A Mathematical Model for Ovine Brucellosis during Dynamic Transportation of Sheep, and Its Applications in Jalaid Banner and Ulanhot City
Abstract
1. Introduction
2. Dynamical Model
- ()
- The susceptible sheep could be infected through touching the exposed and infected sheep, or brucella in the environment.
- ()
- Susceptible people could be infected by exposure to contaminated environments and exposed and infected sheep, but they will not be infected by people with brucellosis.
- ()
- Exposed sheep usually have no symptoms, and they will also be vaccinated. It is assumed that susceptible and exposed sheep have immunity within the validity period after vaccination, that is, they will not be infected with brucellosis.
- ()
- Since the migration of sheep is mainly directional transportation through trade, we consider the directed migration of sheep between patches, but do not consider the spatial dispersal caused by the free movement of individuals.
3. Dynamic Analysis of First Five Equation of System (1) for n = 2
3.1. The Single Patch Model without Sheep Transportation
3.2. The Two Patch Model with the Transportation of Sheep between Two Patches
- (i)
- is equivalent to ,
- (ii)
- is equivalent to .
4. Dynamic Analysis of Last Three Equations of System (1) for n = 2
4.1. The Single Patch Model without Transmission of the Humans
4.2. The Two Patch Model with the Transmission of the Humans between Two Patches
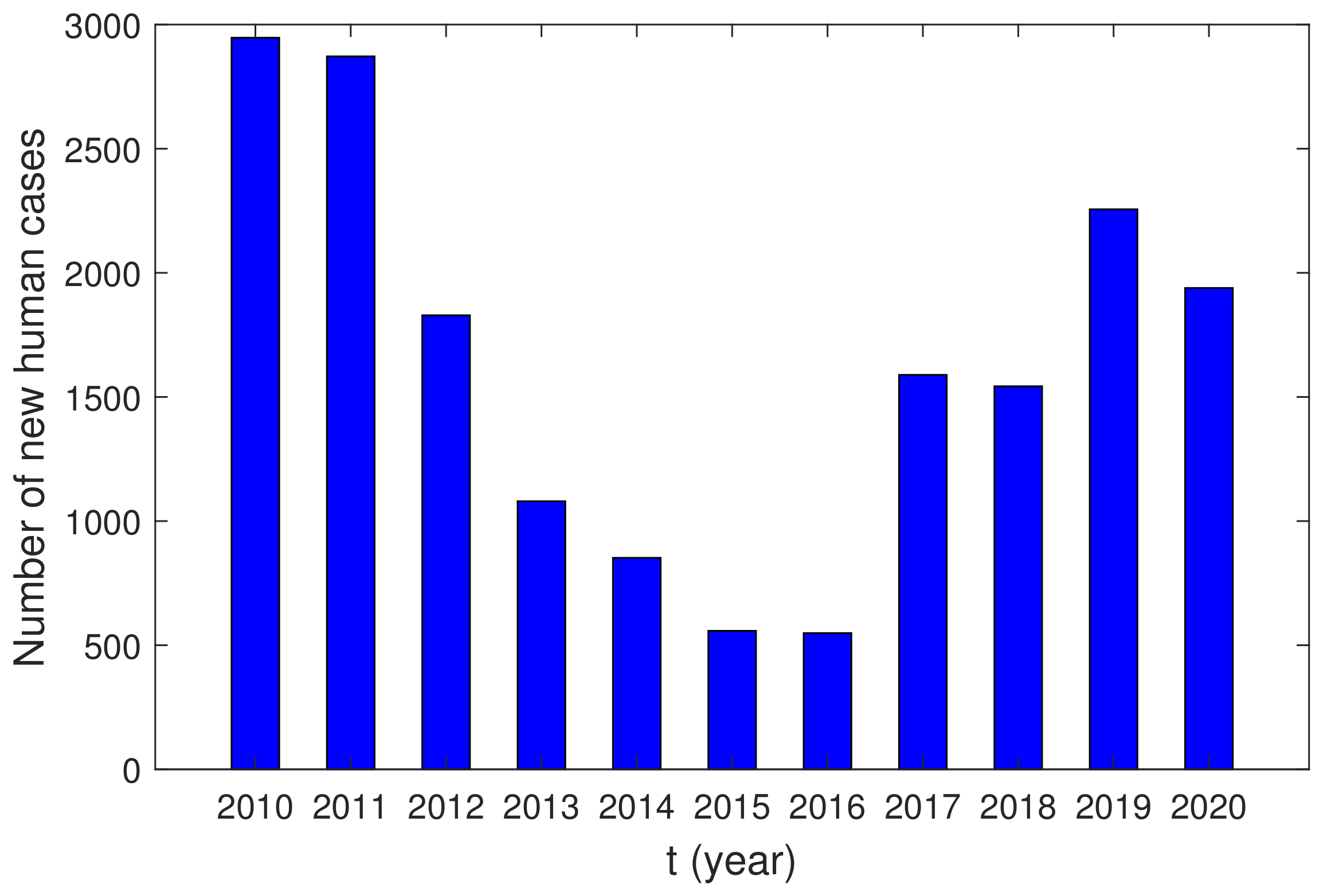
5. Case Study of Brucellosis in Ulanhot and Jalaid
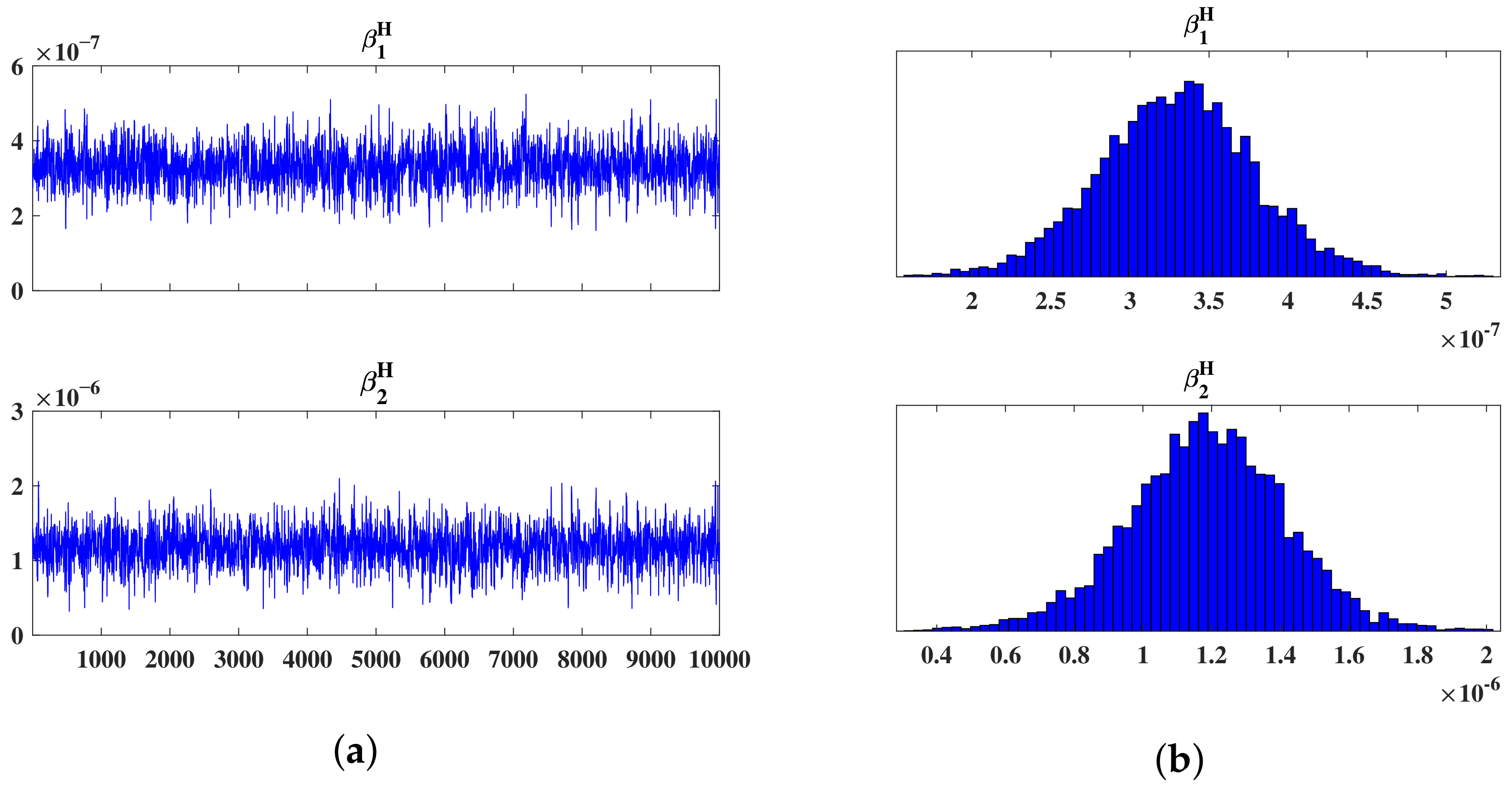
5.1. Parameter Estimation
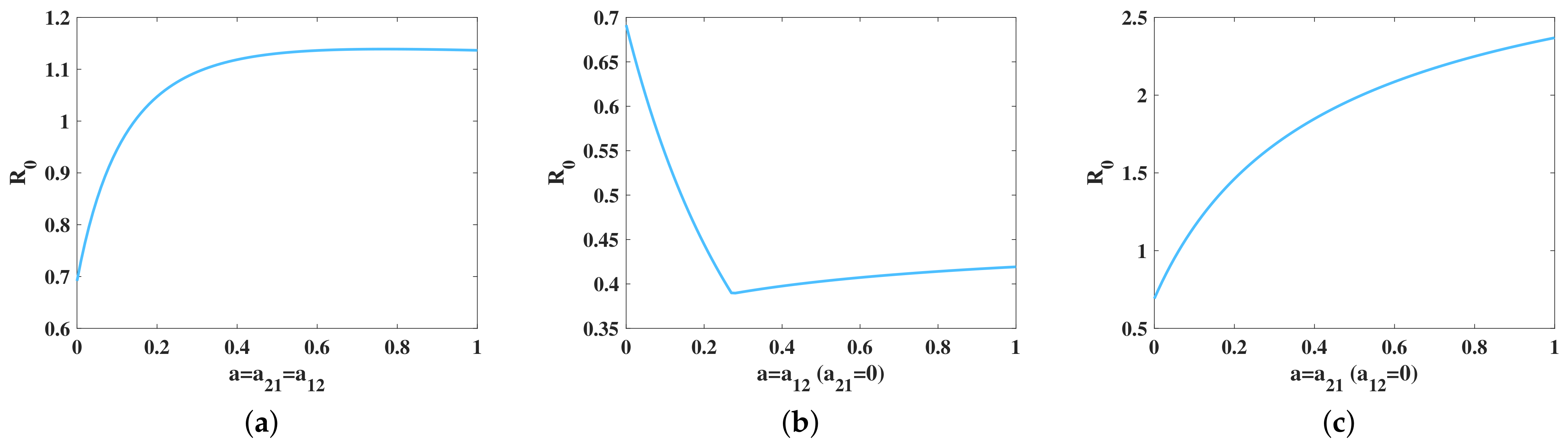
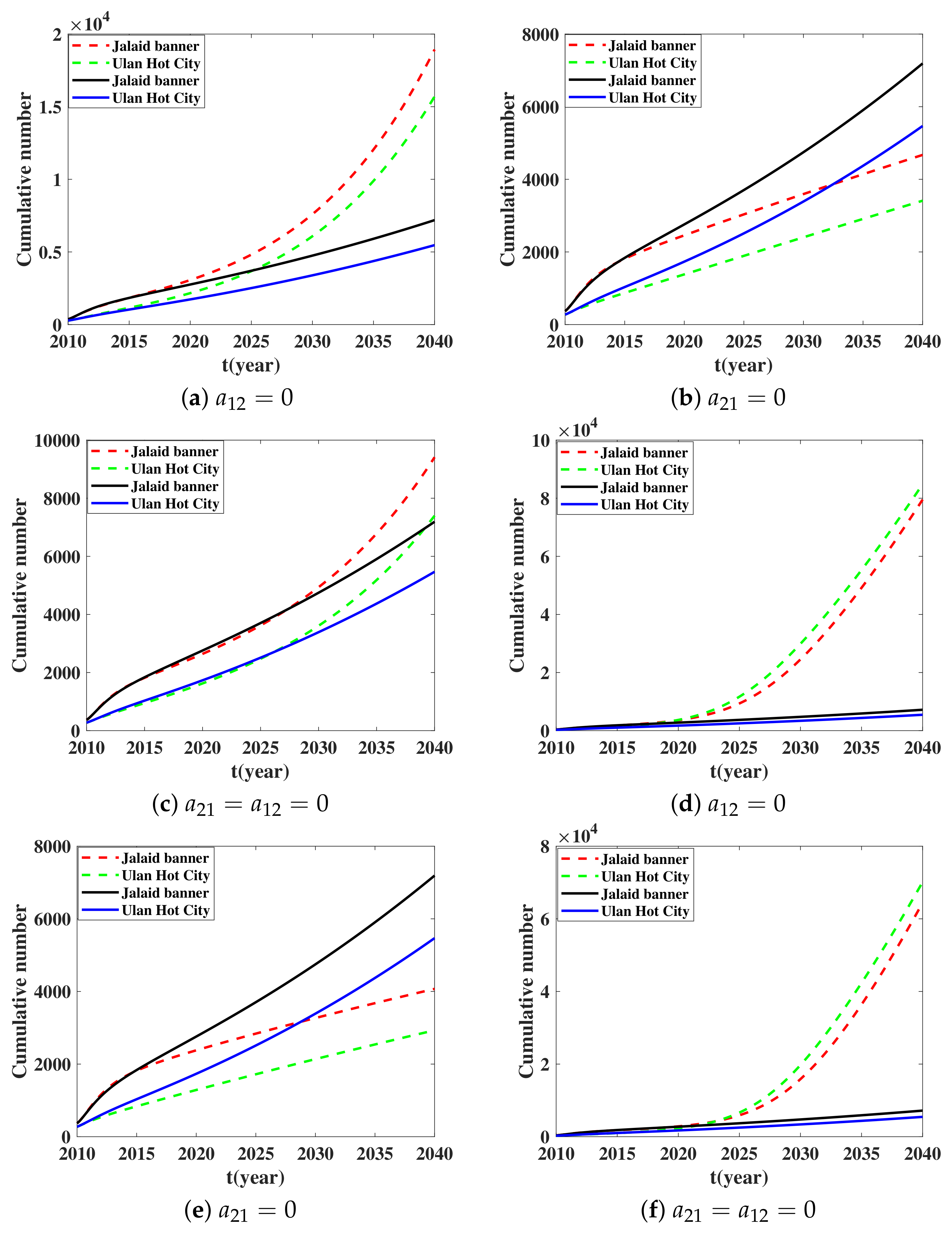
5.2. Influence of Transportation Restriction on Brucellosis Transmission Dynamic
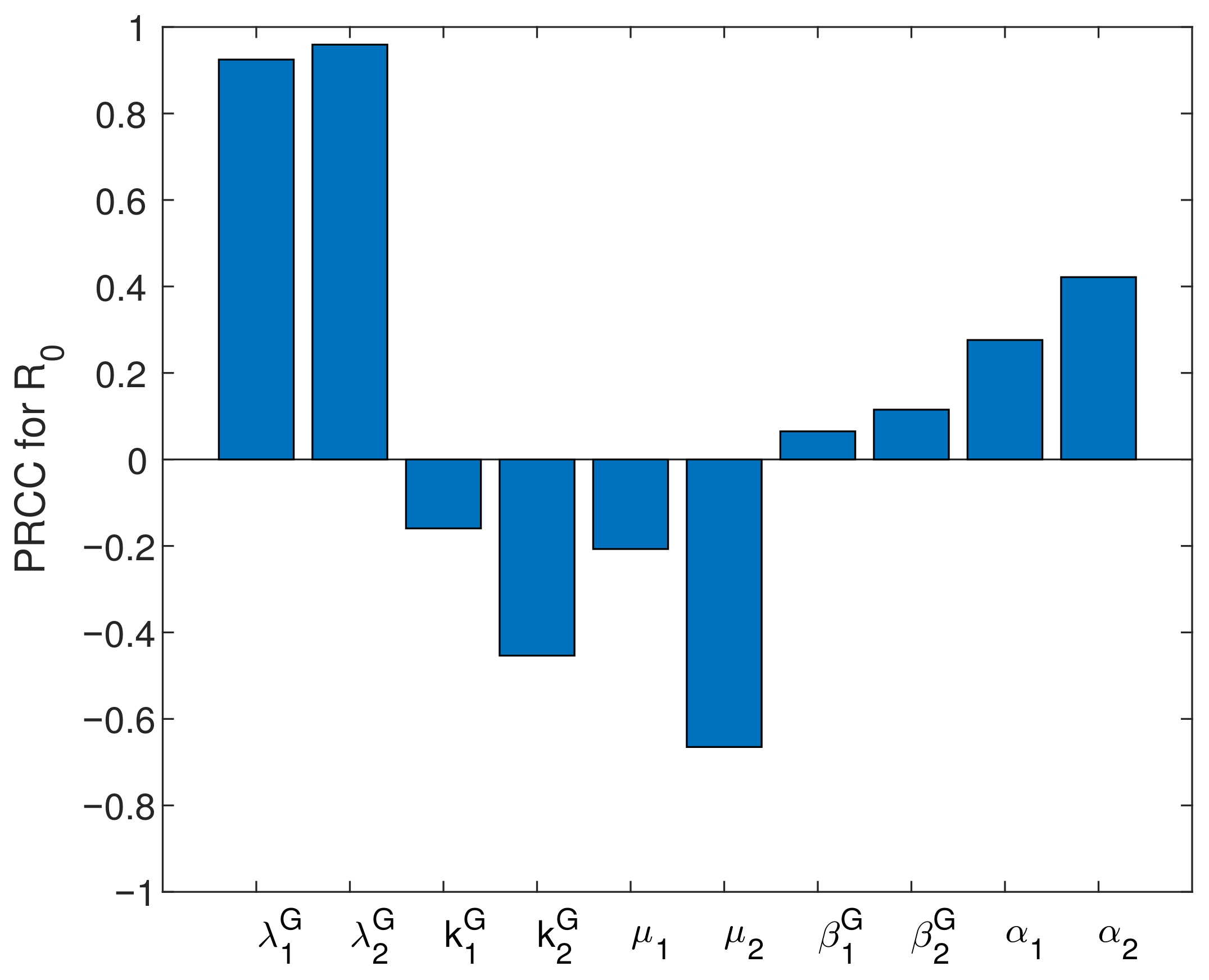
5.3. Sensitivity Analysis
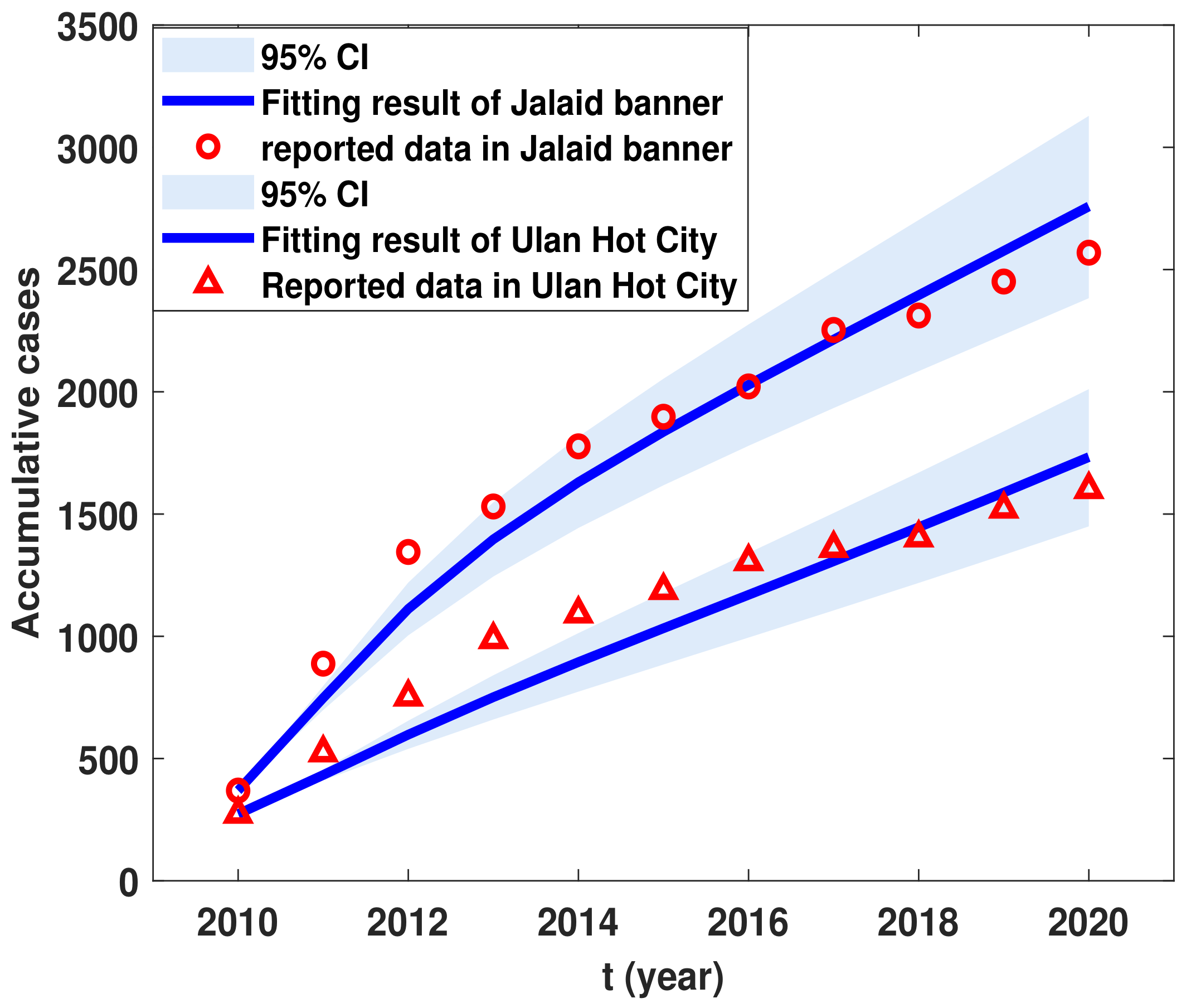
6. Validation of Theories
7. Discussion
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Boschiroli, M.L.; Foulongne, V.; O’Callaghan, D. Brucellosis: A worldwide zoonosis. Curr. Opin. Microbiol. 2001, 4, 58–64. [Google Scholar] [CrossRef]
- Sun, G.; Li, M.; Zhang, J.; Zhang, W.; Pei, X.; Jin, Z. Transmission dynamics of brucellosis: Mathematical modelling and applications in China. Comput. Struct. Biotechnol. J. 2020, 18, 3843–3860. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.T.; Wang, C.; Liu, L.N.; Wang, D.; Li, D.; Li, Z.; Liu, Z. Epidemiologically characteristics of human brucellosis and antimicrobial susceptibility pattern of Brucella melitensis in Hinggan League of the Inner Mongolia Autonomous Region, China. Infect. Dis. Poverty 2020, 9, 79. [Google Scholar] [CrossRef] [PubMed]
