Discrimination of Myrtle Ecotypes from Different Geographic Areas According to Their Morphological Characteristics and Anthocyanins Composition
Abstract
:1. Introduction
2. Results and Discussion
2.1. Morphological Description
2.2. Correlations between the Morphological Traits
2.3. Correlation between the Morphological and the Chemical Traits
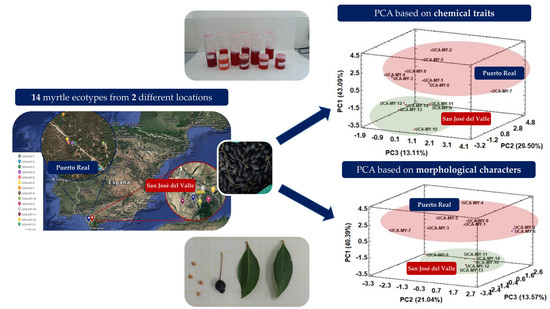
2.4. Correlation between Morphological Traits, Chemical Traits and Geographic Location
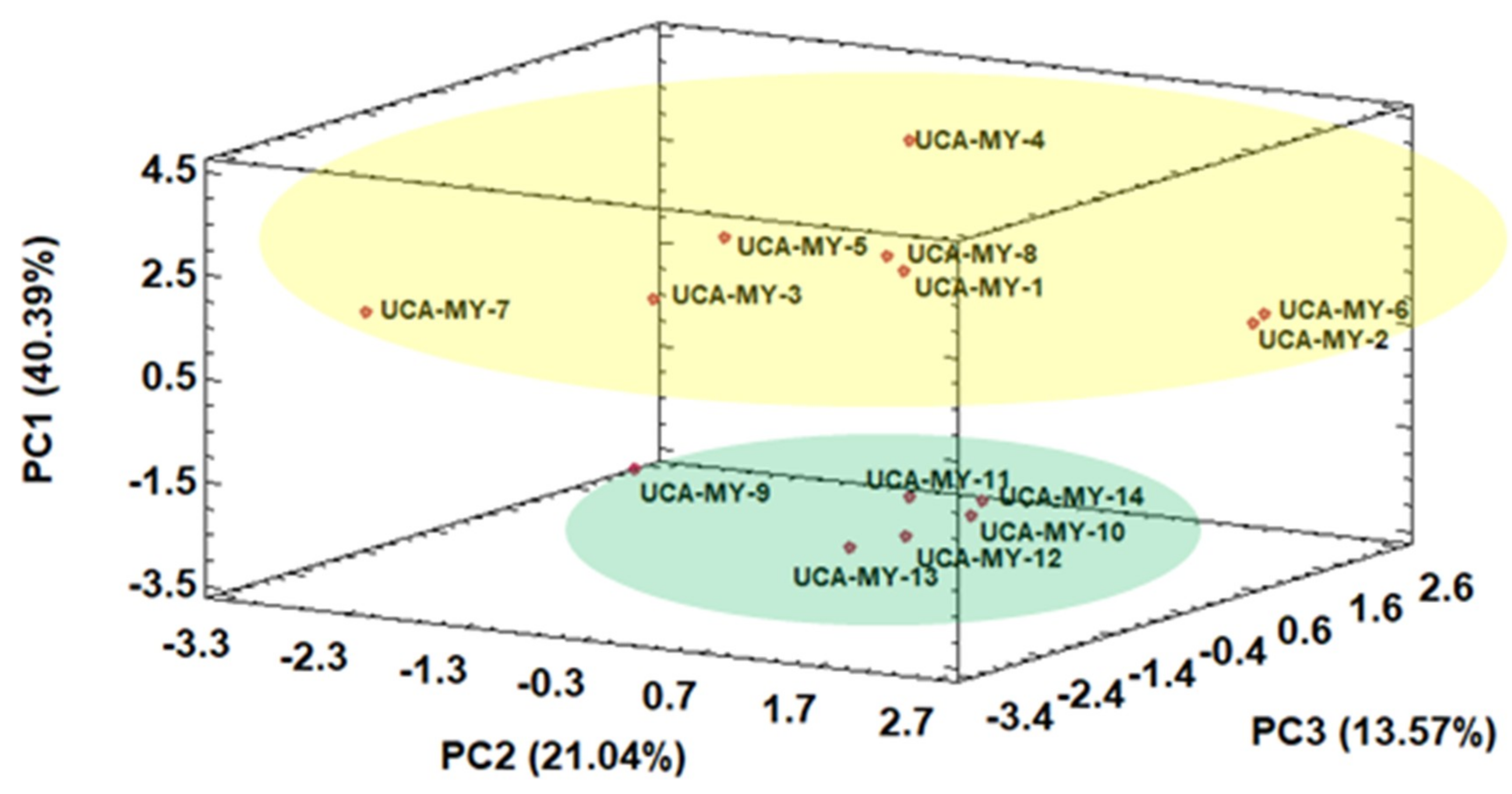
2.4.1. Exploratory Chemometric Study of Morphological Data
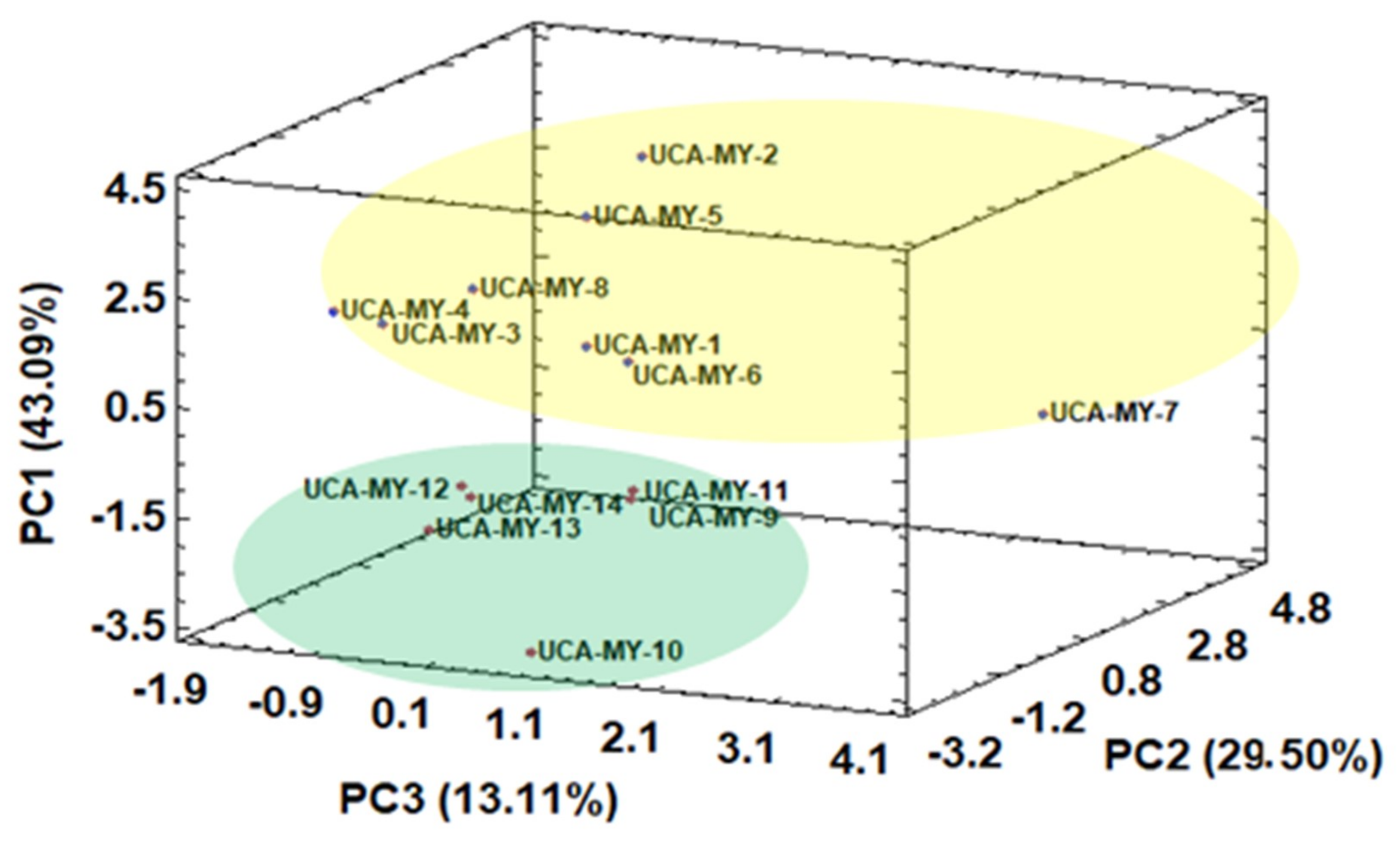
2.4.2. Exploratory Chemometric Study of Chemical Data
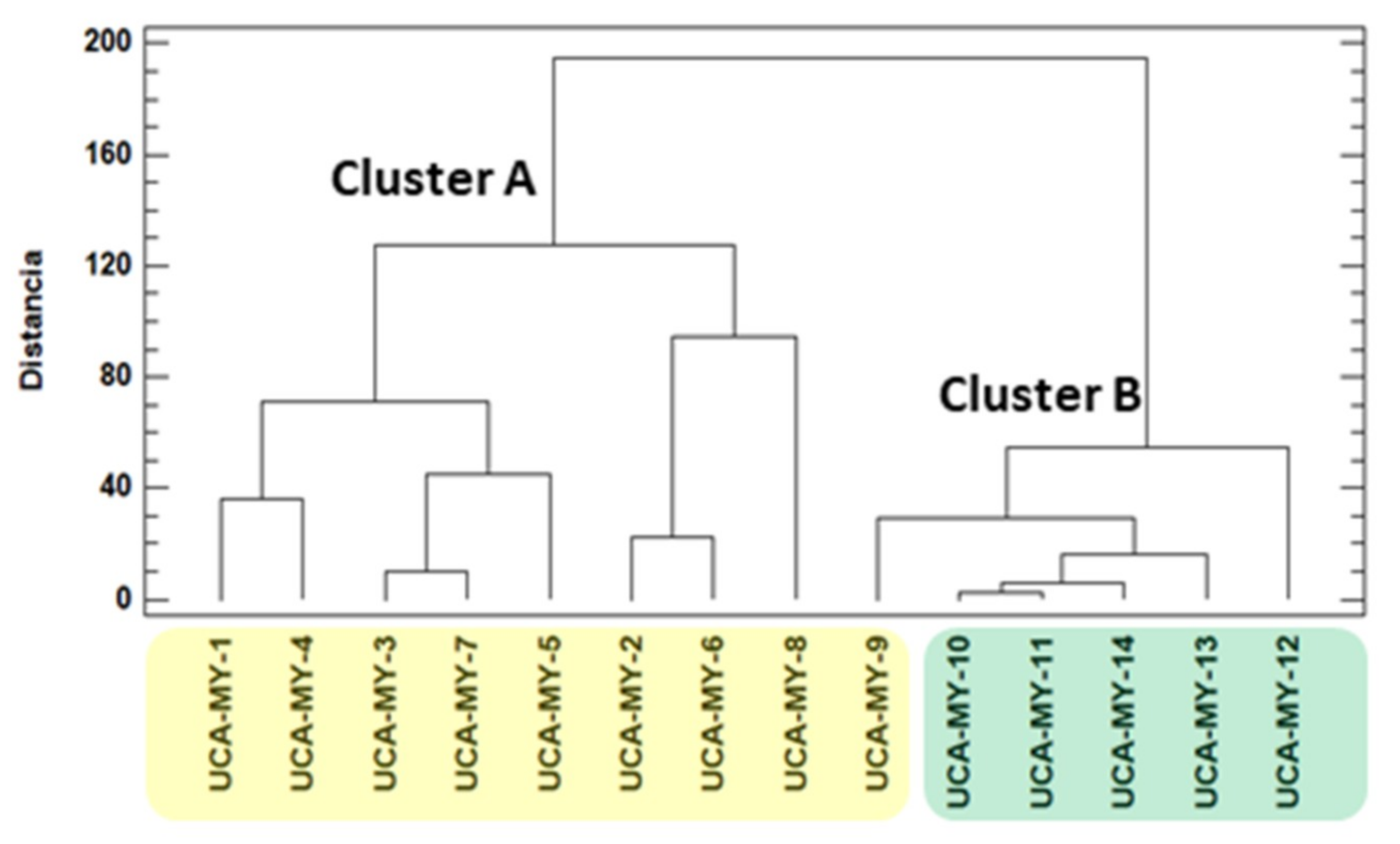
2.4.3. Influence of Ecotype Location on Morphological and Chemical Characteristics
3. Materials and Methods
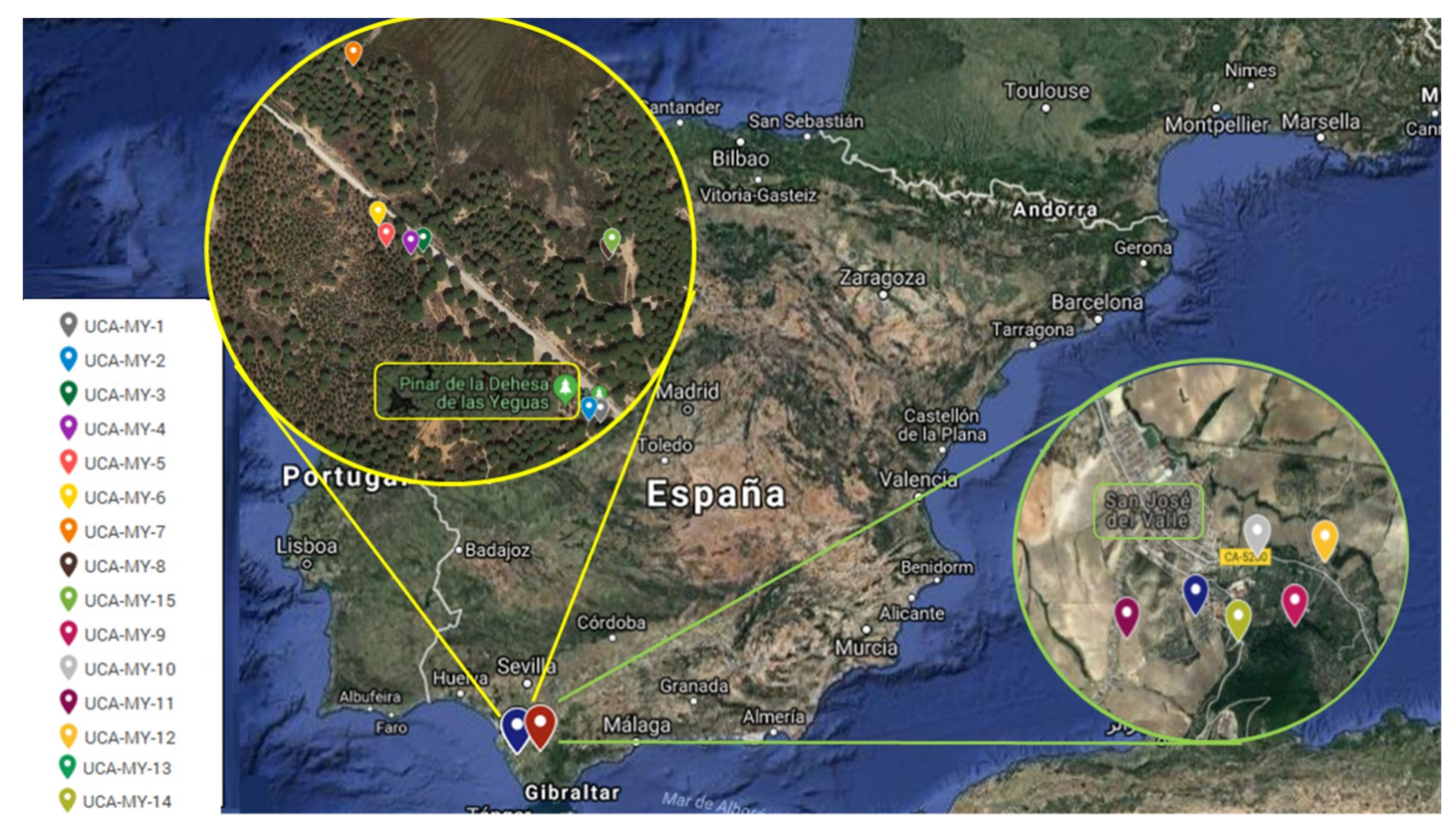
3.1. Plant Material and Study Area
3.2. Morphological Analysis
3.3. Chemical Analysis
3.4. Data Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| BBD | Box–Behnken design |
| CD | calyx diameter |
| CS | calyx shapes |
| CV | coefficient of variation |
| FL | fruit length |
| FS | fruit shapes |
| FWD | fruit width |
| FWG | fruit weight |
| HCA | hierarchical cluster analyses |
| HPLC | high-performance liquid chromatography |
| LL | leaf length |
| LW | leaf width |
| MAE | microwave-assisted extraction |
| PCA | principal component analysis |
| PL | peduncle length |
| Q-ToF-MS | quadrupole-time-of-flight mass spectrometry |
| RSM | response surface methodology |
| SL | seed length |
| SN | seed number |
| SWD | seed width |
| SWG | seed weight |
| TA | total anthocyanins |
| TPC | total phenolic compounds |
| TSWG | total seed weight |
| UHPLC | ultra-high performance liquid chromatography |
References
- Snow, N.; McFadden, J.; Evans, T.M.; Salywon, A.M.; Wojciechowski, M.F.; Wilson, P.G. Morphological and molecular evidence of polyphyly in Rhodomyrtus (Myrtaceae: Myrteae). Syst. Bot. 2011, 36, 390–404. [Google Scholar] [CrossRef]
- Mimica-Dukić, N.; Bugarin, D.; Grbović, S.; Mitić-Ćulafić, D.; Vuković-Gačić, B.; Orčić, D.; Jovin, E.; Couladis, M. Essential oil of Myrtus communis L. as a potential antioxidant and antimutagenic agents. Molecules 2010, 15, 2759–2770. [Google Scholar] [CrossRef]
- Berka-Zougali, B.; Ferhat, M.-A.; Hassani, A.; Chemat, F.; Allaf, K.S.; Berka-Zougali, B.; Ferhat, M.-A.; Hassani, A.; Chemat, F.; Allaf, K.S. Comparative study of essential oils extracted from Algerian Myrtus communis L. leaves using microwaves and hydrodistillation. Int. J. Mol. Sci. 2012, 13, 4673–4695. [Google Scholar] [CrossRef]
- Traveset, A.; Riera, N.; Mas, R.E. Ecology of fruit-colour polymorphism in Myrtus communis and differential effects of birds and mammals on seed germination and seedling growth. J. Ecol. 2001, 89, 749–760. [Google Scholar] [CrossRef]
