Hydrogen Peroxide and Superoxide Anion Radical Photoproduction in PSII Preparations at Various Modifications of the Water-Oxidizing Complex
Abstract
1. Introduction
2. Results
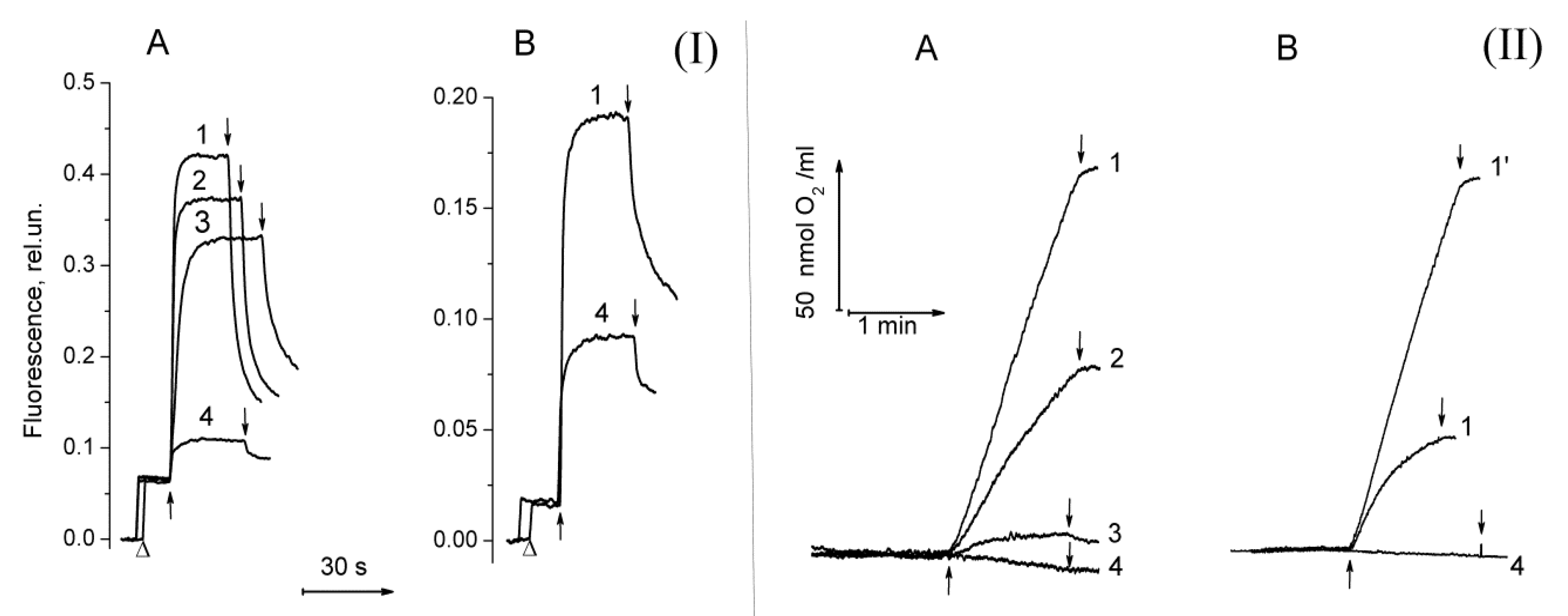
2.1. Functional Activity in PSII Preparations at Various Modifications of the WOC
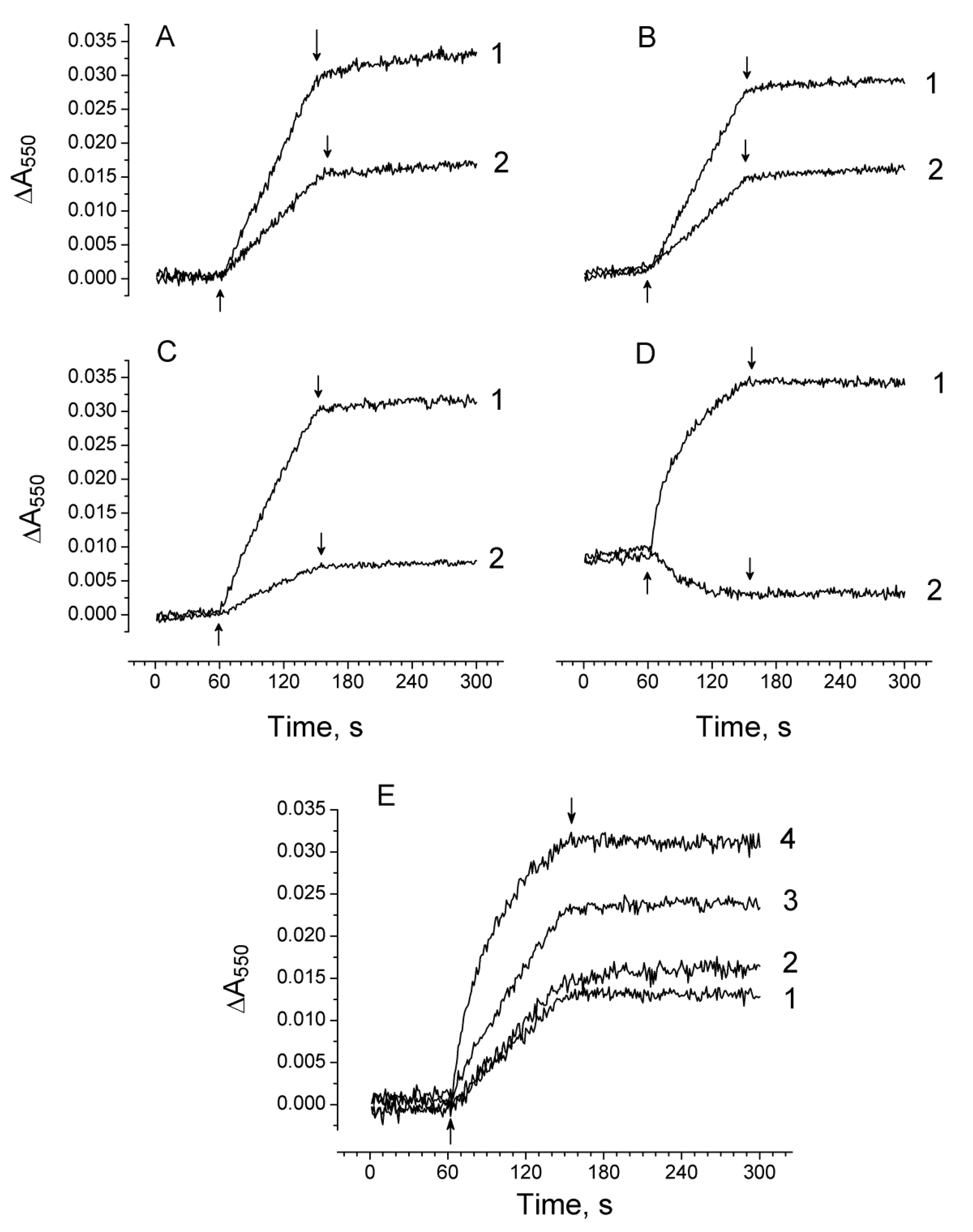
2.2. The Ratio in Redox Forms of Cyt b559 in PSII Preparations at Various Modifications of the WOC
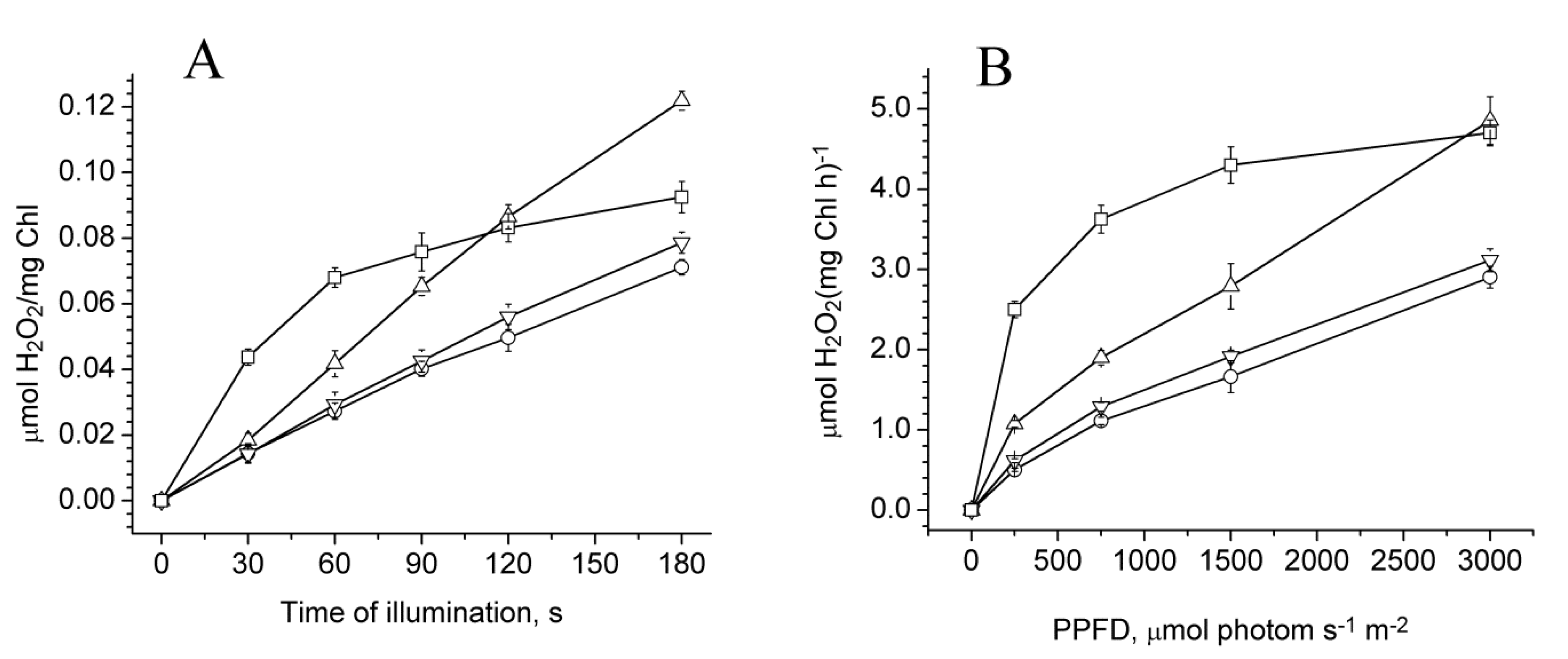
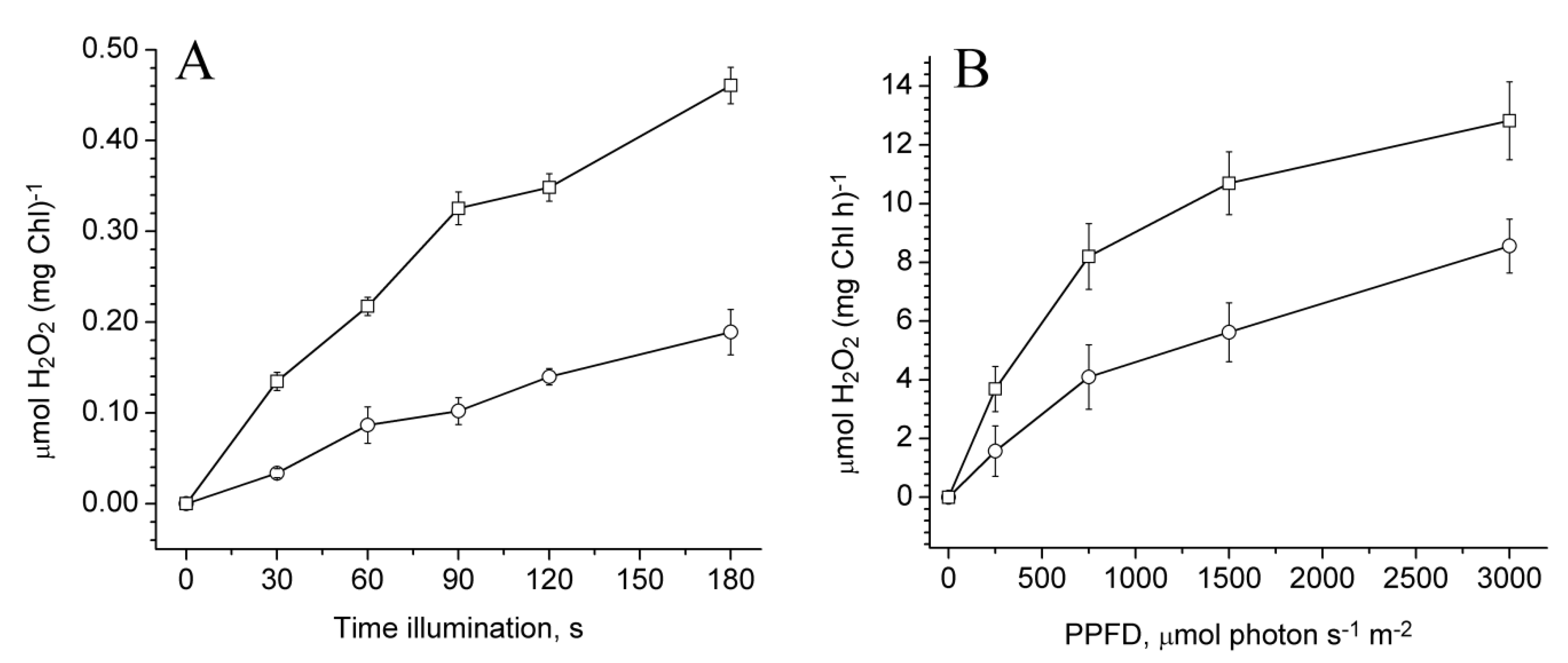
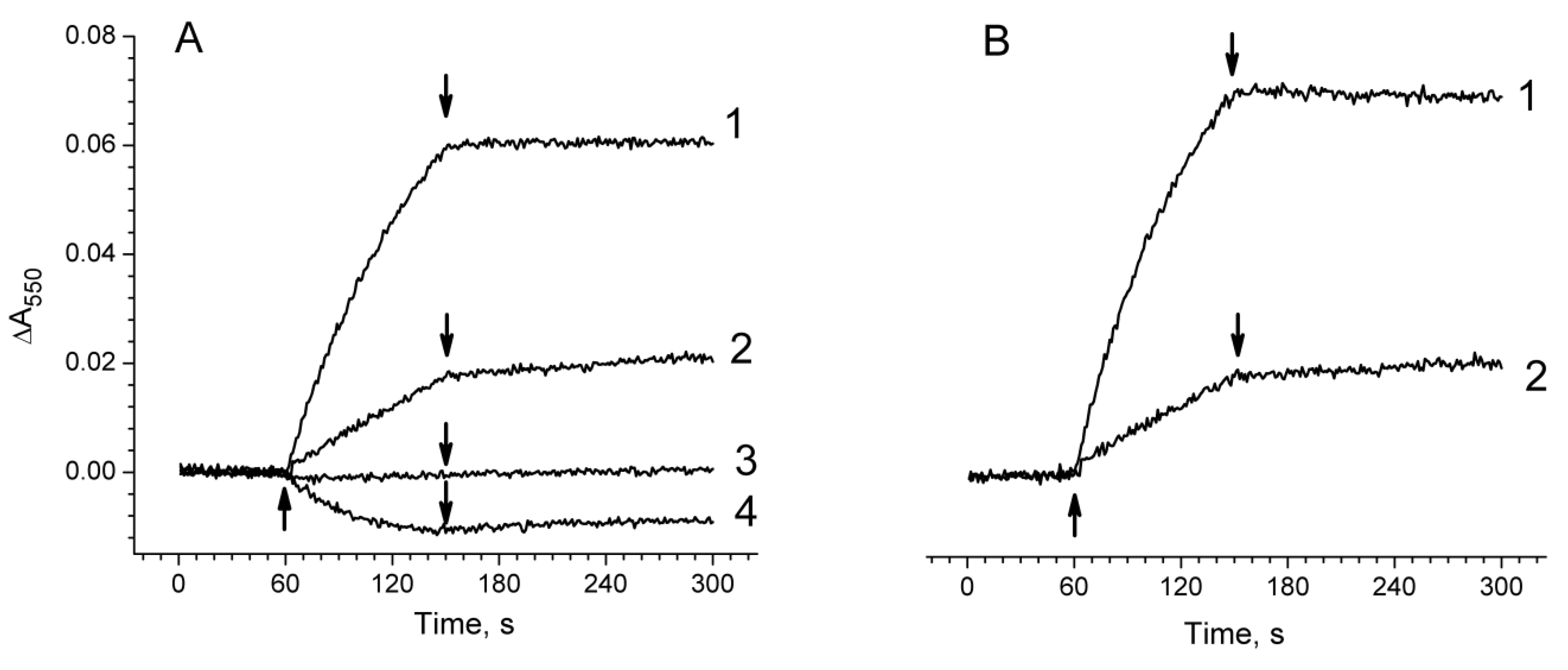
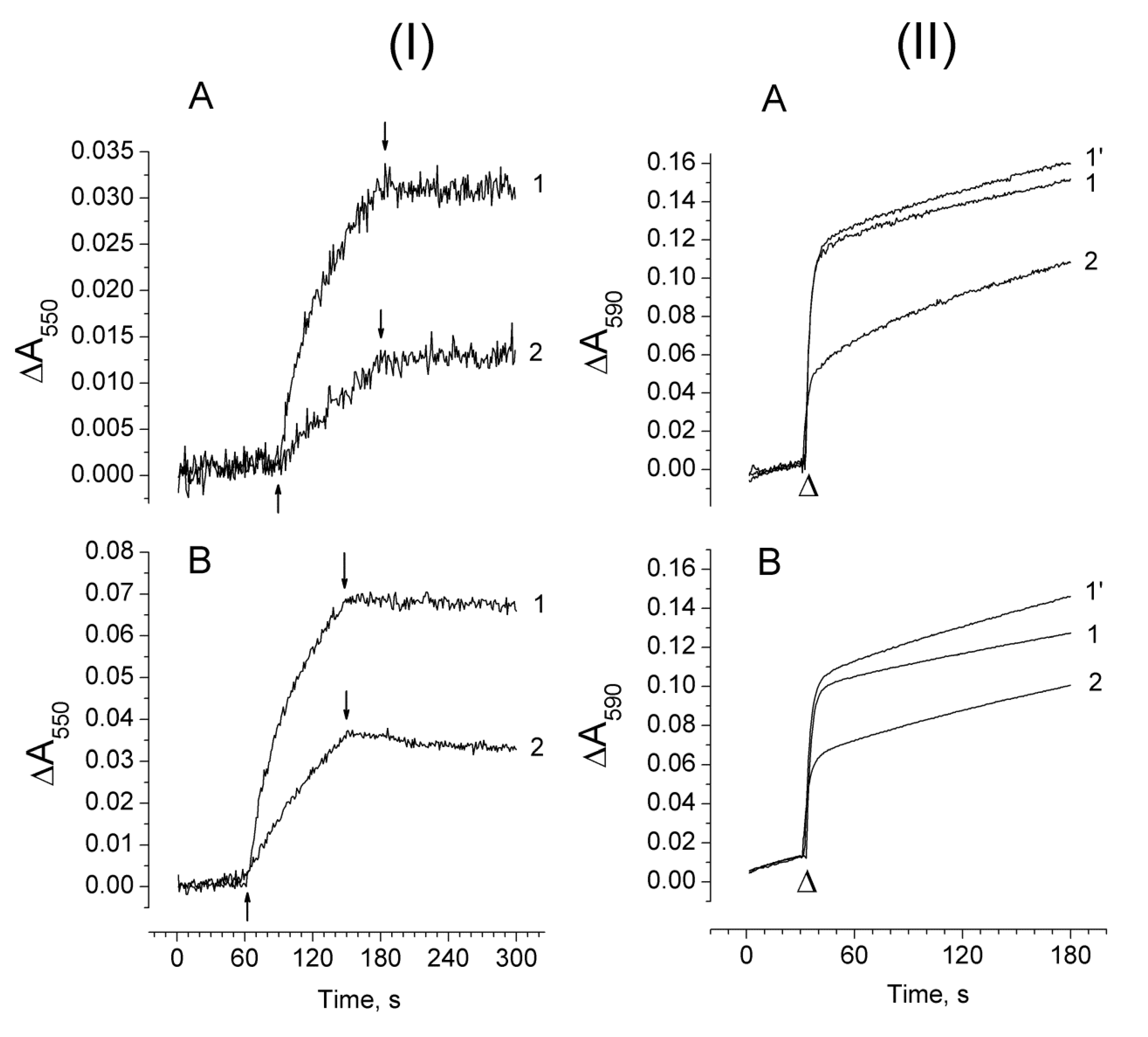
2.3. Photoproduction of H2O2 in PSII Preparations at Various Modifications of the WOC
2.4. Photoproduction of O2−• in PSII Preparations at Various Modification of the WOC
2.5. Effect of Exogenous Electron Donors on the Photoproduction of O2−• and H2O2 in Mn-Depleted PSII Preparations
3. Discussion
4. Materials and Methods
4.1. Isolation of PS II Membranes and PSII Core Complexes
4.2. Preparation of PSII Membranes with a Different Degree of Disassembly of the WOC and Mn-Depleted PSII Core Complexes
4.3. Measurements of Functional Activity of PSII Preparations
4.4. Determination of H2O2 Photoproduction by PSII Preparations
4.5. Determination of O2−• Photoproduction by PSII Preparations
4.6. Analysis of Redox Forms of Cyt b559 in PSII Preparations
Supplementary Materials
Funding
Acknowledgments
Conflicts of Interest
References
- Loll, B.; Kern, J.; Saenger, W.; Zouni, A.; Biesiadka, J. Lipids in photosystem II: Interactions with protein and cofactors. Biochim. Biophys. Acta 2007, 1767, 509–519. [Google Scholar] [CrossRef]
- Guskov, A.; Kern, J.; Gabdulkhakov, A.; Broser, M.; Zouni, A.; Saenger, W. Cyanobacterial photosytems II at 2.9-Å resolution and the role of quinones, lipids, channels and chloride. Nat. Struct. Mol. Biol. 2009, 16, 334–342. [Google Scholar] [CrossRef]
- Umena, Y.; Kawakami, K.; Shen, J.-R.; Kamiya, N. Crystal structure of oxygen-evolving photosystem II at a resolution of 1.9 Å. Nature 2011, 473, 55–61. [Google Scholar] [CrossRef]
- Klimov, V.V.; Allakhverdiev, S.I.; Demeter, S.; Krasnovsky, A.A. Photoreduction of pheophytin in chloroplast photosystem II as a function of the redox potential of the medium. Dokl. Akad. Nauk SSSR 1979, 249, 227–230. [Google Scholar]