- Memish, Z.A.; Mah, M.W.; Mahmoud, S.A.; Shaalan, M.A.; Khan, M.Y. Brucella bacteraemia: Clinical and laboratory observations in 160 patients. J. Infect. 2000, 40, 59–63. [Google Scholar] [CrossRef]
- Jia, B.; Zhang, F.; Lu, Y.; Zhang, W.; Li, J.; Zhang, Y.; Ding, J. The clinical features of 590 patients with brucellosis in Xinjiang, China with the emphasis on the treatment of complications. PLoS Neglected Trop. Dis. 2017, 11, e0005577. [Google Scholar] [CrossRef]
- Singh, B.B.; Khatkar, M.S.; Aulakh, R.S.; Gill, J.P.S.; Dhand, N.K. Estimation of the health and economic burden of human brucellosis in India. Prev. Vet. Med. 2018, 154, 148–155. [Google Scholar] [CrossRef]
- Ciftci, G.; Yigit, Ö.; Çiftçi, A. The effects of the conjunctival Brucella vaccine on some biochemical parameters in sheep. Trop. Anim. Health Prod. 2018, 51, 355–361. [Google Scholar] [CrossRef]
- Daugaliyeva, A.; Sultanov, A.A.; Usserbayev, B.; Baramova, S.A.; Modesto, P.; Adambayeva, A.; Acutis, P.L.; Peletto, S. Genotyping of Brucella melitensis and Brucella abortus strains in Kazakhstan using MLVA-15. Infect. Genet. Evol. J. Mol. Epidemiol. Evol. Genet. Infect. Dis. 2018, 58, 135–144. [Google Scholar] [CrossRef]