- Bouzabata, A.; Casanova, J.; Bighelli, A.; Cavaleiro, C.; Salgueiro, L.; Tomi, F. The genus Myrtus L. in Algeria: Composition and biological aspects of essential oils from M. communis and M. nivellei: A Review. Chem. Biodivers. 2016, 13, 672–680. [Google Scholar] [CrossRef]
- Agrimonti, C.; Bianchi, R.; Bianchi, A.; Ballero, M.; Poli, F.; Marmiroli, N. Understanding biological conservation strategies: A molecular-genetic approach to the case of myrtle (Myrtus communis L.) in two Italian regions: Sardinia and Calabria. Conserv. Genet. 2007, 8, 385–396. [Google Scholar] [CrossRef]
- Aleksic, V.; Knezevic, P. Antimicrobial and antioxidative activity of extracts and essential oils of Myrtus communis L. Microbiol. Res. 2014, 169, 240–254. [Google Scholar] [CrossRef]
- Pereira, P.; Cebola, M.J.; Oliveira, M.C.; Bernardo-Gil, M.G. Supercritical fluid extraction vs. conventional extraction of myrtle leaves and berries: Comparison of antioxidant activity and identification of bioactive compounds. J. Supercrit. Fluids 2016, 113, 1–9. [Google Scholar] [CrossRef]
- Tuberoso, C.I.G.; Melis, M.; Angioni, A.; Pala, M.; Cabras, P. Myrtle hydroalcoholic extracts obtained from different selections of Myrtus communis L. Food Chem. 2007, 101, 806–811. [Google Scholar] [CrossRef]
- Bamba, B.; Shi, J.; Tranchant, C.; Xue, S.; Forney, C.; Lim, L.-T.; Bamba, B.S.; Shi, J.; Tranchant, C.; Xue, S.J.; et al. Influence of extraction conditions on ultrasound-assisted recovery of bioactive phenolics from Blueberry Pomace and their antioxidant activity. Molecules 2018, 23, 1685. [Google Scholar] [CrossRef]
- Yildirim, F.; San, B.; Yildirim, A.N.; Polat, M.; Ercişli, S. Mineral composition of leaves and fruit in some myrtle (Myrtus communis L.) genotypes. Erwerbs-Obstbau 2015, 57, 149–152. [Google Scholar] [CrossRef]
- Covarrubias-Cárdenas, A.; Martínez-Castillo, J.; Medina-Torres, N.; Ayora-Talavera, T.; Espinosa-Andrews, H.; García-Cruz, N.; Pacheco, N. Antioxidant capacity and UPLC-PDA ESI-MS phenolic profile of stevia rebaudiana dry powder extracts obtained by ultrasound assisted extraction. Agronomy 2018, 8, 170. [Google Scholar] [CrossRef]
- Aidi Wannes, W.; Mhamdi, B.; Sriti, J.; Ben Jemia, M.; Ouchikh, O.; Hamdaoui, G.; Kchouk, M.E.; Marzouk, B. Antioxidant activities of the essential oils and methanol extracts from myrtle (Myrtus communis var. italica L.) leaf, stem and flower. Food Chem. Toxicol. 2010, 48, 1362–1370. [Google Scholar] [CrossRef]
- Tuberoso, C.I.G.; Barra, A.; Cabras, P. Effect of different technological processes on the chemical composition of myrtle (Myrtus communis L.) alcoholic extracts. Eur. Food Res. Technol. 2008, 226, 801–808. [Google Scholar] [CrossRef]
