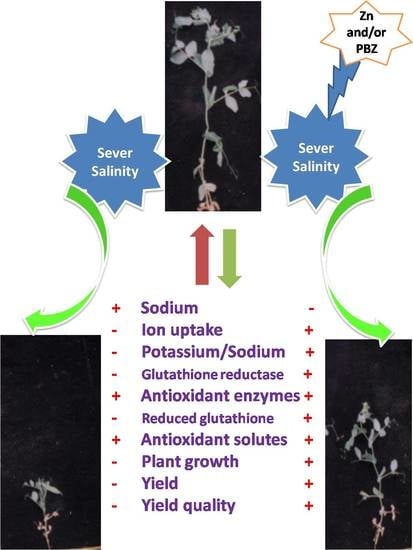
Zinc and Paclobutrazol Mediated Regulation of Growth, Upregulating Antioxidant Aptitude and Plant Productivity of Pea Plants under Salinity
Abstract
1. Introduction
2. Results
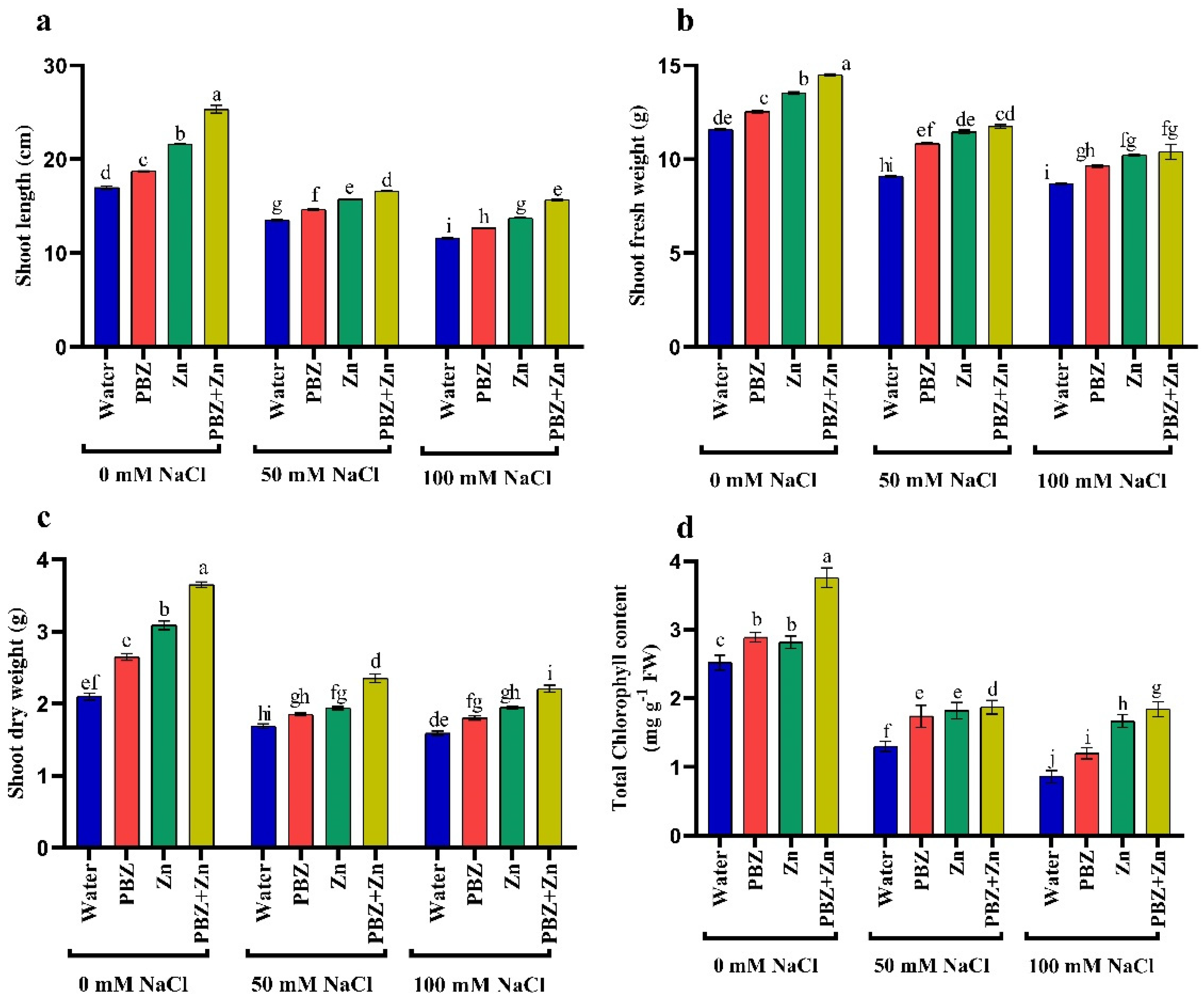
2.1. Plant Growth
2.2. Chlorophyll
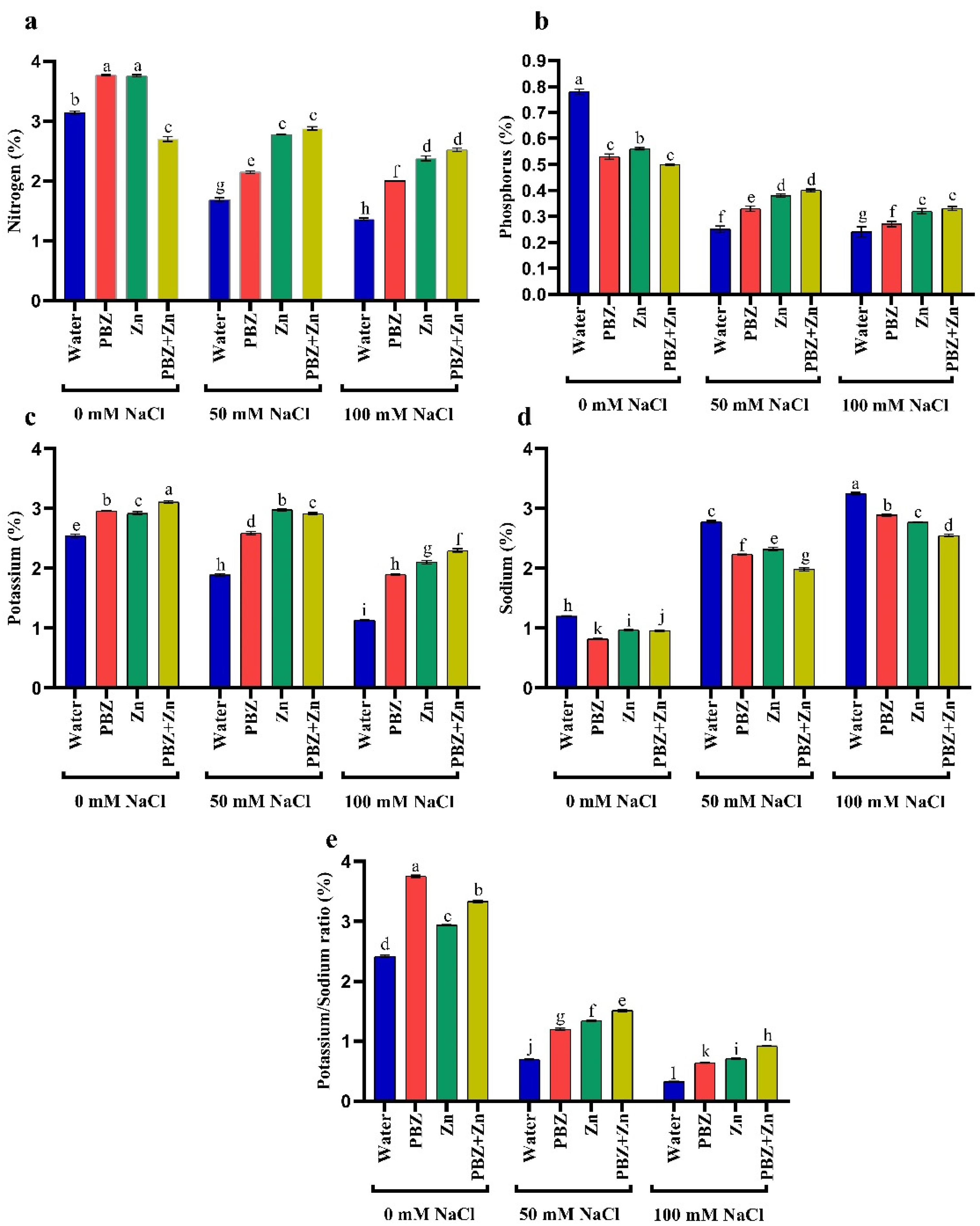
2.3. Ion Percentage
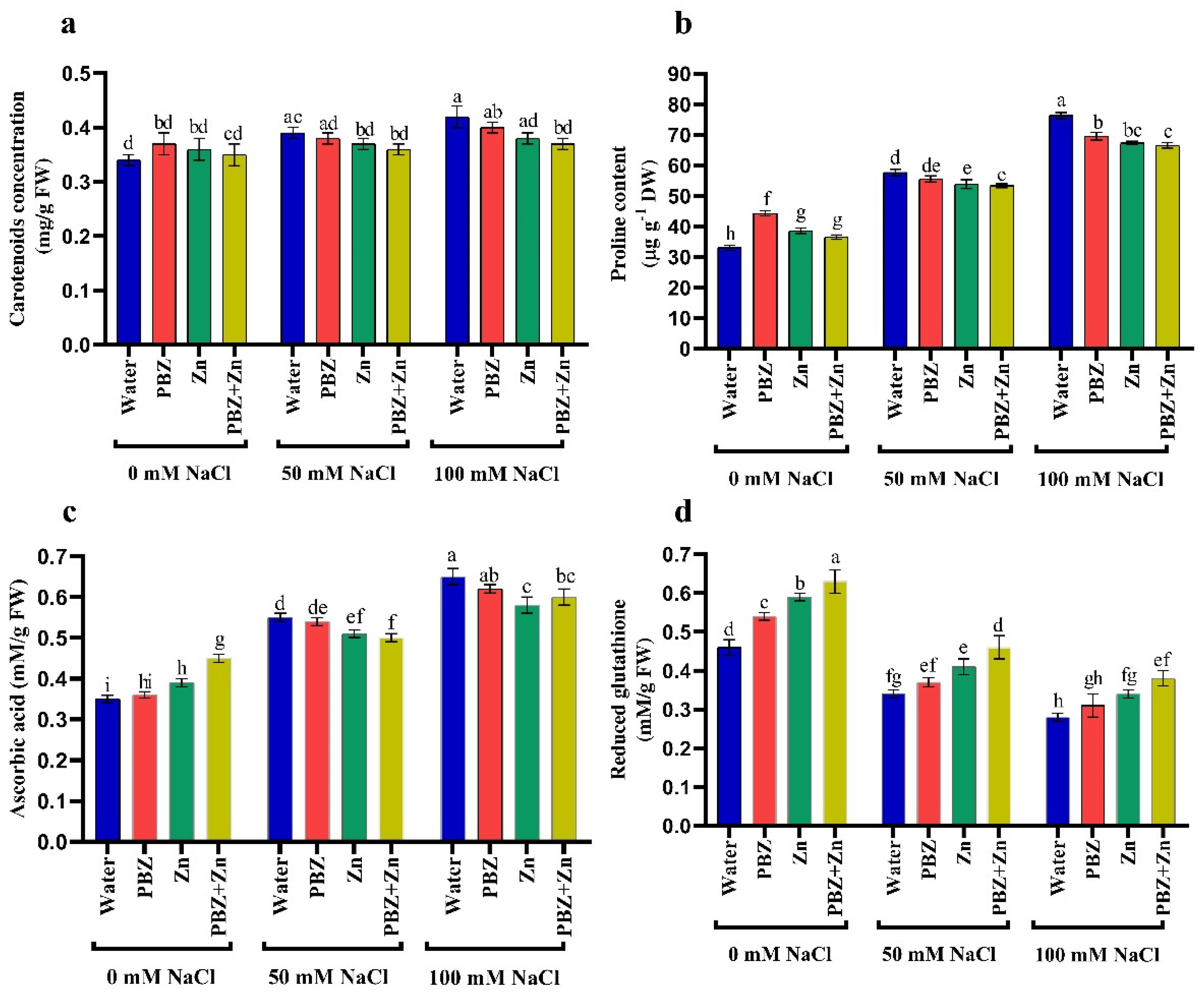
2.4. Antioxidant Solutes
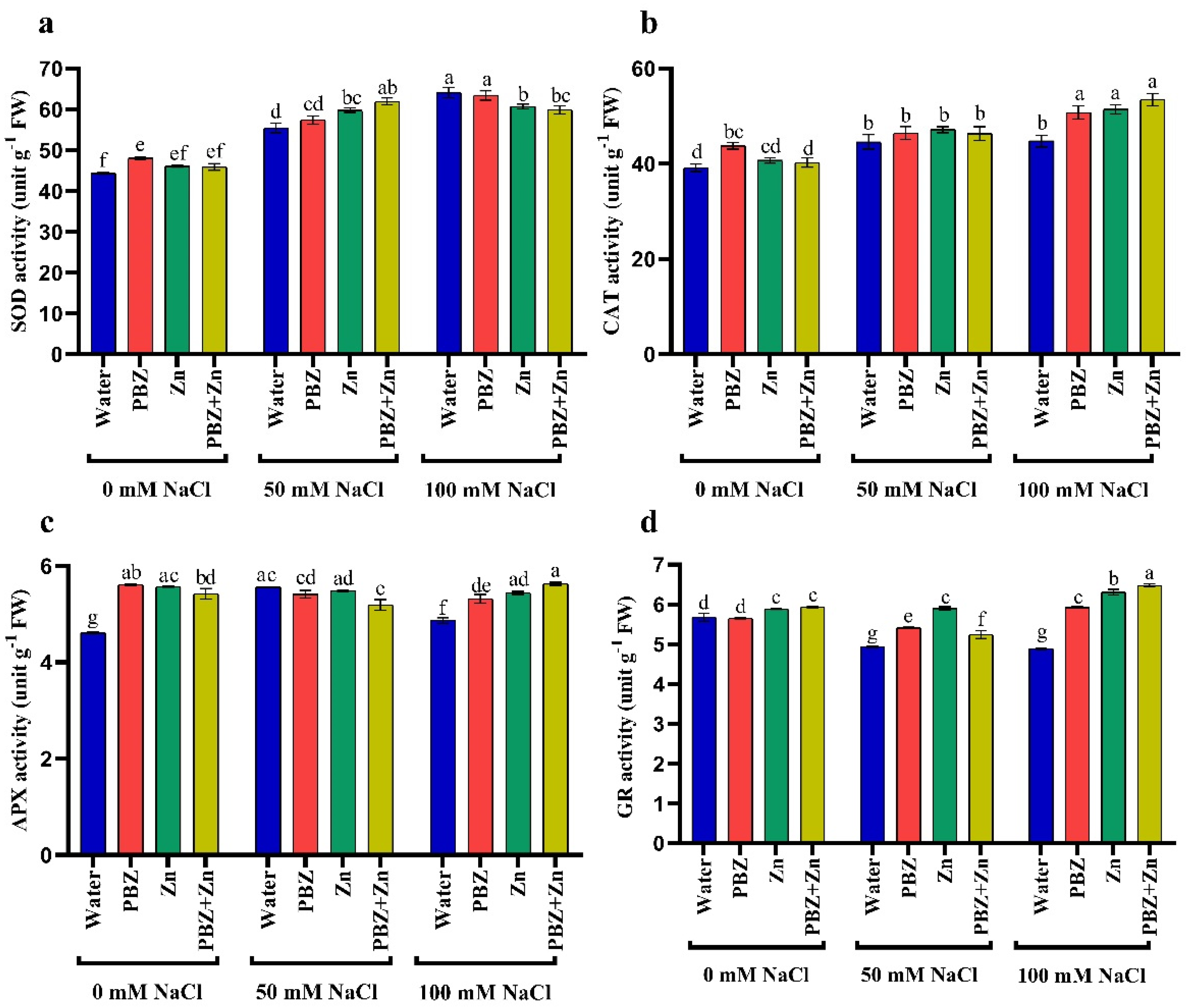
2.5. Activities of Antioxidant Enzymes
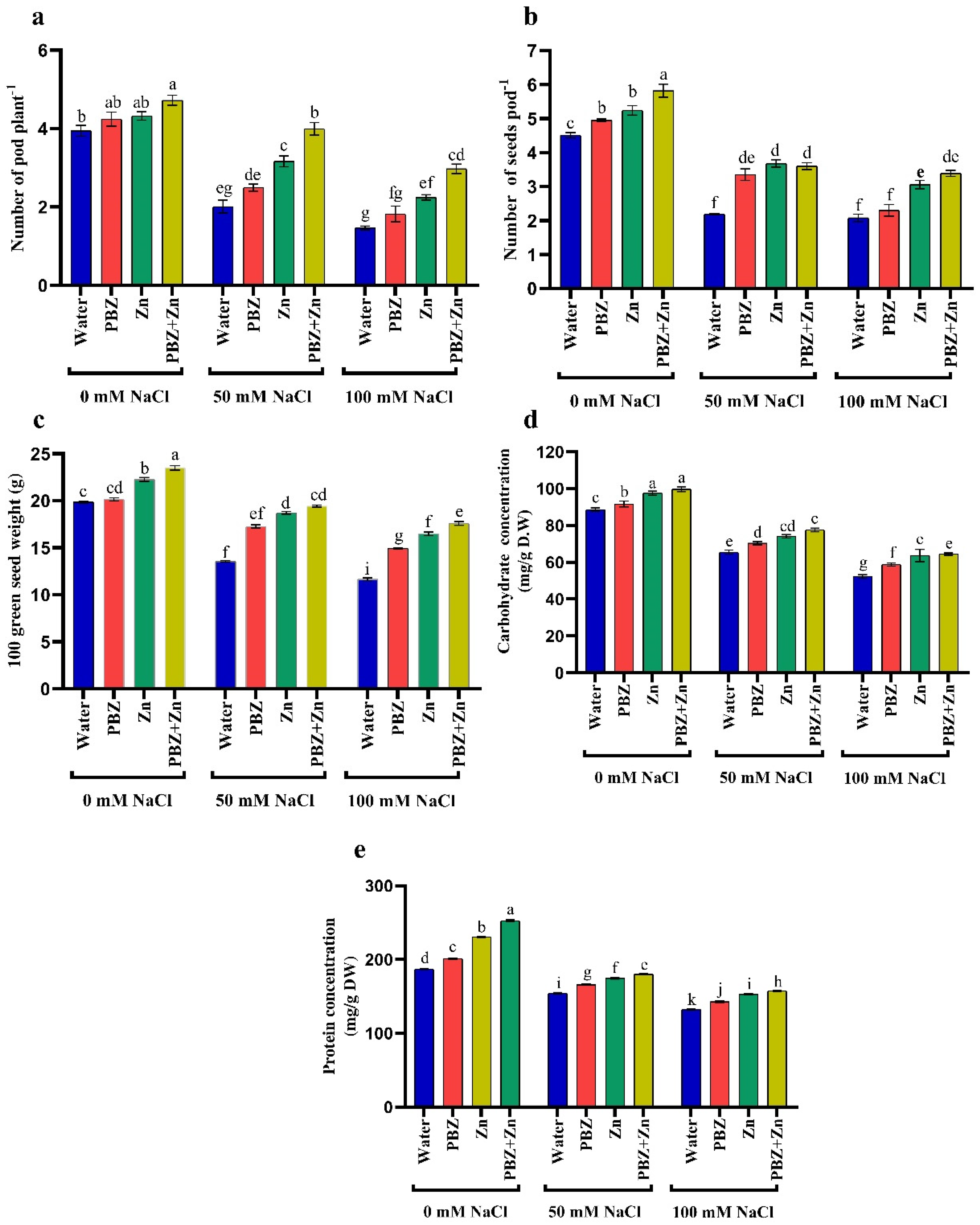
2.6. Yield Attributes
3. Discussion
4. Materials and Methods
4.1. Experimental Design
4.2. Measurement of Vegetative Attributes
4.3. Physiological and Biochemical Trials
4.4. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Cao, C.F.; Li, X.J.; Yu, L.R.; Shi, X.K.; Chen, L.M.; Yu, B.J. Foliar 2,3-dihydroporphyrin iron (III) spray confers ameliorative antioxidation, ion redistribution and seed traits of salt-stressed soybean plants. Soil Sci. Plant Nutr. 2018, 18, 1048–1064. [Google Scholar] [CrossRef]
- Gelaye, K.K.; Zehetner, F.; Loiskandl, W.; Klik, A. Comparison of growth of annual crops used for salinity bioremediation in the semi-arid irrigation area. Plant Soil Environ. 2019, 65, 165–171. [Google Scholar] [CrossRef]
- Farouk, S.; Al-Amri, S.M. Exogenous zinc forms counteract NaCl-induced damage by regulating the antioxidant system, osmotic adjustment substances, and ions in canola (Brassica napus L. cv. Pactol) plants. J. Soil Sci. Plant Nutr. 2019, 19, 887–899. [Google Scholar] [CrossRef]
- Siddiqui, M.; Alamri, S.; Al-Khaishany, M.; Khan, M.; Al-Amri, A.; Ali, H.; Alaraidh, I.; Alsahli, A. Exogenous melatonin counteracts NaCl-induced damage by regulating the antioxidant system, proline and carbohydrates metabolism in tomato seedlings. Int. J. Mol. Sci. 2019, 20, 353. [Google Scholar] [CrossRef] [PubMed]
- Sofy, M.R.; Elhawat, N.; Tarek, A. Glycine betaine counters salinity stress by maintaining high K+/Na+ ratio and antioxidant defense via limiting Na+ uptake in common bean (Phaseolus vulgaris L.). Ecotoxicol. Environ. Saf. 2020, 200, 110732. [Google Scholar] [CrossRef] [PubMed]
