Sustainability Perspectives of Vigna unguiculata L. Walp. Cultivation under No Tillage and Water Stress Conditions
Abstract
1. Introduction
2. Material and Methods
2.1. Experimental Design and Treatments
2.2. Biomass and Morphophysiological Sampling and Analysis
2.3. Measurement of Soil Physical Properties
2.4. Chemical Characterization of V. unguiculata Seeds
2.4.1. Evaluation of Total Starch Content (TSC)
2.4.2. Total Protein Content (TPC)
2.4.3. Amino Acidic Content and Characterization
2.5. Plant Morphometry, Photosynthetic Efficiency, and Phytochemistry
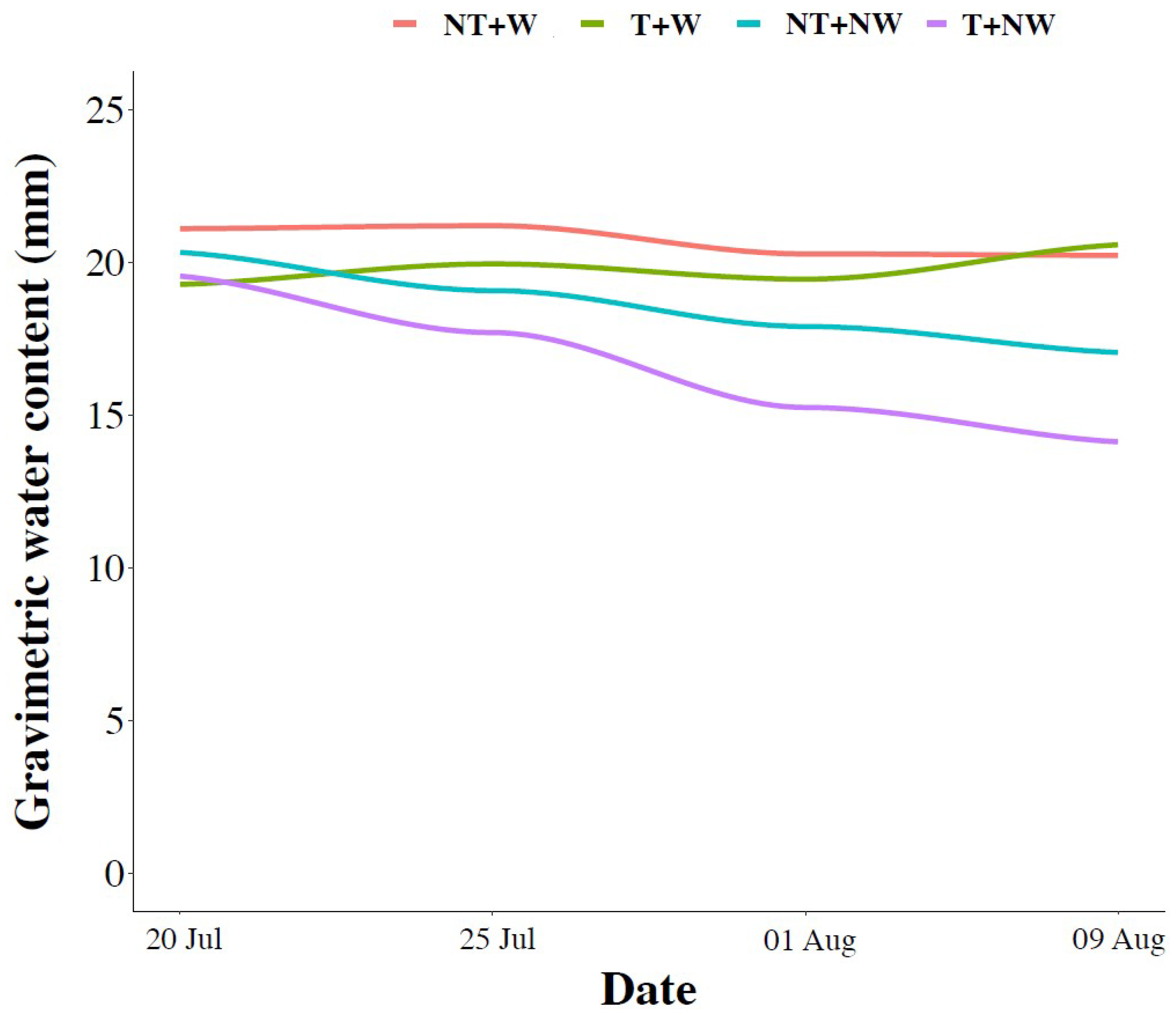
2.6. Soil Properties
3. Results
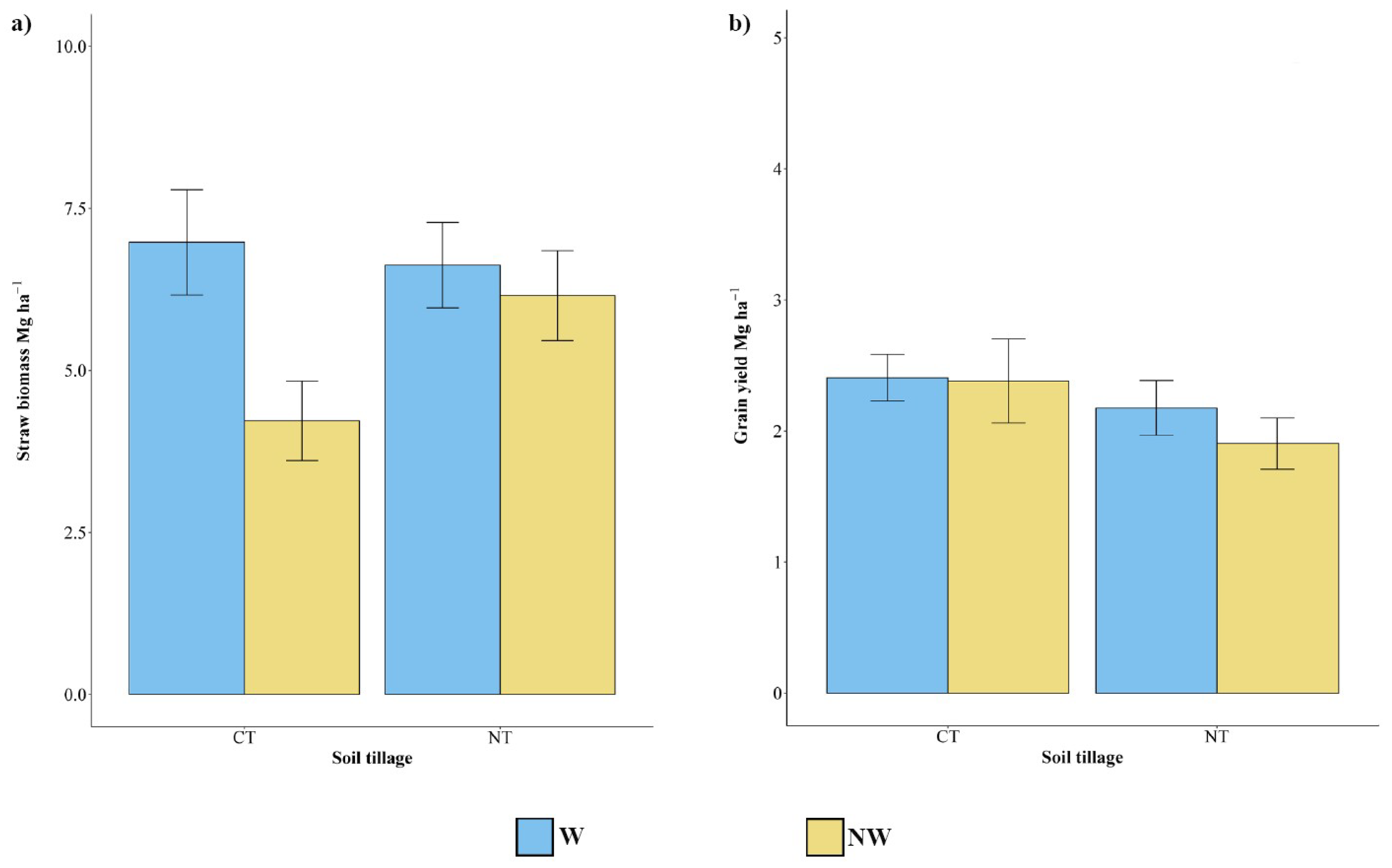
3.1. Biomass and Grain Yield
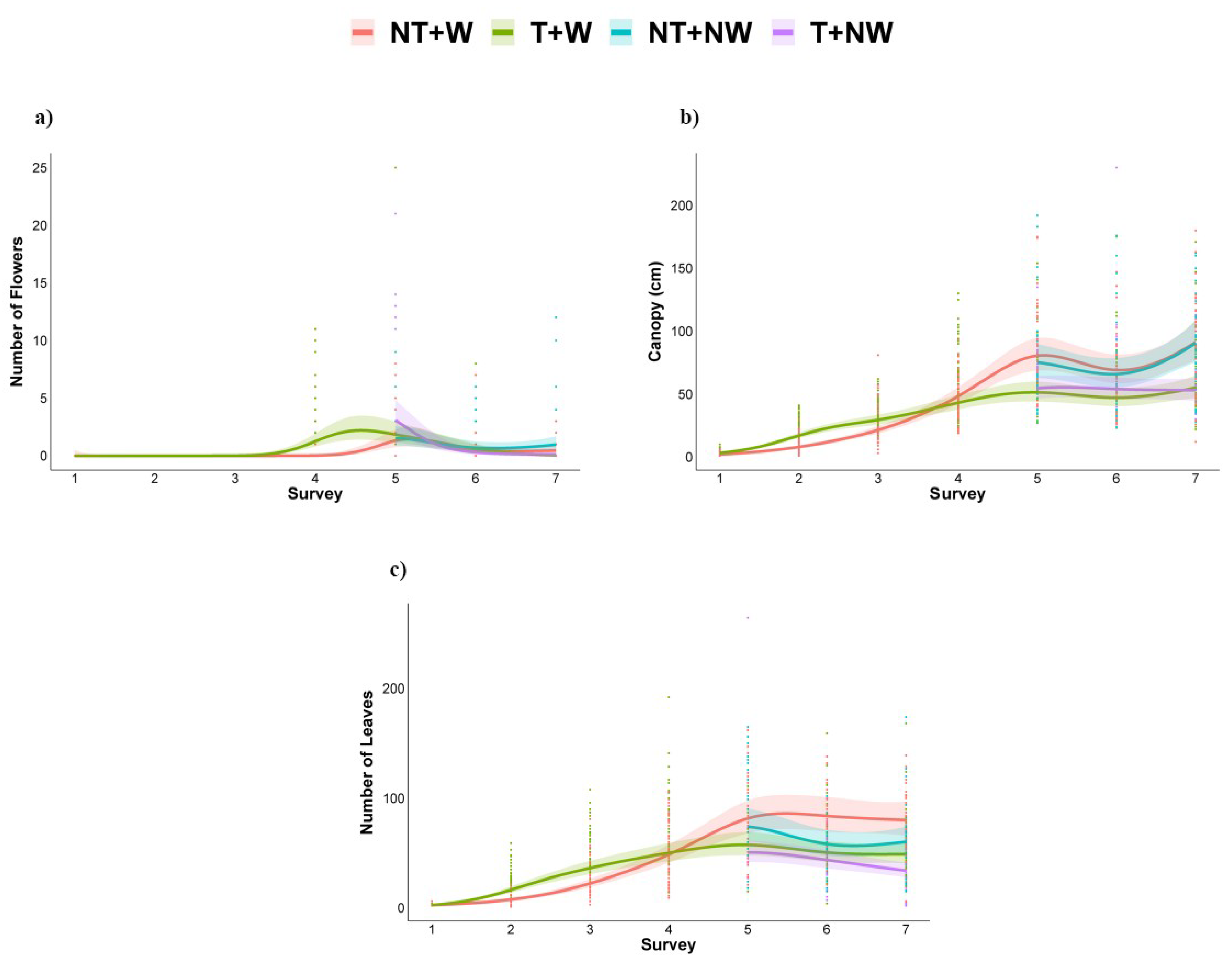
3.2. Morphometrics and Growth Parameters
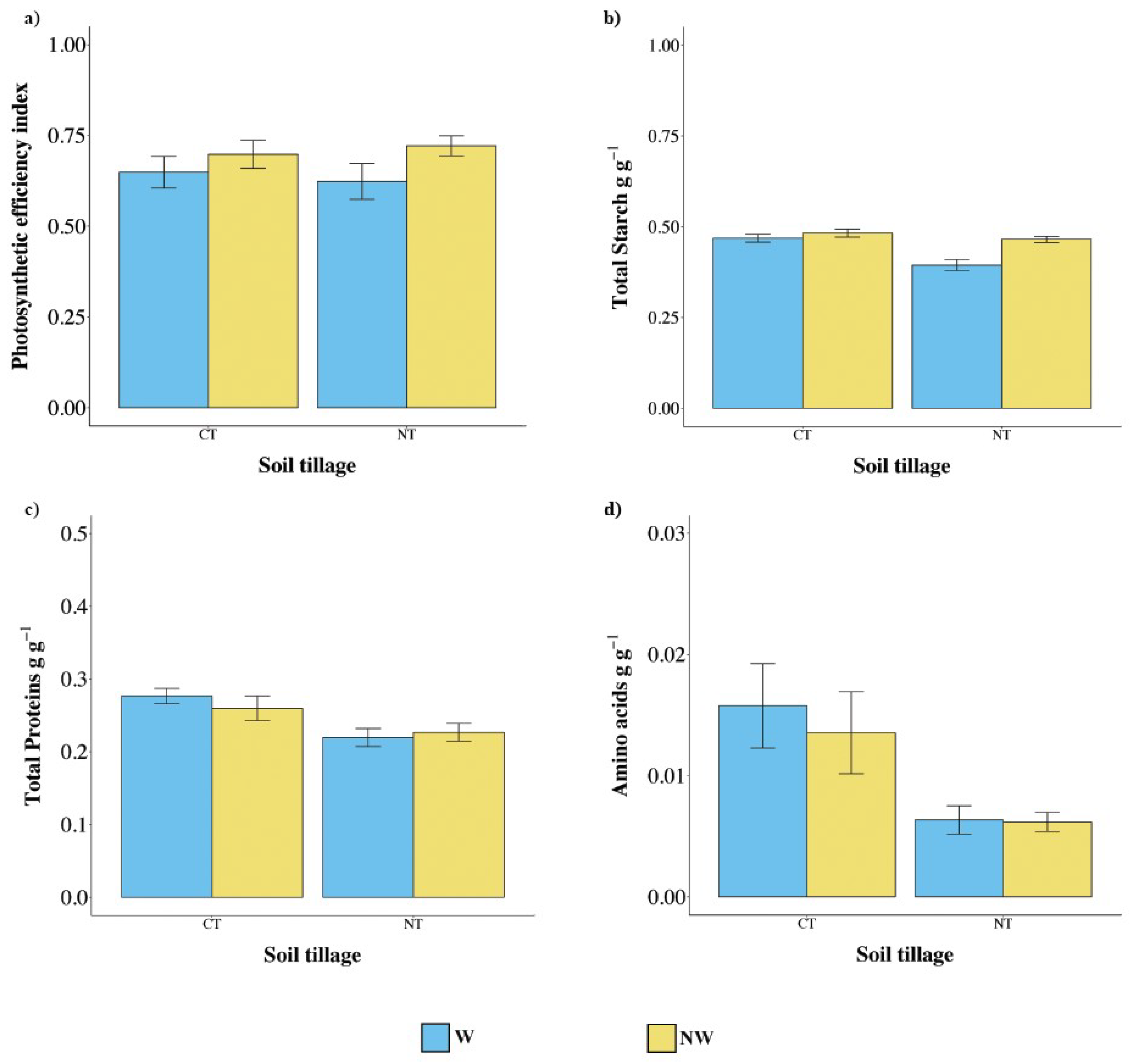
3.3. Efficiency of Photosynthesis and Metabolic Profile
3.4. Soil Organic Carbon and Total Nitrogen
4. Discussion
4.1. Suitability of V. unguiculata for Mediterranean CA
4.2. Conservative Cultivation of V. unguiculata Enhances Soil Fertility
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Evenson, R.E.; Gollin, D. Assessing the impact of the Green Revolution, 1960 to 2000. Science 2003, 300, 758–762. [Google Scholar] [CrossRef]
- Carvalho, F.P. Agriculture, pesticides, food security and food safety. Environ. Sci. Policy 2006, 9, 685–692. [Google Scholar] [CrossRef]
- De Fraiture, C.; Wichelns, D. Satisfying future water demands for agriculture. Agric. Water Manag. 2010, 4, 502–551. [Google Scholar] [CrossRef]
- FAO. Investing in Sustainable Crop Intensification: The Case for Soil Health. In Report of the International Technical Workshop; FAO: Rome, Italy, 2008. [Google Scholar]
