Isolation and Characterization of Serum Extracellular Vesicles (EVs) from Atlantic Salmon Infected with Piscirickettsia Salmonis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Bacteria, Media, and Growth Conditions
2.2. Fish Husbandry and Challenge
2.3. Serum Exosome Isolation
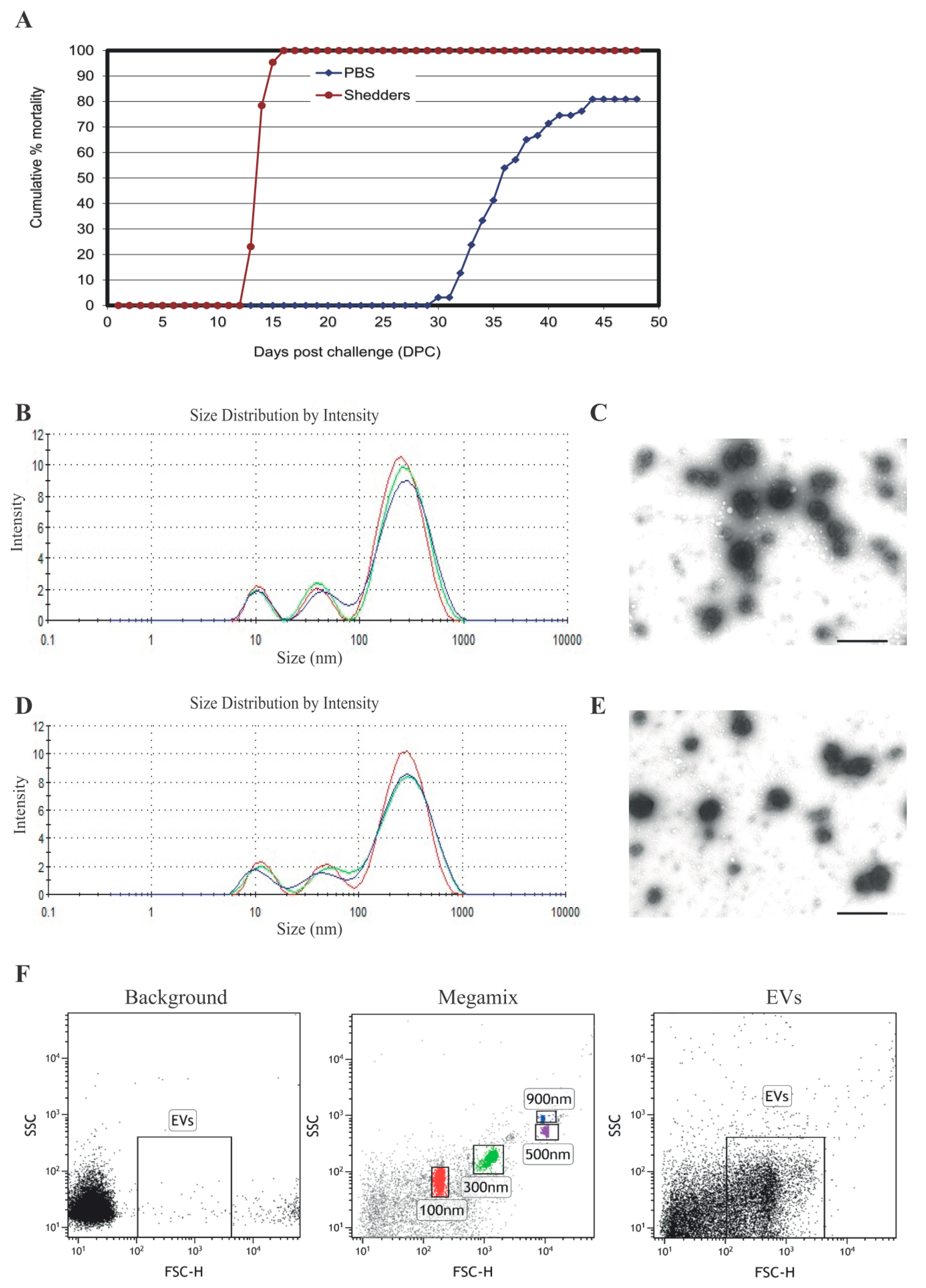
2.4. Nanoparticle Tracking Analysis
2.5. Flow Cytometry
2.6. Transmission Electron Microscopy (TEM)
2.7. In-Solution Digestion and Protein Sequence Analysis by LC-MS/MS
2.8. Proteomic Data Analysis
2.9. Ethics Statement
3. Results
3.1. Serum EV Characterization
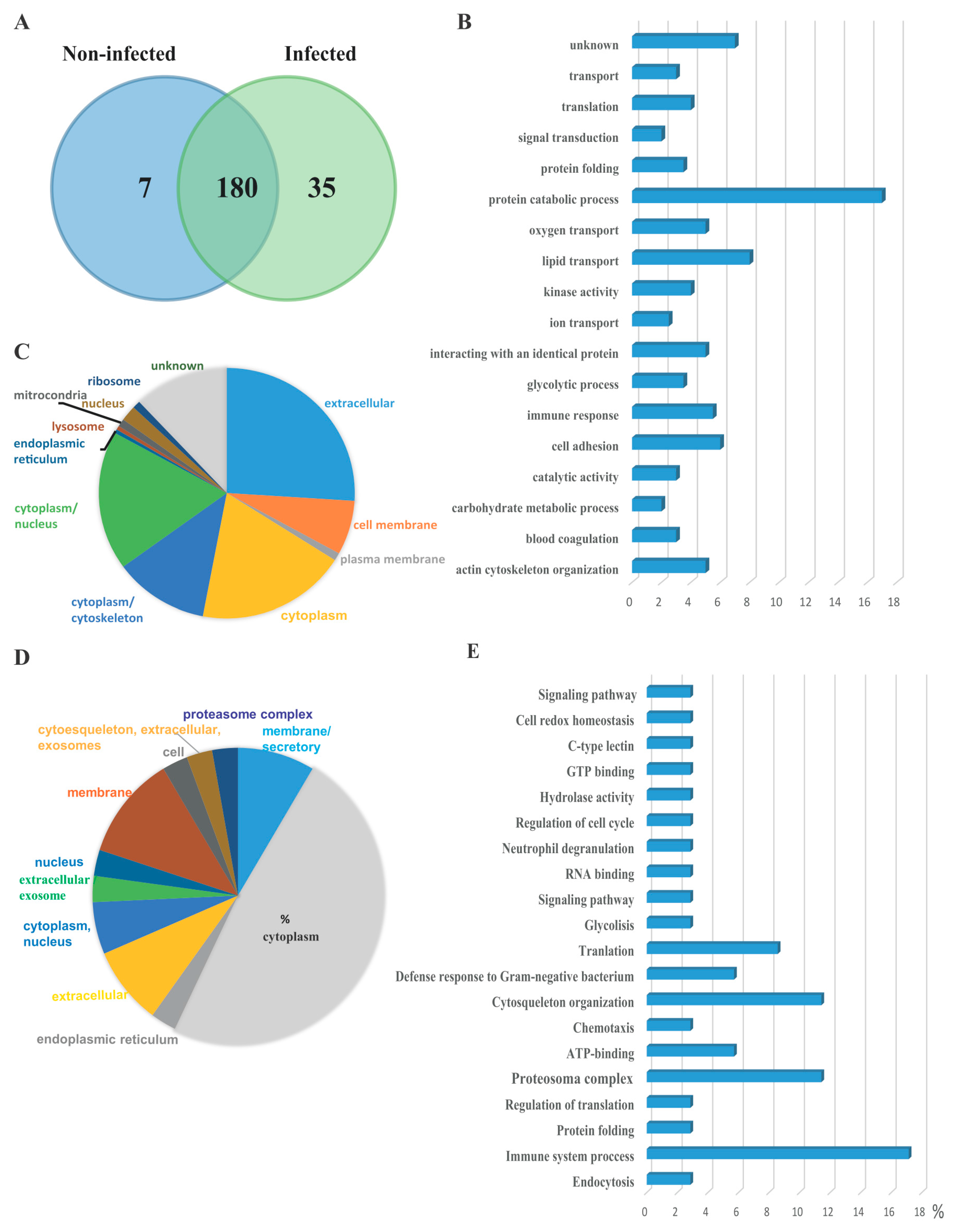
3.2. Serum EVs Proteome
4. Discussion
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Garcia-Contreras, M.; Brooks, R.W.; Boccuzzi, L.; Robbins, P.D.; Ricordi, C. Exosomes as biomarkers and therapeutic tools for type 1 diabetes mellitus. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 2940–2956. [Google Scholar] [PubMed]
- Zhang, W.; Ni, M.; Su, Y.; Wang, H.; Zhu, S.; Zhao, A.; Li, G. Micrornas in serum exosomes as potential biomarkers in clear-cell renal cell carcinoma. Eur. Urol. Focus 2016, in press. [Google Scholar] [CrossRef] [PubMed]
- Lawson, C.; Kovacs, D.; Finding, E.; Ulfelder, E.; Luis-Fuentes, V. Extracellular vesicles: Evolutionarily conserved mediators of intercellular communication. Yale J. Biol. Med. 2017, 90, 481–491. [Google Scholar] [PubMed]
- Hessvik, N.P.; Llorente, A. Current knowledge on exosome biogenesis and release. Cell. Mol. Life Sci. CMLS 2017. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Rana, S.; Giese, N.; Buchler, M.W.; Zoller, M. Tspan8, cd44v6 and alpha6beta4 are biomarkers of migrating pancreatic cancer-initiating cells. Int. J. Cancer 2013, 133, 416–426. [Google Scholar] [CrossRef] [PubMed]
- Andreu, Z.; Yanez-Mo, M. Tetraspanins in extracellular vesicle formation and function. Front. Immunol. 2014, 5, 442. [Google Scholar] [CrossRef] [PubMed]
- Thery, C.; Boussac, M.; Veron, P.; Ricciardi-Castagnoli, P.; Raposo, G.; Garin, J.; Amigorena, S. Proteomic analysis of dendritic cell-derived exosomes: A secreted subcellular compartment distinct from apoptotic vesicles. J. Immunol. 2001, 166, 7309–7318. [Google Scholar] [CrossRef] [PubMed]
- McCready, J.; Sims, J.D.; Chan, D.; Jay, D.G. Secretion of extracellular hsp90alpha via exosomes increases cancer cell motility: A role for plasminogen activation. BMC Cancer 2010, 10, 294. [Google Scholar] [CrossRef] [PubMed]
