Retrocardiac Pneumomediastinum: Description of an Unusual Case and Review of Literature
Abstract
1. Introduction
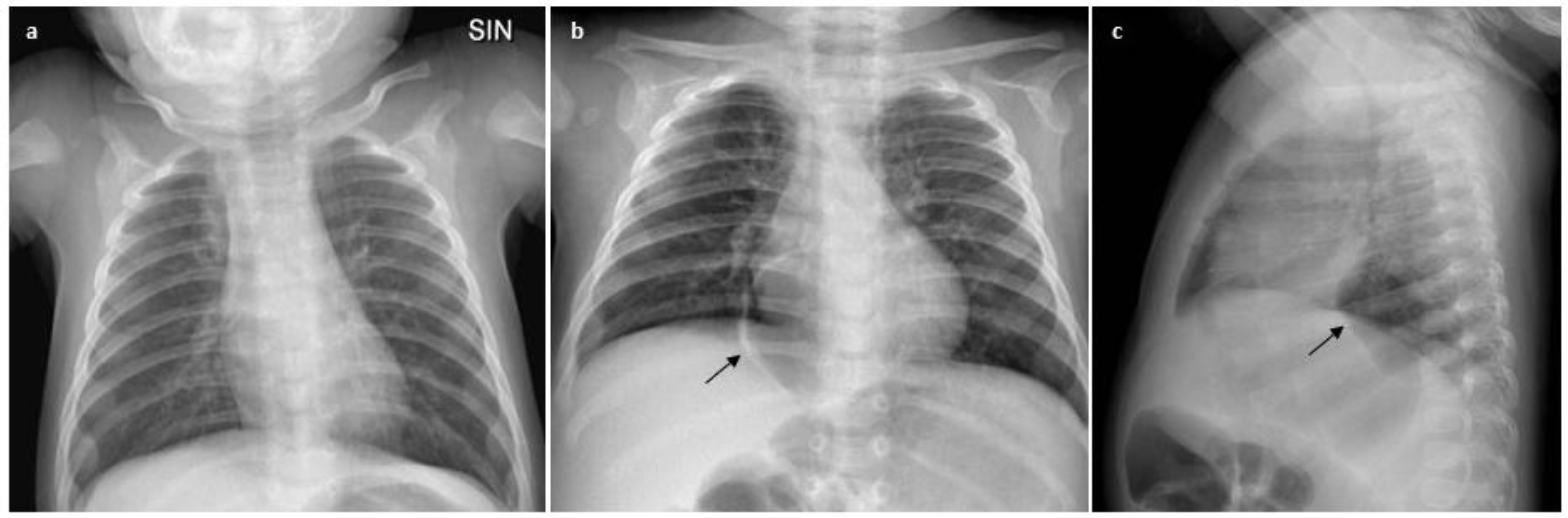
2. Case Description
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Leinert, J.L.; Perez Ortiz, A.; Rafat, N. Spontaneous Pneumomediastinum in Children with Viral Infections: Report of Three Cases Related to Rhinovirus or Respiratory Syncytial Virus Infection. Children 2022, 9, 1040. [Google Scholar] [CrossRef] [PubMed]
- Dixit, A.; Uvaise, M.; Canet-Tarres, A.; Lillie, J. Spontaneous Massive Pneumomediastinum in a Previously Well Infant with COVID-19. Pediatrics 2021, 148, e2021051904. [Google Scholar] [CrossRef] [PubMed]
- Monaco, F.; Barone, M.; Manfredi, V.G.; Marando, R.; Nunnari, F.; David, A.; Monaco, M.; Cascio, A. Pneumomediastinum as a complication of critical pertussis. Clin. Respir. J. 2016, 10, 772–776. [Google Scholar] [CrossRef] [PubMed]
- Kouritas, V.K.; Papagiannopoulos, K.; Lazaridis, G.; Baka, S.; Mpoukovinas, I.; Karavasilis, V.; Lampaki, S.; Kioumis, I.; Pitsiou, G.; Papaiwannou, A.; et al. Pneumomediastinum. J. Thorac. Dis. 2015, 7 (Suppl. S1), S44–S49. [Google Scholar]
- Noorbakhsh, K.A.; Williams, A.E.; Langham, J.J.W.; Wu, L.; Krafty, R.T.; Furtado, A.D.; Zuckerbraun, N.S.; Manole, M.D. Management and Outcomes of Spontaneous Pneumomediastinum in Children. Pediatr. Emerg. Care 2021, 37, e1051–e1056. [Google Scholar] [CrossRef]
- Hassan, H.; Ferguson, L. Spontaneous Pneumomediastinum in a Healthy Pediatric Patient. Cureus 2021, 13, e17847. [Google Scholar] [CrossRef]
- Alahmari, A.K.; Alhelali, A.A.; Alahmari, A.K.; Ahmed, N.J.; Alkathiri, A.A.; Ardi, K.T.; Baali, M.H.; Mubarki, M.H.; Alhamoud, M.A. Pneumothorax/pneumomediastinum as a complication of foreign body inhalation in 3 pediatric patients: A case series. Medicine 2022, 101, e31073. [Google Scholar] [CrossRef]
- Ma, L.; Yin, M.; Yang, X.L.; Xu, W. Risk factors for air leakage during invasive mechanical ventilation in pediatric intensive care units. Eur. J. Med. Res. 2022, 27, 218. [Google Scholar] [CrossRef]
- Hauri-Hohl, A.; Baenziger, O.; Frey, B. Pneumomediastinum in the neonatal and paediatric intensive care unit. Eur. J. Pediatr. 2008, 167, 415–418. [Google Scholar] [CrossRef]
- Cagle, K.J. Pneumomediastinum in the neonate. Neonatal Netw. 2014, 33, 275–282. [Google Scholar] [CrossRef]
- Volberg, F.M.; Everett, C.J.; Brill, P.W. Radiologic features of inferior pulmonary ligament air collections in neonates with respiratory distress. Radiology 1979, 130, 357–360. [Google Scholar] [CrossRef] [PubMed]
- Bowen, A.; Quattromani, F.L. Infraazygous pneumomediastinum in the newborn. AJR Am. J. Roentgenol. 1980, 135, 1017–1021. [Google Scholar] [CrossRef] [PubMed]