- Kassam, A.; Friedrich, T.; Shaxson, F.; Pretty, J. The spread of conservation agriculture: Justification, sustainability and uptake. Int. J. Agric. Sustain. 2009, 7, 292–320. [Google Scholar] [CrossRef]
- Pittelkow, C.M.; Liang, X.; Linquist, B.A.; van Groenigen, L.J.; Lee, J.; Lundy, M.E.; van Gestel, N.; Six, J.; Venterea, R.T.; van Kessel, C. Productivity limits and potentials of the principles of conservation agriculture. Nature 2015, 517, 365–368. [Google Scholar] [CrossRef] [PubMed]
- Derpsch, R. Conservation Tillage, No-Tillage and Related Technologies. In Conservation Agriculture; García-Torres, L., Benites, J., Martínez-Vilela, A., Holgado-Cabrera, A., Eds.; Springer: Dordrecht, The Netherlands, 2003; pp. 181–190. [Google Scholar] [CrossRef]
- Zdruli, P. Land resources of the Mediterranean: Status, pressures, trends and impacts on future regional development. Land Degrad. Dev. 2014, 25, 373–384. [Google Scholar] [CrossRef]
- Kassam, A.; Friedrich, T.; Derpsch, R.; Lahmar, R.; Mrabet, R.; Basch, G.; González-Sánchez, E.J.; Serraj, R. Conservation agriculture in the dry Mediterranean climate. Field Crops Res. 2012, 132, 7–17. [Google Scholar] [CrossRef]
- Ward, P.R.; Flower, K.C.; Cordingley, N.; Weeks, C.; Micin, S.F. Soil water balance with cover crops and conservation agriculture in a Mediterranean climate. Field Crops Res. 2012, 132, 33–39. [Google Scholar] [CrossRef]
- Perego, A.; Rocca, A.; Cattivelli, V.; Tabaglio, V.; Fiorini, A.; Barbieri, S.; Schillaci, C.; Chiodini, M.E.; Brenna, S.; Acutis, M. Agro-environmental aspects of conservation agriculture compared to conventional systems: A 3-year experience on 20 farms in the Po valley (Northern Italy). Agric. Syst. 2019, 168, 73–87. [Google Scholar] [CrossRef]
