Comparing Standardized and Parent-Reported Motor Outcomes of Extremely Preterm Infants
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
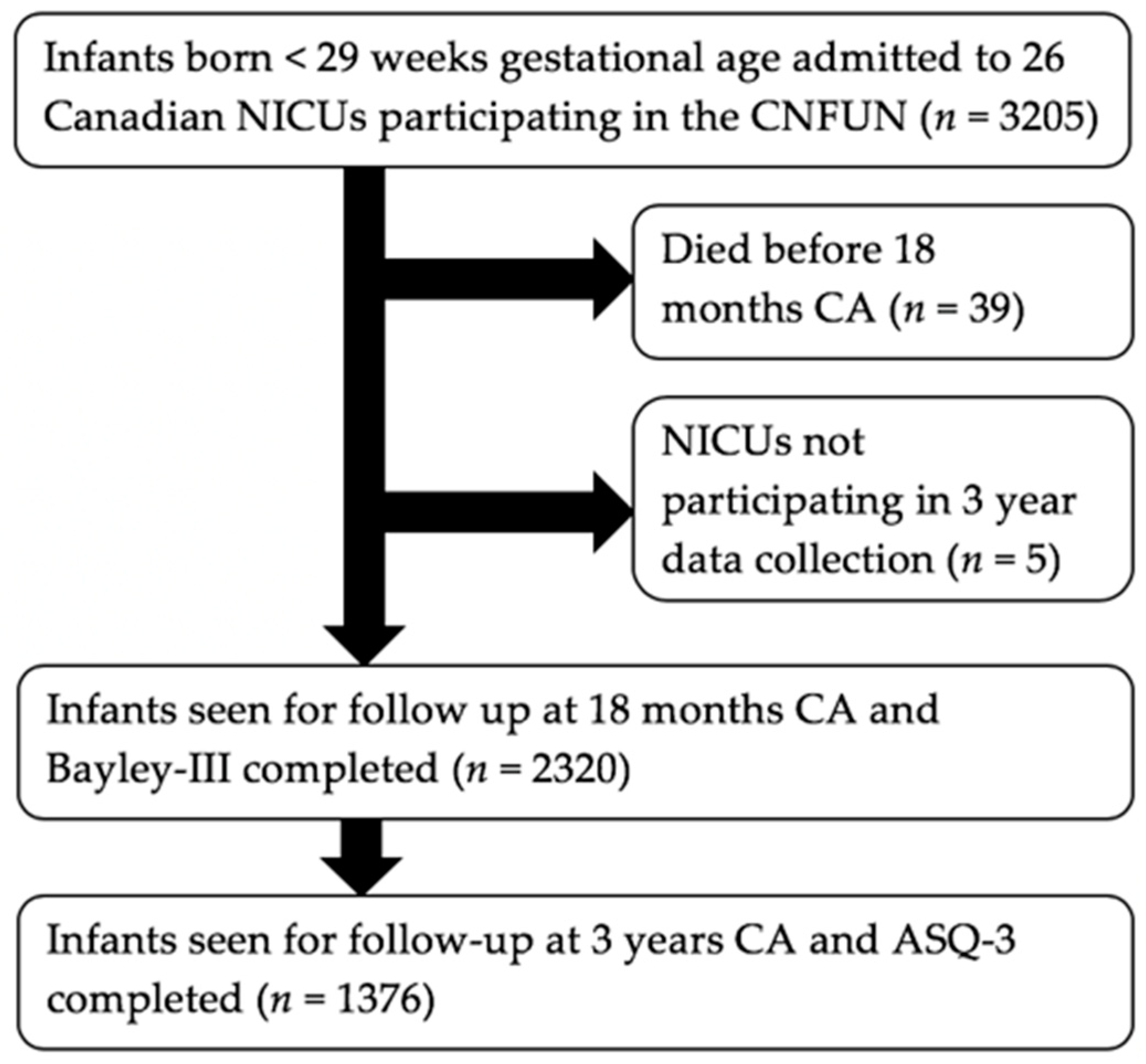
2.2. Study Participants
2.3. Data Collection Protocol
2.4. Statistical Analyses
3. Results
4. Discussion
4.1. Bayley-III Scores at 18 Months CA and ASQ Scores at 3 Years CA
4.2. Correlation of Bayley-III Motor Scores at 18 Months CA and ASQ Motor Scores at 3 Years CA
4.3. Predictive Validity of Bayley-III Motor Scores at 18 Months CA to ASQ Motor SCORES at 3 years CA
4.4. Study Limitations
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Ishii, N.; Kono, Y.; Yonemoto, N.; Kusuda, S.; Fujimura, M. Outcomes of infants born at 22 and 23 weeks’ gestation. Pediatrics 2013, 132, 62–71. [Google Scholar] [CrossRef]
- Serenius, F.; Källén, K.; Blennow, M.; Ewald, U.; Fellman, V.; Holmström, G.; Group, F.T. Neurodevelopmental outcome in extremely preterm infants at 2.5 years after active perinatal care in Sweden. J. Am. Med. Assoc. 2013, 309, 1810–1820. [Google Scholar] [CrossRef]
- Spittle, A.J.; Orton, J. Cerebral palsy and developmental coordination disorder in children born preterm. Semin. Fetal Neonatal Med. 2014, 19, 84–89. [Google Scholar] [CrossRef]
- Edwards, J.; Berube, M.; Erlandson, K.; Haug, S.; Johnstone, H.; Meagher, M.; Sarkodee-Adoo, S.; Zwicker, J.G. Developmental coordination disorder in school-aged children born very preterm and/or at very low birth weight. J. Dev. Behav. Pediatr. 2011, 32, 678–687. [Google Scholar] [CrossRef]
- Synnes, A.R.; Lefebvre, F.; Cake, H.A. Current status of neonatal follow-up in Canada. Pediatr. Child Health 2006, 11, 271–274. [Google Scholar]
- Synnes, A.; Luu, T.; Moddemann, D.; Church, P.; Lee, D.; Vincer, M.; Ballantyne, M.; Majnemer, A.; Creighton, D.; Yang, J.; et al. Determinants of developmental outcomes in a very preterm Canadian cohort. Arch. Dis. Child. Fetal Neonatal Ed. 2017, 102, 235–243. [Google Scholar] [CrossRef]
- Bayley, N. Bayley Scales of Infant and Toddler Development, 3rd ed.; Psychological Corporation: San Antonio, TX, USA, 2006. [Google Scholar]