- Iliev, D.B.; Jorgensen, S.M.; Rode, M.; Krasnov, A.; Harneshaug, I.; Jorgensen, J.B. Cpg-induced secretion of mhciibeta and exosomes from salmon (Salmo salar) apcs. Dev. Comp. Immunol. 2010, 34, 29–41. [Google Scholar] [CrossRef] [PubMed]
- Haraszti, R.A.; Didiot, M.C.; Sapp, E.; Leszyk, J.; Shaffer, S.A.; Rockwell, H.E.; Gao, F.; Narain, N.R.; DiFiglia, M.; Kiebish, M.A.; et al. High-resolution proteomic and lipidomic analysis of exosomes and microvesicles from different cell sources. J. Extracell. Vesicles 2016, 5, 32570. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, K. The proteasome: Overview of structure and functions. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2009, 85, 12–36. [Google Scholar] [CrossRef] [PubMed]
- Seiliez, I.; Dias, K.; Cleveland, B.M. Contribution of the autophagy-lysosomal and ubiquitin-proteasomal proteolytic systems to total proteolysis in rainbow trout (Oncorhynchus mykiss) myotubes. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2014, 307, R1330–R1337. [Google Scholar] [CrossRef] [PubMed]
- Gogliettino, M.; Balestrieri, M.; Riccio, A.; Facchiano, A.; Fusco, C.; Palazzo, V.C.; Rossi, M.; Cocca, E.; Palmieri, G. Uncommon functional properties of the first piscine 26s proteasome from the antarctic notothenioid trematomus bernacchii. Biosci. Rep. 2016, 36, e00320. [Google Scholar] [CrossRef] [PubMed]
- Wowk, P.F.; Zardo, M.L.; Miot, H.T.; Goldenberg, S.; Carvalho, P.C.; Morking, P.A. Proteomic profiling of extracellular vesicles secreted from toxoplasma gondii. Proteomics 2017, 17. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Schorey, J.S. Exosomes carrying mycobacterial antigens can protect mice against mycobacterium tuberculosis infection. Eur. J. Immunol. 2013, 43, 3279–3290. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, S.; Grace, P.S.; Ernst, J.D. Antigen export reduces antigen presentation and limits t cell control of m. Tuberculosis. Cell Host Microbe 2016, 19, 44–54. [Google Scholar] [CrossRef] [PubMed]
- Saavedra, J.; Hernandez, N.; Osses, A.; Castillo, A.; Cancino, A.; Grothusen, H.; Navas, E.; Henriquez, P.; Bohle, H.; Bustamante, F.; et al. Prevalence, geographic distribution and phenotypic differences of piscirickettsia salmonis em-90-like isolates. J. Fish Dis. 2017, 40, 1055–1063. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, U.M.; Bron, J.E.; Brown, L.; Pourahmad, F.; Bricknell, I.R.; Thompson, K.D.; Adams, A.; Ellis, A.E. Survival and replication of piscirickettsia salmonis in rainbow trout head kidney macrophages. Fish Shellfish Immunol. 2008, 25, 477–484. [Google Scholar] [CrossRef] [PubMed]