- Ishikita, H.; Loll, B.; Biesiadka, J.; Saenger, W.; Knapp, E.-W. Redox potentials of chlorophylls in the photosystem II reaction center. Biochemistry 2005, 44, 4118–4124. [Google Scholar] [CrossRef]
- Müh, F.; Zouni, A. Cytochrome b559 in photosystem II. In Cytochrome Complexes: Evolution, Structures, Energy Transduction, and Signaling; Cramer, A., Kallas, T., Eds.; Springer: Dordrecht, The Netherlands, 2016; Volume 41, pp. 143–175. [Google Scholar]
- Shuvalov, V.A.; Schreiber, U.; Heber, U. Spectral and thermodynamic properties of the two hemes of the D1D2cytochrome b-559 complex of spinach. FEBS Lett. 1994, 337, 226–230. [Google Scholar] [CrossRef]
- Kaminskaya, O.; Kurreck, J.; Irrgang, K.D.; Renger, G.; Shuvalov, V.A. Redox and spectral properties of cytochrome b559 in different preparations of Photosystem II. Biochemistry 1999, 38, 16223–16235. [Google Scholar] [CrossRef]
- Mizusawa, N.; Miyao, M.; Yamashita, T. Restoration of the high-potential form of cytochrome b-559 by electron transport reactions through photosystem II in Tris-treated photosystem II membranes. Biochim. Biophys. Acta 1997, 1318, 145–158. [Google Scholar] [CrossRef]
- Mamedov, F.; Gadjieva, R.; Styring, S. Oxygen-induced changes in the redox state of the cytochrome b559 in photosystem II depend on the integrity of the Mn cluster. Physiol. Plant. 2007, 131, 41–49. [Google Scholar] [CrossRef]
- Crofts, J.; Horton, P. Dissipation of excitation energy by Photosystem II particles at low pH. Biochim. Biophys. Acta 1991, 1058, 187–193. [Google Scholar] [CrossRef]
- Barber, J.; De Las Rivas, J. A functional model for the role of cytochrome b559 in the protection against donor and acceptor side photoinhibition. Proc. Natl. Acad. Sci. USA 1993, 90, 10942–10946. [Google Scholar] [CrossRef]
- Thompson, L.K.; Brudvig, G.W. Cytochrome b-559 may function to protect Photosystem II from photoinhibition. Biochemistry 1988, 27, 6653–6658. [Google Scholar] [CrossRef]
- Faller, P.; Fufezan, C.; Rutherford, A.W. Side path electron donors: Cytochrome b559, chlorophyll Z and β-carotene. In Photosystem II: The Light-Driven Water: Plastoquinone Oxidoreductase; Wydrzynski, T., Satoh, K., Eds.; Springer: Dordrecht, The Netherlands, 2005; pp. 347–365. [Google Scholar]
