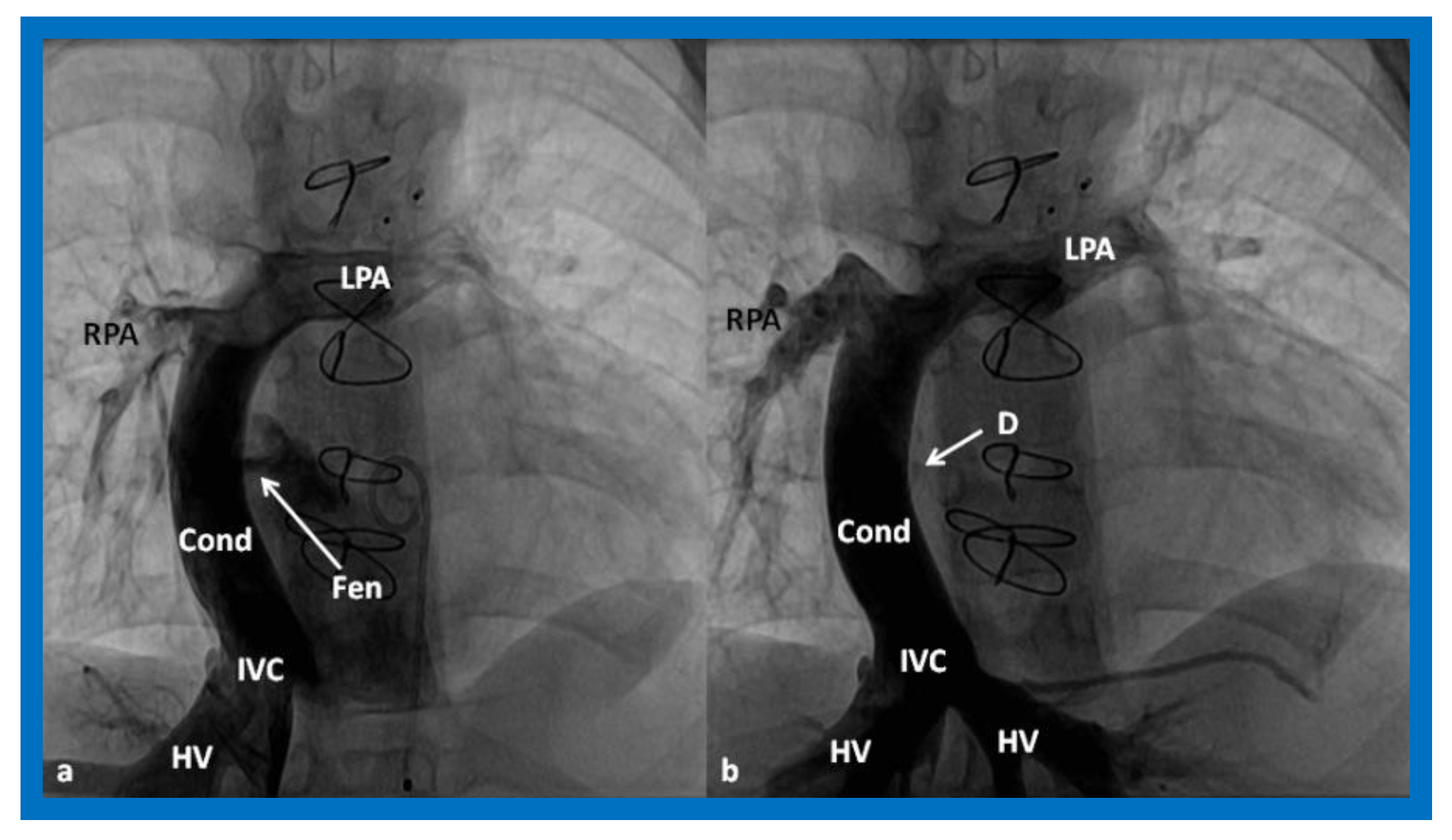
4.3.1. Stage IIIA
4.3.2. Stage IIIB
5. Inter-Stage Mortality
5.1. Restrictive Atrial Communication
5.2. Obstruction of the Aortic Arch
5.3. Distortion/Stenosis of the Pulmonary Arteries
5.4. Atrio-Ventricular Valve Insufficiency
5.5. Shunt Blockage
5.6. Inter-Current Illnesses
6. Post-Fontan Issues
7. Alternative Approaches to Fontan Circulation
7.1. One and One-Half Ventricle Repair
7.2. Primary Biventricular Repair
7.3. Staged Biventricular Repair
7.4. Results of Biventricular Repair
7.5. Conversion from Single-Ventricle to Two-Ventricle Repair
7.6. Comparison of Various Methods of Intervention
8. Other Issues
8.1. Role of Socioeconomic Status, Racial Classification, and Geographical Location in the Management of Single Ventricle Patients
8.2. Is There a Need for Centralization of Surgical Care of Complex CHD
8.3. Hybrid Approach for HLHS
8.4. Cardiac Transplantation
9. Summary and Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rao, P.S. Diagnosis and management of cyanotic congenital heart disease: Part II. Indian. J. Pediatr. 2009, 76, 297–308. [Google Scholar] [CrossRef]
- Noonan, J.A.; Nadas, A.S. The hypoplastic left heart syndrome. Pediat. Clin. N. Am 1958, 5, 1029–1056. [Google Scholar] [CrossRef]
- Rao, P.S.; Striepe, V.; Kambam, J. Hypoplastic left heart syndrome. In Cardiac Anesthesia for Infants and Children; Kambam, J., Ed.; Mosby-Year Book: St. Louis, MO, USA, 1994; pp. 296–309. [Google Scholar]
- Alapati, S.; Rao, P.S. Hypoplastic Left Heart Syndrome in the Neonate. Neonatol. Today 2011, 6, 1–9. [Google Scholar]
- Rao, P.S.; Alapati, S. Hypoplastic left heart syndrome. In A Comprehensive Approach to Management of Congenital Heart Diseases; Vijayalakshmi, I.B., Rao, P.S., Chugh, R., Eds.; Jaypee Publications: New Delhi, India, 2013; pp. 662–678. [Google Scholar]
- Rao, P.S. A unified classification for tricuspid atresia. Am. Heart J. 1980, 99, 799–804. [Google Scholar] [CrossRef]
- Rao, P.S. (Ed.) Terminology: Tricuspid atresia or univentricular heart? In Tricuspid Atresia; Futura Publishing Co.: Mount Kisco, NY, USA, 1982; pp. 3–6. [Google Scholar]
- Rao, P.S. (Ed.) Tricuspid Atresia; Futura Publishing Co.: Mount Kisco, NY, USA, 1982. [Google Scholar]
- Rao, P.S. (Ed.) Tricuspid Atresia, 2nd ed.; Futura Publishing Co.: Mount Kisco, NY, USA, 1992. [Google Scholar]
- Rao, P.S. Pediatric Tricuspid Atresia. Medscape Drugs & Diseases. Available online: http://emedicine.medscape.com/article/900832-overview (accessed on 17 January 2017).
- Rao, P.S. Tricuspid atresia. In Pediatric Cardiovascular Medicine, 2nd ed.; Moller, J.H., Hoffman, J.I.E., Eds.; Wiley-Blackwell/A John Wiley & Sons Ltd.: Hoboken, NJ, USA, 2012; pp. 487–508. [Google Scholar]
- Rao, P.S. Tricuspid atresia. In A Comprehensive Approach to Management of Congenital Heart Diseases; Vijayalakshmi, I.B., Rao, P.S., Chugh, R., Eds.; Jaypee Publications: New Delhi, India, 2013; pp. 397–413. [Google Scholar]
- Rao, P.S. Natural history of the ventricular septal defect in tricuspid atresia and its surgical implications. Br Heart J. 1977, 39, 276–288. [Google Scholar] [CrossRef]
- Rao, P.S. Further observations on the spontaneous closure of physiologically advantageous ventricular septal defects in tricuspid atresia: Surgical implications. Ann. Thorac. Surg. 1983, 35, 121–131. [Google Scholar] [CrossRef]
- Rao, P.S. Other cyanotic heart defects in the neonate. In Perinatal Cardiology: A Multidisciplinary Approach; Rao, P.S., Vidyasagar, D., Eds.; Cardiotext Publishing: Minneapolis, MN, USA, 2015. [Google Scholar]
- Titus, J.L.; Rastelli, G.C. Anatomic features of persistent common atrioventricular canal. In Artioventricular Canal Defects; Feldt, R.H., Ed.; WB Saunders Company: Philadelphia, PA, USA, 1976; pp. 13–35. [Google Scholar]