- Ramirez, R.; Gomez, F.A.; Marshall, S.H. The infection process of piscirickettsia salmonis in fish macrophages is dependent upon interaction with host-cell clathrin and actin. FEMS Microbiol. Lett. 2015, 362, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Gomez, F.A.; Tobar, J.A.; Henriquez, V.; Sola, M.; Altamirano, C.; Marshall, S.H. Evidence of the presence of a functional dot/icm type iv-b secretion system in the fish bacterial pathogen piscirickettsia salmonis. PLoS ONE 2013, 8, e54934. [Google Scholar] [CrossRef] [PubMed]
- Smith, V.L.; Cheng, Y.; Bryant, B.R.; Schorey, J.S. Exosomes function in antigen presentation during an in vivo mycobacterium tuberculosis infection. Sci. Rep. 2017, 7, 43578. [Google Scholar] [CrossRef] [PubMed]
- Tandberg, J.; Oliver, C.; Lagos, L.; Gaarder, M.; Yanez, A.J.; Ropstad, E.; Winther-Larsen, H.C. Membrane vesicles from piscirickettsia salmonis induce protective immunity and reduce development of salmonid rickettsial septicemia in an adult zebrafish model. Fish Shellfish Immunol. 2017, 67, 189–198. [Google Scholar] [CrossRef] [PubMed]
- Vizcaino, J.A.; Csordas, A.; Del-Toro, N.; Dianes, J.A.; Griss, J.; Lavidas, I.; Mayer, G.; Perez-Riverol, Y.; Reisinger, F.; Ternent, T.; et al. 2016 update of the pride database and its related tools. Nucleic Acids Res. 2016, 44, 11033. [Google Scholar] [CrossRef] [PubMed]
- Lotvall, J.; Hill, A.F.; Hochberg, F.; Buzas, E.I.; Di Vizio, D.; Gardiner, C.; Gho, Y.S.; Kurochkin, I.V.; Mathivanan, S.; Quesenberry, P.; et al. Minimal experimental requirements for definition of extracellular vesicles and their functions: A position statement from the international society for extracellular vesicles. J. Extracell. Vesicles 2014, 3, 26913. [Google Scholar] [CrossRef] [PubMed]
- Mehaffy, C.; Dobos, K.M.; Nahid, P.; Kruh-Garcia, N.A. Second generation multiple reaction monitoring assays for enhanced detection of ultra-low abundance mycobacterium tuberculosis peptides in human serum. Clin. Proteom. 2017, 14, 21. [Google Scholar] [CrossRef] [PubMed]
- Mauel, M.J.; Miller, D.L. Piscirickettsiosis and piscirickettsiosis-like infections in fish: A review. Vet. Microbiol. 2002, 87, 279–289. [Google Scholar] [CrossRef]
- De Chastellier, C.; Berche, P. Fate of listeria monocytogenes in murine macrophages: Evidence for simultaneous killing and survival of intracellular bacteria. Infect. Immun. 1994, 62, 543–553. [Google Scholar] [PubMed]