- Graham, P.H.; Vance, C.P. Legumes: Importance and Constraints to Greater Use. Plant Physiol. 2003, 131, 872–877. [Google Scholar] [CrossRef]
- IAASTD. Agriculture at a Crossroads: The Synthesis Report. In International Assessment of Agricultural Knowledge, Science and Technology for Development; Island Press: Washington, DC, USA, 2008. [Google Scholar] [CrossRef]
- Baulcombe, D.; Crute, I.; Davies, B.; Dunwell, J.; Gale, M.; Jones, J.; Pretty, J.; Sutherland, W.; Toulmin, C. Reaping the Benefits: Science and the Sustainable Intensification of Global Agriculture; The Royal Society: London, UK, 2009; Available online: https://royalsociety.org/-/media/Royal_Society_Content/policy/publications/2009/4294967719.pdf (accessed on 1 November 2019).
- FAO. Food, agriculture and cities. Save and grow. In A Policymaker’s Guide to the Sustainable Intensification of Smallholder Crop Production; FAO: Rome, Italy, 2011. [Google Scholar]
- Campanaro, A.; Tommasi, N.; Guzzetti, L.; Galimberti, A.; Bruni, I.; Labra, M. DNA barcoding to promote social awareness and identity of neglected, underutilized plant species having valuable nutritional properties. Food Res. Int. 2019, 115, 1–9. [Google Scholar] [CrossRef]
- Jayathilake, C.; Visvanathan, R.; Deen, A.; Bangamuwage, R.; Jayawardana, B.C.; Nammi, S.; Lianage, R. Cowpea: An overview on its nutritional facts and health benefits. J. Sci. Food Agric. 2018, 98, 4793–4806. [Google Scholar] [CrossRef]
- Powlson, D.S.; Stirling, C.M.; Jat, M.L.; Gerard, B.G.; Palm, C.A.; Sanchez, P.A.; Cassman, K.G. Limited potential of no-till agriculture for climate change mitigation. Nat. Clim. Chang. 2014, 4, 678–683. [Google Scholar] [CrossRef]
