Appropriate Use of Antibiotic and Principles of Antimicrobial Stewardship in Children
Abstract
1. Introduction
2. What about Antimicrobial Resistance?
Antimicrobial Stewardship
3. Good Use of Antibiotics in Children
3.1. Patient Selection
3.2. Sample Collection for Cultures and Baseline Evaluation
3.3. Which Antibiotic Should Be Prescribed?
- -
- Previous microbiological isolates from the same patient, if available;
- -
- Local data on antimicrobial resistance, whether information is available;
- -
- To choose the antibiotic with the narrowest antimicrobial spectrum (the range of microorganisms an antimicrobic agent can kill or inhibit), related to the patient clinical condition;
- -
- Avoid multi-agent prescription, i.e., two or more molecules at the same time; when multiple therapies are necessary, avoid possible overlapping of antibacterial spectra (antibiotics effective against the same microbial agents);
- -
- Prescribe the appropriate dosage, paying attention that the antibiotic is not under dosed. Administration of suboptimal dosage, with the aim of reducing the risk of side effects, is not a good choice, since what is really reduced is only the probability to be effective. For example, prescription of amoxicillin is often underdosed, with dosage inferior to 60 mg/kg/die [27,28,29];
- -
- Choose the most appropriate way of administration: parenteral administration should be the first choice in septic patients, especially if with hemodynamic instability, or in case of difficult oral administration (vomiting, gastrointestinal dysfunction, or unconsciousness). In every other situation, oral administration could be the first choice given its non-inferiority in many infectious diseases, such as hospitalized children with severe not complicated pneumonia [27,30], bone and joint infection [31,32,33], acute pyelonephritis in children older than 3 months old, without urinary tract abnormalities [34,35].
3.3.1. Local Information on Microbial Resistances
3.3.2. AWaRe Classification
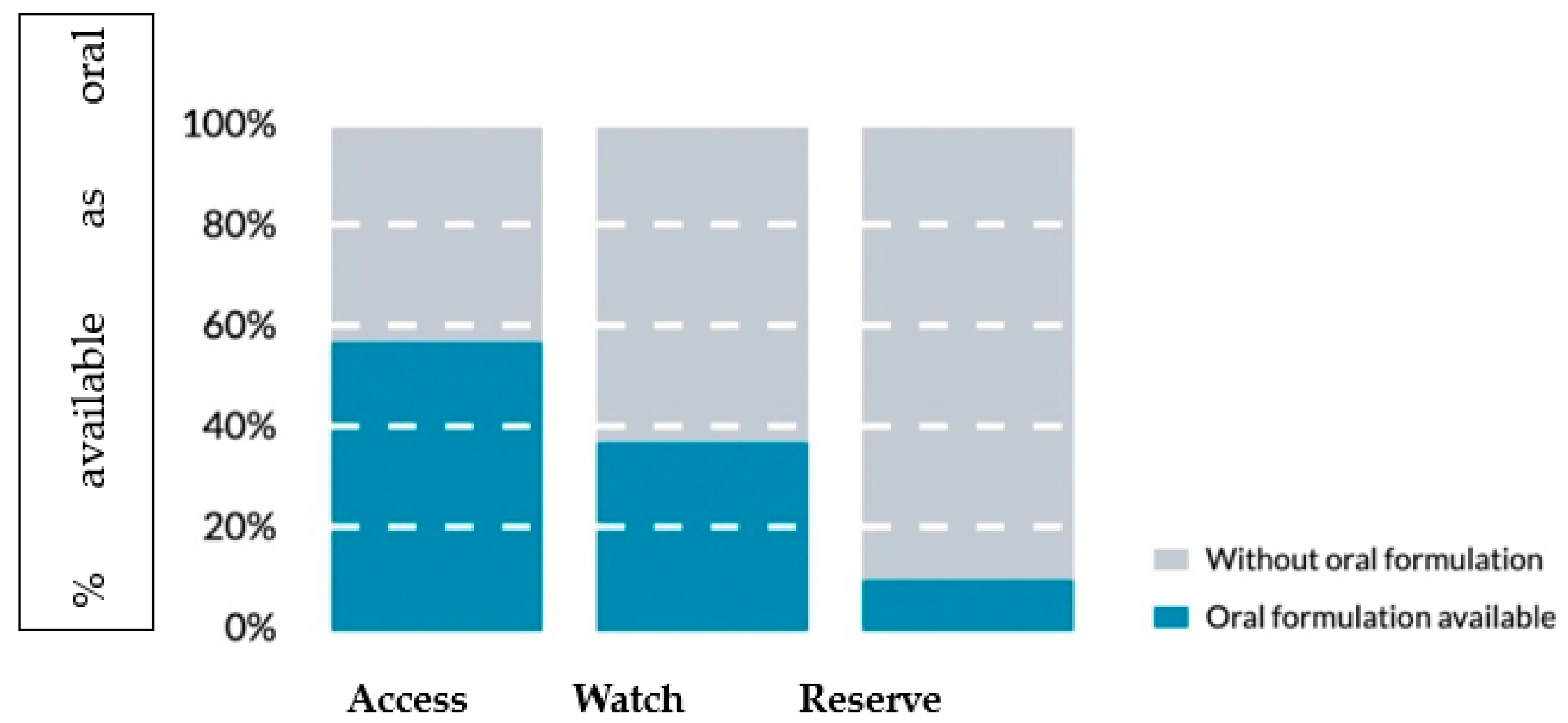
- The Access group includes first and second-choice antibiotics to treat the most common conditions with the best therapeutic value, minimizing the potential to induce resistance. Frequently for these antibiotics, oral administration is possible (Figure 1).
