IoT-Enabled Gas Sensors: Technologies, Applications, and Opportunities
Abstract
1. Introduction
- An analysis of the gas-sensing technologies evolution;
- A deep literature review on the most promising technologies to sense environmental gases through wireless sensors;
- The analysis of the most promising wireless-based solutions for ambient gas monitoring;
- The identification of open research issues on gas-sensing technologies and wireless gas sensors;
- The lessons learned from this study on gas sensors are shared.
2. Background on Environmental Gases
3. Internet of Things (IoT) Verticals Opportunities and Economic Impact
4. Evolution of Gas-Sensing Technologies
5. Sensing Technologies
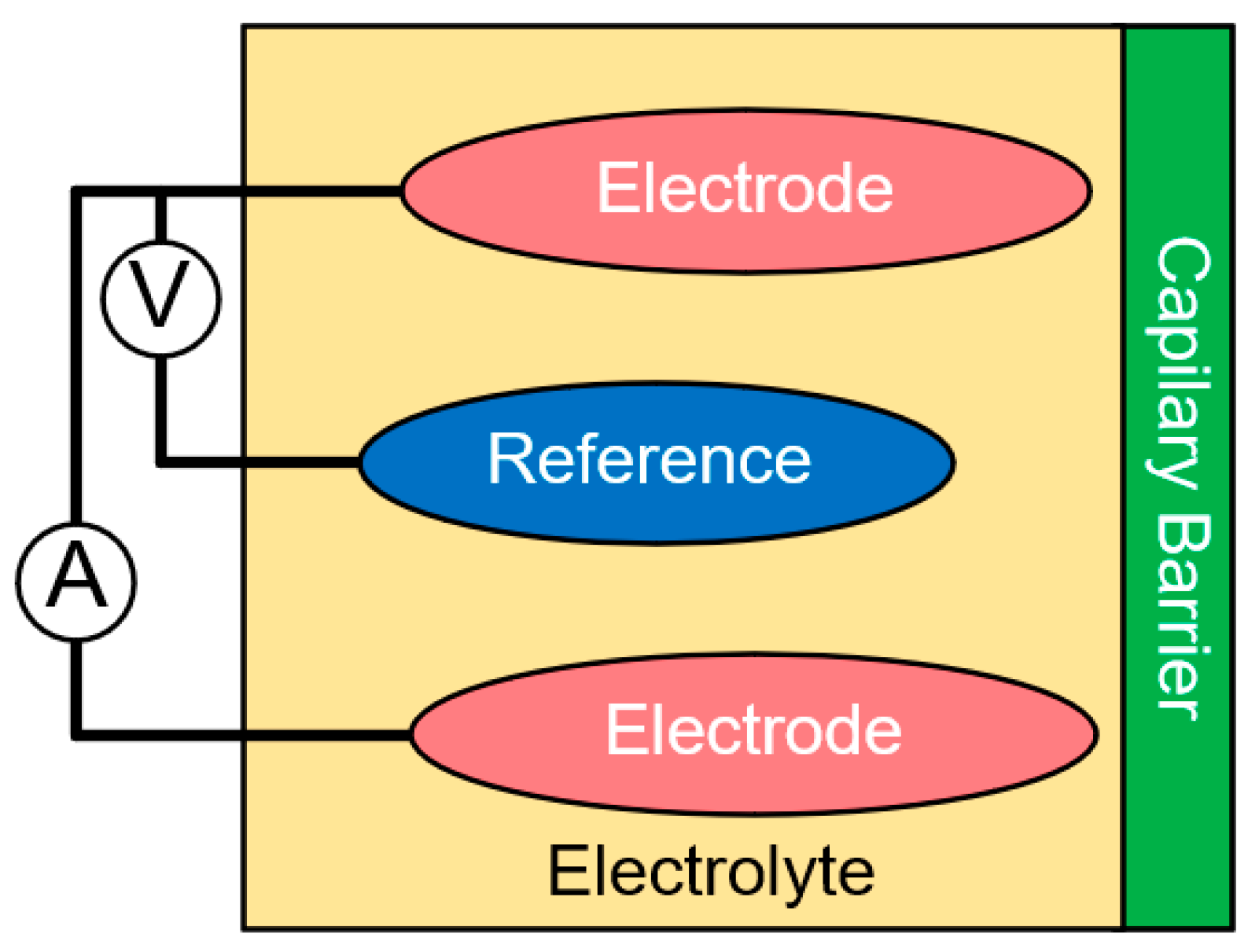