- Pospíšil, P. Enzymatic function of cytochrome b559 in photosystem II. J. Photochem. Photobiolb. 2011, 104, 341–347. [Google Scholar] [CrossRef]
- Klimov, V.V.; Ananyev, G.M.; Zastryzhnaya, O.M.; Wydrzynski, T.; Renger, G. Photoproduction of hydrogen peroxide in Photosystem II membrane fragments: A comparison of four signals. Photosynth. Res. 1993, 38, 409–416. [Google Scholar] [CrossRef]
- Zastrizhnaya, O.M.; Khorobrykh, A.A.; Khristin, M.S.; Klimov, V.V. Photoinduced production of hydrogen peroxide at the acceptor side of photosystem II. Biochemistry 1997, 62, 357–362. [Google Scholar]
- Ananyev, G.M.; Renger, G.; Wacker, U.; Klimov, V.V. The photoproduction of superoxide radicals and the superoxide dismutase activity of Photosystem II. The possible involvement of cytochrome b559. Photosynth. Res. 1994, 41, 327–338. [Google Scholar] [CrossRef]
- Ananyev, G.; Wydrzynski, T.; Renger, G.; Klimov, V. Transient peroxide formation by the manganese-containing redox-active donor side of photosystem II upon inhibition of O2 evolution with lauroylcholine chloride. Biochim. Biophys. Acta 1992, 1100, 303–311. [Google Scholar] [CrossRef]
- Khorobrykh, S.A.; Ivanov, B.N. Oxygen reduction in a plastoquinone pool of isolated pea thylakoids. Photosynth. Res. 2002, 71, 209–219. [Google Scholar] [CrossRef]
- Khorobrykh, S.A.; Mubarakshina, M.; Ivanov, B.N. Photosystem I is not solely responsible for oxygen reduction in isolated thylakoids. Biochim. Biophys. Acta 2004, 1657, 164–167. [Google Scholar] [CrossRef]
- Kruk, J.; Strzałka, K. Dark reoxidation of the plastoquinone-pool is mediated by the low potential form of cytochrome b559 in spinach thylakoids. Photosynth. Res. 1999, 62, 273–279. [Google Scholar] [CrossRef]
- Pospišil, P.; Šnyrychova, I.; Kruk, J.; Strzałka, K.; Nauš, J. Evidence that cytochrome b559 is involved in superoxide production in Photosystem II: Effect of synthetic short-chain plastoquinones in a cytochrome b559 tobacco mutant. Biochem. J. 2006, 397, 321–327. [Google Scholar]
- Pospísil, P. Production of reactive oxygen species by photosystem II. Biochim. Biophys. Acta 2009, 1787, 1151–1160. [Google Scholar] [CrossRef]
- Pospíšil, P. Molecular mechanisms of production and scavenging of reactive oxygen species by photosystem II. Biochim. Biophys. Acta 2012, 1817, 218–231. [Google Scholar] [CrossRef]