- The Watch group includes a selection of antibiotic molecules that should be used for specific and limited infections either as a first or second-choice agent. Their prescription is generally the target of antimicrobial stewardship programs for their higher potential to induce AMR.
- The Reserve group should be regarded as the final resource against multi-resistant bacteria when all previous antibiotic treatments have already failed. They are intended only for hospital use, and their prescription is usually under strict control, single-patient oriented, and better with specialist consultation.
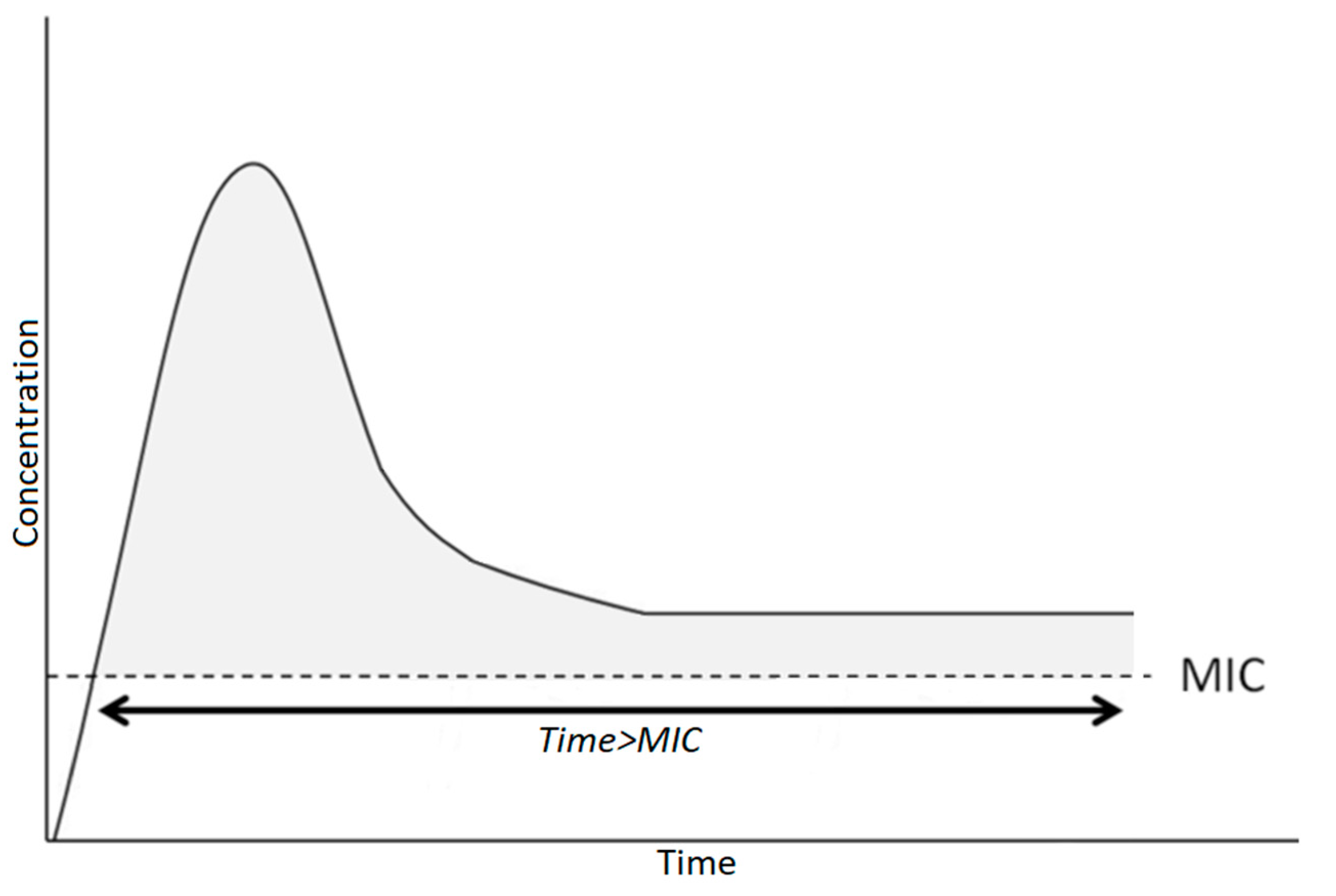
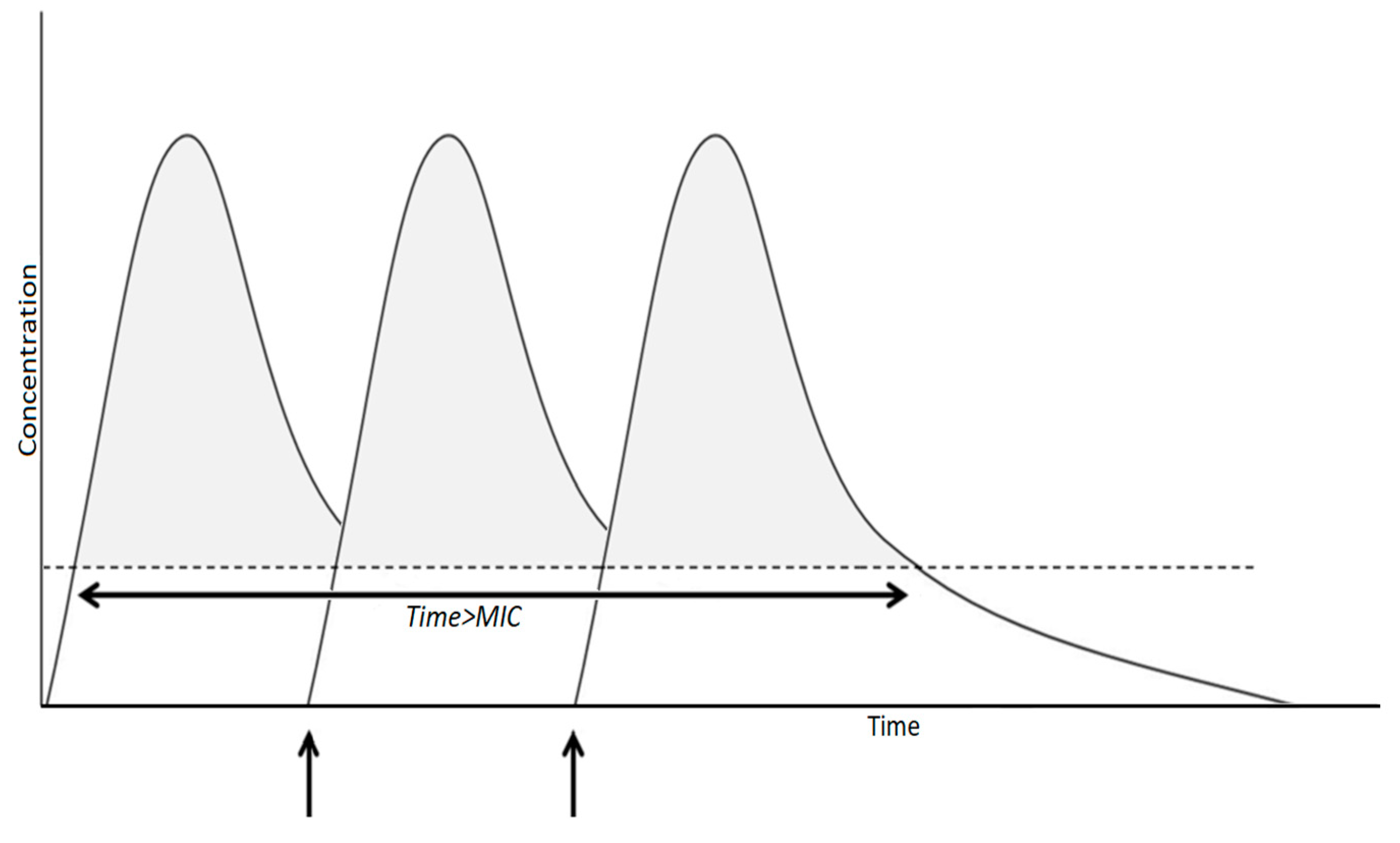
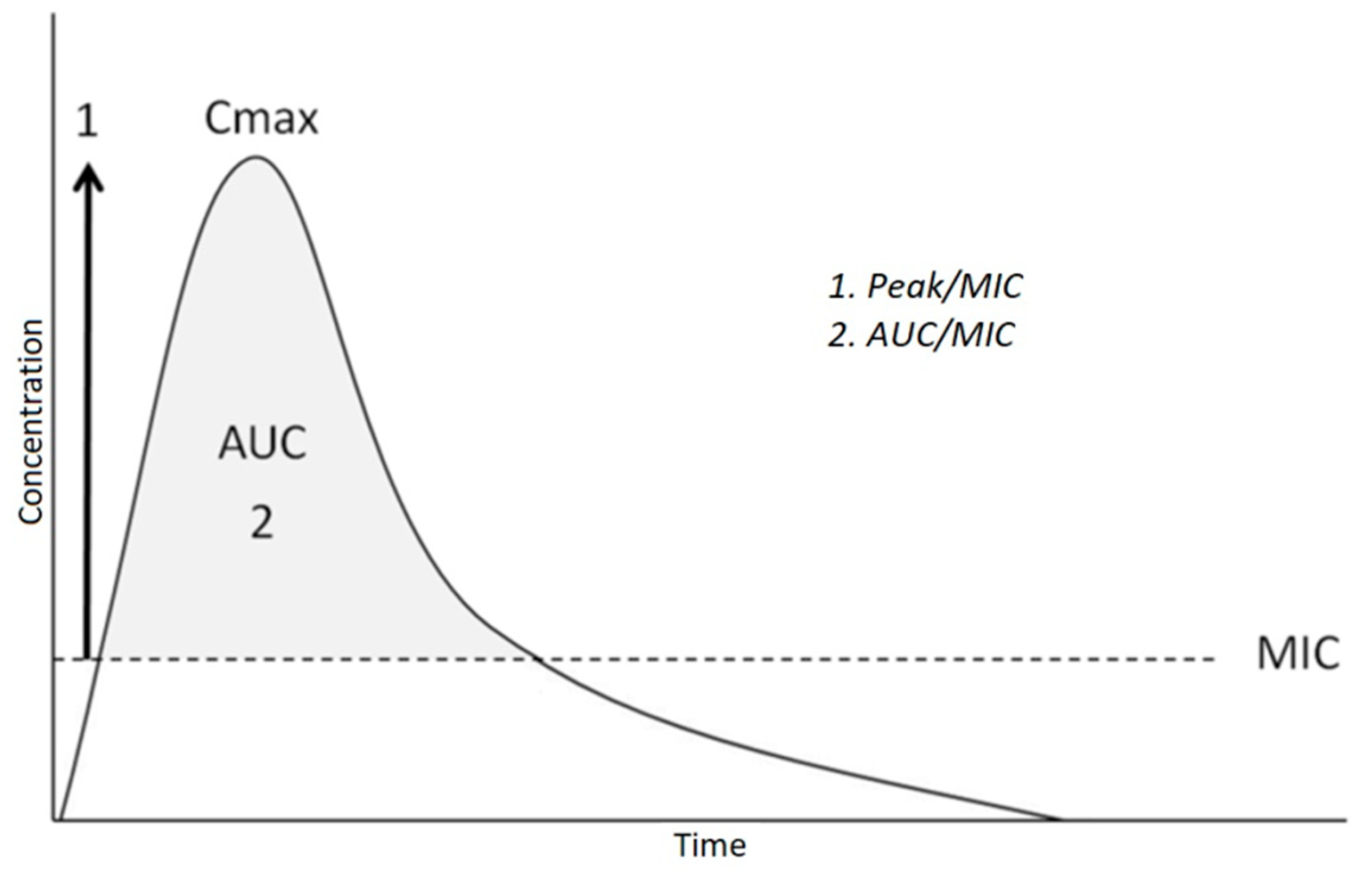