- Fiorini, A.; Boselli, R.; Amaducci, S.; Tabaglio; V. Effects of no-till on root architecture and root-soil interactions in a three-year crop rotation. Eur. J. Agron. 2018, 99, 156–166. [Google Scholar] [CrossRef]
- Gómez-Paccard, C.; Hontoria, C.; Mariscal-Sancho, I.; Pérez, J.; León, P.; González, P.; Espejo, R. Soil-water relationships in the upper soil layer in a Mediterranean Palexerult as affected by no-tillage under excess water conditions—Influence on crop yield. Soil Tillage Res. 2015, 146, 303–312. [Google Scholar] [CrossRef]
- Olaleke, A.M.; Olorunfemi, O.; Emmanuel, A.T. A comparative study on the chemical and amino acid composition of some Nigerian underutilized legume flours. Pak. J. Nutr. 2006, 5, 34–38. [Google Scholar] [CrossRef]
- Zuur, A.; Ieno, E.N.; Walker, N.; Saveliev, A.A.; Smith, G.M. Mixed Effects Models and Extensions in Ecology with R; Springer: New York, NY, USA, 2009. [Google Scholar]
- Gomez, K.A.; Gomez, A.A. Statistical Procedures for Agricultural Research, 2nd ed.; John Wiley and Sons Inc.: New York, NY, USA, 1984. [Google Scholar]
- Smith, M.R.; Veneklaas, E.; Polania, J.; Rao, I.M.; Beebe, S.E.; Merchant, A. Field drought conditions impact yield but not nutritional quality of the seed in common bean (Phaseolus vulgaris L.). PLoS ONE 2019, 14. [Google Scholar] [CrossRef]
- Tabaglio, V.; Gavazzi, C.; Menta, C. Physico-chemical indicators and microarthropod communities as influenced by no-till, conventional tillage and nitrogen fertilisation after four years of continuous maize. Soil Tillage Res. 2009, 105, 135–142. [Google Scholar] [CrossRef]