- Schröder, W.P.; Åkerlund, H.E. Hydrogen Peroxide Production in Photosystem II Preparations. In Current Research in Photosynthesis; Baltscheffsky, M., Ed.; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1990; Volume I, pp. 901–904. [Google Scholar]
- Hillier, W.; Wydrzynski, T. Increases in peroxide formation by the Photosystem II oxygen evolving reactions upon removal of the extrinsic 16, 22 and 33 kDa proteins are reversed by CaCl2 addition. Photosynth. Res. 1993, 38, 417–423. [Google Scholar] [CrossRef]
- Klimov, V.V.; Allakhverdiev, S.I.; Shuvalov, V.A.; Krasnovsky, A.A. Effect of extraction and re-addition of manganese on light reactions of photosystem II preparations. FEBS Lett. 1982, 148, 307–312. [Google Scholar] [CrossRef]
- Khorobrykh, S.A.; Khorobrykh, A.A.; Klimov, V.V.; Ivanov, B.N. Photoconsumption of oxygen in photosystem II preparations under impairment of the water-oxidizing complex. Biochemistry 2002, 67, 683–688. [Google Scholar]
- Yanykin, D.V.; Khorobrykh, A.A.; Khorobrykh, S.A.; Klimov, V.V. Photoconsumption of molecular oxygen on both donor and acceptor sides of photosystem II in Mn-depleted subchloroplast membrane fragments. Biochim. Biophys. Acta 2010, 1797, 516–523. [Google Scholar] [CrossRef]
- Khorobrykh, S.A.; Khorobrykh, A.A.; Yanykin, D.V.; Ivanov, B.N.; Klimov, V.V.; Mano, J. Photoproduction of catalase-insensitive peroxides on the donor side of manganese-depleted photosystem II: Evidence with a specific fluorescent probe. Biochemistry 2011, 50, 10658–10665. [Google Scholar] [CrossRef]
- Ghanotakis, D.F.; Babcock, G.T.; Yocum, C.F. Calcium reconstitutes high rates of oxygen evolution in polypeptide depleted Photosystem II preparations. FEBS Lett. 1984, 167, 127–130. [Google Scholar] [CrossRef]
- Sugiura, M.; Minagawa, J.; Inoue, Y. Properties of chlamydomonas photosystem II core complex with a His-tag at the C-Terminus of the D2 protein. Plant. Cell Physiol. 1999, 40, 311–318. [Google Scholar] [CrossRef][Green Version]
- Ono, T.; Inoue, Y. Ca2+ dependent restoration of evolving activity in CaCl2 washed PSII particles depleted of 33, 24 and 16 kDa proteins. FEBS Lett. 1984, 168, 281–286. [Google Scholar] [CrossRef]