- Rojas, V.; Galanti, N.; Bols, N.C.; Jimenez, V.; Paredes, R.; Marshall, S.H. Piscirickettsia salmonis induces apoptosis in macrophages and monocyte-like cells from rainbow trout. J. Cell. Biochem. 2010, 110, 468–476. [Google Scholar] [PubMed]
- Akiyama, K.; Kagawa, S.; Tamura, T.; Shimbara, N.; Takashina, M.; Kristensen, P.; Hendil, K.B.; Tanaka, K.; Ichihara, A. Replacement of proteasome subunits x and y by lmp7 and lmp2 induced by interferon-gamma for acquirement of the functional diversity responsible for antigen processing. FEBS Lett. 1994, 343, 85–88. [Google Scholar] [CrossRef]
- Ferrington, D.A.; Gregerson, D.S. Immunoproteasomes: Structure, function, and antigen presentation. Prog. Mol. Biol. Transl. Sci. 2012, 109, 75–112. [Google Scholar] [PubMed]
- Kaur, G.; Batra, S. Emerging role of immunoproteasomes in pathophysiology. Immunol. Cell Biol. 2016, 94, 812–820. [Google Scholar] [CrossRef] [PubMed]
- Eskandari, S.K.; Seelen, M.A.J.; Lin, G.; Azzi, J.R. The immunoproteasome: An old player with a novel and emerging role in alloimmunity. Am. J. Transplant. 2017, 17, 3033–3039. [Google Scholar] [CrossRef] [PubMed]
- Tacchi, L.; Bron, J.E.; Taggart, J.B.; Secombes, C.J.; Bickerdike, R.; Adler, M.A.; Takle, H.; Martin, S.A. Multiple tissue transcriptomic responses to piscirickettsia salmonis in atlantic salmon (Salmo salar). Physiol. Genom. 2011, 43, 1241–1254. [Google Scholar] [CrossRef] [PubMed]
- Cui, Z.; Hwang, S.M.; Gomes, A.V. Identification of the immunoproteasome as a novel regulator of skeletal muscle differentiation. Mol. Cell. Biol. 2014, 34, 96–109. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, A.; Maekawa, Y.; Uehara, H.; Izumi, K.; Kawachi, I.; Nishizawa, M.; Toyoshima, Y.; Takahashi, H.; Standley, D.M.; Tanaka, K.; et al. A mutation in the immunoproteasome subunit psmb8 causes autoinflammation and lipodystrophy in humans. J. Clin. Investig. 2011, 121, 4150–4160. [Google Scholar] [CrossRef] [PubMed]
- Hanington, P.C.; Barreda, D.R.; Belosevic, M. A novel hematopoietic granulin induces proliferation of goldfish (Carassius auratus L.) macrophages. J. Biol. Chem. 2006, 281, 9963–9970. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Li, Y.; Li, X.; Wei, L.L.; Pan, Z.; Jiang, T.T.; Chen, Z.L.; Wang, C.; Cao, W.M.; Zhang, X.; et al. Serum protein s100a9, sod3, and mmp9 as new diagnostic biomarkers for pulmonary tuberculosis by itraq-coupled two-dimensional lc-ms/ms. Proteomics 2015, 15, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Gunaldi, M.; Okuturlar, Y.; Gedikbasi, A.; Akarsu, C.; Karabulut, M.; Kural, A. Diagnostic importance of s100a9 and s100a12 in breast cancer. Biomed. Pharmacother. 