PANDAS/PANS in the COVID-19 Age: Autoimmunity and Epstein–Barr Virus Reactivation as Trigger Agents?
Abstract
1. Introduction
2. COVID-19 Impact on Children and Adolescents
3. PANS/PANDAS
Pathogenic Mechanism of PANS/PANDAS
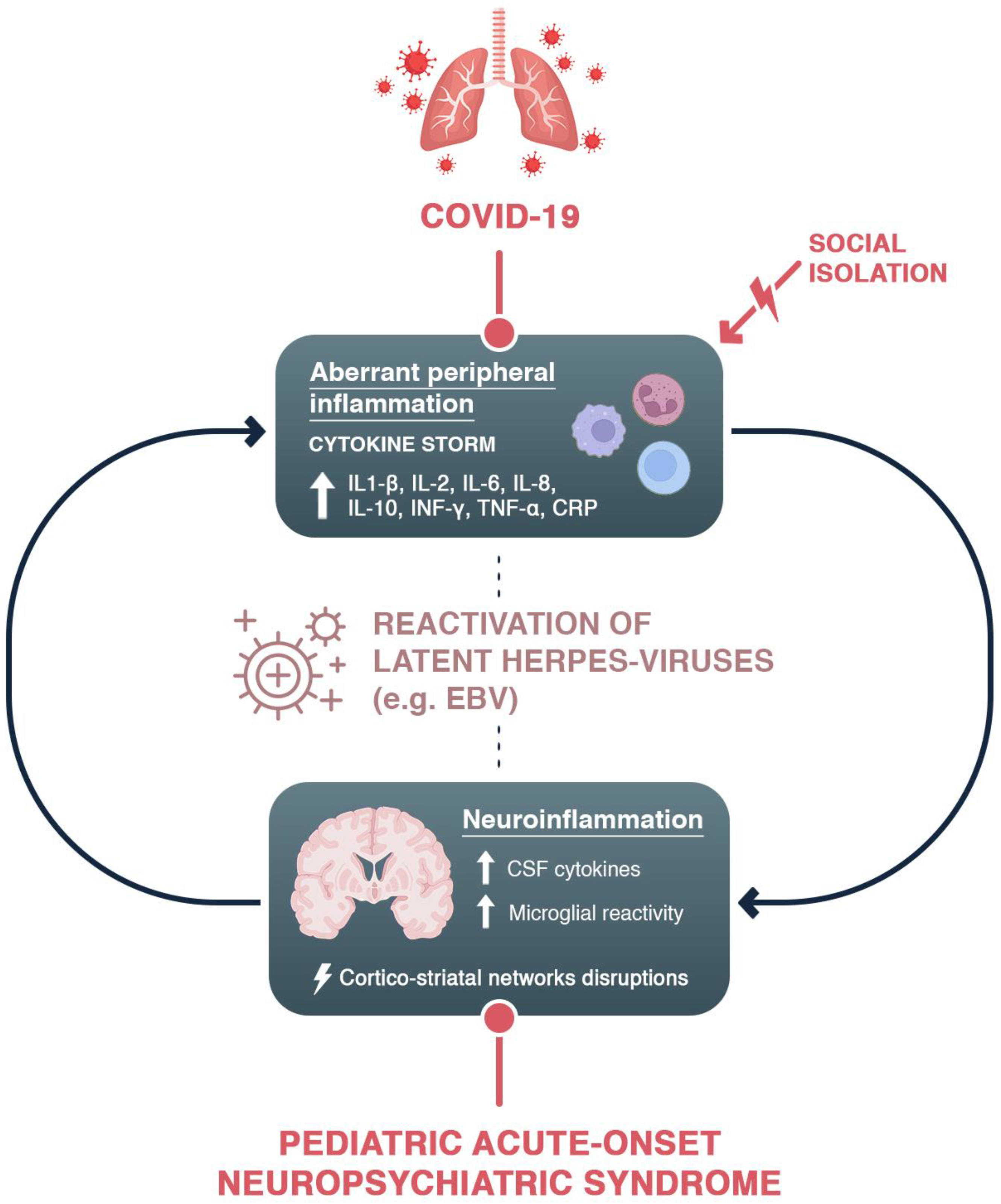
4. PANS and COVID-19
4.1. New Onset or Exacerbation of Symptoms
Cognitive Function Alterations
4.2. Vaccine Hesitancy and Vaccine-Related Neuroinflammation
5. A Case Series
6. Possible Mechanisms: Inflammation and Epstein–Barr Virus Reactivation
7. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Nalbandian, A.; Sehgal, K.; Gupta, A.; Madhavan, M.V.; McGroder, C.; Stevens, J.S.; Cook, J.R.; Nordvig, A.S.; Shalev, D.; Sehrawat, T.S.; et al. Post-acute COVID-19 syndrome. Nat. Med. 2021, 27, 601–615. [Google Scholar] [CrossRef]
- Ellul, M.A.; Benjamin, L.; Singh, B.; Lant, S.; Michael, B.D.; Easton, A.; Kneen, R.; Defres, S.; Sejvar, J.; Solomon, T. Neurological associations of COVID-19. Lancet Neurol. 2020, 19, 767–783. [Google Scholar] [CrossRef]
- Pallanti, S.; Grassi, E.; Makris, N.; Gasic, G.P.; Hollander, E. Neurocovid-19: A clinical neuroscience-based approach to reduce SARS-CoV-2 related mental health sequelae. J. Psychiatr. Res. 2020, 130, 215–217. [Google Scholar] [CrossRef] [PubMed]
- Chou, S.H.; Beghi, E.; Helbok, R.; Moro, E.; Sampson, J.; Altamirano, V.; Mainali, S.; Bassetti, C.; Suarez, J.I.; McNett, M.; et al. Global Incidence of Neurological Manifestations Among Patients Hospitalized With COVID-19-A Report for the GCS-NeuroCOVID Consortium and the ENERGY Consortium. JAMA Netw. Open 2021, 4, e2112131. [Google Scholar] [CrossRef] [PubMed]
- Al-Sarraj, S.; Troakes, C.; Hanley, B.; Osborn, M.; Richardson, M.P.; Hotopf, M.; Bullmore, E.; Everall, I.P. Invited Review: The spectrum of neuropathology in COVID-19. Neuropathol. Appl. Neurobiol. 2021, 47, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Guadarrama-Ortiz, P.; Choreño-Parra, J.A.; Sánchez-Martínez, C.M.; Pacheco-Sánchez, F.J.; Rodríguez-Nava, A.I.; García-Quintero, G. Neurological Aspects of SARS-CoV-2 Infection: Mechanisms and Manifestations. Front. Neurol. 2020, 11, 1039. [Google Scholar] [CrossRef] [PubMed]
- Speth, M.M.; Singer-Cornelius, T.; Oberle, M.; Gengler, I.; Brockmeier, S.J.; Sedaghat, A.R. Mood, Anxiety and Olfactory Dysfunction in COVID-19: Evidence of Central Nervous System Involvement? Laryngoscope 2020, 130, 2520–2525. [Google Scholar] [CrossRef] [PubMed]
- Premraj, L.; Kannapadi, N.V.; Briggs, J.; Seal, S.M.; Battaglini, D.; Fanning, J.; Suen, J.; Robba, C.; Fraser, J.; Cho, S.M. Mid and long-term neurological and neuropsychiatric manifestations of post-COVID-19 syndrome: A meta-analysis. J. Neurol. Sci. 2022, 434, 120162. [Google Scholar] [CrossRef]
- Venkatesan, P. NICE guideline on long COVID. Lancet Respir. Med. 2021, 9, 129. [Google Scholar] [CrossRef]
- Lam, M.H.; Wing, Y.K.; Yu, M.W.; Leung, C.M.; Ma, R.C.; Kong, A.P.; So, W.Y.; Fong, S.Y.; Lam, S.P. Mental morbidities and chronic fatigue in severe acute respiratory syndrome survivors: Long-term follow-up. Arch. Intern. Med. 2009, 169, 2142–2147. [Google Scholar] [CrossRef]