- Rao, P.S.; Harris, A.D. Recent advances in managing septal defects: Ventricular septal defects and atrioventricular septal defects. F1000 Res. 2018, 7. [Google Scholar] [CrossRef] [PubMed]
- Balaguru, D.; Rao, P.S. Mitral Atresia (With Normal Aortic Root). In A Comprehensive Approach to Management of Congenital Heart Diseases; Vijayalakshmi, I.B., Rao, P.S., Chugh, R., Eds.; Jaypee Publications: New Delhi, India, 2013; pp. 458–467. [Google Scholar]
- Norwood, W.I.; Kirklin, J.K.; Sanders, S.P. Hypoplastic left heart syndrome: Experience with palliative surgery. Am. J. Cardiol. 1980, 45, 87–91. [Google Scholar] [CrossRef]
- Norwood, W.I.; Lang, P.; Hansen, D.D. Physiologic repair of aortic atresia-hypoplastic left heart syndrome. N. Engl. J. Med. 1983, 308, 23–26. [Google Scholar] [CrossRef]
- Rao, P.S. Comprehensive management of pulmonary atresia with intact ventricular septum. Ann. Thorac. Surg. 1985, 40, 409–413. [Google Scholar] [CrossRef]
- Siblini, G.; Rao, P.S.; Singh, G.K.; Tinker, K.; Balfour, I.C. Transcatheter management of neonates with pulmonary atresia and intact ventricular septum. Cathet. Cardiovasc. Diagn. 1997, 42, 395–402. [Google Scholar] [CrossRef]
- Rao, P.S. Pulmonary atresia with intact ventricular septum. Curr. Treat. Options Cardiovasc. Med. 2002, 4, 321–336. [Google Scholar] [CrossRef] [PubMed]
- Balaguru, D.; Rao, P.S. Pulmonary Atresia with Intact Ventricular Septum. In Perinatal Cardiology: A Multidisciplinary Approach; Rao, P.S., Vidyasagar, D., Eds.; Cardiotext Publishing: Minneapolis, MN, USA, 2015. [Google Scholar]
- Rao, P.S. Tricuspid valve abnormalities other than tricuspid atresia. In Fetal and Neonatal Cardiology; Long, W.A., Ed.; W.B. Saunders: Philadelphia, PA, USA, 1990; pp. 541–550. [Google Scholar]
- Balaguru, D.; Rao, P.S. Ebstein’s anomaly of the tricuspid valve. In Perinatal Cardiology: A Multidisciplinary Approach; Rao, P.S., Vidyasagar, D., Eds.; Cardiotext Publishing: Minneapolis, MN, USA, 2015. [Google Scholar]
- Balaguru, D.; Rao, P.S. Diseases of the tricuspid valve (Ebstein’s anomaly, tricuspid stenosis and regurgitation). In A Comprehensive Approach to Management of Congenital Heart Diseases, 2nd ed.; Vijayalakshmi, I.B., Rao, P.S., Chugh, R., Eds.; Jaypee Publications: New Delhi, India, 2019; Chapter 29. [Google Scholar]
- Rao, P.S. Cardiac Malpositions including Heterotaxy Syndromes. In Perinatal Cardiology: A Multidisciplinary Approach; Rao, P.S., Vidyasagar, D., Eds.; Cardiotext Publishing: Minneapolis, MN, USA, 2015; Chapter 36. [Google Scholar]
- Rao, P.S. Cardiac Malposition. In PG Textbook of Pediatrics, 2nd ed.; Gupta, P., Menon, P.S.N., Ramji, S., Lodha, R., Eds.; Jaypee Brothers Medical Publishers (P) Ltd.: New Delhi, India, 2018. [Google Scholar]
- Yarrabolu, T.; Douglas, W.I. Single ventricle physiology. In Perinatal Cardiology: A Multidisciplinary Approach; Rao, P.S., Vidyasagar, D., Eds.; Cardiotext Publishing: Minneapolis, MN, USA, 2015. [Google Scholar]
- Chopra, P.S.; Rao, P.S. Corrective surgery for tricuspid atresia: Which modifications of Fontan-Kreutzer procedure should be used? A review. Am. Heart. J. 1992, 123, 758–767. [Google Scholar] [CrossRef]
- Rao, P.S.; Chopra, P.S. Modification of Fontan-Kreutzer procedure for tricuspid atresia: Can a choice be made? In Tricuspid Atresia, 2nd ed.; Rao, P.S., Ed.; Futura Publishing Co.: Mount Kisco, NY, USA, 1992; pp. 361–375. [Google Scholar]
- Rao, P.S. Fontan operation: Indications, short and long term outcomes. Indian J. Pediatr. 2015, 82, 1147–1156. [Google Scholar] [CrossRef]
- Rao, P.S. Fontan operation: A comprehensive review. In Fontan Surgery; Khan, I., Ed.; InTechOpen: Rijeka, Croatia, 2020. [Google Scholar] [CrossRef]