- Palm, C.; Blanco-Canqui, H.; DeClerck, F.; Gatere, L.; Grace, P. Conservation agriculture and ecosystem services. An overview. Agric. Ecosyst. Environ. 2014, 187, 87–105. [Google Scholar] [CrossRef]
- Vogeler, I.; Rogasik, J.; Funder, U.; Panten, K.; Schnug, E. Effect of tillage systems and P-fertilization on soil physical and chemical properties: Crop yield and nutrient uptake. Soil Tillage Res. 2009, 103, 137–143. [Google Scholar] [CrossRef]
- Guan, D.; Al-Kaisi, M.M.; Zhang, Y.; Duan, L.; Tan, W. Tillage practices affect biomass and grain yield through regulating root growth, root-bleeding sap and nutrients uptake in summer maize. Field Crops Res. 2014, 157, 89–97. [Google Scholar] [CrossRef]
- Cantero-Martinez, C.; Angas, P.; Lampurlanes, J. Growth, yield and water productivity of barley (Hordeum vulgare L.) affected by tillage and N fertilization in Mediterranean semiarid, rainfed conditions of Spain. Field Crops Res. 2003, 84, 341–357. [Google Scholar] [CrossRef]
- Pittelkow, C.M.; Linquist, B.A.; Lundy, M.E.; Liang, X.; van Groenigen, K.J.; Lee, J.; van Gestel, N.; Six, J.; Venterea, R.T.; van Kessel, K. When does no-till yield more? A global meta-analysis. Field Crops Res. 2015, 156–168. [Google Scholar] [CrossRef]
- Plaza-Bonilla, D.; Cantero-Martínez, C.; Bareche, J.; Arrúe, J.L.; Lampurlanés, J.; Álvaro-Fuentes, J. Do no-till and pig slurry application improve barley yield and water and nitrogen use efficiencies in rainfed Mediterranean conditions? Field Crops Res. 2017, 203, 74–85. [Google Scholar] [CrossRef]
- Moroke, T.S.; Schwartz, R.C.; Brown, K.W.; Juo, A.S.R. Water use efficiency of dryland cowpea, sorghum and sunflower under reduced tillage. Soil Tillage Res. 2011, 112, 76–84. [Google Scholar] [CrossRef]
