Label-Free Sensing in Microdroplet-Based Microfluidic Systems
Abstract
1. Introduction
2. Microdroplet Sensing Platforms
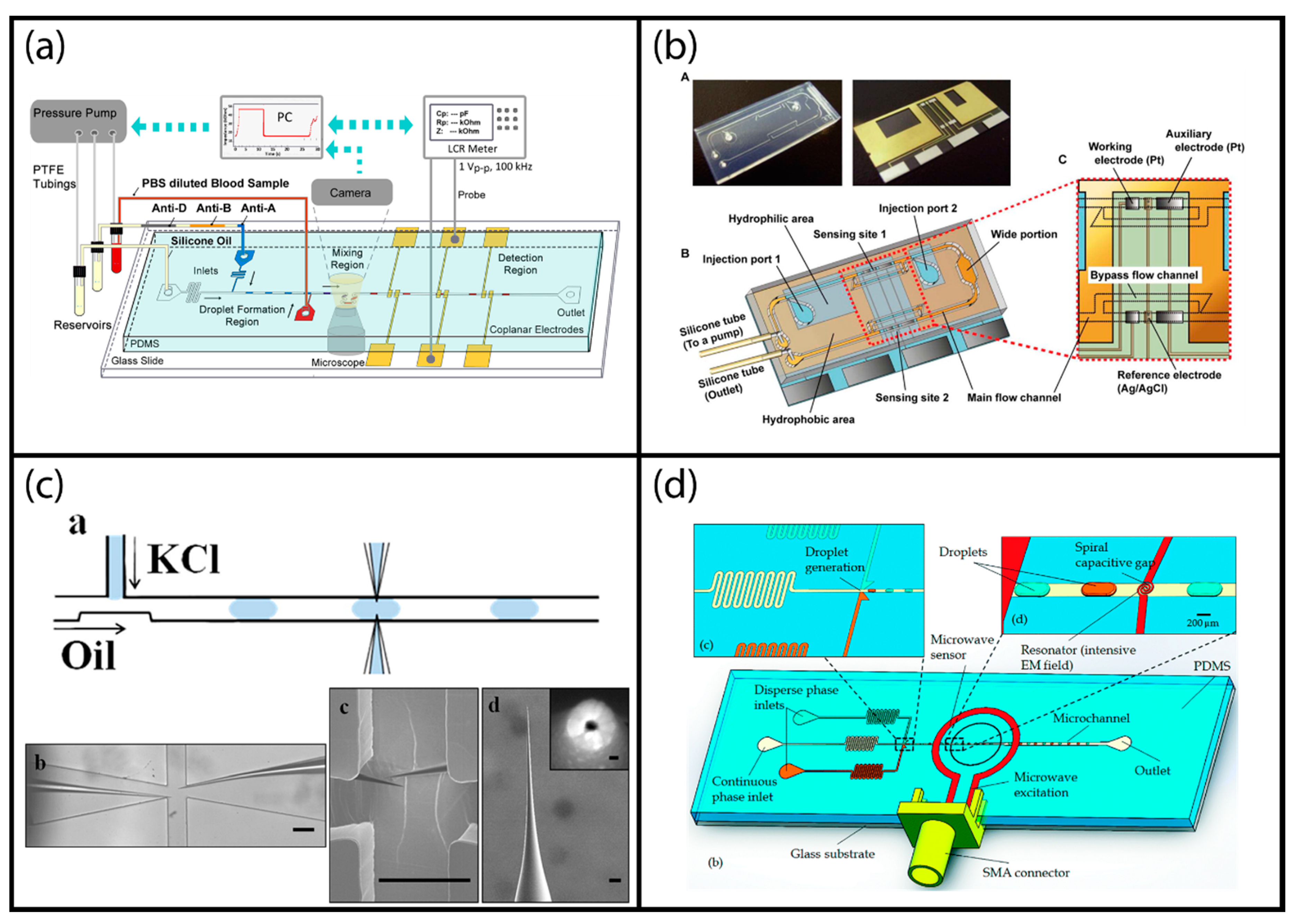
2.1. Dispensed Droplet System
2.2. Slip-Chip System
2.3. Digital Droplet System
2.4. Segmented Flow Droplet Systems
3. Label-Free Sensing Methodologies for Droplet Systems
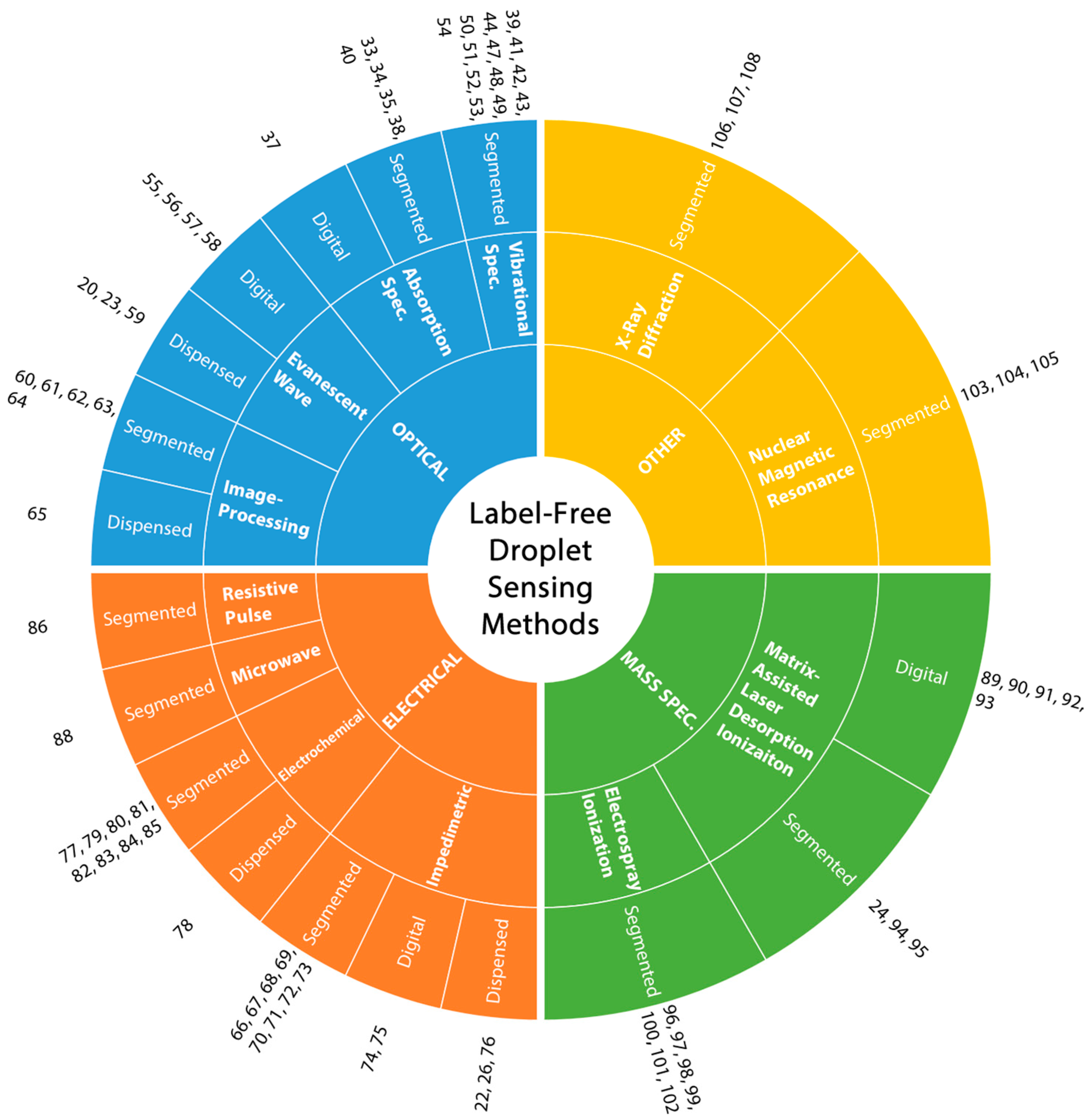
4. Optical Sensing of Droplets
4.1. Absorption Spectroscopy
4.2. Vibrational Spectroscopy
4.2.1. Infrared (IR) Spectroscopy
4.2.2. Raman Spectroscopy
4.3. Evanescent Field Sensing
4.4. Image Processing-Based Sensing
5. Electrical Sensing of Droplets
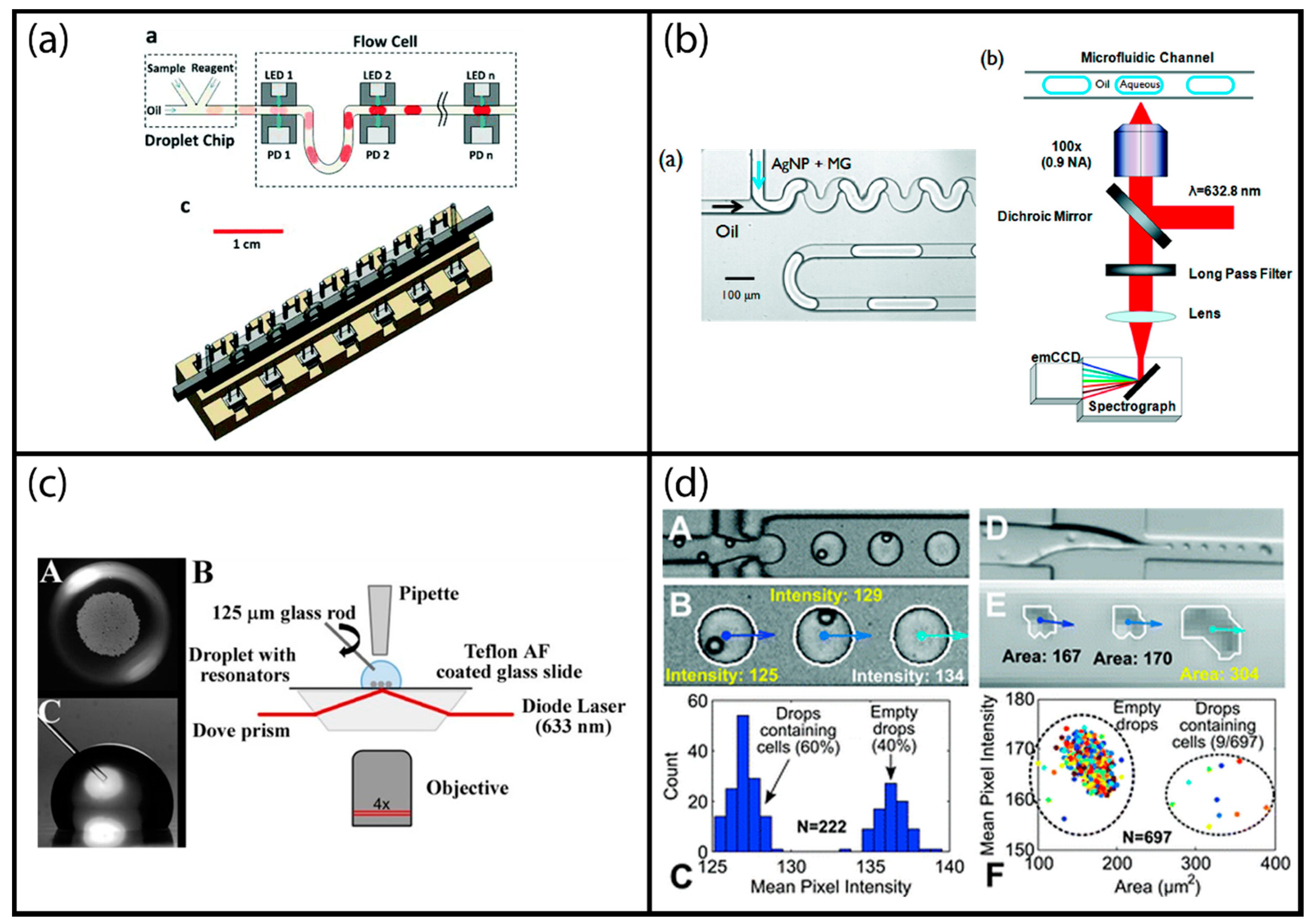
5.1. Impedimetric Sensing
5.2. Electrochemical Sensing
5.3. Resistive Pulse Sensing
5.4. Microwave Sensing
6. Mass Spectrometry
6.1. Matrix-Assisted Laser Desorption/Ionization (MALDI) MS
6.2. Electrospray Ionization (ESI) MS
7. Other Label-free Droplet Sensing Methods
7.1. Nuclear Magnetic Resonance (NMR) Spectroscopy
7.2. X-ray Diffraction/Scattering Analysis
8. Discussion and Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Whitesides, G.M. The origins and the future of microfluidics. Nature 2006, 442, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Squires, T.M.; Quake, S.R. Microfluidics: Fluid physics at the nanoliter scale. Rev. Mod. Phys. 2005, 77, 977–1026. [Google Scholar] [CrossRef]
- Theberge, A.B.; Courtois, F.; Schaerli, Y.; Fischlechner, M.; Abell, C.; Hollfelder, F.; Huck, W.T.S. Microdroplets in microfluidics: An evolving platform for discoveries in chemistry and biology. Angew. Chem. Int. Ed. 2010, 49, 5846–5868. [Google Scholar] [CrossRef] [PubMed]
- Teh, S.Y.; Lin, R.; Hung, L.H.; Lee, A.P. Droplet microfluidics. Lab Chip 2008, 8, 198–220. [Google Scholar] [CrossRef] [PubMed]
- Dressler, O.J.; Maceiczyk, R.M.; Chang, S.I.; deMello, A.J. Droplet-based microfluidics: Enabling impact on drug discovery. J. Biomol. Screen. 2014, 19, 483–496. [Google Scholar] [CrossRef] [PubMed]
- Cao, L.; Cui, X.Y.; Hu, J.; Li, Z.D.; Choi, J.R.; Yang, Q.Z.; Lin, M.; Ying, H.L.; Xu, F. Advances in digital polymerase chain reaction (DPCR) and its emerging biomedical applications. Biosens. Bioelectron. 2017, 90, 459–474. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.H.; Meng, Y.; Sui, Z.W.; Wang, J.; Wu, L.Q.; Fu, B.Q. Comparison of four digital PCR platforms for accurate quantification of DNA copy number of a certified plasmid DNA reference material. Sci. Rep. 2015, 5, 13174. [Google Scholar] [CrossRef] [PubMed]
- Shang, L.; Cheng, Y.; Zhao, Y. Emerging droplet microfluidics. Chem. Rev. 2017, 117, 7964–8040. [Google Scholar] [CrossRef] [PubMed]
- Christopher, G.F.; Anna, S.L. Microfluidic methods for generating continuous droplet streams. J. Phys. D Appl. Phys. 2007, 40, R319. [Google Scholar] [CrossRef]
- Vladisavljević, G.; Kobayashi, I.; Nakajima, M. Production of uniform droplets using membrane, microchannel and microfluidic emulsification devices. Microfluid. Nanofluid. 2012, 13, 151–178. [Google Scholar] [CrossRef]
- Zhu, P.; Wang, L. Passive and active droplet generation with microfluidics: A review. Lab Chip 2017, 17, 34–75. [Google Scholar] [CrossRef] [PubMed]
- Pit, A.M.; Duits, M.H.; Mugele, F. Droplet manipulations in two phase flow microfluidics. Micromachines 2015, 6, 1768–1793. [Google Scholar] [CrossRef]
- Xi, H.-D.; Zheng, H.; Guo, W.; Gañán-Calvo, A.M.; Ai, Y.; Tsao, C.-W.; Zhou, J.; Li, W.; Huang, Y.; Nguyen, N.-T. Active droplet sorting in microfluidics: A review. Lab Chip 2017, 17, 751–771. [Google Scholar] [CrossRef] [PubMed]
