Biological Activity Evaluation and In Silico Studies of Polyprenylated Benzophenones from Garcinia celebica
Abstract
:1. Introduction
2. Materials and Methods
2.1. General Experimental Procedures
2.2. Plant Material
2.3. Preparation of Root Bark Extracts
2.4. Extraction and Isolation
2.5. Total Phenolic and Flavonoid Contents
2.5.1. Total Phenolic Content (TPC)
2.5.2. Total Flavonoid Content (TFC)
2.6. Antioxidant Activities Evaluation
2.6.1. DPPH Radical Scavenging Assay
2.6.2. ABTS Radical Scavenging Assay
2.6.3. FRAP Assay
2.7. In Vitro Cytotoxicity Assay
2.8. In Vitro Antiplasmodial Assay
2.9. In Silico Molecular Docking Study and ADMET Prediction
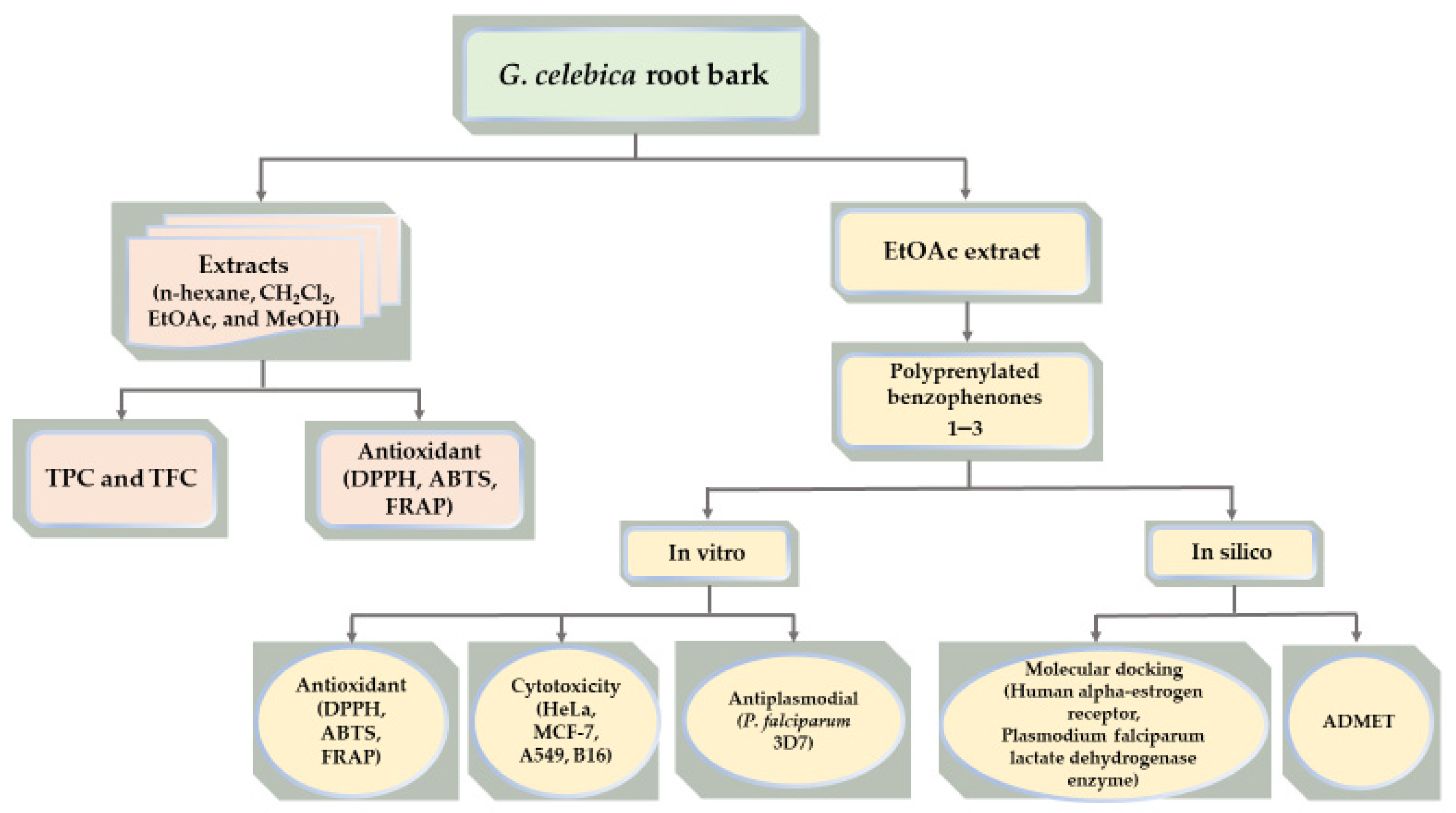
3. Results and Discussion
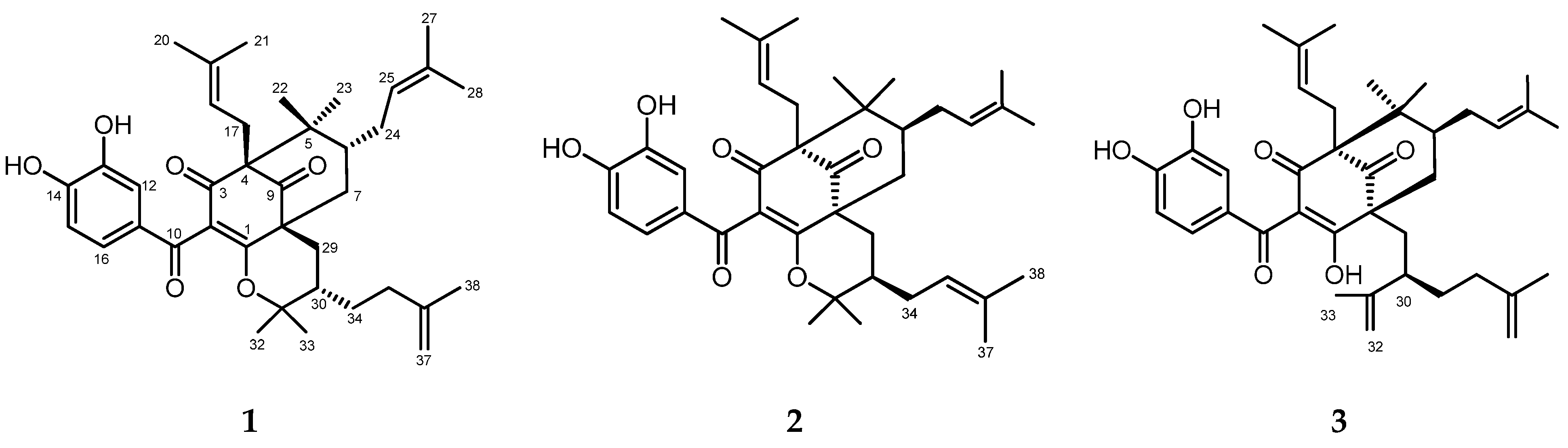
3.1. Identification of the Compounds
3.1.1. (-)-Cycloxanthochymol (1)
3.1.2. Isoxanthochymol (2)
3.1.3. Xanthochymol (3)
3.2. Total Phenolic and Flavonoid Contents of the G. celebica Extracts
3.3. Antioxidant Activity
3.4. In Vitro Cytotoxic Activity
3.5. In Vitro Antiplasmodial Activity
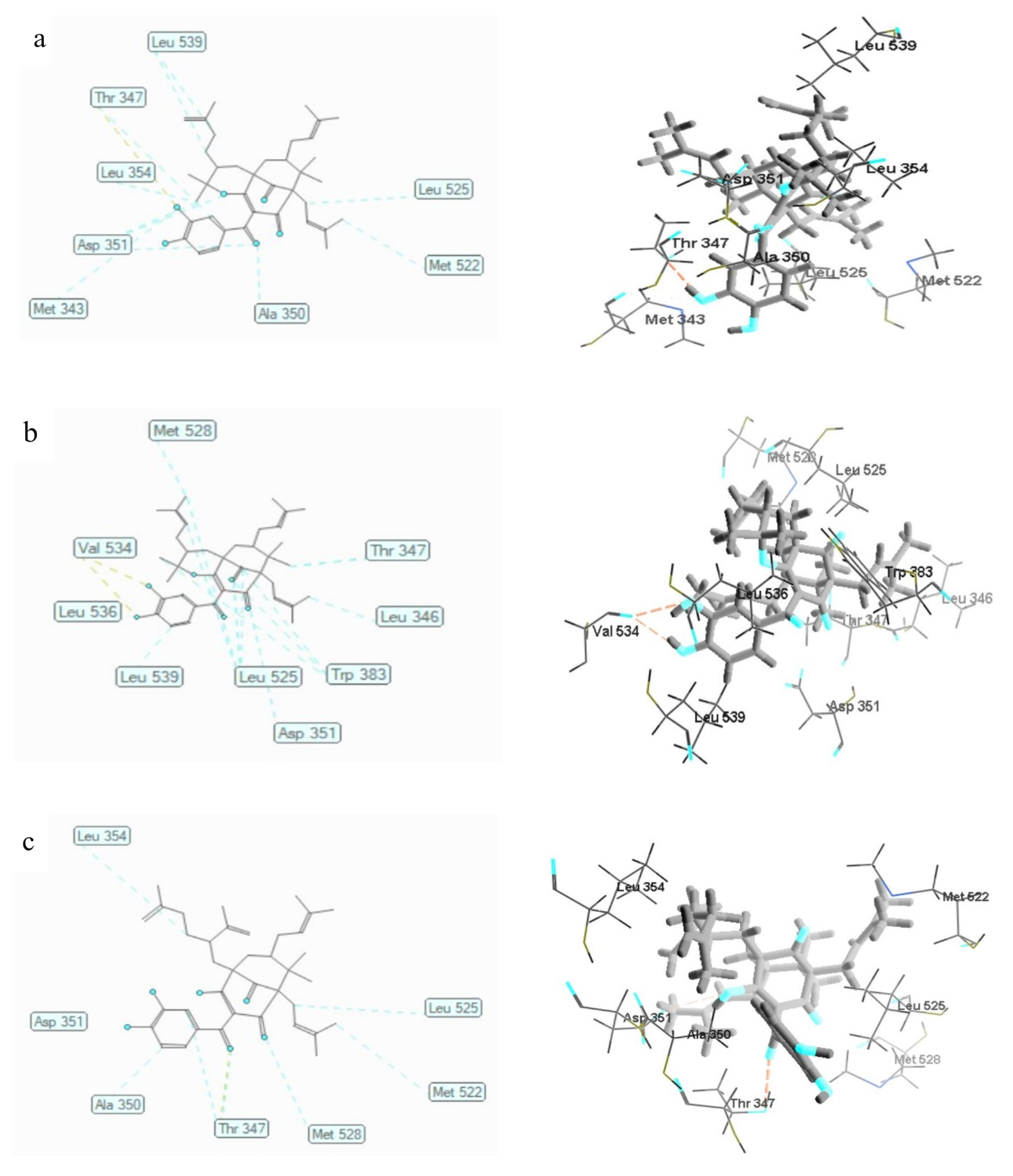
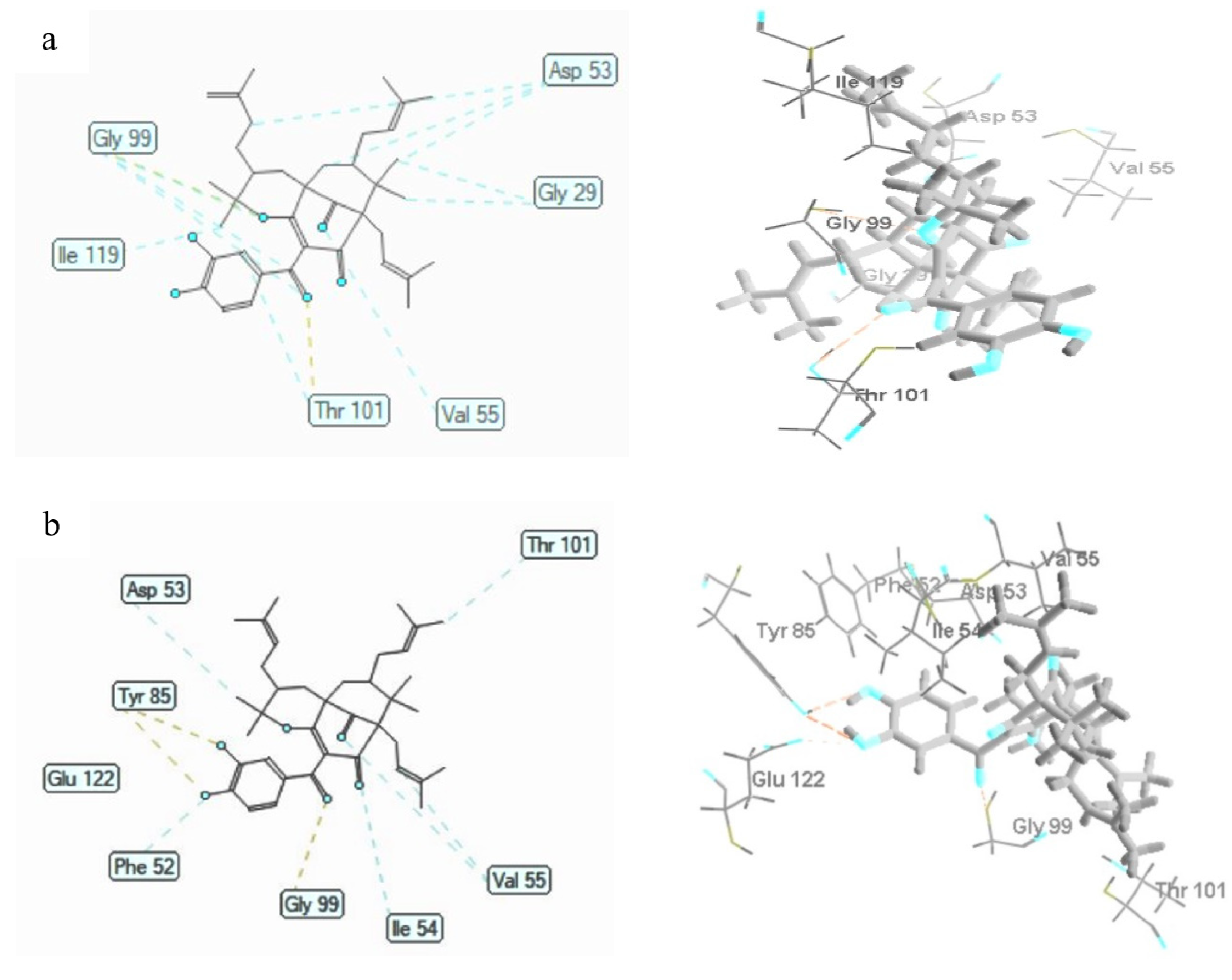
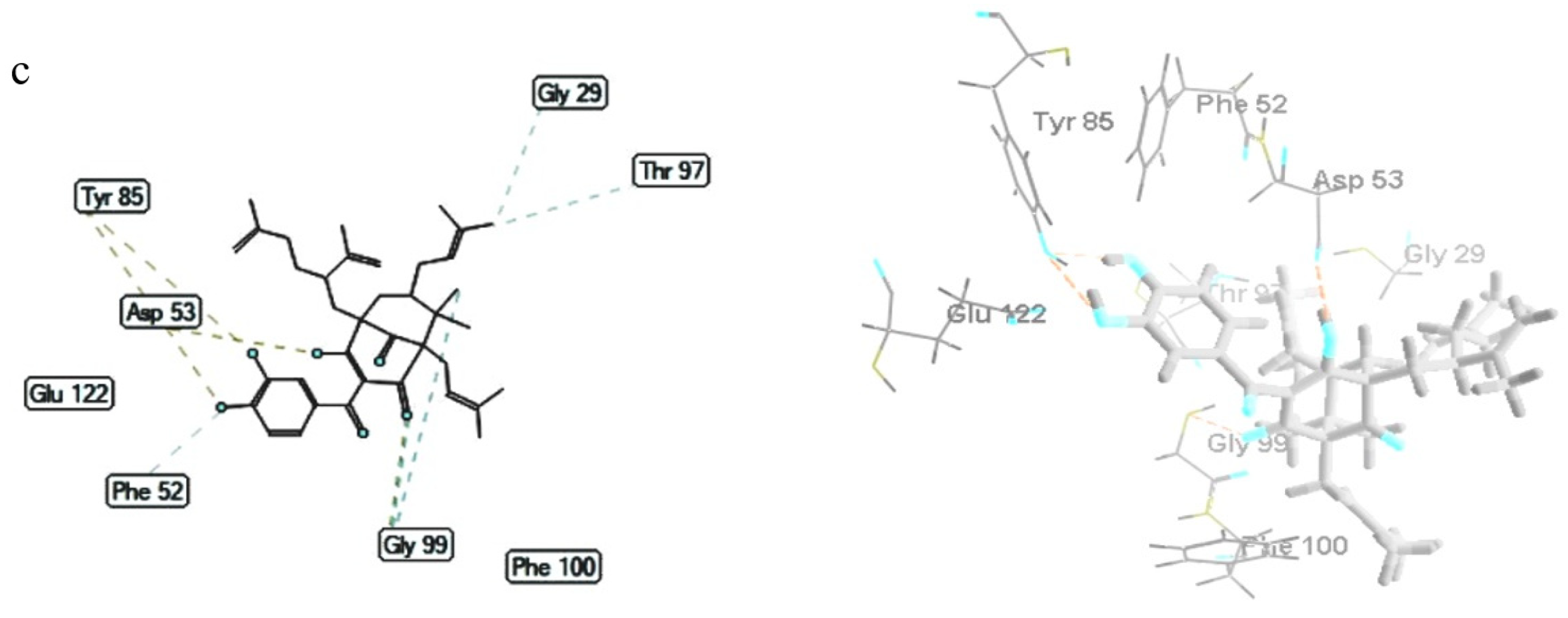
3.6. Molecular Docking Studies
3.6.1. Human Alpha-Estrogen Receptor
3.6.2. Plasmodium falciparum Lactate Dehydrogenase Enzyme
3.6.3. ADMET Profiles
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ABTS | 2,2″-azinobis(3-ethyl benzothiazoline-6-sulfonic acid) |
| ADMET | Absorption Distribution Metabolism Excretion Toxicity |
| DPPH | 1,1-diphenyl-2-picrylhydrazyl |
| FRAP | ferric reducing antioxidant power |
| PDB | protein data bank |
| pkCSM | small-molecule pharmacokinetics prediction |
| TPTZ | 2,4,6-Tris(2-pyridyl)-s-triazine |
References
- World Health Organization. Available online: https://www.who.int/news-room/fact-sheets/detail/malaria (accessed on 5 June 2021).