- Ahamefule, E.H.; Peter, P.C. Cowpea (Vigna unguiculata L. Walp) response to phosphorus fertilizer under two tillage and mulch treatments. Soil Tillage Res. 2014, 136, 70–75. [Google Scholar] [CrossRef]
- Supit, I. Predicting national wheat yields using a crop simulation and trend models. Agric. Forest Meteorol. 1997, 88, 199–214. [Google Scholar] [CrossRef]
- Omer, A.; Pascual, U.; Russell, N.P. Biodiversity conservation and productivity in intensive agricultural systems. J. Agric. Econ. 2007, 58, 308–329. [Google Scholar] [CrossRef]
- Pascual, U.; Perrings, C. Developing incentives and economic mechanisms for in situ biodiversity conservation in agricultural landscapes. Agric. Ecosyst. Environ. 2007, 121, 256–268. [Google Scholar] [CrossRef]
- Loi, A.; Howieson, J.G.; Nutt, B.J.; Carr, S.J. A second generation of annual pasture legumes and their potential for inclusion in Mediterranean-type farming systems. Aust. J. Exp. Agric. 2005, 45, 289–299. [Google Scholar] [CrossRef]
- European Commission. Agriculture and Rural Development Oilseeds and Protein Crops Market Situation. In Committee for the Common Organization of Agricultural Markets; European Commission: Brussels, Belgium, 2017; Available online: https://ec.europa.eu/agriculture/sites/agriculture/files/cereals/presentations/cereals-oilseeds/market-situation-oilseeds_en.pdf (accessed on 1 November 2019).
- European Commission. Development of Plant Proteins in the European Union. 9 October 2018. Available online: https://ec.europa.eu/info/events/development-plant-proteins-europe-opportunities-and-challenges-2018-nov-22_en (accessed on 1 November 2019).
