Development of a Cost-Effective Sensing Platform for Monitoring Phosphate in Natural Waters
Abstract
:1. Introduction
2. Materials and Methods
2.1. Reagents and Standard Preparation
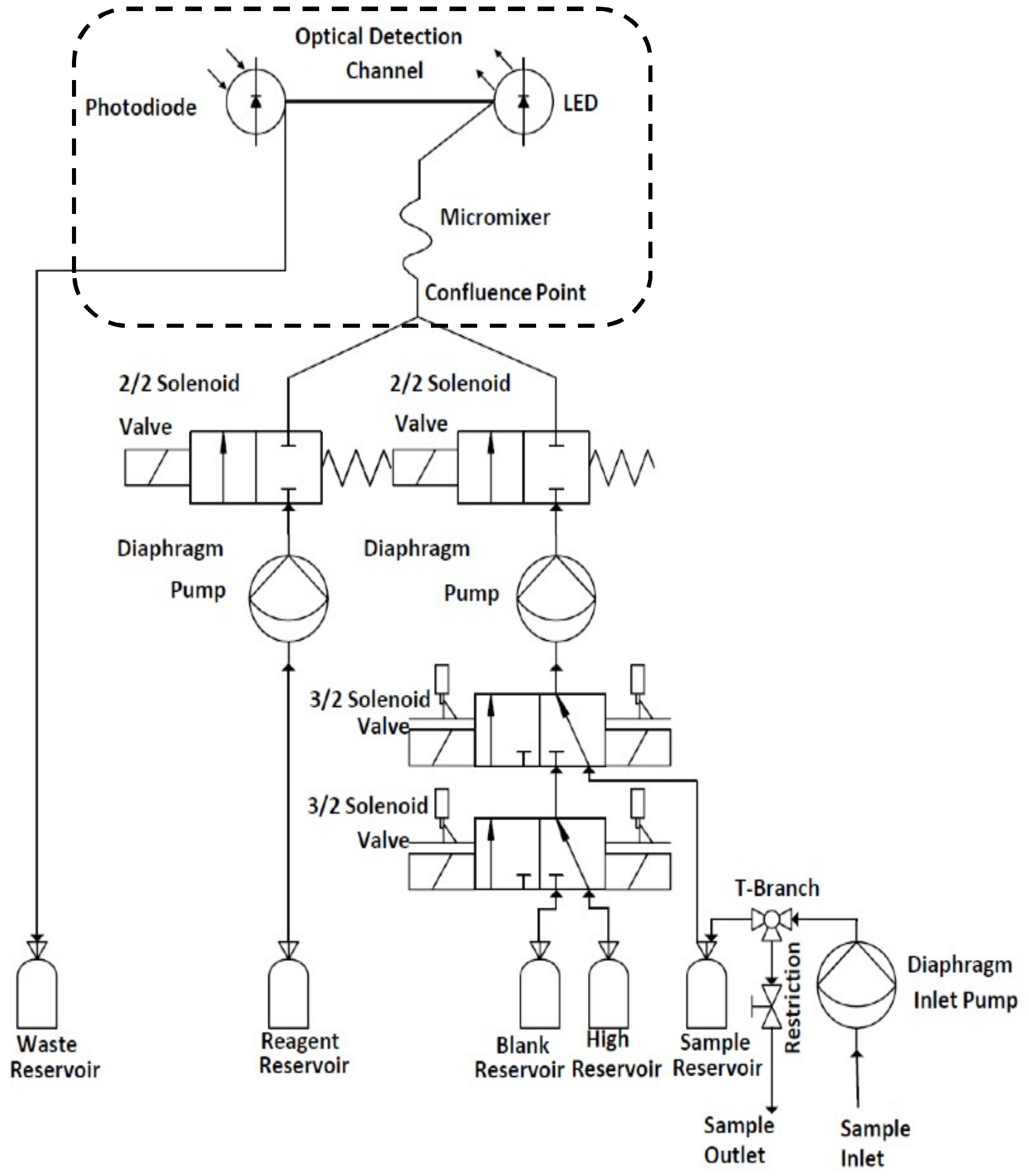
2.2. Fluidic Handling
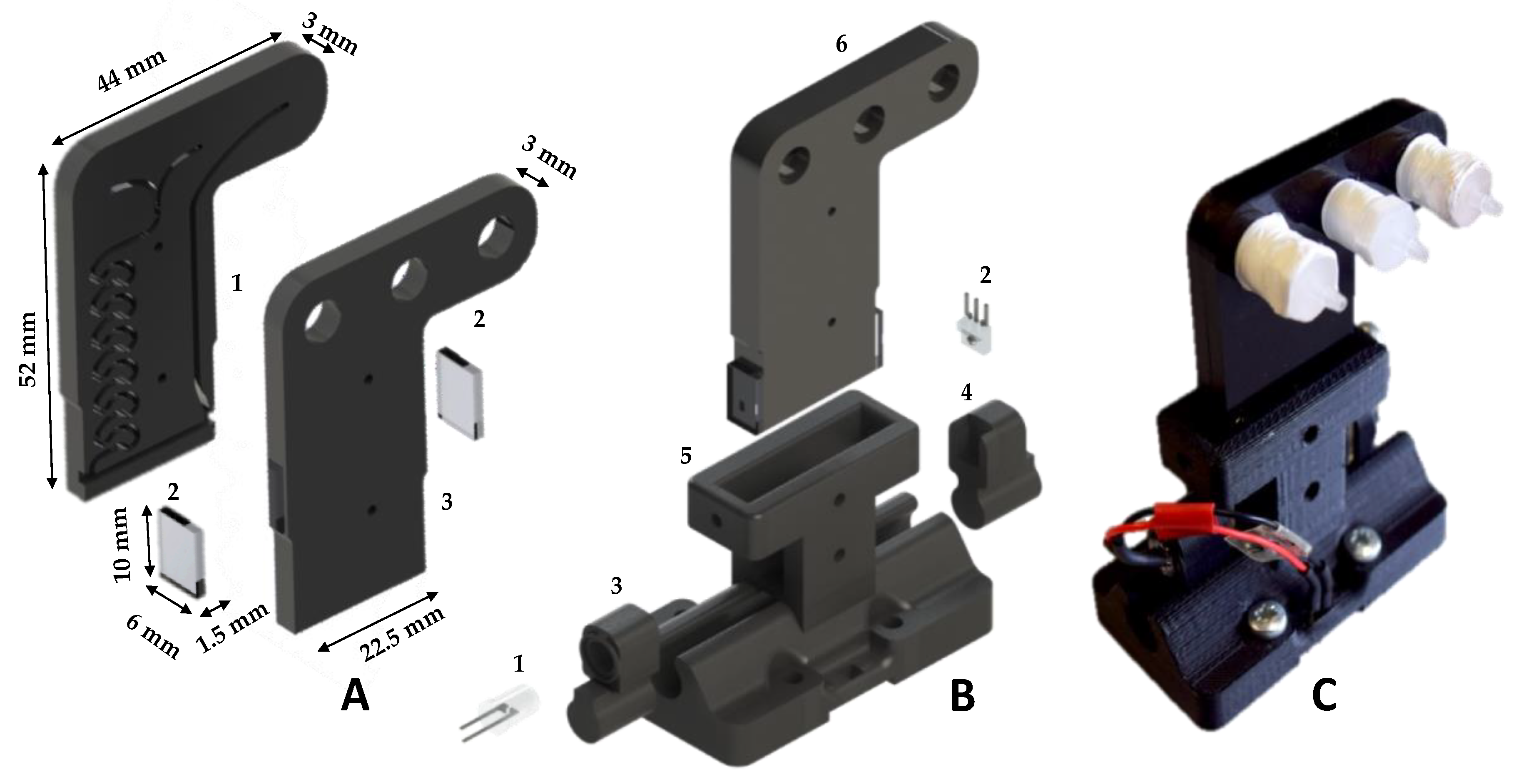
2.3. Fluidic Chip and Optical Detection
2.4. Chassis, Housing and Tubing
2.5. Electrical Control and Transmittance
2.6. Analytical Performance
2.7. Long-Term Laboratory Assessment of the Sensing Platform in Fully Autonomous Mode
2.8. Field Deployment
3. Results and Discussion
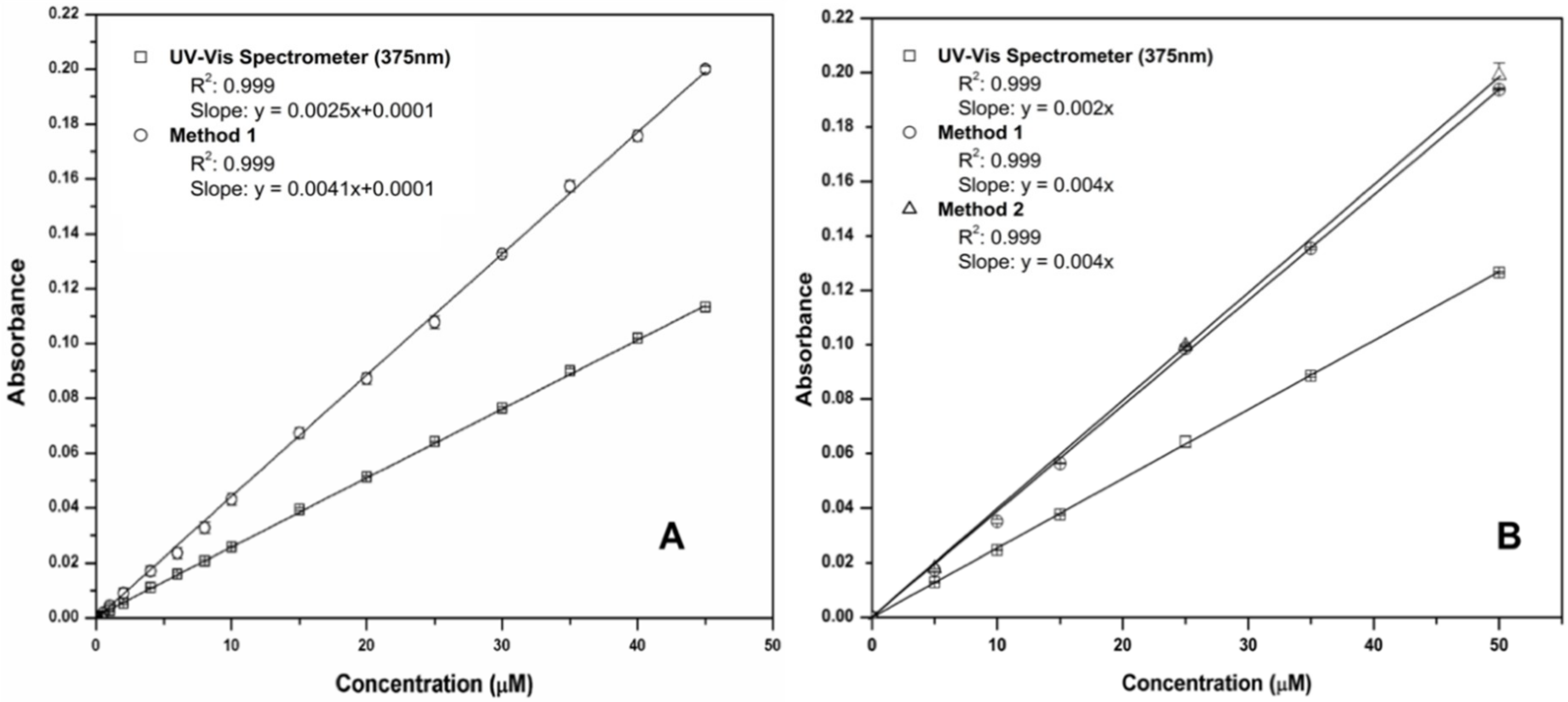
3.1. Validation of Sampling and Optical Detection (Methods 1 and 2)
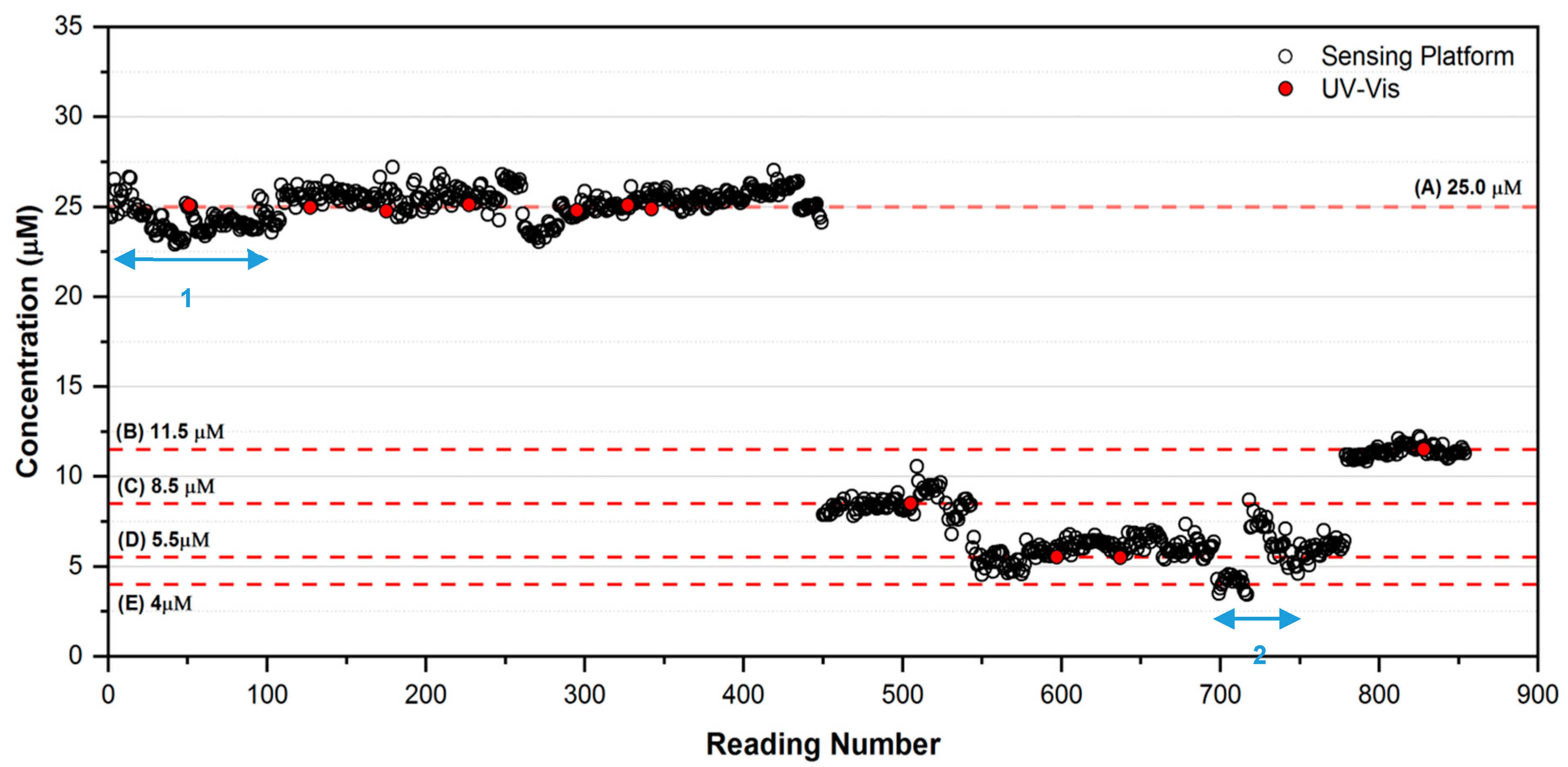
3.2. Long-Term Laboratory Assessment of the Sensing Platform in Fully Autonomous Mode
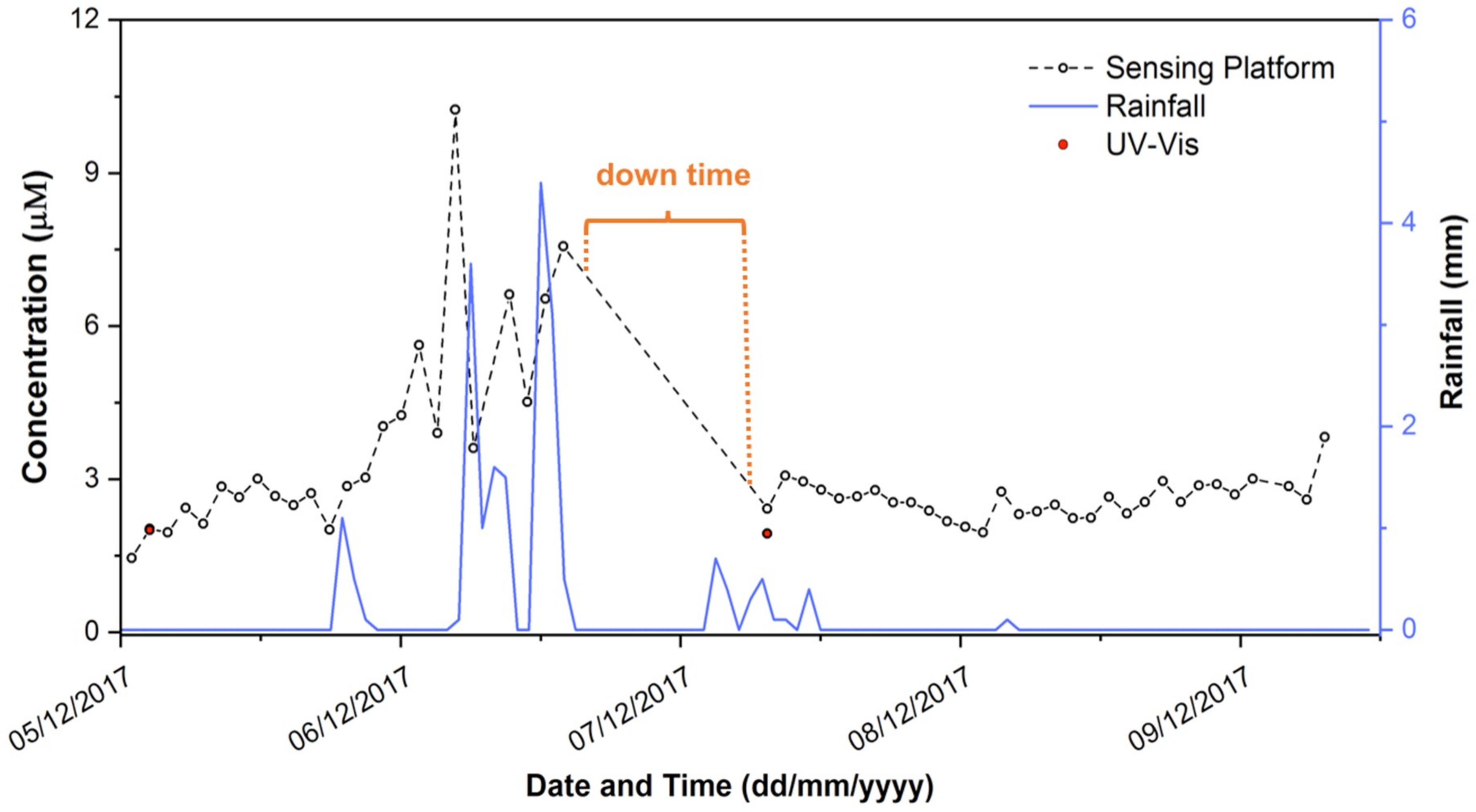