- Kementerian Kesehatan Republik Indonesia. Available online: https://www.kemkes.go.id/article/view/19020100003/hari-kanker-sedunia-2019.html (accessed on 14 July 2021).
- Fajariyani, S.B. Laporan Situasi Terkini Perkembangan Program Pengendalian Malaria di Indonesia Tahun 2019; Kementerian Kesehatan Republik Indonesia: Jakarta, Indonesia, 2020; pp. 1–4. [Google Scholar]
- Newman, D.J.; Crag, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Srivastava, N.; Singh, A.; Kumari, P.; Nishad, J.H.; Gautam, V.S.; Yadav, M.; Bharti, R.; Kumar, D.; Kharwar, R.N. Advanced in extraction technologies: Isolation and purification of bioactive compounds from biological materials. In Natural Bioactive Compounds, 1st ed.; Sinha, R.P., Hader, D.-P., Eds.; Elsevier: London, UK, 2021; pp. 409–433. [Google Scholar]
- Hay, A.-E.; Aumond, M.-C.; Mallet, S.; Dumontet, V.; Litaudon, M.; Rondeau, D.; Richomme, P. Antioxidant Xanthones from Garcinia vieillardii. J. Nat. Prod. 2004, 67, 707–709. [Google Scholar] [CrossRef]
- Yamaguchi, F.; Ariga, T.; Yoshimura, Y.; Nakazawa, H. Antioxidative and Anti-Glycation Activity of Garcinol from Garcinia indica Fruit Rind. J. Agric. Food Chem. 2000, 48, 180–185. [Google Scholar] [CrossRef] [PubMed]
- Fouotsa, H.; Lannang, A.; Dzoyem, J.; Tatsimo, S.; Neumann, B.; Mbazoa, C.; Razakarivony, A.; Nkengfack, A.; Eloff, J.; Sewald, N. Antibacterial and Antioxidant Xanthones and Benzophenone from Garcinia smeathmannii. Planta Med. 2015, 81, 594–599. [Google Scholar] [CrossRef] [Green Version]
- Liu, C.; Ho, P.C.-L.; Wong, F.C.; Sethi, G.; Wang, L.Z.; Goh, B.C. Garcinol: Current status of its anti-oxidative, anti-inflammatory and anti-cancer effects. Cancer Lett. 2015, 362, 8–14. [Google Scholar] [CrossRef] [PubMed]
- Tan, W.-N.; Khairuddean, M.; Wong, K.-C.; Tong, W.-Y.; Ibrahim, D. Antioxidant compounds from the stem bark of Garcinia atroviridis. J. Asian Nat. Prod. Res. 2016, 18, 804–811. [Google Scholar] [CrossRef] [PubMed]
- Mohamed, G.A.; Ibrahim, S.R.M. New benzophenones and a dihydroflavanonol from Garcinia mangostana pericarps and their antioxidant and cytotoxic activities. Phytochem. Lett. 2020, 39, 43–48. [Google Scholar] [CrossRef]
- Tang, Y.-X.; Fu, W.-W.; Wu, R.; Tan, H.-S.; Shen, Z.-W.; Xu, H.-X. Bioassay-Guided Isolation of Prenylated Xanthone Derivatives from the Leaves of Garcinia oligantha. J. Nat. Prod. 2016, 79, 1752–1761. [Google Scholar] [CrossRef]
- Bui, T.Q.; Bui, A.T.; Nguyen, K.T.; Nguyen, V.T.; Trinh, B.T.D.; Nguyen, L.-H.D. A depsidone and six triterpenoids from the bark of Garcinia celebica. Tetrahedron Lett. 2016, 57, 2524–2529. [Google Scholar] [CrossRef]
- Youn, U.J.; Sripisut, T.; Miklossy, G.; Turkson, J.; Laphookhieo, S.; Chang, L.C. Bioactive polyprenylated benzophenone derivatives from the fruits extracts of Garcinia xanthochymus. Bioorg. Med. Chem. Lett. 2017, 27, 3760–3765. [Google Scholar] [CrossRef]
- Sukandar, E.R.; Kaennakam, S.; Rassamee, K.; Siripong, P.; Fatmawati, S.; Ersam, T.; Tip-pyang, S. Xanthones and biphenyls from the stems of Garcinia cylindrocarpa and their cytotoxicity. Fitoterapia 2018, 130, 112–117. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.-J.; Fu, W.-W.; Wu, R.; Yang, J.-L.; Yao, C.-Y.; Yan, B.-X.; Tan, H.-S.; Zheng, C.-W.; Song, Z.-J.; Xu, H.-X. Bioactive scalemic caged xanthones from the leaves of Garcinia bracteata. Bioorg. Chem. 2019, 82, 274–283. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Fu, W.; Xiang, Q.; Tang, Y.; Wu, R.; Zheng, C.; Lu, Y.; Zhou, H.; Xu, H. Cytotoxic xanthone derivatives from the twigs of Garcinia oligantha. Phytochemistry 2020, 174, 112329. [Google Scholar] [CrossRef]
- Hatami, E.; Jaggi, M.; Chauhan, S.C.; Yallapu, M.M. Gambogic acid: A shining natural compound to nanomedicine for cancer therapeutics. Biochim. Biophys. Acta BBA Rev. Cancer 2020, 1874, 188381. [Google Scholar] [CrossRef]
- Protiva, P.; Hopkins, M.E.; Baggett, S.; Yang, H.; Lipkin, M.; Holt, P.R.; Kennelly, E.J.; Bernard, W.I. Growth inhibition of colon cancer cells by polyisoprenylated benzophenones is associated with induction of the endoplasmic reticulum response: Garcinia xanthochymus Benzophenones Induce Stress. Int. J. Cancer 2008, 123, 687–694. [Google Scholar] [CrossRef]