- Khorobrykh, A.A.; Yanykin, D.V.; Klimov, V.V. Photooxidation and photoreduction of exogenous cytochrome c by photosystem II preparations after various modifications of the water-oxidizing complex. Photosynthetica 2018, 56, 244–253. [Google Scholar] [CrossRef]
- Boussac, A.; Picaud, M.; Etienne, A.-L. Effect of potassium iridic chloride on the electron donation by Mn to photosystem II particles. Photobiochem. Photobiophys. 1986, 10, 201–211. [Google Scholar]
- Inoue, H.; Akahori, H.; Noguchi, M. Activation of Electron Donation from Hydrogen Peroxide by Manganese in Non-oxygen evolving Photosystem II Particles. Plant. Cell Physiol. 1987, 28, 1339–1343. [Google Scholar] [CrossRef]
- Klimov, V.V.; Shafiev, M.A.; Allakhverdiev, S.I. Photoinactivation of the reactivation capacity of photosystem II in pea subchloroplast particles after a complete removal of manganese. Photosynth. Res. 1990, 23, 59–65. [Google Scholar] [CrossRef]
- Telfer, A.; De Las Rivas, J.; Barber, J. β-Carotene within the isolated photosystem II reaction centre: Photooxidation and irreversible bleaching of this chromophore by oxidised P680. Biochim. Biophys. Acta 1991, 1060, 106–114. [Google Scholar] [CrossRef]
- Telfer, A.; Frolov, D.; Barber, J.; Robert, B.; Pascal, A. Oxidation of the two β-carotene molecules in the photosystem II reaction center. Biochemistry 2003, 42, 1008–1015. [Google Scholar] [CrossRef]
- Hanley, J.; Deligiannakis, Y.; Pascal, A.; Faller, P.; Rutherford, A.W. Carotenoid oxidation in photosystem II. Biochemistry 1999, 38, 8189–8195. [Google Scholar] [CrossRef]
- Tracewell, C.A.; Vrettos, J.S.; Bautista, J.A.; Frank, H.A.; Brudvig, G.W. Carotenoid photooxidation in photosystem II. Arch. Biochem. Biophys. 2001, 385, 61–69. [Google Scholar] [CrossRef]
- Mizusawa, N.; Wada, H. The role of lipids in photosystem II. Biochim. Biophys. Acta 2012, 1817, 194–208. [Google Scholar] [CrossRef]
- Yanykin, D.V.; Khorobrykh, A.A.; Terentyev, V.V.; Klimov, V.V. Two pathways of photoproduction of organic hydroperoxides on the donor side of photosystem 2 in subchloroplast membrane fragments. Photosynth. Res. 2017, 133, 129–138. [Google Scholar] [CrossRef]
- Kurreck, J.; Schödel, R.; Renger, G. Investigation of the plastoquinone pool size and fluorescence quenching in thylakoid membranes and Photosystem II (PS II) membrane fragments. Photosynth Res. 2000, 63, 171–182. [Google Scholar] [CrossRef]
