A New Proximal Femur Reconstruction Technique after Bone Tumor Resection in a Very Small Patient: An Exemplificative Case
Abstract
1. Background
- (1)
- Expandable prosthesis: this can be used only in older children where the residual bone is sufficiently long for a prosthesis with an elongation system to be inserted. Expandable prostheses are potentially able to compensate for the bone shortening after tumor resection, but results are poor due to loosening and breakage [9].
- (2)
- Custom-made prosthesis: this is considered the most common treatment when modular prostheses are not available. Their use is very common in children. Custom-made prostheses can provoke aseptic loosening, and cause loss of bone stock, making revision surgery difficult and obtaining low functional results [11].
- (3)
- Vascularized fibular flap: this technique was proposed by Manfrini et al., who replaced the femur by modeling the autogenous fibula, reproducing the femoral shape. The fibular epiphysis was used to imitate the femoral head [12]. The advantage of this technique is the potential growth of the fibula and its remodeling; nevertheless, it is a very difficult technique, and the success rate is quite low. Indeed, there are no case series in the literature.
- (4)
- Extracorporeal irradiated autograft: this technique consists of reimplantation of the resected specimen after irradiation and soft tissue removal. It has the advantage of a perfect anatomical correspondence, although non-union is common [13]; moreover, it does not furnish complete information about histology and tumor necrosis after neoadjuvant chemotherapy. Although the hypothetical risk of local recurrence is present, it seems similar to that of other techniques which do not include the reimplantation of the specimen [14].
- (5)