- Sukandar, E.R.; Kaennakam, S.; Aree, T.; Nöst, X.; Rassamee, K.; Bauer, R.; Siripong, P.; Ersam, T.; Tip-pyang, S. Picrorhizones A–H, Polyprenylated Benzoylphloroglucinols from the Stem Bark of Garcinia picrorhiza. J. Nat. Prod. 2020, 83, 2102–2111. [Google Scholar] [CrossRef]
- Auranwiwat, C.; Laphookhieo, S.; Rattanajak, R.; Kamchonwongpaisan, S.; Pyne, S.G.; Ritthiwigrom, T. Antimalarial polyoxygenated and prenylated xanthones from the leaves and branches of Garcinia mckeaniana. Tetrahedron 2016, 72, 6837–6842. [Google Scholar] [CrossRef] [Green Version]
- Lyles, J.; Negrin, A.; Khan, S.; He, K.; Kennelly, E. In Vitro Antiplasmodial Activity of Benzophenones and Xanthones from Edible Fruits of Garcinia Species. Planta Med. 2014, 80, 676–681. [Google Scholar] [CrossRef] [Green Version]
- Wairata, J.; Sukandar, E.R.; Fadlan, A.; Purnomo, A.S.; Taher, M.; Ersam, T. Evaluation of the antioxidant, antidiabetic, and antiplasmodial activities of xanthones 2 isolated from Garcinia forbesii and their in silico studies. Biomedicines 2021, 9, 1380. [Google Scholar] [CrossRef]
- Elfita, E.; Muharni, M.; Latief, M.; Darwati, D.; Widiyantoro, A.; Supriyatna, S.; Bahti, H.H.; Dachriyanus, D.; Cos, P.; Maes, L. Antiplasmodial and other constituents from four Indonesian Garcinia spp. Phytochemistry 2009, 70, 907–912. [Google Scholar] [CrossRef]
- Subarnas, A.; Diantini, A.; Abdulah, R.; Zuhrotun, A.; Nugraha, P.A.; Hadisaputri, Y.E.; Puspitasari, I.M.; Yamazaki, C.; Kuwano, H.; Koyama, H. Apoptosis-mediated antiproliferative activity of friedolanostane triterpenoid isolated from the leaves of Garcinia celebica against MCF-7 human breast cancer cell lines. Biomed. Rep. 2016, 4, 79–82. [Google Scholar] [CrossRef] [Green Version]
- Armania, N.; Yazan, L.S.; Musa, S.N.; Ismail, I.S.; Foo, J.B.; Chan, K.W.; Noreen, H.; Hisyam, A.H.; Zulfahmi, S.; Ismail, M. Dillenia suffruticosa exhibited antioxidant and cytotoxic activity through induction of apoptosis and G2/M cell cycle arrest. J. Ethnopharmacol. 2013, 146, 525–535. [Google Scholar] [CrossRef]
- Abubakar, A.R.; Haque, M. Preparation of Medicinal Plants: Basic Extraction and Fractionation Procedures for Experimental Purposes. J. Pharm. Bioallied Sci. 2020, 12, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Brighente, I.M.C.; Dias, M.; Verdi, L.G.; Pizzolatti, M.G. Antioxidant Activity and Total Phenolic Content of Some Brazilian Species. Pharm. Biol. 2007, 45, 156–161. [Google Scholar] [CrossRef]
- Dudonné, S.; Vitrac, X.; Coutière, P.; Woillez, M.; Mérillon, J.-M. Comparative Study of Antioxidant Properties and Total Phenolic Content of 30 Plant Extracts of Industrial Interest Using DPPH, ABTS, FRAP, SOD, and ORAC Assays. J. Agric. Food Chem. 2009, 57, 1768–1774. [Google Scholar] [CrossRef]
- Lulan, T.Y.K.; Fatmawati, S.; Santoso, M.; Ersam, T. α-VINIFERIN as a potential antidiabetic and antiplasmodial extracted from Dipterocarpus littoralis. Heliyon 2020, 6, e04102. [Google Scholar] [CrossRef]
- Benzie, I.F.F.; Devaki, M. The ferric reducing/antioxidant power (FRAP) assay for non-enzymatic antioxidant capacity: Concepts, procedures, limitations and applications. In Measurement of Antioxidant Activity & Capacity, 1st ed.; Apak, R., Capanoglu, E., Shahidi, F., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2017; pp. 77–106. [Google Scholar] [CrossRef]
- Nurlelasari, R.I.; Salam, S.; Safari, A.; Harneti, D.; Maharani, R.; Hidayat, A.T.; Tanjung, M.; Retnowati, R.; Shiono, Y.; Supratman, U. A new havanensin-type limonoid from Chisocheton macrophyllus. Appl. Biol. Chem. 2021, 64, 35. [Google Scholar] [CrossRef]
- Wang, X.; Miyazaki, Y.; Inaoka, D.K.; Hartuti, E.D.; Watanabe, Y.-I.; Shiba, T.; Harada, S.; Saimoto, H.; Burrows, J.N.; Benito, F.J.G.; et al. Identification of Plasmodium falciparum Mitochondrial Malate: Quinone Oxidoreductase Inhibitors from the Pathogen Box. Genes 2019, 10, 471. [Google Scholar] [CrossRef] [Green Version]
- Zhang, L.-J.; Chiou, C.-T.; Cheng, J.-J.; Huang, H.-C.; Kuo, L.-M.Y.; Liao, C.-C.; Bastow, K.F.; Lee, K.-H.; Kuo, Y.-H. Cytotoxic Polyisoprenyl Benzophenonoids from Garcinia subelliptica. J. Nat. Prod. 2010, 73, 557–562. [Google Scholar] [CrossRef] [Green Version]