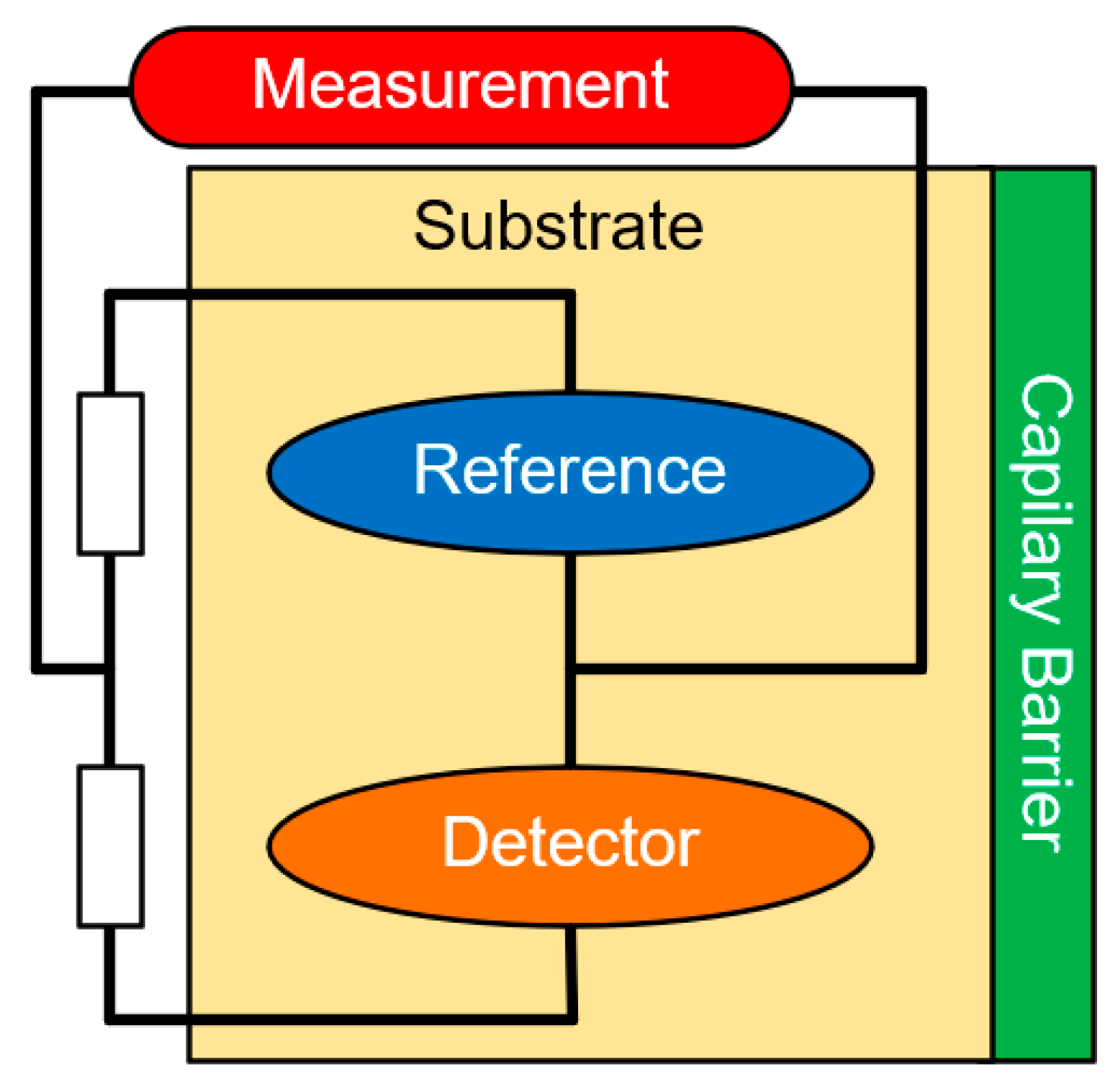
5.1. Electrochemical
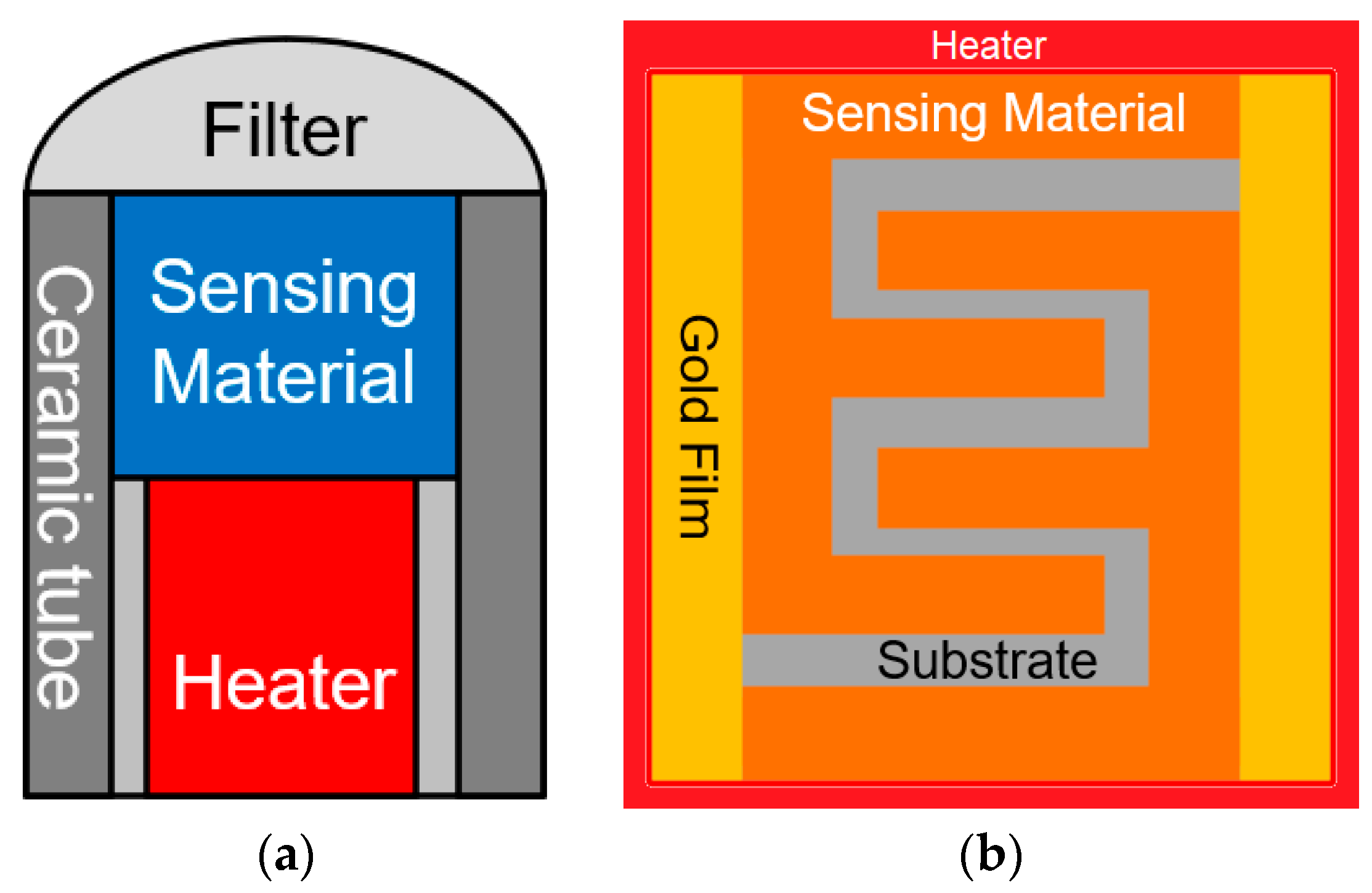
5.2. Metal Oxide Semiconductors
5.3. Catalytic
5.4. Polymers
5.5. Carbon Nanotubes (CNTs)
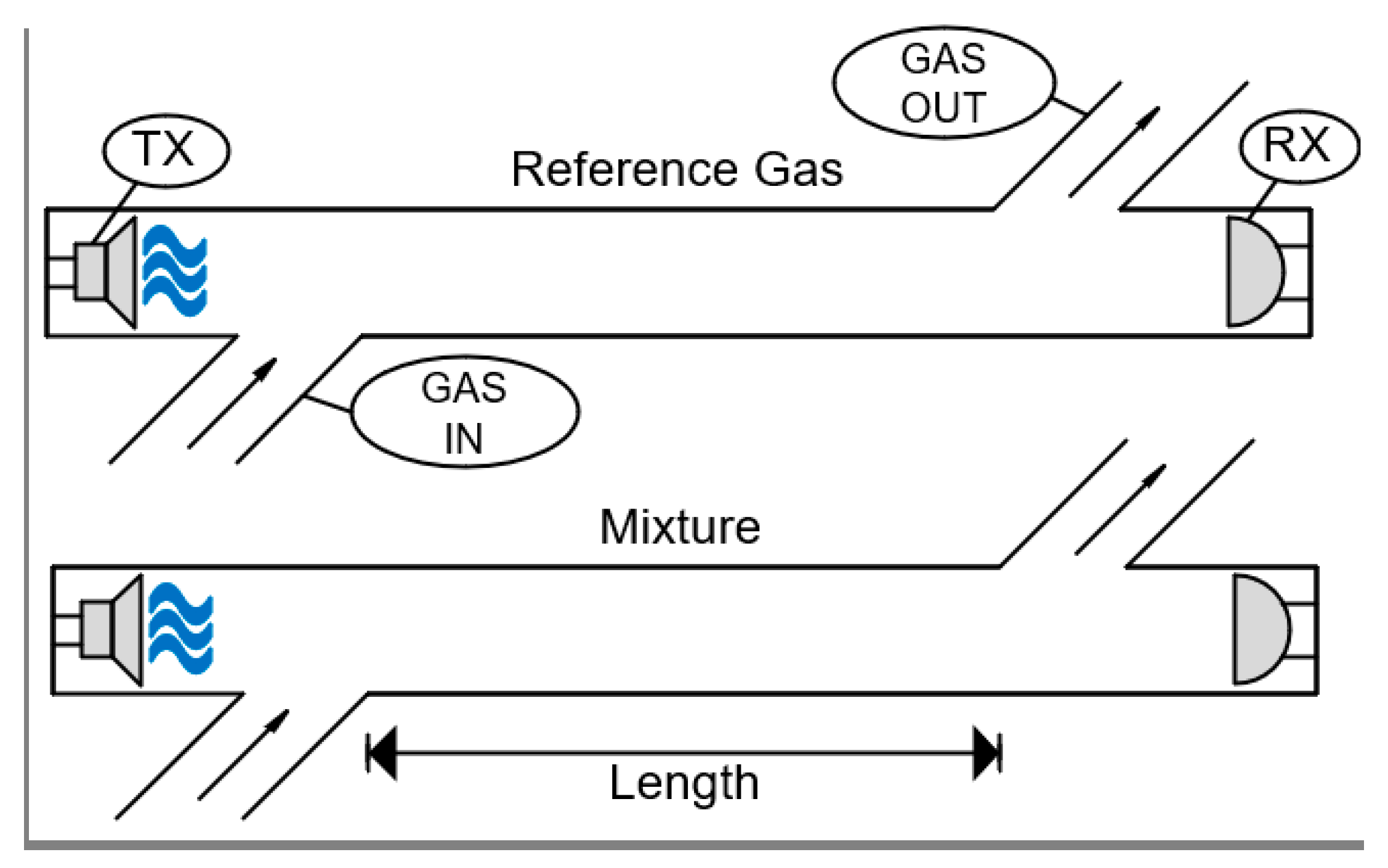
5.6. Acoustic
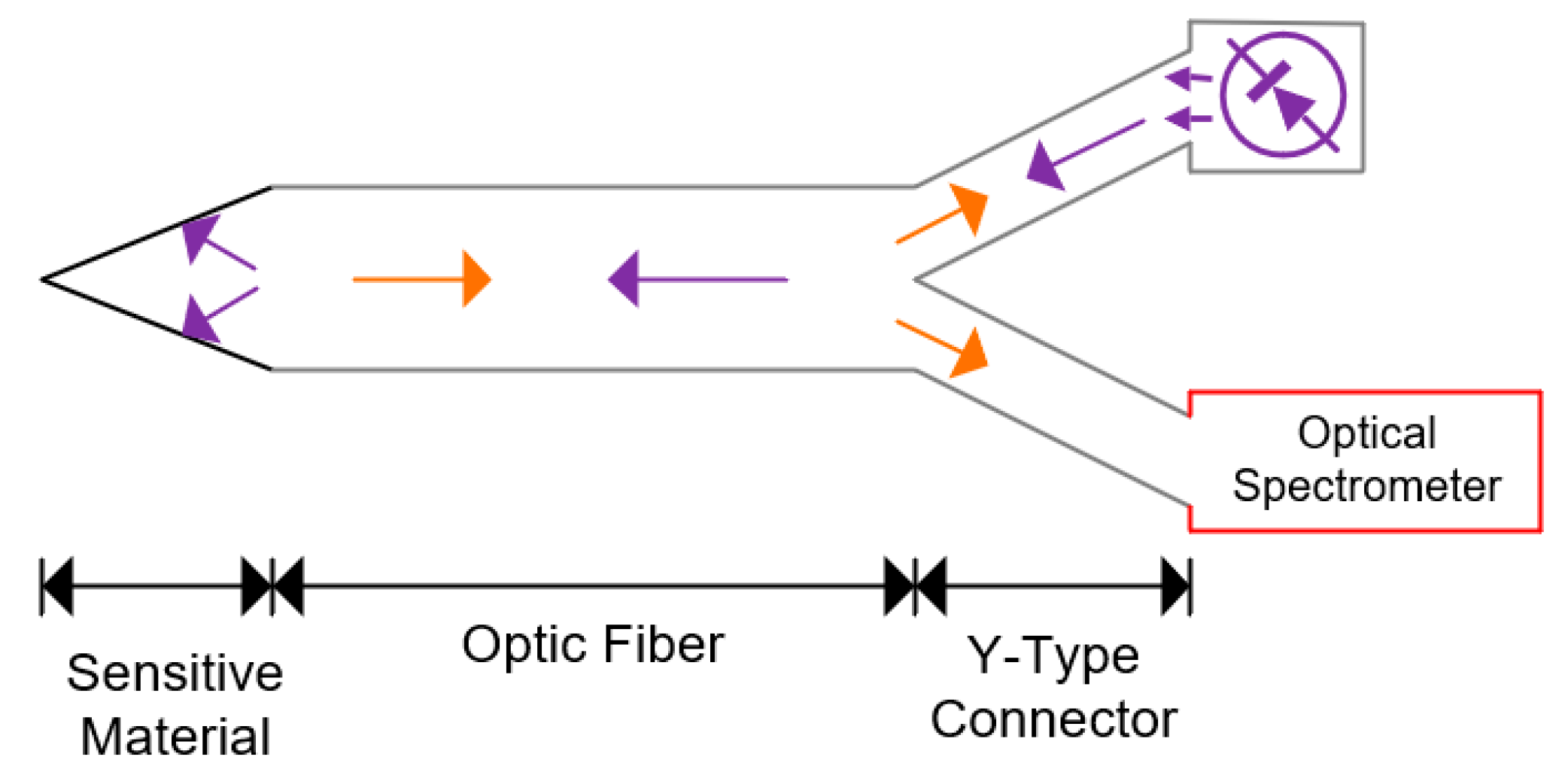
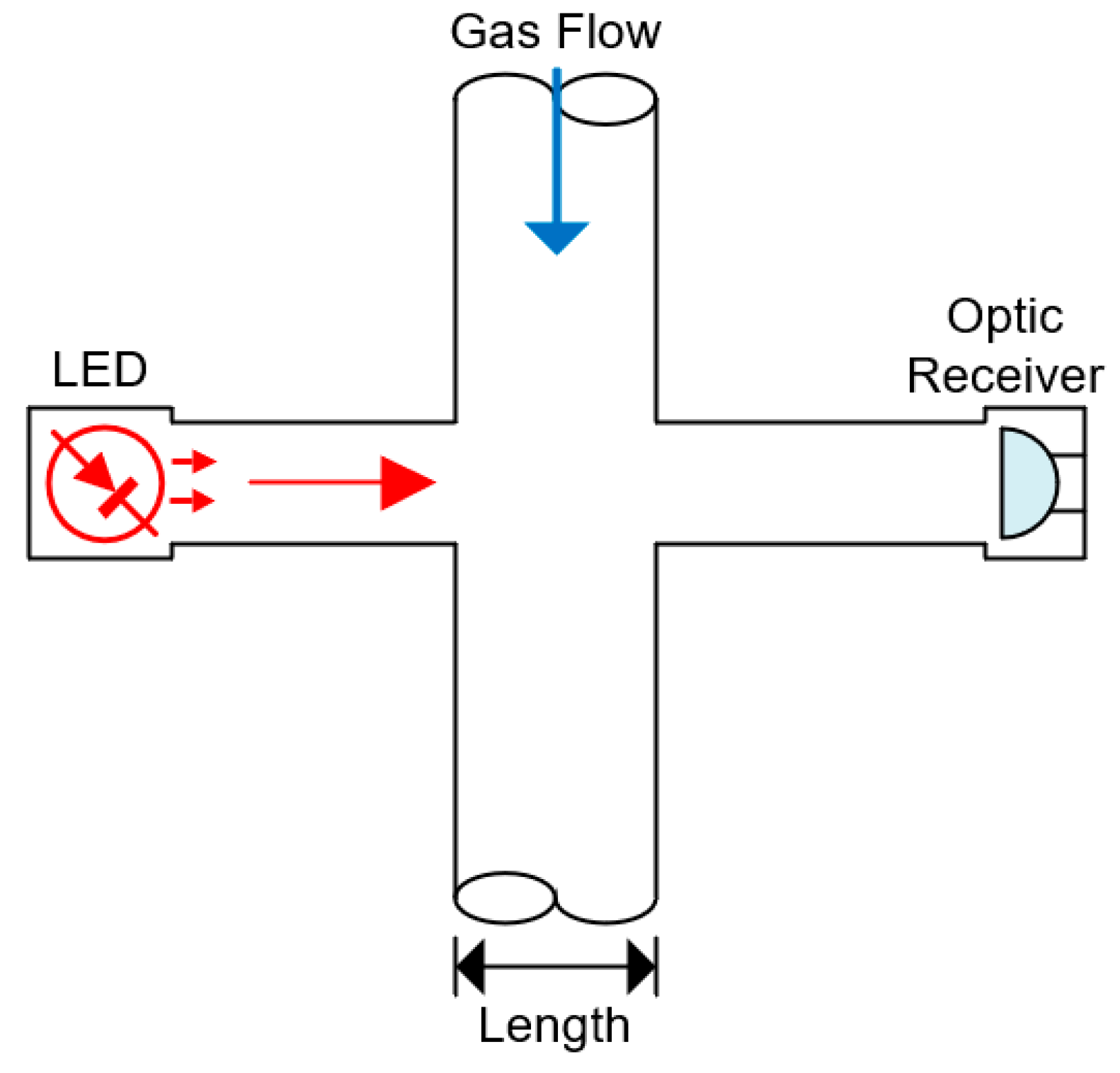
5.7. Optic
6. IoT-Based Wireless Gas Sensors
6.1. Sensing Requirements for IoT-Based Gas Sensors
6.2. Gas-Sensing Solutions for IoT and Wireless Sensors and Actuators Networks (WSANs)
7. Discussion and Open Issues
7.1. Sensing Technologies
7.2. Wireless Gas Sensors
7.3. Open Issues
- Gas sensing could provide valuable data to diverse applications, using the IoT paradigm, offering important data for decisions taken by smart devices. They can provide better experiences to users.
- The improvement of sensing characteristics, miniaturization of transducers and combination of sensing technologies are topics with great potential for research.
- Creation of customized multi-gas smart sensors since, to the best of the authors’ knowledge, there are no these kind of solutions in the literature.
- Proposals following a plug-and-play approach based on IoT focusing on the end-user empowerment to properly configure these devices according to their needs.
- Performance evaluation, demonstration, and validation of available gas transducer proposals in real environments since they only were studied through theoretical and laboratory prototype approaches.
