Comparison of Growth Rate and Nutrient Content of Five Microalgae Species Cultivated in Greenhouses
Abstract
1. Introduction
2. Materials and Methods
2.1. Microalgae Cultivation
2.2. Measurements
2.3. Nutrient Composition Analyses
2.4. Statistical Analysis
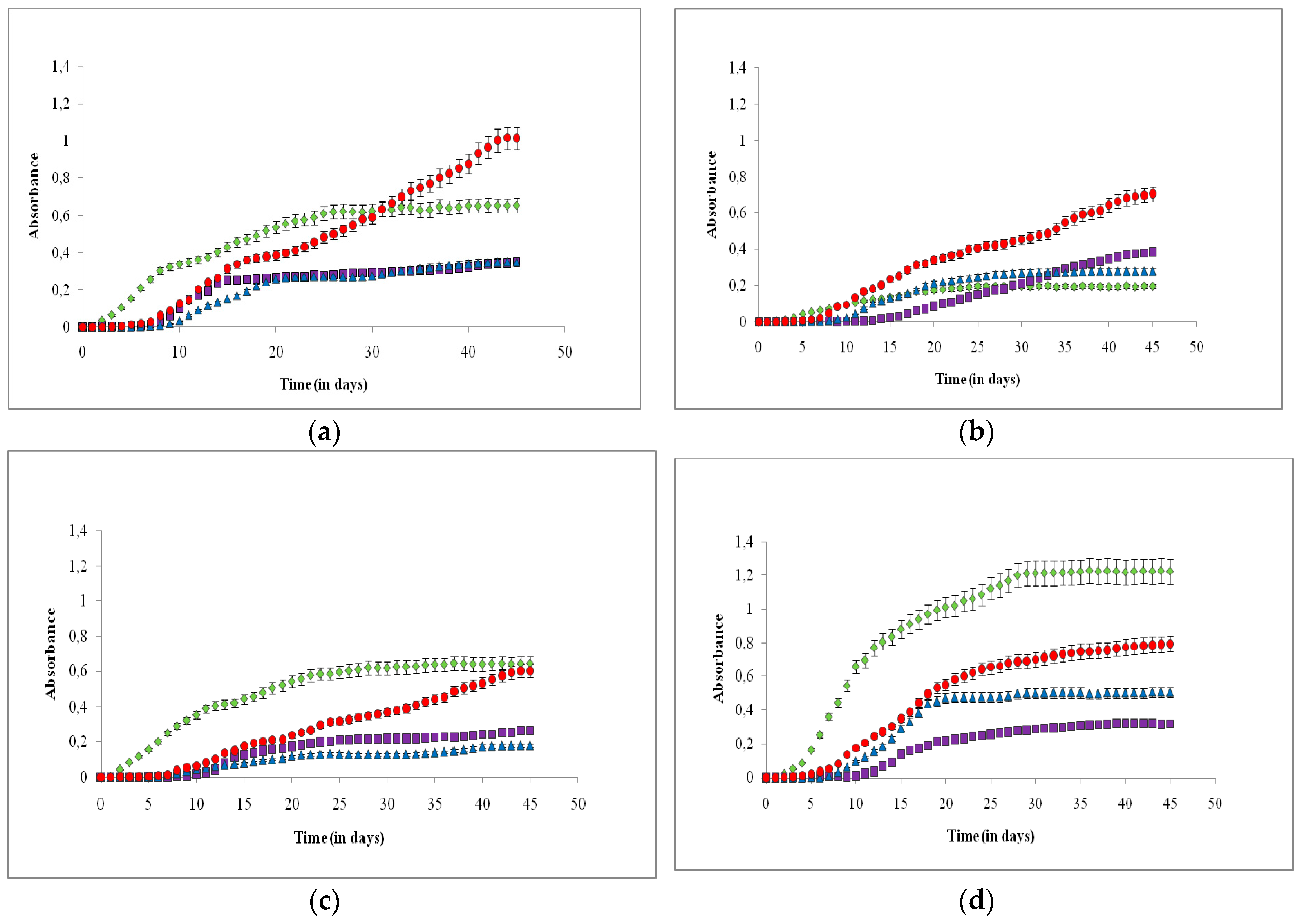
3. Results
4. Discussion
4.1. Effects on Growth Rate
4.2. Comparison of Macronutrient Composition
4.3. Comparison of Total Phenolic Content
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pulz, O.; Gross, W. Valuable products from biotechnology of microalgae. Appl. Microbiol. Biotechnol. 2004, 65, 635–648. [Google Scholar] [CrossRef]
- Brown, M.R.; Jeffrey, S.W.; Volkman, J.K.; Dunstan, G.A. Nutritional properties of microalgae for mariculture. Aquaculture 1997, 151, 315–331. [Google Scholar] [CrossRef]
- Brown, M.R.; Mular, M.; Miller, I.; Trenerry, C.; Farmer, C. The vitamin content of microalgae used in aquaculture. J. Appl. Phycol. 1999, 11, 247–255. [Google Scholar] [CrossRef]
- Lubián, L.M.; Montero, O.; Moreno-Garrido, I.; Huertas, I.E.; Sobrino, C.; González-Del Valle, M.; Parés, G. Nannochloropsis (Eustigmatophyceae) as source of commercially valuable pigments. J. Appl. Phycol. 2000, 12, 249–255. [Google Scholar] [CrossRef]
- Tibbetts, S.M.; Milley, J.E.; Lall, S.P. Chemical composition and nutritional properties of freshwater and marine microalgal biomass cultured in photobioreactors. J. Appl. Phycol. 2015, 27, 1109–1119. [Google Scholar] [CrossRef]
- Hajimahmoodi, M.; Faramarzi, M.A.; Mohammadi, N.; Soltani, N.; Oveisi, M.R.; Nafissi-Varcheh, N. Evaluation of antioxidant properties and total phenolic contents of some strains of microalgae. J. Appl. Phycol. 2010, 22, 43–50. [Google Scholar] [CrossRef]
- Machu, L.; Misurcova, L.; Ambrozova, J.V.; Orsavova, J.; Mlcek, J.; Sochor, J.; Jurikova, T. Phenolic content and antioxidant capacity in algal food products. Molecules 2015, 20, 1118–1133. [Google Scholar] [CrossRef]
- Papapolymerou, G.; Karayannis, V.; Besios, A.; Riga, A.; Gougoulias, N.; Spiliotis, X. Scaling-up sustainable Chlorella vulgaris microalgal biomass cultivation from laboratory to pilot-plant photobioreactor, towards biofuel. Glob. Nest J. 2019, 21, 37–42. [Google Scholar]
- Li, Y.; Zhou, W.; Hu, B.; Min, M.; Chen, P.; Ruan, R.R. Effect of light intensity on algal biomass accumulation and biodiesel production for mixotrophic strains Chlorella kessleri and Chlorella protothecoide cultivated in highly concentrated municipal wastewater. Biotechnol. Bioeng. 2012, 109, 2222–2229. [Google Scholar] [CrossRef]
- López, C.V.; Cerón, M.C.; Acién, F.G.; Segovia, C.; Chisti, Y.; Fernández, J.M. Protein measurements of microalgal and cyanobacterial biomass. Bioresour. Technol. 2010, 101, 7587–7591. [Google Scholar] [CrossRef]
- Wijffels, R.H.; Barbosa, M.J.; Eppink, M.H.M. Microalgae for the production of bulk chemicals and biofuels. Biofuels Bioprod. Biorefining 2010, 4, 287–295. [Google Scholar] [CrossRef]
