Forest Contribution to Climate Change Mitigation: Management Oriented to Carbon Capture and Storage
Abstract
1. Introduction
2. The Global Carbon Balance
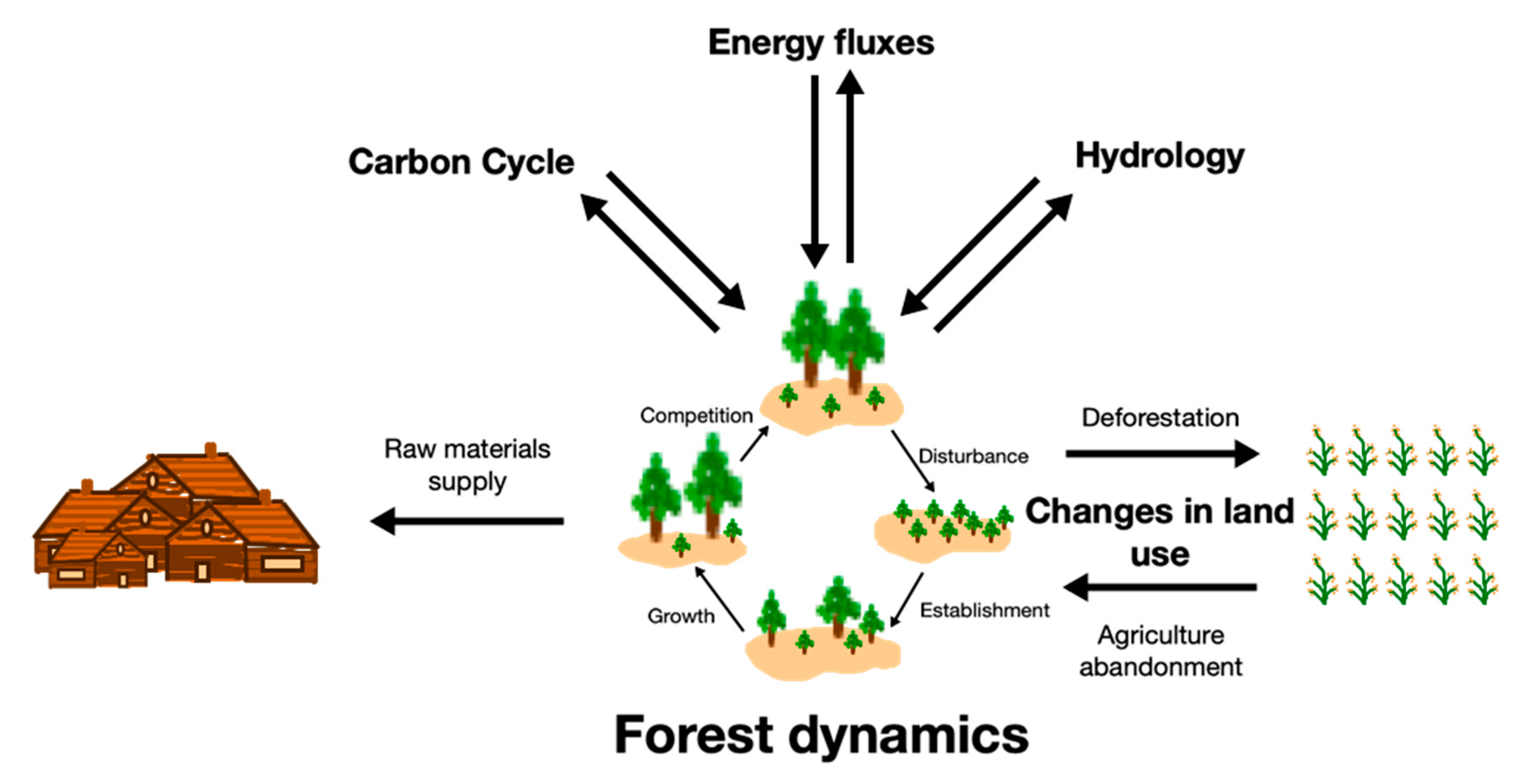
3. Carbon Flows in Terrestrial Ecosystems
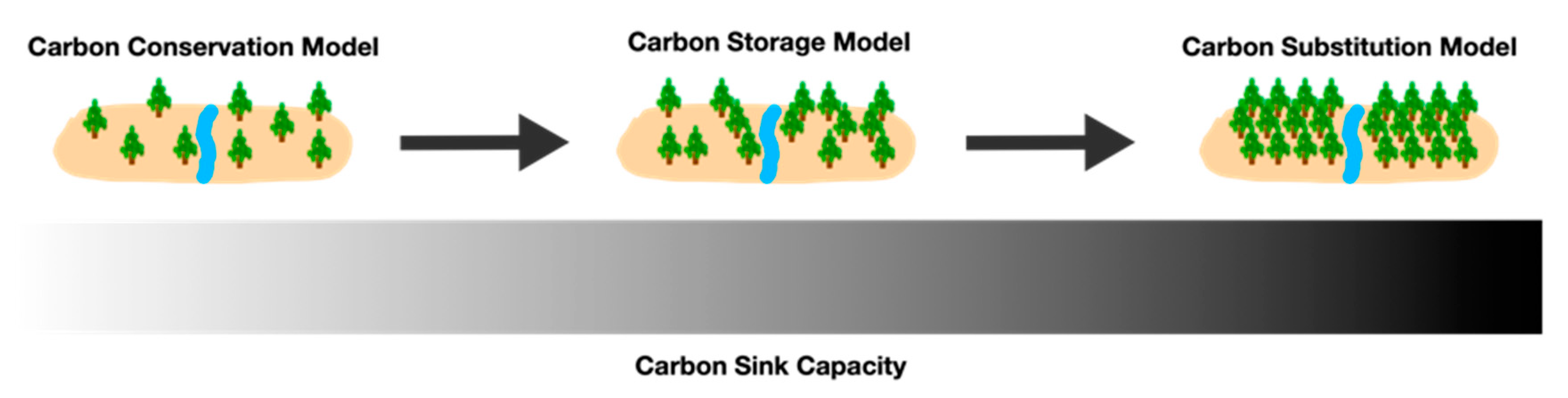
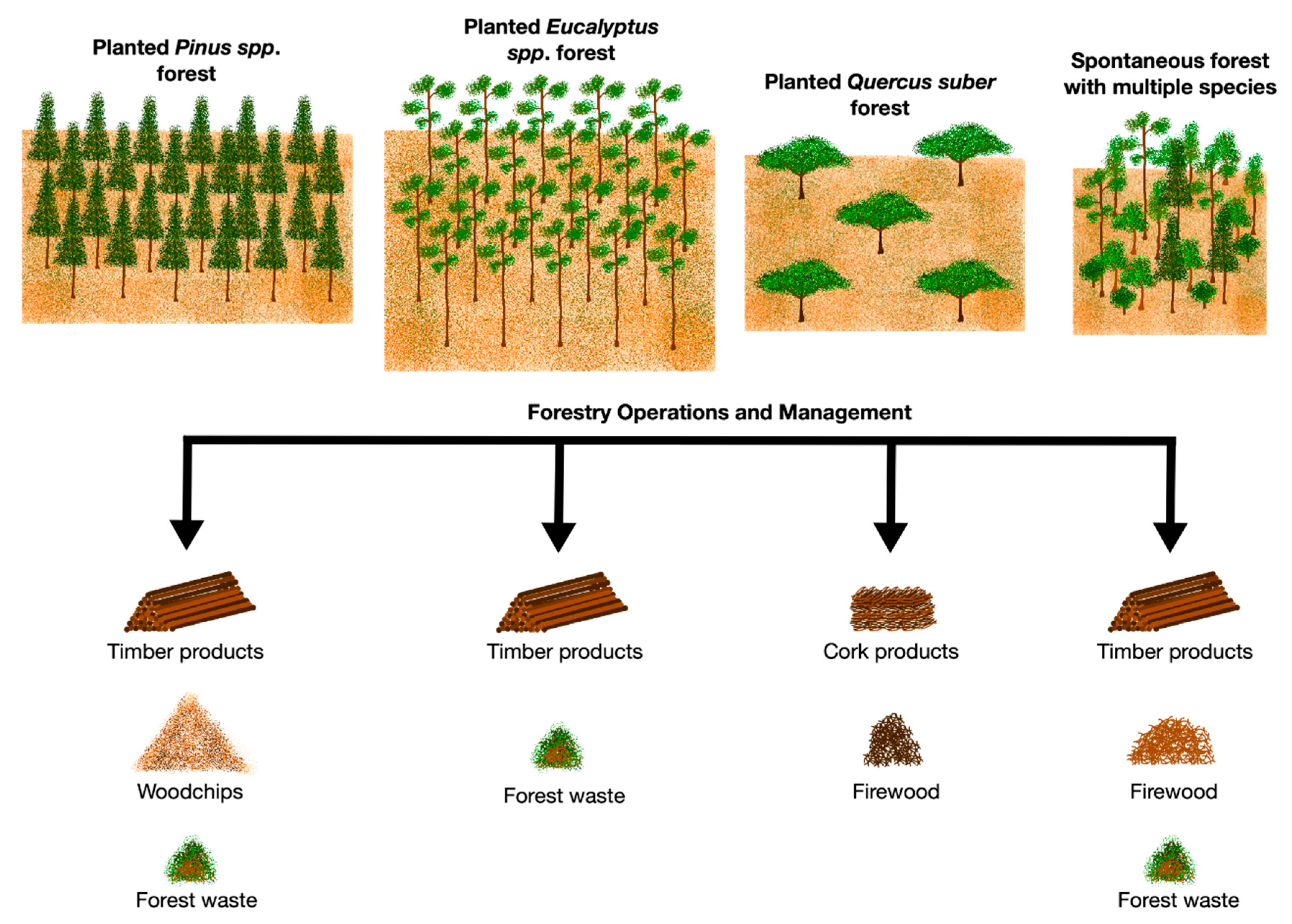
4. Forest Management based on Carbon Capture and Sequestration Capacity
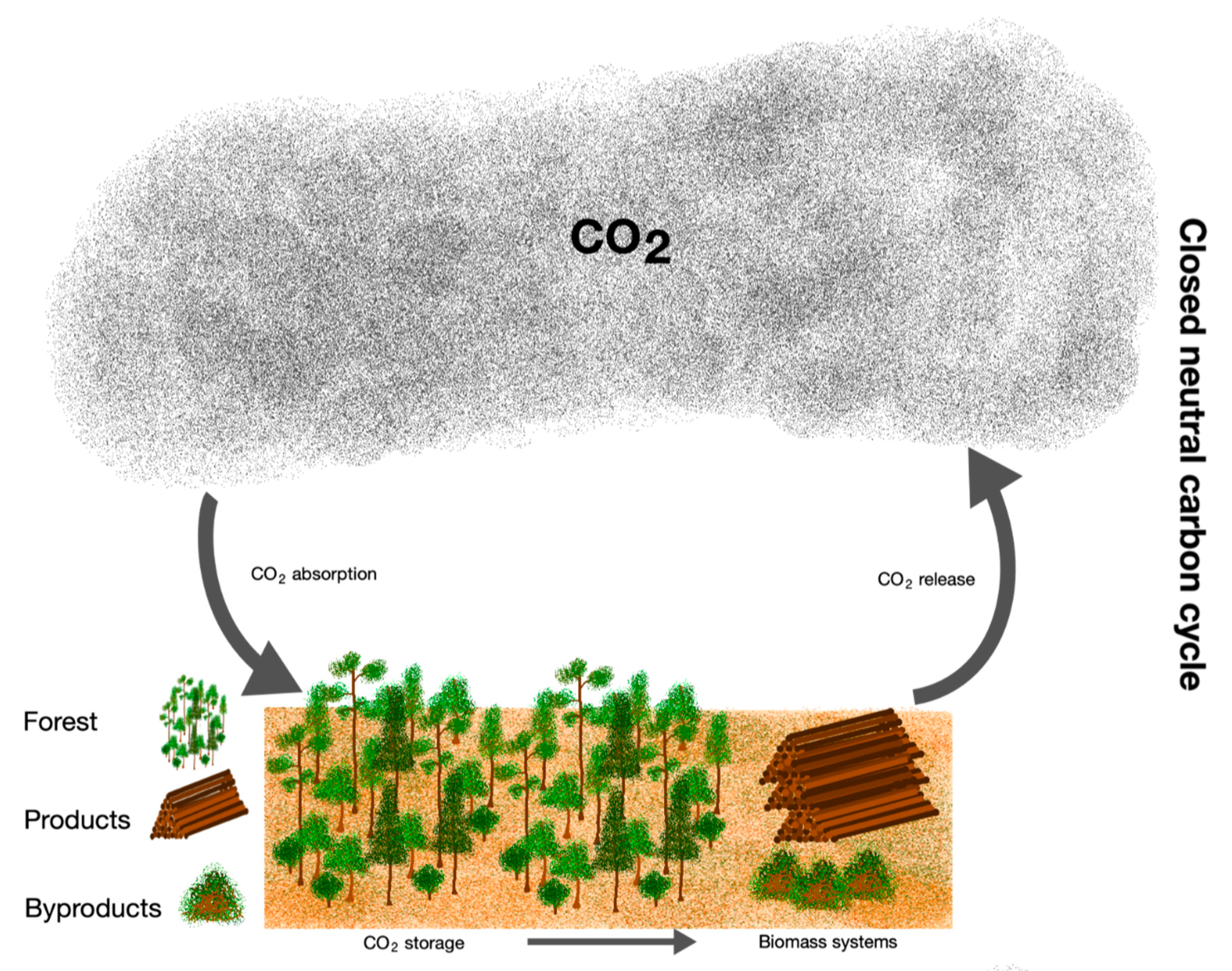
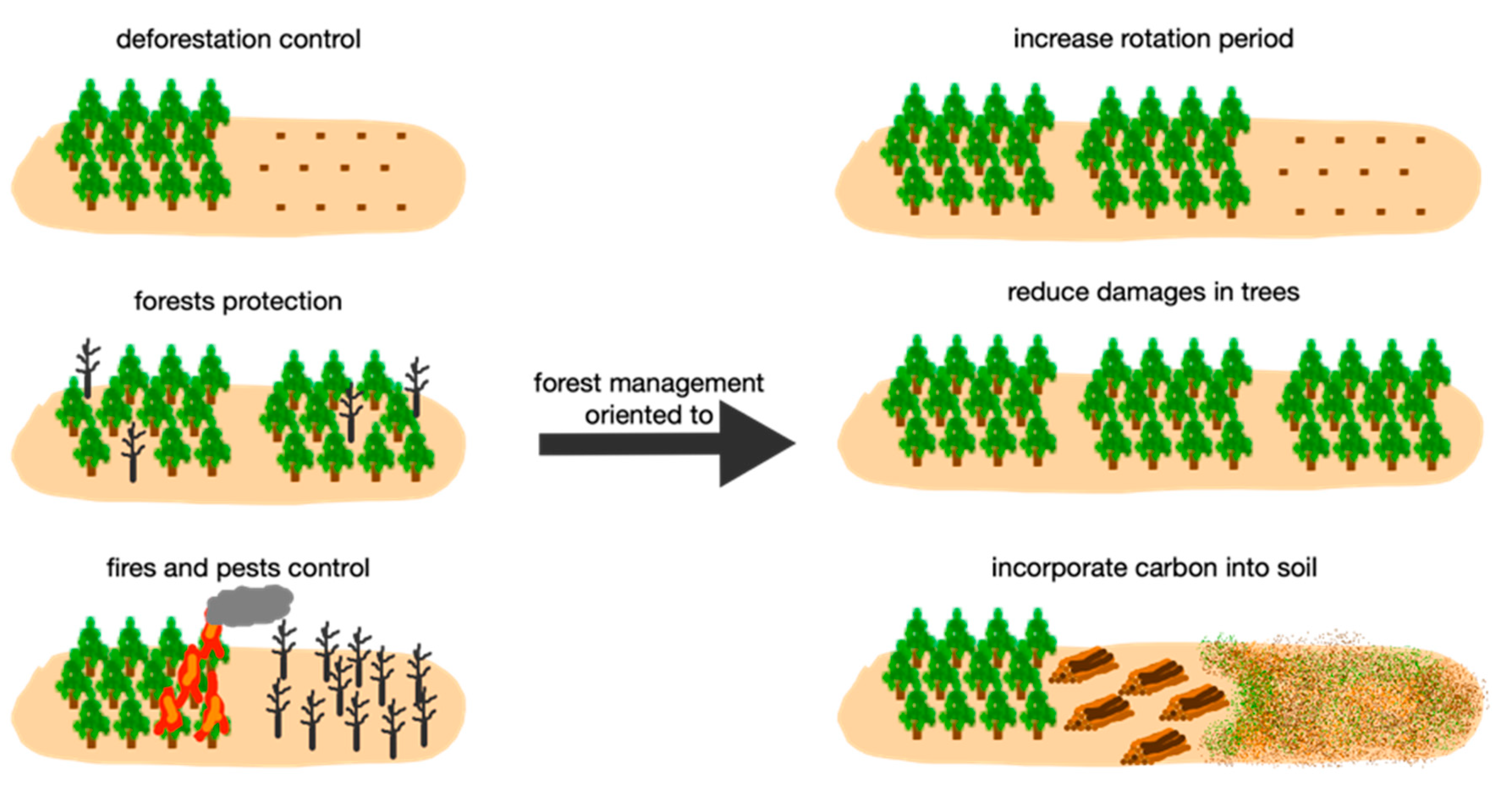
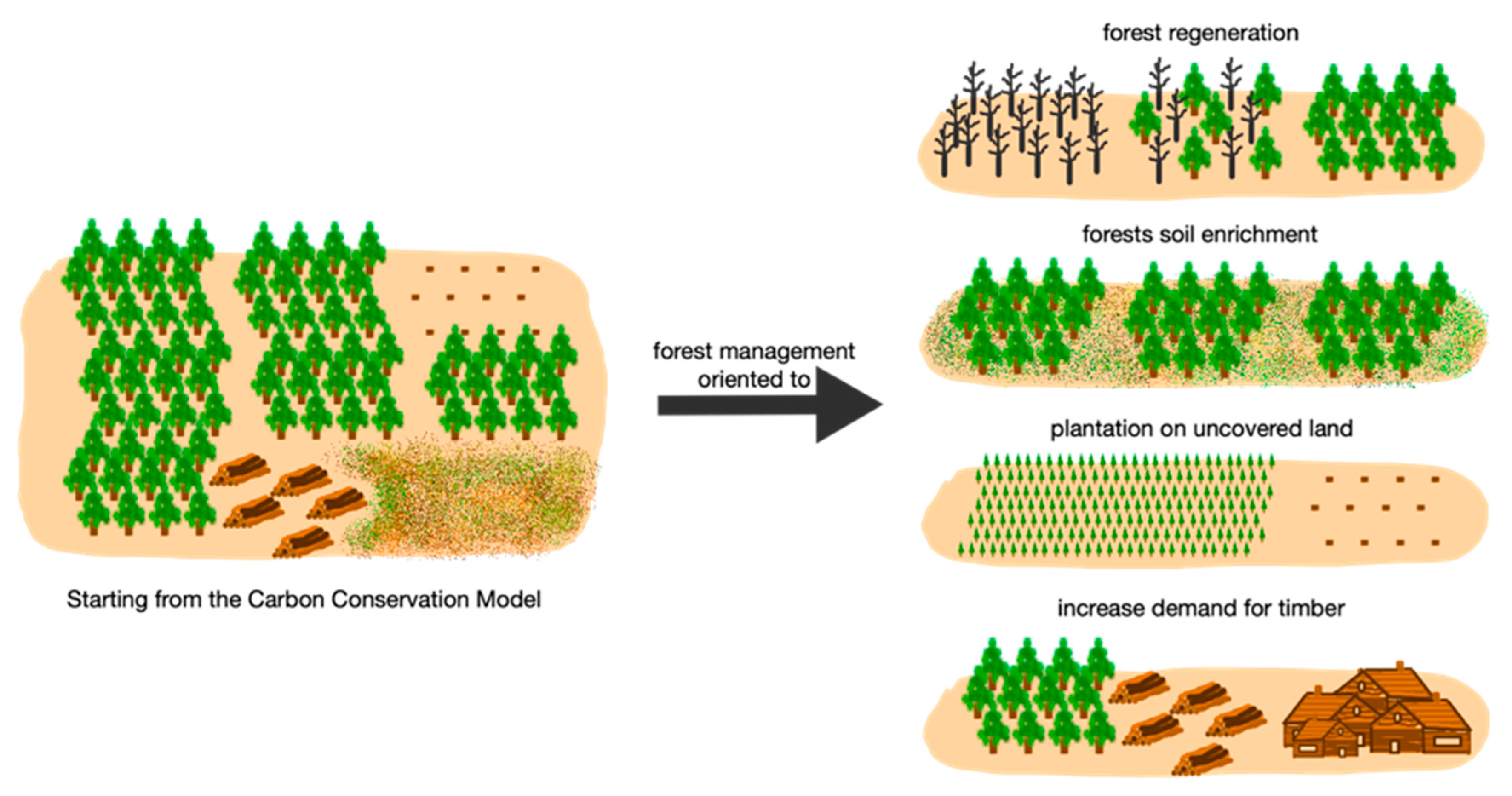
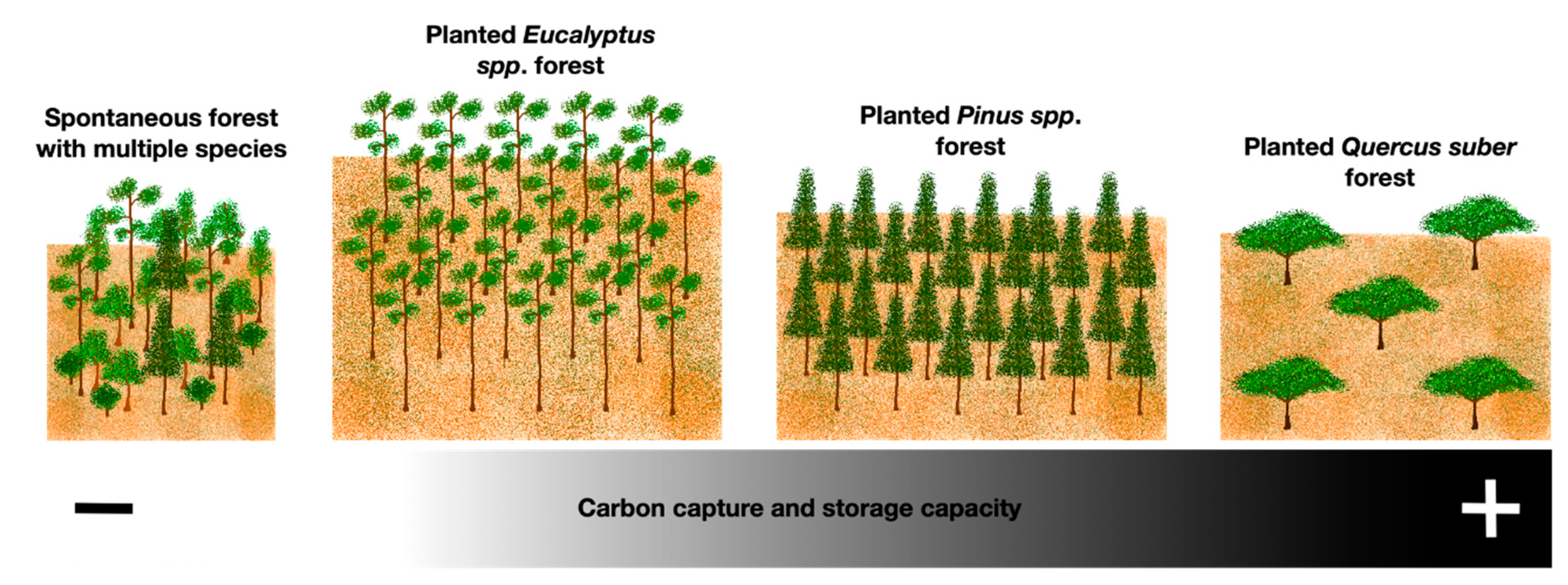
4.1. Carbon Conservation Model
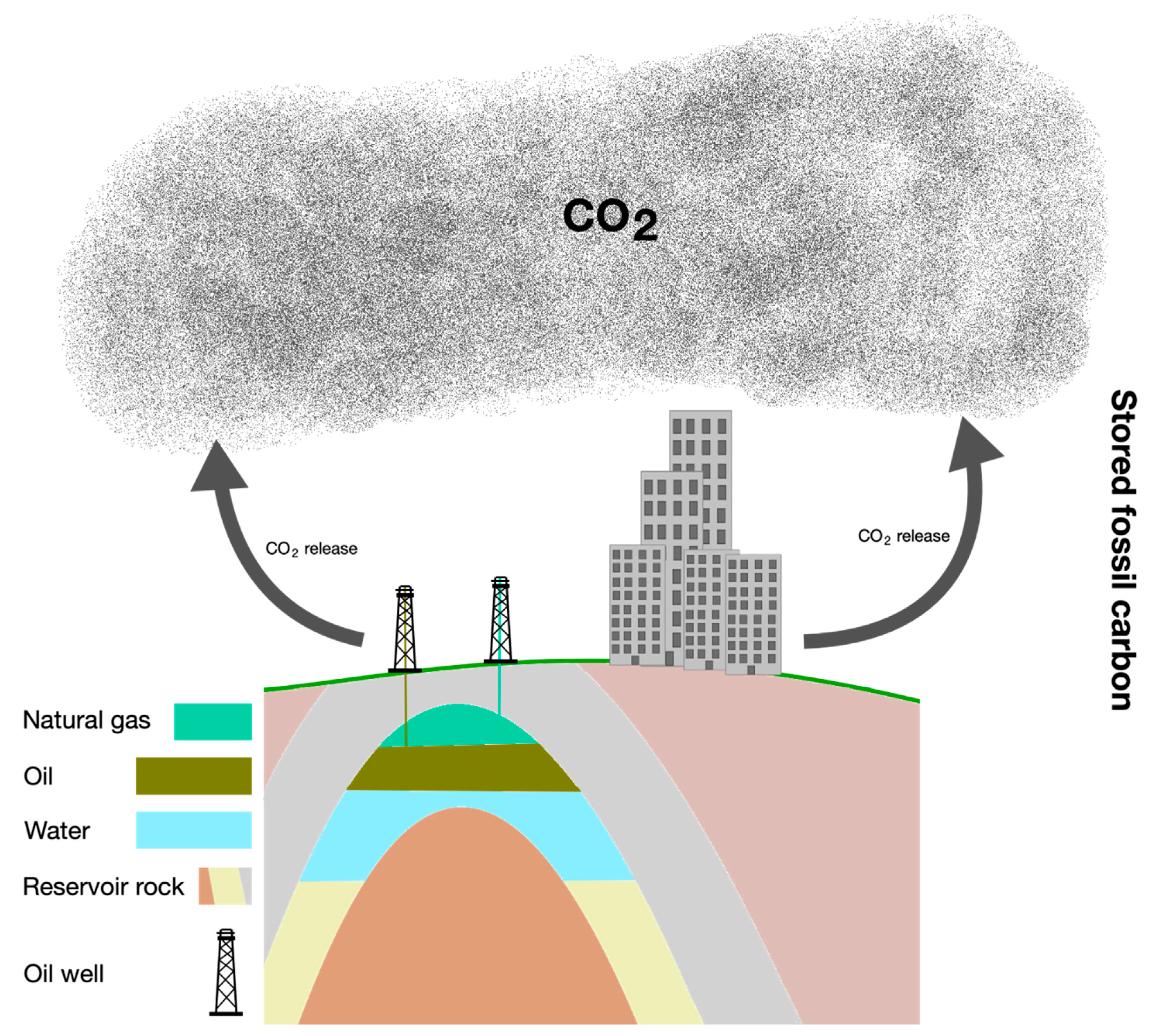
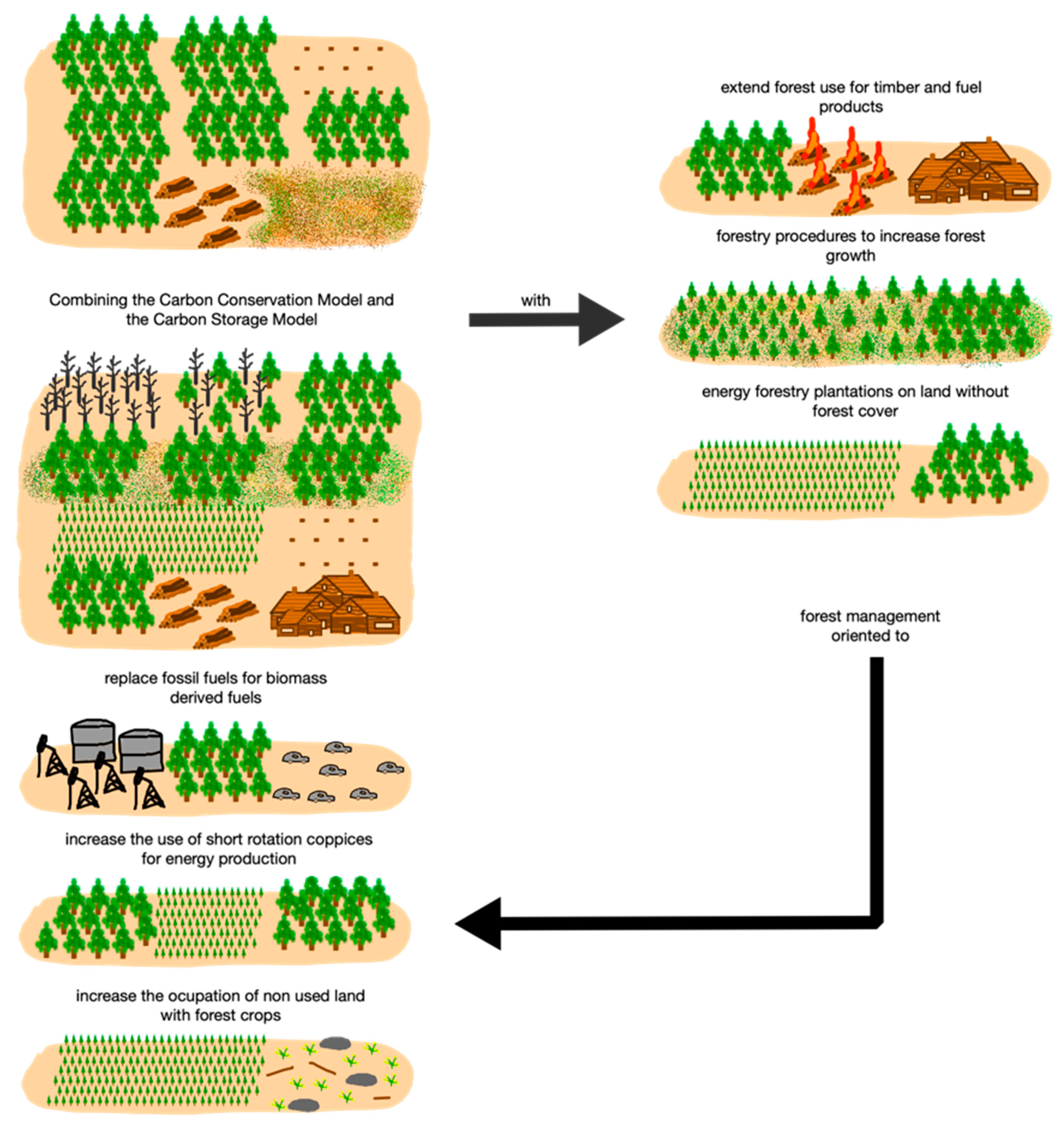
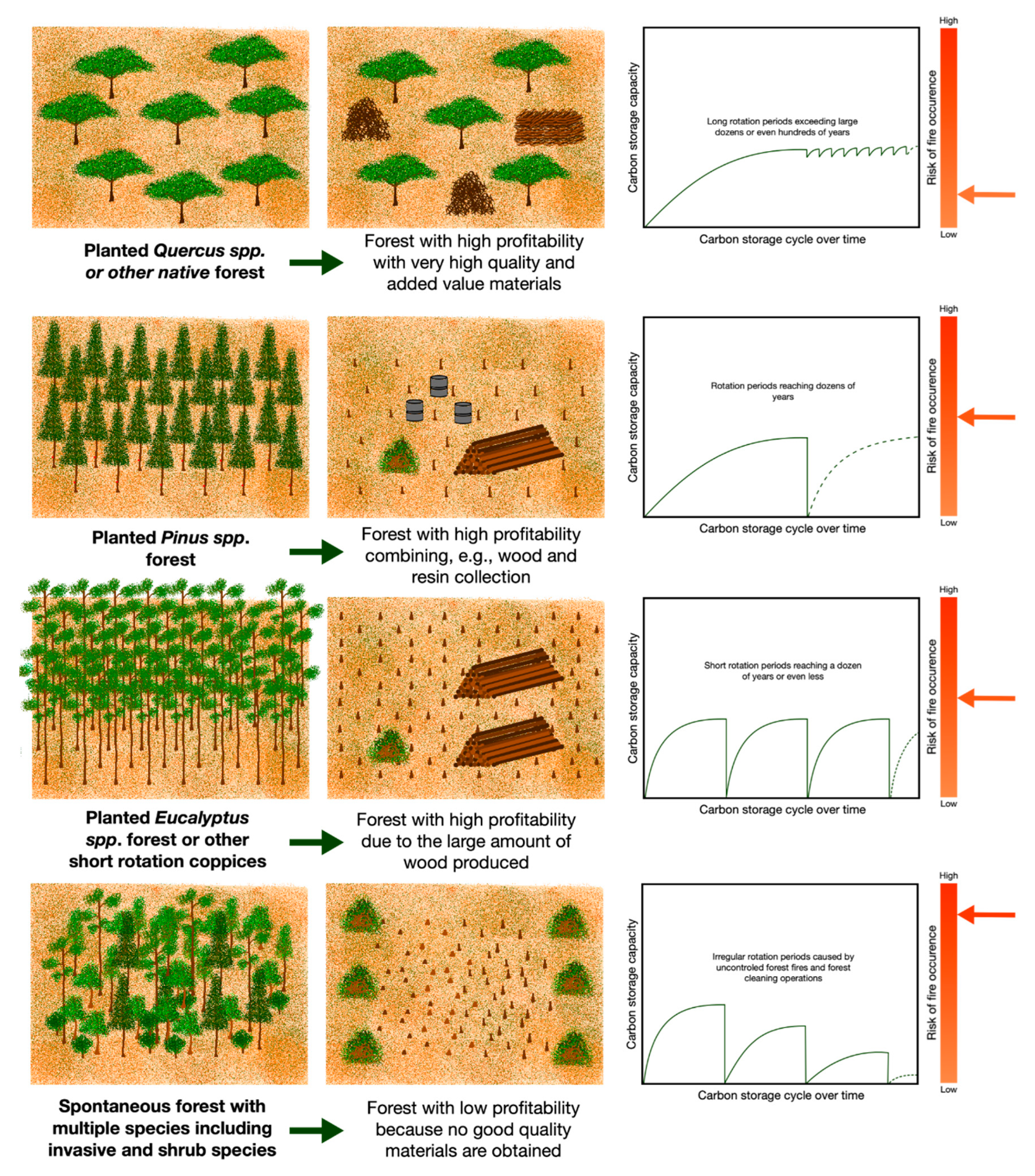
4.2. Carbon Storage Model
4.3. Carbon Substitution Model
5. Discussion and Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Senge, P.M.; Smith, B.; Kruschwitz, N.; Laur, J.; Schley, S. The Necessary Revolution: How Individuals and Organizations Are Working Together to Create a Sustainable World; Crown Business: New York, NY, USA, 2008. [Google Scholar]
