The Use of Primary Care Electronic Health Records for Research: Lipid Medications and Mortality in Elderly Patients
Abstract
1. Introduction
2. Materials and Methods
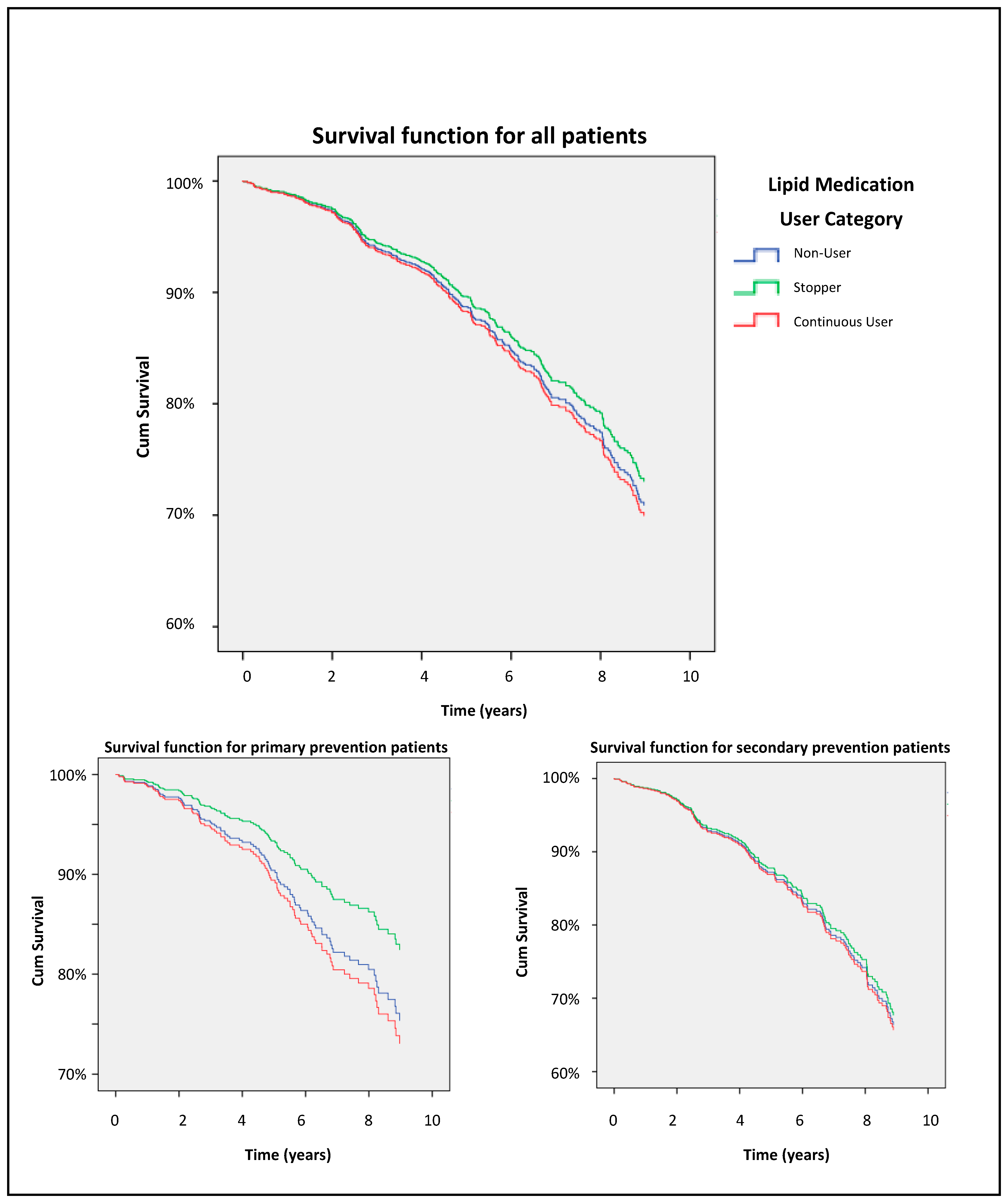
3. Results
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Muller, S. Electronic medical records: The way forward for primary care research? Fam. Pract. 2014, 31, 127–129. [Google Scholar] [CrossRef]
- Stange, K.C.; Miller, W.L.; McWhinney, I. Developing the knowledge base of family practice. Fam. Med. 2001, 33, 286–297. [Google Scholar] [PubMed]
- Hurwitz, J.T.; Brown, M.; Graff, J.S.; Peters, L.; Malone, D.C. Is Real-World Evidence Used in P&T Monographs and Therapeutic Class Reviews? J. Manag. Care Spec. Pharm. 2017, 23, 613–620. [Google Scholar]
- Cowie, M.R.; Blomster, J.I.; Curtis, L.H.; Duclaux, S.; Ford, I.; Fritz, F.; Goldman, S.; Janmohamed, S.; Kreuzer, J.; Leenay, M.; et al. Electronic health records to facilitate clinical research. Clin. Res. Cardiol. 2017, 106, 1–9. [Google Scholar] [CrossRef]
- Tran, B.; Straka, P.; Falster, M.O.; Douglas, K.A.; Britz, T.; Jorm, L.R. Overcoming the data drought: Exploring general practice in Australia by network analysis of big data. Med. J. Aust. 2018, 209, 68–73. [Google Scholar] [CrossRef] [PubMed]
- Casey, J.A.; Schwartz, B.S.; Stewart, W.F.; Adler, N.E. Using Electronic Health Records for Population Health Research: A Review of Methods and Applications. Annu. Rev. Public Health 2016, 37, 61–81. [Google Scholar] [CrossRef] [PubMed]
- Hodgkins, A.J.; Bonney, A.; Mullan, J.; Mayne, D.J.; Barnett, S. Survival analysis using primary care electronic health record data: A systematic review of the literature. Health Inf. Manag. J. 2018, 47, 6–16. [Google Scholar] [CrossRef] [PubMed]