- Fontan, F.; Baudet, E. Surgical repair of tricuspid atresia. Thorax 1971, 26, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Kreutzer, G.; Bono, H.; Galindez, E. Una operacion para la correccion de la atresia tricuspidea. In Proceedings of the Ninth Argentinean Congress of Cardiology, Buenos Aires, Argentina, 31 October–6 November 1971. [Google Scholar]
- De Leval, M.R.; Kilner, P.; Gewillig, M.; Bull, C.; McGoon, D.C. Total cavopulmonary connection: A logical alternative to atriopulmonary connection for complex Fontan operation. J. Thorac. Cardiovasc. Surg. 1988, 96, 682–695. [Google Scholar] [CrossRef]
- Rao, P.S.; Covitz, W.; Chopra, P.S. Principles of palliative management of patients with tricuspid atresia. In Tricuspid Atresia, 2nd ed.; Rao, P.S., Ed.; Futura Publishing Co.: Mt. Kisco, NY, USA, 1992; pp. 297–320. [Google Scholar]
- De Leval, M.; McKay, R.; Jones, M.; Stark, J.; McCartney, F.J. Modified Blalock-Taussig shunt: Use of subclavian orifice as a flow regulator in prosthetic systemic-pulmonary artery shunts. J. Thorac. Cardiovasc. Surg. 1981, 18, 112–119. [Google Scholar] [CrossRef]
- Gibbs, J.L.; Orhan, U.; Blackburn, M.E.C.; Wren, C.; Hamilton, J.R.; Watterson, K.G. Fate of stented arterial duct. Circulation 1999, 99, 2621–2625. [Google Scholar] [CrossRef]
- Alwi, M.; Choo, K.K.; Latiff, H.A.; Kandavello, G.; Hasri Samion, H.; Mulyadi, M.D. Initial results and medium-term follow-up of stent implantation of patent ductus arteriosus in duct-dependent pulmonary circulation. J. Amer. Coll. Cardiol. 2004, 44, 438–445. [Google Scholar] [CrossRef]
- Rao, P.S.; Brais, M. Balloon pulmonary valvuloplasty for congenital cyanotic heart defects. Amer. Heart. J. 1988, 115, 1105–1110. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.S.; Wilson, A.D.; Thapar, M.K.; Brais, M. Balloon pulmonary valvuloplasty in the management of cyanotic congenital heart defects. Cathet. Cardiovasc. Diagn. 1992, 25, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.S. Pulmonary valve disease: Pulmonary valve in cyanotic heart defects with pulmonary oligemia. In Interventions in Structural, Valvular and Congenital Heart Disease; Sievert, H., Qureshi, S.A., Wilson, N., Hijazi, Z., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 297–308. [Google Scholar]
- Alapati, S.; Rao, P.S. Tetralogy of Fallot. In A Multidisciplinary Approach to Perinatal Cardiology; Rao, P.S., Vidyasagar, D., Eds.; Cambridge Scholars Publishing: New Castle Upon Tyne, UK, 2021; Volume 2, pp. 203–246. [Google Scholar]
- Rao, P.S. Congenital heart disease. In Conn’s Current Therapy; Rakel, R.E., Ed.; WB Saunders Co: Philadelphia, PA, USA, 1989; pp. 201–213. [Google Scholar]
- Muller, W.H., Jr.; Danimann, J.F., Jr. The treatment of certain congenital malformations of the heart by the creation of pulmonic stenosis to reduce pulmonary hypertension and excessive pulmonary blood flow; a preliminary report. Surg. Gynec. Obst. 1952, 95, 213–219. [Google Scholar]
- Sano, S.; Ishino, K.; Kawada, M.; Arai, S.; Kasahara, S.; Asai, T.; Masuda, Z.; Takeuchi, M.; Ohtsuki, S. Right ventricle-pulmonary artery shunt in first-stage palliation of hypoplastic left heart syndrome. J. Thorac. Cardiovasc. Surg. 2003, 126, 504–509. [Google Scholar] [CrossRef]
- Sano, S.; Ishino, K.; Kado, H.; Shiokawa, Y.; Sakamoto, K.; Yokota, M.; Kawada, M. Outcome of right ventricle-to-pulmonary artery shunt in first-stage palliation of hypoplastic left heart syndrome: A multi-institutional study. Ann. Thorac. Surg. 2004, 78, 1951–1957, discussion 1957–1958. [Google Scholar] [CrossRef] [PubMed]
- Bailey, L.; Concepcion, W.; Shattuk, H.; Huang, L. Method of heart transplantation for treatment of hypoplastic left heart syndrome. J. Thorac. Cardiovasc. Surg. 1986, 92, 1–9. [Google Scholar] [CrossRef]
- Rao, P.S. Principles of management of the neonate with congenital heart disease. Neonatol. Today 2007, 2, 1–10. [Google Scholar]