- Hart, S.L. Capitalism at the Crossroads: The Unlimited Business Opportunities in Solving the World’s Most Difficult Problems; Pearson Education: New York, NY, USA, 2005. [Google Scholar]
- Keohane, R.O.; Victor, D.G. The Regime Complex for Climate Change. Perspect. Polit. 2011, 9, 7–23. [Google Scholar] [CrossRef]
- Ziman, J.M. Public Knowledge: An Essay Concerning the Social Dimension of Science; CUP Archive: Cambridge, UK, 1968. [Google Scholar]
- Buttel, F.H.; Dickens, P.; Dunlap, R.E.; Gijswijt, A. Sociological theory and the environment: An overview and introduction. In Sociological Theory and the Environment: Classical Foundations, Contemporary Insights; Rowman & Littlefield Publishers: Lanham, MD, USA, 2002; pp. 3–32. [Google Scholar]
- Brulle, R.J.; Dunlap, R.E. Sociology and global climate change. Clim. Chang. Soc. Sociol. Perspect. 2015, 1, 1–31. [Google Scholar]
- Rosenzweig, C.; Iglesias, A.; Yang, X.; Epstein, P.R.; Chivian, E. Climate Change and Extreme Weather Events; Implications for Food Production, Plant Diseases, and Pests. Glob. Chang. Hum. Heal. 2001, 2, 90–104. [Google Scholar] [CrossRef]
- Flannigan, M.D.; Krawchuk, M.A.; De Groot, W.J.; Wotton, B.M.; Gowman, L.M. Implications of changing climate for global wildland fire. Int. J. Wildland Fire 2009, 18, 483–507. [Google Scholar] [CrossRef]
- Hansen, A.J.; Neilson, R.P.; Dale, V.H.; Flather, C.H.; Iverson, L.R.; Currie, D.J.; Shafer, S.; Cook, R.; Bartlein, P.J. Global change in forests: Responses of species, communities, and biomes: Interactions between climate change and land use are projected to cause large shifts in biodiversity. BioScience 2001, 51, 765–779. [Google Scholar] [CrossRef]
- Pautasso, M.; Döring, T.F.; Garbelotto, M.; Pellis, L.; Jeger, M.J. Impacts of climate change on plant diseases—opinions and trends. Eur. J. Plant Pathol. 2012, 133, 295–313. [Google Scholar] [CrossRef]
- Schneider, S.H. The greenhouse effect: Science and policy. Science 1989, 243, 771–781. [Google Scholar] [CrossRef] [PubMed]
- Fleming, J.R. Fixing the Sky: The Checkered History of Weather and Climate Control; Columbia University Press: New York, NY, USA, 2010. [Google Scholar]
- Oreskes, N. The scientific consensus on climate change: How do we know we’re not wrong? In Climate Change: What It Means for Us, Our Children, and Our Grandchildren; MIT Press: Cambridge, UK, 2007; pp. 65–99. [Google Scholar]
- Yamasaki, A. An Overview of CO2 Mitigation Options for Global Warming-Emphasizing CO2 Sequestration Options. J. Chem. Eng. Jpn. 2003, 36, 361–375. [Google Scholar] [CrossRef]
- Finkl, C.W. (Ed.) Carbon cycle. In Encyclopedia of Earth Sciences Series; Springer: Berlin, Germany, 2018. [Google Scholar]
- Kellogg, W.W. Climate Change and Society: Consequences Increasing Atmospheric Carbon Dioxide; Routledge: Abingdon, UK, 2019. [Google Scholar]
- Seidel, S. Can We Delay a Greenhouse Warming?: The Effectiveness and Feasibility of Options to Slow a Build-up of Carbon Dioxide in the Atmosphere; United States Environmental Protection Agency: Washington, DC, USA, 1983.