- Briones-Labarca, V.; Plaza-Morales, M.; Giovagnoli-Vicuña, C.; Jamett, F. High hydrostatic pressure and ultrasound extractions of antioxidant compounds, sulforaphane and fatty acids from Chilean papaya (Vasconcellea pubescens) seeds: Effects of extraction conditions and methods. LWT Food Sci. Technol. 2015, 60, 525–534. [Google Scholar] [CrossRef]
- Dahmoune, F.; Nayak, B.; Moussi, K.; Remini, H.; Madani, K. Optimization of microwave-assisted extraction of polyphenols from Myrtus communis L. leaves. Food Chem. 2015, 166, 585–595. [Google Scholar] [CrossRef]
- Messaoud, C.; Boussaid, M. Myrtus communis berry color morphs: A comparative analysis of essential oils, fatty acids, phenolic compounds, and antioxidant activities. Chem. Biodivers. 2011, 8, 300–310. [Google Scholar] [CrossRef]
- Fadda, A.; Mulas, M. Chemical changes during myrtle (Myrtus communis L.) fruit development and ripening. Sci. Hortic. 2010, 125, 477–485. [Google Scholar] [CrossRef]
- Messaoud, C.; Zaouali, Y.; Salah, A.B.; Khoudja, M.L.; Boussaid, M. Myrtus communis in Tunisia: Variability of the essential oil composition in natural populations. Flavour Fragr. J. 2005, 20, 577–582. [Google Scholar] [CrossRef]
- Fadil, M.; Farah, A.; Ihssane, B.; Haloui, T.; Lebrazi, S.; Zghari, B.; Rachiq, S. Chemometric investigation of light-shade effects on essential oil yield and morphology of Moroccan Myrtus communis L. SpringerPlus 2016, 5, 1062. [Google Scholar] [CrossRef]
- Aksic, M.F.; Tosti, T.; Sredojevic, M.; Milivojevic, J.; Meland, M.; Natic, M. Comparison of sugar profile between leaves and fruits of blueberry and strawberry cultivars grown in organic and integrated production system. Plants 2019, 8, 2015. [Google Scholar] [CrossRef]
- Medina-Torres, N.; Ayora-Talavera, T.; Espinosa-Andrews, H.; Sánchez-Contreras, A.; Pacheco, N. Ultrasound assisted extraction for the recovery of phenolic compounds from vegetable sources. Agronomy 2017, 7, 47. [Google Scholar] [CrossRef]
- Spigno, G.; De Faveri, D.M.M. Microwave-assisted extraction of tea phenols: A phenomenological study. J. Food Eng. 2009, 93, 210–217. [Google Scholar] [CrossRef]
- Espada-Bellido, E.; Ferreiro-González, M.; Carrera, C.; Palma, M.; Barroso, C.G.; Barbero, G.F. Optimization of the ultrasound-assisted extraction of anthocyanins and total phenolic compounds in mulberry (Morus nigra) pulp. Food Chem. 2017, 219, 23–32. [Google Scholar] [CrossRef]
- González de Peredo, A.V.; Vázquez-Espinosa, M.; Espada-Bellido, E.; Jiménez-Cantizano, A.; Ferreiro-González, M.; Amores-Arrocha, A.; Palma, M.; Barroso, C.G.; Barbero, G. Development of new analytical microwave-assisted extraction methods for bioactive compounds from myrtle (myrtus communis L.). Molecules 2018, 23, 2992. [Google Scholar] [CrossRef]