- Isiksacan, Z.; Guler, M.T.; Kalantarifard, A.; Asghari, M.; Elbuken, C. Lab-on-a-Chip Platforms for Disease Detection and Diagnosis. In Biosensors and Nanotechnology: Applications in Health Care Diagnostics; Altintas, Z., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2017; pp. 155–181. [Google Scholar] [CrossRef]
- Joensson, H.N.; Andersson Svahn, H. Droplet microfluidics—A tool for single-cell analysis. Angew. Chem. Int. Ed. 2012, 51, 12176–12192. [Google Scholar] [CrossRef] [PubMed]
- Lagus, T.P.; Edd, J.F. A review of the theory, methods and recent applications of high-throughput single-cell droplet microfluidics. J. Phys. D Appl. Phys. 2013, 46, 114005. [Google Scholar] [CrossRef]
- Dressler, O.; Casadevall i Solvas, X.; deMello, A.J. Chemical and biological dynamics using droplet-based microfluidics. Ann. Rev. Anal. Chem. 2017, 10, 1–24. [Google Scholar] [CrossRef] [PubMed]
- Gach, P.C.; Iwai, K.; Kim, P.W.; Hillson, N.J.; Singh, A.K. Droplet microfluidics for synthetic biology. Lab Chip 2017, 17, 3388–3400. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Cordero, J.L.; Fan, Z.H. Sessile droplets for chemical and biological assays. Lab Chip 2017, 17, 2150–2166. [Google Scholar] [CrossRef] [PubMed]
- Wildgen, S.M.; Dunn, R.C. Whispering gallery mode resonators for rapid label-free biosensing in small volume droplets. Biosensors 2015, 5, 118–130. [Google Scholar] [CrossRef] [PubMed]
- Yin, L.; Wang, S.; Shan, X.; Zhang, S.; Tao, N. Quantification of protein interaction kinetics in a micro droplet. Rev. Sci. Instrum. 2015, 86, 114101. [Google Scholar] [CrossRef] [PubMed]
- Ebrahimi, A.; Dak, P.; Salm, E.; Dash, S.; Garimella, S.V.; Bashir, R.; Alam, M.A. Nanotextured superhydrophobic electrodes enable detection of attomolar-scale DNA concentration within a droplet by non-faradaic impedance spectroscopy. Lab Chip 2013, 13, 4248–4256. [Google Scholar] [CrossRef] [PubMed]
- Valentino, J.; Troian, S.; Wagner, S. Microfluidic Detection and Analysis by Integration of Evanescent Wave Sensing with Thermocapillary Actuation. In Proceedings of the 18th IEEE International Conference on Micro Electro Mechanical Systems (MEMS), Miami Beach, FL, USA, 30 January–3 February 2005; pp. 730–733. [Google Scholar]
- Pereira, F.; Niu, X. A nano lc-maldi mass spectrometry droplet interface for the analysis of complex protein samples. PLoS ONE 2013, 8, e63087. [Google Scholar] [CrossRef] [PubMed]
- Jarocka, U.; Radecka, H.; Malinowski, T.; Michalczuk, L.; Radecki, J. Detection of prunus necrotic ringspot virus in plant extracts with impedimetric immunosensor based on glassy carbon electrode. Electroanalysis 2013, 25, 433–438. [Google Scholar] [CrossRef]
- Ebrahimi, A.; Alam, M.A. Evaporation-induced stimulation of bacterial osmoregulation for electrical assessment of cell viability. Proc. Natl. Acad. Sci. USA 2016, 113, 7059–7064. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Fang, Q. Analytical detection techniques for droplet microfluidics—A review. Anal. Chim. Acta 2013, 787, 24–35. [Google Scholar] [CrossRef] [PubMed]
- Du, W.; Li, L.; Nichols, K.P.; Ismagilov, R.F. Slipchip. Lab Chip 2009, 9, 2286–2292. [Google Scholar] [CrossRef] [PubMed]
- Malic, L.; Brassard, D.; Veres, T.; Tabrizian, M. Integration and detection of biochemical assays in digital microfluidic loc devices. Lab Chip 2010, 10, 418–431. [Google Scholar] [CrossRef] [PubMed]