- Rashkind, W.J.; Miller, W.W. Creation of an atrial septal defect without thoracotomy. J. Am. Med. Assoc. 1966, 196, 991–992. [Google Scholar] [CrossRef]
- Park, S.C.; Neches, W.H.; Zuberbuhler, J.R.; Lenox, C.C.; Mathews, R.A.; Fricker, F.J.; Zoltun, R.A. Clinical use of blade septostomy. Circulation 1978, 58, 600–606. [Google Scholar] [CrossRef]
- Shrivatsava, S.; Radhakrishnan, S.; Dev, V.; Singh, L.S.; Rajani, M. Balloon dilatation of atrial septum in complete transposition of great arteries—A new technique. Indian Heart J. 1987, 39, 298–300. [Google Scholar]
- Rao, P.S. Static balloon dilation of atrial septum (Editorial). Am. Heart. J. 1993, 125, 1826–1827. [Google Scholar]
- Gewillig, D.; Boshoff, L.; Mertens, L. Creation with a stent of an unrestrictive lasting atrial communication. Cardiol. Young 2002, 12, 404–407. [Google Scholar] [CrossRef] [PubMed]
- Eicken, A.; Gildein, H.P.; Schreiber, C.; Balling, G.; Hess, J. Stenting of a restrictive foramen ovale in a patient with hypoplastic left heart syndrome. Intern. J. Cardiol. 2006, 113, 254–256. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.S. Neonatal Catheter Interventions. In Cardiac Catheterization and Imaging (From Pediatrics to Geriatrics); Vijayalakshmi, I.B., Ed.; Jaypee Publications: New Delhi, India, 2015; pp. 388–432. [Google Scholar]
- Atz, A.M.; Feinstein, J.A.; Jonas, R.A.; Perry, S.B.; Wessel, D.L. Preoperative management of pulmonary venous hypertension in hypoplastic left heart syndrome with restrictive atrial septal defect. Am. J. Cardiol. 1999, 83, 1224–1228. [Google Scholar] [CrossRef]
- Justino, H.; Benson, L.N.; Nykanen, D.G. Transcatheter creation of an atrial septal defect using radiofrequency perforation. Catheter. Cardiovasc. Interv. 2001, 54, 83–87. [Google Scholar] [CrossRef]
- Rao, P.S. Catheter interventions in the neonate. In A Multidisciplinary Approach to Perinatal Cardiology; Rao, P.S., Vidyasagar, D., Eds.; Cambridge Scholars Publishing: New Castle Upon Tyne, UK, 2021; Volume 1, pp. 646–742. [Google Scholar]
- Rao, P.S.; Moore, H.V.; Strong, W.B. Surgery for mitral atresia and interatrial obstruction without pulmonic stenosis (Letter). Circulation 1980, 62, 201–202. [Google Scholar]
- Rao, P.S.; Kulangara, R.J.; Moore, H.V.; Strong, W.B. Syndrome of single ventricle without pulmonic stenosis but with left atrioventricular valve atresia and interatrial obstruction: Palliative management with simultaneous atrial septostomy and pulmonary artery banding. J. Thorac. Cardiovasc. Surg. 1981, 81, 127–130. [Google Scholar] [CrossRef]
- Rao, P.S. Physiologically advantageous ventricular septal defects (Letter). Pediatr. Cardiol. 1983, 4, 59–62. [Google Scholar] [CrossRef]
- Rao, P.S. Subaortic obstruction after pulmonary artery banding in patients with tricuspid atresia and double-inlet left ventricle and ventriculoarterial discordance (Letter). J. Am. Coll. Cardiol. 1991, 18, 1885–1886. [Google Scholar] [CrossRef]
- Stansel, H.C., Jr. A new operation for d- transposition of the great vessels. Ann. Thorac. Surg. 1975, 19, 565–567. [Google Scholar] [CrossRef]
- Rao, P.S. Coarctation of the aorta. In Secondary Forms of Hypertension; Seminars in Nephrology; Kurtzman, N.A., Ram, C.V.S., Eds.; W.B. Saunders: Philadelphia, PA, USA, 1995; Volume 15, pp. 81–105. [Google Scholar]
- Rao, P.S.; Thapar, M.K.; Galal, O.; Wilson, A.D. Follow-up results of balloon angioplasty of native coarctation in neonates and infants. Am. Heart. J. 1990, 120, 1310–1319. [Google Scholar] [CrossRef]
- Rao, P.S. Current status of balloon angioplasty for neonatal and infant aortic coarctation. Prog. Pediat. Cardiol. 2001, 14, 35–44. [Google Scholar]
- Rao, P.S.; Jureidini, S.B.; Balfour, I.C.; Singh, G.K.; Chen, S.C. Severe aortic coarctation in infants less than 3 months: Successful palliation by balloon angioplasty. J. Interv. Cardiol. 2003, 15, 203–208. [Google Scholar]