- Bachu, S. Screening and ranking of sedimentary basins for sequestration of CO2 in geological media in response to climate change. Environ. Earth Sci. 2003, 44, 277–289. [Google Scholar] [CrossRef]
- Gorte, R.W. Carbon Sequestration in Forests; DIANE Publishing: Collingdale, PA, USA, 2009. [Google Scholar]
- Metz, B.; Davidson, O.; De Coninck, H. Carbon Dioxide Capture and Storage: Special Report of the Intergovernmental Panel on Climate Change; Cambridge University Press: Cambridge, UK, 2005. [Google Scholar]
- Perry, D.A.; Oren, R.; Hart, S.C. Forest Ecosystems; JHU Press: Baltimore, MD, USA, 2008. [Google Scholar]
- Plasynski, S.I.; Litynski, J.T.; McIlvried, H.G.; Srivastava, R.D. Progress and New Developments in Carbon Capture and Storage. Crit. Rev. Plant Sci. 2009, 28, 123–138. [Google Scholar] [CrossRef]
- Shahbazi, A.; Nasab, B.R. Carbon Capture and Storage (CCS) and its Impacts on Climate Change and Global Warming. J. Pet. Environ. Biotechnol. 2016, 7, 291. [Google Scholar] [CrossRef]
- Pires, J.C.M.; da Cunha Goncalves, A.L. Bioenergy with Carbon Capture and Storage: Using Natural Resources for Sustainable Development; Academic Press: Cambridge, UK, 2019. [Google Scholar]
- Savill, S.P. The Silviculture of Trees Used in British Forestry; CAB International: Wallingford, UK, 1991. [Google Scholar]
- Bartlett, D.; Nicolescu, V.-N.; Pyttel, P. Evolution and Perspectives of Coppice Forests in European Countries and South Africa. Available online: Gala.gre.ac.uk/14009/5/14009_BARTLETT_FPS-COST_Coppice_Forests_Final_2015.pdf (accessed on 27 November 2019).
- Daniels, T.L. Integrating Forest Carbon Sequestration Into a Cap-and-Trade Program to Reduce Net CO2Emissions. J. Am. Plan. Assoc. 2010, 76, 463–475. [Google Scholar] [CrossRef]
- Burley, J.; Ebeling, J.; Costa, P.; Freer-Smith, P.; Broadmeadow, M.; Lynch, J. 5 Carbon sequestration as a forestry opportunity in a changing climate. In Forestry and Climate Change; CABI: Oxfordshire, UK, 2007; p. 31. [Google Scholar]
- Bastin, J.-F.; Finegold, Y.; Garcia, C.; Mollicone, D.; Rezende, M.; Routh, D.; Zohner, C.M.; Crowther, T.W. The global tree restoration potential. Science 2019, 365, 76–79. [Google Scholar] [CrossRef]
- Cacho, O.J.; Marshall, G.R.; Milne, M. Transaction and abatement costs of carbon-sink projects in developing countries. Environ. Dev. Econ. 2005, 10, 597–614. [Google Scholar] [CrossRef]
- Wunder, S. Payments for Environmental Services: Some Nuts and Bolts; CIFOR Occasional Paper No. 42; Center for International Forestry Research: Bogor, Indonesia, 2005. [Google Scholar]
- Laurance, W.F. A crisis in the making: Responses of Amazonian forests to land use and climate change. Trends Ecol. Evol. 1998, 13, 411–415. [Google Scholar] [CrossRef]
- Kirschbaum, M.U.F. Can Trees Buy Time? An Assessment of the Role of Vegetation Sinks as Part of the Global Carbon Cycle. Clim. Chang. 2003, 58, 47–71. [Google Scholar] [CrossRef]
- Grace, J. Understanding and managing the global carbon cycle. J. Ecol. 2004, 92, 189–202. [Google Scholar] [CrossRef]
- Le, H.D.; Smith, C.; Herbohn, J. What drives the success of reforestation projects in tropical developing countries? The case of the Philippines. Glob. Environ. Chang. 2014, 24, 334–348. [Google Scholar] [CrossRef]
- Ravindranath, N.; Chaturvedi, R.K.; Murthy, I.K. Forest conservation, afforestation and reforestation in India: Implications for forest carbon stocks. Curr. Sci. 2008, 216–222. [Google Scholar]
- De Jesus, R.M. The need for reforestation. In Proceedings of the International Workshop on Large-Scale Reforestation, Corvallis, Oregon, 9–10 May 1990; p. 81. [Google Scholar]
- Jindal, R.; Swallow, B.; Kerr, J. Forestry-based carbon sequestration projects in Africa: Potential benefits and challenges. Nat. Resour. Forum 2008, 32, 116–130. [Google Scholar] [CrossRef]
- Reynolds, T.W. Institutional Determinants of Success among Forestry-Based Carbon Sequestration Projects in Sub-Saharan Africa. World Dev. 2012, 40, 542–554. [Google Scholar] [CrossRef]
- McLymont, R. Toward Clean and Green. Network J. 2010, 17, 20. [Google Scholar]
- Reid, H. Climate change and biodiversity in Europe. Conserv. Soc. 2006, 4, 84. [Google Scholar]
- Curiel-Esparza, J.; Gonzalez-Utrillas, N.; Canto-Perello, J.; Martin-Utrillas, M. Integrating climate change criteria in reforestation projects using a hybrid decision-support system. Environ. Res. Lett. 2015, 10, 94022. [Google Scholar] [CrossRef]
- Ivetić, V.; Devetaković, J. Reforestation challenges in Southeast Europe facing climate change. REFORESTA 2016, 1, 178–220. [Google Scholar] [CrossRef]
- Lopes, J.A.; Santos, M. Facilitating Financing for Sustainable Forest Management in Small Islands Developing States and Low Forest Cover Countries; An analytical report prepared by Indufor for the United Nations Forum on Forests; Country Case Study: Helsinki, Finland, August 2010; 27p. [Google Scholar]
- Nijnik, M.; Bizikova, L. Responding to the Kyoto Protocol through forestry: A comparison of opportunities for several countries in Europe. For. Policy Econ. 2008, 10, 257–269. [Google Scholar] [CrossRef]
- Olschewski, R.; Benítez, P.C. Optimizing joint production of timber and carbon sequestration of afforestation projects. J. For. Econ. 2010, 16, 1–10. [Google Scholar] [CrossRef]
- Major, I.; Haszpra, L.; Rinyu, L.; Futó, I.; Bihari, Á.; Hammer, S.; Jull, A.J.T.; Molnár, M. Temporal Variation of Atmospheric Fossil and Modern CO2 Excess at a Central European Rural Tower Station between 2008 and 2014. Radiocarb. 2018, 60, 1285–1299. [Google Scholar] [CrossRef]
- Möller, D.; Oelßner, W. Environmental CO2 monitoring. In Carbon Dioxide Sensing: Fundamentals, Principles, and Applications; John Wiley & Sons: Hoboken, NJ, USA, 2019; p. 275. [Google Scholar]
- Peñuelas, J.; Sardans, J.; Estiarte, M.; Ogaya, R.; Carnicer, J.; Coll, M.; Barbeta, A.; Rivas-Ubach, A.; Llusià, J.; Garbulsky, M.; et al. Evidence of current impact of climate change on life: A walk from genes to the biosphere. Glob. Chang. Boil. 2013, 19, 2303–2338. [Google Scholar] [CrossRef]
- Van Aalst, M.K. The impacts of climate change on the risk of natural disasters. Disasters 2006, 30, 5–18. [Google Scholar] [CrossRef]
- Pachauri, R.K.; Reisinger, A. IPCC Fourth Assessment Report; IPCC: Geneva, Switzerland, 2007. [Google Scholar]
- Bogner, J.; Pipatti, R.; Hashimoto, S.; Diaz, C.; Mareckova, K.; Diaz, L.; Kjeldsen, P.; Monni, S.; Faaij, A.; Gao, Q.; et al. Mitigation of global greenhouse gas emissions from waste: Conclusions and strategies from the Intergovernmental Panel on Climate Change (IPCC) Fourth Assessment Report. Working Group III (Mitigation). Waste Manag. Res. 2008, 26, 11–32. [Google Scholar] [CrossRef] [PubMed]