- Kaminski, T.S.; Garstecki, P. Controlled droplet microfluidic systems for multistep chemical and biological assays. Chem. Soc. Rev. 2017, 46, 6210–6226. [Google Scholar] [CrossRef] [PubMed]
- Cole, R.H.; Tang, S.Y.; Siltanen, C.A.; Shahi, P.; Zhang, J.Q.; Poust, S.; Gartner, Z.J.; Abate, A.R. Printed droplet microfluidics for on demand dispensing of picoliter droplets and cells. Proc. Natl. Acad. Sci. USA 2017, 114, 8728–8733. [Google Scholar] [CrossRef] [PubMed]
- Luka, G.; Ahmadi, A.; Najjaran, H.; Alocilja, E.; DeRosa, M.; Wolthers, K.; Malki, A.; Aziz, H.; Althani, A.; Hoorfar, M. Microfluidics integrated biosensors: A leading technology towards lab-on-a-chip and sensing applications. Sensors 2015, 15, 30011–30031. [Google Scholar] [CrossRef] [PubMed]
- Neil, S.R.; Rushworth, C.M.; Vallance, C.; Mackenzie, S.R. Broadband cavity-enhanced absorption spectroscopy for real time, in situ spectral analysis of microfluidic droplets. Lab Chip 2011, 11, 3953–3955. [Google Scholar] [CrossRef] [PubMed]
- Mao, Z.; Guo, F.; Xie, Y.; Zhao, Y.; Lapsley, M.I.; Wang, L.; Mai, J.D.; Costanzo, F.; Huang, T.J. Label-free measurements of reaction kinetics using a droplet-based optofluidic device. J. Lab. Autom. 2015, 20, 17–24. [Google Scholar] [CrossRef] [PubMed]
- Hung, L.-H.; Choi, K.M.; Tseng, W.-Y.; Tan, Y.-C.; Shea, K.J.; Lee, A.P. Alternating droplet generation and controlled dynamic droplet fusion in microfluidic device for CDS nanoparticle synthesis. Lab Chip 2006, 6, 174–178. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, V.; Pamula, V.K.; Fair, R.B. Droplet-based microfluidic lab-on-a-chip for glucose detection. Anal. Chim. Acta 2004, 507, 145–150. [Google Scholar] [CrossRef]
- Srinivasan, V.; Pamula, V.K.; Fair, R.B. An integrated digital microfluidic lab-on-a-chip for clinical diagnostics on human physiological fluids. Lab Chip 2004, 4, 310–315. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.-U.; Nightingale, A.M.; Niu, X. Continuous measurement of enzymatic kinetics in droplet flow for point-of-care monitoring. Analyst 2016, 141, 3266–3273. [Google Scholar] [CrossRef] [PubMed]
- Bunaciu, A.A.; Aboul-Enein, H.Y.; Fleschin, Ş. Vibrational spectroscopy in clinical analysis. Appl. Spectrosc. Rev. 2015, 50, 176–191. [Google Scholar] [CrossRef]
- Chan, K.A.; Niu, X.; DeMello, A.; Kazarian, S. Generation of chemical movies: Ft-ir spectroscopic imaging of segmented flows. Anal. Chem. 2011, 83, 3606–3609. [Google Scholar] [CrossRef] [PubMed]
- Müller, T.; Ruggeri, F.S.; Kulik, A.J.; Shimanovich, U.; Mason, T.O.; Knowles, T.P.; Dietler, G. Nanoscale spatially resolved infrared spectra from single microdroplets. Lab Chip 2014, 14, 1315–1319. [Google Scholar] [CrossRef] [PubMed]
- Barnes, S.E.; Cygan, Z.T.; Yates, J.K.; Beers, K.L.; Amis, E.J. Raman spectroscopic monitoring of droplet polymerization in a microfluidic device. Analyst 2006, 131, 1027–1033. [Google Scholar] [CrossRef] [PubMed]
- Cristobal, G.; Arbouet, L.; Sarrazin, F.; Talaga, D.; Bruneel, J.-L.; Joanicot, M.; Servant, L. On-line laser raman spectroscopic probing of droplets engineered in microfluidic devices. Lab Chip 2006, 6, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Sarrazin, F.; Salmon, J.-B.; Talaga, D.; Servant, L. Chemical reaction imaging within microfluidic devices using confocal raman spectroscopy: The case of water and deuterium oxide as a model system. Anal. Chem. 2008, 80, 1689–1695. [Google Scholar] [CrossRef] [PubMed]
