Abstract
During plant invasions, exotic species have to face new environmental challenges and are affected by interacting components of global change, which may include more stressful environmental conditions. We investigated an invasive species of New Zealand grasslands, commonly exposed to two concomitant and limiting abiotic factors—high levels of ultraviolet-B radiation and drought. The extent to which Verbascum thapsus may respond to these interacting stress factors via adaptive responses was assessed in a greenhouse experiment comprising native German plants and plants of exotic New Zealand origins. Plants from both origins were grown within four treatments resulting from the crossed combinations of two levels of UV-B and drought. Over twelve weeks, we recorded growth, morphological characteristics, physiological responses and productivity. The results showed that drought stress had the strongest effect on biomass, morphology and physiology. Significant effects of UV-B radiation were restricted to variables of leaf morphology and physiology. We found neither evidence for additive effects of UV-B and drought nor origin-dependent stress responses that would indicate local adaptation of native or exotic populations. We conclude that drought-resistant plant species might be predisposed to handle high UV-B levels, but emphasize the importance of setting comparable magnitudes in stress levels when testing experimentally for antagonistic interaction effects between two manipulated factors.
1. Introduction
Biological plant invasions are a key aspect of global change [1] and their mechanisms and preconditions have been frequently investigated to date [2,3,4]. A species has to overcome a number of barriers before it can be considered invasive elsewhere [5], among them biotic and abiotic conditions in the invaded range (see also [6]). Several mechanisms, including plastic and adaptive responses, which enable plant species to handle novel environmental conditions, have been repeatedly addressed. High phenotypic plasticity allows a genotype to develop different phenotypes in response to heterogeneous environments and is an often-observed advantageous property of invasive species, e.g., [7,8]. By contrast, pre-adaptation to particular environmental factors represented in single populations in the native range [9], as well as more recent adaptive evolution to novel environments following natural selection in the invaded range, can strongly contribute to a species’ invasive potential [10,11].
Addressing the role of large-scale abiotic factors as environmental barriers during plant invasions has so far mostly dealt with climatic conditions in native and invaded ranges and associated climatic niches of invasive species based on temperature and humidity tolerance [12,13]. Overall irradiation and biologically active UV-B radiation levels are equally subjected to climate change and are becoming more important for both resident plant communities and plant invasions [14]. However, these factors have been largely neglected in plant invasion research (see however: [15,16,17]). The effects of global change have largely been under consideration to date as ‘one-factor-only’ approaches. However, more recent research ambitions have identified the importance of testing for the role of interacting environmental factors [18,19,20,21].
The single effects of two environmental factors might be more or less linked so that their combined effect on plants cannot be directly extrapolated from plant response to each stress applied individually [22]. Two abiotic factors generally interact either in an additive, synergistic or antagonistic way [21,23]. A detrimental synergy of two limiting factors occurs, for instance, if the magnitude of the combined effect of both stressors exceeds the sum of the single stressor effects, as it has been observed, e.g., for jointly applied drought and heat stress on plants (see [22]). Another presumable scenario is an antagonistic interaction of stressors—a so-called cross-resistance—to both stresses by a decrease of sensitivity to one environmental factor during exposure to the other as it was previously described, e.g., in the field of biotic interactions of plants with herbivores and pathogens [24]. In natural habitats, some climatic factors are typically coupled, e.g., high solar radiation and high temperature [25]. The individual and combined contributions of these factors to plant responses can only be quantified experimentally in controlled environments.
High radiation levels and high growing season temperatures are characteristic for temperate grasslands at the global scale and they often occur in combination with low water availability. Distinctively higher levels of UV-B radiation additionally apply to the Southern hemisphere when compared to comparable sites in the Northern hemisphere where many invasive plants originate [26]. High levels of UV-B affect several plant responses more rapidly and with stronger effects in herbaceous plant species than in woody species [27]. Therefore, in particular, grassland ecosystems can be supposed to show strong responses to UV-B and should receive more attention as UV-B radiation levels continue to vary and thus to be a component of global change [28].
UV-B radiation causes interferences at different organizational levels of plants, including DNA damage, limitation of photosynthesis and morphological changes due to decreasing phytohormone concentrations (e.g., IAA [29]). Consequently, UV-B-exposed plants suffer from reductions in biomass, height and leaf area [29,30,31] and experience changes in functional leaf traits, e.g., an increasing leaf dry matter content [16]. Effective UV-B protection can be provided by strengthening epidermal or cuticular structures and trichomes on the upper leaf surface [32,33], as well as by the incorporation of UV-B absorbing flavonoids and anthocyanins [29]. These protection measures can also be advantageous in regulating plants’ water balance [34]. In their review of effects of drought stress in plants, Jaleel et al. [35] identified drought as one of the most important abiotic environmental stress factors and described several effects on plants that are similar to consequences of UV-B, including biomass reduction, decreases in plant height and leaf area and changes in dry matter content and photosynthetic pigments.
The effects of drought and high UV-B intensities on plants have been frequently investigated individually, but have been addressed in combination less often. Additive detrimental effects of UV-B radiation and drought were shown, for example, for Populus cathayana [36] in terms of plant height and leaf area reduction, such as for total biomass decrease of the shrub Hippophae rhamnoides [37]. In a soybean (Glycine max) study, there was no evidence for additive effects of both abiotic factors in growth response and seed yield [38]. However, results of other studies indicated antagonistic effects of UV-B and drought in crops [39], in European heathland species [40] and even in conifer species [41,42]. Both environmental factors provoke an oxidative burst and, thus, can jointly induce protective measures. Moreover, several studies even revealed different adaptive responses of congeneric species or distinct populations within species dependent on local conditions of their origin. Comparing high altitude and low altitude Populus species (P. kangdingensis, P. cathayana) or Hippophae rhamnoides populations, respectively, high altitude individuals exhibited higher tolerance to drought in the presence of UV-B, whereas low altitude individuals showed additive damaging effects [36,37]. Consistent with these results, Hofmann et al. [43] found higher physiological acclimation capacity of stress-adapted slow growing Trifolium repens ecotypes under high UV-B radiation compared to other populations. UV-B × drought interactions have been predominantly examined for crops and woody species, but not yet considered with regard to plant invasions into grasslands.
In the present study, we compared native (German) and invasive (New Zealand) populations of the grassland species Verbascum thapsus L. in response to combined drought stress and UV-B radiation in a greenhouse experiment (Figure 1). We tested for pre-adaptation to UV-B radiation in native populations from Germany as being induced by high drought tolerance, and for recent local adaptation of exotic populations from New Zealand in growth and physiological responses. We addressed the hypotheses that i) the combined stress of UV-B and drought has an antagonistic effect on plants and ii) New Zealand populations are better adapted to high UV-B levels providing evidence of recent adaptation. To the best of our knowledge, this is the first study addressing the role of combined environmental stress of UV-B and drought for native and exotic origins in the context of plant invasion processes in the southern hemisphere.
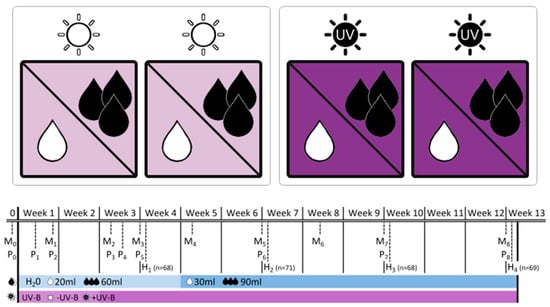
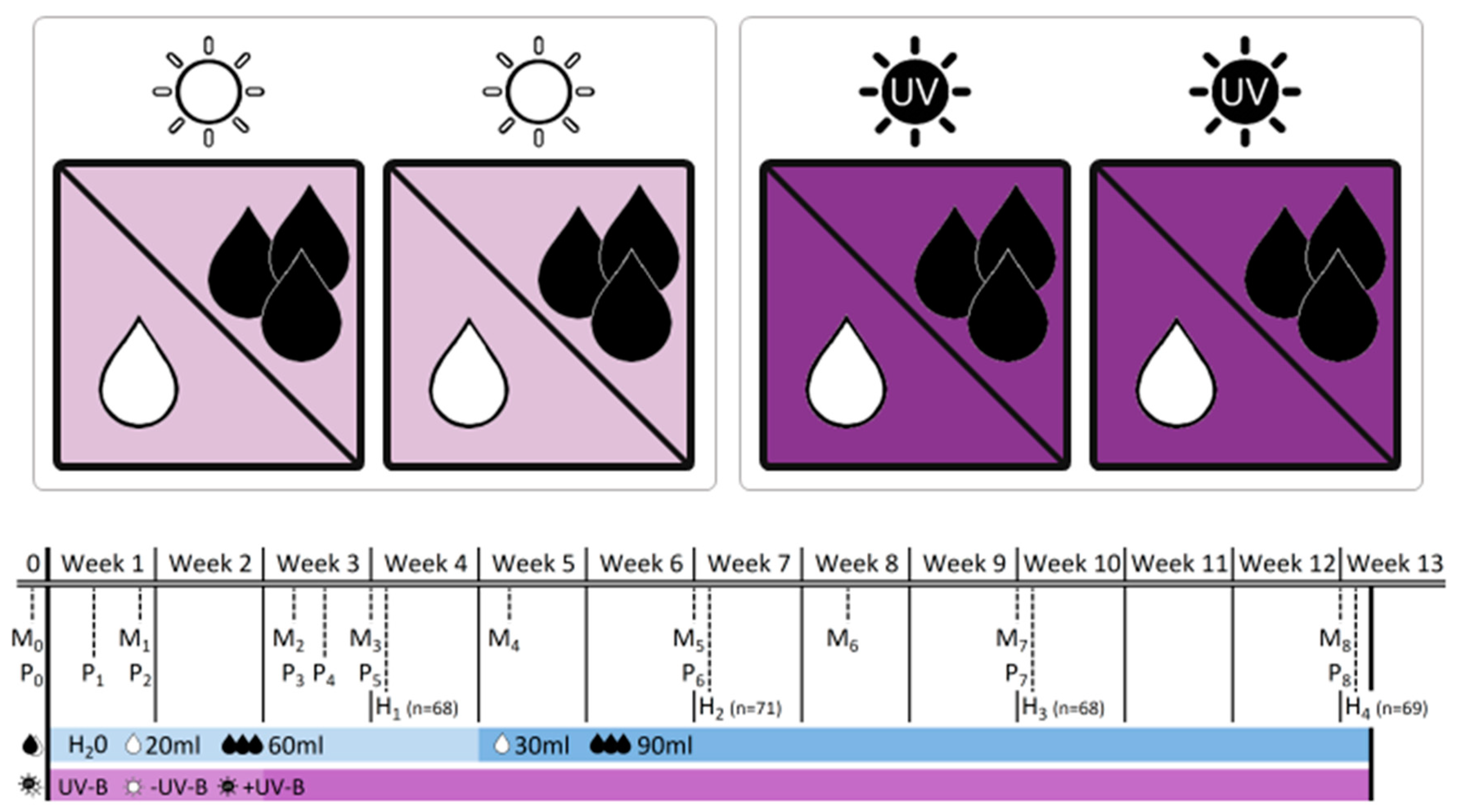
Figure 1.