- Luo, X.F.; Jin, Z.; He, D.; Li, L. The impact of contact patterns of sexual networks on Zika virus spread: A case study in Costa Rica. Appl. Math. Comput. 2021, 393, 125765. [Google Scholar] [CrossRef]
- Zhang, W.; Zhang, J.; Wu, Y.P.; Li, L. Dynamical analysis of the SEIB model for Brucellosis transmission to the dairy cows with immunological threshold. Complexity 2019, 2019, 6526589. [Google Scholar] [CrossRef]
- Liang, J.; Liu, C.; Sun, G.Q.; Li, L.; Zhang, L.; Hou, M.; Wang, H.; Wang, Z. Nonlocal interactions between vegetation induce spatial patterning. Appl. Math. Comput. 2022, 428, 127061. [Google Scholar] [CrossRef]
- Li, J.; Sun, G.Q.; Jin, Z. Interactions of time delay and spatial diffusion induce the periodic oscillation of the vegetation system. Discret. Contin. Dyn. Syst.-B 2022, 27, 2147. [Google Scholar] [CrossRef]
- Sun, G.Q.; Zhang, H.T.; Song, Y.L.; Li, L.; Jin, Z. Dynamic analysis of a plant-water model with spatial diffusion. J. Differ. Equ. 2022, 329, 395–430. [Google Scholar] [CrossRef]