- Bringer, M.R.; Gerdts, C.J.; Song, H.; Tice, J.D.; Ismagilov, R.F. Microfluidic systems for chemical kinetics that rely on chaotic mixing in droplets. Philos. Trans. R. Soc. A 2004, 362, 1087–1104. [Google Scholar] [CrossRef] [PubMed]
- Jahn, I.J.; Zukovskaja, O.; Zheng, X.S.; Weber, K.; Bocklitz, T.W.; Cialla-May, D.; Popp, J. Surface-enhanced raman spectroscopy and microfluidic platforms: Challenges, solutions and potential applications. Analyst 2017, 142, 1022–1047. [Google Scholar] [CrossRef] [PubMed]
- Marz, A.; Henkel, T.; Cialla, D.; Schmitt, M.; Popp, J. Droplet formation via flow-through microdevices in raman and surface enhanced raman spectroscopy-concepts and applications. Lab Chip 2011, 11, 3584–3592. [Google Scholar] [CrossRef] [PubMed]
- Strehle, K.R.; Cialla, D.; Rösch, P.; Henkel, T.; Köhler, M.; Popp, J. A reproducible surface-enhanced raman spectroscopy approach. Online sers measurements in a segmented microfluidic system. Anal. Chem. 2007, 79, 1542–1547. [Google Scholar] [CrossRef] [PubMed]
- Ackermann, K.R.; Henkel, T.; Popp, J. Quantitative online detection of low-concentrated drugs via a sers microfluidic system. Chem. Phys. Chem. 2007, 8, 2665–2670. [Google Scholar] [CrossRef] [PubMed]
- Walter, A.; März, A.; Schumacher, W.; Rösch, P.; Popp, J. Towards a fast, high specific and reliable discrimination of bacteria on strain level by means of SERS in a microfluidic device. Lab Chip 2011, 11, 1013–1021. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Lim, C.; Chen, L.; Chon, H.; Choo, J.; Hong, J. Surface-enhanced raman scattering in nanoliter droplets: Towards high-sensitivity detection of mercury (ii) ions. Anal. Bioanal. Chem. 2009, 394, 1827–1832. [Google Scholar] [CrossRef] [PubMed]
- Gao, R.; Choi, N.; Chang, S.-I.; Kang, S.H.; Song, J.M.; Cho, S.I.; Lim, D.W.; Choo, J. Highly sensitive trace analysis of paraquat using a surface-enhanced raman scattering microdroplet sensor. Anal. Chim. Acta 2010, 681, 87–91. [Google Scholar] [CrossRef] [PubMed]
- Cecchini, M.P.; Hong, J.; Lim, C.; Choo, J.; Albrecht, T.; Demello, A.J.; Edel, J.B. Ultrafast surface enhanced resonance raman scattering detection in droplet-based microfluidic systems. Anal. Chem. 2011, 83, 3076–3081. [Google Scholar] [CrossRef] [PubMed]
- Syme, C.D.; Martino, C.; Yusvana, R.; Sirimuthu, N.M.; Cooper, J.M. Quantitative characterization of individual microdroplets using surface-enhanced resonance raman scattering spectroscopy. Anal. Chem. 2012, 84, 1491–1495. [Google Scholar] [CrossRef] [PubMed]
- Malic, L.; Veres, T.; Tabrizian, M. Two-dimensional droplet-based surface plasmon resonance imaging using electrowetting-on-dielectric microfluidics. Lab Chip 2009, 9, 473–475. [Google Scholar] [CrossRef] [PubMed]
- Malic, L.; Veres, T.; Tabrizian, M. Biochip functionalization using electrowetting-on-dielectric digital microfluidics for surface plasmon resonance imaging detection of DNA hybridization. Biosens. Bioelectron. 2009, 24, 2218–2224. [Google Scholar] [CrossRef] [PubMed]
- Arce, C.L.; Witters, D.; Puers, R.; Lammertyn, J.; Bienstman, P. Silicon photonic sensors incorporated in a digital microfluidic system. Anal. Bioanal. Chem. 2012, 404, 2887–2894. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.B.; Su, L.Y.; Fu, C.S.; Huang, C.S.; Hsu, W. Droplet-based label-free detection system based on guided-mode resonance and electrowetting-on-dielectric for concentration measurement. Jpn. J. Appl. Phys. 2017, 56. [Google Scholar] [CrossRef]