Experimental design of the greenhouse experiment indicating the four experimental boxes and the crossed application of ultraviolet-B (UV-B levels:  −UV-B/
−UV-B/ +UV-B) and water treatment (water levels:
+UV-B) and water treatment (water levels:  low/
low/ well-watered). The table provides information on the dates of monitoring (Mx), physiological measurements (Px) and biomass harvests (Hx) during the 12-week experimental period, and adds information on the treatment intensity applied. Note that levels of water addition were adjusted after 4 weeks to account for rapid initial increase in plant size.
well-watered). The table provides information on the dates of monitoring (Mx), physiological measurements (Px) and biomass harvests (Hx) during the 12-week experimental period, and adds information on the treatment intensity applied. Note that levels of water addition were adjusted after 4 weeks to account for rapid initial increase in plant size.
 −UV-B/
−UV-B/ +UV-B) and water treatment (water levels:
+UV-B) and water treatment (water levels:  low/
low/ well-watered). The table provides information on the dates of monitoring (Mx), physiological measurements (Px) and biomass harvests (Hx) during the 12-week experimental period, and adds information on the treatment intensity applied. Note that levels of water addition were adjusted after 4 weeks to account for rapid initial increase in plant size.
well-watered). The table provides information on the dates of monitoring (Mx), physiological measurements (Px) and biomass harvests (Hx) during the 12-week experimental period, and adds information on the treatment intensity applied. Note that levels of water addition were adjusted after 4 weeks to account for rapid initial increase in plant size.
2. Results
2.1. Biomass Data (Harvest Data Hx)
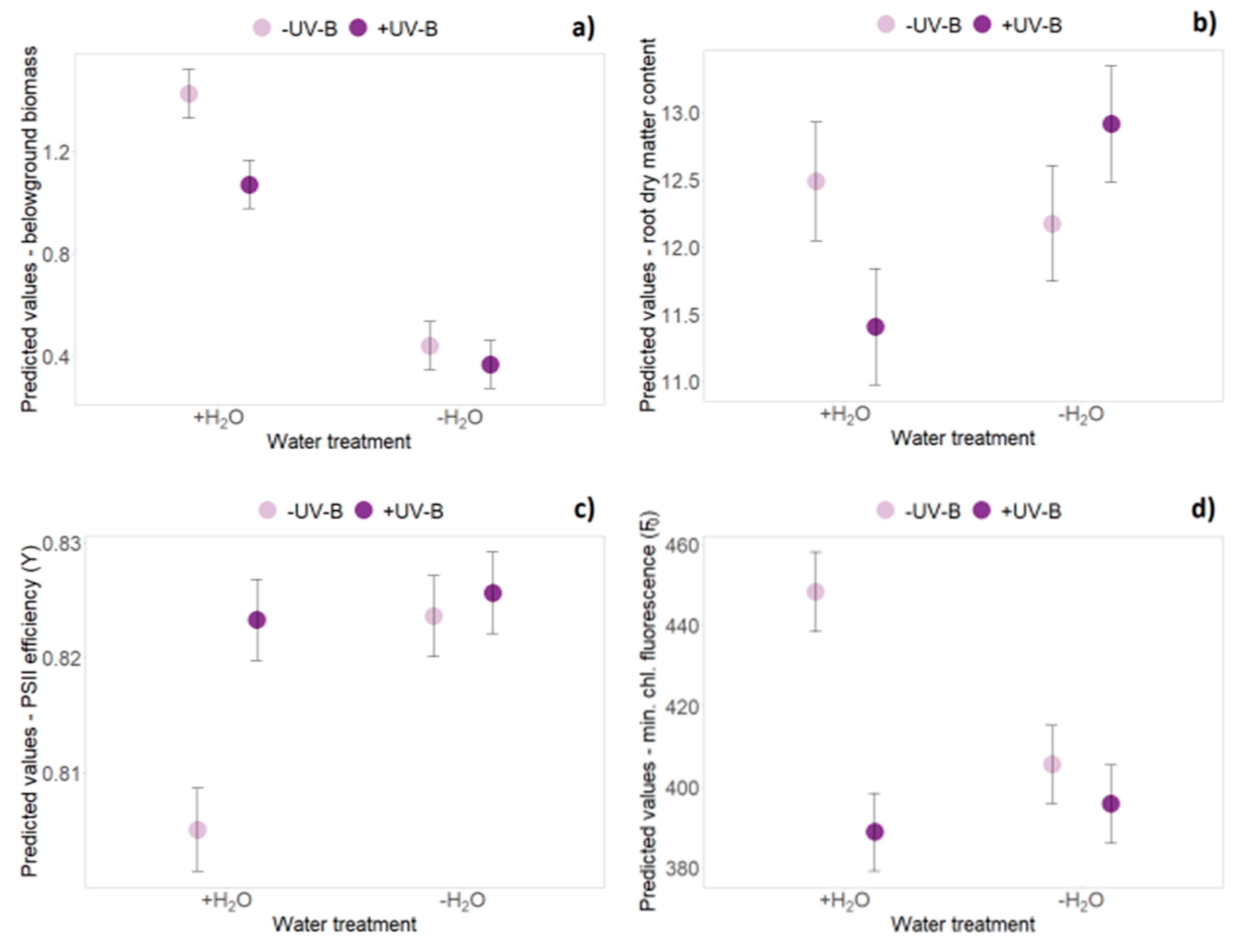
At the first harvest, aboveground, belowground and total biomasses of German individuals were significantly higher (+26% on average), than biomass of exotic individuals (p < 0.05, Table 1 and Table S1). Overall, productivity was not significantly affected by UV-B radiation at any time point. Aboveground, belowground and total biomass were significantly reduced by about 40%–50% due to limited water availability at all four harvest dates (p < 0.001, Table 1 and Table S1). By contrast, water limitation significantly increased the shoot:mass ratio among all harvest dates, as well as root dry matter content at the first and the third harvest (Table 1). At the fourth harvest, German individuals showed a more pronounced decrease in belowground and total biomass, and a consequently stronger increase in the shoot:mass ratio under limited water availability conditions compared with New Zealand individuals, as evidenced by a significant origin × water treatment interaction (p < 0.05, Table 1 and Table S1). Significant interaction effects of UV-B treatment and water treatment occurred only at the second harvest date (Table 1): when averaging over native and exotic origin, an UV-B-induced reduction of belowground biomass was less apparent under additional water limitation (p = 0.012, Figure 2a, Table S1). In well-watered conditions, a decrease of root dry matter content (−9% on average) was observed under UV-B exposure, whereas an increase of root dry matter content (+6% on average) was caused by the combined application of UV-B and limited water availability (p = 0.032, Figure 2b, Table S1).

Table 1.
Fixed-effect results of the harvest data analysis. “UV-B” and “Water” depict the effect of treatments and “Origin” refers to the effect of German vs. New Zealand provenance. Degrees of freedom (dfN = numerator, dfD = denominator), F statistics (F) and significance values (p) are provided. Significant p-values (* p < 0.05; ** p < 0.01; *** p < 0.001) and marginal effects (. p < 0.1) are indicated.
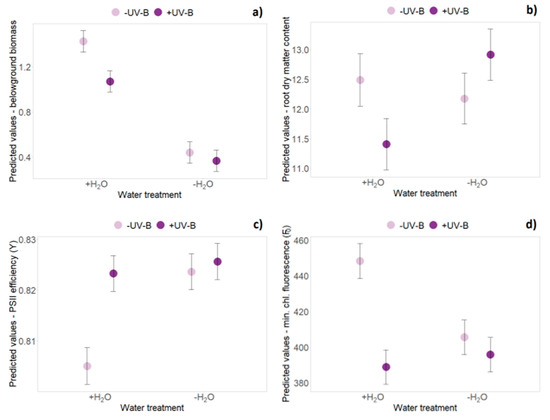
Figure 2.
Interaction effects of UV-B and water treatment when averaging over origin. Predicted values ± SE of (a) belowground biomass, (b) root dry matter content, (c) PSII efficiency (Y) and (d) minimum chlorophyll fluorescence (F0) are shown across all treatment combinations (+H2O|-UV-B, +H2O|+UV-B, -H2O|-UV-B, -H2O|+UV-B).