- Ghonaim, M.M.; Mohamed, H.I.; Omran, A.A.A. Evaluation of wheat (Triticum aestivum L.) salt stress tolerance using physiological parameters and retrotransposon-based markers. Genet. Resour. Crop Evol. 2020. [Google Scholar] [CrossRef]
- Akyol, T.; Yilmaz, O.; Uzilday, B.; Uzilday, R.; Türkan, İ. Plant response to salinity: An analysis of ROS formation, signaling, and antioxidant defense. Turk. J. Bot. 2020, 44, 1–13. [Google Scholar]
- Tang, X.; Mu, X.; Shao, H.; Wang, H.; Brestic, M. Global plant-responding mechanisms to salt stress: Physiological and molecular levels and implications in biotechnology. Crit. Rev. Biotechnol. 2015, 35, 425–437. [Google Scholar] [CrossRef]
- Nguyen, D.; Rieu, I.; Mariani, C.; van Dam, N.M. How plants handle multiple stresses: Hormonal interactions underlying responses to abiotic stress and insect herbivory. Plant Mol. Biol. 2016, 91, 727–740. [Google Scholar] [CrossRef]
- Sofy, A.R.; Dawoud, R.A.; Sofy, M.R.; Mohamed, H.I.; Hmed, A.A.; El-Dougdoug, N.K. Improving regulation of enzymatic and non-enzymatic antioxidants and stress-related gene stimulation in cucumber mosaic cucumovirus-infected cucumber plants treated with glycine betaine, chitosan and combination. Molecules 2020, 25, 2341. [Google Scholar] [CrossRef]
- Sofy, A.R.; Hmed, A.A.; Alnaggar, A.E.M.; Dawoud, R.A.; Elshaarawy, R.F.M.; Sofy, M.R. Mitigating effects of Bean yellow mosaic virus infection in faba bean using new carboxymethyl chitosan-titania nanobiocomposites. Int. J. Biol. Macromol. 2020, 163, 1261–1275. [Google Scholar] [CrossRef] [PubMed]
- Sofy, M.R.; Seleiman, M.F.; Alhammad, B.A.; Alharbi, B.M.; Mohamed, H.I. Minimizing adverse effects of Pb on maize plants by combined treatment with jasmonic, salicylic acids and proline. Agronomy 2020, 10, 699. [Google Scholar] [CrossRef]
- Abiala, M.A.; Abdelrahman, M.; Burritt, D.J.; Tran, L.P. Salt stress tolerance mechanisms and potential applications of legumes for sustainable reclamation of salt-degraded soils. Land Degrad. Dev. 2018, 29, 3812–3822. [Google Scholar] [CrossRef]