3.3.3. Schedule of Antibiotic Administration
3.4. Clinical and Laboratory Re-Evaluation: Therapeutic De-Escalation
3.4.1. Early Switch to Oral Administration
3.4.2. Re-Evaluation Based on Microbiologic Isolations
3.5. When to Stop Antimicrobial Treatment?
4. Conclusions
- Inappropriate or “too early” start of antibiotic therapy even in a clinical picture suggesting a viral infection;
- Post-operative prophylaxis not indicated or continued beyond 24 h.
- Lack of microbiological analysis supporting antibiotic treatment;
- Non-optimal antibiotic selection: starting with a broad-spectrum antibiotic when another with narrower spectrum (i.e., amoxicillin—clavulanic acid or ceftriaxone vs. amoxicillin) could be used;
- Suboptimal dosage and administration schedule for the considered infectious disease; parenteral administration even if oral route is feasible and equally effective;
- No therapeutic de-escalation, i.e., no modification of broad-spectrum therapy with a narrow-spectrum one;
- Duration of antibiotic therapy based on a pre-established “antibiotic course”.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- The Medicines Utilisation Monitoring Centre. National Report on Antibiotics Use in Italy. Year 2021; Italian Medicines Agency: Rome, Italy, 2023.
- Available online: https://assr.regione.emilia-romagna.it/pubblicazioni/rapporti-documenti/antibiotici-in-pediatria-2020 (accessed on 24 August 2022).