2.2. Growth Data (Monitoring Data)
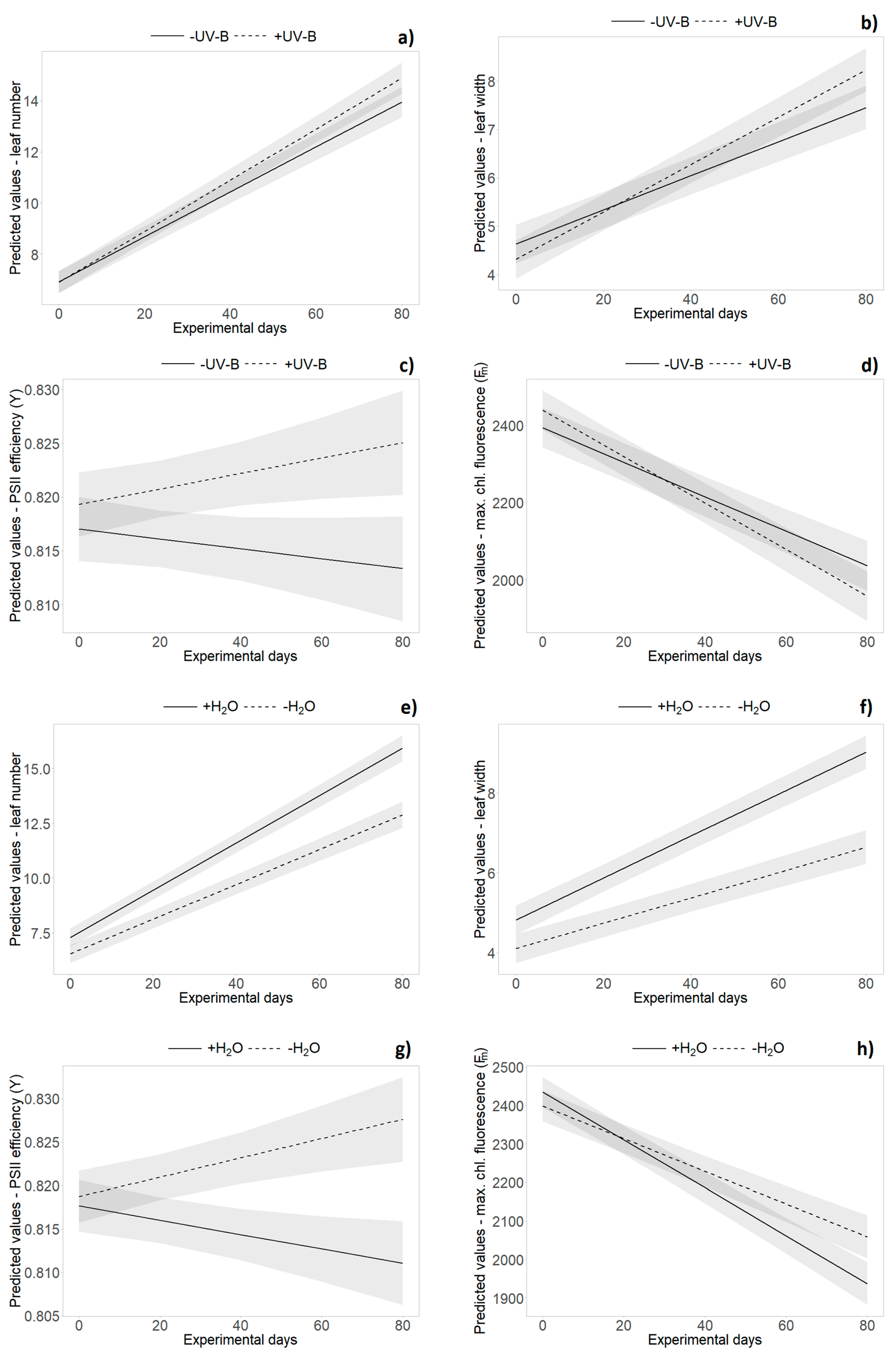
The repeated measures analysis across all monitoring dates during the experiment revealed an overall significantly higher leaf number, longer and wider leaves and bigger rosettes in German individuals compared to New Zealand plants (Table 2 and Table S2). Significant origin × time interactions revealed relatively faster leaf growth and rosette expansion of New Zealand individuals during the course of the experiment, whereas German plants were bigger in the beginning of the experiment (Table 2 and Table S2). The repeated measures analysis did not reveal significant main effects of “UV-B treatment” on any of the tested variables, but did reveal significant interaction effects with “time” for leaf number and leaf width. Leaf number and leaf width showed a stronger increase in the presence of UV-B radiation during the experiment (Table 2 and Table S2, Figure 3a,b). Single UV-B effects (e.g., reduced maximum leaf length (H1), rosette area (H1) and leaf number (H2)) or interaction effects with the origin (percentage of dead leaves (H1) and leaf number (H3)) were not consistent over time (see Tables S1 and S3).

Table 2.
Fixed-effect results of the repeated measures analysis. “UV-B” and “Water” depict the effect of treatments and “Origin” refers to the effect of German vs. New Zealand provenance. Degrees of freedom (dfN = numerator, dfD = denominator), F statistics (F) and significance values (p) are provided. Significant p-values (* p < 0.05; ** p < 0.01; *** p < 0.001) and marginal effects (. p < 0.1) are indicated.
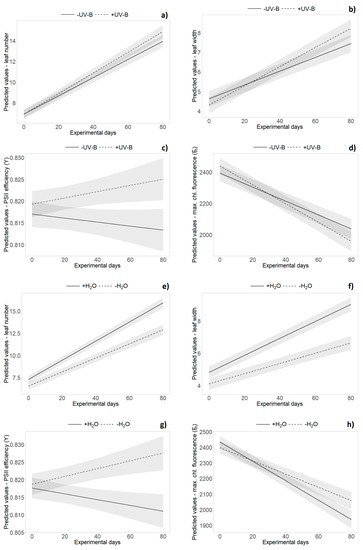
Figure 3.
Effects of UV-B (a–d) and water treatment (e–h) over time. Predicted values ± SE of (a,e) leaf number, (b,f) leaf width, (c,g) PSII efficiency (Y) and (d,h) maximum chlorophyll fluorescence (Fm) are shown in (a–d) absence of UV-B (solid line) and in presence of UV-B (dashed line), as well as under (e–h) well-watered conditions (solid line) and under drought (dashed line).
Overall, leaf number and the proportions of dead leaves, leaf length and leaf width, as well as rosette size, were significantly decreased by the water limitation treatment (p < 0.001, Table 2 and Table S2, Figure 3e,f). Water level × origin interaction effects were only found at separate monitoring dates for various variables (e.g., leaf length (H2, H4), leaf width (H3) and leaf number (H4), see Table S3). The repeated measures analysis did not reveal significant interaction effects of “UV-B treatment” and “water treatment”, nor was there a three-way interaction with “origin” or “time” (Table 2).
2.3. Functional Leaf Traits and Physiology
Significant origin effects were found at separate harvest dates and as consistent effects evidenced over time: leaves of German individuals showed an about 10% higher leaf dry matter content (LDMC) than leaves of New Zealand individuals at the first two harvests (Table 1 and Table S1). Overall, PSII efficiency (Y) was significantly higher in German individuals (p = 0.004, Table 2 and Table S2), while minimum chlorophyll fluorescence (F0) was higher in New Zealand individuals (p = 0.005, Table 2 and Table S2). Maximum chlorophyll fluorescence (Fm) decreased during the experiment with a stronger decline in German plants (p = 0.004, Table 2 and Table S2).