- Wahid, N.; Chkichekh, A.; Bakry, M. Morphological traits and essential oil yield variation of three myrtus communis l. populations: Implication for domestication. J. Agric. Food Sci. 2016, 4, 199–207. [Google Scholar]
- Aliaño-González, M.; Ferreiro-González, M.; Barbero, G.; Palma, M.; Barroso, C. Application of headspace gas chromatography-ion mobility spectrometry for the determination of ignitable liquids from fire debris. Separations 2018, 5, 41. [Google Scholar] [CrossRef]
- Héberger, K. Chemoinformatics—multivariate mathematical–statistical methods for data evaluation. Med. Appl. Mass Spectrom. 2008, 141–169. [Google Scholar] [CrossRef]
- Piñeiro, Z.; Marrufo-Curtido, A.; Vela, C.; Palma, M. Microwave-assisted extraction of stilbenes from woody vine material. Food Bioprod. Process 2017, 103, 18–26. [Google Scholar] [CrossRef]
- Linic, I.; Samec, D.; Grúz, J.; Bok, V.V.; Strnad, M.; Salopek-Sondi, B. Involvement of phenolic acids in short-term adaptation to salinity stress is species-specific among brassicaceae. Plants 2019, 8, 155. [Google Scholar] [CrossRef]
- Hosseini, A.S.; Akramian, M.; Khadivi, A.; Salehi-Arjmand, H. Phenotypic and chemical variation of black mulberry (Morus nigra) genotypes. Ind. Crops Prod. 2018, 117, 260–271. [Google Scholar] [CrossRef]
- Aliaño-González, M.J.; Ferreiro-González, M.; Espada-Bellido, E.; Palma, M.; Barbero, G.F. A screening method based on headspace-ion mobility spectrometry to identify adulterated honey. Sensors 2019, 19, 1621. [Google Scholar] [CrossRef]
- Aliaño-González, M.J.; Ferreiro-González, M.; Barbero, G.F.; Palma, M. Novel method based on ion mobility spectrometry sum spectrum for the characterization of ignitable liquids in fire debris. Talanta 2019, 199, 189–194. [Google Scholar] [CrossRef]
- Ferrari, S.M.; Fallahi, P.; Politti, U.; Materazzi, G.; Baldini, E.; Ulisse, S. A user’s guide to principle components. Clin. Cancer Res. 2012, 18, 1–100. [Google Scholar] [CrossRef]
- Farzaneh, V.; Carvalho, I.S. Modelling of microwave assisted extraction (MAE) of anthocyanins (TMA). J. Appl. Res. Med. Aromat. Plants 2017, 6, 92–100. [Google Scholar] [CrossRef]
- González-Varo, J.P.; Albaladejo, R.G.; Aparicio, A. Mating patterns and spatial distribution of conspecific neighbours in the Mediterranean shrub Myrtus communis (Myrtaceae). Plant Ecol. 2009, 203, 207–215. [Google Scholar] [CrossRef]
- Mulas, M.; Cani, M.R. Germplasm evaluation of spontaneous myrtle (Myrtus communis L.) for cultivar selection and crop development. J. Herbs Spices Med. Plants 1999, 17, 31–49. [Google Scholar] [CrossRef]