- Subramaniam, G.; Girish, M. Antibiotic Resistance-A Cause for Reemergence of Infections. Indian J. Pediatr. 2020, 87, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Souza da Cunha, S.; Santorelli, G.; Pearce, N.; Wright, J.; Oddie, S.; Petherick, E.; Pembrey, L. Evidence for causal associations between prenatal and postnatal antibiotic exposure and asthma in children, England. Clin. Exp. Allergy 2021, 51, 1438–1448. [Google Scholar] [CrossRef]
- Kelderer, F.; Mogren, I.; Eriksson, C.; Silfverdal, S.A.; Domellöf, M.; West, C.E. Associations between pre- and postnatal antibiotic exposures and early allergic outcomes: A population-based birth cohort study. Pediatr. Allergy Immunol. 2022, 33, e13848. [Google Scholar] [CrossRef]
- Bailey, L.C.; Forrest, C.B.; Zhang, P.; Richards, T.M.; Livshits, A.; DeRusso, P.A. Association of antibiotics in infancy with early childhood obesity. JAMA Pediatr. 2014, 168, 1063–1069. [Google Scholar] [CrossRef] [PubMed]
- McManus, M.C. Mechanisms of bacterial resistance to antimicrobial agents. Am. J. Health Syst. Pharm. 1997, 54, 1420–1433. [Google Scholar] [CrossRef] [PubMed]
- Munita, J.M.; Arias, C.A. Mechanisms of Antibiotic Resistance. Microbiol. Spectr. 2016, 4, 464–473. [Google Scholar] [CrossRef] [PubMed]
- Global Action Plan on Antimicrobial Resistance. Available online: https://www.who.int/publications/i/item/9789241509763 (accessed on 23 October 2022).
- Antimicrobial Resistance Collaborators. Global burden of bacterial antimicrobial resistance in 2019: A systematic analysis. Lancet 2022, 399, 629–655. [Google Scholar] [CrossRef]
- Aarestrup, F.M.; Wegener, H.C.; Collignon, P. Resistance in bacteria of the food chain: Epidemiology and control strategies. Expert Rev. Anti Infect. Ther. 2008, 6, 733–750. [Google Scholar] [CrossRef]
- Morley, V.J.; Woods, R.J.; Read, A.F. Bystander Selection for Antimicrobial Resistance: Implications for Patient Health. Trends Microbiol. 2019, 27, 864–877. [Google Scholar] [CrossRef] [PubMed]
- Nasrin, D.; Collignon, P.J.; Roberts, L.; Wilson, E.J.; Pilotto, L.S.; Douglas, R.M. Effect of beta lactam antibiotic use in children on pneumococcal resistance to penicillin: Prospective cohort study. BMJ 2002, 324, 28–30. [Google Scholar] [CrossRef]
- Malhotra-Kumar, S.; Lammens, C.; Coenen, S.; Van Herck, K.; Goossens, H. Effect of azithromycin and clarithromycin therapy on pharyngeal carriage of macrolide-resistant streptococci in healthy volunteers: A randomized, double-blind, placebo-controlled study. Lancet 2007, 369, 482–490. [Google Scholar] [CrossRef] [PubMed]
- The World Bank. Drug-Resistant Infections. A Threat to Our Economic Future. 2017. Available online: http://documents1.worldbank.org/curated/en/323311493396993758/pdf/final-report.pdf (accessed on 4 September 2022).
- Rahbi, F.A.; Salmi, I.A.; Khamis, F.; Balushi, Z.A.; Pandak, N.; Petersen, E.; Hannawi, S. Physicians’ attitudes, knowledge, and practices regarding antibiotic prescriptions. J. Glob. Antimicrob. Resist. 2022, 32, 58–65. [Google Scholar] [CrossRef]
- Cunha, C.B. Antimicrobial Stewardship Programs: Principles and Practice. Med. Clin. N. Am. 2018, 102, 797–803. [Google Scholar] [CrossRef] [PubMed]
- Donà, D.; Barbieri, E.; Daverio, M.; Lundin, R.; Giaquinto, C.; Zaoutis, T.; Sharland, M. Implementation and impact of pediatric antimicrobial stewardship programs: A systematic scoping review. Antimicrob. Resist. Infect. Control 2020, 9, 3. [Google Scholar] [CrossRef] [PubMed]
- Davey, P.; Marwick, C.A.; Scott, C.L.; Charani, E.; McNeil, K.; Brown, E.; Gould, I.M.; Ramsay, C.R.; Michie, S. Interventions to improve antibiotic prescribing practices for hospital inpatients. Cochrane Database Syst. Rev. 2017, 2, CD003543. [Google Scholar] [CrossRef] [PubMed]
- American Academy of Pediatrics. Antimicrobial Resistence and Antimicrobial Stewardship: Appropriate and Judicious Use of Antimicrobial Agents. In Red Book: 2021. Report of Committee on Infectious Disease; Kimberlin, D.W., Barnett, E.D., Lynfield, R., Sawyer, M.H., Eds.; American Academy of Pediatrics: Itasca, IL, USA, 2021; pp. 868–872. [Google Scholar]