- Iinuma, M.; Tosa, H.; Tanaka, T.; Kanamaru, S.; Asai, F.; Kobayashi, Y.; Miyauchi, K.; Shimano, R. Antibacterial Activity of Some Garcinia Benzophenone Derivatives against Methicilin-Resistant Staphylococcus aureus. Biol. Pharm. Bull. 1996, 19, 311–314. [Google Scholar] [CrossRef] [Green Version]
- Baggett, S.; Mazzola, E.P.; Kennelly, E.J. The benzophenones: Isolation, structural elucidation and biological activities. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2005; pp. 721–771. [Google Scholar] [CrossRef]
- Ramirez, C.; Gil, J.H.; Marín-Loaiza, J.C.; Rojano, B.; Durango, D. Chemical constituents and antioxidant activity of Garcinia madruno (Kunth) Hammel. J. King Saud. Univ. Sci. 2019, 31, 1283–1289. [Google Scholar] [CrossRef]
- Menon, L.N.; Satheesh, S.K.K.; Panicker, S.P.; Rameshkumar, K.B. Antiproliferative activity of caged xanthones from the leaves of Garcinia wightii T. Anderson. Fitoterapia 2020, 143, 104592. [Google Scholar] [CrossRef] [PubMed]
- Baggett, S.; Protiva, P.; Mazzola, E.P.; Yang, H.; Ressler, E.T.; Basile, M.J.; Weinstein, I.B.; Kennely, E.J. Bioactive Benzophenones from Garcinia xanthochymus Fruits. J. Nat. Prod. 2005, 68, 354–360. [Google Scholar] [CrossRef] [PubMed]
- Floegel, A.; Kim, D.-O.; Chung, S.-J.; Koo, S.I.; Chun, O.K. Comparison of ABTS/DPPH assays to measure antioxidant capacity in popular antioxidant-rich US foods. J. Food Compos. Anal. 2011, 24, 1043–1048. [Google Scholar] [CrossRef]
- Kim, D.-O.; Lee, K.W.; Lee, H.J.; Lee, C.Y. Vitamin C Equivalent Antioxidant Capacity (VCEAC) of Phenolic Phytochemicals. J. Agric. Food Chem. 2002, 50, 3713–3717. [Google Scholar] [CrossRef]
- Liang, N.; Kitts, D. Antioxidant Property of Coffee Components: Assessment of Methods that Define Mechanisms of Action. Molecules 2014, 19, 19180–19208. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Essien, E.E.; Thomas, P.S.; Ekanem, I.R.; Choudhary, M.I. Isolation and characterization of 5-hydroxymethylfurfural, antiglycation, antihyperglycaemic, antioxidant, and cytotoxic effects of Garcinia kola Heckel roots extract and fractions. S. Afr. J. Bot. 2021, 140, 62–67. [Google Scholar] [CrossRef]
- Prasad, K.N.; Chew, L.Y.; Khoo, H.E.; Kong, K.W.; Azlan, A.; Ismail, A. Antioxidant Capacities of Peel, Pulp, and Seed Fractions of Canarium odontophyllum Miq. Fruit. J. Biomed. Biotechnol. 2010, 2010, 871379. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, X.; Tian, D.; Fu, Y.; Li, Y.; Huang, L.; Gu, W.; Song, J.; Li, Y.; Ben-David, Y.; Wen, M.; et al. Synthesis of novel guttiferone E and xanthochymol derivatives with cytotoxicities by inducing cell apoptosis and arresting the cell cycle phase. Eur. J. Med. Chem. 2019, 162, 765–780. [Google Scholar] [CrossRef]
- Matsumoto, K.; Akao, Y.; Kobayashi, E.; Ito, T.; Ohguchi, K.; Tanaka, T.; Iinuma, M.; Nozawa, Y. Cytotoxic Benzophenone Derivatives from Garcinia Species Display a Strong Apoptosis-Inducing Effect against Human Leukemia Cell Lines. Biol. Pharm. Bull. 2003, 26, 569–571. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, S.; Chattopadhyay, S.; Darokar, M.; Garg, A.; Khanuja, S. Cytotoxic Activities of Xanthochymol and Isoxanthochymol Substantiated by LC-MS/MS. Planta Med. 2007, 73, 1452–1456. [Google Scholar] [CrossRef]
- Kuete, V.; Tchakam, P.D.; Wiench, B.; Ngameni, B.; Wabo, H.K.; Tala, M.F.; Moungang, M.L.; Ngadjui, B.T.; Murayama, T.; Efferth, T. Cytotoxicity and modes of action of four naturally occuring benzophenones: 2,2′,5,6′-Tetrahydroxybenzophenone, guttiferone E, isogarcinol and isoxanthochymol. Phytomedicine 2013, 20, 528–536. [Google Scholar] [CrossRef]
- Wu, S.-B.; Long, C.; Kennelly, E.J. Structural diversity and bioactivities of natural benzophenones. Nat. Prod. Rep. 2014, 31, 1158–1174. [Google Scholar] [CrossRef]
- Phang, Y.; Wang, X.; Lu, Y.; Fu, W.; Zheng, C.; Xu, H. Bicyclic polyprenylated acylphloroglucinols and their derivatives: Structural modification, structure-activity relationship, biological activity and mechanism of action. Eur. J. Med. Chem. 2020, 205, 112646. [Google Scholar] [CrossRef]
- Lannang, A.; Louh, G.; Biloa, B.; Komguem, J.; Mbazoa, C.; Sondengam, B.; Naesens, L.; Pannecouque, C.; De Clercq, E.; Sayed El Ashry, E. Cytotoxicity of Natural Compounds Isolated from the Seeds of Garcinia afzelii. Planta Med. 2010, 76, 708–712. [Google Scholar] [CrossRef]