- Melito, S.; La Bella, S.; Martinelli, F.; Cammalleri, I.; Tuttolomondo, T.; Leto, C.; Fadda, A.; Molinu, M.G.; Mulas, M. Morphological, chemical, and genetic diversity of wild myrtle (Myrtus communis L.) populations in Sicily. Turk. J. Agric. For. 2016, 40, 249–261. [Google Scholar] [CrossRef]
- Chaikovskaia, M.J. Effect of external and internal factors on the reproduction of geese. Zh. Obshch. Biol. 2012, 10, 332–347. [Google Scholar]
- Szakiel, A.; Pączkowski, C.; Henry, M. Influence of environmental abiotic factors on the content of saponins in plants. Phytochem. Rev. 2011, 10, 471–491. [Google Scholar] [CrossRef]
- Farzaneh, V.; Gominho, J.; Pereira, H.; Carvalho, I.S. Screening of the antioxidant and enzyme inhibition potentials of portuguese Pimpinella anisum L. seeds by GC-MS. Food Anal. Methods 2018, 11, 2645–2656. [Google Scholar] [CrossRef]
- González de Peredo, A.V.; Vázquez-Espinosa, M.; Espada-Bellido, E.; Ferreiro-González, M.; Amores-Arrocha, A.; Palma, M.; Barroso, C.G.; Barbero, G.; Jiménez-Cantizano, A. Alternative ultrasound-assisted method for the extraction of the bioactive compounds present in myrtle (Myrtus communis L.). Molecules 2019, 24, 882. [Google Scholar] [CrossRef]
- Espada-Bellido, E.; Ferreiro-González, M.; Barbero, G.F.; Carrera, C.; Palma, M.; Barroso, C.G. Alternative extraction method of bioactive compounds from mulberry (Morus nigra L.) pulp using pressurized-liquid extraction. Food Anal. Methods 2018, 11, 2384–2395. [Google Scholar] [CrossRef]
- Vázquez-Espinosa, M.; Espada-Bellido, E.; González de Peredo, A.V.; Ferreiro-González, M.; Carreira, C.; Palma, M.; Barroso, C.G.; Barbero, G.F. Optimization of microwave-assisted extraction for the recovery of bioactive compounds from the chilean superfruit (Aristotelia chilensis (Mol.) Stuntz). Agronomy 2018, 8, 240. [Google Scholar] [CrossRef]
- Aryal, S.; Baniya, M.K.; Danekhu, K.; Kunwar, P.; Gurung, R.; Koirala, N. Total Phenolic content, flavonoid content and antioxidant potential of wild vegetables from Western Nepal. Plants 2019, 8, 96. [Google Scholar] [CrossRef]
| Ecotype | Measures | FL (mm) | FWD (mm) | FWG (g) | PL (mm) | CD (mm) | SN | SL (mm) | SWD (mm) | SWG (g) | TSWG (g) | LL (mm) | LW (mm) | FS | CS |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ecotype 1 | Mean | 11.92 | 9.02 | 0.4377 | 17.29 | 4.77 | 8.28 | 4.00 | 3.04 | 0.0164 | 0.1241 | 36.63 | 17.25 | 1 | 2 |
| CV | 10.03 | 6.82 | 15.43 | 17.06 | 5.78 | 40.29 | 10.65 | 12.99 | 27.75 | 22.49 | 10.24 | 13.17 | |||
| Ecotype 2 | Mean | 13.40 | 9.88 | 0.6835 | 19.58 | 4.68 | 7.32 | 4.33 | 3.28 | 0.0213 | 0.1384 | 28.93 | 12.70 | 2 | 2 |
| CV | 12.39 | 9.32 | 22.87 | 17.93 | 8.99 | 36.51 | 8.96 | 16.13 | 17.77 | 38.86 | 10.27 | 33.48 | |||