- Keeling, C.D.; Bacastow, R.B.; Bainbridge, A.E.; Ekdahl, C.A., Jr.; Guenther, P.R.; Waterman, L.S.; Chin, J.F. Atmospheric carbon dioxide variations at Mauna Loa observatory, Hawaii. Tellus 1976, 28, 538–551. [Google Scholar]
- Stocker, T.F.; Qin, D.; Plattner, G.K.; Tignor, M.; Allen, S.K.; Boschung, J.; Nauels, A.; Xia, Y.; Bex, V.; Midgley, P.M. Climate Change 2013: The Physical Science Basis; IPCC: Geneva, Switzerland, 2013. [Google Scholar]
- Etheridge, D.M.; Steele, L.P.; Langenfelds, R.L.; Francey, R.J.; Barnola, J.-M.; Morgan, V.I. Natural and anthropogenic changes in atmospheric CO2over the last 1000 years from air in Antarctic ice and firn. J. Geophys. Res. Space Phys. 1996, 101, 4115–4128. [Google Scholar] [CrossRef]
- Pearman, G.I.; Etheridge, D.; De Silva, F.; Fraser, P.J. Evidence of changing concentrations of atmospheric CO2, N2O and CH4 from air bubbles in Antarctic ice. Nature 1986, 320, 248–250. [Google Scholar] [CrossRef]
- Barnola, J.M.; Pimienta, P.; Raynaud, D.; Korotkevich, Y.S. CO2-climate relationship as deduced from the Vostok ice core: A re-examination based on new measurements and on a re-evaluation of the air dating. Tellus B 1991, 43, 83–90. [Google Scholar] [CrossRef]
- Seinfeld, J.H.; Pandis, S.N. Atmospheric Chemistry and Physics: From Air Pollution to Climate Change; John Wiley & Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Sun, Y.; Frankenberg, C.; Wood, J.D.; Schimel, D.S.; Jung, M.; Guanter, L.; Drewry, D.T.; Verma, M.; Porcar-Castell, A.; Griffis, T.J.; et al. OCO-2 advances photosynthesis observation from space via solar-induced chlorophyll fluorescence. Science 2017, 358, eaam5747. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Yang, D.; Cai, Z. A retrieval algorithm for TanSat XCO2 observation: Retrieval experiments using GOSAT data. Chin. Sci. Bull. 2013, 58, 1520–1523. [Google Scholar] [CrossRef]
- Butz, A.; Guerlet, S.; Hasekamp, O.; Schepers, D.; Galli, A.; Aben, I.; Frankenberg, C.; Hartmann, J.-M.; Tran, H.; Kuze, A.; et al. Toward accurate CO2and CH4observations from GOSAT. Geophys. Res. Lett. 2011, 38, 38. [Google Scholar] [CrossRef]
- Taylor, T.E.; O’dell, C.W.; Frankenberg, C.; Partain, P.T.; Cronk, H.Q.; Savtchenko, A.; Nelson, R.R.; Rosenthal, E.J.; Chang, A.Y.; Fisher, B.; et al. Orbiting Carbon Observatory-2 (OCO-2) cloud screening algorithms: Validation against collocated MODIS and CALIOP data. Atmospheric Meas. Tech. 2016, 9, 973–989. [Google Scholar] [CrossRef]
- Canadell, J.G.; Ciais, P.; Gurney, K.; Le Quéré, C.; Piao, S.; Raupach, M.R.; Sabine, C.L. An International Effort to Quantify Regional Carbon Fluxes. Eos 2011, 92, 81–82. [Google Scholar] [CrossRef]
- Sitch, S.; Friedlingstein, P.; Gruber, N.; Jones, S.D.; Murray-Tortarolo, G.; Ahlström, A.; Doney, S.C.; Graven, H.; Heinze, C.; Huntingford, C.; et al. Recent trends and drivers of regional sources and sinks of carbon dioxide. Biogeosciences 2015, 12, 653–679. [Google Scholar] [CrossRef]
- Peylin, P.; Law, R.; Gurney, K.; Chevallier, F.; Jacobson, A.; Maki, T.; Niwa, Y.; Patra, P.; Peters, W.; Rayner, P. Global atmospheric carbon budget: Results from an ensemble of atmospheric CO2 inversions. Biogeosciences 2013, 10, 6699–6720. [Google Scholar] [CrossRef]
- Allam, S.; D EL Ouadghiri, M.; EL Ouahbi, R. Carbon Dioxide Information Analysis Center CDIAC: Databases analysis (December 2015). Adv. Inf. Technol. Theory Appl. 2016, 1. Available online: https://cdiac.ess-dive.lbl.gov/ (accessed on 3 December 2019).
- Moberg, R.; Stewart, D.; Stachniak, D. The IEA Weyburn CO2 Monitoring and Storage Project. In Greenhouse Gas Control Technologies—6th International Conference; Elsevier BV: Amsterdam, The Netherlands, 2003; pp. 219–224. [Google Scholar]
- McDonald, R.I.; Fargione, J.; Kiesecker, J.; Miller, W.M.; Powell, J. Energy Sprawl or Energy Efficiency: Climate Policy Impacts on Natural Habitat for the United States of America. PLOS ONE 2009, 4, e6802. [Google Scholar] [CrossRef] [PubMed]
- Houghton, R.A.; House, J.; Pongratz, J.; Van Der Werf, G.; DeFries, R.; Hansen, M.; Quéré, C.L.; Ramankutty, N. Carbon emissions from land use and land-cover change. Biogeosciences 2012, 9, 5125–5142. [Google Scholar] [CrossRef]
- Le Quéré, C.; Moriarty, R.; Andrew, R.M.; Peters, G.P.; Ciais, P.; Friedlingstein, P.; Jones, S.D.; Sitch, S.; Tans, P.; Arneth, A.; et al. Global carbon budget 2014. Earth Syst. Sci. Data 2015, 7, 47–85. [Google Scholar] [CrossRef]
- Bustamante, M.; Robledo-Abad, C.; Harper, R.; Mbow, C.; Ravindranat, N.H.; Sperling, F.; Haberl, H.; Pinto, A.D.S.; Smith, P. Co-benefits, trade-offs, barriers and policies for greenhouse gas mitigation in the agriculture, forestry and other land use (AFOLU) sector. Glob. Chang. Boil. 2014, 20, 3270–3290. [Google Scholar] [CrossRef]
- McKinley, G.A.; Fay, A.R.; Takahashi, T.; Metzl, N. Convergence of atmospheric and North Atlantic carbon dioxide trends on multidecadal timescales. Nat. Geosci. 2011, 4, 606–610. [Google Scholar] [CrossRef]
- Le Quéré, C.; Moriarty, R.; Andrew, R.M.; Canadell, J.G.; Sitch, S.; Korsbakken, J.I.; Friedlingstein, P.; Peters, G.P.; Andres, R.J.; Boden, T.A.; et al. Global Carbon Budget 2015. Earth Syst. Sci. Data 2015, 7, 349–396. [Google Scholar] [CrossRef]
- Le Quéré, C.; Andrew, R.M.; Canadell, J.G.; Sitch, S. Global carbon budget 2016. Earth Syst. Sci. Data 2016, 8, 605–649. [Google Scholar] [CrossRef]
- Fay, A.; McKinley, G. Global trends in surface ocean pCO2 from in situ data. Glob. Biogeochem. Cycles 2013, 27, 541–557. [Google Scholar] [CrossRef]
- Post, W.M.; Peng, T.-H.; Emanuel, W.R.; King, A.W.; Dale, V.H.; DeAngelis, D.L. The global carbon cycle. Am. Sci. 1990, 78, 310–326. [Google Scholar]
- Neff, J.C.; Asner, G.P. Dissolved Organic Carbon in Terrestrial Ecosystems: Synthesis and a Model. Ecosystems 2001, 4, 29–48. [Google Scholar] [CrossRef]
- Baldocchi, D.; Valentini, R.; Running, S.; Oechel, W.; Dahlman, R. Strategies for measuring and modelling carbon dioxide and water vapour fluxes over terrestrial ecosystems. Glob. Chang. Boil. 1996, 2, 159–168. [Google Scholar] [CrossRef]
- Baldocchi, D.; Falge, E.; Gu, L.; Olson, R.; Hollinger, D.; Running, S.; Anthoni, P.; Bernhofer, C.; Davis, K.; Evans, R. FLUXNET: A new tool to study the temporal and spatial variability of ecosystem-scale carbon dioxide, water vapor, and energy flux densities. Bull. Am. Meteorol. Soc. 2001, 82, 2415–2434. [Google Scholar] [CrossRef]
- Baldocchi, D. ‘Breathing’of the terrestrial biosphere: Lessons learned from a global network of carbon dioxide flux measurement systems. Aust. J. Bot. 2008, 56, 1–26. [Google Scholar] [CrossRef]
- U.S. Department of Energy. About the FLUXDATA Network. Available online: https://fluxnet.fluxdata.org/about/ (accessed on 27 November 2019).