- Barnett, S.; Henderson, J.; Hodgkins, A.; Harrison, C.; Ghosh, A.; Dijkmans-Hadley, B.; Britt, H.; Bonney, A. A valuable approach to the use of electronic medical data in primary care research: Panning for gold. Health Inf. Manag. J. 2017, 46, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Hari, P.; Romanus, D.; Luptakova, K.; Blazer, M.; Yong, C.; Raju, A.; Farrelly, E.; Labotka, R.; Morrison, V.A. The impact of age and comorbidities on practice patterns and outcomes in patients with relapsed/refractory multiple myeloma in the era of novel therapies. J. Geriatr. Oncol. 2018, 9, 138–144. [Google Scholar] [CrossRef]
- PBS Information Management Section: Pharmaceutical Policy Branch Expenditure and Prescriptions Twelve Months to 30 June 2015. Available online: http://www.pbs.gov.au/statistics/2014-2015-files/exp-prs-book-01-2014-15.pdf (accessed on 13 October 2016).
- Mancini, G.B.J.; Baker, S.; Bergeron, J.; Fitchett, D.; Frohlich, J.; Genest, J.; Gupta, M.; Hegele, R.A.; Ng, D.; Pearson, G.J.; et al. Diagnosis, Prevention, and Management of Statin Adverse Effects and Intolerance: Canadian Consensus Working Group Update (2016). Can. J. Cardiol. 2016, 32, S35–S65. [Google Scholar] [CrossRef]
- Absolute risk|The Heart Foundation. Available online: https://heartfoundation.org.au/for-professionals/clinical-information/absolute-risk (accessed on 12 October 2016).
- National Vascular Disease Prevention Alliance Guidelines for the Management of Absolute Cardiovascular Disease Risk. Available online: https://www.heartfoundation.org.au/images/uploads/publications/Absolute-CVD-Risk-Full-Guidelines.pdf (accessed on 12 October 2016).