- Alidu, M.S. Genetic Variability for Flowering Time, Maturity and Drought Tolerance in Cowpea [Vigna unguiculata (L.) Walp.]: A Review Paper. J. Agric. Ecol. Res. Int. 2019, 17, 1–18. [Google Scholar] [CrossRef]
- Boutraa, T.; Sanders, F.E. Influence of water stress on grain yield and vegetative growth of two cultivars of bean (Phaseolus vulgaris L.). J. Agron. Crop Sci. 2001, 187, 251–257. [Google Scholar] [CrossRef]
- Del Pozo, A.; Brunel-Saldias, N.; Engler, A.; Ortega-Farias, S.; Acevedo-Opazo, C.; Lobos, G.A.; Jara-Rojas, R.; Molina-Montenegro, M.A. Climate Change Impacts and Adaptation Strategies of Agriculture in Mediterranean-Climate Regions (MCRs). Sustainability 2019, 11, 2769. [Google Scholar] [CrossRef]
- Souza, R.P.; Machado, E.C.; Silva, J.A.B.; Lagôa, A.M.M.A.; Silveira, J.A.G. Photosynthetic gas exchange, chlorophyll fluorescence and some associated metabolic changes in cowpea (Vigna unguiculata) during water stress and recovery. Environ. Exp. Bot. 2004, 51, 45–56. [Google Scholar] [CrossRef]
- Bacelar, E.A.; Santos, D.L.; Moutinho-Pereira, J.M.; Gonçalves, B.C.; Ferreira, H.F.; Correia, C.M. Immediate responses and adaptative strategies of three olive cultivars under contrasting water availability regimes: Changes on structure and chemical composition of foliage and oxidative damage. Plant Sci. 2006, 170, 596–605. [Google Scholar] [CrossRef]
- Carvalho, M.; Lino-Neto, T.; Rosa, E.; Carnide, V. Cowpea: A legume crop for a challenging environment. J. Sci. Food Agric. 2017, 97, 4273–4284. [Google Scholar] [CrossRef]
- Castro-Quezada, I.; Román-Viñas, B.; Serra-Majem, L. The Mediterranean diet and nutritional adequacy: A review. Nutrients 2014, 6, 231–248. [Google Scholar] [CrossRef]
- Lal, R. Carbon sequestration. Philos. Trans. R. Soc. B Biol. Sci. 2008, 363, 815–830. [Google Scholar] [CrossRef]
- West, T.O.; Post, W.M. Soil Organic Carbon Sequestration Rates by Tillage and Crop Rotation: A Global Data Analysis. Soil Sci. Soc. Am. J. 2002, 66, 1930–1946. [Google Scholar] [CrossRef]
- Aguilera, E.; Lassaletta, L.; Gattinger, A.; Benjamín, S.G. Managing soil carbon for climate change mitigation and adaptation in Mediterranean cropping systems: A meta-analysis. Agric. Ecosyst. Environ. 2013, 168, 25–36. [Google Scholar] [CrossRef]
- Alvarez, R.; Steinbach, H.S. A review of the effects of tillage systems on some soil physical properties, water content, nitrate availability and crops yield in the Argentine Pampas. Soil Tillage Res. 2009, 104, 1–5. [Google Scholar] [CrossRef]
- Lal, R. Soil carbon sequestration impacts on global climate change and food security. Science 2004, 304, 1623–1627. [Google Scholar] [CrossRef] [PubMed]
- Halvorson, A.D.; Wienhold, B.J.; Black, A.L. Tillage, nitrogen and cropping system effects on soil carbon sequestration. Soil Sci. Soc. Am. J. 2002, 66, 906–912. [Google Scholar] [CrossRef]
- Mazzoncini, M.; Di Bene, C.; Coli, A.; Antichi, D.; Petri, M.; Bonari, E. Rainfed Wheat and Soybean Productivity in a Long-Term Tillage Experiment in Central Italy. Agron. J. 2008, 100. [Google Scholar] [CrossRef]
- Fabrizzi, K.P.; García, F.O.; Costa, J.L.; Picone, L.I. Soil water dynamics, physical properties and corn and wheat responses to minimum and no-tillage systems in the southern Pampas of Argentina. Soil Tillage Res. 2005, 81, 57–69. [Google Scholar] [CrossRef]