- Spittle, A.J.; Spencer-Smith, M.M.; Eeles, A.L.; Lee, K.J.; Lorefice, L.E.; Anderson, P.J.; Doyle, L.W. Does the Bayley-III motor scale at 2 years predict motor outcome at 4 years in very preterm children? Dev. Med. Child Neurol. 2013, 55, 448–452. [Google Scholar] [CrossRef] [PubMed]
- De Kieviet, J.F.; Piek, J.P.; Aarnoudse-Moens, C.S.; Oosterlaan, J. Motor development in very preterm and very low-birth-weight children from birth to adolescence: A meta-analysis. J. Am. Med. Assoc. 2009, 302, 2235–2242. [Google Scholar] [CrossRef] [PubMed]
- Duncan, A.F.; Bann, C.; Boatman, C.; Hintz, S.R.; Vaucher, Y.E.; Vohr, B.R.; Yolton, K.; Heyne, R.J. Do currently recommended Bayley-III cutoffs overestimate motor impairment in infants born <27 weeks gestation? J. Perinatol. 2015, 35, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Anderson, P.J.; De Luca, C.R.; Hutchinson, E.; Roberts, G.; Doyle, L.W. The Victorian Infant Collaborative Group. Underestimation of developmental delay by the new Bayley-III Scale. Arch. Pediatr. Adolesc. Med. 2010, 164, 352–356. [Google Scholar] [CrossRef] [PubMed]
- Aylward, G. Continuing issues with the Bayley-III: Where to go from here. J. Dev. Behav. Pediatr. 2013, 34, 697–701. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Yeh, J.C.; Blanchard, S.B. Ages and Stages Questionnaire: A global screening scale. Bol. Med. Hosp. Infant. Mex. 2017, 74, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Squires, J.; Twombly, E.; Bricker, D.; Potter, L. The ASQ-3 User’s Guide: Ages & Stages Questionnaires, 3rd ed.; Paul, H., Ed.; Brookes Pub. Co.: Baltimore, MD, USA, 2009. [Google Scholar]
- Pontoppidan, M.; Niss, N.K.; Pejtersen, J.H.; Julian, M.M.; Væver, M.S. Parent report measures of infant and toddler social-emotional development: A systematic review. Fam. Pract. 2017, 34, 127–137. [Google Scholar] [CrossRef] [PubMed]
- Blaggan, S.; Guy, A.; Boyle, E.; Spata, E.; Manktelow, B.; Wolke, D.; Johnson, S. A parent questionnaire for developmental screening in infants born late and moderately preterm. Pediatrics 2014, 134, 55–62. [Google Scholar] [CrossRef] [PubMed]
| Birth Data | Median (IQR) or N (%) |
| Male | 741 (54) |