- Williams, C.Y.K.; Townson, A.T.; Kapur, M.; Ferreira, A.F.; Nunn, R.; Galante, J.; Phillips, V.; Gentry, S.; Usher-Smith, J.A. Interventions to reduce social isolation and loneliness during COVID-19 physical distancing measures: A rapid systematic review. PLoS ONE 2021, 16, e0247139. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Rai, M. Social isolation in Covid-19: The impact of loneliness. Int. J. Soc. Psychiatry 2020, 66, 525–527. [Google Scholar] [CrossRef] [PubMed]
- Abba-Aji, A.; Li, D.; Hrabok, M.; Shalaby, R.; Gusnowski, A.; Vuong, W.; Surood, S.; Nkire, N.; Li, X.M.; Greenshaw, A.J.; et al. COVID-19 Pandemic and Mental Health: Prevalence and Correlates of New-Onset Obsessive-Compulsive Symptoms in a Canadian Province. Int. J. Environ. Res. Public Health 2020, 17, 6986. [Google Scholar] [CrossRef] [PubMed]
- Van Ameringen, M.; Patterson, B.; Turna, J.; Lethbridge, G.; Goldman Bergmann, C.; Lamberti, N.; Rahat, M.; Sideris, B.; Francisco, A.P.; Fineberg, N.; et al. Obsessive-compulsive disorder during the COVID-19 pandemic. J. Psychiatr. Res. 2022, 149, 114–123. [Google Scholar] [CrossRef] [PubMed]
- Loades, M.E.; Chatburn, E.; Higson-Sweeney, N.; Reynolds, S.; Shafran, R.; Brigden, A.; Linney, C.; McManus, M.N.; Borwick, C.; Crawley, E. Rapid Systematic Review: The Impact of Social Isolation and Loneliness on the Mental Health of Children and Adolescents in the Context of COVID-19. J. Am. Acad. Child Adolesc. Psychiatry 2020, 59, 1218–1239.e3. [Google Scholar] [CrossRef] [PubMed]
- Panda, P.K.; Gupta, J.; Chowdhury, S.R.; Kumar, R.; Meena, A.K.; Madaan, P.; Sharawat, I.K.; Gulati, S. Psychological and Behavioral Impact of Lockdown and Quarantine Measures for COVID-19 Pandemic on Children, Adolescents and Caregivers: A Systematic Review and Meta-Analysis. J. Trop. Pediatr. 2021, 67, fmaa122. [Google Scholar] [CrossRef]
- Lopez-Leon, S.; Wegman-Ostrosky, T.; Ayuzo Del Valle, N.C.; Perelman, C.; Sepulveda, R.; Rebolledo, P.A.; Cuapio, A.; Villapol, S. Long-COVID in children and adolescents: A systematic review and meta-analyses. Sci. Rep. 2022, 12, 9950. [Google Scholar] [CrossRef]
- Steardo, L., Jr.; Steardo, L.; Verkhratsky, A. Psychiatric face of COVID-19. Transl. Psychiatry 2020, 10, 261. [Google Scholar] [CrossRef]
- Troyer, E.A.; Kohn, J.N.; Hong, S. Are we facing a crashing wave of neuropsychiatric sequelae of COVID-19? Neuropsychiatric symptoms and potential immunologic mechanisms. Brain Behav. Immun. 2020, 87, 34–39. [Google Scholar] [CrossRef]
- Racine, N.; Cooke, J.E.; Eirich, R.; Korczak, D.J.; McArthur, B.; Madigan, S. Child and adolescent mental illness during COVID-19: A rapid review. Psychiatry Res. 2020, 292, 113307. [Google Scholar] [CrossRef]
- Racine, N.; McArthur, B.A.; Cooke, J.E.; Eirich, R.; Zhu, J.; Madigan, S. Global Prevalence of Depressive and Anxiety Symptoms in Children and Adolescents During COVID-19: A Meta-analysis. JAMA Pediatr. 2021, 175, 1142–1150. [Google Scholar] [CrossRef] [PubMed]