- Yin, L.L.; Wang, S.P.; Shan, X.N.; Zhang, S.T.; Tao, N.J. Quantification of protein interaction kinetics in a micro droplet. Rev. Sci. Instrum. 2015, 86, 114101. [Google Scholar] [CrossRef] [PubMed]
- Glawdel, T.; Elbuken, C.; Ren, C.L. Droplet formation in microfluidic t-junction generators operating in the transitional regime. I. Experimental observations. Phys. Rev. E 2012, 85, 016322. [Google Scholar] [CrossRef] [PubMed]
- Glawdel, T.; Elbuken, C.; Ren, C.L. Droplet formation in microfluidic t-junction generators operating in the transitional regime. II. Modeling. Phys. Rev. E 2012, 85, 016323. [Google Scholar] [CrossRef] [PubMed]
- Basu, A.S. Droplet morphometry and velocimetry (DMV): A video processing software for time-resolved, label-free tracking of droplet parameters. Lab Chip 2013, 13, 1892–1901. [Google Scholar] [CrossRef] [PubMed]
- Zang, E.; Brandes, S.; Tovar, M.; Martin, K.; Mech, F.; Horbert, P.; Henkel, T.; Figge, M.T.; Roth, M. Real-time image processing for label-free enrichment of actinobacteria cultivated in picolitre droplets. Lab Chip 2013, 13, 3707–3713. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, T.W.; Hänselmann, S.; Janiesch, J.-W.; Rademacher, A.; Böhm, C.H. Applying microdroplets as sensors for label-free detection of chemical reactions. Lab Chip 2012, 12, 916–922. [Google Scholar] [CrossRef] [PubMed]
- Liao, T.-C.; Yeh, J.A. Microdroplet protein sensors on a gold surface with a self-assembled monolayer treatment. Int. J. Autom. Smart Technol. 2012, 2, 43–48. [Google Scholar] [CrossRef]
- Chen, J.Z.; Darhuber, A.A.; Troian, S.M.; Wagner, S. Capacitive sensing of droplets for microfluidic devices based on thermocapillary actuation. Lab Chip 2004, 4, 473–480. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, N.; Burns, M.A. Electronic drop sensing in microfluidic devices: Automated operation of a nanoliter viscometer. Lab Chip 2006, 6, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Elbuken, C.; Glawdel, T.; Chan, D.; Ren, C.L. Detection of microdroplet size and speed using capacitive sensors. Sens. Actuators A Phys. 2011, 171, 55–62. [Google Scholar] [CrossRef]
- Isgor, P.K.; Marcali, M.; Keser, M.; Elbuken, C. Microfluidic droplet content detection using integrated capacitive sensors. Sens. Actuators B Chem. 2015, 210, 669–675. [Google Scholar] [CrossRef]
- Kemna, E.W.; Segerink, L.I.; Wolbers, F.; Vermes, I.; van den Berg, A. Label-free, high-throughput, electrical detection of cells in droplets. Analyst 2013, 138, 4585–4592. [Google Scholar] [CrossRef] [PubMed]
- Simon, M.G.; Lin, R.; Lopez-Prieto, J.; Lee, A.P. Label-Free Detection of DNA Amplification in Droplets Using Electrical Impedance. In Proceedings of the 15th International Conference on Miniaturized Systems for Chemistry and Life Sciences, Seattle, WA, USA, 2–6 October 2011. [Google Scholar]
- Lee, A.P.; Lopez-Prieto, J.; Lin, R.; Simon, M.; Martin, N. Real-Time, Label-Free Detection of Nucleic Acid Amplification in Droplets Using Impedance Spectroscopy and Solid-Phase Substrate. U.S. Patent US9030215B2, 12 May 2015. [Google Scholar]
- Marcali, M.; Elbuken, C. Impedimetric detection and lumped element modelling of a hemagglutination assay in microdroplets. Lab Chip 2016, 16, 2494–2503. [Google Scholar] [CrossRef] [PubMed]
- Sadeghi, S.; Ding, H.J.; Shah, G.J.; Chen, S.P.; Keng, P.Y.; Kim, C.J.; van Dam, R.M. On chip droplet characterization: A practical, high-sensitivity measurement of droplet impedance in digital microfluidics. Anal. Chem. 2012, 84, 1915–1923. [Google Scholar] [CrossRef] [PubMed]