- Reddington, A.N.; Booth, P.; Shore, D.F.; Rigby, M.L. Primary balloon dilatation of coarctation in neonates. Br Heart J. 1990, 64, 277–281. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.S.; Galal, O.; Smith, P.A.; Wilson, A.D. Five-to-nine-year follow-up results of balloon angioplasty of native aortic coarctation in infants and children. J. Am. Coll. Cardiol. 1996, 27, 462–470. [Google Scholar] [CrossRef]
- Rao, P.S. Should balloon angioplasty be used as a treatment of choice for native aortic coarctations? J. Invasive Cardiol. 1996, 8, 301–308. [Google Scholar] [PubMed]
- Sharma, S.K.; Rao, P.S. Interrupted aortic arch. In The Encyclopedic Reference of Molecular Mechanisms of Disease; Springer: Berlin/Heidelberg, Germany; New York, NY, USA; Tokyo, Japan, 2008. [Google Scholar]
- Celoria, G.C.; Patton, R.B. Congenital absence of the aortic arch. Am. Heart. J. 1959, 58, 407–413. [Google Scholar] [CrossRef]
- Hopkins, R.A.; Armstrong, S.E.; Serwer, G.A.; Peterson, R.J.; Oldham, H.N., Jr. Physiologic rationale for a bidirectional cavopulmonary shunt: A versatile complement to the Fontan principle. J. Thorac. Cardiovasc. Surg. 1985, 90, 391–398. [Google Scholar] [CrossRef]
- Puga, F.J.; Chiavarelli, M.; Hagler, D.J. Modifications of the Fontan operation applicable to patients with left atrioventricular valve atresia or single atrioventricular valve. Circulation 1987, 76 (Suppl. III), III53–III60. [Google Scholar] [PubMed]
- Jonas, R.A.; Castaneda, A.R. Modified Fontan procedure: Atrial baffle and systemic venous to pulmonary artery anastomotic techniques. J. Cardiac Surg. 1988, 3, 91–96. [Google Scholar] [CrossRef]
- Marcelletti, C.; Corno, A.; Giannico, S.; Marino, B. Inferior vena cava-pulmonary artery extra-cardiac conduit. A new form of right heart bypass. J. Thorac. Cardiovasc. Surg. 1990, 100, 228–232. [Google Scholar] [CrossRef]
- Marcelletti, C.; Iorio, F.S.; Abella, R.F. Late results of extra-cardiac Fontan repair. Semin. Thorac. Cardiovasc. Surg. Pediatr. Card. Surg. Annu. 1999, 2, 131–141. [Google Scholar] [CrossRef]
- Bridges, N.D.; Lock, J.E.; Castaneda, A.R. Baffle fenestration with subsequent transcatheter closure. Modification of the Fontan operation for patients at increased risk. Circulation 1990, 82, 1681–1689. [Google Scholar] [CrossRef] [PubMed]
- Laks, H.; Pearl, J.M.; Haas, G.S.; Drinkwater, D.C.; Milgalter, E.; Jarmakani, J.M.; Isabel-Jones, J.; George, B.L.; Williams, R.G. Partial Fontan: Advantages of an adjustable interatrial communication. Ann. Thorac. Surg. 1991, 52, 1084–1094, discussion 1094–1095. [Google Scholar] [CrossRef]
- Rao, P.S.; Chandar, J.S.; Sideris, E.B. Role of inverted buttoned device in transcatheter occlusion of atrial septal defects or patent foramen ovale with right-to-left shunting associated with previously operated complex congenital cardiac anomalies. Am. J. Cardiol. 1997, 80, 914–921. [Google Scholar] [CrossRef]
- Goff, D.A.; Blume, E.D.; Gauvreau, K.; Mayer, J.E.; Lock, J.E.; Jenkins, K.J. Clinical Advances in Complex Valvular Disease 24 outcome of fenestrated Fontan patients after closure: The first 10 years. Circulation 2000, 102, 2094–2099. [Google Scholar] [CrossRef] [PubMed]
- Boudjemline, Y.; Bonnet, D.; Sidi, D.; Agnoletti, G. Closure of extracardiac Fontan fenestration by using the Amplatzer duct occluder. Arch. des Mal. du Coeur et des Vaiss. 2005, 98, 449–454. [Google Scholar]
- Tweddell, J.S.; Hoffman, G.M.; Mussatto, K.A.; Fedderly, R.T.; Berger, S.; Jaquiss, R.D.; Ghanayem, N.S.; Frisbee, S.J.; Litwin, S.B. Improved survival of patients undergoing palliation of hypoplastic left heart syndrome: Lessons learned from 115 consecutive patients. Circulation 2002, 106, 82–89. [Google Scholar] [CrossRef]