- Jiao, W.Y.; Wang, L.N.; Liu, J.; Fang, S.F.; Jiao, F.Y.; Pettoello-Mantovani, M.; Somekh, E. Behavioral and Emotional Disorders in Children during the COVID-19 Epidemic. J. Pediatr. 2020, 221, 264–266.e1. [Google Scholar] [CrossRef] [PubMed]
- Tanir, Y.; Karayagmurlu, A.; Kaya, İ.; Kaynar, T.B.; Türkmen, G.; Dambasan, B.N.; Meral, Y.; Coşkun, M. Exacerbation of obsessive compulsive disorder symptoms in children and adolescents during COVID-19 pandemic. Psychiatry Res. 2020, 293, 113363. [Google Scholar] [CrossRef] [PubMed]
- Nissen, J.B.; Højgaard, D.R.M.A.; Thomsen, P.H. The immediate effect of COVID-19 pandemic on children and adolescents with obsessive compulsive disorder. BMC Psychiatry 2020, 20, 511. [Google Scholar] [CrossRef]
- Conti, E.; Sgandurra, G.; De Nicola, G.; Biagioni, T.; Boldrini, S.; Bonaventura, E.; Buchignani, B.; Della Vecchia, S.; Falcone, F.; Fedi, C.; et al. Behavioural and Emotional Changes during COVID-19 Lockdown in an Italian Paediatric Population with Neurologic and Psychiatric Disorders. Brain Sci. 2020, 10, 918. [Google Scholar] [CrossRef]
- Cusinato, M.; Iannattone, S.; Spoto, A.; Poli, M.; Moretti, C.; Gatta, M.; Miscioscia, M. Stress, Resilience, and Well-Being in Italian Children and Their Parents during the COVID-19 Pandemic. Int. J. Environ. Res. Public Health 2020, 17, 8297. [Google Scholar] [CrossRef]
- Zhou, S.J.; Zhang, L.G.; Wang, L.L.; Guo, Z.C.; Wang, J.Q.; Chen, J.C.; Liu, M.; Chen, X.; Chen, J.X. Prevalence and socio-demographic correlates of psychological health problems in Chinese adolescents during the outbreak of COVID-19. Eur. Child Adolesc. Psychiatry 2020, 29, 749–758. [Google Scholar] [CrossRef]
- Buonsenso, D.; Sali, M.; Pata, D.; De Rose, C.; Sanguinetti, M.; Valentini, P.; Delogu, G. Children and COVID-19: Microbiological and immunological insights. Pediatr. Pulmonol. 2020, 55, 2547–2555. [Google Scholar] [CrossRef]
- Savino, R.; Polito, A.N.; Arcidiacono, G.; Poliseno, M.; Lo Caputo, S. Neuropsychiatric Disorders in Pediatric Long COVID-19: A Case Series. Brain Sci. 2022, 12, 514. [Google Scholar] [CrossRef]
- Stoian, A.; Stoian, M.; Bajko, Z.; Maier, S.; Andone, S.; Cioflinc, R.A.; Motataianu, A.; Barcutean, L.; Balasa, R. Autoimmune encephalitis in COVID-19 infection: Our experience and systematic review of the literature. Biomedicines 2022, 10, 774. [Google Scholar] [CrossRef]
- Swedo, S.E.; Leonard, H.L.; Garvey, M.; Mittleman, B.; Allen, A.J.; Perlmutter, S.; Dow, S.; Zamkoff, J.; Dubbert, B.K.; Lougee, L. Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections: Clinical description of the first 50 cases. Am. J. Psychiatry 1998, 155, 264–271. [Google Scholar] [CrossRef]
- Swedo, S.E.; Leckman, J.F.; Rose, N.R. From research subgroup to clinical syndrome: Modifying the PANDAS criteria to describe PANS (pediatric acute-onset neuropsychiatric syndrome). Pediatr Ther. 2012, 2, 113. [Google Scholar] [CrossRef]