- Mathew, S.; Abraham, T.E.; Zakaria, Z.A. Reactivity of phenolic compounds towards free radicals under in vitro conditions. J. Food Sci. Technol. 2015, 52, 5790–5798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saha Roy, S.; Vadlamudi, R.K. Role of Estrogen Receptor Signaling in Breast Cancer Metastasis. Int. J. Breast Cancer 2012, 2012, 654698. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hayes, E.L.; Lewis-Wambi, J.S. Mechanisms of endocrine resistance in breast cancer: An overview of the proposed roles of noncoding RNA. Breast Cancer Res. 2015, 17, 40. [Google Scholar] [CrossRef] [Green Version]
- Hua, H.; Zhang, H.; Kong, Q.; Jiang, Y. Mechanisms for estrogen receptor expression in human cancer. Exp. Hematol. Oncol. 2018, 7, 24. [Google Scholar] [CrossRef] [Green Version]
- Srivastava, V.; Yadav, A.; Sarkar, P. Molecular docking and ADMET study of bioactive compounds of Glycyrrhiza glabra against main protease of SARS-CoV2. Mater. Today Proc. 2020. [Google Scholar] [CrossRef]
- Pires, D.E.V.; Blundell, T.L.; Ascher, D.B. pkCSM: Predicting Small-Molecule Pharmacokinetic and Toxicity Properties Using Graph-Based Signatures. J. Med. Chem. 2015, 58, 4066–4072. [Google Scholar] [CrossRef]
- Da Paixão, V.G.; da Rocha Pita, S.S. In silico identification and evaluation of new Trypanosoma cruzi trypanothione reductase (TcTR) inhibitors obtained from natural products database of the Bahia semi-arid region (NatProDB). Comput. Biol. Chem. 2019, 79, 36–47. [Google Scholar] [CrossRef] [PubMed]
| Position | 1 (CD3OD) | 2 (CD3OD) | 3 (CD3OD) | |||
|---|---|---|---|---|---|---|
| δC | δH (J in Hz) | δC | δH (J in Hz) | δC | δH (J in Hz) | |
| 1 | 173.7 | 172.9 | 195.8 b | |||
| 2 | 126.8 | 128.0 | 117.9 | |||
| 3 | 196.4 | 196.0 | - c | |||
| 4 | 69.5 | 71.7 | - c | |||
| 5 | 47.1 | 46.9 | 50.2 | |||
| 6 | 47.4 | 1.52, m | 42.4 | 1.98, m | 47.9 | 1.50 a |
| 7 | 39.7 | 2.27, m | 43.3 | 2.17, dd (14.1, 4.5) | 43.7 | 2.05, m |
| 2.04, m | 1.56, t (13.0) | 2.05, m | ||||
| 8 | 52.9 | 54.2 | - c | |||
| 9 | 207.9 | 207.2 | 210.6 | |||
| 10 | 194.2 | 195.1 | 195.8 | |||
| 11 | 131.1 | 130.7 | 129.5 | |||
| 12 | 115.9 | 7.28, d (2.1) | 116.3 | 7.29, d (2.1) | 117.3 | 7.19, d (2.2) |
| 13 | 146.7 | 146.7 | 146.3 | |||
| 14 | 152.6 | 152.8 | 152.5 | |||
| 15 | 115.4 | 6.70, d (8.0) | 115.6 | 6.75, d (8.3) | 115.1 | 6.71, d (8.3) |
| 16 | 124.6 | 6.95, dd (8.0, 2.1) | 124.8 | 7.06, dd (8.2, 2.1) | 125.2 | 6.98, dd (8.3, 2.2) |
| 17 | 26.6 | 2.46, dd (13.5, 6.0) | 25.8 | 2.44, dd (13.8, 5.8) | 27.0 | 2.71, dd (13.30, 9.45) |
| 2.64, dd (14.0, 6.0) | 2.63, dd (13.6, 8.0) | 2.57, m | ||||
| 18 | 121.2 | 4.91 a | 121.4 | 4.86 a | 121.4 | 5.05, m |
| 19 | 135.5 | 135.1 | 135.8 | |||
| 20 | 26.6 | 1.59, s | 26.4 | 1.61, s | 26.5 | 1.74, s |
| 21 | 18.3 | 1.59, s | 18.3 | 1.60, s | 18.3 | 1.69, s |
| 22 | 22.9 | 1.15, s | 22.6 | 1.09, s | 23.2 | 1.15, s |
| 23 | 27.1 | 0.99, s | 16.3 | 0.77, s | 27.3 | 1.00, s |
| 24 | 30.5 | 2.15, m | 30.6 | 1.85, m | 30.3 | 2.05, m |
| 2.67, m | 2.07, m | 2.05, m | ||||
| 25 | 126.3 | 4.92 a | 122.9 | 5.21, t | 125.6 | 4.90 a |
| 26 | 134.0 | 134.7 | 133.6 | |||
| 27 | 26.1 | 1.69, s | 26.0 | 1.79, s | 26.0 | 1.66, s |
| 28 | 18.6 | 1.67, s | 18.1 | 1.65, s | 18.2 | 1.50, s |
| 29 | 28.8 | 1.07, br d (13.5) | 28.5 | 1.04, t (13.6) | 37.7 | 1.90, m |
| 3.09, dd (14.0, 3.4) | 3.03, dd (14.2, 3.65) | 1.90, m | ||||
| 30 | 42.9 | 1.33, m | 44.6 | 1.40, tt (13.5) | 44.7 | 2.57, m |
| 31 | 88.5 | 88.5 | 148.9 | |||
| 32 | 28.8 | 0.84, s | 21.5 | 1.24, s | 113.6 | 4.50, br s |
| 33 | 21.5 | 1.24, s | 29.0 | 0.98, s | 17.8 | 1.59, s |
| 34 | 29.3 | 1.15 a | 28.7 | 1.79 a | 32.7 | 1.50 a |
| 1.52, m | 2.23, d (14.5) | 1.42, m | ||||
| 35 | 36.2 | 2.15, m | 123.9 | 5.16, t | 36.8 | 1.83, m |
| 2.27, m | 1.83, m | |||||
| 36 | 146.0 | 134.2 | 146.9 | |||
| 37 | 111.9 | 4.79, s | 26.0 | 1.73, s | 110.5 | 4.64, d (7.3) |
| 38 | 22.3 | 1.73, s | 18.1 | 1.62, s | 22.8 | 1.69, s |
| Extract | TPC (mg GAE/g Extract) | TFC (mg QE/g Extract) |
|---|---|---|
| n-Hexane | 188.00 ± 1.78 | 33.07 ± 0.76 |
| CH2Cl2 | 164.35 ± 1.19 | 31.03 ± 2.28 |
| EtOAc | 180.06 ± 1.07 | 35.73 ± 0.13 |
| MeOH | 112.84 ± 1.89 | 27.11 ± 0.24 |