- Yadav, D.K.; Prasad, A.; Kruk, J.; Pospíšil, P. Evidence for the involvement of loosely bound plastosemiquinones in superoxide anion radical production in photosystem II. PLoS ONE 2014, 9, e0130244. [Google Scholar] [CrossRef]
- Tomo, T.; Okubo, T.; Akimoto, S.; Yokono, M.; Miyashita, H.; Tsuchiya, T.; Noguchi, T.; Mimuro, M. Identification of the special pair of photosystem II in a chlorophyll d-dominated cyanobacterium. Proc. Natl. Acad. Sci. USA 2007, 104, 7283–7288. [Google Scholar] [CrossRef]
- Rappaport, F.; Guergova-Kuras, M.; Nixon, P.J.; Diner, B.A.; Lavergne, J. Kinetics and pathways of charge recombination in photosystem II. Biochemistry 2002, 41, 8518–8527. [Google Scholar] [CrossRef]
- Allakhverdiev, S.I.; Tsuchiya, T.; Watabe, K.; Kojima, A.; Los, D.A.; Tomo, T.; Klimov, V.V.; Mimuro, M. Redox potentials of primary electron acceptor quinone molecule (Qa) and conserved energetics of photosystem II in cyanobacteria with chlorophyll a and chlorophyll d. Proc. Natl. Acad. Sci. USA 2011, 108, 8054–8058. [Google Scholar] [CrossRef]
- Afanas’ev, I.B. Superoxide Ion: Chemistry and Biological Implications; CRC Press: Boca Raton, FL, USA, 1989; Volume 1, pp. 1–279. [Google Scholar]
- Krieger, A.; Rutherford, A.W.; Johnson, G.N. On the determination of redox midpoint potential of the primary quinone electron acceptor, QA, in photosystem II. Biochim. Biophys. Acta 1995, 1229, 193–201. [Google Scholar] [CrossRef]
- Shibamoto, T.; Kato, Y.; Nagao, R.; Yamazaki, T.; Tomo, T.; Watanabe, T. Species-dependence of the redox potential of the primary quinone electron acceptor QA in photosystem II verified by spectroelectrochemistry. FEBS Lett. 2010, 584, 1526–1530. [Google Scholar] [CrossRef]
- Brinkert, K.; De Causmaecker, S.; Krieger-Liszkay, A.; Fantuzzi, A.; Rutherford, A.W. Bicarbonate-induced redox tuning in Photosystem II for regulation and protection. Proc. Natl. Acad. Sci. USA 2016, 113, 12144–12149. [Google Scholar] [CrossRef]
- Krieger, A.; Weis, E. Energy-dependent quenching of chlorophyll-a- fluorescence: The involvement of proton-calcium exchange at photosystem II. Photosynthetica 1992, 27, 89–98. [Google Scholar]