- Shih, S.C.C.; Barbulovic-Nad, I.; Yang, X.N.; Fobel, R.; Wheeler, A.R. Digital microfluidics with impedance sensing for integrated cell culture and analysis. Biosens. Bioelectron. 2013, 42, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Ernst, A.; Streule, W.; Schmitt, N.; Zengerle, R.; Koltay, P. A capacitive sensor for non-contact nanoliter droplet detection. Sens. Actuators A Phys. 2009, 153, 57–63. [Google Scholar] [CrossRef]
- Luo, C.X.; Yang, X.J.; Fu, O.; Sun, M.H.; Ouyang, Q.; Chen, Y.; Ji, H. Picoliter-volume aqueous droplets in oil: Electrochemical detection and east electroporation. Electrophoresis 2006, 27, 1977–1983. [Google Scholar] [CrossRef] [PubMed]
- Cai, X.X.; Klauke, N.; Glidle, A.; Cobbold, P.; Smith, G.L.; Cooper, J.M. Ultra-low-volume, real-time measurements of lactate from the single heart cell using microsystems technology. Anal. Chem. 2002, 74, 908–914. [Google Scholar] [CrossRef] [PubMed]
- Han, Z.Y.; Li, W.T.; Huang, Y.Y.; Zheng, B. Measuring rapid enzymatic kinetics by electrochemical method in droplet-based microfluidic devices with pneumatic valves. Anal. Chem. 2009, 81, 5840–5845. [Google Scholar] [CrossRef] [PubMed]
- Gu, S.; Lu, Y.; Ding, Y.; Li, L.; Zhang, F.; Wu, Q. Droplet-based microfluidics for dose–response assay of enzyme inhibitors by electrochemical method. Anal. Chim. Acta 2013, 796, 68–74. [Google Scholar] [CrossRef] [PubMed]
- Sassa, F.; Laghzali, H.; Fukuda, J.; Suzuki, H. Coulometric detection of components in liquid plugs by microfabricated flow channel and electrode structures. Anal. Chem. 2010, 82, 8725–8732. [Google Scholar] [CrossRef] [PubMed]
- Lin, X.Y.; Hu, X.Q.; Bai, Z.Q.; He, Q.H.; Chen, H.W.; Yan, Y.Z.; Ding, Z.H. A microfluidic chip capable of switching w/o droplets to vertical laminar flow for electrochemical detection of droplet contents. Anal. Chim. Acta 2014, 828, 70–79. [Google Scholar] [CrossRef] [PubMed]
- Itoh, D.; Sassa, F.; Nishi, T.; Kani, Y.; Murata, M.; Suzuki, H. Droplet-based microfluidic sensing system for rapid fish freshness determination. Sens. Actuators B Chem. 2012, 171, 619–626. [Google Scholar] [CrossRef]
- Itoh, D.; Koyachi, E.; Yokokawa, M.; Murata, Y.; Murata, M.; Suzuki, H. Microdevice for on-site fish freshness checking based on k-value measurement. Anal. Chem. 2013, 85, 10962–10968. [Google Scholar] [CrossRef] [PubMed]
- Rattanarat, P.; Suea-Ngam, A.; Ruecha, N.; Siangproh, W.; Henry, C.S.; Monpichar, S.A.; Chailapakul, O. Graphene-polyaniline modified electrochemical droplet-based microfluidic sensor for high-throughput determination of 4-aminophenol. Anal. Chim. Acta 2016, 925, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Gibb, T.R.; Ivanov, A.P.; Edel, J.B.; Albrecht, T. Single molecule ionic current sensing in segmented flow microfluidics. Anal. Chem. 2014, 86, 1864–1871. [Google Scholar] [CrossRef] [PubMed]
- Somerville, J.A.; Willmott, G.R.; Eldridge, J.; Griffiths, M.; McGrath, K.M. Size and charge characterisation of a submicrometre oil-in-water emulsion using resistive pulse sensing with tunable pores. J. Colloid Interface Sci. 2013, 394, 243–251. [Google Scholar] [CrossRef] [PubMed]