- Sofy, M.R. Effect of gibberellic acid, paclobutrazol and zinc on growth, physiological attributes and the antioxidant defense system of soybean (Glycine max) under salinity stress. Int. J. Plant Res. 2016, 6, 64–87. [Google Scholar]
- Sofy, M.R.; Sharaf, A.E.M.; Osman, M.S.; Sofy, A.R. Physiological changes, antioxidant activity, lipid peroxidation and yield characters of salt stressed barely plant in response to treatment with Sargassum extract. Int. J. Adv. Res. Biol. Sci. 2017, 4, 90–109. [Google Scholar]
- Sharaf, A.E.M.; Farghal, I.I.; Sofy, M.R. Response of broad bean and lupin plants to foliar treatment with boron and zinc. Aust. J. Basic Appl. Sci. 2009, 3, 2226–2231. [Google Scholar]
- Hassanpouraghdam, M.B.; Mehrabani, L.V.; Tzortzakis, N. Foliar application of nano-zinc and iron affects physiological attributes of Rosmarinus officinalis and quietens NaCl salinity depression. J. Soil Sci. Plant Nutr. 2019, 20, 335–345. [Google Scholar] [CrossRef]
- ÖKtem, A.G. Effects of different zinc levels on grain yield and some phenological characteristics of red lentil (Lens culinaris Medic.) under arid conditions. Turk. J. Agric. For. 2019, 43, 360–367. [Google Scholar] [CrossRef]
- Palai, J.B.; Jena, J.; Lenka, S.K. Growth, yield and nutrient uptake of maize as affected by zinc application—A review. Ind. J. Pure App. Biosci. 2020, 8, 332–339. [Google Scholar] [CrossRef]
- Tesfahun, W.; Yildiz, F. A review on: Response of crops to paclobutrazol application. Cogent Food Agric. 2018, 4, 1525169. [Google Scholar] [CrossRef]
- Kamran, M.; Ahmad, S.; Ahmad, I.; Hussain, I.; Meng, X.; Zhang, X.; Javed, T.; Ullah, M.; Ding, R.; Xu, P. Paclobutrazol application favors yield improvement of maize under semiarid regions by delaying leaf senescence and regulating photosynthetic capacity and antioxidant system during grain-filling stage. Agronomy 2020, 10, 187. [Google Scholar] [CrossRef]