- Morrison, S.C.; Fletcher, B.D. Infra-azygous pneumomediastinum versus pulmonary ligament air collection: CT evaluation. Pediatr. Radiol. 1985, 15, 129–130. [Google Scholar] [CrossRef] [PubMed]
- Purohit, D.M.; Lorenzo, R.L.; Smith, C.D.; Bradford, B.F. Bronchial laceration in a newborn with persistent posterior pneumomediastinum. J. Pediatr. Surg. 1985, 20, 82–85. [Google Scholar] [CrossRef]
- Amodio, J.B.; Berdon, W.E.; Abramson, S.J.; Oh, K.S.; Oudjhane, K.; Wung, J.T. Retrocardiac pneumomediastinum in association with tracheal and esophageal perforations. Pediatr. Radiol. 1986, 16, 380–383. [Google Scholar] [CrossRef] [PubMed]
- Rosenfeld, D.L.; Cordell, C.E.; Jadeja, N. Retrocardiac pneumomediastinum: Radiographic finding and clinical implications. Pediatrics 1990, 85, 92–97. [Google Scholar] [CrossRef]
- Pollack, A.; Lask, P.; Kassner, E.G.; Wood, B.P. Radiological case of the month. Retrocardiac pneumomediastinum. Am. J. Dis. Child. 1992, 146, 831–832. [Google Scholar]
- Newman, B.; Oh, K.S. Iatrogenic tracheobronchial perforation in infants. J. Thorac. Imaging. 1994, 9, 269–272. [Google Scholar] [CrossRef]
- Kyle, A.; Veldtman, G.; Stanton, M.; Weeden, D.; Baral, V. Barotrauma-associated posterior tension pneumomediastinum, a rare cause of cardiac tamponade in a ventilated neonate: A case report and review of the literature. Acta Paediatr. 2012, 101, e142–e144. [Google Scholar] [CrossRef]
- Beckstrom, A.C.; Ricca, R.L.; Gow, K.W.; McAdams, R.M. Persistent posterior pneumomediastinum in a neonate. Pediatr. Int. Off. J. Jpn. Pediatr. Soc. 2012, 54, 441–442. [Google Scholar] [CrossRef]
- Ponkowski, M.J.; Yadav, B.; Meagher, E.; Ho, A.; Vellody, R. Minimally invasive treatment of retrocardiac tension pneumomediastinum in an extremely low birth weight infant. Pediatr. Radiol. 2020, 50, 1624–1628. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, D.; Meyer, K.M.; Thompson, A.R.; Speck, K.E. Pediatric pneumomediastinum: Symptom-based management. J. Pediatr. Surg. 2023, 58, 427–431. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.J.; Liao, C.H.; Shen, T.C. Bronchogenic cyst. QJM Mon. J. Assoc. Physicians 2018, 111, 905. [Google Scholar] [CrossRef] [PubMed]
- Hermelijn, S.M.; Elders, B.B.L.J.; Ciet, P.; Wijnen, R.M.H.; Tiddens, H.A.W.M.; Schnater, J.M. A clinical guideline for structured assessment of CT-imaging in congenital lung abnormalities. Paediatr. Respir. Rev. 2021, 37, 80–88. [Google Scholar] [CrossRef]
- Hegde, S.; Prodhan, P. Serious air leak syndrome complicating high-flow nasal cannula therapy: A report of 3 cases. Pediatrics 2013, 131, e939–e944. [Google Scholar] [CrossRef]