3.3. Preliminary Field Assessment of the Sensing Platform
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Zamparas, M.; Zacharias, I. Restoration of eutrophic freshwater by managing internal nutrient loads. A review. Sci. Total Environ. 2014, 496, 551–562. [Google Scholar] [CrossRef] [PubMed]
- Johnson, A.G.; Glenn, C.R.; Burnett, W.C.; Peterson, R.N.; Lucey, P.G. Aerial infrared imaging reveals large nutrient-rich groundwater inputs to the ocean. Geophys. Res. Lett. 2008, 35. [Google Scholar] [CrossRef] [Green Version]
- Schierenbeck, T.M.; Smith, M.C. Path to Impact for Autonomous Field Deployable Chemical Sensors: A Case Study of in Situ Nitrite Sensors. Environ. Sci. Technol. 2017, 51, 4755–4771. [Google Scholar] [CrossRef] [PubMed]
- Warwick, C.; Guerreiro, A.; Soares, A. Sensing and analysis of soluble phosphates in environmental samples: A review. Biosens. Bioelectron. 2013, 41, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Blaen, P.J.; Khamis, K.; Lloyd, C.E.M.; Bradley, C.; Hannah, D.; Krause, S. Real-time monitoring of nutrients and dissolved organic matter in rivers: Capturing event dynamics, technological opportunities and future directions. Sci. Total Environ. 2016, 569–570, 647–660. [Google Scholar] [CrossRef] [PubMed]
- Sanseverino, I.; Conduto, D.; Pozzoli, L.; Dobricic, S.; Lettieri, T.; European Commission. Joint Research Centre: Algal Bloom and Its Economic Impact; Publications Office: Luxembourg, 2016. [Google Scholar]
- Beaton, A.D.; Cardwell, C.L.; Thomas, R.S.; Sieben, V.J.; Legiret, F.-E.; Waugh, E.M.; Statham, P.J.; Mowlem, M.C.; Morgan, H. Lab-on-Chip Measurement of Nitrate and Nitrite for In Situ Analysis of Natural Waters. Environ. Sci. Technol. 