- Teng, M.; Lin, L.; Zhao, Y.J.; Khoo, A.L.; Davis, B.R.; Yong, Q.W.; Yeo, T.C.; Lim, B.P. Statins for Primary Prevention of Cardiovascular Disease in Elderly Patients: Systematic Review and Meta-Analysis. Drugs Aging 2015, 32, 649–661. [Google Scholar] [CrossRef] [PubMed]
- Zoungas, S.; Curtis, A.; Tonkin, A.; McNeil, J. Statins in the elderly: An answered question? Curr. Opin. Cardiol. 2014, 29, 372–380. [Google Scholar] [CrossRef] [PubMed]
- LaRosa, J.C. Treatment of cholesterol in the elderly: Statins and beyond. Curr. Atheroscler. Rep. 2014, 16, 385. [Google Scholar] [CrossRef]
- Shepherd, J.; Blauw, G.J.; Murphy, M.B.; Bollen, E.L.E.M.; Buckley, B.M.; Cobbe, S.M.; Ford, I.; Gaw, A.; Hyland, M.; Jukema, J.W.; et al. Pravastatin in elderly individuals at risk of vascular disease (PROSPER): A randomised controlled trial. Lancet 2002, 360, 1623–1630. [Google Scholar] [CrossRef]
- Gurwitz, J.H.; Go, A.S.; Fortmann, S.P. Statins for primary prevention in older adults: Uncertainty and the need for more evidence. JAMA 2016, 316, 1971–1972. [Google Scholar] [CrossRef] [PubMed]
- Staff, M.; Roberts, C.; March, L.M. Using aggregated general practice data to evaluate primary care interventions. Med. J. Aust. 2017, 206, 242–243. [Google Scholar] [CrossRef] [PubMed]
- Pyefinch, F. Best Practice Software; Best Practice Software Pty Ltd.: Bundaberg, Australia, 2018. [Google Scholar]
- RACGP Standards for General Practices (4th Edition), Glossary of Terms. Available online: http://www.racgp.org.au/your-practice/standards/standards4thedition/appendices/glossary-of-terms/ (accessed on 13 November 2015).
- Huntley, A.L.; Johnson, R.; Purdy, S.; Valderas, J.M.; Salisbury, C. Measures of Multimorbidity and Morbidity Burden for Use in Primary Care and Community Settings: A Systematic Review and Guide. Ann. Fam. Med. 2012, 10, 134–141. [Google Scholar] [CrossRef]
- Brilleman, S.L.; Salisbury, C. Comparing measures of multimorbidity to predict outcomes in primary care: A cross sectional study. Fam. Pract. 2013, 30, 172–178. [Google Scholar] [CrossRef]
- IBM Corp. IBM SPSS Statistics for Windows, version 23.0; IBM Corp: Armonk, NY, USA, 2015. [Google Scholar]
- Caruana, E.J.; Roman, M.; Hernández-Sánchez, J.; Solli, P. Longitudinal studies. J. Thorac. Dis. 2015, 7, E537–E540. [Google Scholar]
- Rodriguez, F.; Maron, D.J.; Knowles, J.W.; Virani, S.S.; Lin, S.; Heidenreich, P.A. Association between intensity of statin therapy and mortality in patients with atherosclerotic cardiovascular disease. JAMA Cardiol. 2017, 2, 47–54. [Google Scholar] [CrossRef]
- Ramos, R.; Comas-Cufí, M.; Martí-Lluch, R.; Balló, E.; Ponjoan, A.; Alves-Cabratosa, L.; Blanch, J.; Marrugat, J.; Elosua, R.; Grau, M.; et al. Statins for primary prevention of cardiovascular events and mortality in old and very old adults with and without type 2 diabetes: Retrospective cohort study. BMJ 2018, 362, k3359. [Google Scholar] [CrossRef] [PubMed]
- Han, B.H.; Sutin, D.; Williamson, J.D.; Davis, B.R.; Piller, L.B.; Pervin, H.; Pressel, S.L.; Blaum, C.S. Effect of Statin Treatment vs. Usual Care on Primary Cardiovascular Prevention Among Older Adults The ALLHAT-LLT Randomized Clinical Trial. JAMA Intern. Med. 2017, 177, 955. [Google Scholar] [CrossRef] [PubMed]