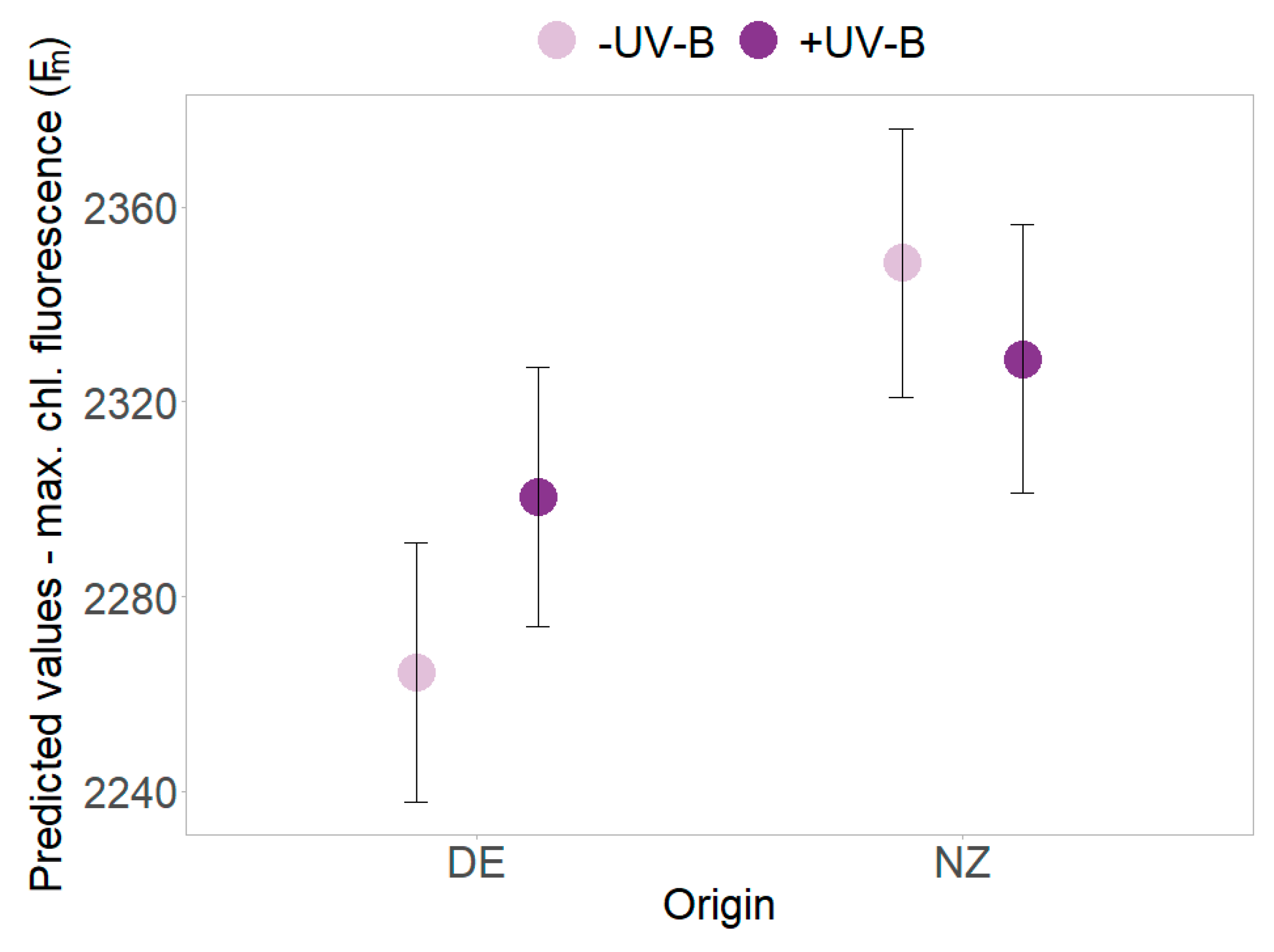
UV-B effects were found in PSII efficiency (Y) that decreased in the absence of UV-B but increased for plants under UV-B exposure (p = 0.018, Table 2 and Table S2, Figure 3c). The decrease of minimum and maximum chlorophyll fluorescence over time was more pronounced in presence of UV-B radiation (Table 2 and Table S2, Figure 3d). In presence of UV-B, overall higher maximum chlorophyll fluorescence (Fm) of New Zealand individuals (slightly) decreased, whereas an increase was found for German plants (p = 0.011, Table 2 and Table S2, Figure 4). Further UV-B × origin interaction effects were found for single harvest dates (see Tables S1 and S3).
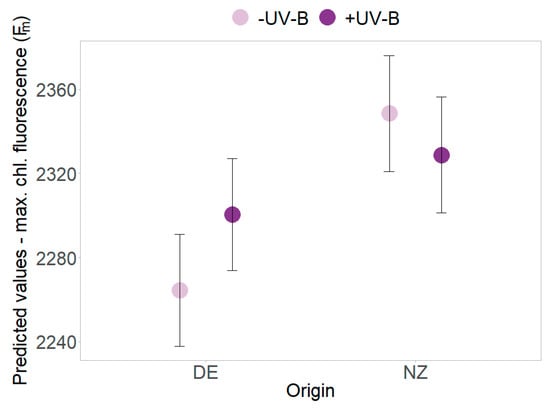
Figure 4.
Interaction effects of UV-B and origin. Predicted values ± SE of maximum chlorophyll fluorescence (Fm) are shown in the absence of UV-B (pale violet) and in the presence of UV-B (dark violet) for native German (DE) and exotic New Zealand individuals.
Water limitation significantly increased leaf dry matter content and PSII efficiency among all harvests. Overall, maximum chlorophyll fluorescence (Fm) was increased under dry conditions, whereas higher values of minimum chlorophyll fluorescence (F0) were found for well-watered plants (p < 0.05, Table 2 and Table S2). PSII efficiency (Y) decreased with sufficient water availability but increased for plants under water limitation (p < 0.001, Table 2 and Table S2, Figure 3g). The decrease of maximum chlorophyll fluorescence (Fm) over time was more pronounced in well-watered individuals (p < 0.001, Table 2 and Table S2, Figure 3h). For most of the variables, there was no significant interaction effect of origin and water, while insufficient water supply induced an increase of specific leaf area (H4) in New Zealand individuals only (see Table S3).
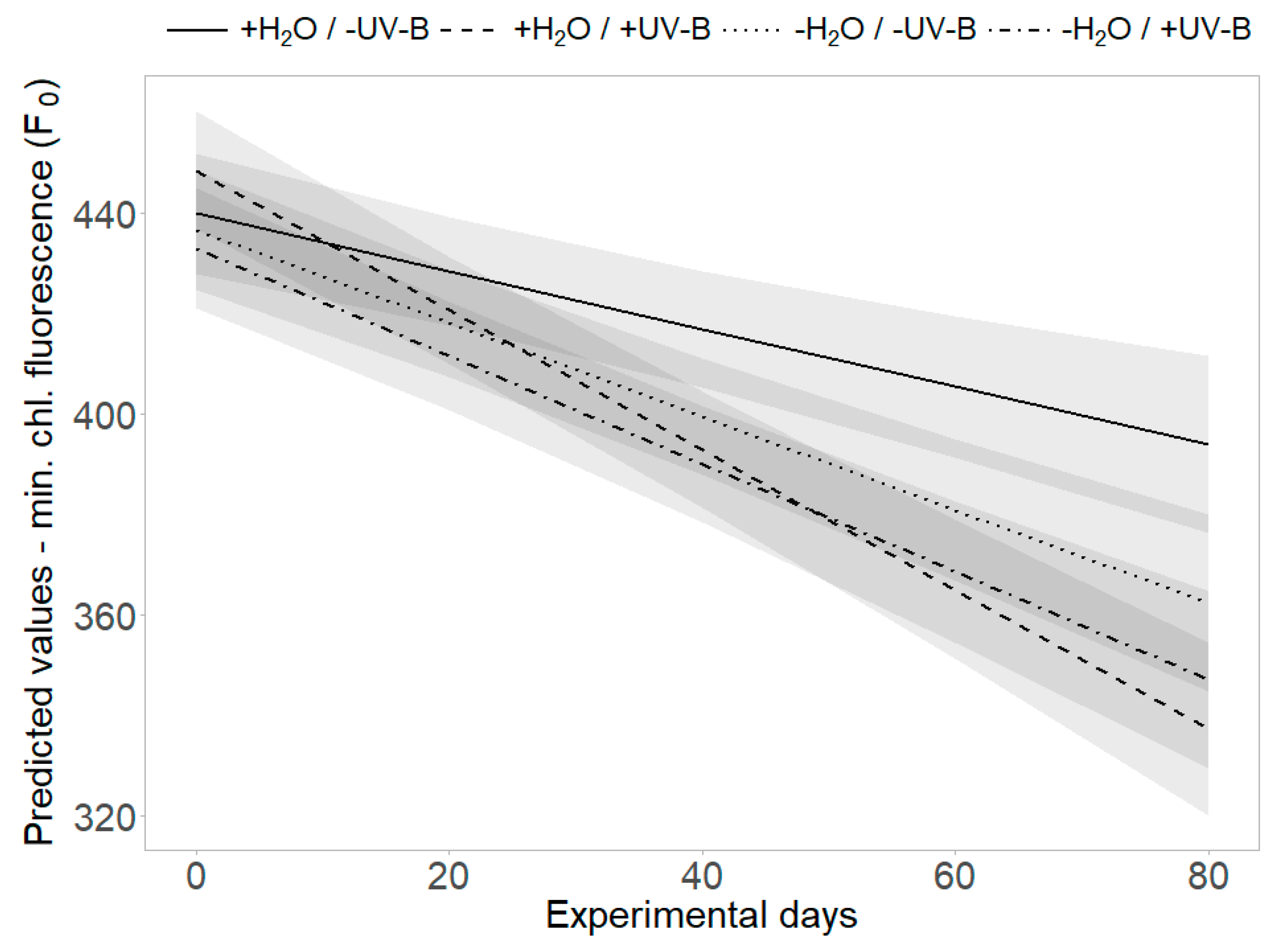
The threefold interaction effect of “UV-B treatment”, “water treatment” and “time” revealed a significant difference in minimum chlorophyll fluorescence (F0): the strongest decrease of F0 was found in the presence of UV-B radiation and the decrease was lessened by limited water availability, applied separately or in combination with UV-B radiation (p = 0.011, Table 2, Figure 5). At the second harvest, PSII efficiency (Y) was significantly increased and minimum chlorophyll fluorescence (F0) significantly decreased by UV-B radiation and low water availability individually, but this effect was not additionally enhanced by the joint presence of both factors (p < 0.01, Figure 2c,d).
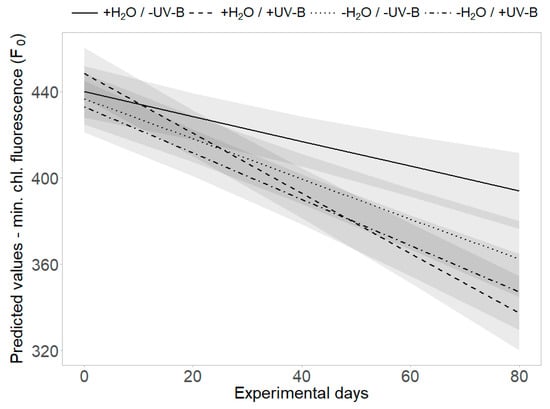
Figure 5.
Interaction effects of UV-B and water treatment. Predicted values of minimum chlorophyll fluorescence (F0) are shown across all treatment combinations (+H2O|-UV-B, +H2O|+UV-B, -H2O|-UV-B, -H2O|+UV-B). Shaded areas depict the respective SE confidence intervals.