2012, 46, 9548–9556. [Google Scholar] [CrossRef] [PubMed]
- Nightingale, A.M.; Beaton, A.D.; Mowlem, M.C. Trends in microfluidic systems for in situ chemical analysis of natural waters. Sens. Actuators B Chem. 2015, 221, 1398–1405. [Google Scholar] [CrossRef] [Green Version]
- Voulvoulis, N.; Arpon, K.D.; Giakoumis, T. The EU Water Framework Directive: From great expectations to problems with implementation. Sci. Total Environ. 2017, 575, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Ribotti, A.; Magni, P.; Mireno, B.; Schroeder, K.; Barton, J.; McCaul, M.; Diamond, D. New cost-effective, interoperable sensors tested on existing ocean observing platforms in application of European directives: The COMMON SENSE European project. In Proceedings of the OCEANS 2015, Genova, Italy, 18–21 May 2015; pp. 1–9. [Google Scholar]
- Cleary, J.; Slater, C.; McGraw, C.; Diamond, D. An autonomous microfluidic sensor for phosphate: On-site analysis of treated wastewater. IEEE Sens. J. 2008, 8, 508–515. [Google Scholar] [CrossRef]
- Slater, C.; Cleary, J.; Lau, K.-T.; Snakenborg, D.; Corcoran, B.; Kutter, J.P.; Diamond, D. Validation of a fully autonomous phosphate analyser based on a microfluidic lab-on-a-chip. Water Sci. Technol. 2010, 61, 1811–1818. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bowden, M.; Diamond, D. The determination of phosphorus in a microfluidic manifold demonstrating long-term reagent lifetime and chemical stability utilising a colorimetric method. Sens. Actuators B Chem. 2003, 90, 170–174. [Google Scholar] [CrossRef]
- O’Toole, M.; Lau, K.T.; Shepherd, R.; Slater, C.; Diamond, D. Determination of phosphate using a highly sensitive paired emitter–detector diode photometric flow detector. Anal. Chim. Acta 2007, 597, 290–294. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cleary, J.; Slater, C.; McGraw, C.; Diamond, D. An autonomous microfluidic sensor for phosphate: On-site analysis of treated wastewater. IEEE Sens. J. 2008, 8, 508–515. [Google Scholar] [CrossRef]
- McCarthy, V.; Irvine, K. A test of stoichiometry across six Irish lakes of low-moderate nutrient status and contrasting hardness. J. Plankton Res. 2010, 32, 15–29. [Google Scholar] [CrossRef]
- Galway County Council. Available online: http://www.galway.ie/en/services/environment/environmentalprotectionbeachesburialgrounds/beaches/blueflagandgreencoastawards/ (accessed on 26 June 2018).