| Ecotype 3 | Mean | 9.40 | 7.16 | 0.2822 | 20.78 | 4.86 | 4.28 | 4.07 | 3.08 | 0.0196 | 0.0770 | 38.17 | 14.90 | 1 | 1 |
| CV | 8.29 | 8.61 | 21.39 | 17.88 | 4.83 | 41.31 | 7.02 | 11.34 | 14.75 | 34.31 | 12.02 | 10.93 | |||
| Ecotype 4 | Mean | 10.95 | 10.68 | 0.6500 | 21.12 | 4.60 | 6.6 | 4.02 | 2.92 | 0.0204 | 0.1179 | 45.19 | 18.45 | 2 | 3 |
| CV | 16.00 | 8.44 | 25.14 | 20.22 | 10.63 | 61.08 | 8.36 | 14.51 | 17.39 | 49.02 | 13.47 | 9.68 | |||
| Ecotype 5 | Mean | 12.31 | 8.54 | 0.4515 | 19.50 | 5.12 | 3.32 | 3.74 | 2.99 | 0.0187 | 0.0628 | 46.21 | 18.00 | 3 | 2 |
| CV | 5.87 | 8.61 | 18.71 | 19.29 | 10.98 | 44.10 | 8.17 | 12.01 | 17.71 | 38.46 | 9.13 | 11.28 | |||
| Ecotype 6 | Mean | 14.49 | 8.91 | 0.5162 | 18.29 | 3.87 | 6.96 | 4.67 | 3.02 | 0.0240 | 0.1486 | 38.30 | 13.17 | 2 | 1 |
| CV | 7.57 | 5.46 | 12.80 | 15.70 | 15.51 | 31.99 | 8.45 | 13.25 | 22.43 | 22.08 | 13.49 | 13.50 | |||
| Ecotype 7 | Mean | 8.80 | 6.96 | 0.1998 | 21.29 | 4.46 | 2.96 | 3.36 | 2.78 | 0.0167 | 0.0390 | 36.45 | 14.44 | 2 | 1 |
| CV | 12.08 | 13.43 | 36.19 | 11.76 | 10.28 | 37.11 | 8.44 | 13.26 | 103.16 | 37.34 | 13.70 | 29.27 | |||
| Ecotype 8 | Mean | 11.79 | 11.29 | 0.5942 | 16.06 | 3.73 | 10.08 | 3.40 | 2.50 | 0.0106 | 0.1022 | 33.36 | 12.27 | 3 | 3 |
| CV | 5.39 | 17.54 | 15.31 | 24.48 | 18.80 | 29.75 | 20.43 | 13.08 | 17.18 | 25.17 | 11.26 | 15.47 | |||
| Ecotype 9 | Mean | 12.6 | 6.51 | 0.2334 | 20.6 | 4.24 | 2.28 | 3.95 | 3.13 | 0.0184 | 0.04 | 32.65 | 13.94 | 3 | 2 |
| CV | 14.12 | 15.89 | 34.01 | 18.23 | 9.84 | 51.46 | 14.50 | 14.58 | 19.12 | 45.92 | 7.34 | 7.48 | |||
| Ecotype 10 | Mean | 16.22 | 7.52 | 0.4396 | 17.54 | 4.36 | 3.28 | 4.10 | 3.33 | 0.0201 | 0.0618 | 30.92 | 11.82 | 3 | 1 |
| CV | 6.67 | 14.65 | 18.29 | 17.23 | 8.53 | 39.87 | 11.99 | 12.85 | 15.00 | 38.56 | 20.40 | 18.15 | |||
| Ecotype 11 | Mean | 13.86 | 7.19 | 0.3189 | 15.54 | 4.49 | 4.00 | 3.96 | 3.67 | 0.0206 | 0.0733 | 33.38 | 11.92 | 1 | 2 |
| CV | 5.38 | 10.21 | 17.61 | 16.11 | 7.81 | 40.16 | 10.12 | 11.32 | 14.21 | 30.21 | 18.76 | 16.52 | |||
| Ecotype 12 | Mean | 16.24 | 8.27 | 0.4798 | 20.31 | 4.42 | 3.00 | 3.18 | 4.3 | 0.0212 | 0.0563 | 28.25 | 10.55 | 1 | 1 |
| CV | 6.10 | 6.10 | 12.31 | 12.31 | 6.78 | 38.76 | 11.12 | 13.56 | 17.21 | 29.87 | 16.54 | 14.32 | |||
| Ecotype 13 | Mean | 12.98 | 6.18 | 0.2562 | 17.58 | 4.3 | 2.00 | 4.75 | 4.47 | 0.021 | 0.0381 | 30.44 | 13.6 | 1 | 2 |
| CV | 5.41 | 1.23 | 10.29 | 11.23 | 7.56 | 40.97 | 8.76 | 13.21 | 16.79 | 29.01 | 16.70 | 15.20 | |||
| Ecotype 14 | Mean | 15.03 | 6.74 | 0.4252 | 19.12 | 4.97 | 4.00 | 4.47 | 3.9 | 0.0204 | 0.0704 | 31.86 | 11.58 | 1 | 1 |
| CV | 6.35 | 2.43 | 15.16 | 14.56 | 6.78 | 38.72 | 8.11 | 14.21 | 16.72 | 19.67 | 14.32 | 13.29 | |||