- Gellert, C.; Schöttker, B.; Brenner, H. Smoking and all-cause mortality in older people: Systematic review and meta-analysis. Arch. Intern. Med. 2012, 172, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Reeve, E.; Thompson, W.; Farrell, B. Deprescribing: A narrative review of the evidence and practical recommendations for recognizing opportunities and taking action. Eur. J. Intern. Med. 2017, 38, 3–11. [Google Scholar] [CrossRef] [PubMed]
| Continuous Variables (Mean ± Standard Deviation) | |||||
|---|---|---|---|---|---|
| Excluded Cases | Complete Cases | Non-User | Stopper | Continuous User | |
| Age at commencement of study (years) | 81.36 (±6.12) | 79.15 (±4.83) | 80.54 (±5.48) | 78.89 (±4.07) | 77.65 (±4.07) |
| Duration of follow-up (years) | 2.90 (±2.66) | 4.62 (±3.06) | 4.45 (±3.03) | 5.63 (±2.83) | 4.36 (±3.09) |
| Prescriptions per year | 19.87 (±24.93) | 19.41 (±16.47) | 18.62 (±17.71) | 18.45 (±16.92) | 20.78 (±14.59) |
| Categorical Variables (number, percentage) | |||||
| Excluded Cases | Complete Cases | Non-User | Stopper | Continuous User | |
| Outcome | |||||
| Death | 66 (15.3%) | 265 (17.9%) | 131 (19.8%) | 51 (20.0%) | 83 (14.7%) |
| Censored | 365 (84.7%) | 1215 (82.1%) | 529 (80.2%) | 204 (80.0%) | 482 (85.3%) |
| Gender | |||||
| Male | 173 (40.2%) | 641 (43.3%) | 292 (44.2%) | 91 (35.7%) | 258 (45.7%) |
| Female | 257 (59.8%) | 839 (56.7%) | 368 (55.8%) | 164 (64.3%) | 307 (54.3%) |
| Smoking status | |||||
| Non-smoker | 40 (37.0%) | 645 (43.6%) | 279 (42.3%) | 119 (46.7%) | 247 (43.7%) |
| Former smoker | 60 (56.5%) | 780 (52.7%) | 355 (53.8%) | 124 (48.9%) | 301 (53.3%) |
| Current smoker | 7 (6.5%) | 55 (3.7%) | 26 (3.9%) | 12 (4.7%) | 17 (3.0%) |
| Marital status | |||||
| Married or de facto | 41 (36.9%) | 763 (51.6%) | 314 (47.6%) | 124 (48.6%) | 325 (57.5%) |
| Single or separated | 28 (25.2%) | 159 (10.7%) | 79 (12.0%) | 20 (7.8%) | 60 (10.6%) |
| Widowed | 42 (37.8%) | 558 (37.7%) | 267 (40.5%) | 111 (43.5%) | 180 (31.9%) |
| Cardiovascular disease | |||||
| No | 348 (80.7%) | 1062 (71.8%) | 573 (86.8%) | 154 (60.4%) | 335 (59.3%) |
| Yes | 83 (19.3%) | 418 (28.2%) | 87 (13.2%) | 101 (39.6%) | 230 (40.7%) |
| Cerebrovascular disease | |||||
| No | 380 (88.2%) | 1226 (82.8%) | 570 (86.4%) | 195 (76.5%) | 461 (81.6%) |
| Yes | 51 (11.8%) | 254 (17.2%) | 90 (13.6%) | 60 (23.5%) | 104 (18.4%) |
| Peripheral vascular disease | |||||
| No | 421 (97.7%) | 1400 (94.6%) | 640 (95.5%) | 235 (92.2%) | 525 (92.9%) |
| Yes | 10 (2.3%) | 80 (5.4%) | 30 (4.5%) | 20 (7.8%) | 40 (7.1%) |
| Diabetes mellitus | |||||
| No | 361 (83.8%) | 1163 (78.6%) | 584 (88.5%) | 173 (67.8%) | 406 (71.9%) |
| Yes | 70 (16.2%) | 317 (21.4%) | 76 (11.5%) | 82 (32.2%) | 159 (28.1%) |
| Lipid medication use | |||||
| Never | 286 (66.4%) | 660 (44.6%) | 660 (100%) | - | - |
| Ceased | 54 (12.5%) | 255 (17.2%) | - | 255 (100%) | - |
| Current | 91 (21.1%) | 565 (38.2%) | - | - | 565 (100%) |
| All Patients | Primary Prevention | Secondary Prevention | ||||
|---|---|---|---|---|---|---|
| Unadjusted HR (95% CI) | Adjusted HR (95% CI) | Unadjusted HR (95% CI) | Adjusted HR (95% CI) | Unadjusted HR (95% CI) | Adjusted HR (95% CI) | |