- Sun, G.Q.; Zhang, H.T.; Chang, L.L.; Jin, Z.; Wang, H.; Ruan, S. On the Dynamics of a Diffusive Foot-and-Mouth Disease Model with Nonlocal Infections. SIAM J. Appl. Math. 2022, 82, 1587–1610. [Google Scholar] [CrossRef]
- Zhang, J.; Sun, G.; Sun, X.; Hou, Q.; Li, M.; Huang, B.; Wang, H.; Jin, Z. Prediction and Control of Brucellosis Transmission of Dairy Cattle in Zhejiang Province, China. PLoS ONE 2014, 9, e108592. [Google Scholar] [CrossRef] [PubMed]
- Hou, Q.; Sun, X.; Zhang, J.; Liu, Y.; Wang, Y.; Jin, Z. Modeling the transmission dynamics of sheep brucellosis in Inner Mongolia Autonomous Region, China. Math. Biosci. 2013, 242 1, 51–58. [Google Scholar] [CrossRef]
- Sun, G.Q.; Zhang, Z.K. Global stability for a sheep brucellosis model with immigration. Appl. Math. Comput. 2014, 246, 336–345. [Google Scholar] [CrossRef]
- Zhang, J.; Ruan, S.; Sun, G.; Sun, X.; Jin, Z. Analysis of a multi - patch dynamical model about cattle brucellosis. J. Shanghai Norm. Univ. Nat. Sci. Math. 2014, 43, 441–455. [Google Scholar]
- Li, M.; Sun, G.; Zhang, J.; Jin, Z.; Sun, X.; Wang, Y.; Huang, B.; Zheng, Y. Transmission dynamics and control for a brucellosis model in Hinggan League of Inner Mongolia, China. Math. Biosci. Eng. 2014, 11, 1115. [Google Scholar] [CrossRef]