- Saleh, E.A.; Schroeder, D.R.; Hanson, A.C.; Banerjee, R. Guideline-concordant antibiotic prescribing for pediatric outpatients with otitis media, community-acquired pneumonia, and skin and soft tissue infections in a large multispecialty healthcare system. Clin. Res. Infect. Dis. 2015, 2, 1010. [Google Scholar] [PubMed]
- McMullan, B.; Bryant, P.A.; Duffy, E.; Bielicki, J.; De Cock, P.; Science, M.; Zembles, T.; Timberlake, K.; Monsees, E.; Hamdy, R.F.; et al. Multinational consensus antimicrobial stewardship recommendations for children managed in hospital settings. Lancet Infect. Dis. 2022. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.J.; Gerber, J.S.; Hersh, A.L. Inpatient antimicrobial stewardship in pediatrics: A systematic review. J. Pediatric. Infect. Dis. Soc. 2015, 4, e127–e135. [Google Scholar] [CrossRef]
- Taylor, M.G.; Palazzi, D.L. Antimicrobial Stewardship in the Pediatric Primary Care Setting. Pediatr. Ann. 2022, 51, e196–e201. [Google Scholar] [CrossRef]
- Principi, N.; Esposito, S. Antimicrobial stewardship in paediatrics. BMC Infect. Dis. 2016, 16, 424. [Google Scholar] [CrossRef] [PubMed]
- Available online: https://www.choosingwisely.org/societies/american-academy-of-pediatrics-committee-on-infectious-diseases-and-the-pediatric-infectious-diseases-society/ (accessed on 10 March 2023).
- Tang, B.H.; Wu, Y.E.; Kou, C.; Qi, Y.J.; Qi, H.; Xu, H.Y.; Leroux, S.; Huang, X.; Zhou, Y.; Zheng, Y.; et al. Population Pharmacokinetics and Dosing Optimization of Amoxicillin in Neonates and Young Infants. Antimicrob. Agents Chemother. 2019, 63, e02336-18. [Google Scholar] [CrossRef]
- Lonsdale, D.O.; Kipper, K.; Baker, E.H.; Barker, C.I.S.; Oldfield, I.; Philips, B.J.; Johnston, A.; Rhodes, A.; Sharland, M.; Standing, J.F. β-Lactam antimicrobial pharmacokinetics and target attainment in critically ill patients aged 1 day to 90 years: The ABDose study. J. Antimicrob. Chemother. 2020, 75, 3625–3634. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, Y.; Tang, B.; Dong, L.; Li, X.; Zhang, W.; Li, D.; Tian, L.; Anker, J.D.; You, D.; et al. Population Pharmacokinetics and Dosing Optimization of Amoxicillin in Chinese Infants. J. Clin. Pharmacol. 2021, 61, 538–546. [Google Scholar] [CrossRef]
- Rojas, M.X.; Granados, C. Oral antibiotics versus parenteral antibiotics for severe pneumonia in children. Cochrane Database Syst. Rev. 2006, 2006, CD004979. [Google Scholar] [CrossRef]
- Scarborough, M.; Li, H.K.; Rombach, I.; Zambellas, R.; Walker, A.S.; McNally, M.; Atkins, B.; Kümin, M.; Lipsky, B.A.; Hughes, H.; et al. Oral versus intravenous antibiotics for bone and joint infections: The OVIVA non-inferiority RCT. Health Technol. Assess. 2019, 23, 1–92. [Google Scholar] [CrossRef]
- Azamgarhi, T.; Shah, A.; Warren, S. Clinical Experience of Implementing Oral Versus Intravenous Antibiotics (OVIVA) in a Specialist Orthopedic Hospital. Clin. Infect. Dis. 2021, 73, e2582–e2588. [Google Scholar] [CrossRef]
- Keren, R.; Shah, S.S.; Srivastava, R.; Rangel, S.; Bendel-Stenzel, M.; Harik, N.; Hartley, J.; Lopez, M.; Seguias, L.; Tieder, J.; et al. Comparative effectiveness of intravenous vs oral antibiotics for postdischarge treatment of acute osteomyelitis in children. JAMA Pediatr. 2015, 169, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Montini, G.; Toffolo, A.; Zucchetta, P.; Dall’Amico, R.; Gobber, D.; Calderan, A.; Maschio, F.; Pavanello, L.; Molinari, P.P.; Scorrano, D.; et al. Antibiotic treatment for pyelonephritis in children: Multicentre randomised controlled non-inferiority trial. BMJ 2007, 335, 386. [Google Scholar] [CrossRef]