3. Discussion
3.1. Single and Combined Effects of UV-B and Drought
The applied UV-B radiation treatment to both origins of V. thapsus aimed at simulating midsummer UV-B levels of the invaded range in New Zealand. In consequence, the applied UV-B intensity was familiar to the level exotic populations experience but novel to native individuals only. The observed limiting effects of UV-B radiation on leaf number, leaf length and rosette area confirm previous studies, which also reported UV-B-induced growth and biomass reduction [17,19,29,44]. A UV-B-induced increase in PSII efficiency was determined in the present study and caused by a decrease of minimum and maximum chlorophyll fluorescence (see also [45]). PSII efficiency response to UV-B has been previously identified for several species and was mostly found to decrease as a result of increasing minimum and maximum chlorophyll fluorescence [30,33,46] (but see also [47]). Especially an increase of minimum chlorophyll indicates photoinhibition and direct damage or an inactivation of PSII reaction centers as a result of a disconnection of light-harvesting antennae from their reaction centers [48,49,50]. A concomitant decrease of maximum and minimum chlorophyll fluorescence, as observed in the present study, was previously linked to thermal dissipation in PSII reaction centers, which displays a key photoprotective process [48]. In consequence, reactive oxygen species (ROS) production in response to a moderate dose of UV-B radiation in our experiment might be avoided and might even explain a temporary increase of PSII efficiency in the context of efficient repair mechanisms.
The applied water treatment presumably induced drought stress in the individuals of the “low” water treatment level, as those regularly responded with wilting of leaves at the latest on the day of watering. We therefore assume that a physiological stress response was provoked in the plants by water limitation. An overall reduced biomass, leaf number, leaf area and higher leaf dry matter content confirm this assumption and have been previously observed in response to water limitation (e.g., [35,44,51]). In line with the effects of UV-B, the maximum photosynthetic quantum yield of all plants was higher under drought conditions. This effect was initially caused by a decrease of minimum chlorophyll fluorescence, and in the later experimental phase due to an increase of maximum chlorophyll fluorescence. Previous studies on drought effects predominantly revealed decreasing PSII efficiency due to water limitation [52,53]. The opposite effect in the present study might be the consequence of the existing drought tolerance and resulting protection measures of Verbascum thapsus, that are known to naturally occur on very dry and disturbed sites [54].
We found no evidence for detrimental synergy effects or generally additive effects of drought and UV-B radiation as they had been reported before [36,37]. Other studies found enhanced drought tolerance in the presence of UV-B radiation, since certain stress avoidance mechanisms turned out to be of advantage under both abiotic stresses, e.g., leaf area reduction, increase of leaf cuticle thickness or stomatal closure [38,40,55]. Relevant antagonistic effects can be also provoked at the physiological level by common metabolic responses to drought and UV-B, e.g., an increase of anthocyanins, phenolics, prolin and other antioxidants to decrease ROS production and consequently maintain photosynthetic capacity and carbon assimilation rate [39,41,42,43].
In the present study, the only significant interaction effects of UV-B and drought were found after six weeks of the experiment (H2) and might have been the temporary consequence of the UV-B radiation dose increase after the initial two-week UV-B acclimation phase. Belowground biomass moderately decreased in response to UV-B radiation [16,56] but was highly sensitive to drought with a strong overall decrease [44] that was not additionally aggravated by supplementary UV-B exposure. By contrast, root dry matter content did not change significantly with drought in the absence of UV-B, but increased under combined stress application and decreased under UV-B exposure in well-watered conditions, thus displaying strong interaction effects. Dry matter distribution towards the roots has been previously shown for plant species under abiotic stress conditions [57,58], and might be only induced in response to the combined application of drought and UV-B in our study. Furthermore, the PSII efficiency was increased by UV-B and drought to a similar extent, whether applied separately or jointly. This points to a similar and non-additive effect size of both abiotic stresses at the physiological level. Interestingly, the minimum chlorophyll fluorescence (F0) appeared to be more sensitive in response to UV-B radiation at the second harvest (H2) and in the repeated measures analysis. The only explanation for a decrease of F0 might be a higher number of unimpaired PSII reaction centers that could be provided by activation of efficient photoprotection and repair mechanisms. Those might be induced to a higher level by UV-B compared to drought, as radiation displays the more immediate trigger for PSII damage. Therefore, we could conclude that the plant physiology of V. thapsus is affected by both UV-B and drought to a similar extent, but the respective effect is induced by different underlying mechanisms.
3.2. Origin Differentiation and Origin-Specific Response to UV-B and Drought
Differences in plant performance or functional plant traits between native and exotic origins might hint at genetic differentiation as a result of founder effects or evolutionary processes during the invasion of novel habitats in New Zealand [59]. As the set of investigated German and New Zealand populations does not represent the entire native and invaded range, respectively, other sources of variation among populations may have also contributed to significant differences between origins: among them are population-specific differences in elevation, microclimate or other environmental factors that are able to induce geographical clines within ranges [54,60]. Thus, complementary experiments with further seed material from other parts of the native and invaded range along larger latitudinal gradients would be necessary to draw more general conclusions.
In the present study, native individuals from Germany started with higher biomass and larger leaves, but exotic individuals showed stronger increase in time regarding leaf area and respective size of rosettes. Therefore, the initial advantage of native plants disappeared during the experimental runtime. Higher productivity/relative growth rates of exotic populations in comparison to native origins has been repeatedly reported in the past, especially in the context of altered resource allocation as a result of the release from native biotic and abiotic stresses [2,54,61]. Nevertheless, in our study, the New Zealand populations of V. thapsus appeared to be less successful in the early establishment (personal observation). The difference in seed age of the German and New Zealand populations and the conditions during seed transfer from New Zealand to Germany might be a reason for differences in germination and establishment success. Further explanations for the initial disadvantage of exotic populations may comprise a potentially reduced genetic diversity of exotic populations due to founder effects or the importance of range identity with regard to covarying effects of different biotic and abiotic conditions, as discussed by Dieskau et al. [62]. This might be particularly important, since other native–invasive comparisons revealed early invasive superiority when testing V. thapsus performance in other parts of the species’ exotic range [54,63,64]. PSII efficiency was found to be generally higher in native plants with a stronger decrease of maximum chlorophyll fluorescence (Fm) in German individuals during the experiment and higher values of minimum chlorophyll fluorescence (F0) in New Zealand plants since the second harvest (H2) independent of water availability and UV-B treatment. Higher physiological performance of native individuals could also be a consequence of their early establishment success prior to the application of environmental stress by drought and UV-B radiation, which might have led to stronger and more resilient plants.
In the presence of UV-B, New Zealand plants showed the described reduction of maximum chlorophyll fluorescence, whereas an observed increase of maximum chlorophyll fluorescence in German individuals might be linked to an impaired electron transfer or secondary electron acceptor of PSII [49]. In contrast to German plants, New Zealand individuals also showed a pronounced decrease of minimum chlorophyll fluorescence and an increase of leaf number and dead leaf proportion in the early stage of the experiment. Therefore, the higher photoprotection abilities and growth of exotic individuals under UV-B radiation might indicate an evolved reduced sensitivity to UV-B in consequence to the experienced higher radiation levels in New Zealand.
Drought stress is known to limit invasibility of habitats, as drier sites appear to be less invaded and non-natives turned out to be more abundant in wetter years [65]. Nevertheless, previous studies did not agree on the question if drought tolerance of native and non-native species differs and thus is subject to evolutionary changes in plant invasions [66,67,68]. In the present study, non-native plants from New Zealand responded with measurable changes in leaf morphology to low water availability, whereas native German plants experienced a stronger decrease in growth estimates in the late experimental phase. We could therefore assume that non-native genotypes are able to respond with functional changes at the leaf level in order to sustain overall growth under drought conditions. This ability might be the result of evolutionary processes in response to environmental conditions in the invaded range or overall higher phenotypic plasticity [69]. Interestingly, previous studies on drought tolerance of native and non-native populations of grassland species assumed a trade-off between rapid growth and drought tolerance, since they revealed more resilient native populations under drought conditions, although non-native populations appeared to be more vigorous and fast-growing in other environments [61,70].
Furthermore, we found no evidence for the importance of population origin to the combined stress effects on plants. Previous studies on different woody plant species revealed different resistance of low and high altitude populations to a treatment combining UV-B radiation and drought [36,37]: whereas low altitude populations experienced additive detrimental effects of both abiotic stressors on productivity and growth traits, high altitude populations responded with higher tolerance to the combined application of drought and UV-B, and thus appeared to be better adapted. By contrast, testing for adaptation to elevational constraints in multiple exotic plant species gradient, Watermann et al. [31] did not find any evidence for combined UV-B × drought interactions with low and high altitude populations. However, neither native nor non-native populations of Verbascum thapsus had an advantage in the presence of combined abiotic stress by drought and UV-B radiation in the present experiment. As both origins are expected to be similarly adapted to drought but experience different levels of UV-B in their home ranges, we assume that origin differences in stress response may be more precisely carved out by moderate water limitation and moderate or elevated UV-B intensity. While severe levels of stress usually lead to direct negative effects on plant metabolism and growth, moderate abiotic stress triggers physiological and biochemical defense mechanisms, which are of advantage under harmful conditions [44].