- Liu, J.; Jia, Y.; Zhang, T. Analysis of a rabies transmission model with population dispersal. Nonlinear Anal. Real World Appl. 2017, 35, 229–249. [Google Scholar] [CrossRef]
- Zhong, Z.; Yu, S.; Wang, X.; Dong, S.; Xu, J.; Wang, Y.; Chen, Z.; Ren, Z.; Peng, G. Human brucellosis in the People’s Republic of China during 2005–2010. Int. J. Infect. Dis. 2013, 17 5, e289–e292. [Google Scholar] [CrossRef]
- Zhang, W.Y.; Guo, W.D.; Sun, S.H.; Jiang, J.F.; Sun, H.L.; Li, S.L.; Liu, W.; Cao, W.C. Human brucellosis, Inner Mongolia, China. Emerg. Infect. Dis. 2010, 16, 2001. [Google Scholar] [CrossRef] [PubMed]
- Tao, Z.; Chen, Q.; Chen, Y.; Li, Y.; Mu, D.; Yang, H.; Yin, W. Epidemiological characteristics of human brucellosis—China, 2016– 2019. China CDC Wkly. 2021, 3, 114. [Google Scholar] [PubMed]
- Ma, S.; Liu, Z.; Zhu, X.; Zhao, Z.Z.; Guo, Z.; Wang, M.; Cui, B.Y.; Li, J.Y.; Li, Z. Molecular epidemiology of Brucella abortus strains from cattle in Inner Mongolia, China. Prev. Vet. Med. 2020, 183, 105080. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Liu, R. Hinggan League 2001–2009 human brucellosis epidemiological analysis(in chinese). Med. Inf. 2010, 5, 473–474. [Google Scholar]
- Available online: https://www.phsciencedata.cn/Share/edtShareNew.jsp?id=39308 (accessed on 3 June 2022).