| Gestational age (weeks) | 27 (25, 28) |
| Birth weight (grams) | 910 (770, 1090) |
| Multiple births | 387 (28) |
| 18 Month CA Outcomes | Mean (SD) or N (%) |
| Motor Composite Score | |
| Mean | 91.5 (15) |
| ≥85 | 1002 (73) |
| 70–84 | 198 (14) |
| <70 | 92 (7) |
| Not performed (untestable) | 84 (6) |
| Definite Cerebral Palsy | 78 (6) |
| Gross Motor Subscale | |
| Mean | 7.7 (3) |
| ≥7 | 946 (69) |
| 4–6 | 249 (18) |
| <4 | 104 (7) |
| Not performed (untestable) | 77 (6) |
| Fine Motor Subscale | |
| Mean | 9.6 (3) |
| ≥7 | 1141 (83) |
| 4–6 | 121 (9) |
| <4 | 38 (3) |
| Not performed (untestable) | 76 (5) |
| 3-year CA Outcomes | Mean (SD) or N (%) |
| Gross Motor | |
| Mean | 48.70 (14) |
| Monitoring Zone (−1SD) | 184 (14) |
| Cut off Zone (−2SD) | 235 (18) |
| Fine Motor | |
| Mean | 40.97 (17) |
| Monitoring Zone (−1SD) | 216 (16) |
| Cut off Zone (−2SD) | 142 (11) |
| ASQ GM Normal N (%) | ASQ GM Monitoring N (%) | ASQ GM Abnormal N (%) | X2 | p-Value | |
|---|---|---|---|---|---|
| Bayley Motor ≥85 | 745 (78) | 126 (13) | 89 (9) | ||
| Bayley Motor 70–84 | 88 (46) | 41 (21) | 63 (33) | 285.2 | <0.01 |
| Bayley Motor <70 | 17 (19) | 8 (9) | 65 (72) | ||
| ASQ FM Normal N (%) | ASQ FM Monitoring N (%) | ASQ FM Abnormal N (%) | X2 | p-Value | |
|---|---|---|---|---|---|
| Bayley Motor ≥85 | 751 (80) | 131 (14) | 59 (6) | ||
| Bayley-Motor 70–84 | 115 (61) | 43 (23) | 30 (16) | 139.4 | <0.01 |
| Bayley Motor <70 | 33 (37) | 19 (21) | 37 (42) | ||
| ASQ GM Normal N (%) | ASQ GM Monitoring N (%) | ASQ GM Abnormal N (%) | X2 | p-Value | |
|---|---|---|---|---|---|
| Bayley Gross Motor ≥7 | 709 (78) | 117 (13) | 79 (9) | ||
| Bayley Gross Motor 4–6 | 130 (54) | 48 (20) | 64 (26) | 295.9 | <0.01 |
| Bayley Gross Motor <4 | 18 (17) | 10 (10) | 74 (73) | ||
| ASQ FM Normal N (%) | ASQ FM Monitoring N (%) | ASQ FM Abnormal N (%) | X2 | p-Value | |
|---|---|---|---|---|---|
| Bayley Fine Motor ≥7 | 841 (78) | 162 (15) | 70 (7) | ||
| Bayley Fine Motor 4–6 | 55 (48) | 28 (24) | 32 (28) | 192.3 | <0.01 |
| Bayley Fine Motor <4 | 6 (16) | 7 (19) | 24 (65) | ||
| ASQ-3 Gross Motor | ASQ-3 Fine Motor | |||
|---|---|---|---|---|