- Osteoarticular homograft: this is rarely used in isolation for the inferior limb, principally because it collapses under body weight and cannot articulate with the acetabulum, undergoing precocious resorption [15]. Moreover, these are not available for children, due to the absence of donors.
- (6)
- Composite prosthesis (association of a joint prosthesis and a cemented homograft): this has the advantage of increasing the bone stock [16]; the prosthetic component should also guarantee a good articular motion. The corresponding homograft segment would be the best solution, but unfortunately, it is impossible to have child donors, so homografts from adult donors have to be adapted for young patients [10]. The homograft medullary canal is completely filled with cement in order to obtain higher resistance in weight-bearing.
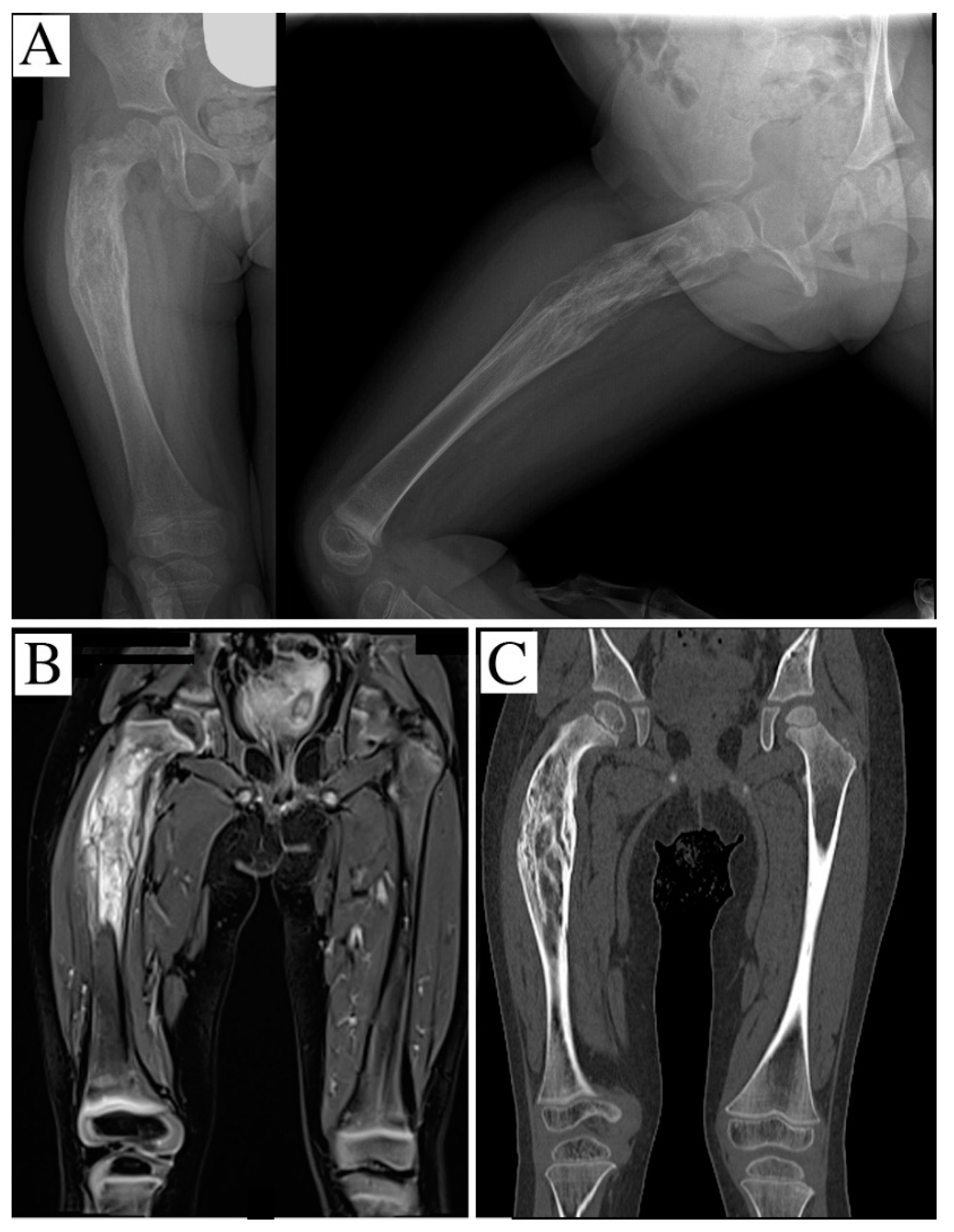
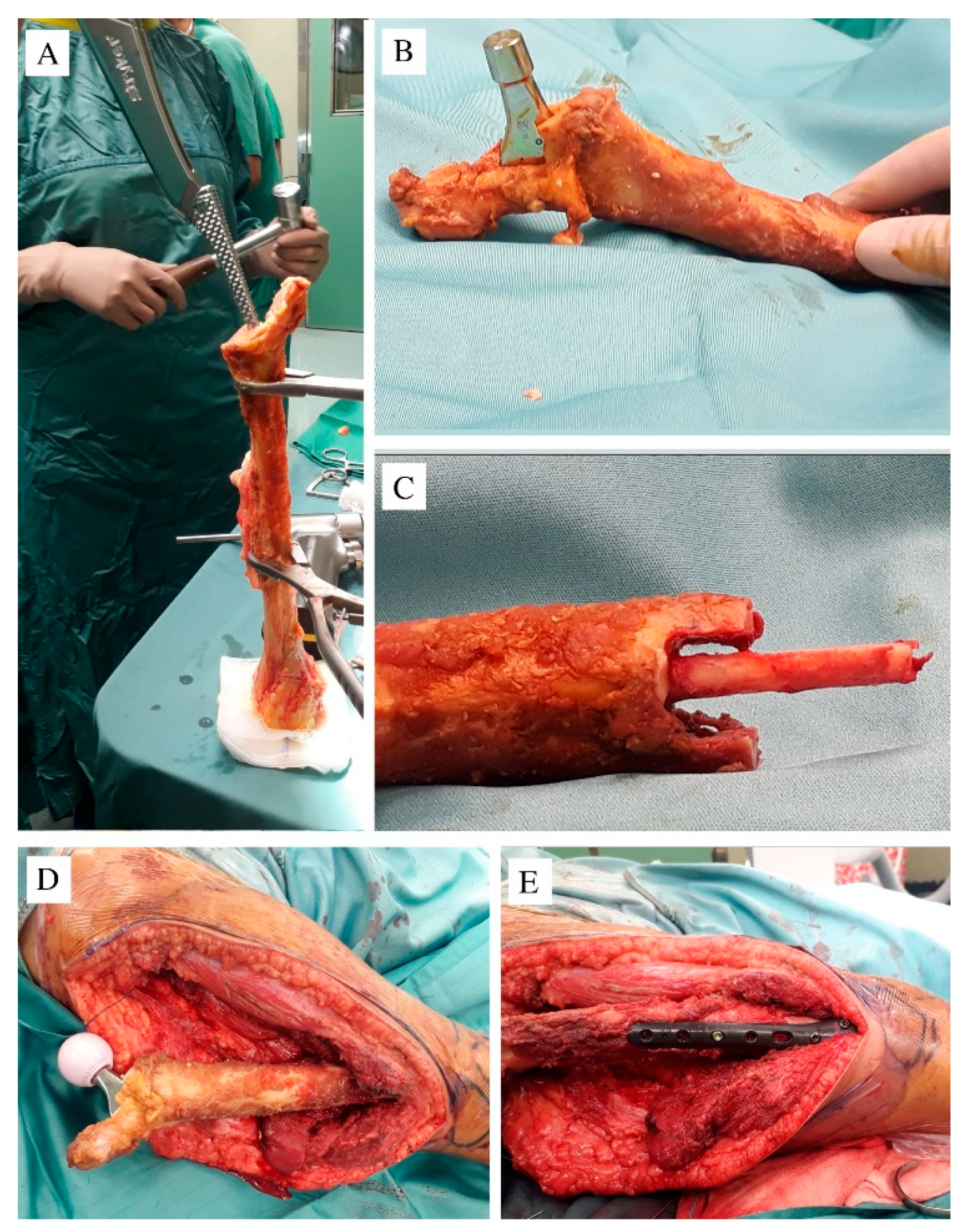
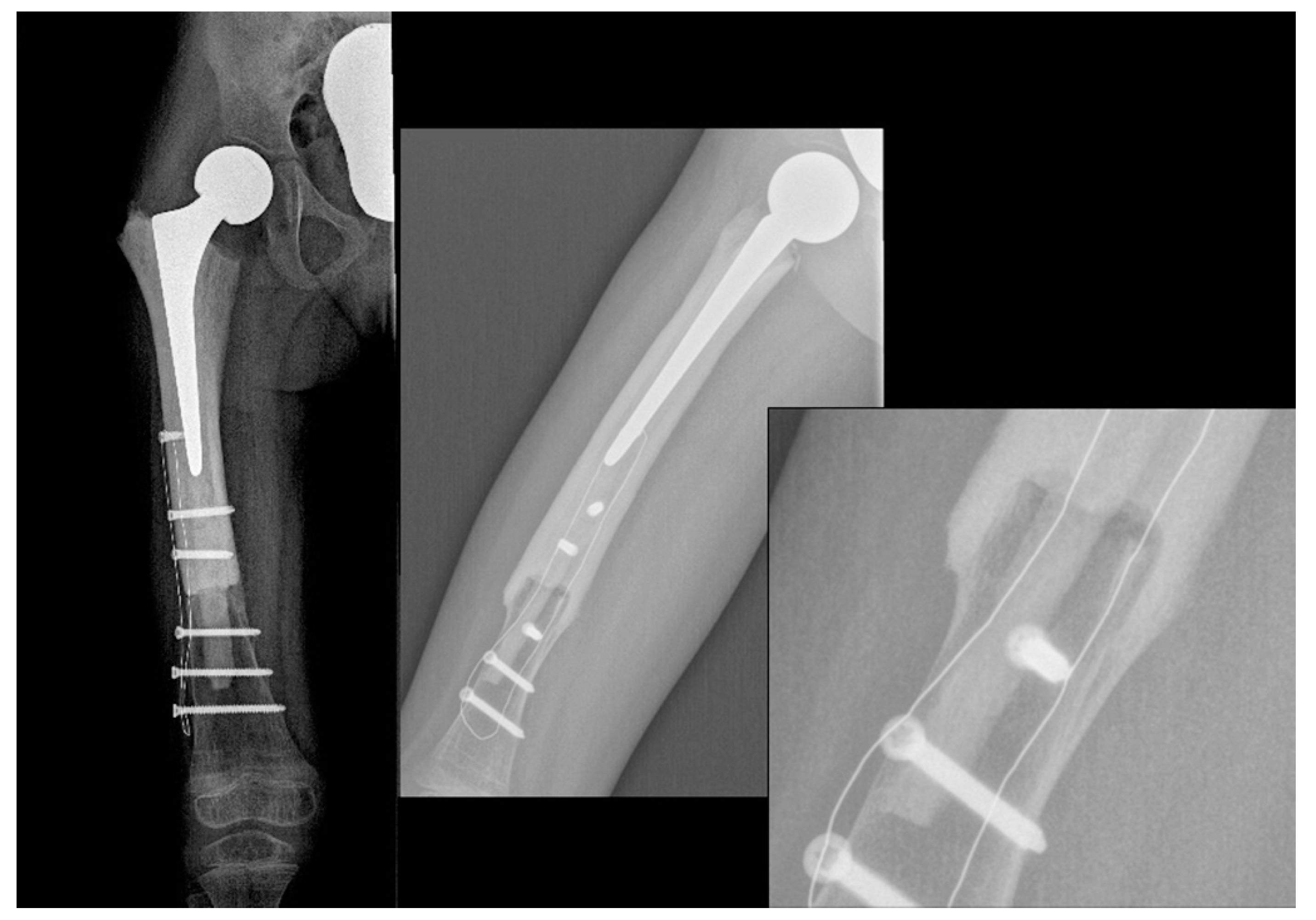
2. Case Report
3. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Granowetter, L.; Womer, R.; Devidas, M.; Krailo, M.; Wang, C.; Bernstein, M.; Marina, N.; Leavey, P.; Gebhardt, M.; Healey, J.; et al. Dose-intensified compared with standard chemotherapy for nonmetastatic ewing sarcoma family of tumors: A children’s oncology group study. J. Clin. Oncol. 2009, 27, 2536–2541. [Google Scholar] [CrossRef]
- Cirstoiu, C.; Cretu, B.; Serban, B.; Panti, Z.; Nica, M. Current review of surgical management options for extremity bone sarcomas. EFORT Open Rev. 2019, 4, 174–182. [Google Scholar] [CrossRef]
- Hesla, A.C.; Papakonstantinou, A.; Tsagkozis, P. Current status of management and outcome for patients with ewing sarcoma. Cancers 2021, 13, 1202. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, S.; Palmerini, E. Adjuvant and neoadjuvant combination chemotherapy for osteogenic sarcoma. Curr. Opin. Oncol. 2007, 19, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Goryń, T.; Pieńkowski, A.; Szostakowski, B.; Zdzienicki, M.; Ługowska, I.; Rutkowski, P. Functional outcome of surgical treatment of adults with extremity osteosarcoma after megaprosthetic reconstruction-Single-center experience. J. Orthop. Surg. Res. 2019, 14. [Google Scholar] [CrossRef] [PubMed]