- van den Driessche, P.; Watmough, J. Reproduction numbers and sub-threshold endemic equilibria for compartmental models of disease transmission. Math. Biosci. 2002, 180, 29–48. [Google Scholar] [CrossRef]
- Smith, H.L.; Waltman, P. The Theory of the Chemostat: Dynamics of Microbial Competition; Cambridge University Press: Cambridge, UK, 1995; Volume 13. [Google Scholar]
- Thieme, H.R. Convergence results and a Poincaré-Bendixson trichotomy for asymptotically autonomous differential equations. J. Math. Biol. 1992, 30, 755–763. [Google Scholar] [CrossRef]
- Li, M.T.; Sun, G.Q.; Wu, Y.F.; Zhang, J.; Jin, Z. Transmission dynamics of a multi-group brucellosis model with mixed cross infection in public farm. Appl. Math. Comput. 2014, 237, 582–594. [Google Scholar] [CrossRef]
- Thieme, H.R. Persistence under relaxed point-dissipativity (with application to an endemic model). Siam J. Math. Anal. 1993, 24, 407–435. [Google Scholar] [CrossRef]
- Zhao, X.Q. Uniform persistence and periodic coexistence states in infinite-dimensional periodic semiflows with applications. Canad. Appl. Math. Quart 1995, 3, 473–495. [Google Scholar]
- Li, J.; Sun, G.Q.; Guo, Z.G. Bifurcation analysis of an extended Klausmeier–Gray–Scott model with infiltration delay. Stud. Appl. Math. 2022, 148, 1519–1542. [Google Scholar] [CrossRef]
- Pan, Z.; Wang, Z.Q.; Xia, R.H.; Tian, X.R.; Shi, R.L.; Zhang, J.H.; Fen, Z.Z.; Li, S.Q.; Qian, H.W.; Wang, F.; et al. 2015–2020 Inner Mongolia Bureau of Statistics Yearbook; China Statistics Press: Beijing, China, 2020. [Google Scholar]
- Li, Y.; Li, Y.J.; Mou, Y.J.; Niu, X.H.; Bi, Y.B.; Zhang, T.Z.; Liu, X.G.; Xu, Y.; Li, X.H.; Ma, N.; et al. 2015–2020 Hinggan League of Statistics Yearbook; China Statistics Press: Beijing, China, 2020. [Google Scholar]
- Sun, T.; Wu, Z.; Pang, X. Prevention measures and countermeasures on brucellosis in Inner Mongolia. Neimenggu Prev. Med. 2000, 1, 136–139. [Google Scholar]
- Mi, J.; Zhang, Q.; Wei, R.; Song, L.; Zheng, Z. The epidemiological characteristics of human brucellosis in Inner Mongolia. Chin. J. Control Endem. Dis. 2010, 25, 34–36. [Google Scholar]
- The National Brucellosis Control Plan (2016–2020). Available online: http://dkxy.shzu.edu.cn/2016/1122/c3182a85716/page.htm (accessed on 3 June 2022).