| Sensitivity (%) | Specificity (%) | Sensitivity (%) | Specificity (%) | |
| Bayley Motor Composite <85 | 45 | 88 | 40 | 84 |
| Bayley Motor Composite <84 | 45 | 88 | 40 | 84 |
| Bayley Motor Composite <83 | 45 | 88 | 40 | 84 |
| Bayley Motor Composite <82 | 38 | 92 | 34 | 88 |
| Bayley Motor Composite <81 | 38 | 92 | 34 | 88 |
| Bayley Motor Composite <80 | 38 | 92 | 34 | 88 |
| Bayley Motor Composite <79 | 30 | 95 | 27 | 92 |
| Bayley Motor Composite <78 | 30 | 95 | 27 | 92 |
| Bayley Motor Composite <77 | 30 | 95 | 27 | 92 |
| Bayley Motor Composite <76 | 26 | 96 | 25 | 94 |
| Bayley Motor Composite <75 | 26 | 96 | 25 | 94 |
| Bayley Motor Composite <74 | 26 | 96 | 25 | 94 |
| Bayley Motor Composite <73 | 23 | 98 | 21 | 95 |
| Bayley Motor Composite <72 | 23 | 98 | 21 | 95 |
| Bayley Motor Composite <71 | 23 | 98 | 21 | 95 |
| Bayley Motor Composite <70 | 23 | 98 | 18 | 96 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morgan-Feir, M.; Abbott, A.; Synnes, A.; Creighton, D.; Pillay, T.; Zwicker, J.G., on behalf of the Canadian Neonatal Follow-Up Network. Comparing Standardized and Parent-Reported Motor Outcomes of Extremely Preterm Infants. Children 2019, 6, 90. https://doi.org/10.3390/children6080090
Morgan-Feir M, Abbott A, Synnes A, Creighton D, Pillay T, Zwicker JG on behalf of the Canadian Neonatal Follow-Up Network. Comparing Standardized and Parent-Reported Motor Outcomes of Extremely Preterm Infants. Children. 2019; 6(8):90. https://doi.org/10.3390/children6080090
Chicago/Turabian StyleMorgan-Feir, Maeve, Andrea Abbott, Anne Synnes, Dianne Creighton, Thevanisha Pillay, and Jill G. Zwicker on behalf of the Canadian Neonatal Follow-Up Network. 2019. "Comparing Standardized and Parent-Reported Motor Outcomes of Extremely Preterm Infants" Children 6, no. 8: 90. https://doi.org/10.3390/children6080090
APA StyleMorgan-Feir, M., Abbott, A., Synnes, A., Creighton, D., Pillay, T., & Zwicker, J. G., on behalf of the Canadian Neonatal Follow-Up Network. (2019). Comparing Standardized and Parent-Reported Motor Outcomes of Extremely Preterm Infants. Children, 6(8), 90. https://doi.org/10.3390/children6080090