| Mean CV | 8.69 | 9.20 | 19.68 | 16.71 | 9.51 | 40.86 | 10.36 | 13.31 | 24.9 | 32.93 | 13.40 | 15.84 | |||
| FL | FWD | FWG | PL | CD | SN | SL | SWD | SWG | TSWG | LL | LW | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FL | 1.000 | |||||||||||
| FWD | 0.065 | 1.000 | ||||||||||
| FWG | 0.442 | 0.874 * | 1.000 | |||||||||
| PL | −0.116 | −0.368 | −0.183 | 1.000 | ||||||||
| CD | −0.294 | −0.233 | −0.167 | 0.311 | 1.000 | |||||||
| SN | 0.129 | 0.845 * | 0.738 * | −0.339 | −0.368 | 1.000 | ||||||
| SL | 0.562 | 0.0019 | 0.376 | 0.058 | −0.019 | 0.115 | 1.000 | |||||
| SWD | 0.558 | −0.432 | 0.003 | 0.256 | 0.388 | −0.371 | 0.689 * | 1.000 | ||||
| SWG | 0.395 | −0.253 | 0.156 | 0.390 | 0.218 | −0.285 | 0.849 * | 0.730 * | 1.000 | |||
| TSWG | 0.320 | 0.695 * | 0.814 * | −0.149 | −0.183 | 0.819 * | 0.616 | 0.060 | 0.305 | 1.000 | ||
| LL | −0.316 | 0.138 | 0.048 | 0.288 | 0.415 | −0.090 | −0.088 | −0.272 | 0.143 | 0.017 | 1.000 | |
| LW | −0.263 | 0.093 | 0.051 | 0.476 | 0.612 | −0.0005 | −0.085 | −0.048 | 0.088 | 0.078 | 0.821 * | 1.000 |
| TA | TPC | |
|---|---|---|
| TA | 1 | |
| TPC | 0.632 * | 1 |
| FWD | 0.679 * | 0.509 |
| FL | −0.541 * | −0.307 |
| FWG | 0.526 | 0.441 |
| PL | 0.272 | 0.043 |
| CD | 0.150 | 0.078 |
| SN | 0.598 * | 0.562 * |
| SL | −0.109 | −0.039 |
| SWD | −0.467 | −0.493 |
| SWG | −0.217 | −0.225 |
| TSWG | 0.562 * | 0.545 * |
| LL | 0.428 | 0.051 |
| LW | 0.458 | 0.159 |
| PC1 | PC2 | PC3 | PC4 | |
|---|---|---|---|---|
| FWD | 0.332545 | 0.261105 | 0.016976 | −0.250766 |
| FL | −0.238809 | 0.374353 | 0.0722094 | −0.23456 |
| FWG | 0.21349 | 0.378674 | 0.218042 | −0.354937 |
| PL | 0.0300118 | −0.332878 | 0.216556 | −0.524048 |
| CD | −0.041398 | −0.268718 | 0.38474 | −0.239024 |
| SN | 0.313662 | 0.312652 | −0.0277603 | 0.0991843 |
| SL | −0.113674 | 0.159997 | 0.440774 | 0.583266 |
| SWD | −0.344552 | 0.0811821 | 0.103164 | −0.126136 |
| SWG | −0.204153 | 0.0776921 | 0.515538 | −0.0716526 |
| TSWG | 0.249142 | 0.340977 | 0.292583 | 0.074471 |
| LL | 0.252782 | −0.265527 | 0.291788 | 0.1241 |
| LW | 0.234908 | −0.305274 | 0.306359 | 0.158535 |
| PC1 | PC2 | PC3 | |
|---|---|---|---|
| TPC | 0.276762 | −0.215631 | 0.345485 |
| Delphinidin 3,5-O-diglucoside | 0.357497 | −0.158785 | −0.0858731 |
| Delphinidin 3-O-glucoside | 0.400637 | 0.0174325 | 0.0435637 |
| Cyanidin 3-O-galactoside | 0.230934 | 0.313119 | −0.289586 |
| Cyanidin 3-O-glucoside | 0.340067 | −0.0419079 | 0.366281 |
| Cyanidin 3-O-arabinoside | 0.026038 | 0.359961 | 0.483078 |
| Petunidin 3-O-glucoside | 0.330595 | −0.247933 | −0.143641 |
| Delphinidin 3-O-arabinoside | 0.102898 | 0.475758 | 0.0623436 |
| Peonidin 3-O-glucoside | 0.336837 | 0.0944947 | 0.286337 |
| Malvidin 3-O-glucoside | 0.157008 | −0.280399 | −0.360425 |