8. Lessons Learned
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Gopal, M.; Singh, V. Control Systems Engineering; Wiley: New York, NY, USA, 2008; Volume SMC-6, No. 9. [Google Scholar]
- Díaz-Cacho, M.; Delgado, E.; Prieto, J.A.G.; López, J. Network adaptive deadband: NCS data flow control for shared networks. Sensors 2012, 12, 16591–16613. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Lin, H.J. Design and Implementation of Smart Home Control Systems Based on Wireless Sensor Networks and Power Line Communications. IEEE Trans. Ind. Electron. 2015, 62, 4430–4442. [Google Scholar] [CrossRef]
- Santos, I.L.; Pirmez, L.; Carmo, L.R.; Pires, P.F.; Delicato, F.C.; Khan, S.U.; Zomaya, A.Y. A Decentralized Damage Detection System for Wireless Sensor and Actuator Networks. IEEE Trans. Comput. 2016, 65, 1363–1376. [Google Scholar] [CrossRef]
- Rawat, P.; Singh, K.D.; Chaouchi, H.; Bonnin, J.M. Wireless sensor networks: A survey on recent developments and potential synergies. J. Supercomput. 2014, 68, 1–48. [Google Scholar] [CrossRef]
- Borges, L.M.; Velez, F.J.; Lebres, A.S. Survey on the characterization and classification of wireless sensor network applications. IEEE Commun. Surv. Tutor. 2014, 16, 1860–1890. [Google Scholar] [CrossRef]
- Oliveira, L.M.L.; Rodrigues, J.J.P.C.; Elias, A.G.F.; Zarpelão, B.B. Ubiquitous monitoring solution for Wireless Sensor Networks with push notifications and end-to-end connectivity. Mob. Inf. Syst. 2014, 10, 19–35. [Google Scholar] [CrossRef]
- Kiumarsi, B.; Vamvoudakis, K.G.; Modares, H.; Lewis, F.L. Optimal and Autonomous Control Using Reinforcement Learning: A Survey. IEEE Trans. Neural Netw. Learn. Syst. 2018, 29, 2042–2062. [Google Scholar] [CrossRef]
- Al Dakheel, J.; Tabet Aoul, K. Building Applications, Opportunities and Challenges of Active Shading Systems: A State-of-the-Art Review. Energies 2017, 10, 1672. [Google Scholar] [CrossRef]
- Eaton, C.; Chong, E.; Maciejewski, A. Multiple-Scenario Unmanned Aerial System Control: A Systems Engineering Approach and Review of Existing Control Methods. Aerospace 2016, 3, 1. [Google Scholar] [CrossRef]
- Al-Fuqaha, A.; Guizani, M.; Mohammadi, M.; Aledhari, M.; Ayyash, M. Internet of Things: A Survey on Enabling Technologies, Protocols, and Applications. IEEE Commun. Surv. Tutor. 2015, 17, 2347–2376. [Google Scholar] [CrossRef]
- Karagiannis, J.; Chatzimisios, V.; Vazquez-Gallego, P.; Alonso-Zarate, F. A Survey on Application Layer Protocols for Internet of Things. Trans. IoT Cloud Comput. 2015, 3, 11–17. [Google Scholar]
- Da Cruz, M.A.A.; Rodrigues, J.J.P.C.; Sangaiah, A.K.; Al-Muhtadi, J.; Korotaev, V. Performance evaluation of IoT middleware. J. Netw. Comput. Appl. 2018, 109, 53–65. [Google Scholar] [CrossRef]
- Oliveira, L.; Rodrigues, J.J.P.C.; Kozlov, S.A.; Rabêlo, R.A.L.; de Albuquerque, V.H.C. MAC layer protocols for internet of things: A survey. Future Internet 2019, 11, 16. [Google Scholar] [CrossRef]
- Barry, M. Canaries in the coal mine. Eur. Respir. J. 2013, 42, 1469–1471. [Google Scholar] [CrossRef] [PubMed]
- Battaglia, M.; Frey, M.; Passetti, E. Accidents at Work and Costs Analysis: A Field Study in a Large Italian Company. Ind. Health 2014, 52, 354–366. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Turgut, P.; Arif Gurel, M.; Kadir Pekgokgoz, R. LPG explosion damage of a reinforced concrete building: A case study in Sanliurfa, Turkey. Eng. Fail. Anal. 2013, 32, 220–235. [Google Scholar] [CrossRef]
- Park, K.; Sam Mannan, M.; Do Jo, Y.; Kim, J.Y.; Keren, N.; Wang, Y. Incident analysis of Bucheon LPG filling station pool fire and BLEVE. J. Hazard. Mater. 2006, 137, 62–67. [Google Scholar] [CrossRef]
- Galada, H.C.; Gurian, P.L.; Corella-Barud, V.; Pérez, F.G.; Velázquez-Angulo, G.; Flores, S.; Montoya, T. Applying the mental models framework to carbon monoxide risk in northern Mexico. Rev. Panam. Salud Pública 2009, 25, 242–253. [Google Scholar] [CrossRef]
- Gupta Chatterjee, S.; Chatterjee, S.; Ray, A.K.; Chakraborty, A.K. Graphene-metal oxide nanohybrids for toxic gas sensor: A review. Sens. Actuators B Chem. 2015, 221, 1170–1181. [Google Scholar] [CrossRef]
- Mirzaei, A.; Leonardi, S.G.; Neri, G. Detection of hazardous volatile organic compounds (VOCs) by metal oxide nanostructures-based gas sensors: A review. Ceram. Int. 2016, 42, 15119–15141. [Google Scholar] [CrossRef]
- Sun, Y.F.; Liu, S.B.; Meng, F.L.; Liu, J.Y.; Jin, Z.; Kong, L.T.; Liu, J.H. Metal oxide nanostructures and their gas sensing properties: A review. Sensors 2012, 12, 2610–2631. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhou, Z.; Yang, Z.; Chen, X.; Xu, D.; Zhang, Y. Gas sensors based on deposited single-walled carbon nanotube networks for DMMP detection. Nanotechnology 2009, 20, 345502. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.S.; Lo, Y.L.; Sung, T.W. Review on recent developments of fluorescent oxygen and carbon dioxide optical fiber sensors. Photonic Sens. 2011, 1, 234–250. [Google Scholar] [CrossRef]
- Llobet, E. Gas sensors using carbon nanomaterials: A review. Sens. Actuators B Chem. 2013, 179, 32–45. [Google Scholar] [CrossRef]
- Lakard, B.; Carquigny, S.; Segut, O.; Patois, T.; Lakard, S. Gas Sensors Based on Electrodeposited Polymers. Metals 2015, 5, 1371–1386. [Google Scholar] [CrossRef]
- Fratoddi, I.; Venditti, I.; Cametti, C.; Russo, M.V. Chemiresistive polyaniline-based gas sensors: A mini review. Sens. Actuators B Chem. 2015, 220, 534–548. [Google Scholar] [CrossRef]
- Liu, X.; Cheng, S.; Liu, H.; Hu, S.; Zhang, D.; Ning, H. A survey on gas sensing technology. Sensors 2012, 12, 9635–9665. [Google Scholar] [CrossRef]
- Chou, C.; Wu, Y.; Lin, C. High performance oxygen sensor utilizing ultraviolet irradiation assisted ZnO nanorods under low operation temperature. In Proceedings of the the 8th Annual IEEE International Conference on Nano/Micro Engineered and Molecular Systems, Suzhou, China, 7–10 April 2013; pp. 72–75. [Google Scholar]
- Sari, W.; Smith, P.; Leigh, S.; Covington, J. Oxygen Sensors Based on Screen Printed Platinum and Palladium Doped Indium Oxides. Proc. Eurosens. 2017, 1, 401. [Google Scholar] [CrossRef]
- Shuk, P.; Jantz, R. Oxygen gas sensing technologies: A comprehensive review. In Proceedings of the 2015 9th International Conference on Sensing Technology, ICST, Auckland, New Zealand, 8–10 December2015; pp. 12–17. [Google Scholar]
- Sieber, A.; Enoksson, P.; Krozer, A. Smart electrochemical oxygen sensor for personal protective equipment. IEEE Sens. J. 2012, 12, 1846–1852. [Google Scholar] [CrossRef]
- Beaubien, S.E.; Ciotoli, G.; Lombardi, S. Carbon dioxide and radon gas hazard in the Alban Hills area (central Italy). J. Volcanol. Geotherm. Res. 2003, 123, 63–80. [Google Scholar] [CrossRef]
- Jeyakkannan, N.; Nagaraj, B. Online monitoring of geological methane storage and leakage based on wireless sensor networks. Asian J. Chem. 2014, 26, S23–S26. [Google Scholar] [CrossRef]
- Al Rasyid, M.U.H.; Nadhori, I.U.; Alnovinda, Y.T. CO and CO2 pollution monitoring based on wireless sensor network. In Proceedings of the 2015 IEEE International Conference on Aerospace Electronics and Remote Sensing (IEEE ICARES 2015), Bali, Indonesia, 3–5 December 2015. [Google Scholar]
- Hampson, N.B. Cost of accidental carbon monoxide poisoning: A preventable expense. Prev. Med. Rep. 2016, 3, 21–24. [Google Scholar] [CrossRef] [PubMed]