- Vitousek, P.M. Global environmental change: An introduction. Annu. Rev. Ecol. Syst. 1992, 23, 1–14. [Google Scholar] [CrossRef]
- Vitousek, P.M. Beyond global warming: Ecology and global change. Ecology 1994, 75, 1861–1876. [Google Scholar] [CrossRef]
- Vitousek, P.M.; D’Antonio, C.M.; Loope, L.L.; Westbrooks, R. Biological invasions as global environmental change. Am. Sci. 1996, 84, 468–478. [Google Scholar]
- Vitousek, P.M.; D’antonio, C.M.; Loope, L.L.; Rejmanek, M.; Westbrooks, R. Introduced species: A significant component of human-caused global change. N. Z. J. Ecol. 1997, 21, 1–16. [Google Scholar]
- Henry, H.A.L.; Juarez, J.D.; Field, C.B.; Vitousek, P.M. Interactive effects of elevated CO2, N deposition and climate change on extracellular enzyme activity and soil density fractionation in a California annual grassland. Glob. Chang. Boil. 2005, 11, 1808–1815. [Google Scholar] [CrossRef]
- Vitousek, S.; Barnard, P.L.; Limber, P.; Erikson, L.; Cole, B. A model integrating longshore and cross-shore processes for predicting long-term shoreline response to climate change. J. Geophys. Res. Earth Surf. 2017, 122, 782–806. [Google Scholar] [CrossRef]
- De Noblet-Ducoudré, N.; Boisier, J.-P.; Pitman, A.; Bonan, G.B.; Brovkin, V.; Cruz, F.; Delire, C.; Gayler, V.; Hurk, B.J.J.M.V.D.; Lawrence, P.J.; et al. Determining Robust Impacts of Land-Use-Induced Land Cover Changes on Surface Climate over North America and Eurasia: Results from the First Set of LUCID Experiments. J. Clim. 2012, 25, 3261–3281. [Google Scholar] [CrossRef]
- Bonan, G.B. Forests and Climate Change: Forcings, Feedbacks, and the Climate Benefits of Forests. Science 2008, 320, 1444–1449. [Google Scholar] [CrossRef] [PubMed]
- Oleson, K.W.; Bonan, G.B.; Feddema, J.; Vertenstein, M.; Grimmond, C.S.B. An Urban Parameterization for a Global Climate Model. Part I: Formulation and Evaluation for Two Cities. J. Appl. Meteorol. Clim. 2008, 47, 1038–1060. [Google Scholar] [CrossRef]
- Jackson, R.B.; Canadell, J.G.; Avissar, R.; Baldocchi, D.D.; Bonan, G.B.; Caldeira, K.; Diffenbaugh, N.S.; Field, C.B.; A Hungate, B.; Jobbágy, E.G.; et al. Protecting climate with forests. Environ. Res. Lett. 2008, 3, 044006. [Google Scholar] [CrossRef]
- Ridgwell, A.; Zeebe, R. The role of the global carbonate cycle in the regulation and evolution of the Earth system. Earth Planet. Sci. Lett. 2005, 234, 299–315. [Google Scholar] [CrossRef]
- Kump, L.R.; Brantley, S.L.; Arthur, M.A. Chemical Weathering, Atmospheric CO2, and Climate. Annu. Rev. Earth Planet. Sci. 2000, 28, 611–667. [Google Scholar] [CrossRef]
- Martin, J.B. Carbonate minerals in the global carbon cycle. Chem. Geol. 2017, 449, 58–72. [Google Scholar] [CrossRef]
- Adams, H.D.; Macalady, A.K.; Breshears, D.D.; Allen, C.D.; Stephenson, N.L.; Saleska, S.R.; Huxman, T.E.; McDowell, N.G. Climate-Induced Tree Mortality: Earth System Consequences. Eos 2010, 91, 153. [Google Scholar] [CrossRef]
- Bonan, G. Ecological Climatology: Concepts and Applications; Cambridge University Press: Cambridge, UK, 2015. [Google Scholar]
- Allen, C.D.; Macalady, A.K.; Chenchouni, H.; Bachelet, M.; McDowell, N.; Vennetier, M.; Kitzberger, T.; Rigling, A.; Breshears, D.D.; Hogg, E.H.; et al. A global overview of drought and heat-induced tree mortality reveals emerging climate change risks for forests. For. Ecol. Manag. 2010, 259, 660–684. [Google Scholar] [CrossRef]
- Graybill, D.A.; Idso, S.B. Detecting the aerial fertilization effect of atmospheric CO2enrichment in tree-ring chronologies. Glob. Biogeochem. Cycles 1993, 7, 81–95. [Google Scholar] [CrossRef]
- Wullschleger, S.D.; Post, W.; King, A. On the potential for a CO2 fertilization effect in forests: Estimates of the biotic growth factor based on 58 controlled-exposure studies. In Biotic Feedbacks in the Global Climatic System; Woodwell, G.M., Mackenzie, F.T., Eds.; Oxford University Press: Oxford, UK, 1995; pp. 85–107. [Google Scholar]
- Cao, M.; Woodward, F.I. Net primary and ecosystem production and carbon stocks of terrestrial ecosystems and their responses to climate change. Glob. Chang. Boil. 1998, 4, 185–198. [Google Scholar] [CrossRef]
- Seneviratne, S.I.; Corti, T.; Davin, E.L.; Hirschi, M.; Jaeger, E.B.; Lehner, I.; Orlowsky, B.; Teuling, A.J. Investigating soil moisture–climate interactions in a changing climate: A review. Earth-Science Rev. 2010, 99, 125–161. [Google Scholar] [CrossRef]
- Van Mantgem, P.J.; Stephenson, N.L.; Byrne, J.C.; Daniels, L.D.; Franklin, J.F.; Fulé, P.Z.; Harmon, M.E.; Larson, A.J.; Smith, J.M.; Taylor, A.H.; et al. Widespread Increase of Tree Mortality Rates in the Western United States. Science 2009, 323, 521–524. [Google Scholar] [CrossRef]
- Broecker, W.S.; Takahashi, T.; Simpson, H.J.; Peng, T.-H. Fate of Fossil Fuel Carbon Dioxide and the Global Carbon Budget. Science 1979, 206, 409–418. [Google Scholar] [CrossRef]
- Revelle, R.; Suess, H.E. Carbon Dioxide Exchange Between Atmosphere and Ocean and the Question of an Increase of Atmospheric CO2 during the Past Decades. Tellus 1957, 9, 18–27. [Google Scholar] [CrossRef]
- Yuan-Hui, L. Geochemical cycles of elements and human perturbation. Geochim. et Cosmochim. Acta 1981, 45, 2073–2084. [Google Scholar] [CrossRef]
- NOAA. Atmospheric Carbon Dioxide Concentrations and Global Annual Average Temperatures over the Years 1880 to 2009. Available online: https://ja.wikipedia.org/wiki/%E3%83%95%E3%82%A1%E3%82%A4%E3%83%AB:Atmospheric_carbon_dioxide_concentrations_and_global_annual_average_temperatures_over_the_years_1880_to_2009.png (accessed on 25 August 2018).
- Trenberth, K.E.; Stepaniak, D.P. The flow of energy through the earth’s climate system. Q. J. R. Meteorol. Soc. 2004, 130, 2677–2701. [Google Scholar] [CrossRef]
- Ramanathan, V.; Barkstrom, B.R.; Harrison, E.F. Climate and the Earth’s Radiation Budget. Phys. Today 1989, 42, 22. [Google Scholar] [CrossRef]
- Pecl, G.T.; Araújo, M.B.; Bell, J.D.; Blanchard, J.; Bonebrake, T.C.; Chen, I.-C.; Clark, T.D.; Colwell, R.K.; Danielsen, F.; Evengård, B.; et al. Biodiversity redistribution under climate change: Impacts on ecosystems and human well-being. Science 2017, 355, eaai9214. [Google Scholar] [CrossRef]
- Perlack, R.D.; Wright, L.L.; Turhollow, A.F.; Graham, R.L.; Stokes, B.J.; Erbach, D.C. Biomass as Feedstock for a Bioenergy and Bioproducts Industry: The Technical Feasibility of a Billion-Ton Annual Supply; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 2005. [Google Scholar]
- Nair, P.K.R.; Kumar, B.M.; Nair, V.D. Agroforestry as a strategy for carbon sequestration. J. Plant Nutr. Soil Sci. 2009, 172, 10–23. [Google Scholar] [CrossRef]
- Fang, J.; Chen, A.; Peng, C.; Zhao, S.; Ci, L. Changes in Forest Biomass Carbon Storage in China Between 1949 and 1998. Science 2001, 292, 2320–2322. [Google Scholar] [CrossRef] [PubMed]
- Nowak, D.J.; Crane, D.E. Carbon storage and sequestration by urban trees in the USA. Environ. Pollut. 2002, 116, 381–389. [Google Scholar] [CrossRef]
- Nelson, E.; Polasky, S.; Lewis, D.J.; Plantinga, A.J.; Lonsdorf, E.; White, D.; Bael, D.; Lawler, J.J. Efficiency of incentives to jointly increase carbon sequestration and species conservation on a landscape. Proc. Natl. Acad. Sci. USA 2008, 105, 9471–9476. [Google Scholar]
- Alvarez, R. A review of nitrogen fertilizer and conservation tillage effects on soil organic carbon storage. Soil Use Manag. 2005, 21, 38–52. [Google Scholar] [CrossRef]
- Pękala, Ł.M.; Tan, R.R.; Foo, D.C.; Jeżowski, J.M. Optimal energy planning models with carbon footprint constraints. Appl. Energy 2010, 87, 1903–1910. [Google Scholar] [CrossRef]
- Luyssaert, S.; Schulze, E.-D.; Börner, A.; Knohl, A.; Hessenmöller, D.; Law, B.E.; Ciais, P.; Grace, J. Old-growth forests as global carbon sinks. Nature 2008, 455, 213–215. [Google Scholar] [CrossRef]
- Lewis, S.L.; Lopez-Gonzalez, G.; Sonké, B.; Affum-Baffoe, K.; Baker, T.R.; Ojo, L.O.; Phillips, O.L.; Reitsma, J.M.; White, L.; Comiskey, J.A.; et al. Increasing carbon storage in intact African tropical forests. Nature 2009, 457, 1003–1006. [Google Scholar] [CrossRef]
- Mackey, B.; Prentice, I.C.; Steffen, W.; House, J.I.; Lindenmayer, D.; Keith, H.; Berry, S. Untangling the confusion around land carbon science and climate change mitigation policy. Nat. Clim. Chang. 2013, 3, 552–557. [Google Scholar] [CrossRef]