- Yesiloz, G.; Boybay, M.S.; Ren, C.L. Label-free high-throughput detection and content sensing of individual droplets in microfluidic systems. Lab Chip 2015, 15, 4008–4019. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, A.R.; Moon, H.; Kim, C.J.; Loo, J.A.; Garrell, R.L. Electrowetting-based microfluidics for analysis of peptides and proteins by matrix-assisted laser desorption/ionization mass spectrometry. Anal. Chem. 2004, 76, 4833–4838. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, A.R.; Moon, H.; Bird, C.A.; Loo, R.R.O.; Kim, C.J.; Loo, J.A.; Garrell, R.L. Digital microfluidics with in-line sample purification for proteomics analyses with MALDI-MS. Anal. Chem. 2005, 77, 534–540. [Google Scholar] [CrossRef] [PubMed]
- Moon, H.; Wheeler, A.R.; Garrell, R.L.; Loo, J.A.; Kim, C.J. An integrated digital microfluidic chip for multiplexed proteomic sample preparation and analysis by MALDI-MS. Lab Chip 2006, 6, 1213–1219. [Google Scholar] [CrossRef] [PubMed]
- Nichols, K.P.; Gardeniers, H.J.G.E. A digital microfluidic system for the investigation of pre-steady-state enzyme kinetics using rapid quenching with MALDI-TOF mass spectrometry. Anal. Chem. 2007, 79, 8699–8704. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Luk, V.N.; Abeigawad, M.; Barbulovic-Nad, I.; Wheeler, A.R. A world-to-chip interface for digital microfluidics. Anal. Chem. 2009, 81, 1061–1067. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, T.; Chen, D.L.; Ismagilov, R.F. Microgram-scale testing of reaction conditions in solution using nanoliter plugs in microfluidics with detection by MALDI-MS. J. Am. Chem. Soc. 2006, 128, 2518–2519. [Google Scholar] [CrossRef] [PubMed]
- Küster, S.K.; Fagerer, S.R.; Verboket, P.E.; Eyer, K.; Jefimovs, K.; Zenobi, R.; Dittrich, P.S. Interfacing droplet microfluidics with matrix-assisted laser desorption/ionization mass spectrometry: Label-free content analysis of single droplets. Anal. Chem. 2013, 85, 1285–1289. [Google Scholar] [CrossRef] [PubMed]
- Fidalgo, L.M.; Whyte, G.; Ruotolo, B.T.; Benesch, J.L.; Stengel, F.; Abell, C.; Robinson, C.V.; Huck, W.T. Coupling microdroplet microreactors with mass spectrometry: Reading the contents of single droplets online. Angew. Chem. Int. Ed. 2009, 48, 3665–3668. [Google Scholar] [CrossRef] [PubMed]
- Kelly, R.T.; Page, J.S.; Marginean, I.; Tang, K.; Smith, R.D. Dilution-free analysis from picoliter droplets by nano-electrospray ionization mass spectrometry. Angew. Chem. Int. Ed. 2009, 48, 6832–6835. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Fang, Q. Integrated droplet analysis system with electrospray ionization-mass spectrometry using a hydrophilic tongue-based droplet extraction interface. Anal. Chem. 2010, 82, 8361–8366. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Pei, J.; Song, P.; Kennedy, R.T. Fraction collection from capillary liquid chromatography and off-line electrospray ionization mass spectrometry using oil segmented flow. Anal. Chem. 2010, 82, 5260–5267. [Google Scholar] [CrossRef] [PubMed]
- Pei, J.; Li, Q.; Kennedy, R.T. Rapid and label-free screening of enzyme inhibitors using segmented flow electrospray ionization mass spectrometry. J. Am. Soc. Mass Spectrom. 2010, 21, 1107–1113. [Google Scholar] [CrossRef] [PubMed]
- Sun, S.W.; Slaney, T.R.; Kennedy, R.T. Label free screening of enzyme inhibitors at femtomole scale using segmented flow electrospray ionization mass spectrometry. Anal. Chem. 2012, 84, 5794–5800. [Google Scholar] [CrossRef] [PubMed]
- Smith, C.A.; Li, X.; Mize, T.H.; Sharpe, T.D.; Graziani, E.I.; Abell, C.; Huck, W.T.S. Sensitive, high throughput detection of proteins in individual, surfactant-stabilized picoliter droplets using nanoelectrospray ionization mass spectrometry. Anal. Chem. 2013, 85, 3812–3816. [Google Scholar] [CrossRef] [PubMed]