- Lu, Z.X.; He, J.F.; Zhang, Y.C.; Bing, D.J. Composition, physicochemical properties of pea protein and its application in functional foods. Crit. Rev. Food Sci. Nutr. 2019, 60, 2593–2605. [Google Scholar] [CrossRef] [PubMed]
- Mahmoud, A.W.M.; Abdeldaym, E.A.; Abdelaziz, S.M.; El-Sawy, M.B.I.; Mottaleb, S.A. Synergetic effects of zinc, boron, silicon, and zeolite nanoparticles on confer tolerance in potato plants subjected to salinity. Agronomy 2019, 10, 19. [Google Scholar] [CrossRef]
- Cregg, B.; Ellison-Smith, D. Application of paclobutrazol to mitigate environmental stress of urban street trees. Forests 2020, 11, 355. [Google Scholar] [CrossRef]
- Chang, J.; Cheong, B.E.; Natera, S.; Roessner, U. Morphological and metabolic responses to salt stress of rice (Oryza sativa L.) cultivars which differ in salinity tolerance. Plant Physiol. Biochem. 2019, 144, 427–435. [Google Scholar] [CrossRef] [PubMed]
- Farouk, S.; Arafa, S.A. Mitigation of salinity stress in canola plants by sodium nitroprusside application. Span. J. Agric. Res. 2018, 16, e0802. [Google Scholar] [CrossRef]
- Arif, Y.; Singh, P.; Siddiqui, H.; Bajguz, A.; Hayat, S. Salinity induced physiological and biochemical changes in plants: An omic approach towards salt stress tolerance. Plant Physiol. Biochem. 2020, 156, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Alamer, K.; Ali, E.; Al-Thubaiti, M.; Al-Ghamdi, M. Zinc nutrition and its activated roles on growth, inflorescences attributes and some physiological parameters of Tagetes erecta L. Plants. Pak. J. Biol. Sci. 2020, 23, 35–44. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Singh, P.; Shukla, A.K.; Behera, S.K.; Tiwari, P.K. Zinc application enhances superoxide dismutase and carbonic anhydrase activities in zinc-efficient and zinc-inefficient wheat genotypes. J. Soil Sci. Plant Nutr. 2019, 19, 477–487. [Google Scholar] [CrossRef]
- Mohamed, H.I.; Akladious, S.; El-Beltagi, H. Mitigation the harmful effect of salt stress on physiological, biochemical and anatomical traits by foliar spray with trehalose on wheat cultivars. Fresenius Environ. Bull. 2018, 27, 7054–7076. [Google Scholar]