- Pala, E.; Trovarelli, G.; Angelini, A.; Maraldi, M.; Berizzi, A.; Ruggieri, P. Megaprosthesis of the knee in tumor and revision surgery. Acta Biomed. 2017, 88, 129–138. [Google Scholar] [CrossRef]
- Dukan, R.; Mascard, E.; Langlais, T.; Ouchrif, Y.; Glorion, C.; Pannier, S.; Bouthors, C. Long-term outcomes of non-invasive expandable endoprostheses for primary malignant tumors around the knee in skeletally-immature patients. Arch. Orthop. Trauma Surg. 2021. [Google Scholar] [CrossRef] [PubMed]
- Torner, F.; Segur, J.M.; Ullot, R.; Soldado, F.; Domenech, P.; DeSena, L.; Knorr, J. Non-invasive expandable prosthesis in musculoskeletal oncology paediatric patients for the distal and proximal femur. First results. Int. Orthop. 2016, 40, 1683–1688. [Google Scholar] [CrossRef]
- Baumgart, R.; Lenze, U. Expandable endoprostheses in malignant bone tumors in children: Indications and limitations. Recent Results Cancer Res. 2009, 179, 59–73. [Google Scholar]
- Campanacci, L.; Alì, N.; Casanova, J.M.P.S.; Kreshak, J.; Manfrini, M. Resurfaced allograft-prosthetic composite for proximal tibial reconstruction in children: Intermediate-term results of an original technique. J. Bone Jt. Surg. Am. Vol. 2015, 97, 241–250. [Google Scholar] [CrossRef]
- Schindler, O.S.; Cannon, S.R.; Briggs, T.W.R.; Blunn, G.W. Stanmore custom-made extendible distal femoral replacements. Clinical experience in children with primary malignant bone tumours. J. Bone Jt. Surg. Ser. B 1997, 79, 927–937. [Google Scholar] [CrossRef]
- Manfrini, M.; Innocenti, M.; Ceruso, M.; Mercuri, M. Original biological reconstruction of the hip in a 4-year-old girl. Lancet 2003, 361, 140–142. [Google Scholar] [CrossRef]