2015, 76, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Prieto, D.; Sotelo, N.; Seija, N.; Sernbo, S.; Abreu, C.; Duran, R.; Gil, M.; Sicco, E.; Irigoin, V.; Oliver, C.; et al. S100-a9 protein in exosomes from chronic lymphocytic leukemia cells promotes nf-kappab activity during disease progression. Blood 2017, 130, 777–788. [Google Scholar] [CrossRef] [PubMed]
- Blanc, L.; Vidal, M. New insights into the function of rab gtpases in the context of exosomal secretion. Small GTPases 2017, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Machuca, A.; Martinez, V. Transcriptome analysis of the intracellular facultative pathogen piscirickettsia salmonis: Expression of putative groups of genes associated with virulence and iron metabolism. PLoS ONE 2016, 11, e0168855. [Google Scholar] [CrossRef] [PubMed]
- Tandberg, J.I.; Lagos, L.X.; Langlete, P.; Berger, E.; Rishovd, A.L.; Roos, N.; Varkey, D.; Paulsen, I.T.; Winther-Larsen, H.C. Comparative analysis of membrane vesicles from three piscirickettsia salmonis isolates reveals differences in vesicle characteristics. PLoS ONE 2016, 11, e0165099. [Google Scholar] [CrossRef] [PubMed]
- Enderle, D.; Spiel, A.; Coticchia, C.M.; Berghoff, E.; Mueller, R.; Schlumpberger, M.; Sprenger-Haussels, M.; Shaffer, J.M.; Lader, E.; Skog, J.; et al. Characterization of rna from exosomes and other extracellular vesicles isolated by a novel spin column-based method. PLoS ONE 2015, 10, e0136133. [Google Scholar] [CrossRef] [PubMed]
- Coumans, F.A.W.; Brisson, A.R.; Buzas, E.I.; Dignat-George, F.; Drees, E.E.E.; El-Andaloussi, S.; Emanueli, C.; Gasecka, A.; Hendrix, A.; Hill, A.F.; et al. Methodological guidelines to study extracellular vesicles. Circ. Res. 2017, 120, 1632–1648. [Google Scholar] [CrossRef] [PubMed]
- Faught, E.; Henrickson, L.; Vijayan, M.M. Plasma exosomes are enriched in hsp70 and modulated by stress and cortisol in rainbow trout. J. Endocrinol. 2017, 232, 237–246. [Google Scholar] [CrossRef] [PubMed]
| Protein ID | Gen | Identified Proteins | Function |
|---|---|---|---|
| B5XFB2 | blvrb | Flavin reductase | Reductase activity |
| B5X3Z5 | fgl2 | Fibroleukin | Peptidase activity |
| Q9DDI8 | gdf-8 | Myostatin 1b | Receptor binding |
| B5XF63 | natte | Nattectin | Carbohydrate binding |
| C0H8M3 | tco2 | Transcobalamin-2 | Cobalamin binding |
| B5X5M2 | fabph | Fatty acid-binding protein, heart | Transport activity |
| B9EMN9 | leca | Lectin | Ion binding |
| Protein | Identified Proteins | Function | Localization | Process |