- Tahjib-UI-Arif, M.; Sohag, A.A.M.; Afrin, S.; Bashar, K.K.; Afrin, T.; Mahamud, A.S.U.; Polash, M.A.S.; Hossain, M.T.; Sohel, M.A.T.; Brestic, M. Differential response of sugar beet to long-term mild to severe salinity in a soil–pot culture. Agriculture 2019, 9, 223. [Google Scholar] [CrossRef]
- Szafrańska, K.; Reiter, R.J.; Posmyk, M.M. Melatonin improves the photosynthetic apparatus in pea leaves stressed by paraquat via chlorophyll breakdown regulation and its accelerated de novo synthesis. Front. Plant Sci. 2017, 8. [Google Scholar] [CrossRef]
- Liu, D.; Kong, D.D.; Fu, X.K.; Ali, B.; Xu, L.; Zhou, W.J. Influence of exogenous 5-aminolevulinic acid on chlorophyll synthesis and related gene expression in oilseed rape de-etiolated cotyledons under water-deficit stress. Photosynthetica 2016, 54, 468–474. [Google Scholar] [CrossRef]
- Mohamed, H.I.; Elsherbiny, E.A.; Abdelhamid, M.T. Physiological and biochemical responses of Vicia Faba plants to foliar application of zinc and iron. Gesunde Pflanz. 2016, 68, 201–212. [Google Scholar] [CrossRef]
- Akladious, S.A.; Mohamed, H.I. Physiological role of exogenous nitric oxide in improving performance, yield and some biochemical aspects of sunflower plant under zinc stress. Acta Biol. Hung. 2017, 68, 101–114. [Google Scholar] [CrossRef]
- Weisany, W.; Sohrabi, Y.; Heidari, G.; Siosemardeh, A.; Badakhshan, H. Effects of zinc Application on Growth, Absorption and Distribution of mineral nutrients under salinity stress in soybean (Glycine Max L.). J. Plant Nutr. 2014, 37, 2255–2269. [Google Scholar] [CrossRef]
- Dewi, U.; Rizkika, Z.A.; Farida, N. Effects of blue light and paclobutrazol on seed germination, vegetative growth and yield of black rice (Oryza Sativa L. ‘CempoIreng’). Biotropia 2016, 23, 85–96. [Google Scholar] [CrossRef]
- Othman, Y.; Al-Karaki, G.; Al Tawaha, A.; Al-Horani, A. Variation in germination and ion uptake in barley genotypes under salinity conditions. World J. Agric. Sci. 2006, 2, 11–15. [Google Scholar]