- Yang, S.S.; Tsai, J.D.; Kanematsu, A.; Han, C.H. Asian guidelines for urinary tract infection in children. J. Infect. Chemother. 2021, 27, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- MacGregor, R.R.; Graziani, A.L. Oral administration of antibiotics: A rational alternative to the parenteral route. Clin. Infect. Dis. 1997, 24, 457–467. [Google Scholar] [CrossRef]
- Ruebner, R.; Keren, R.; Coffin, S.; Chu, J.; Horn, D.; Zaoutis, T.E. Complications of central venous catheters used for the treatment of acute hematogenous osteomyelitis. Pediatrics 2006, 117, 1210–1215. [Google Scholar] [CrossRef]
- Kovacich, A.; Tamma, P.D.; Advani, S.; Popoola, V.O.; Colantuoni, E.; Gosey, L.; Milstone, A.M. Peripherally Inserted Central Venous Catheter Complications in Children Receiving Outpatient Parenteral Antibiotic Therapy (OPAT). Infect. Control Hosp. Epidemiol. 2016, 37, 420–424. [Google Scholar] [CrossRef] [PubMed]
- Rangel, S.J.; Anderson, B.R.; Srivastava, R.; Shah, S.S.; Ishimine, P.; Srinivasan, M.; Keren, R. Intravenous Versus Oral Antibiotics for the Prevention of Treatment Failure in Children With Complicated Appendicitis: Has the Abandonment of Peripherally Inserted Catheters Been Justified? Ann. Surg. 2016, 266, 361–368. [Google Scholar] [CrossRef]
- Shah, S.S.; Srivastava, R.; Wu, S.; Colvin, J.D.; Williams, D.J.; Rangel, S.J.; Samady, W.; Rao, S.; Miller, C.; Cross, C.; et al. Intravenous Versus Oral Antibiotics for Postdischarge Treatment of Complicated Pneumonia. Pediatrics 2016, 138, e20161692. [Google Scholar] [CrossRef]
- WHO Regional Office for Europe/European Centre for Disease Prevention and Control. Antimicrobial Resistance Surveillance in Europe 2022–2020 Data; WHO Regional Office for Europe: Copenhagen, Denmark, 2022. [Google Scholar]
- Versporten, A.; Bielicki, J.; Drapier, N.; Sharland, M.; Goossens, H.; ARPEC project group. The Worldwide Antibiotic Resistance and Prescribing in European Children (ARPEC) point prevalence survey: Developing hospital-quality indicators of antibiotic prescribing for children. J. Antimicrob. Chemother. 2016, 71, 1106–1117. [Google Scholar] [CrossRef]
- Available online: https://aware.essentialmeds.org/groups (accessed on 15 March 2023).
- Imburgia, T.A.; Kussin, M.L. A Review of Extended and Continuous Infusion Beta-Lactams in Pediatric Patients. J. Pediatr. Pharmacol. Ther. 2022, 27, 214–227. [Google Scholar] [CrossRef] [PubMed]
- Hurst, A.L.; Baumgartner, C.; MacBrayne, C.E.; Child, J. Experience with Continuous Infusion Vancomycin Dosing in a Large Pediatric Hospital. J. Pediatr. Infect. Dis. Soc. 2019, 8, 174–179. [Google Scholar] [CrossRef]
- Llewelyn, M.J.; Fitzpatrick, J.M.; Darwin, E.; Tonkin-Crine, S.; Gorton, C.; Paul, J.; Peto, T.E.A.; Yardley, L.; Hopkins, S.; Walker, A.S. The antibiotic course has had its day. BMJ 2017, 358, j3418. [Google Scholar] [CrossRef]
- Aricò, M.O. Quando “meno” è abbastanza: La terapia antibiotica breve. Quad. ACP 2022, 295, 197–199. [Google Scholar] [CrossRef]
- Hikmat, S.; Lawrence, J.; Gwee, A. Short Intravenous Antibiotic Courses for Urinary Infections in Young Infants: A Systematic Review. Pediatrics 2022, 149, e2021052466. [Google Scholar] [CrossRef]
- Fox, M.T.; Amoah, J.; Hsu, A.J.; Herzke, C.A.; Gerber, J.S.; Tamma, P.D. Comparative Effectiveness of Antibiotic Treatment Duration in Children with Pyelonephritis. JAMA Netw. Open 2020, 3, e203951. [Google Scholar] [CrossRef]
- Clinical efficacy of 3 days versus 5 days of oral amoxicillin for treatment of childhood pneumonia: A multicentre double-blind trial. Lancet 2002, 360, 835–841. [CrossRef] [PubMed]
- Haider, B.A.; Saeed, M.A.; Bhutta, Z.A. Short-course versus long-course antibiotic therapy for non-severe community-acquired pneumonia in children aged 2 months to 59 months. Cochrane Database Syst. Rev. 2008, CD005976. [Google Scholar] [CrossRef] [PubMed]
- Sutijono, D.; Hom, J.; Zehtabchi, S. Efficacy of 3-day versus 5-day antibiotic therapy for clinically diagnosed nonsevere pneumonia in children from developing countries. Eur. J. Emerg. Med. 2011, 18, 244–250. [Google Scholar] [CrossRef] [PubMed]