- Hampson, N.B.; Dunn, S.L. Carbon monoxide poisoning from portable electrical generators. J. Emerg. Med. 2015, 49, 125–129. [Google Scholar] [CrossRef] [PubMed]
- Jin, H.; Zhou, H.; Zhang, Y. Insight into the mechanism of CO oxidation on WO3(001) surfaces for gas sensing: A DFT study. Sensors 2017, 17, 1898. [Google Scholar] [CrossRef]
- Garzón, J.P.; Huertas, J.I.; Magaña, M.; Huertas, M.E.; Cárdenas, B.; Watanabe, T.; Maeda, T.; Wakamatsu, S.; Blanco, S. Volatile organic compounds in the atmosphere of Mexico City. Atmos. Environ. 2015, 119, 415–429. [Google Scholar] [CrossRef]
- Kamal, M.S.; Razzak, S.A.; Hossain, M.M. Catalytic oxidation of volatile organic compounds (VOCs)—A review. Atmos. Environ. 2016, 140, 117–134. [Google Scholar] [CrossRef]
- Colindres, S.C.; Aguir, K.; Sodi, F.C.; Vargas, L.V.; Salazar, J.M.; Febles, V.G. Ozone sensing based on palladium decorated carbon nanotubes. Sensors 2014, 14, 6806–6818. [Google Scholar] [CrossRef]
- Duvall, R.M.; Long, R.W.; Beaver, M.R.; Kronmiller, K.G.; Wheeler, M.L.; Szykman, J.J. Performance evaluation and community application of low-cost sensors for ozone and nitrogen dioxide. Sensors 2016, 16, 1698. [Google Scholar] [CrossRef]
- Wang, R.; Tsow, F.; Zhang, X.; Peng, J.H.; Forzani, E.S.; Chen, Y.; Crittenden, J.C.; Destaillats, H.; Tao, N. Real-time ozone detection based on a microfabricated quartz crystal tuning fork sensor. Sensors 2009, 9, 5655–5663. [Google Scholar] [CrossRef]
- Yagura, T.; Makita, K.; Yamamoto, H.; Menck, C.F.M.; Schuch, A.P. Biological sensors for solar ultraviolet radiation. Sensors 2011, 11, 4277–4294. [Google Scholar] [CrossRef]
- Rocha, M.; Sthel, M.; Lima, G.; da Silva, M.; Schramm, D.; Miklós, A.; Vargas, H. A sulfur hexafluoride sensor using quantum cascade and CO2 laser-based photoacoustic spectroscopy. Sensors 2010, 10, 9359–9368. [Google Scholar] [CrossRef]
- Xiao, X.; Xu, Y.; Dong, Z. Thermodynamic modeling and analysis of an optical electric-field sensor. Sensors 2015, 15, 7125–7135. [Google Scholar] [CrossRef]
- Dong, M.; Zhang, C.; Ren, M.; Albarracín, R.; Ye, R. Electrochemical and infrared absorption spectroscopy detection of SF6 decomposition products. Sensors 2017, 17, 2627. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Huang, R.; Gui, Y.; Zeng, H. Gas sensing analysis of ag-decorated graphene for sulfur hexafluoride decomposition products based on the density functional theory. Sensors 2016, 16, 1830. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Yu, L.; Tie, J.; Dong, X. Gas sensitivity and sensing mechanism studies on Au-doped TiO2 nanotube arrays for detecting SF6 decomposed components. Sensors 2014, 14, 19517–19532. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Cui, H.; Gui, Y. Synthesis of graphene-based sensors and application on detecting SF6 decomposing products: A review. Sensors 2017, 17, 363. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Luo, C.; Tang, J. Sensitivity characteristic analysis of adsorbent-mixed carbon nanotube sensors for the detection of SF6 decomposition products under PD conditions. Sensors 2013, 13, 15209–15220. [Google Scholar] [CrossRef]
- Zhang, X.; Li, X.; Luo, C.; Dong, X.; Zhou, L. Analysis of the sensitivity of K-type molecular sieve-deposited MWNTs for the detection of SF6 decomposition gases under partial discharge. Sensors 2015, 15, 28367–28384. [Google Scholar] [CrossRef]
- Garnacho, F.; Khamlichi, A.; Rovira, J. The design and characterization of a prototype wideband voltage sensor based on a resistive divider. Sensors 2017, 17, 2657. [Google Scholar] [CrossRef]
- Blanco-Rodríguez, P.; Fernández-Serantes, L.A.; Otero-Pazos, A.; Calvo-Rolle, J.L.; de Cos Juez, F.J. Radon Mitigation Approach in a Laboratory Measurement Room. Sensors 2017, 17, 1090. [Google Scholar] [CrossRef]
- Ting, D.S. Who Handbook on Indoor Radon: A Public Health Perspective; World Health Organization: Geneva, Switzerland, 2010; Volume 67. [Google Scholar]
- Nikezic, D.; Yu, K.N. Are radon gas measurements adequate for epidemiological studies and case control studies of radon-induced lung cancer? Radiat. Prot. Dosim. 2005, 113, 233–235. [Google Scholar] [CrossRef] [PubMed]
- Field, R.W.; Steck, D.J.; Smith, B.J.; Brus, C.P.; Fisher, E.F.; Neuberger, J.S.; Lynch, C.F. The Iowa radon lung cancer study-phase I: Residential radon gas exposure and lung cancer. Sci. Total Environ. 2001, 272, 67–72. [Google Scholar] [CrossRef]
- Darby, S.; Hill, D.; Auvinen, A.; Barros-Dios, J.M.; Baysson, H.; Bochicchio, F.; Deo, H.; Falk, R.; Forastiere, F.; Hakama, M.; et al. Radon in homes and risk of lung cancer: Collaborative analysis of individual data from 13 European case-control studies. Br. Med. J. 2005, 330, 223–226. [Google Scholar] [CrossRef] [PubMed]
- Ioannides, K.; Papachristodoulou, C.; Stamoulis, K.; Karamanis, D.; Pavlides, S.; Chatzipetros, A.; Karakala, E. Soil gas radon: A tool for exploring active fault zones. Appl. Radiat. Isot. 2003, 59, 205–213. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, L.; Wang, H.; Xiong, M.; Yang, T.; Zakharova, G.S. Highly sensitive and selective ammonia gas sensors based on PbS quantum dots/TiO2 nanotube arrays at room temperature. Sens. Actuators B Chem. 2016, 236, 529–536. [Google Scholar] [CrossRef]
- Liu, W.; Liu, Y.Y.; Do, J.S.; Li, J. Highly sensitive room temperature ammonia gas sensor based on Ir-doped Pt porous ceramic electrodes. Appl. Surf. Sci. 2016, 390, 929–935. [Google Scholar] [CrossRef]
- Renganathan, B.; Sastikumar, D.; Gobi, G.; X Rajeswari Yogamalar, N.; Chandra Bose, A. Nanocrystalline ZnO coated fiber optic sensor for ammonia gas detection. Opt. Laser Technol. 2011, 43, 1398–1404. [Google Scholar] [CrossRef]
- Gautam, M.; Jayatissa, A.H. Ammonia gas sensing behavior of graphene surface decorated with gold nanoparticles. Solid-State Electron. 2012, 78, 159–165. [Google Scholar] [CrossRef]
- Hakimi, M.; Salehi, A.; Boroumand, F.A.; Mosleh, N. Fabrication of a Room Temperature Ammonia Gas Sensor Based on Polyaniline with N-Doped Graphene Quantum Dots. IEEE Sens. J. 2018, 18, 2245–2252. [Google Scholar] [CrossRef]
- Gatty, H.K.; Leijonmarck, S.; Antelius, M.; Stemme, G.; Roxhed, N. An amperometric nitric oxide sensor with fast response and ppb-level concentration detection relevant to asthma monitoring. Sens. Actuators B Chem. 2015, 209, 639–644. [Google Scholar] [CrossRef]
- Gatty, H.K.; Stemme, G.; Roxhed, N. A wafer-level liquid cavity integrated amperometric gas sensor with ppb-level nitric oxide gas sensitivity. J. Micromech. Microeng. 2015, 25, 105013. [Google Scholar] [CrossRef]
- Sluis, W.W.; Allaart, M.A.F.; Piters, A.J.M.; Gast, L.F.L. The development of a nitrogen dioxide sonde. Atmos. Meas. Tech. 2010, 3, 1753–1762. [Google Scholar] [CrossRef]
- Shu, J.H.; Wikle, H.C.; Chin, B.A. Passive chemiresistor sensor based on iron (II) phthalocyanine thin films for monitoring of nitrogen dioxide. Sens. Actuators B Chem. 2010, 148, 498–503. [Google Scholar] [CrossRef]
- Tabassum, R.; Pavelyev, V.S.; Moskalenko, A.S.; Tukmakov, K.N.; Islam, S.S.; Mishra, P. A Highly Sensitive Nitrogen Dioxide Gas Sensor Using Horizontally Aligned SWCNTs Employing MEMS and Dielectrophoresis Methods. IEEE Sens. Lett. 2017, 2, 1–4. [Google Scholar] [CrossRef]