4. Materials and Methods
4.1. Study Species
Verbascum thapsus L. (Scrophulariaceae) is a typical component of temperate dry grasslands and ruderal habitats in the investigated ranges in Germany and New Zealand, and is characterized by high drought tolerance and a strong prevalence in open, unshaded habitats [71]. The species is monocarpic, generally biennial and develops a long tap root to access remote nutrient and water resources in deeper soil layers [72]. The plant’s surface is typically piliferous, i.e., leafs and stems are densely covered with woolly, branched stellate trichomes, which provide a reliable protection against herbivory, frost and drought [73] and may be also advantageous under high radiation levels. The native distribution of V. thapsus ranges from Europe to Central Asia. To date, it is furthermore naturalized in North America, Hawaii, Australia and New Zealand. In the present study, we used ten native and eight invasive populations of V. thapsus from Germany and New Zealand, respectively (for population details see Table S4).
4.2. Experimental Design
The experiment was conducted in the summer of 2013 in the greenhouse cabinets of the Martin Luther University Halle–Wittenberg. Seeds were germinated in the greenhouse under standard conditions within seedling trays on a soil-sand mixture (2:1) and transferred into pots (9 × 9 × 10 cm) with the same substrate about six weeks later. At the age of ten weeks, plants were assigned to the experimental setting: four treatments resulting from fully crossed combinations of two water levels (“low” vs. “well-watered”) and two UV-B levels (“−UV-B” vs. “+UV-B”) were applied to four individuals of all 18 populations (totaling 288 individuals). Therefore, plants were arranged within four identical boxes (120 × 120 × 70 cm), which served as self-contained UV-B environments (Figure 1). All boxes were equipped with white chipboard to the left and the right side and with white fleece at the front and the back, allowing the implementation of a UV-B radiation source from the top to the plants and to ensure ventilation within the boxes to minimize uncontrolled microclimatic effects. Each of the four boxes was equipped with a greenhouse PAR lamp (HQI 400 W, Philips) on the top. Additionally, in two boxes, three UV-B tubes (TL 20W/12 RS SLV, Philips) were implemented. The two boxes without UV-B tubes served as a “no UV-B” control. Photosynthetically active radiation (PAR) was applied 16 h a day, whereas UV-B lamps were switched on for eight hours within this period. Initially, UV-B lamps had a distance of 80 cm to the plant individuals, resulting in a UV-B intensity of 0.014-0.052 mW cm−2 dependent on pot position. After two weeks we reduced the distance between the lamps and the plants in order to increase UV-B radiation to 0.096–0.159 mW cm−2, thereby approaching the midsummer UV-B level on the South Island, New Zealand [17].
Half of the plants in each box received sufficient water supply, whereas the other half was exposed to drought (Figure 1). Both treatment groups were watered every second day with water amounts in a ratio of 3:1 (week 1–4: 60 mL/20 mL, week 5–12: 90 mL/30 mL, for well-watered and drought treatments, respectively). Measurements of soil moisture using a time-domain reflectometer (TDR) revealed water contents of 15%–20% in well-watered pots and 3%–8% in pots mimicking situations of drought. Based on a visual assessment, the latter group generally reached the wilting point within 48 h but was kept from being permanently damaged.
Individuals of each population were equally represented at each UV-B level and water level. Initially, all individuals were randomly assigned to and positioned within the boxes, but within-box randomization was subsequently repeated every 7–10 days during the experiment. Due to the occurrence of some mortality in the early phase of the study, we received data of 276 individuals of 18 populations (10 DE, 8 NZ) within an experimental period of 12 weeks.
4.3. Data Collection
Biometrical variables were determined for each individual on a weekly basis in the beginning, and later every ten days (Figure 1): rosette diameter, length and width of the longest leaf and the number of intact and dead leaves were recorded ten times during the experiment. Rosette area (A) was calculated for each individual as an ellipse using measured rosette diameters (d1, d2):
A = π × (d1/2) × (d2/2),
In order to assess repeated productivity data and growth rates, one individual per population and treatment was harvested every three weeks, resulting in four harvests during the experimental period (n = 68–71, Figure 1). We determined aboveground, belowground and dead biomass, leaf dry matter content (LDMC), root dry matter content (RDMC), specific leaf area (SLA) and the shoot:root ratio for each individual harvested in the different subsets. The selective sampling for harvest reduced the total amount of individuals available for monitoring of biometrical variables over time.
At the physiological level, we recorded maximum quantum yield of photochemical energy conversion (Y) as a measure of photosystem II efficiency, such as minimum and maximum fluorescence yield (F0, Fm) in response to the initial UV-B application and the enhancement of UV-B intensity after a two-week acclimatization, totaling eight times during the experiment (Figure 1). Therefore, one fully developed and healthy leaf per individual was dark-adapted for about ten minutes and subsequently measured once with a hand-held fluorometer (Mini-PAM, Heinz Walz GmbH) without removal from the experimental boxes or interruption of the UV-B treatment.
4.4. Statistical Analysis
A repeated measures analysis of the monitoring data was done to test for the effects of origin, UV-B treatment and water treatment on plant performance over time in R (Version 3.5.3, R CORE TEAM 2019). We therefore applied a linear mixed effect model (function “lmer”, package “lmerTest”, [74]) containing “origin” (DE vs. NZ), “UV-B” (-UV-B vs. +UV-B) and “water” (low vs. well-watered) as fixed factors and “time” (eight dates of monitoring and physiological measurements), as well as all of their interactions. The following nested random effect terms were additionally included in the repeated measures analysis: “box:UV-B” and “population:origin”, while fitting a random slope model with “time|plant ID:UV-B:water:origin”. Due to the partial harvests during the experiment, the repeated measures analysis of the monitoring data naturally experienced a decrease in sample size over time. Thereby, the number of replicates within populations was reduced from four to one during the experimental duration. As the statistical analyses aim to test for differences between origins (DE vs. NZ), all remaining individuals of the ten German populations served as replicates for the origin level “DE” and all remaining individuals of the eight New Zealand populations were considered replicates for the origin level “NZ”.
Furthermore, data of the four partial biomass harvests was separately analyzed per date by linear mixed effect models containing “origin” (DE vs. NZ), “UV-B” (-UV-B vs. +UV-B) and “water” (low vs. well-watered), as well as their interactions as fixed factors. We additionally included the individual leaf number at the time of the experimental start as a covariate and again determined “box:UV-B” and “population:origin” as nested random effect terms in the separate mixed effects models.
5. Conclusions
Generally, interaction effects of UV-B and drought depend on species-specific sensitivity, stress factor intensity, exposure duration and operation mode [44]. In our study, the strong effect of water treatment might potentially mask UV-B effects on plants, as the induced water limitation level is likely to display a more restrictive condition for plant metabolism than the applied UV-B radiation level. This observation points at the importance of setting comparable stress levels in abiotic interaction experiments, as otherwise one of the abiotic environmental factors dominates the results and potential antagonistic effects are difficult to detect. Moreover, application of artificial UV-B under greenhouse conditions partly excludes photosynthetically active radiation (PAR) that is known to have a mitigating effect on plants under UV-B and might have additionally induced mechanisms of drought resistance [44,75]. Nevertheless, our results point at similar physiological responses to drought and UV-B radiation and an absence of detrimental synergy effects of both environmental factors. Therefore, we assume that drought-tolerant plant species might also be more resilient to higher levels of UV-B radiation. To adequately test and identify cross-resistance mechanisms in plant invasions and the potential impact of local adaptation on this characteristic, we recommend attaching great importance to the application of suitable and relevant environmental stress gradients derived from respective native and/or invaded ranges in future experimental studies.
Supplementary Materials
The following are available online at https://www.mdpi.com/2223-7747/9/2/269/s1, Table S1: Effect directions of harvest data analysis; Table S2: Effect directions of repeated measures analysis; Table S3: Fixed-effect results of the harvest data analysis; Table S4: Location and sampling information of native (German) and exotic (New Zealand) populations included in the experiment.
Author Contributions
Conceptualization, M.H. and A.E.; Data curation, C.P. and M.S.; Formal analysis, M.H.; Investigation, C.P. and M.S.; Methodology, M.H. and A.E.; Project administration, M.H.; Supervision, A.E.; Writing—original draft, M.H.; Writing—review and editing, C.P., M.S. and A.E. All authors have read and agreed to the published version of the manuscript.
Funding
We acknowledge financial support by Land Schleswig-Holstein within the funding programme Open Access Publikationsfonds.