- Six, J.; Feller, C.; Denef, K.; Ogle, S.M.; de Moraes Sa, J.C.; Albrecht, A. Soil organic matter, biota and aggregation in temperate and tropical soils—Effects of no-tillage. Agronomie 2002, 22, 755–775. [Google Scholar] [CrossRef]
- Dixit, A.K.; Agrawal, R.K.; Das, S.K.; Sahay, C.S.; Choudhary, M.; Rai, A.K.; Kumar, S.; Kantua, S.R.; Palsaniya, D.R. Soil properties, crop productivity and energetics under different tillage practices in fodder + sorghum cowpea–wheat cropping system. Arch. Agron. Soil Sci. 2019, 65, 492–506. [Google Scholar] [CrossRef]
- Montanaro, G.; Xiloyannis, C.; Nuzzo, V.; Dichio, B. Orchard management, soil organic carbon and ecosystem services in Mediterranean fruit tree crops. Sci. Hortic. 2017, 92–101. [Google Scholar] [CrossRef]
| Condition | Code | C Concentration 0–5 cm (g C kg−1 soil) | C Concentration 5–30 cm (g C kg−1 soil) | C Stock (Mg ha−1) |
|---|---|---|---|---|
| Tillage | CT | 12.49 ± 1.48 | 12.39 ± 0.88 | 48.56 ± 3.37 |
| NT | 19.92 ± 0.73 | 12.5 ± 0.76 | 51.15 ± 2.24 | |
| Signif. | *** | n.s. | * | |
| Water | W | 16.17 ± 4.06 | 12.49 ± 0.76 | 49.98 ± 2.81 |
| NW | 16.24 ± 4.22 | 12.4 ± 0.88 | 49.73 ± 3.51 | |
| Signif. | n.s. | n.s. | n.s. | |
| Interaction | CT-W | 12.46 ± 0.92 | 12.43 ± 1.03 | 48.69 ± 3.53 |
| CT-NW | 12.51 ± 2.07 | 12.34 ± 0.87 | 48.43 ± 3.74 | |
| NT-NW | 19.88 ± 0.97 | 12.54 ± 0.54 | 51.27 ± 1.24 | |
| NT-NW | 19.96 ± 0.55 | 12.45 ± 1.02 | 51.03 ± 3.19 | |
| Signif. | n.s. | n.s. | n.s. |
| Condition | Code | N Concentration 0–5 cm (g N kg−1 soil) | N Concentration 5–30 cm (g N kg−1 soil) | N Stock (Mg ha−1) |
|---|---|---|---|---|
| Tillage | CT | 1.49 ± 0.21 | 1.49 ± 0.1 | 5.84 ± 0.46 |
| NT | 1.9 ± 0.21 | 1.57 ± 0.1 | 6.1 ± 0.37 | |
| Signif. | ** | n.s. | n.s. | |
| Water | W | 1.72 ± 0.37 | 1.55 ± 0.11 | 6.04 ± 0.39 |
| NW | 1.67 ± 0.22 | 1.52 ± 0.12 | 5.91 ± 0.48 | |
| Signif. | n.s. | n.s. | n.s. | |
| Interaction | CT-W | 1.41 ± 0.09 | 1.48 ± 0.1 | 5.76 ± 0.37 |
| CT-NW | 1.57 ± 0.28 | 1.5 ± 0.12 | 5.93 ± 0.58 | |
| NT-NW | 2.03 ± 0.22 | 1.62 ± 0.06 | 6.32 ± 0.08 | |
| NT-NW | 1.76 ± 0.08 | 1.53 ± 0.13 | 5.88 ± 0.43 | |
| Signif. | n.s. | n.s. | n.s. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guzzetti, L.; Fiorini, A.; Panzeri, D.; Tommasi, N.; Grassi, F.; Taskin, E.; Misci, C.; Puglisi, E.; Tabaglio, V.; Galimberti, A.; et al. Sustainability Perspectives of Vigna unguiculata L. Walp. Cultivation under No Tillage and Water Stress Conditions. Plants 2020, 9, 48. https://doi.org/10.3390/plants9010048
Guzzetti L, Fiorini A, Panzeri D, Tommasi N, Grassi F, Taskin E, Misci C, Puglisi E, Tabaglio V, Galimberti A, et al. Sustainability Perspectives of Vigna unguiculata L. Walp. Cultivation under No Tillage and Water Stress Conditions. Plants. 2020; 9(1):48. https://doi.org/10.3390/plants9010048
Chicago/Turabian StyleGuzzetti, Lorenzo, Andrea Fiorini, Davide Panzeri, Nicola Tommasi, Fabrizio Grassi, Eren Taskin, Chiara Misci, Edoardo Puglisi, Vincenzo Tabaglio, Andrea Galimberti, and et al. 2020. "Sustainability Perspectives of Vigna unguiculata L. Walp. Cultivation under No Tillage and Water Stress Conditions" Plants 9, no. 1: 48. https://doi.org/10.3390/plants9010048
APA StyleGuzzetti, L., Fiorini, A., Panzeri, D., Tommasi, N., Grassi, F., Taskin, E., Misci, C., Puglisi, E., Tabaglio, V., Galimberti, A., & Labra, M. (2020). Sustainability Perspectives of Vigna unguiculata L. Walp. Cultivation under No Tillage and Water Stress Conditions. Plants, 9(1), 48. https://doi.org/10.3390/plants9010048