- Peterson, P.J.D.; Aujla, A.; Grant, K.H.; Brundle, A.G.; Thompson, M.R.; Vande Hey, J.; Leigh, R.J. Practical use of metal oxide semiconductor gas sensors for measuring nitrogen dioxide and ozone in urban environments. Sensors 2017, 17, 1653. [Google Scholar] [CrossRef] [PubMed]
- Gao, X.; Sun, Y.; Zhu, C.; Li, C.; Ouyang, Q.; Chen, Y. Highly sensitive and selective H2S sensor based on porous ZnFe2O4 nanosheets. Sens. Actuators B Chem. 2017, 246, 662–672. [Google Scholar] [CrossRef]
- Kumar, A.; Samanta, S.; Singh, A.; Roy, M.; Singh, S.; Basu, S.; Chehimi, M.M.; Roy, K.; Ramgir, N.; Navaneethan, M.; et al. Fast Response and High Sensitivity of ZnO Nanowires—Cobalt Phthalocyanine Heterojunction Based H2S Sensor. ACS Appl. Mater. Interfaces 2015, 7, 17713–17724. [Google Scholar] [CrossRef]
- Malek Alaie, M.; Jahangiri, M.; Rashidi, A.M.; Haghighi Asl, A.; Izadi, N. A novel selective H2S sensor using dodecylamine and ethylenediamine functionalized graphene oxide. J. Ind. Eng. Chem. 2015, 29, 97–103. [Google Scholar] [CrossRef]
- Van Dang, T.; Duc Hoa, N.; Van Duy, N.; Van Hieu, N. Chlorine Gas Sensing Performance of On-Chip Grown ZnO, WO3, and SnO2 Nanowire Sensors. ACS Appl. Mater. Interfaces 2016, 8, 4828–4837. [Google Scholar] [CrossRef]
- Massa, C.B.; Scott, P.; Abramova, E.; Gardner, C.; Laskin, D.L.; Gow, A.J. Acute chlorine gas exposure produces transient inflammation and a progressive alteration in surfactant composition with accompanying mechanical dysfunction. Toxicol. Appl. Pharmacol. 2014, 278, 53–64. [Google Scholar] [CrossRef]
- Govier, P.; Coulson, J.M. Civilian exposure to chlorine gas: A systematic review. Toxicol. Lett. 2018, 293, 249–252. [Google Scholar] [CrossRef] [PubMed]
- DeCoste, J.B.; Browe, M.A.; Wagner, G.W.; Rossin, J.A.; Peterson, G.W. Removal of chlorine gas by an amine functionalized metal–organic framework via electrophilic aromatic substitution. Chem. Commun. 2015, 51, 12474–12477. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.W.; Chiang, C.C. Sandwiched long-period fiber grating fabricated by MEMS process for CO2 gas detection. Micromachines 2016, 7, 35. [Google Scholar] [CrossRef] [PubMed]
- Çengel, Y.A.; Boles, M.A. Thermodynamics: An Engineering Approach with Student Resources DVD; McGraw-Hill Science/Engineering/Math: New York, NY, USA, 2010. [Google Scholar]
- Cengel, Y.A. Heat Transfer: A Practical Approach; McGraw-Hill Science/Engineering/Math: New York, NY, USA, 2003. [Google Scholar]
- Genta, C.; Marotta, C.; Migliardini, F. Study and Development of a Complete System for Recovery, Recycle, and Disposal of Refrigerant Gas from Existent Plants. J. Eng. (USA) 2017, 2017, 1–9. [Google Scholar] [CrossRef]
- Ghandehari, M.; Aghamohamadnia, M.; Dobler, G.; Karpf, A.; Buckland, K.; Qian, J.; Koonin, S. Mapping Refrigerant Gases in the New York City Skyline. Sci. Rep. 2017, 7, 2735. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.I.; Lin, C.C. A study of storage tank accidents. J. Loss Prev. Process Ind. 2006, 19, 51–59. [Google Scholar] [CrossRef]
- Barrett, K.; Brooks, H.; Boitano, S.; Barman, S. Ganong’s Review of Medical Physiology, 23rd ed.; McGraw-Hill Medical: New York, NY, USA, 2010. [Google Scholar]
- Gardner, J.W.; Guha, P.K.; Udrea, F.; Covington, J.A. CMOS interfacing for integrated gas sensors: A review. IEEE Sens. J. 2010, 10, 1833–1848. [Google Scholar] [CrossRef]
- Li, H.; Sam Boling, C.; Mason, A.J. CMOS Amperometric ADC with High Sensitivity, Dynamic Range and Power Efficiency for Air Quality Monitoring. IEEE Trans. Biomed. Circuits Syst. 2016, 10, 817–827. [Google Scholar] [CrossRef]
- Adgate, J.L.; Goldstein, B.D.; McKenzie, L.M. Potential public health hazards, exposures and health effects from unconventional natural gas development. Environ. Sci. Technol. 2014, 48, 8307–8320. [Google Scholar] [CrossRef]
- Fthenakis, V.M. Overview of potential hazards. In McEvoy’s Handbook of Photovoltaics: Fundamentals and Applications; Elsevier: Amsterdam, The Netherlands, 2017; pp. 1195–1212. [Google Scholar]
- Da Cruz, M.A.A.; Rodrigues, J.J.P.C.; Al-Muhtadi, J.; Korotaev, V.V.; de Albuquerque, V.H.C. A Reference Model for Internet of Things Middleware. IEEE Internet Things J. 2018, 5, 871–883. [Google Scholar] [CrossRef]
- Li, H.; Mu, X.; Yang, Y.; Mason, A.J. Low power multimode electrochemical gas sensor array system for wearable health and safety monitoring. IEEE Sens. J. 2014, 14, 3391–3399. [Google Scholar] [CrossRef]
- Wang, L.; Wu, Y.; Yin, P.; Cheng, P.; Liu, Y.; Schwebel, D.C.; Qi, J.; Ning, P.; Liu, J.; Cheng, X.; et al. Poisoning deaths in China, 2006–2016. Bull. World Health Organ. 2018, 96, 314–326A. [Google Scholar] [CrossRef] [PubMed]
- Chalk, R. The miners’ canary. Bull. Atomic Sci. 1982, 38, 16–22. [Google Scholar] [CrossRef]
- Jelicic, V.; Magno, M.; Brunelli, D.; Paci, G.; Benini, L. Context-adaptive multimodal wireless sensor network for energy-efficient gas monitoring. IEEE Sens. J. 2013, 13, 328–338. [Google Scholar] [CrossRef]
- Saharudin, S.; Isha, K.M.; Mahmud, Z.; Herman, S.H.; Noor, U.M. Performance evaluation of optical fiber sensor using different oxygen sensitive nano-materials. In Proceedings of the 4th International Conference on Photonics (ICP 2013), Melaka, Malaysia, 28–30 October 2013; pp. 309–312. [Google Scholar]
- Niazi, A.; Anthony, C.J. Development of Oxygen Sensor by Integrating the Low Cost Printed Circuit Board Technology and Solid Electrolyte Membrane. In Proceedings of the International Conference on Biomedical Engineering and Systems (ICBES 2014), Prague, Czech Republic, 14–15 August 2014; Volume 137, pp. 1–7. [Google Scholar]
- Shuk, P.; Jantz, R.; Guth, H.U. Oxygen sensor with advanced oxide electrode materials. Int. J. Smart Sens. Intell. Syst. 2012, 5, 233–245. [Google Scholar] [CrossRef]
- Chen, R.; Formenti, F.; McPeak, H.; Obeid, A.N.; Hahn, C.E.W.; Farmery, A.D. Optimizing design for polymer fiber optic oxygen sensors. IEEE Sens. J. 2014, 14, 3358–3364. [Google Scholar] [CrossRef]
- Verma, R.; Kamble, S.S.; Radhakrishnan, J.K. Tunable Diode Laser Absorption Spectroscopy based Oxygen Sensor. In Proceedings of the 2012 Sixth International Conference on Sensing Technology (ICST), Kolkata, India, 18–21 December 2012; Volume 4, pp. 130–135. [Google Scholar]
- Yan, S.; Shan, M. An acoustic method on sulfur hexafluoride concentration detection. In Proceedings of the 2014 IEEE Workshop on Electronics, Computer and Applications, IWECA 2014, Ottawa, ON, Canada, 8–9 May 2014; pp. 485–488. [Google Scholar]
- Sonoyama, M.; Kato, Y.; Fujita, H. Application of ultrasonic to a hydrogen sensor. In Proceedings of the IEEE Sensors, Kona, HI, USA, 1–4 November 2010; pp. 2141–2144. [Google Scholar]
- Shan, M.; Li, X.; Zhu, C.; Zhang, J. Gas concentration detection using ultrasonic based on wireless sensor networks. In Proceedings of the 2nd International Conference on Information Science and Engineering (ICISE 2010), Hangzhou, China, 3–5 December 2010; pp. 2101–2106. [Google Scholar]
- Petculescu, A.; Hall, B.; Fraenzle, R.; Phillips, S.; Lueptow, R.M. A prototype acoustic gas sensor based on attenuation. J. Acoust. Soc. Am. 2006, 120, 1779–1782. [Google Scholar] [CrossRef]
- Ortiz Perez, A.; Bierer, B.; Scholz, L.; Wöllenstein, J.; Palzer, S. A Wireless Gas Sensor Network to Monitor Indoor Environmental Quality in Schools. Sensors 2018, 18, 4345. [Google Scholar] [CrossRef]