- Johnson, G.N.; Rutherford, A.W.; Krieger, A. A change in the midpoint potential of the quinone QA in Photosystem II associated with photoactivation of oxygen evolution. Biochim. Biophys. Acta 1995, 1229, 201–207. [Google Scholar] [CrossRef]
- Hsu, B.D.; Lee, J.Y.; Pan, R.L. The two binding sites for DCMU in photosystem II. Biochem. Biophys. Res. Commun. 1986, 141, 682–688. [Google Scholar] [CrossRef]
- Klimov, V.V.; Shuvalov, V.A.; Heber, U. Photoreduction of pheophytin as a result of electron donation from the water-splitting system to Photosystem-II reaction centers. Biochim. Biophys. Acta 1985, 809, 345–350. [Google Scholar] [CrossRef]
- Kaminskaya, O.; Shuvalov, V.A.; Renger, G. Evidence for a novel quinone-binding site in the Photosystem II (PS II) complex that regulates the redox potential of cytochrome b559. Biochemistry 2007, 46, 1091–1105. [Google Scholar] [CrossRef]
- Ford, R.C.; Evans, M.C.W. Isolation of a photosystem 2 preparation from higher plants with highly enriched oxygen evolution activity. FEBS Lett. 1983, 160, 159–164. [Google Scholar] [CrossRef]
- Van Leeuwen, P.J.; Nieveen, M.C.; van de Meent, E.J.; Dekker, J.P.; van Gorkom, H.J. Rapid and simple isolation of pure photosystem II core and reaction center particles from spinach. Photosynth. Res. 1991, 28, 149–153. [Google Scholar] [CrossRef]
- Lichtenthaler, H.K. Chlorophylls and carotenoids: Pigments of photosynthetic biomembranes. Methods Enzymol. 1987, 148, 350–382. [Google Scholar]
- Miyao, M.; Murata, N. Partial disintegration and reconstitution of the photosynthetic oxygen evolution system. Binding of 24 kilodalton and 18 kilodalton polypeptides. Biochim. Biophys. Acta 1983, 725, 87–93. [Google Scholar] [CrossRef]
- Ono, T.; Inoue, Y. Mn-preserving extraction of 33-, 24- and 16 kDa proteins from O2-evolving PS II particles by divalent salt-washing. FEBS Lett. 1983, 164, 255–260. [Google Scholar] [CrossRef]
- Tamura, N.; Cheniae, G.M. Photoactivation of the water-oxidizing complex in Photosystem II membranes depleted of Mn and extrinsic proteins. I. Biochemical and kinetic characterization. Biochim. Biophys. Acta 1987, 890, 179–194. [Google Scholar] [CrossRef]
- Ruch, W.; Cooper, P.H.; Baggiolini, M. Assay of H2O2 production by macrophages and neutrophils with homovanillic acid and horse-radish peroxidase. J. Immunol. Methods 1983, 63, 347–357. [Google Scholar] [CrossRef]