- Körner, C. A matter of tree longevity. Science 2017, 355, 130–131. [Google Scholar] [CrossRef]
- Griscom, B.W.; Adams, J.; Ellis, P.W.; Houghton, R.A.; Lomax, G.; Miteva, D.A.; Schlesinger, W.H.; Shoch, D.; Siikamäki, J.V.; Smith, P.; et al. Natural climate solutions. Proc. Natl. Acad. Sci. USA 2017, 114, 11645–11650. [Google Scholar]
- Kirschbaum, M.U. To sink or burn? A discussion of the potential contributions of forests to greenhouse gas balances through storing carbon or providing biofuels. Biomass Bioenergy 2003, 24, 297–310. [Google Scholar] [CrossRef]
- Asner, G.P.; Powell, G.V.N.; Mascaro, J.; Knapp, D.E.; Clark, J.K.; Jacobson, J.; Kennedy-Bowdoin, T.; Balaji, A.; Paez-Acosta, G.; Victoria, E.; et al. High-resolution forest carbon stocks and emissions in the Amazon. Proc. Natl. Acad. Sci. USA 2010, 107, 16738–16742. [Google Scholar]
- Pearson, T.R.H.; Brown, S.; Murray, L.; Sidman, G. Greenhouse gas emissions from tropical forest degradation: An underestimated source. Carbon Balance Manag. 2017, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Putz, F.E.; Zuidema, P.A.; Synnott, T.; Peña-Claros, M.; Pinard, M.A.; Sheil, D.; Vanclay, J.K.; Sist, P.; Gourlet-Fleury, S.; Griscom, B.; et al. Sustaining conservation values in selectively logged tropical forests: The attained and the attainable. Conserv. Lett. 2012, 5, 296–303. [Google Scholar] [CrossRef]
- Dean, C.; Wardell-Johnson, G.W.; Kirkpatrick, J.B. Are there any circumstances in which logging primary wet-eucalypt forest will not add to the global carbon burden? Agric. For. Meteorol. 2012, 161, 156–169. [Google Scholar] [CrossRef]
- Bryan, J.; Shearman, P.; Ash, J.; Kirkpatrick, J.B. Impact of logging on aboveground biomass stocks in lowland rain forest, Papua New Guinea. Ecol. Appl. 2010, 20, 2096–2103. [Google Scholar] [CrossRef] [PubMed]
- Körner, C. ATMOSPHERIC SCIENCE: Slow in, Rapid out--Carbon Flux Studies and Kyoto Targets. Science 2003, 300, 1242–1243. [Google Scholar] [CrossRef] [PubMed]
- Moomaw, W.R.; Masino, S.A.; Faison, E.K. Intact Forests in the United States: Proforestation Mitigates Climate Change and Serves the Greatest Good. Front. For. Glob. Chang. 2019, 2, 27. [Google Scholar] [CrossRef]
- Dixon, R.K.; Solomon, A.M.; Brown, S.; Houghton, R.A.; Trexier, M.C.; Wiśniewski, J. Carbon Pools and Flux of Global Forest Ecosystems. Science 1994, 263, 185–190. [Google Scholar] [CrossRef]
- Carle, J.; Holmgren, P. Wood from planted forests. For. Prod. J. 2008, 58, 6. [Google Scholar]
- Zhang, H.; Guan, D.; Song, M. Biomass and carbon storage of Eucalyptus and Acacia plantations in the Pearl River Delta, South China. For. Ecol. Manag. 2012, 277, 90–97. [Google Scholar] [CrossRef]
- Giardina, C.P.; Ryan, M.G. Total Belowground Carbon Allocation in a Fast-growing Eucalyptus Plantation Estimated Using a Carbon Balance Approach. Ecosystems 2002, 5, 487–499. [Google Scholar] [CrossRef]
- Keith, H.; MacKey, B.G.; Lindenmayer, D.B. Re-evaluation of forest biomass carbon stocks and lessons from the world’s most carbon-dense forests. Proc. Natl. Acad. Sci. USA 2009, 106, 11635–11640. [Google Scholar] [CrossRef] [PubMed]
- Liski, J.; Pussinen, A.; Pingoud, K.; Mäkipää, R.; Karjalainen, T. Which rotation length is favourable to carbon sequestration? Can. J. For. Res. 2001, 31, 2004–2013. [Google Scholar] [CrossRef]
- Brown, S. Bosques y Cambio Climático y la Función de Los Bosques Como Sumideros de Carbono. 1997. Available online: https://www.typsa.com/files/pdf/Bosques.pdf (accessed on 3 December 2019).
- Cox, P.M.; Betts, R.A.; Jones, C.D.; Spall, S.A.; Totterdell, I.J. Acceleration of global warming due to carbon-cycle feedbacks in a coupled climate model. Nature 2000, 408, 184–187. [Google Scholar] [CrossRef]
- Cooper, C.F. Carbon storage in managed forests. Can. J. For. Res. 1983, 13, 155–166. [Google Scholar] [CrossRef]
- Harmon, M.E.; Ferrell, W.K.; Franklin, J.F. Effects on carbon storage of conversion of old-growth forests to young forests. Science 1990, 247, 699–702. [Google Scholar] [CrossRef]
- Sohngen, B.; Mendelsohn, R. An Optimal Control Model of Forest Carbon Sequestration. Am. J. Agric. Econ. 2003, 85, 448–457. [Google Scholar] [CrossRef]
- Palma, J.H.N.; Paulo, J.A.; Tomé, M. Carbon sequestration of modern Quercus suber L. silvoarable agroforestry systems in Portugal: A YieldSAFE-based estimation. Agrofor. Syst. 2014, 88, 791–801. [Google Scholar] [CrossRef]
- Ogaya, R.; Peñuelas, J. Tree growth, mortality, and above-ground biomass accumulation in a holm oak forest under a five-year experimental field drought. Plant Ecol. 2007, 189, 291–299. [Google Scholar] [CrossRef]
- Marland, G.; Schlamadinger, B. Forests for carbon sequestration or fossil fuel substitution? A sensitivity analysis. Biomass Bioenergy 1997, 13, 389–397. [Google Scholar] [CrossRef]
- Perez-Garcia, J.; Lippke, B.; Comnick, J.; Manriquez, C. An assessment of carbon pools, storage, and wood products market substitution using life-cycle analysis results. Wood Fiber Sci. 2007, 37, 140–148. [Google Scholar]
- Chulze, E.-D.; Körner, C.; Law, B.E.; Haberl, H.; Luyssaert, S. Large-scale bioenergy from additional harvest of forest biomass is neither sustainable nor greenhouse gas neutral. GCB Bioenergy 2012, 4, 611–616. [Google Scholar] [CrossRef]
- Gustavsson, L.; Haus, S.; Lundblad, M.; Lundström, A.; Ortiz, C.A.; Sathre, R.; Le Truong, N.; Wikberg, P.-E. Climate change effects of forestry and substitution of carbon-intensive materials and fossil fuels. Renew. Sustain. Energy Rev. 2017, 67, 612–624. [Google Scholar] [CrossRef]
- Baral, A. Trees for carbon sequestration or fossil fuel substitution: The issue of cost vs. carbon benefit. Biomass Bioenergy 2004, 27, 41–55. [Google Scholar] [CrossRef]
- Bird, D.N. Estimating the Displacement of Energy and Materials by Woody Biomass in Austria; Joanneum Research Resources: Graz, Austria, 2013. [Google Scholar]
- Ingerson, A. Carbon storage potential of harvested wood: Summary and policy implications. Mitigation Adapt. Strateg. Glob. Chang. 2011, 16, 307–323. [Google Scholar] [CrossRef]
- E Harmon, M. Have product substitution carbon benefits been overestimated? A sensitivity analysis of key assumptions. Environ. Res. Lett. 2019, 14, 065008. [Google Scholar] [CrossRef]
- Marras, T.; Petroselli, A.; Vessella, F.; Damiani, G.; Schirone, B. Noble biomass: Restore, recycle, profit using cork oak (Quercus suber L.). Appl. Math. Sci. 2014, 8, 6495–6513. [Google Scholar] [CrossRef]
- Aronson, J.; Santos-Pereira, J.; Pausas, J.G. (Eds.) Cork Oak Woodlands on the Edge: Ecology, Adaptive Management, and Restoration; Island Press: Washington, DC, USA, 2009; pp. 1–10. [Google Scholar]
- A Coomes, D.; Allen, R.B.; A Scott, N.; Goulding, C.; Beets, P. Designing systems to monitor carbon stocks in forests and shrublands. For. Ecol. Manag. 2002, 164, 89–108. [Google Scholar] [CrossRef]
- Zylstra, P.J. Flammability dynamics in the Australian Alps. Austral Ecol. 2018, 43, 578–591. [Google Scholar] [CrossRef]
- Taylor, C.; McCarthy, M.A.; Lindenmayer, D.B. Nonlinear Effects of Stand Age on Fire Severity. Conserv. Lett. 2014, 7, 355–370. [Google Scholar] [CrossRef]
- Lohmann, L. Carbon trading. Dev. dialogue 2006, 48, 1–359. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nunes, L.J.R.; Meireles, C.I.R.; Pinto Gomes, C.J.; Almeida Ribeiro, N.M.C. Forest Contribution to Climate Change Mitigation: Management Oriented to Carbon Capture and Storage. Climate 2020, 8, 21. https://doi.org/10.3390/cli8020021
Nunes LJR, Meireles CIR, Pinto Gomes CJ, Almeida Ribeiro NMC. Forest Contribution to Climate Change Mitigation: Management Oriented to Carbon Capture and Storage. Climate. 2020; 8(2):21. https://doi.org/10.3390/cli8020021
Chicago/Turabian StyleNunes, Leonel J.R., Catarina I.R. Meireles, Carlos J. Pinto Gomes, and Nuno M.C. Almeida Ribeiro. 2020. "Forest Contribution to Climate Change Mitigation: Management Oriented to Carbon Capture and Storage" Climate 8, no. 2: 21. https://doi.org/10.3390/cli8020021
APA StyleNunes, L. J. R., Meireles, C. I. R., Pinto Gomes, C. J., & Almeida Ribeiro, N. M. C. (2020). Forest Contribution to Climate Change Mitigation: Management Oriented to Carbon Capture and Storage. Climate, 8(2), 21. https://doi.org/10.3390/cli8020021