- Thienemann, M.; Murphy, T.; Leckman, J.; Shaw, R.; Williams, K.; Kapphahn, C.; Frankovich, J.; Geller, D.; Bernstein, G.; Chang, K.; et al. Clinical Management of Pediatric Acute-Onset Neuropsychiatric Syndrome: Part I-Psychiatric and Behavioral Interventions. J. Child Adolesc. Psychopharmacol. 2017, 27, 566–573. [Google Scholar] [CrossRef] [PubMed]
- Ercan, T.E.; Ercan, G.; Severge, B.; Arpaozu, M.; Karasu, G. Mycoplasma pneumoniae infection and obsessive-compulsive disease: A case report. J. Child Neurol. 2008, 23, 338–340. [Google Scholar] [CrossRef] [PubMed]
- Caruso, J.M.; Tung, G.A.; Gascon, G.G.; Rogg, J.; Davis, L.; Brown, W.D. Persistent preceding focal neurologic deficits in children with chronic Epstein-Barr virus encephalitis. J. Child Neurol. 2000, 15, 791–796. [Google Scholar] [CrossRef] [PubMed]
- Fallon, B.A.; Kochevar, J.M.; Gaito, A.; Nields, J.A. The underdiagnosis of neuropsychiatric Lyme disease in children and adults. Psychiatr. Clin. North Am. 1998, 21, 693–703. [Google Scholar] [CrossRef] [PubMed]
- Clapp, M.; Aurora, N.; Herrera, L.; Bhatia, M.; Wilen, E.; Wakefield, S. Gut microbiota’s effect on mental health: The gut-brain axis. Clin. Pract. 2017, 7, 987. [Google Scholar] [CrossRef]
- Molina, V.; Shoenfeld, Y. Infection, vaccines and other environmental triggers of autoimmunity. Autoimmunity 2005, 38, 235–245. [Google Scholar] [CrossRef]
- Zibordi, F.; Zorzi, G.; Carecchio, M.; Nardocci, N. CANS: Childhood acute neuropsychiatric syndromes. Eur. J. Paediatr. Neurol. 2018, 22, 316–320. [Google Scholar] [CrossRef]
- Garvey, M.; Jay, G.; Swedo, S. Topical review: PANDAS: The search for environmental triggers of pediatric neuropsychiatric disorders. Lessons from rheumatic fever. J. Child Neurol. 1998, 13, 413–423. [Google Scholar] [CrossRef]
- Murphy, T.K.; Patel, P.D.; McGuire, J.F.; Kennel, A.; Mutch, P.J.; Parker-Athill, E.C.; Hanks, C.E.; Lewin, A.B.; Storch, E.A.; Toufexis, M.D.; et al. Characterization of the pediatric acute-onset neuropsychiatric syndrome phenotype. J. Child Adolesc. Psychopharmacol. 2015, 25, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Dantzer, R.; Kelley, K.W. Twenty years of research on cytokine-induced sickness behavior. Brain Behav. Immun. 2007, 21, 153–160. [Google Scholar] [CrossRef]
- Williams, K.A.; Swedo, S.E. Post-infectious autoimmune disorders: Sydenham’s chorea, PANDAS and beyond. Brain Res. 2015, 1617, 144–154. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Williams, M.T.; Chugani, H.T. Evaluation of basal ganglia and thalamic inflammation in children with pediatric autoimmune neuropsychiatric disorders associated with streptococcal infection and tourette syndrome: A positron emission tomographic (PET) study using 11C-[R]-PK11195. J. Child Neurol. 2015, 30, 749–756. [Google Scholar] [CrossRef] [PubMed]
- Gray, S.M.; Bloch, M.H. Systematic review of proinflammatory cytokines in obsessive-compulsive disorder. Curr. Psychiatry Rep. 2012, 14, 220–228. [Google Scholar] [CrossRef] [PubMed]