- Yan, W.; Yanhong, Y.; Wenyu, Y.; Taiwen, Y.; Weiguo, L.; Xiaochun, W. Responses of root growth and nitrogen transfer metabolism to uniconazole, a growth retardant, during the seedling stage of soybean under relay strip intercropping system. Commun. Soil Sci. Plant Anal. 2013, 44, 3267–3280. [Google Scholar] [CrossRef]
- Ahmad, P.; Abass, A.M.; Nasser, A.M.; Wijaya, L.; Alam, P.; Ashraf, M. Mitigation of sodium chloride toxicity in Solanum lycopersicum L. by supplementation of jasmonic acid and nitric oxide. J. Plant Interact. 2018, 13, 64–72. [Google Scholar] [CrossRef]
- Smirnoff, N. Ascorbic acid metabolism and functions: A comparison of plants and mammals. Free Radic. Biol. Med. 2018, 122, 116–129. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Sofy, M.R.; Aldaej, M.I.; Mohamed, H.I. Silicon alleviates copper toxicity in flax plants by up-regulating antioxidant defense and secondary metabolites and decreasing oxidative damage. Sustainability 2020, 12, 4732. [Google Scholar] [CrossRef]
- Maughan, S.; Foyer, C.H. Engineering and genetic approaches to modulating the glutathione network in plants. Physiol. Plant 2006, 126, 382–397. [Google Scholar] [CrossRef]
- Anjum, N.; Umar, S.; Ahmad, A.; Iqbal, M.; Khan, N. Sulphur protects mustard (Brassica campestris L.) from cadmium toxicity by improving leaf ascorbate and glutathione. Plant Growth Regul. 2007, 54, 271–279. [Google Scholar] [CrossRef]
- Foyer, C.H. Reactive oxygen species, oxidative signaling and the regulation of photosynthesis. Environ. Exp. Bot. 2018, 154, 134–142. [Google Scholar] [CrossRef]
- El-Beltagi, H.S.; Mohamed, H.I.; Sofy, M.R. Role of ascorbic acid, glutathione and proline applied as singly or in sequence combination in improving chickpea plant through physiological change and antioxidant defense under different levels of irrigation intervals. Molecules 2020, 25, 1702. [Google Scholar] [CrossRef]
- Stahl, W.; Sies, H. Antioxidant activity of carotenoids. Mol. Asp. Med. 2003, 24, 345–351. [Google Scholar] [CrossRef]