- Oike, N.; Kawashima, H.; Ogose, A.; Hatano, H.; Ariizumi, T.; Kaidu, M.; Aoyama, H.; Endo, N. Long-term outcomes of an extracorporeal irradiated autograft for limb salvage operations in musculoskeletal tumours: Over ten years’ observation. Bone Jt. J. 2019, 101-B, 1151–1159. [Google Scholar] [CrossRef] [PubMed]
- Mihara, A.; Muramatsu, K.; Hashimoto, T.; Iwanaga, R.; Ihara, K.; Sakai, T. Combination of extracorporeally-irradiated autograft and vascularized bone graft for reconstruction of malignant musculoskeletal tumor. Anticancer Res. 2020, 40, 1637–1643. [Google Scholar] [CrossRef]
- Roque, P.J.; Mankin, H.J.; Malchau, H. Proximal femoral allograft: Prognostic indicators. J. Arthroplasty 2010, 25, 1028–1033. [Google Scholar] [CrossRef]
- Takeuchi, A.; Yamamoto, N.; Hayashi, K.; Matsubara, H.; Miwa, S.; Igarashi, K.; Tsuchiya, H. Joint-preservation surgery for pediatric osteosarcoma of the knee joint. Cancer Metastasis Rev. 2019, 38, 709–722. [Google Scholar] [CrossRef]
- Picci, P.; Böhling, T.; Bacci, G.; Ferrari, S.; Sangiorgi, L.; Mercuri, M.; Ruggieri, P.; Manfrini, M.; Ferraro, A.; Casadei, R.; et al. Chemotherapy-induced tumor necrosis as a prognostic factor in localized Ewing’s sarcoma of the extremities. J. Clin. Oncol. 1997, 15, 1553–1559. [Google Scholar] [CrossRef]
- Weisstein, J.S.; Goldsby, R.E.; O’Donnell, R.J. Oncologic approaches to pediatric limb preservation. J. Am. Acad. Orthop. Surg. 2005, 13, 544–554. [Google Scholar] [CrossRef]
- Lim, Z.; Strike, S.A.; Puhaindran, M.E. Sarcoma of the lower limb: Reconstructive surgeon’s perspective. Indian J. Plast. Surg. 2019, 52, 55–61. [Google Scholar] [CrossRef]
- Finn, H.A.; Simon, M.A. Limb-salvage surgery in the treatment of osteosarcoma in skeletally immature individuals. Clin. Orthop. Relat. Res. 1991, 108–118. [Google Scholar] [CrossRef]
- Herring, J.A. Tachdjian’s Pediatric Orthopaedics: From the Texas Scottish Rite Hospital for Children, 3rd ed.; Herring, J.A., Ed.; Saunders: Philadelphia, PA, USA, 2002. [Google Scholar]