- Walls, A.; Cubangbang, M.; Wang, H.; Raiji, M.; Knight, J.; Steehler, M.; Latimer, E.; Harley, E.H. Pediatric autoimmune neuropsychiatric disorder associated with streptococcus immunology: A pilot study. Otolaryngol. Head Neck Surg. 2015, 153, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Frankovich, J.; Cooperstock, M.; Cunningham, M.W.; Latimer, M.E.; Murphy, T.K.; From the PANS Collaborative Consortium. Clinical evaluation of youth with pediatric acute-onset neuropsychiatric syndrome (PANS): Recommendations from the 2013 PANS Consensus Conference. J. Child Adolesc. Psychopharmacol. 2015, 25, 3–13. [Google Scholar] [CrossRef]
- Frankovich, J.; Thienemann, M.; Rana, S.; Chang, K. Five youth with pediatric acute-onset neuropsychiatric syndrome of differing etiologies. J. Child Adolesc. Psychopharmacol. 2015, 25, 31–37. [Google Scholar] [CrossRef]
- O’Dor, S.L.; Zagaroli, J.S.; Belisle, R.M.; Hamel, M.A.; Downer, O.M.; Homayoun, S.; Williams, K.A. The COVID-19 pandemic and children with PANS/PANDAS: An evaluation of symptom severity, telehealth, and vaccination hesitancy. Child Psychiatry Human Develop. 2022, 1–9. [Google Scholar] [CrossRef]
- Guido, C.A.; Loffredo, L.; Zicari, A.M.; Pavone, P.; Savasta, S.; Gagliano, A.; Brindisi, G.; Galardini, G.; Bertolini, A.; Spalice, A. The Impact of the COVID-19 Epidemic During the Lockdown on Children with the Pediatric Acute-Onset Neuropsychiatric Syndrome (PANDAS/PANS): The Importance of Environmental Factors on Clinical Conditions. Front. Neurol. 2021, 12, 1310. [Google Scholar] [CrossRef]
- Efe, A. SARS-CoV-2/COVID-19 Associated Pediatric Acute-onset Neuropsychiatric Syndrome A Case Report of Female Twin Adolescents. Psychiatry Res. Case Rep. 2022, 1, 100074. [Google Scholar] [CrossRef] [PubMed]
- Pavone, P.; Ceccarelli, M.; Marino, S.; Caruso, D.; Falsaperla, R.; Berretta, M.; Rullo, E.V.; Nunnari, G. SARS-CoV-2 related paediatric acute-onset neuropsychiatric syndrome. Lancet Child Adolesc. Health 2021, 5, e19–e21. [Google Scholar] [CrossRef] [PubMed]
- Raviv, T.; Warren, C.M.; Washburn, J.J.; Kanaley, M.K.; Eihentale, L.; Goldenthal, H.J.; Russo, J.; Martin, C.P.; Lombard, L.S.; Tully, J.; et al. Caregiver perceptions of children’s psychological well-being during the COVID-19 pandemic. JAMA Netw. Open 2021, 4, e2111103. [Google Scholar] [CrossRef]
- Crivelli, L.; Palmer, K.; Calandri, I.; Guekht, A.; Beghi, E.; Carroll, W.; Frontera, J.; García-Azorín, D.; Westenberg, E.; Winkler, A.S.; et al. Changes in cognitive functioning after COVID-19: A systematic review and meta-analysis. Alzheimer’s Dement. 2022, 18, 1047–1066. [Google Scholar] [CrossRef] [PubMed]
- Lewin, A.B.; Storch, E.A.; Mutch, P.J.; Murphy, T.K. Neurocognitive functioning in youth with pediatric autoimmune neuropsychiatric disorders associated with streptococcus. J. Neuropsychiatry Clin. Neurosci. 2011, 23, 391–398. [Google Scholar] [CrossRef] [PubMed]