- Baudin, F.; Gagnon, S.; Crulli, B.; Proulx, F.; Jouvet, P.; Emeriaud, G. Modalities and Complications Associated with the Use of High-Flow Nasal Cannula: Experience in a Pediatric ICU. Respir. Care 2016, 61, 1305–1310. [Google Scholar] [CrossRef]
- Hishikawa, K.; Goishi, K.; Fujiwara, T.; Kaneshige, M.; Ito, Y.; Sago, H. Pulmonary air leak associated with CPAP at term birth resuscitation. Arch. Dis. Child. Fetal Neonatal Ed. 2015, 100, F382–F387. [Google Scholar] [CrossRef]
- Hung, S.C.; Hsu, H.C.; Chang, S.C. Cerebral air embolism complicating bilevel positive airway pressure therapy. Eur. Respir. J. 1998, 12, 235–237. [Google Scholar] [CrossRef]
- Hazkani, I.; Siong, T.C.; Hill, R.; Dautel, J.; Patel, M.D.; Vaughn, W.; Patzer, R.; Raol, N. The safety of respiratory positive pressure support immediately following pediatric tonsillectomy. Int. J. Pediatr. Otorhinolaryngol. 2023, 167, 111487. [Google Scholar] [CrossRef]


| Authors | N. Preterm/Term Patients | Total Patients’ n. | Primary Cause | Symptoms | Treatment | Outcomes |
|---|---|---|---|---|---|---|
| Volberg, 1979 [11] | 12 pt 2 t | 14 | 14 IMV | RD | n.a. | n.a. |
| Bowen, 1980 [12] | 6 pt | 6 | 6 IMV | RD | n.a. | 2 Dead 3 Alive 1 n.a. |
| Morrison, 1985 [13] | 1 t | 1 | 1 IMV | RD | Conservative | Alive |
| Purohit, 1985 [14] | n.a. | 1 | n.a. | RD | Conservative | Alive |
| Amodio, 1986 [15] | 2 pt | 2 | n.a. | n.a. | n.a. | n.a. |
| Rosenfeld, 1990 [16] | 13 pt 1 t | 14 | 1 IMV 12 HMD 1 HL 1 MA | RD | n.a. | 6 Dead 8 Alive |
| Pollack, 1992 [17] | n.a. | n.a. | n.a. | n.a. | n.a. | n.a. |
| Newman, 1994 [18] | 4 pt | 4 | IMV | RD | 2 Drainage 2 Conservative | 2 Dead 2 Alive |
| Kyle, 2011 [19] | 1 pt | 1 | IMV | RD | Drainage | Alive |
| Beckstrom, 2012 [20] | 1 pt | 1 | IMV | RD | Drainage | Alive |
| Ponkowski, 2020 [21] | 1 pt | 1 | IMV | RD | Drainage | Alive |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Porcaro, F.; Onofri, A.; Allegorico, A.; Tomà, P.; Cutrera, R. Retrocardiac Pneumomediastinum: Description of an Unusual Case and Review of Literature. Children 2023, 10, 649. https://doi.org/10.3390/children10040649
Porcaro F, Onofri A, Allegorico A, Tomà P, Cutrera R. Retrocardiac Pneumomediastinum: Description of an Unusual Case and Review of Literature. Children. 2023; 10(4):649. https://doi.org/10.3390/children10040649
Chicago/Turabian StylePorcaro, Federica, Alessandro Onofri, Annalisa Allegorico, Paolo Tomà, and Renato Cutrera. 2023. "Retrocardiac Pneumomediastinum: Description of an Unusual Case and Review of Literature" Children 10, no. 4: 649. https://doi.org/10.3390/children10040649
APA StylePorcaro, F., Onofri, A., Allegorico, A., Tomà, P., & Cutrera, R. (2023). Retrocardiac Pneumomediastinum: Description of an Unusual Case and Review of Literature. Children, 10(4), 649. https://doi.org/10.3390/children10040649