- Kautz, R.A.; Goetzinger, W.K.; Karger, B.L. High-throughput microcoil nmr of compound libraries using zero-dispersion segmented flow analysis. J. Comb. Chem. 2005, 7, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.Q.; Schiavo, S.; Orjala, J.; Vouros, P.; Kautz, R. Microscale LC-MS-NMR platform applied to the identification of active cyanobacterial metabolites. Anal. Chem. 2008, 80, 8045–8054. [Google Scholar] [CrossRef] [PubMed]
- Kautz, R.; Wang, P.G.; Giese, R.W. Nuclear magnetic resonance at the picomole level of a DNA adduct. Chem. Res. Toxicol. 2013, 26, 1424–1429. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Tice, J.D.; Roach, L.S.; Ismagilov, R.F. A droplet-based, composite PDMS/Glass capillary microfluidic system for evaluating protein crystallization conditions by microbatch and vapor-diffusion methods with on-chip x-ray diffraction. Angew. Chem. Int. Ed. 2004, 43, 2508–2511. [Google Scholar] [CrossRef] [PubMed]
- Zheng, B.; Gerdts, C.J.; Ismagilov, R.F. Using nanoliter plugs in microfluidics to facilitate and understand protein crystallization. Curr. Opin. Struct. Biol. 2005, 15, 548–555. [Google Scholar] [CrossRef] [PubMed]
- Stehle, R.; Goerigk, G.; Wallacher, D.; Ballauff, M.; Seiffert, S. Small-angle x-ray scattering in droplet-based microfluidics. Lab Chip 2013, 13, 1529–1537. [Google Scholar] [CrossRef] [PubMed]
- Song, H.; Chen, D.L.; Ismagilov, R.F. Reactions in droplets in microfluidic channels. Angew. Chem. Int. Ed. 2006, 45, 7336–7356. [Google Scholar] [CrossRef] [PubMed]
- Charwat, V.; Purtscher, M.; Tedde, S.F.; Hayden, O.; Ertl, P. Standardization of microfluidic cell cultures using integrated organic photodiodes and electrode arrays. Lab Chip 2013, 13, 785–797. [Google Scholar] [CrossRef] [PubMed]
- Jansen-van Vuuren, R.D.; Armin, A.; Pandey, A.K.; Burn, P.L.; Meredith, P. Organic photodiodes: The future of full color detection and image sensing. Adv. Mater. 2016, 28, 4766–4802. [Google Scholar] [CrossRef] [PubMed]
- Schutt, J.; Ibarlucea, B.; Illing, R.; Zorgiebel, F.; Pregl, S.; Nozaki, D.; Weber, W.M.; Mikolajick, T.; Baraban, L.; Cuniberti, G. Compact nanowire sensors probe microdroplets. Nano Lett. 2016, 16, 4991–5000. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Feng, L.; Panaitov, G.; Kireev, D.; Mayer, D.; Offenhausser, A.; Krause, H.J. Biosensing near the neutrality point of graphene. Sci. Adv. 2017, 3, e1701247. [Google Scholar] [CrossRef] [PubMed]
- Schwierz, F. Graphene transistors. Nat. Nanotechnol. 2010, 5, 487–496. [Google Scholar] [CrossRef] [PubMed]




© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kalantarifard, A.; Saateh, A.; Elbuken, C. Label-Free Sensing in Microdroplet-Based Microfluidic Systems. Chemosensors 2018, 6, 23. https://doi.org/10.3390/chemosensors6020023
Kalantarifard A, Saateh A, Elbuken C. Label-Free Sensing in Microdroplet-Based Microfluidic Systems. Chemosensors. 2018; 6(2):23. https://doi.org/10.3390/chemosensors6020023
Chicago/Turabian StyleKalantarifard, Ali, Abtin Saateh, and Caglar Elbuken. 2018. "Label-Free Sensing in Microdroplet-Based Microfluidic Systems" Chemosensors 6, no. 2: 23. https://doi.org/10.3390/chemosensors6020023
APA StyleKalantarifard, A., Saateh, A., & Elbuken, C. (2018). Label-Free Sensing in Microdroplet-Based Microfluidic Systems. Chemosensors, 6(2), 23. https://doi.org/10.3390/chemosensors6020023