- Hirschtritt, M.E.; Hammond, C.J.; Luckenbaugh, D.; Buhle, J.; Thurm, A.E.; Casey, B.J.; Swedo, S.E. Executive and attention functioning among children in the PANDAS subgroup. Child Neuropsychol. 2009, 15, 179–194. [Google Scholar] [CrossRef] [PubMed]
- Gamucci, A.; Uccella, S.; Sciarretta, L.; D’Apruzzo, M.; Calevo, M.G.; Mancardi, M.M.; Veneselli, E.; De Grandis, E. PANDAS and PANS: Clinical, Neuropsychological, and Biological Characterization of a Monocentric Series of Patients and Proposal for a Diagnostic Protocol. J. Child Adolesc. Psychopharmacol. 2019, 29, 305–312. [Google Scholar] [CrossRef]
- Colvin, M.K.; Erwin, S.; Alluri, P.R.; Laffer, A.; Pasquariello, K.; Williams, K.A. Cognitive, Graphomotor, and Psychosocial Challenges in Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS). J. Neuropsychiatry Clin. Neurosci. 2021, 33, 90–97. [Google Scholar] [CrossRef]
- Pallanti, S.; Di Ponzio, M.; Gasic, G.; Gavazzi, G.; Benchester, B.; Heller, C.; Kikinis, Z.; Makris, N.; Kikinis, R. From ‘mental fog’ to ost-acute covid-19 syndrome’s executive function alteration: Implications for clinical approach. Submitted.
- Toniolo, S.; Di Lorenzo, F.; Scarioni, M.; Frederiksen, K.S.; Nobili, F. Is the Frontal Lobe the Primary Target of SARS-CoV-2? J. Alzheimer’s Dis. 2021, 81, 75–81. [Google Scholar] [CrossRef]
- Goldman, R.D.; Marneni, S.R.; Seiler, M.; Brown, J.C.; Klein, E.J.; Cotanda, C.P.; Gelernter, R.; Yan, T.D.; Hoeffe, J.; Davis, A.L.; et al. Caregivers’ willingness to accept expedited vaccine research during the COVID-19 pandemic: A cross-sectional survey. Clin. Ther. 2020, 42, 2124–2133. [Google Scholar] [CrossRef] [PubMed]
- Epstein, S.; Xia, Z.; Lee, A.J.; Dahl, M.; Edwards, K.; Levit, E.; Longbrake, E.E.; Perrone, C.; Kavak, K.; Weinstock-Guttman, B.; et al. Vaccination against SARS-CoV-2 in neuroinflammatory disease: Early safety/tolerability data. Mult. Scler. Relat. Disord. 2022, 57, 103433. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Xu, Z.; Wang, P.; Li, X.M.; Shuai, Z.W.; Ye, D.Q.; Pan, H.F. New-onset autoimmune phenomena post-COVID-19 vaccination. Immunology 2022, 165, 386–401. [Google Scholar] [CrossRef]
- Li, X.; Gao, L.; Tong, X.; Chan, V.K.; Chui, C.S.; Lai, F.T.; Wong, C.K.; Wan, E.Y.; Chan, E.W.; Lau, K.K.; et al. Autoimmune conditions following mRNA (BNT162b2) and inactivated (CoronaVac) COVID-19 vaccination: A descriptive cohort study among 1.1 million vaccinated people in Hong Kong. J. Autoimmun. 2022, 130, 102830. [Google Scholar] [CrossRef] [PubMed]
- Brusaferri, L.; Alshelh, Z.; Martins, D.; Kim, M.; Weerasekera, A.; Housman, H.; Morrisey, E.; Knight, P.; Castro, K.; Albrecht, D.; et al. The pandemic brain: Neuroinflammation in non-infected individuals during the COVID-19 pandemic. Brain Behav. Immun. 2022, 102, 89–97. [Google Scholar] [CrossRef]
- Lin, J.E.; Asfour, A.; Sewell, T.B.; Hooe, B.; Pryce, P.; Earley, C.; Shen, M.Y.; Kerner-Rossi, M.; Thakur, K.T.; Vargas, W.S.; et al. Neurological issues in children with COVID-19. Neurosci. Lett. 2021, 743, 135567. [Google Scholar] [CrossRef]