- Sofy, M.R.; Sharaf, A.M.; Fouda, H.M. Effect of foliar application of proline and zinc on growth, yield and some metabolic activities of Chenopodium quinoa Plants. Int. J. Adv. Res. 2016, 4, 1701–1717. [Google Scholar]
- Parida, A.K.; Das, A.B.; Mohanty, P. Defense potentials to NaCl in a mangrove, Bruguiera parviflora: Differential changes of isoforms of some antioxidative enzymes. J. Plant Physiol. 2004, 161, 531–542. [Google Scholar] [CrossRef]
- Jacob, O.O.; Francis, N.I. Effect of different levels of NaCl and Na2SO4 salinity on dry matter and ionic content of cowpea (Vigna unguiculata L.). Afr. J. Agric. Res. 2015, 10, 1239–1243. [Google Scholar]
- Lindskog, S. Structure and mechanism of carbonic anhydrase. Pharmacol. Ther. 1997, 74, 1–20. [Google Scholar] [CrossRef]
- Aly, A.A.; Mahmoud, T.M.; Mohamed, H.I.; Kamel, A.A. Examination of correlations between several biochemical components and powdery mildew resistance of flax cultivars. Plant Pathol. 2012, 28, 149–155. [Google Scholar] [CrossRef]
- Mohamed, H.I.; Akladious, S.A. Changes in antioxidants potential, secondary metabolites and plant hormones induced by different fungicides treatment in cotton plants. Pestic. Physiol. Biochem. 2017, 142, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar] [CrossRef]
- Motsara, M.R.; Roy, R.N. Guide to Laboratory Establishment for Plant Nutrient Analysis; Food and Agriculture Organization of the United Nations: Rome, Italy, 2008; Volume 19. [Google Scholar]
- Chaudhary, M.T.; Wainwright, S.J.; Merrett, M.J. Comparative NaCl tolerance of Lucerne plants regenerated from salt-selected suspension cultures. Plant Sci. 1996, 114, 221–232. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Deepa, N.; Kaur, C.; Singh, B.; Kapoor, H.C. Antioxidant activity in some red sweet pepper cultivars. J. Food Compos. Anal. 2006, 19, 572–578. [Google Scholar] [CrossRef]
- Guri, A.S.A.F. Variation in glutathione and ascorbic acid content among selected cultivars of Phaseolus vulgaris prior to and after exposure to ozone. Can. J. Plant Sci. 1983, 63, 733–737. [Google Scholar] [CrossRef]