|---|---|---|---|---|
| B5X3W8 | Galectin-3-binding protein | Receptor mediated endocytosis | membrane | Endocytosis |
| Q9DD33 | Proteasome subunit beta type-9 | Proteolysis | cytoplasm | Immune system proccess |
| B5X0V5 | Calreticulin | Calcium ion binding | endoplasmic reticulum | Protein folding |
| C0HBQ7 | Caprin-1 | RNA binding | cytoplasmic | Regulation of translation |
| A7KDZ9 | Proteasome subunit beta type | Proteolysis | cytoplasm | Proteosoma complex |
| B5RI16 | Proteasome, 26S subunit, ATPase, 6 | Protein catabolic proccess | cytoplasm | ATP-binding |
| B5X6D8 | Leukocyte cell-derived chemotaxin 2 | Protein binding | extracellular | Chemotaxis |
| B5XF14 | Proteasome subunit alpha type | Protein catabolic proccess | cytoplasm | Endopeptidase activity |
| C0PU67 | Gelsolin (Fragment) | Actin filament binding | cytoplasm | Actin nucleation |
| Q49TU5 | Cathelicidin antimicrobial peptide | Protein binding | extracellular | Defense response to Gram-negative bacterium |
| B5X0W4 | Eukaryotic translation initiation factor 3 subunit I | Translation | cytoplasm | Translation |
| A7KE01 | Proteasome subunit beta type-6-A like protein | Endopeptidase activity | cytoplasm, nucleus | Immune system proccess |
| B5RI28 | Proteasome, 26S subunit, ATPase 1a (Fragment) | Protein catabolic proccess | cytoplasm | ATP-binding |
| B5X3K2 | Glyceraldehyde-3-phosphate dehydrogenase | Oxidoreductasa | cytoplasm | Glycolisis |
| B5XAP5 | Protein S100 | Calcium binding | extracellular | Immune system process |
| B5XDU3 | 14-3-3 protein zeta | Protein domain specific binding | cytoplasm | Signaling pathway |
| C0H9S0 | Eukaryotic translation initiation factor 3 subunit A | Translation | cytoplasm | RNA binding |
| C0PUP2 | Granulins (Fragment) | Protein binding | extracellular/exosome | Neutrophil degranulation |
| B5X2B1 | Histone-binding protein RBBP4 | Protein binding | nucleus | Regulation of cell cycle |
| A2VA22 | MHC class II antigen beta chain (Fragment) | Immune response | membrane | Antigen proccessing nad presentation |
| C0PUI9 | Fructose-1,6-bisphosphatase 1 (Fragment) | Carbohydrate metabolic proccess | membrane | Hydrolase activity |
| Q5UT54 | Toll-like leucine-rich repeat protein | LPS binding | secreted or membrane components | Immune response |
| B5X141 | Ras-related protein Rab-10 | GTPase activity | cytoplasmic | GTP binding |
| B5X1G4 | Cysteinyl-tRNA synthetase | Nucleotide binding | cytoplasmic | tRNA aminoacylation for protein translation |
| B5X2E1 | Adenylyl cyclase-associated protein | Actin binding | membrane | Cytosqueleton organization |