- Yates, M.C.; Rao, P.S. Pediatric cardiac emergencies. Emerg. Med. 2013, 3, 164. [Google Scholar] [CrossRef]
- Bartram, U.; Grunenfelder, J.; Van Praagh, R. Causes of death after the modified Norwood procedure: A study of 122 postmortem cases. Ann. Thorac. Surg. 1997, 64, 1795–1802. [Google Scholar] [CrossRef]
- Rao, P.S. The role of interventional cardiology in neonate. In Pediatric Cardiology: How It Has Evolved over the Last 50 Years; Rao, P.S., Ed.; Cambridge Scholars Publishing: New Castle upon Tyne, UK, 2020; pp. 560–603. [Google Scholar]
- Rao, P.S. Role of interventional cardiology in neonates: Part II—Balloon angioplasty/valvuloplasty. Congenital. Cardiol. Today 2008, 6, 1–14. [Google Scholar]
- Rao, P.S.; Wilson, A.D.; Chopra, P.S. Immediate and follow-up results of balloon angioplasty of postoperative recoarctation in infants and children. Am. Heart J. 1990, 120, 1315–1320. [Google Scholar] [CrossRef]
- Rao, P.S.; Chopra, P.S. Role of balloon angioplasty in the treatment of aortic coarctation. Ann. Thorac. Surg. 1991, 52, 621–631. [Google Scholar] [CrossRef]
- Rao, P.S. (Ed.) Balloon angioplasty for aortic recoarctation following previous surgery. In Trans-Catheter Therapy in Pediatric Cardiology; Wiley-Liss: New York, NY, USA, 1993; pp. 197–212. [Google Scholar]
- Siblini, G.; Rao, P.S.; Nouri, S.; Ferdman, B.; Jureidini, S.B.; Wilson, A.D. Long-term follow-up results of balloon angioplasty of postoperative aortic recoarctation. Am. J. Cardiol. 1998, 81, 61–67. [Google Scholar] [CrossRef]
- Rao, P.S.; Balfour, I.C.; Singh, G.K.; Jureidini, S.B.; Chen, S. Bridge stents in the management of obstructive vascular lesions in children. Am. J. Cardiol. 2001, 88, 699–702. [Google Scholar] [CrossRef]
- Rao, P.S. Stents in the management of congenital heart disease in the pediatric and adult patients. Indian Heart J. 2001, 53, 714–730. [Google Scholar] [PubMed]
- Sahu, R.; Rao, P.S. Transcatheter stent therapy in children: An update. Pediatrics Ther. 2012, S5. [Google Scholar] [CrossRef]
- Rao, P.S. Stents in the Management of Vascular Obstructive Lesions Associated with Congenital Heart Disease. In Cardiac Catheterization and Imaging (From Pediatrics to Geriatrics); Vijayalakshmi, I.B., Ed.; Jaypee Publications: New Delhi, India, 2015; pp. 573–598. [Google Scholar]
- Rao, P.S. Stents. In Pediatric Cardiology: How It Has Evolved Over The Last 50 Years; Rao, P.S., Ed.; Cambridge Scholars Publishing: New Castle upon Tyne, UK, 2020; pp. 511–559. ISBN 9781527548886. [Google Scholar]
- Tsounias, E.; Rao, P.S. Stent therapy for clotted Blalock-Taussig shunts. Congenit. Cardiol. Today 2010, 8, 1–9. [Google Scholar]
- Schaff, H.V.; Danielson, G.K. Corrective surgery for tricuspid atresia. In Tricuspid Atresia; Rao, P.S., Ed.; Futura Publishing Co.: Mount Kisco, NY, USA, 1982; pp. 275–292. [Google Scholar]
- Freedom, R.M.; Gow, R.; Caspi, J. The Fontan procedure for patients with tricuspid atresia: Longterm follow-up. In Tricuspid Atresia, 2nd ed.; Rao, P.S., Ed.; Futura Publishing Co.: Mt. Kisco, NY, USA, 1992; pp. 377–386. [Google Scholar]
- Kaulitz, R.; Ziemer, G.; Luhmer, I.; Kallfelz, H. Modified Fontan operation in functionally univentricular hearts: Preoperative risk factors and intermediate results. J. Thorac. Cardiovasc. Surg. 1996, 112, 658–664. [Google Scholar] [CrossRef]
- Mosca, R.S.; Kulik, T.J.; Goldberg, C.S.; Vermilion, R.P.; Charpie, J.R.; Crowley, D.C.; Bove, E.L. Early results of the Fontan procedure in one hundred consecutive patients with hypoplastic left heart syndrome. J. Thorac. Cardiovasc. Surg. 2000, 199, 1110–1118. [Google Scholar] [CrossRef]
- Yoshimura, N.; Yamaguchi, M.; Oshima, Y.; Oka, S.; Ootaki, Y.; Tei, T.; Kido, S. Risk factors influencing early and late mortality after total cavopulmonary connection. Eur. J Cardio-Thorac. Surg. 2001, 20, 598–602. [Google Scholar] [CrossRef][Green Version]