- Ngo, T.T.; Lenhoff, H.M. A sensitive and versatile chromogenic assay for peroxidase and peroxidase-coupled reactions. Anal. Biochem. 1980, 105, 389–397. [Google Scholar] [CrossRef]
- Fridovich, I. Quantitative aspects of the production of superoxide anion radical by milk xanthine oxidase. J. Biol. Chem. 1970, 245, 4053–4057. [Google Scholar]
- Chen, G.X.; Kazimir, J.; Cheniae, G.M. Photoinhibition of hydroxylamine extracted photosystem II membranes: Studies of the mechanism. Biochemistry 1992, 31, 11072–11083. [Google Scholar] [CrossRef]
| Redox Forms of Cyt b559 | PSII Membranes | PSII Core Complexes | ||||
|---|---|---|---|---|---|---|
| Untreated | NaCl-Treated | CaCl2-Treated | Mn-Depleted | Untreated | Mn-Depleted | |
| HP | 56.6 | 43.7 | 19.8 | 17.4 | 12 | 11.8 |
| IP | 8.6 | 21.7 | 34.7 | 30.6 | 45.2 | 21 |
| LP | 34.8 | 34.6 | 45.5 | 52 | 42.8 | 67.2 |
| The Rate of Photoproduction, µmol (mg Chl h)−1 | PSII Membranes | PSII Core Complexes | ||||
|---|---|---|---|---|---|---|
| Untreated | NaCl-Treated | CaCl2-Treated | Mn-Depleted | Untreated | Mn-Depleted | |
| H2O2 | 1.7 ± 0.36 | 1.7 ± 0.3 | 2.2 ± 0.25 | 5.25 ± 0.3 | 4.1 ± 0.6 | 16.2 ± 1.2 |
| O2−• | 2.7 ± 0.1 | 2.6 ± 0.2 | 5.3 ± 0.15 | 11.3 ± 0.3 | 6.9 ± 0.25 | 37.8 ± 0.5 |
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Khorobrykh, A. Hydrogen Peroxide and Superoxide Anion Radical Photoproduction in PSII Preparations at Various Modifications of the Water-Oxidizing Complex. Plants 2019, 8, 329. https://doi.org/10.3390/plants8090329
Khorobrykh A. Hydrogen Peroxide and Superoxide Anion Radical Photoproduction in PSII Preparations at Various Modifications of the Water-Oxidizing Complex. Plants. 2019; 8(9):329. https://doi.org/10.3390/plants8090329
Chicago/Turabian StyleKhorobrykh, Andrey. 2019. "Hydrogen Peroxide and Superoxide Anion Radical Photoproduction in PSII Preparations at Various Modifications of the Water-Oxidizing Complex" Plants 8, no. 9: 329. https://doi.org/10.3390/plants8090329
APA StyleKhorobrykh, A. (2019). Hydrogen Peroxide and Superoxide Anion Radical Photoproduction in PSII Preparations at Various Modifications of the Water-Oxidizing Complex. Plants, 8(9), 329. https://doi.org/10.3390/plants8090329