Acknowledgments
We thank Susanne Both and Julia Dieskau for seed sampling of New Zealand and German Verbascum thapsus populations. Additional seeds from the Botanical Garden of Martin Luther University Halle-Wittenberg were provided by E. Grimm. We thank three reviewers for their valuable comments.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Pereira, M.G.; Calado, T.J.; DaCamara, C.C.; Calheiros, T. Effects of regional climate change on rural fires in Portugal. Clim. Res. 2013, 57, 187–200. [Google Scholar] [CrossRef]
- Keane, R.M.; Crawley, M.J. Exotic plant invasions and the enemy release hypothesis. Trends Ecol. Evol. 2002, 17, 164–170. [Google Scholar] [CrossRef]
- Theoharides, K.A.; Dukes, J.S. Plant invasion across space and time: Factors affecting nonindigenous species success during four stages of invasion. New Phytol. 2007, 176, 256–273. [Google Scholar] [CrossRef] [PubMed]
- Erfmeier, A. Constraints and release at different scales—The role of adaptation in biological invasions. Basic Appl. Ecol. 2013, 14, 281–288. [Google Scholar] [CrossRef]
- Blackburn, T.M.; Pyšek, P.; Bacher, S.; Carlton, J.T.; Duncan, R.P.; Jarošík, V.; Wilson, J.R.U.; Richardson, D.M. A proposed unified framework for biological invasions. Trends Ecol. Evol. 2011, 26, 333–339. [Google Scholar] [CrossRef]
- Richardson, D.M.; Pyšek, P.; Rejmánek, M.; Barbour, M.G.; Panetta, F.D.; West, C.J. Naturalization and invasion of alien plants: Concepts and definitions. Divers. Distrib. 2000, 6, 93–107. [Google Scholar] [CrossRef]
- Sharma, G.P.; Esler, K.J. Phenotypic plasticity among Echium plantagineum populations in different habitats of Western Cape, South Africa. S. Afr. J. Bot. 2008, 74, 746–749. [Google Scholar] [CrossRef]
- Davidson, A.M.; Jennions, M.; Nicotra, A.B. Do invasive species show higher phenotypic plasticity than native species and, if so, is it adaptive? A meta-analysis. Ecol. Lett. 2011, 14, 419–431. [Google Scholar] [CrossRef]
- Schlaepfer, D.R.; Glättli, M.; Fischer, M.; van Kleunen, M. A multi-species experiment in their native range indicates pre-adaptation of invasive alien plant species. New Phytol. 2010, 185, 1087–1099. [Google Scholar] [CrossRef]
- Colautti, R.I.; Eckert, C.G.; Barrett, S.C.H. Evolutionary constraints on adaptive evolution during range expansion in an invasive plant. Proc. R. Soc. B Biol. Sci. 2010, 277, 1799–1806. [Google Scholar] [CrossRef]
- Buswell, J.M.; Moles, A.T.; Hartley, S. Is rapid evolution common in introduced plant species? J. Ecol. 2011, 99, 214–224. [Google Scholar] [CrossRef]
- Broennimann, O.; Treier, U.A.; Müller-Schärer, H.; Thuiller, W.; Peterson, A.T.; Guisan, A. Evidence of climatic niche shift during biological invasion. Ecol. Lett. 2007, 10, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Petitpierre, B.; Kueffer, C.; Broennimann, O.; Randin, C.; Daehler, C.; Guisan, A. Climatic niche shifts are rare among terrestrial plant invaders. Science 2012, 335, 1344–1348. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, G.K.; Gwynn-Jones, D.; Callaghan, T.V.; Sleep, D.; Lee, J.A. Effects of global change on a sub-Arctic heath: Effects of enhanced UV-B radiation and increased summer precipitation. J. Ecol. 2001, 89, 256–267. [Google Scholar] [CrossRef]
- Qaderi, M.M.; Yeung, E.C.; Reid, D.M. Growth and physiological responses of an invasive alien species, Silene noctiflora, during two developmental stages to four levels of ultraviolet-B radiation. Ecoscience 2008, 15, 150–159. [Google Scholar] [CrossRef]
- Beckmann, M.; Hock, M.; Bruelheide, H.; Erfmeier, A. The role of UV-B radiation in the invasion of Hieracium pilosella—A comparison of German and New Zealand plants. Environ. Exp. Bot. 2012, 75, 173–180. [Google Scholar] [CrossRef]
- Hock, M.; Hofmann, R.W.; Müller, C.; Erfmeier, A. Exotic plant species are locally adapted but not to high ultraviolet-B radiation: A reciprocal multispecies experiment. Ecology 2019, 100, e02665. [Google Scholar] [CrossRef]
- Norby, R.J.; Luo, Y. Evaluating ecosystem responses to rising atmospheric CO2 and global warming in a multi-factor world. New Phytol. 2004, 162, 281–293. [Google Scholar] [CrossRef]
- Caldwell, M.M.; Bornman, J.F.; Ballaré, C.L.; Flint, S.D.; Kulandaivelu, G. Terrestrial ecosystems, increased solar ultraviolet radiation, and interactions with other climate change factors. Photochem. Photobiol. Sci. 2007, 6, 252–266. [Google Scholar] [CrossRef]
- Alba, C.; Fahey, C.; Flory, S.L. Global change stressors alter resources and shift plant interactions from facilitation to competition over time. Ecology 2019, 100, e02859. [Google Scholar] [CrossRef]
- Côté, I.M.; Darling, E.S.; Brown, C.J. Interactions among ecosystem stressors and their importance in conservation. Proc. R. Soc. B Biol. Sci. 2016, 283, 20152592. [Google Scholar] [CrossRef] [PubMed]
- Mittler, R. Abiotic stress, the field environment and stress combination. Trends Plant Sci. 2006, 11, 15–19. [Google Scholar] [CrossRef] [PubMed]
- Galic, N.; Sullivan, L.L.; Grimm, V.; Forbes, V.E. When things don’t add up: Quantifying impacts of multiple stressors from individual metabolism to ecosystem processing. Ecol. Lett. 2018, 21, 568–577. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, C.M.; Dicke, M. Plant interactions with microbes and insects: From molecular mechanisms to ecology. Trends Plant Sci. 2007, 12, 564–569. [Google Scholar] [CrossRef]
- Mauro, R.P.; Monaco, A.L.; Lombardo, S.; Restuccia, A.; Mauromicale, G. Eradication of Orobanche/Phelipanche spp. seedbank by soil solarization and organic supplementation. Sci. Hortic. 2015, 193, 62–68. [Google Scholar] [CrossRef]
- McKenzie, R.L.; Aucamp, P.J.; Bais, A.F.; Björn, L.O.; Ilyas, M. Changes in biologically-active ultraviolet radiation reaching the Earth’s surface. Photochem. Photobiol. Sci. 2007, 6, 218–231. [Google Scholar] [CrossRef]
- Li, F.R.; Peng, S.L.; Chen, B.M.; Hou, Y.P. A meta-analysis of the responses of woody and herbaceous plants to elevated ultraviolet-B radiation. Acta Oecol. 2010, 36, 1–9. [Google Scholar] [CrossRef]
- Watanabe, S.; Sudo, K.; Nagashima, T.; Takemura, T.; Kawase, H.; Nozawa, T. Future projections of surface UV-B in a changing climate. J. Geophys. Res. Atmos. 2011, 116. [Google Scholar] [CrossRef]
- Jansen, M.A.K.; Gaba, V.; Greenberg, B.M. Higher plants and UV-B radiation: Balancing damage, repair and acclimation. Trends Plant Sci. 1998, 3, 131–135. [Google Scholar] [CrossRef]
- Hofmann, R.W.; Campbell, B.D.; Fountain, D.W.; Jordan, B.R.; Greer, D.H.; Hunt, D.Y.; Hunt, C.L. Multivariate analysis of intraspecific responses to UV-B radiation in white clover (Trifolium repens L.). Plant Cell Environ. 2001, 24, 917–927. [Google Scholar] [CrossRef]
- Watermann, L.Y.; Hock, M.; Blake, C.; Erfmeier, A. Plant invasions into high elevations implies adaptation to high UV-B environments—A multi-species experiment. Biol. Invasions 2019. [Google Scholar] [CrossRef]
- Holmes, M.G.; Keiller, D.R. Effects of pubescence and waxes on the reflectance of leaves in the ultraviolet and photosynthetic wavebands: A comparison of a range of species. Plant Cell Enviorn. 2002, 25, 85–93. [Google Scholar] [CrossRef]
- Manetas, Y. The importance of being hairy: The adverse effects of hair removal on stem photosynthesis of Verbascum speciosum are due to solar UV-B radiation. New Phytol. 2003, 158, 503–508. [Google Scholar] [CrossRef]
- Micco, V.D.; Aronne, G. Morpho-Anatomical Traits for Plant Adaptation to Drought. In Plant Responses to Drought Stress; Aroca, R., Ed.; Springer: Berlin/Heidelberg, Germany, 2012; pp. 37–61. [Google Scholar]
- Jaleel, C.A.; Manivannan, P.; Wahid, A.; Farooq, M.; Al-Juburi, H.J.; Somasundaram, R.; Panneerselvam, R. Drought stress in plants: A review on morphological characteristics and pigments composition. Int. J. Agric. Biol. 2009, 11, 100–105. [Google Scholar]
- Ren, J.; Dai, W.; Xuan, Z.; Yao, Y.; Korpelainen, H.; Li, C. The effect of drought and enhanced UV-B radiation on the growth and physiological traits of two contrasting poplar species. For. Ecol. Manag. 2007, 239, 112–119. [Google Scholar] [CrossRef]
- Yang, Y.; Yao, Y.; Xu, G.; Li, C. Growth and physiological responses to drought and elevated ultraviolet-B in two contrasting populations of Hippophae rhamnoides. Physiol. Plant. 2005, 124, 431–440. [Google Scholar] [CrossRef]
- Sullivan, J.H.; Teramura, A.H. Field Study of the Interaction between Solar Ultraviolet-B Radiation and Drought on Photosynthesis and Growth in Soybean. Plant Physiol. 1990, 92, 141–146. [Google Scholar] [CrossRef]
- Alexieva, V.; Sergiev, I.; Mapelli, S.; Karanov, E. The effect of drought and ultraviolet radiation on growth and stress markers in pea and wheat. Plant Cell Environ. 2001, 24, 1337–1344. [Google Scholar] [CrossRef]