- Chrysargyris, A.; Xylia, P.; Botsaris, G.; Tzortzakis, N. Antioxidant and antibacterial activities, mineral and essential oil composition of spearmint (Mentha spicata L.) affected by the potassium levels. Ind. Crop. Prod. 2017, 103, 202–212. [Google Scholar] [CrossRef]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Zhu, Z.; Wei, G.; Li, J.; Qian, Q.; Yu, J. Silicon alleviates salt stress and increases antioxidant enzymes activity in leaves of salt-stressed cucumber (Cucumis sativus L.). Plant Sci. 2004, 167, 527–533. [Google Scholar] [CrossRef]
- Foyer, C.H.; Halliwell, B. The presence of glutathione and glutathione reductase in chloroplasts: A proposed role in ascorbic acid metabolism. Planta 1976, 133, 21–25. [Google Scholar] [CrossRef] [PubMed]
- AOAC. Official Methods of Analysis of the Association of Official Analytical Chemists; AOAC: Washington, DC, USA, 2000. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sofy, M.R.; Elhindi, K.M.; Farouk, S.; Alotaibi, M.A. Zinc and Paclobutrazol Mediated Regulation of Growth, Upregulating Antioxidant Aptitude and Plant Productivity of Pea Plants under Salinity. Plants 2020, 9, 1197. https://doi.org/10.3390/plants9091197
Sofy MR, Elhindi KM, Farouk S, Alotaibi MA. Zinc and Paclobutrazol Mediated Regulation of Growth, Upregulating Antioxidant Aptitude and Plant Productivity of Pea Plants under Salinity. Plants. 2020; 9(9):1197. https://doi.org/10.3390/plants9091197
Chicago/Turabian StyleSofy, Mahmoud R., Khalid M. Elhindi, Saad Farouk, and Majed A. Alotaibi. 2020. "Zinc and Paclobutrazol Mediated Regulation of Growth, Upregulating Antioxidant Aptitude and Plant Productivity of Pea Plants under Salinity" Plants 9, no. 9: 1197. https://doi.org/10.3390/plants9091197
APA StyleSofy, M. R., Elhindi, K. M., Farouk, S., & Alotaibi, M. A. (2020). Zinc and Paclobutrazol Mediated Regulation of Growth, Upregulating Antioxidant Aptitude and Plant Productivity of Pea Plants under Salinity. Plants, 9(9), 1197. https://doi.org/10.3390/plants9091197