- Suh, J.H.; Cho, I.; Kang, K.; Kweon, S.-J.; Lee, M.; Yoo, H.-J.; Park, I. Fully integrated and portable semiconductor-type multi-gas sensing module for IoT applications. Sens. Actuators B Chem. 2018, 265, 660–667. [Google Scholar] [CrossRef]
- Sun, L.; JOE Wong, K.C.; Wei, P.; Ye, S.; Huang, H.; Yang, F.; Westerdahl, D.; Louie, P.K.K.; Luk, C.W.Y.; Ning, Z. Development and application of a next generation air sensor network for the Hong Kong marathon 2015 air quality monitoring. Sensors 2016, 16, 211. [Google Scholar] [CrossRef]
- Al Rasyid, Y.T. M Udin Harun and Nadhori, Isbat Uzzin and Sudarsono, Amang and Alnovinda, Pollution monitoring system using gas sensor based on wireless sensor network. Int. J. Eng. Technol. Innov. 2016, 6, 79–91. [Google Scholar]
- Kumar, A.; Hancke, G.P. Energy efficient environment monitoring system based on the IEEE 802.15.4 standard for low cost requirements. IEEE Sens. J. 2014, 14, 2557–2566. [Google Scholar] [CrossRef]
- Somov, A.; Baranov, A.; Spirjakin, D. A wireless sensor-actuator system for hazardous gases detection and control. Sens. Actuators A Phys. 2014, 210, 157–164. [Google Scholar] [CrossRef]
- Moltchanov, S.; Levy, I.; Etzion, Y.; Lerner, U.; Broday, D.M.; Fishbain, B. On the feasibility of measuring urban air pollution by wireless distributed sensor networks. Sci. Total Environ. 2015, 502, 537–547. [Google Scholar] [CrossRef]
- Dong, S.; Duan, S.; Yang, Q.; Zhang, J.; Li, G.; Tao, R. MEMS-Based Smart Gas Metering for Internet of Things. IEEE Internet Things J. 2017, 4, 1296–1303. [Google Scholar] [CrossRef]
- Baranov, A.; Spirjakin, D.; Akbari, S.; Somov, A.; Passerone, R. POCO: ‘Perpetual’ operation of CO wireless sensor node with hybrid power supply. Sens. Actuators A Phys. 2016, 238, 112–121. [Google Scholar] [CrossRef]
- Choi, S.; Kim, N.; Cha, H.; Ha, R. Micro sensor node for air pollutant monitoring: Hardware and software issues. Sensors 2009, 9, 7970–7987. [Google Scholar] [CrossRef]
- Wan, H.; Yin, H.; Mason, A.J. Rapid measurement of room temperature ionic liquid electrochemical gas sensor using transient double potential amperometry. Sens. Actuators B Chem. 2017, 242, 658–666. [Google Scholar] [CrossRef]
- Miura, N.; Sato, T.; Anggraini, S.A.; Ikeda, H.; Zhuiykov, S. A review of mixed-potential type zirconia-based gas sensors. Ionics 2014, 20, 901–925. [Google Scholar] [CrossRef]
- Chu, C.-S.; Syu, J.-J. The Development of a Highly Sensitive Fiber-Optic Oxygen Sensor. Inventions 2016, 1, 9. [Google Scholar] [CrossRef]
- Eich, S.; Schmälzlin, E.; Löhmannsröben, H.G. Distributed fiber optical sensing of oxygen with optical time domain reflectometry. Sensors 2013, 13, 7170–7183. [Google Scholar] [CrossRef] [PubMed]
- Simões, F.R.; Xavier, M.G. Electrochemical Sensors. Nanosci. Appl. 2017, 74, 155–178. [Google Scholar]
- Wang, H.; Chen, L.; Wang, J.; Sun, Q.; Zhao, Y. A micro oxygen sensor based on a nano sol-gel TiO2 thin film. Sensors 2014, 14, 16423–16433. [Google Scholar] [CrossRef] [PubMed]
- Bertuna, A.; Faglia, G.; Ferroni, M.; Kaur, N.; Munasinghe Arachchige, H.M.M.; Sberveglieri, G.; Comini, E. Metal oxide nanowire preparation and their integration into chemical sensing devices at the sensor lab in Brescia. Sensors 2017, 17, 1000. [Google Scholar] [CrossRef]
- Basu, P.K.; Benedict, S.S.; Kallat, S.; Bhat, N. A Suspended Low Power Gas Sensor with In-Plane Heater. J. Microelectromech. Syst. 2017, 26, 48–50. [Google Scholar] [CrossRef]
- Yeh, L.K.; Luo, J.C.; Chen, M.C.; Wu, C.H.; Chen, J.Z.; Chen, I.C.; Hsu, C.C.; Tian, W.C. A photoactivated gas detector for toluene sensing at room temperature based on new coral-like ZnO nanostructure arrays. Sensors 2016, 16, 1820. [Google Scholar] [CrossRef]
- Binions, R.; Naik, A.J.T. Metal oxide semiconductor gas sensors in environmental monitoring. Semicond. Gas Sens. 2013, 10, 433–466. [Google Scholar]
- Jin, Y.; Zhang, N.; Zhang, B. Fabrication of p-Type ZnO: N films by oxidizing Zn3N2 films in oxygen plasma at low temperature. Materials 2017, 10, 236. [Google Scholar] [CrossRef]
- Stratulat, A.; Serban, B.-C.; de Luca, A.; Avramescu, V.; Cobianu, C.; Brezeanu, M.; Buiu, O.; Diamandescu, L.; Feder, M.; Ali, S.Z.; et al. Low power resistive oxygen sensor based on sonochemical SrTi-0.6-Fe-0.4-O-2.8 (STFO40). Sensors 2015, 15, 17495–17506. [Google Scholar] [CrossRef]
- Lahlalia, A.; Filipovic, L.; Selberherr, S. Modeling and Simulation of Novel Semiconducting Metal Oxide Gas Sensors for Wearable Devices. IEEE Sens. J. 2018, 18, 1960–1970. [Google Scholar] [CrossRef]
- Korotcenkov, G. Handbook of Gas Sensor Materials; Springer: New York, NY, USA, 2014; Volume 2. [Google Scholar]
- Grossel, S.S. Hazardous gas monitoring: A guide for semiconductor and other hazardous occupancies (2000). J. Loss Prev. Process Ind. 2002, 15, 249–250. [Google Scholar] [CrossRef]
- Lutic, D.; Sanati, M.; Spetz, A.L. Gas Sensors. In Synthesis, Properties, and Applications of Oxide Nanomaterials; Sberveglieri, G., Ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2006; pp. 411–450. [Google Scholar]
- Bai, H.; Shi, G. Gas sensors based on conducting polymers. Sensors 2007, 7, 267–307. [Google Scholar] [CrossRef]
- Byshkin, M.S.; Buonocore, F.; Di Matteo, A.; Milano, G. A unified bottom up multiscale strategy to model gas sensors based on conductive polymers. Sens. Actuators B Chem. 2015, 211, 42–51. [Google Scholar] [CrossRef]
- Liu, N.; Liang, W.; Mai, J.D.; Liu, L.; Bin Lee, G.; Li, W.J. Rapid fabrication of nanomaterial electrodes using digitally controlled electrokinetics. IEEE Trans. Nanotechnol. 2014, 13, 245–253. [Google Scholar] [CrossRef]
- Kim, J.H.; Song, M.J.; Kim, K.B.; Jin, J.H.; Min, N.K. Evaluation of surface cleaning procedures in terms of gas sensing properties of spray-deposited CNT film: Thermal- and O2 plasma treatments. Sensors 2017, 17, 73. [Google Scholar] [CrossRef]
- Jacobson, S. New developments in ultrasonic gas analysis and flowmetering. In Proceedings of the 2008 IEEE Ultrasonics Symposium, Beijing, China, 2–5 November 2008; pp. 508–516. [Google Scholar]
- Ong, Y.T.; Ahmad, A.L.; Zein, S.H.S.; Tan, S.H. A review on carbon nanotubes in an environmental protection and green engineering perspective. Braz. J. Chem. Eng. 2010, 27, 227–242. [Google Scholar] [CrossRef]
- De Volder, M.F.L.; Tawfick, S.H.; Baughman, R.H.; Hart, A.J. Carbon nanotubes: Present and future commercial applications. Science 2013, 339, 535–539. [Google Scholar] [CrossRef]
- Gordon, I.E.; Rothman, L.S.; Hill, C.; Kochanov, R.V.; Tan, Y.; Bernath, P.F.; Birk, M.; Boudon, V.; Campargue, A.; Chance, K.V.; et al. The HITRAN2016 molecular spectroscopic database. J. Quant. Spectrosc. Radiative Transf. 2017, 203, 3–69. [Google Scholar] [CrossRef]
- Shao, S.; Huang, Y.; Tao, S. Simultaneous monitoring of ammonia and moisture using a single fiber optoelectrode as a transducer. IEEE Sens. J. 2014, 14, 847–852. [Google Scholar] [CrossRef]
- Iwata, T.; Katagiri, T.; Matsuura, Y. Real-time analysis of isoprene in breath by using ultraviolet-absorption spectroscopy with a hollow optical fiber gas cell. Sensors 2016, 16, 2058. [Google Scholar] [CrossRef]
- Melo, L.; Burton, G.; Davies, B.; Risk, D.; Wild, P. Highly sensitive coated long period grating sensor for CO2 detection at atmospheric pressure. Sens. Actuators B Chem. 2014, 202, 294–300. [Google Scholar] [CrossRef]