- Fursule, A.; Thakur, A.; Garg, P.; Kler, N. Duration of Antibiotic Therapy in Neonatal Gram-negative Bacterial Sepsis-10 Days Versus 14 Days: A Randomized Controlled Trial. Pediatr. Infect. Dis. J. 2022, 41, 156–160. [Google Scholar] [CrossRef] [PubMed]
- Chiappini, E.; Mastrolia, M.V.; Galli, L.; De Martino, M.; Lazzeri, S. Septic arthritis in children in resource limited and non-resource limited countries: An update on diagnosis and treatment. Expert Rev. Anti-Infect. Ther. 2016, 14, 1087–1096. [Google Scholar] [CrossRef] [PubMed]
- Bielicki, J.A.; Stöhr, W.; Barratt, S.; Dunn, D.; Naufal, N.; Roland, D.; Sturgeon, K.; Finn, A.; Rodriguez-Ruiz, J.P.; Malhotra-Kumar, S.; et al. Effect of Amoxicillin Dose and Treatment Duration on the Need for Antibiotic Re-treatment in Children With Community-Acquired Pneumonia: The CAP-IT Randomized Clinical Trial. JAMA 2021, 326, 1713–1724. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.J.; Creech, C.B.; Walter, E.B.; Martin, J.M.; Gerber, J.S.; Newland, J.G.; Howard, L.; Hofto, M.E.; Staat, M.A.; Oler, R.E.; et al. Short- vs Standard-Course Outpatient Antibiotic Therapy for Community-Acquired Pneumonia in Children: The SCOUT-CAP Randomized Clinical Trial. JAMA Pediatr. 2022, 176, 253–261. [Google Scholar] [CrossRef]
| Group | Features | Example |
| Access |
| Amoxicillin, Amoxicillin–clavulanic acid, Cefalexin, Cefazolin, Clindamycin, Cotrimoxazole, Gentamicin; |
| Watch |
| Azithromycin, Cefixime, Ceftriaxone, Vancomycin, Piperacillin–tazobactam, Meropenem |
| Reserve group |
| Ceftazidime–avibactam, Colistin, Daptomycin, Linezolid |
| Step | Key Point | Question |
| Bacterial infection probability | Is antibiotic treatment mandatory for this patient? |
| Clinical re-evaluation after 24–48 h | ||
| Caregivers’ compliance | ||
| Prophylaxis | Clean or clean-contaminated surgery? High-risk patient? | |
| Suspect of invasive bacterial infection | Collect microbiological sample (i.e., blood, urine, swab) |
| Laboratory test | Inflammatory index (WBC, CRP, ESR, PCT) | |
| Local data on AMR | |
| Consider antibiotic with a narrow spectrum | ||
| Avoid combined therapy with more than a single antibiotic molecule, whenever possible | If using more than a single antibiotic is necessary, spectra overlapping should be checked | |
| Appropriate dosage (neither over- nor under-dosed) and schedule of administration | Dosage Number of daily doses | |
| Evaluate if oral administration is possible | Not indicated in sepsis or oral intolerance | |
| Early switch from parenteral to oral route | Switch to oral administration when patient shows clinical and laboratory improvement |
| Optimization of antibiotic treatment upon new microbiological information | Evaluate microorganism susceptibility to antibiotic treatment Consider a narrow spectrum antibiotic Consider to stop multiple antibiotic administration | |
| Avoid prolonging antibiotic treatment to complete “antibiotic course” | Consider treatment interruption if clinical and laboratory resolution |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aricò, M.O.; Valletta, E.; Caselli, D. Appropriate Use of Antibiotic and Principles of Antimicrobial Stewardship in Children. Children 2023, 10, 740. https://doi.org/10.3390/children10040740
Aricò MO, Valletta E, Caselli D. Appropriate Use of Antibiotic and Principles of Antimicrobial Stewardship in Children. Children. 2023; 10(4):740. https://doi.org/10.3390/children10040740
Chicago/Turabian StyleAricò, Melodie O., Enrico Valletta, and Désirée Caselli. 2023. "Appropriate Use of Antibiotic and Principles of Antimicrobial Stewardship in Children" Children 10, no. 4: 740. https://doi.org/10.3390/children10040740
APA StyleAricò, M. O., Valletta, E., & Caselli, D. (2023). Appropriate Use of Antibiotic and Principles of Antimicrobial Stewardship in Children. Children, 10(4), 740. https://doi.org/10.3390/children10040740