| Petunidin 3-O-arabinoside | 0.225104 | 0.385053 | −0.246464 |
| Malvidin 3-O-arabinoside | 0.106503 | 0.403267 | −0.30904 |
| TA | 0.38135 | −0.111159 | −0.165277 |
| Place | Altitude (m Above Sea Level) | Tmax (°C) 1 | Tmin (°C) 2 | Mean Annual Precipitation (mm) |
|---|---|---|---|---|
| Puerto Real | 8 | 30 | 9 | 609 |
| San José del Valle | 141 | 34 | 6 | 720 |
| Morphological Traits | Code | Unit of Measure | Method of Measurement |
|---|---|---|---|
| Fruit traits | |||
| Fruit length | FL | mm | Caliper |
| Fruit width | FWD | mm | Caliper |
| Fruit weight | FWG | g | Precision balance |
| Peduncle length | PL | mm | Caliper |
| Fruit shapes | FS | - | By observation: spherical, elliptic, obovate, pyriform, turbinate |
| Calyx shapes | CS | - | By observation: closed, partially closed, opened |
| Calyx diameter | CD | mm | Caliper |
| Seed traits | |||
| Seed number | SN | - | Counter |
| Seed length | SL | mm | Caliper |
| Seed width | SWG | mm | Caliper |
| Seed weight | SWD | g | Precision balance |
| Total seed weight | TSWG | g | Precision balance |
| Leaves traits | |||
| Leaf length | LL | mm | Caliper |
| Leaf width | LW | mm | Caliper |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
González-de-Peredo, A.V.; Vázquez-Espinosa, M.; Espada-Bellido, E.; Ferreiro-González, M.; Amores-Arrocha, A.; Palma, M.; Barbero, G.F.; Jiménez-Cantizano, A. Discrimination of Myrtle Ecotypes from Different Geographic Areas According to Their Morphological Characteristics and Anthocyanins Composition. Plants 2019, 8, 328. https://doi.org/10.3390/plants8090328
González-de-Peredo AV, Vázquez-Espinosa M, Espada-Bellido E, Ferreiro-González M, Amores-Arrocha A, Palma M, Barbero GF, Jiménez-Cantizano A. Discrimination of Myrtle Ecotypes from Different Geographic Areas According to Their Morphological Characteristics and Anthocyanins Composition. Plants. 2019; 8(9):328. https://doi.org/10.3390/plants8090328
Chicago/Turabian StyleGonzález-de-Peredo, Ana V., Mercedes Vázquez-Espinosa, Estrella Espada-Bellido, Marta Ferreiro-González, Antonio Amores-Arrocha, Miguel Palma, Gerardo F. Barbero, and Ana Jiménez-Cantizano. 2019. "Discrimination of Myrtle Ecotypes from Different Geographic Areas According to Their Morphological Characteristics and Anthocyanins Composition" Plants 8, no. 9: 328. https://doi.org/10.3390/plants8090328
APA StyleGonzález-de-Peredo, A. V., Vázquez-Espinosa, M., Espada-Bellido, E., Ferreiro-González, M., Amores-Arrocha, A., Palma, M., Barbero, G. F., & Jiménez-Cantizano, A. (2019). Discrimination of Myrtle Ecotypes from Different Geographic Areas According to Their Morphological Characteristics and Anthocyanins Composition. Plants, 8(9), 328. https://doi.org/10.3390/plants8090328