| B5X7T8 | Type-2 ice-structuring protein | Carbohydrate binding | secreted or membrane components | C-type lectin |
| B5X8H5 | Peroxiredoxin | Antioxidant activity | cytoplasm | Cell redox homeostasis |
| B5XDE4 | 14-3-3 protein beta/alpha | Protein domain specific binding | cytoplasm | Signaling pathway |
| B9EQN9 | Plastin-2 | Calcium/actin binding | cytoplasm/cytosqueleton | Actin crosslink formation |
| H8PHI0 | Cathelicidin-derived antimicrobial peptide 1 isoform A | LPS binding | cell | Defense response to Gram-negative bacterium |
| B5X340 | Asparaginyl-tRNA synthetase, | Nucleotide binding | cytoplasmic | tRNA aminoacylation for protein translation |
| B5X383 | SUMO-activating enzyme subunit 2 | Nucleotide/metal ion binding | cytoplasmic/nucleus | Protein sumoylation |
| B5DGD5 | Actin related protein 2/3 complex subunit 2 | Actin binding | cytoesqueleton, extracellular exosome | Actin filament polymerization |
| C0HAP0 | Class I histocompatibility antigen, F10 alpha chain | Antigen proccessing and presentation | membrane | Immune response |
| B5X6E1 | Proteasome activator complex subunit 2 | Protein binding | proteasome complex/cytoplasm/nucleosome | Protein polyubiquitination |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lagos, L.; Tandberg, J.; Kashulin-Bekkelund, A.; Colquhoun, D.J.; Sørum, H.; Winther-Larsen, H.C. Isolation and Characterization of Serum Extracellular Vesicles (EVs) from Atlantic Salmon Infected with Piscirickettsia Salmonis. Proteomes 2017, 5, 34. https://doi.org/10.3390/proteomes5040034
Lagos L, Tandberg J, Kashulin-Bekkelund A, Colquhoun DJ, Sørum H, Winther-Larsen HC. Isolation and Characterization of Serum Extracellular Vesicles (EVs) from Atlantic Salmon Infected with Piscirickettsia Salmonis. Proteomes. 2017; 5(4):34. https://doi.org/10.3390/proteomes5040034
Chicago/Turabian StyleLagos, Leidy, Julia Tandberg, Alexander Kashulin-Bekkelund, Duncan J. Colquhoun, Henning Sørum, and Hanne C. Winther-Larsen. 2017. "Isolation and Characterization of Serum Extracellular Vesicles (EVs) from Atlantic Salmon Infected with Piscirickettsia Salmonis" Proteomes 5, no. 4: 34. https://doi.org/10.3390/proteomes5040034
APA StyleLagos, L., Tandberg, J., Kashulin-Bekkelund, A., Colquhoun, D. J., Sørum, H., & Winther-Larsen, H. C. (2017). Isolation and Characterization of Serum Extracellular Vesicles (EVs) from Atlantic Salmon Infected with Piscirickettsia Salmonis. Proteomes, 5(4), 34. https://doi.org/10.3390/proteomes5040034