- Björn, L.O.; Callaghan, T.V.; Johnsen, I.; Lee, J.A.; Manetas, Y.; Paul, N.D.; Sonesson, M.; Wellburn, A.R.; Coop, D.; Heide-Jørgensen, H.S.; et al. The effects of UV-B radiation on European heathland species. In UV-B and Biosphere. Advances in Vegetation Science; Rozema, J., Gieskes, W.W.C., Van De Geijn, S.C., Nolan, C., De Boois, H., Eds.; Springer: Dordrecht, The Netherlands, 1997; Volume 17, pp. 252–264. [Google Scholar]
- Manetas, Y.; Petropoulou, Y.; Stamatakis, K.; Nikolopoulos, D.; Levizou, E.; Psaras, G.; Karabourniotis, G. Beneficial effects of enhanced UV-B radiation under field conditions: Improvement of needle water relations and survival capacity of Pinus pinea L. seedlings during the dry Mediterranean summer. In UV-B and Biosphere. Advances in Vegetation Science; Rozema, J., Gieskes, W.W.C., Van De Geijn, S.C., Nolan, C., De Boois, H., Eds.; Springer: Dordrecht, The Netherlands, 1997; Volume 17, pp. 100–108. [Google Scholar]
- Poulson, M.E.; Donahue, R.A.; Konvalinka, J.; Boeger, M.R.T. Enhanced tolerance of photosynthesis to high-light and drought stress in Pseudotsuga menziesii seedlings grown in ultraviolet-B radiation. Tree Physiol. 2002, 22, 829–838. [Google Scholar] [CrossRef]
- Hofmann, R.W.; Campbell, B.D.; Bloor, S.J.; Swinny, E.E.; Markham, K.R.; Ryan, K.G.; Fountain, D.W. Responses to UV-B radiation in Trifolium repens L—Physiological links to plant productivity and water availability. Plant Cell Enviorn. 2003, 26, 603–612. [Google Scholar] [CrossRef]
- Bandurska, H.; Niedziela, J.; Chadzinikolau, T. Separate and combined responses to water deficit and UV-B radiation. Plant Sci. 2013, 213, 98–105. [Google Scholar] [CrossRef] [PubMed]
- Hofmann, R.W.; Campbell, B.D. Leaf-level responses to ultraviolet-B radiation in Trifolium repens populations under defoliation pressure. Enviorn. Exp. Bot. 2012, 78, 64–69. [Google Scholar] [CrossRef]
- Bacelar, E.; Moutinho-Pereira, J.; Ferreira, H.; Correia, C. Enhanced ultraviolet-B radiation affect growth, yield and physiological processes on triticale plants. Procedia Enviorn. Sci. 2015, 29, 219–220. [Google Scholar] [CrossRef]
- Newsham, K.K.; Robinson, S.A. Responses of plants in polar regions to UVB exposure: A meta-analysis. Glob. Chang. Biol. 2009, 15, 2574–2589. [Google Scholar] [CrossRef]
- Porcar-Castell, A.; Juurola, E.; Nikinmaa, E.; Berninger, F.; Ensminger, I.; Hari, P. Seasonal acclimation of photosystem II in Pinus sylvestris. I. Estimating the rate constants of sustained thermal energy dissipation and photochemistry. Tree Physiol. 2008, 28, 1475–1482. [Google Scholar] [CrossRef] [PubMed]
- Ranjbarfordoei, A.; Samson, R.; Van Damme, P. Photosynthesis performance in sweet almond [Prunus dulcis (Mill) D. Webb] exposed to supplemental UV-B radiation. Photosynthetica 2011, 49, 107. [Google Scholar] [CrossRef]
- Osório, J.; Osório, M.L.; Correia, P.J.; de Varennes, A.; Pestana, M. Chlorophyll fluorescence imaging as a tool to understand the impact of iron deficiency and resupply on photosynthetic performance of strawberry plants. Sci. Hortic. 2014, 165, 148–155. [Google Scholar] [CrossRef]
- Anjum, S.; Xie, X.; Wang, L.; Saleem, M.S.; Man, C.; Lei, W. Morphological, physiological and biochemical responses of plants to drought stress. Afr. J. Agric. Res. 2011, 6, 2026–2032. [Google Scholar]
- Li, R.H.; Guo, P.G.; Michael, B.; Stefania, G.; Salvatore, C. Evaluation of chlorophyll content and fluorescence parameters as indicators of drought tolerance in barley. Agric. Sci. China 2006, 5, 751–757. [Google Scholar] [CrossRef]
- Yao, J.; Sun, D.; Cen, H.; Xu, H.; Weng, H.; Yuan, F.; He, Y. Phenotyping of Arabidopsis drought stress response using kinetic chlorophyll fluorescence and multicolor fluorescence imaging. Front Plant Sci. 2018, 9, 603. [Google Scholar] [CrossRef]
- Alba, C.; Hufbauer, R. Exploring the potential for climatic factors, herbivory, and co-occurring vegetation to shape performance in native and introduced populations of Verbascum thapsus. Biol. Invasions 2012, 14, 2505–2518. [Google Scholar] [CrossRef]
- Hideg, É.; Nagy, T.; Oberschall, A.; Dudits, D.; Vass, I. Detoxification function of aldose/aldehyde reductase during drought and ultraviolet-B (280–320 nm) stresses. Plant Cell Enviorn. 2003, 26, 513–522. [Google Scholar] [CrossRef]
- Rinnan, R.; Keinänen, M.M.; Kasurinen, A.; Asikainen, J.; Kekki, T.K.; Holopainen, T.; Ro-Poulsen, H.; Mikkelsen, T.N.; Michelsen, A. Ambient ultraviolet radiation in the Arctic reduces root biomass and alters microbial community composition but has no effects on microbial biomass. Glob. Chang. Biol. 2005, 11, 564–574. [Google Scholar] [CrossRef]
- Jouili, H.; El Ferjani, E. Changes in antioxidant and lignifying enzyme activities in sunflower roots (Helianthus annuus L.) stressed with copper excess. C. R. Biol. 2003, 326, 639–644. [Google Scholar] [CrossRef]
- Claussen, W. Proline as a measure of stress in tomato plants. Plant Sci. 2005, 168, 241–248. [Google Scholar] [CrossRef]
- Lachmuth, S.; Durka, W.; Schurr, F.M. The making of a rapid plant invader: Genetic diversity and differentiation in the native and invaded range of Senecio inaequidens. Mol. Ecol. 2010, 19, 3952–3967. [Google Scholar] [CrossRef] [PubMed]
- Colautti, R.I.; Maron, J.L.; Barrett, S.C. Common garden comparisons of native and introduced plant populations: Latitudinal clines can obscure evolutionary inferences. Evol. Appl. 2009, 2, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Hodgins, K.A.; Rieseberg, L. Genetic differentiation in life-history traits of introduced and native common ragweed (Ambrosia artemisiifolia) populations. J. Evol. Biol. 2011, 24, 2731–2749. [Google Scholar] [CrossRef]
- Dieskau, J.; Bruelheide, H.; Gutknecht, J.; Erfmeier, A. Biogeographic differences in plant-soil biota relationships contribute to the invasion of Verbascum thapsus. Ecol. Evol. under review.
- Alba, C.; Bowers, D.M.; Blumenthal, D.M.; Hufbauer, R. Evolution of growth but not structural or chemical defense in Verbascum thapsus (common mullein) following introduction to North America. Biol. Invasions 2011, 13, 2379–2389. [Google Scholar] [CrossRef]
- Endriss, S.B.; Alba, C.; Norton, A.P.; Pyšek, P.; Hufbauer, R.A. Breakdown of a geographic cline explains high performance of introduced populations of a weedy invader. J. Ecol. 2018, 106, 699–713. [Google Scholar] [CrossRef]
- Alpert, P.; Bone, E.; Holzapfel, C. Invasiveness, invasibility and the role of environmental stress in the spread of non-native plants. Perspect Plant Ecol. 2000, 3, 52–66. [Google Scholar] [CrossRef]
- te Beest, M.; Elschot, K.; Olff, H.; Etienne, R.S. Invasion success in a marginal habitat: An experimental test of competitive ability and drought tolerance in Chromolaena odorata. PLoS ONE 2013, 8, e68274. [Google Scholar] [CrossRef] [PubMed]
- Ashbacher, A.C.; Cleland, E.E. Native and exotic plant species show differential growth but similar functional trait responses to experimental rainfall. Ecosphere 2015, 6, 1–14. [Google Scholar] [CrossRef]
- Antunes, C.; Pereira, A.J.; Fernandes, P.; Ramos, M.; Ascensão, L.; Correia, O.; Máguas, C. Understanding plant drought resistance in a Mediterranean coastal sand dune ecosystem: Differences between native and exotic invasive species. J. Plant Ecol. 2018, 11, 26–38. [Google Scholar] [CrossRef]
- Erfmeier, A.; Böhnke, M.; Bruelheide, H. Secondary invasion of Acer negundo: The role of phenotypic responses versus local adaptation. Biol. Invasions 2011, 13, 1599–1614. [Google Scholar] [CrossRef]
- Kumschick, S.; Hufbauer, R.A.; Alba, C.; Blumenthal, D.M. Evolution of fast-growing and more resistant phenotypes in introduced common mullein (Verbascum thapsus). J. Ecol. 2013, 101, 378–387. [Google Scholar] [CrossRef]
- Hubbard, J.C.; Wilson, J.B. A survey of the lowland vegetation of the Upper Clutha District of Otago, New Zealand. N. Z. J. Bot. 1988, 26, 21–35. [Google Scholar] [CrossRef]
- Turker, A.U.; Gurel, E. Common mullein (Verbascum thapsus L.): Recent advances in research. Phytother. Res. 2005, 19, 733–739. [Google Scholar] [CrossRef]
- Gross, K.L.; Werner, P.A. The biology of Canadian weeds: 28. Verbascum thapsus L. and V. blattaria L. Can. J. Plant Sci. 1978, 58, 401–413. [Google Scholar] [CrossRef]
- Kuznetsova, A.; Brockhoff, P.B.; Christensen, R.H.B. lmerTest Package: Tests in Linear Mixed Effects Models. J. Stat. Softw. 2017, 82. [Google Scholar] [CrossRef]
- Alexieva, V.; Ivanov, S.; Sergiev, I.; Karanov, E. Interaction between stresses. Bulg. J. Plant Physiol. 2003, 29, 1–17. [Google Scholar]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).