- Jacobs, M.L.; Norwood, W.I., Jr. Fontan operation: Influence of modifications on morbidity and mortality. Ann. Thorac. Surg. 1994, 58, 945–952. [Google Scholar] [CrossRef]
- Gentles, T.L.; Mayer, J.E., Jr.; Gauvreau, K.; Newburger, J.W.; Lock, J.E.; Kupferschmid, J.P.; Burnett, J.; Jonas, R.A.; Castañeda, A.R.; Wernovsky, G. Fontan operation in five hundred consecutive patients: Factors influencing early and late outcome. J. Thorac. Cardiovasc. Surg. 1997, 114, 376–391. [Google Scholar] [CrossRef]
- Gaynor, J.W.; Bridges, N.D.; Cohen, M.I.; Mahle, W.T.; Decampli, W.M.; Steven, J.M.; Nicolson, S.C.; Spray, T.L. Predictors of outcome after the Fontan operation: Is hypoplastic left heart syndrome still a risk factor? J Thorac Cardiovasc. Surg. 2002, 123, 237–245. [Google Scholar] [CrossRef] [PubMed]
- Rao, P.S. Protein-losing enteropathy following the Fontan operation (editorial). J. Invasive Cardiol. 2007, 19, 447–448. [Google Scholar]
- Alsaied, T.; Bokma, J.P.; Engel, M.E.; Kuijpers, J.M.; Hanke, S.P.; Zuhlke, L.; Zhang, B.; Veldtman, G.R. Factors associated with long-term mortality after Fontan procedures: A systematic review. Heart 2017, 103, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Poh, C.L.; d’Udekem, Y. Life after surviving Fontan surgery: A meta-analysis of the incidence and predictors of late death. Heart Lung Circ. 2018, 27, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Kverneland, L.S.; Kramer, P.; Ovroutski, S. Five decades of the Fontan operation: A systematic review of international reports on outcomes after univentricular palliation. Congenit. Heart Dis. 2018, 13, 181–193. [Google Scholar] [CrossRef]
- Wada, N.; Takahashi, Y.; Ando, M.; Park, I.S.; Sasaki, T. Single ventricle repair in children with Down’s syndrome. Gen. Thorac. Cardiovasc. Surg. 2008, 56, 104–108. [Google Scholar] [CrossRef] [PubMed]
- Bartz, P.J.; Driscoll, D.J.; Dearani, J.A.; Puga, F.J.; Danielson, G.K.; O’Leary, P.W.; Earing, M.G.; Warnes, C.A.; Hodge, D.O.; Cetta, F. Early and Late Results of the Modified Fontan Operation for Heterotaxy Syndrome. 30 Years of Experience in 142 Patients. J. Am. Coll. Cardiol. 2006, 48, 2301–2305. [Google Scholar] [CrossRef] [PubMed]
- Gupta-Malhotra, M.; Larson, V.E.V.; Rosengart, R.M.; Guo, H.; Moller, J.H. Mortality after total cavopulmonary connection in children with the Down syndrome. Am. J. Cardiol. 2010, 105, 865–868. [Google Scholar] [CrossRef]
- Schulz, A.; Baird, C.; Emani, S.; Nathan, M. Insights into biventricular repair and biventricular conversion of hearts with hypoplastic left heart structures. In A Multidisciplinary Approach to Perinatal Cardiology; Rao, P.S., Vidyasagar, D., Eds.; Cambridge Scholars Publishing: New Castle upon Tyne, UK, 2021; Volume 2, Chapter 25. [Google Scholar]
- Nathan, M.; Emani, S.; IJsselhof, R.; Liu, H.; Gauvreau, K.; Del Nido, P. Mid-term outcomes in unbalanced complete atrioventricular septal defect: Role of biventricular conversion from single-ventricle palliation. Eur. J. Cardiothorac. Surg. 2017, 52, 565–572. [Google Scholar] [CrossRef]
- Foker, J.E.; Berry, J.M.; Harvey, B.A.; Pyles, L.A. Mitral and tricuspid valve repair and growth in unbalanced atrial ventricular canal defects. J. Thorac. Cardiovasc. Surg. 2012, 143 (Suppl. 4), S29–S32. [Google Scholar] [CrossRef]
- Jegatheeswaran, A.; Pizarro, C.; Caldarone, C.A.; Cohen, M.S.; Baffa, J.M.; Gremmels, D.B.; Mertens, L.; Morell, V.O.; Williams, W.G.; Blackstone, E.H.; et al. Echocardiographic definition and surgical decision-making in unbalanced atrioventricular septal defect: A Congenital Heart Surgeons’ Society multi-institutional study. Circulation 2010, 22 (Suppl. 11), S209–S215. [Google Scholar] [CrossRef]