- Wysokiński, K.; Napierała, M.; Stańczyk, T.; Lipiński, S.; Nasiłowski, T. Study on the sensing coating of the optical fibre CO2 sensor. Sensors 2015, 15, 31888–31903. [Google Scholar] [CrossRef] [PubMed]
- Mi, G.; Horvath, C.; Aktary, M.; Van, V. Silicon microring refractometric sensor for atmospheric CO2 gas monitoring. Opt. Express 2016, 24, 1773. [Google Scholar] [CrossRef] [PubMed]
- Sansen, W.; De Wachter, D.; Callewaert, L.; Lambrechts, M.; Claes, A. A smart sensor for the voltammetric measurement of oxygen or glucose concentrations. Sens. Actuators B Chem. 1990, 1, 298–302. [Google Scholar] [CrossRef]
- Ong, K.G.; Zeng, K.; Grimes, C.A. A wireless, passive carbon nanotube-based gas sensor. IEEE Sens. J. 2002, 2, 82–88. [Google Scholar]
- Dominguez, D.D.; McGill, R.A.; Chung, R.; Nguyen, V. Performance of an embedded SAW sensor for filter bed monitor and the development of a wireless monitoring prototype system. In Proceedings of the 1998 IEEE International Frequency Control Symposium (IEEE IFCS 1998), Pasadena, CA, USA, 29 May 1998; pp. 602–607. [Google Scholar]
- Peng, C.; Qian, K.; Wang, C. Design and Application of a VOC-Monitoring System Based on a ZigBee Wireless Sensor Network. IEEE Sens. J. 2015, 15, 2255–2268. [Google Scholar] [CrossRef]
- Gomes, J.B.A.; Rodrigues, J.J.P.C.; Al-Muhtadi, J.; Arunkumar, N.; Rabelo, R.A.L.; Furtado, V. An IoT-Based Smart Solution for Preventing Domestic CO and LPG Gas Accidents. In Proceedings of the 2018 IEEE 10th Latin-American Conference on Communications (IEEE LATINCOM 2018), Guadalajara, Mexico, 14–16 November 2018; pp. 1–6. [Google Scholar]
- Ortiz Pérez, A.; Kallfaß-de Frenes, V.; Filbert, A.; Kneer, J.; Bierer, B.; Held, P.; Klein, P.; Wöllenstein, J.; Benyoucef, D.; Kallfaß, S.; et al. Odor-Sensing System to Support Social Participation of People Suffering from Incontinence. Sensors 2016, 17, 58. [Google Scholar] [CrossRef]
- Scholz, L.; Ortiz Perez, A.; Bierer, B.; Eaksen, P.; Wollenstein, J.; Palzer, S. Miniature Low-Cost Carbon Dioxide Sensor for Mobile Devices. IEEE Sens. J. 2017, 17, 2889–2895. [Google Scholar] [CrossRef]
- Wittstock, V.; Scholz, L.; Bierer, B.; Perez, A.O.; Wöllenstein, J.; Palzer, S. Design of a LED-based sensor for monitoring the lower explosion limit of methane. Sens. Actuators B Chem. 2017, 247, 930–939. [Google Scholar] [CrossRef]
| Technology | Description | Lifetime | Strengths | Weaknesses |
|---|---|---|---|---|
| Electrochemical | Composed of a membrane, an electrolyte, and electrodes. Reacts with the target gases, generating a variation on the output signal. | 2–5 years |
|
|
| Metal Oxide Semiconductors (MOS) | Consists of a metal oxide semiconductor connected through a wire, involved in a ceramic structure, where it is heated. | 2–5 years |
|
|
| Catalytic | Consists of two elements dispersed on a substrate. The active element reacts with the target gases, generating a variation on its resistance. | Up to 10 years |
|
|
| Polymers | Reacts with the target gases, generating an output signal variation according with the concentration of the target gas; the materials are consumed by the reaction, and have a short lifetime | <6 months |
|
|
| Carbon Nanotubes (CNTs) | The interaction between the nanotubes and gas molecules changes the electron configuration of these nanostructures, allowing the measurement of differences on the output current or tension. | n/a 1 |
|
|
| Acoustic | Relies on sound-propagation characteristics to determine the gases on the sensor. Can be used to measure gas flow in pipes. | 5–10 years |
|
|
| Optic | Relies on wavelength-propagation characteristics to determine the gases in the mixture. It can be used to determine more than one gas | 5–10 years |
|
|
| Ref. | Description | Gas | Sensing Tech. | Wireless | Strengths | Weaknesses |
|---|---|---|---|---|---|---|
| [34] | Remote gas monitoring system. Data collected are transferred to a database and displayed on a webpage. | CO2 | Optic | 2G |
|
|
| [93] | Energy efficient gas sensor for indoor air quality. Nodes exchange information in order to save energy | VOC CO | MOS | ZigBee |
|
|
| [106] | Sensor collects environmental data and transfers it to an online dashboard through a gateway | CO2 | Photoacoustic | Z-Wave |
|
|
| [107] | Portable multi-gas sensor that uses a smartphone as a gateway. Data available on Google spreadsheet | H2S CO | MOS | Wi-Fi Bluetooth |
|
|
| [108] | Development and validation of a multi-gas sensor for remotely monitoring outdoor air quality | CO NO2 | Electrochemical | 2G |
|
|
| [110] | A multi-gas sensor with automatic periodic calibration. | SO2 NO2 CO CO2 O2 | Electrochemical | ZigBee |
|
|
| [104] | Wireless sensor with hybrid power supply to monitor outdoor areas, focusing on energy efficiency | CO | Electrochemical | ZigBee |
|
|
| [111] | Gas sensor to identify gas leakages on industrial environments and gas distribution systems, integrated with an actuator to immediately interrupt the detected leakage | CH4 | Catalytic | ZigBee |
|
|
| [112] | Presents a WSN to collect data on gaseous air pollutants on urban areas. Sensed information is stored on a database | O3 NO2 VOC | MOS | 2G |
|
|
| [103] | Describes the deployment of the sensor in various networks, although no data transfer to online dashboards was reported, nor application layer protocols were described | Natural Gas | MOS | 2G 3G 4G LoRa- WAN Bluetooth |
|
|
| [105] | Multi-gas wireless sensor using one MCU to collect the data from the transducers and another to transfer the information to the network | CO CO2 NO2 CH4 | Optic, Electrochemical, MOS | ZigBee |
|
|
| [145] | Indoor gas sensor with M2M communication data displayed on an online dashboard. The collected data is stored on a local database | VOC | Optic | ZigBee |
|
|
| [146] | CO and LPG sensor for domestic environments in order to prevent accidents. Data is transferred to a local MQTT broker | CO LPG | MOS | 6LoWPAN over IEEE 802.15.4 |
|
|
| [147] | A sensor for incontinence patients to be connected to smartphones, capable of detecting methylmercaptan, mmonia, and dimetylsulfide | NH3 C3SH (CH3)2S | MOS | Bluetooth Low Energy |
|
|
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomes, J.B.A.; Rodrigues, J.J.P.C.; Rabêlo, R.A.L.; Kumar, N.; Kozlov, S. IoT-Enabled Gas Sensors: Technologies, Applications, and Opportunities. J. Sens. Actuator Netw. 2019, 8, 57. https://doi.org/10.3390/jsan8040057
Gomes JBA, Rodrigues JJPC, Rabêlo RAL, Kumar N, Kozlov S. IoT-Enabled Gas Sensors: Technologies, Applications, and Opportunities. Journal of Sensor and Actuator Networks. 2019; 8(4):57. https://doi.org/10.3390/jsan8040057
Chicago/Turabian StyleGomes, João B. A., Joel J. P. C. Rodrigues, Ricardo A. L. Rabêlo, Neeraj Kumar, and Sergey Kozlov. 2019. "IoT-Enabled Gas Sensors: Technologies, Applications, and Opportunities" Journal of Sensor and Actuator Networks 8, no. 4: 57. https://doi.org/10.3390/jsan8040057
APA StyleGomes, J. B. A., Rodrigues, J. J. P. C., Rabêlo, R. A. L., Kumar, N., & Kozlov, S. (2019). IoT-Enabled Gas Sensors: Technologies, Applications, and Opportunities. Journal of Sensor and Actuator Networks, 8(4), 57. https://doi.org/10.3390/jsan8040057