| Extracts/Compounds | Antioxidant Capacity | ||||
|---|---|---|---|---|---|
| DPPH | ABTS | FRAP (μM Fe2+/g) | |||
| IC50 (μg/mL) | IC50 (μM) | IC50 (μg/mL) | IC50 (μM) | ||
| n-Hexane | 7.86 ± 0.07 | - | 5.96 ± 0.00 | - | 35.64 ± 1.56 |
| CH2Cl2 | 7.45 ± 0.04 | - | 6.43 ± 0.02 | - | 34.58 ± 1.67 |
| EtOAc | 7.55 ± 0.06 | - | 6.01 ± 0.01 | - | 66.36 ± 5.05 |
| MeOH | 11.40 ± 0.00 | - | 7.63 ± 0.01 | - | 38.75 ± 2.41 |
| 1 | 3.36 ± 0.03 | 5.58 ± 0.05 | 0.78 ± 0.02 | 1.30 ± 0.04 | 22.61 ± 0.06 |
| 2 | 2.98 ± 0.05 | 4.95 ± 0.08 | 0.70 ± 0.02 | 1.16 ± 0.04 | 24.58 ± 0.29 |
| 3 | 2.33 ± 0.06 | 3.87 ± 0.10 | 0.53 ± 0.01 | 0.88 ± 0.02 | 44.28 ± 0.79 |
| Gallic acid | 0.55 ± 0.00 | 3.22 ± 0.02 | 0.12 ± 0.00 | 0.68 ± 0.00 | Nt |
| Quercetin | 1.34 ± 0.02 | 4.43 ± 0.06 | 0.06 ± 0.00 | 0.19 ± 0.00 | Nt |
| Ascorbic acid | Nt | Nt | 30.62 ± 0.27 | ||
| Compounds | IC50 (μM) | |||
|---|---|---|---|---|
| HeLa | MCF-7 | A549 | B16 | |
| 1 | 13.77 ± 0.07 | 13.54 ± 0.01 | 13.77 ± 0.20 | 12.46 ± 0.08 |
| 2 | 10.18 ± 0.51 | 10.73 ± 0.14 | 13.99 ± 0.09 | 12.39 ± 0.11 |
| 3 | 15.48 ± 0.13 | 14.93 ± 0.52 | 16.10 + 0.18 | 16.94 ± 0.15 |
| Cisplatin | 19 | 53 | 18 | 43 |
| Compound | % Inhibition ± SD (10 μg/mL) | IC50 ± SD (μM) |
|---|---|---|
| 1 | <10 | Nt |
| 2 | 83.97 ± 0.47 | 2.99 ± 0.20 |
| 3 | <10 | Nt |
| chloroquine | 98.8 ± 0.25 | 0.006 ± 0.01 |
| Ligands | MolDock Score (kcal/mol) | Hydrogen Bonding | Steric Interaction | Electrostatic Interaction |
|---|---|---|---|---|
| 1 | −144.638 ± 1.41 | Thr347 | Asp351 (3); Leu539 (2); Ala350; Met343; Leu354; Thr347; Leu525; Met522 | |
| 2 | −139.590 ± 0.37 | Val534 (2) | Leu525 (4); Trp383 (4); Thr347; Leu346; Asp351; Leu539; Leu536; Met528 | |
| 3 | −162.832 ± 2.24 | Thr347 | Thr347 (2); Ala350; Asp351; Leu354; Leu525; Met522; Met528 | |
| 4-hydroxtamoxifen | −165.042 ± 0,48 | Arg394; Glu353 | Ala350 | Asp351 |
| Ligands | MolDock Score (kcal/mol) | Hydrogen Bonding | Steric Interaction |
|---|---|---|---|
| 1 | −154.72 ± 0.48 | Gly99; Thr101 | Gly29 (3); Gly99 (2); Asp53 (3); Val55; Ile119, Thr101 |
| 2 | −147.43 ± 1.16 | Tyr85 (2); Gly99; Glu122 | Ile54 (3); Asp53; Thr101; Val55 (2), Phe52 |
| 3 | −145.56 ± 0.84 | Tyr85 (2); Gly99; Asp53 | Gly29; Gly99 (2); Thr97; Phe52 |
| Chloroquine | −108.99 ± 0.48 | Gly29; Gly99; Ser28 | Gly29; Gly99; Ala98; Asp53 |
| Ligands | Absorption | Distribution | Metabolism | Excretion | Toxicity | |||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Caco-2 Permeability | Intestinal Absorption (Human) | Skin Permeability | VDss (Human) | BBB Permeability | CYP2D6 Inhibitor | CYP3A4 Inhibitor | Total Clearance | Renal OCT2 Substrate | Oral Rat Acute Toxicity (LD50) | Oral Rat Chronic Toxicity (LOAEL) | Hepatotoxicity | |
| 1 | 1.54 | 100.00 | −2.74 | −0.16 | −0.51 | No | No | 0.04 | No | 3.36 | 1.74 | No |
| 2 | 1.26 | 92.65 | −2.73 | −0.46 | −1.39 | No | No | 0.29 | No | 3.80 | 1.72 | No |
| 3 | 1.60 | 100.00 | −2.75 | −0.19 | −0.48 | No | No | −0.02 | No | 3.37 | 1.75 | No |
| 4−Hidroxytamoxifen | 1.03 | 93.54 | −2.74 | 0.31 | −0.29 | Yes | No | 0.59 | No | 2.18 | 1.37 | No |
| Chloroquine | 1.259 | 89.44 | −2.564 | 1.757 | 0.410 | Yes | No | 0.993 | Yes | 2.888 | 0.423 | Yes |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pasaribu, Y.P.; Fadlan, A.; Fatmawati, S.; Ersam, T. Biological Activity Evaluation and In Silico Studies of Polyprenylated Benzophenones from Garcinia celebica. Biomedicines 2021, 9, 1654. https://doi.org/10.3390/biomedicines9111654
Pasaribu YP, Fadlan A, Fatmawati S, Ersam T. Biological Activity Evaluation and In Silico Studies of Polyprenylated Benzophenones from Garcinia celebica. Biomedicines. 2021; 9(11):1654. https://doi.org/10.3390/biomedicines9111654
Chicago/Turabian StylePasaribu, Yenni Pintauli, Arif Fadlan, Sri Fatmawati, and Taslim Ersam. 2021. "Biological Activity Evaluation and In Silico Studies of Polyprenylated Benzophenones from Garcinia celebica" Biomedicines 9, no. 11: 1654. https://doi.org/10.3390/biomedicines9111654
APA StylePasaribu, Y. P., Fadlan, A., Fatmawati, S., & Ersam, T. (2021). Biological Activity Evaluation and In Silico Studies of Polyprenylated Benzophenones from Garcinia celebica. Biomedicines, 9(11), 1654. https://doi.org/10.3390/biomedicines9111654