- Haario, H.; Laine, M.; Mira, A.; Saksman, E. DRAM: Efficient adaptive MCMC. Stat. Comput. 2006, 16, 339–354. [Google Scholar] [CrossRef]
- Gamerman, D.; Lopes, H.F. Markov Chain Monte Carlo: Stochastic Simulation for Bayesian Inference; CRC Press: Boca Raton, FL, USA, 2006. [Google Scholar]
- Marino, S.; Hogue, I.B.; Ray, C.J.; Kirschner, D.E. A methodology for performing global uncertainty and sensitivity analysis in systems biology. J. Theor. Biol. 2008, 254, 178–196. [Google Scholar] [CrossRef]
- Ma, X.; Luo, X.F.; Li, L.; Li, Y.; Sun, G.Q. The influence of mask use on the spread of COVID-19 during pandemic in New York City. Results Phys. 2022, 34, 105224. [Google Scholar] [CrossRef]
- Jiang, H.; O’Callaghan, D.; Ding, J.B. Brucellosis in China: History, progress and challenge. Infect. Dis. Poverty 2020, 9, 101–104. [Google Scholar] [CrossRef] [PubMed]
- Guan, P.; Wu, W.; Huang, D. Trends of reported human brucellosis cases in mainland China from 2007 to 2017: An exponential smoothing time series analysis. Environ. Health Prev. Med. 2018, 23, 1–7. [Google Scholar] [CrossRef]
- Wang, H.; Liu, H.; Zhang, Q.; Lu, X.; Li, D.; Zhang, H.; Wang, Y.A.; Zheng, R.; Zhang, Y.; Fu, Z.; et al. Natural History of and Dynamic Changes in Clinical Manifestation, Serology, and Treatment of Brucellosis, China. Emerg. Infect. Dis. 2022, 28, 1460. [Google Scholar] [CrossRef]


| Parameters | Description |
|---|---|
| the annual birth rate of sheep in patch i | |
| the annual birth rate of humans in patch i | |
| the loss rate of vaccination immunity for sheep in patch i | |
| the transmission coefficient of infectious sheep to susceptible sheep in patch i | |
| the transmission coefficient of infectious sheep to susceptible humans in patch i | |
| the transmission coefficient of polluted environment to susceptible sheep in patch i | |
| natural death and slaughter elimination rate coefficient of sheep in patch i | |
| vaccination rate of sheep | |
| the natural death rate of people in patch i | |
| the rate of clinical outcome of exposed sheep in patch i | |
| disease-related culling rate of infectious sheep in patch i | |
| the amount of brucella per unit time emitted by exposed and infected sheep | |
| brucella decay rate in patch i | |
| the transmission rate coefficient of brucella in environment-to-susceptible human in patch i | |
| the rate of clinical outcome of exposed human in patch i | |
| the migration rate of sheep from patch j to patch i () | |
| the migration rate of people from patch j to patch i () |
| Parameter | Mean Value | Unit | Source |
|---|---|---|---|
| 489,992 | year | [B] | |
| 135,776 | year | [B] | |
| 3840 | year | [A] | |
| 214 | year | [A] | |
| 1/3 | year | [C] | |
| 1/3 | year | [C] | |
| year | [C] | ||
| year | [C] | ||
| year | MCMC | ||
| year | MCMC | ||
| year | [C] | ||
| year | [C] | ||
| 0.3385 | year | [B] | |
| 0.4873 | year | [B] | |
| 0.316 | year | [C] | |
| 0.316 | year | [C] | |
| 7.23‰ | year | [A] | |
| 6‰ | year | [A] | |
| 1 | year | [C] | |
| 1 | year | [C] | |
| 1 | year | [A] | |
| 1 | year | [A] | |
| 15 | year | [C] | |
| 15 | year | [C] | |
| 3.6 | year | [C] | |
| 3.6 | year | [C] | |
| year | [C] | ||
| year | [C] | ||
| 1 | year | [C] | |
| 1 | year | [C] | |
| 50.71% | year | [C] | |
| 40.92% | year | [C] | |
| 27.89% | year | [C] | |
| 5.36% | year | [C] |
| Parameter | Mean Value | Standard | MC Error | Geweke |
|---|---|---|---|---|
| 3.3433 × 10−7 | 4.8114 × 10−8 | 1.1761 × 10−9 | 0.99557 | |
| 1.1681 × 10−6 | 2.2907 × 10−7 | 5.6932 × 10−9 | 0.99545 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, J.; Luo, X.; Zhang, J.; Li, M. A Mathematical Model for Ovine Brucellosis during Dynamic Transportation of Sheep, and Its Applications in Jalaid Banner and Ulanhot City. Mathematics 2022, 10, 3436. https://doi.org/10.3390/math10193436
Guo J, Luo X, Zhang J, Li M. A Mathematical Model for Ovine Brucellosis during Dynamic Transportation of Sheep, and Its Applications in Jalaid Banner and Ulanhot City. Mathematics. 2022; 10(19):3436. https://doi.org/10.3390/math10193436
Chicago/Turabian StyleGuo, Jiaming, Xiaofeng Luo, Juan Zhang, and Mingtao Li. 2022. "A Mathematical Model for Ovine Brucellosis during Dynamic Transportation of Sheep, and Its Applications in Jalaid Banner and Ulanhot City" Mathematics 10, no. 19: 3436. https://doi.org/10.3390/math10193436
APA StyleGuo, J., Luo, X., Zhang, J., & Li, M. (2022). A Mathematical Model for Ovine Brucellosis during Dynamic Transportation of Sheep, and Its Applications in Jalaid Banner and Ulanhot City. Mathematics, 10(19), 3436. https://doi.org/10.3390/math10193436