- Kakodkar, P.; Kaka, N.; Baig, M.N. A Comprehensive Literature Review on the Clinical Presentation, and Management of the Pandemic Coronavirus Disease 2019 (COVID-19). Cureus 2020, 12, e7560. [Google Scholar] [CrossRef]
- Robinson-Agramonte, M.A.; Gonçalves, C.A.; Noris-García, E.; Préndes Rivero, N.; Brigida, A.L.; Schultz, S.; Siniscalco, D.; García García, R.J. Impact of SARS-CoV-2 on neuropsychiatric disorders. World J. Psychiatry 2021, 11, 347–354. [Google Scholar] [CrossRef]
- He, Y.; Yu, R.; Ren, J. The correlation between psychiatric disorders and COVID-19: A narrative review. Psychiatr. Danub. 2021, 33, 76–85. [Google Scholar] [CrossRef]
- Proal, A.D.; VanElzakker, M.B. Long COVID or Post-acute Sequelae of COVID-19 (PASC): An Overview of Biological Factors That May Contribute to Persistent Symptoms. Front. Microbiol. 2021, 12, 698169. [Google Scholar] [CrossRef]
- Boldrini, M.; Canoll, P.D.; Klein, R.S. How COVID-19 affects the brain. JAMA Psychiatry 2021, 78, 682–683. [Google Scholar] [CrossRef] [PubMed]
- Schou, T.M.; Joca, S.; Wegener, G.; Bay-Richter, C. Psychiatric and neuropsychiatric sequelae of COVID-19–A systematic review. Brain Behav. Immun. 2021, 97, 328–348. [Google Scholar] [CrossRef] [PubMed]
- Ngo, B.; Lapp, S.A.; Siegel, B.; Patel, V.; Hussaini, L.; Bora, S.; Philbrook, B.; Weinschenk, K.; Wright, L.; Anderson, E.J.; et al. Cerebrospinal fluid cytokine, chemokine, and SARS-CoV-2 antibody profiles in children with neuropsychiatric symptoms associated with COVID-19. Mult. Scler. Relat. Disord. 2021, 55, 103169. [Google Scholar] [CrossRef] [PubMed]
- Bartley, C.M.; Johns, C.; Ngo, T.T.; Dandekar, R.; Loudermilk, R.L.; Alvarenga, B.D.; Hawes, I.A.; Zamecnik, C.R.; Zorn, K.C.; Alexander, J.R.; et al. Anti-SARS-CoV-2 and Autoantibody Profiles in the Cerebrospinal Fluid of 3 Teenaged Patients With COVID-19 and Subacute Neuropsychiatric Symptoms. JAMA Neurol. 2021, 78, 1503–1509. [Google Scholar] [CrossRef] [PubMed]
- Smatti, M.K.; Cyprian, F.S.; Nasrallah, G.K.; Al Thani, A.A.; Almishal, R.O.; Yassine, H.M. Viruses and autoimmunity: A review on the potential interaction and molecular mechanisms. Viruses 2019, 11, 762. [Google Scholar] [CrossRef] [PubMed]
- Gold, J.E.; Okyay, R.A.; Licht, W.E.; Hurley, D.J. Investigation of Long COVID Prevalence and Its Relationship to Epstein-Barr Virus Reactivation. Pathogens 2021, 10, 763. [Google Scholar] [CrossRef] [PubMed]
- Klein, J.; Wood, J.; Jaycox, J.; Lu, P.; Dhodapkar, R.M.; Gehlhausen, J.R.; Iwasaki, A. Distinguishing features of Long COVID identified through immune profiling. medRxiv 2022. [CrossRef]
- Eisenberger, N.I.; Moieni, M. Inflammation affects social experience: Implications for mental health. World Psychiatry 2020, 19, 109. [Google Scholar] [CrossRef]
- Koyama, Y.; Nawa, N.; Yamaoka, Y.; Nishimura, H.; Sonoda, S.; Kuramochi, J.; Miyazaki, Y.; Fujiwara, T. Interplay between social isolation and loneliness and chronic systemic inflammation during the COVID-19 pandemic in Japan: Results from U-CORONA study. Brain Behav. Immun. 2021, 94, 51–59. [Google Scholar] [CrossRef]