| Age (per year) | 1.12 (1.09–1.14) | 1.14 (1.12–1.17) | 1.12 (1.08–1.15) | 1.13 (1.08–1.17) | 1.13 (1.09–1.16) | 1.15 (1.11–1.19) |
| Gender | ||||||
| Female | REF | REF | REF | REF | REF | REF |
| Male | 1.30 (1.05–1.61) | 1.61 (1.22–2.11) | 1.13 (0.76–1.70) | 1.46 (0.92–2.32) | 1.38 (1.01–1.87) | 1.63 (1.20–2.40) |
| Smoking status | ||||||
| Non-smoker | REF | REF | REF | REF | REF | REF |
| Former smoker | 0.92 (0.72–1.18) | 0.74 (0.57–0.96) | 0.84 (0.56–1.27) | 0.63 (0.40–1.00) | 0.93 (0.68–1.27) | 0.79 (0.56–1.10) |
| Current smoker | 2.23 (1.34–3.71) | 2.91 (1.71–4.95) | 3.50 (1.76–6.95) | 4.58 (2.16–9.75) | 1.41 (0.65–3.07) | 2.04 (0.91–4.59) |
| Marital status | ||||||
| Married or de facto | REF | REF | REF | REF | REF | REF |
| Single or separated | 0.64 (0.39–1.05) | 0.49 (0.29–0.81) | 0.58 (0.25–1.36) | 0.40 (0.16–0.99) | 0.69 (0.37–1.26) | 0.52 (0.28–0.97) |
| Widowed | 1.08 (0.84–1.38) | 0.77 (0.57–1.02) | 1.15 (0.77–1.72) | 0.65 (0.40–1.06) | 1.06 (0.77–1.46) | 0.85 (0.59–1.22) |
| Prescriptions per year | 1.02 (1.02–1.03) | 1.02 (1.02–1.03) | 1.02 (1.01–1.03) | 1.02 (1.01–1.03) | 1.02 (1.01–1.03) | 1.02 (1.01–1.03) |
| Cardiovascular disease | ||||||
| No | REF | REF | REF | REF | ||
| Yes | 1.33 (1.04–1.70) | 1.20 (0.91–1.58) | – | – | 1.25 (0.91–1.72) | 1.09 (0.76–1.57) |
| Cerebrovascular disease | ||||||
| No | REF | REF | REF | REF | ||
| Yes | 1.24 (0.94–1.65) | 1.16 (0.87–1.55) | – | – | 1.13 (0.82–1.55) | 1.09 (0.76–1.55) |
| Peripheral vascular disease | ||||||
| No | REF | REF | REF | REF | ||
| Yes | 1.25 (0.82–1.90) | 1.15 (0.75–1.78) | – | – | 0.56 (0.74–1.75) | 1.18 (0.75–1.84) |
| Diabetes mellitus | ||||||
| No | REF | REF | REF | REF | ||
| Yes | 0.92 (0.68–1.24) | 0.92 (0.67–1.26) | – | – | 0.78 (0.56–1.08) | 0.89 (0.62–1.28) |
| Lipid medication use | ||||||
| Current | REF | REF | REF | REF | REF | REF |
| Never | 1.32 (1.01–1.74) | 0.97 (0.70–1.33) | 1.57 (0.94–2.64) | 0.91 (0.57–1.57) | 1.44 (1.01–2.05) | 0.97 (0.66–1.45) |
| Ceased | 1.01 (0.71–1.42) | 0.87 (0.61–1.25) | 0.92 (0.43–1.94) | 0.59 (0.26–1.30) | 1.05 (0.71–1.56) | 0.92 (0.62–1.38) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hodgkins, A.J.; Mullan, J.; Mayne, D.J.; Bonney, A. The Use of Primary Care Electronic Health Records for Research: Lipid Medications and Mortality in Elderly Patients. Pharmacy 2019, 7, 134. https://doi.org/10.3390/pharmacy7030134
Hodgkins AJ, Mullan J, Mayne DJ, Bonney A. The Use of Primary Care Electronic Health Records for Research: Lipid Medications and Mortality in Elderly Patients. Pharmacy. 2019; 7(3):134. https://doi.org/10.3390/pharmacy7030134
Chicago/Turabian StyleHodgkins, Adam J., Judy Mullan, Darren J. Mayne, and Andrew Bonney. 2019. "The Use of Primary Care Electronic Health Records for Research: Lipid Medications and Mortality in Elderly Patients" Pharmacy 7, no. 3: 134. https://doi.org/10.3390/pharmacy7030134
APA StyleHodgkins, A. J., Mullan, J., Mayne, D. J., & Bonney, A. (2019). The Use of Primary Care Electronic Health Records for Research: Lipid Medications and Mortality in Elderly Patients. Pharmacy, 7(3), 134. https://doi.org/10.3390/pharmacy7030134