- Muscolo, D.L.; Ayerza, M.A.; Aponte-Tinao, L.; Farfalli, G. Allograft reconstruction after sarcoma resection in children younger than 10 years old. In Proceedings of the Clinical Orthopaedics and Related Research; Springer: New York, NY, USA, 2008; Volume 466, pp. 1856–1862. [Google Scholar]
- Soriani, A.; Strigari, L.; Petrongari, M.G.; Anelli, V.; Baldi, J.; Salducca, N.; Biagini, R.; Zoccali, C. The advantages of carbon fiber-based orthopedic devices in patients who have to undergo radiotherapy: An experimental evidence. Acta Biomed. 2020, 91, 1–12. [Google Scholar] [CrossRef]
- Bagheri, Z.S.; El Sawi, I.; Schemitsch, E.H.; Zdero, R.; Bougherara, H. Biomechanical properties of an advanced new carbon/flax/epoxy composite material for bone plate applications. J. Mech. Behav. Biomed. Mater. 2013, 20, 398–406. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zoccali, C.; Careri, S.; Attala, D.; Florio, M.; Milano, G.M.; Giordano, M. A New Proximal Femur Reconstruction Technique after Bone Tumor Resection in a Very Small Patient: An Exemplificative Case. Children 2021, 8, 442. https://doi.org/10.3390/children8060442
Zoccali C, Careri S, Attala D, Florio M, Milano GM, Giordano M. A New Proximal Femur Reconstruction Technique after Bone Tumor Resection in a Very Small Patient: An Exemplificative Case. Children. 2021; 8(6):442. https://doi.org/10.3390/children8060442
Chicago/Turabian StyleZoccali, Carmine, Silvia Careri, Dario Attala, Michela Florio, Giuseppe Maria Milano, and Marco Giordano. 2021. "A New Proximal Femur Reconstruction Technique after Bone Tumor Resection in a Very Small Patient: An Exemplificative Case" Children 8, no. 6: 442. https://doi.org/10.3390/children8060442
APA StyleZoccali, C., Careri, S., Attala, D., Florio, M., Milano, G. M., & Giordano, M. (2021). A New Proximal Femur Reconstruction Technique after Bone Tumor Resection in a Very Small Patient: An Exemplificative Case. Children, 8(6), 442. https://doi.org/10.3390/children8060442