- Smith, K.J.; Gavey, S.; RIddell, N.E.; Kontari, P.; Victor, C. The association between loneliness, social isolation and inflammation: A systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2020, 112, 519–541. [Google Scholar] [CrossRef]
- Xiang, H.R.; Cheng, X.; Li, Y.; Luo, W.W.; Zhang, Q.Z.; Peng, W.X. Efficacy of IVIG (intravenous immunoglobulin) for corona virus disease 2019 (COVID-19): A meta-analysis. Int. Immunopharmacol. 2021, 96, 107732. [Google Scholar] [CrossRef] [PubMed]
- Sigra, S.; Hesselmark, E.; Bejerot, S. Treatment of PANDAS and PANS: A systematic review. Neurosci. Biobehav. Rev. 2018, 86, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Pashei, Y. Drug repurposing of selective serotonin reuptake inhibitors: Could these drugs help fight COVID-19 and save lives? J. Clin. Neurosci. 2021, 88, 163–172. [Google Scholar] [CrossRef] [PubMed]
| Criteria |
|---|
|
|
|
| Reference | Study Type | Sample | Notes |
|---|---|---|---|
| O’Dor et al., 2022 [49] | Survey | 254 minors | Self-reported (by caregivers) worsening of symptoms during the pandemic |
| Guido et al., 2021 [50] | Survey | 108 minors | Self-reported (by caregivers) worsening of symptoms during the pandemic |
| Efe, 2022 [51] | Case series | 2 adolescents | Onset of PANS temporarily associated with COVID-19 infection |
| Pavone et al., 2021 [52] | Case series | 2 adolescents | Onset of PANS temporarily associated with COVID-19 infection |
| Lab Test | Lab Ref Value | Case 1 | Case 2 | Case 3 | Case 4 | Case 5 |
|---|---|---|---|---|---|---|
| EBV anticorpi (VCA IgG) (U/mL) | <20 | 12 | 27 | 42 | 38 | 15 |
| IL-6 (pg/mL) | <4.4 | 8.2 | 5.6 | 7.8 | 12.8 | 5.2 |
| VES mm/h | 2–28 | 12 | 24 | 23 | 18 | 5 |
| TAS Ul/mL | <250 | 112 | 211 | 156 | 167 | 198 |
| CRP mg/dL <1.00 | <1.00 | 1.5 | <1.00 | 1.8 | <1.00 | <1.00 |
| Anticorpi anti-DNASI-B (U/mL) | <200 | 127 | 182 | 98 | 45 | 131 |
| Fibrinogen (mg/dL) | 180–400 | 421 | 736 | 531 | 386 | 350 |
| D-dimer (µg/L) | <500 | 410 | 118 | 257 | 511 | 689 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pallanti, S.; Di Ponzio, M. PANDAS/PANS in the COVID-19 Age: Autoimmunity and Epstein–Barr Virus Reactivation as Trigger Agents? Children 2023, 10, 648. https://doi.org/10.3390/children10040648
Pallanti S, Di Ponzio M. PANDAS/PANS in the COVID-19 Age: Autoimmunity and Epstein–Barr Virus Reactivation as Trigger Agents? Children. 2023; 10(4):648. https://doi.org/10.3390/children10040648
Chicago/Turabian StylePallanti, Stefano, and Michele Di Ponzio. 2023. "PANDAS/PANS in the COVID-19 Age: Autoimmunity and Epstein–Barr Virus Reactivation as Trigger Agents?" Children 10, no. 4: 648. https://doi.org/10.3390/children10040648
APA StylePallanti, S., & Di Ponzio, M. (2023). PANDAS/PANS in the COVID-19 Age: Autoimmunity and Epstein–Barr Virus Reactivation as Trigger Agents? Children, 10(4), 648. https://doi.org/10.3390/children10040648