- Delmo Walter, E.M.; Ewert, P.; Hetzer, R.; Hübler, M.; Alexi-Meskishvili, V.; Lange, P.; Berger, F. Biventricular repair in children with complete atrioventricular septal defect and a small left ventricle. Eur. J. Cardiothorac. Surg. 2008, 33, 40–47. [Google Scholar] [CrossRef]
- Nathan, M.; Liu, H.; Pigula, F.A.; Fynn-Thompson, F.; Emani, S.; Baird, C.A.; Marx, G.; Mayer, J.E.; Del Nido, P.J. Biventricular conversion after single-ventricle palliation in unbalanced atrioventricular canal defects. Ann. Thorac. Surg. 2013, 95, 2086–2095, discussion 2095–2096. [Google Scholar] [CrossRef]
- Emani, S.M.; McElhinney, D.B.; Tworetzky, W.; Myers, P..; Schroeder, B.; Zurakowski, D.; Pigula, F.A.; Marx, G.R.; Lock, J.E.; del Nido, P.J. Staged left ventricular recruitment after single-ventricle palliation in patients with borderline left heart hypoplasia. J. Am. Coll. Cardiol. 2012, 60, 1966–1974. [Google Scholar] [CrossRef]
- Kalish, B.T.; Banka, P.; Lafranchi, T.; Tworetzky, W.; Del Nido, P.; Emani, S.M. Biventricular conversion after single ventricle palliation in patients with small left heart structures: Short-term outcomes. Ann. Thorac. Surg. 2013, 96, 1406–1412. [Google Scholar] [CrossRef] [PubMed]
- Buratto, E.; Ye, X.T.; King, G.; Shi, W.Y.; Weintraub, R.G.; d’Udekem, Y.; Brizard, C.P.; Konstantinov, I.E. Long-term outcomes of single-ventricle palliation for unbalanced atrioventricular septal defects: Fontan survivors do better than previously thought. J. Thorac. Cardiovasc. Surg. 2017, 153, 430–438. [Google Scholar] [CrossRef] [PubMed]
- Nasser, S.A.; Senatore, F.F. Contemporary concepts in access to healthcare: Identification and elimination of disparities in care of minority patients. Prog. Cardiovasc. Dis. 2020, 63, 2–3. [Google Scholar] [CrossRef]
- Akintuerk, H.; Michel-Behnke, I.; Valeske, K.; Mueller, M.; Thul, J.; Bauer, J.; Hagel, K.; Kreuder, J.; Vogt, P.; Schranz, D. Stenting of the arterial duct and banding of the pulmonary arteries: Basis for combined Norwood stage I and II repair in hypoplastic left heart. Circulation 2002, 105, 1099–1103. [Google Scholar] [CrossRef] [PubMed]
- Galantowicz, M.; Cheatham, J.P. Lessons learned from the development of a new hybrid strategy for the management of hypoplastic left heart syndrome. Pediatr. Cardiol. 2005, 26, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Photiadis, J.; Sinzobahamvya, N.; Hraška, V.; Asfour, B. Does bilateral pulmonary banding in comparison to Norwood procedure improve outcome in neonates with hypoplastic left heart syndrome beyond second-stage palliation? A review of the current literature. J. Thorac. Cardiovasc. Surg. 2012, 60, 181–188. [Google Scholar] [CrossRef] [PubMed]
- Hsia, T.Y.; Cosentino, D.; Corsini, C.; Pennati, G.; Dubini, G.; Migliavacca, F. Modeling of Congenital Hearts Alliance (MOCHA) Investigators. Use of mathematical modeling to compare and predict hemodynamic effects between hybrid and surgical Norwood palliations for hypoplastic left heart syndrome. Circulation 2011, 124 (Suppl. 11), S204–S210. [Google Scholar] [CrossRef] [PubMed]
























| 1 | Pulmonary atresia |
| 2 | Tricuspid atresia |
| 3 | Mitral atresia |
| 4 | Hypoplastic left heart syndrome |
| 1 | Tricuspid atresia |
| 2 | Double-inlet left ventricle |
| 3 | Double-outlet right (or left) ventricle |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rao, P.S. Single Ventricle—A Comprehensive Review. Children 2021, 8, 441. https://doi.org/10.3390/children8060441
Rao PS. Single Ventricle—A Comprehensive Review. Children. 2021; 8(6):441. https://doi.org/10.3390/children8060441
Chicago/Turabian StyleRao, P. Syamasundar. 2021. "Single Ventricle—A Comprehensive Review" Children 8, no. 6: 441. https://doi.org/10.3390/children8060441
APA StyleRao, P. S. (2021). Single Ventricle—A Comprehensive Review. Children, 8(6), 441. https://doi.org/10.3390/children8060441