- Historical Data—Met Éireann—The Irish Meteorological Service. Available online: https://www.met.ie/climate/available-data/historical-data (accessed on 14 August 2018).
- Interactive, N. Zoom Earth—Explore Satellite and Aerial Images of the Earth. Available online: https://zoom.earth/ (accessed on 5 November 2018).


| PO43− (µM) | Reference PO43− | Method 1 PO43− | Method 2 PO43− ** | |||
|---|---|---|---|---|---|---|
| Mean (µM) ± %RSD * | %RE | Mean (µM) ± %RSD * | %RE | Mean (µM) ± %RSD * | %RE | |
| 5 | 5.07 ± 1.98 | 1.40 | 4.44 ± 2.23 | −11.16 | 5.03 ± 0.81 | 0.52 |
| 10 | 9.71 ± 0.46 | −2.90 | 8.98 ± 2.03 | −10.19 | - | - |
| 15 | 14.86 ± 0.95 | −0.93 | 14.38 ± 0.38 | −4.12 | - | - |
| 25 | 25.35 ± 3.28 | 1.40 | 25.17 ± 0.84 | 0.69 | 25.04 ± 0.59 | 0.15 |
| 35 | 34.91 ± 0.58 | −0.26 | 34.56 ± 0.34 | −1.25 | - | - |
| 50 | 49.89 ± 0.28 | −0.22 | 49.44 ± 0.12 | −1.13 | 50.01 ± 2.30 | 0.01 |
| PO43− (µM) | Reference PO43− | Sensing Platform PO43− | ||
|---|---|---|---|---|
| Mean (µM) ± %RSD * | %RE | Mean (µM) ± %RSD | %RE | |
| 5.5 | 5.81 ± 3.27 | 5.64 | 5.77 ± 13.45 (n = 223) | 4.91 |
| 8.5 | 8.48 ± 2.82 | −0.24 | 8.49 ± 6.12 (n = 90) | −0.12 |
| 11.5 | 11.49 ± 0.78 | −0.09 | 11.40 ± 2.42 (n = 69) | −0.87 |
| 25.0 | 24.97 ± 0.48 | −0.12 | 25.12 ± 3.36 (n = 462) | 0.48 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donohoe, A.; Lacour, G.; McCluskey, P.; Diamond, D.; McCaul, M. Development of a Cost-Effective Sensing Platform for Monitoring Phosphate in Natural Waters. Chemosensors 2018, 6, 57. https://doi.org/10.3390/chemosensors6040057
Donohoe A, Lacour G, McCluskey P, Diamond D, McCaul M. Development of a Cost-Effective Sensing Platform for Monitoring Phosphate in Natural Waters. Chemosensors. 2018; 6(4):57. https://doi.org/10.3390/chemosensors6040057
Chicago/Turabian StyleDonohoe, Andrew, Gareth Lacour, Peter McCluskey, Dermot Diamond, and Margaret McCaul. 2018. "Development of a Cost-Effective Sensing Platform for Monitoring Phosphate in Natural Waters" Chemosensors 6, no. 4: 57. https://doi.org/10.3390/chemosensors6040057
APA StyleDonohoe, A., Lacour, G., McCluskey, P., Diamond, D., & McCaul, M. (2018). Development of a Cost-Effective Sensing Platform for Monitoring Phosphate in Natural Waters. Chemosensors, 6(4), 57. https://doi.org/10.3390/chemosensors6040057