- Zhu, S.; Wang, Y.; Shang, C.; Wang, Z.; Xu, J.; Yuan, Z. Characterization of lipid and fatty acids composition of Chlorella zofingiensis in response to nitrogen starvation. J. Biosci. Bioeng. 2015, 120, 205–209. [Google Scholar] [CrossRef] [PubMed]
- Metsoviti, M.N.; Katsoulas, N.; Karapanagiotidis, I.T.; Papapolymerou, G. Effect of nitrogen concentration, two-stage and prolonged cultivation on growth rate, lipid and protein content of Chlorella vulgaris. J. Chem. Technol. Biotechnol. 2019, 94, 1466–1473. [Google Scholar] [CrossRef]
- Ruangsomboon, S. Effect of light, nutrient, cultivation time and salinity on lipid production of newly isolated strain of the green microalga, Botryococcus braunii KMITL 2. Bioresour. Technol. 2012, 109, 261–265. [Google Scholar] [CrossRef] [PubMed]
- Sukenik, A.; Carmeli, Y.; Berner, T. Regulation of fatty acid composition by irradiance level in the eustigmatophyte Nannochloropsis sp. 1. J. Phycol. 1989, 25, 686–692. [Google Scholar] [CrossRef]
- George, B.; Pancha, I.; Dessai, C.; Chokshi, K.; Paliwal, C.; Ghosh, T.; Mishra, S. Effects of different media composition, light intensity and photoperiod on morphology and physiology of freshwater microalgae Ankistrodesmus falcatus—A potential strain for bio-fuel production. Bioresour. Technol. 2014, 171, 367–374. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.J.; Lee, Y.K.; Chao, T.M. Effects of temperature and growth phase on lipid and biochemical composition of Isochrysis galbana TK1. J. Phycol. 1997, 9, 451–457. [Google Scholar] [CrossRef]
- Vitova, M.; Bisova, K.; Kawano, S.; Zachleder, V. Accumulation of energy reserves in algae: From cell cycles to biotechnological applications. Biotechnol. Adv. 2015, 33, 1204–1218. [Google Scholar] [CrossRef] [PubMed]
- Converti, A.; Casazza, A.A.; Ortiz, E.Y.; Perego, P.; Borghi, M.D. Effect of temperature and nitrogen concentration on the growth and lipid content of Nannochloropsis oculata and Chlorella vulgaris for biodiesel production. Chem. Eng. Process. 2009, 48, 1146–1151. [Google Scholar] [CrossRef]
- Renaud, S.M.; Thinh, L.V.; Lambrinidis, G.; Parry, D.L. Effect of temperature on growth, chemical composition and fatty acid composition of tropical Australian microalgae grown in batch cultures. Aquaculture 2002, 211, 195–214. [Google Scholar] [CrossRef]
- Chen, C.Y.; Yeh, K.L.; Aisyah, R.; Lee, D.J.; Chang, J.S. Cultivation, photobioreactor design and harvesting of microalgae for biodiesel production: A critical review. Bioresour. Technol. 2011, 102, 71–81. [Google Scholar] [CrossRef]
- Perez, L.; Salgueiro, J.L.; Maceiras, R.; Cancela, A.; Sanchez, A. An effective method for harvesting of marine microalgae: pH induced flocculation. Biomass Bioenergy 2017, 97, 20–26. [Google Scholar] [CrossRef]
- Safi, C.; Zebib, B.; Merah, O.; Pontalier, P.Y.; Vaca-Garcia, C. Morphology, composition, production, processing and applications of Chlorella vulgaris: A review. Renew. Sustain. Energy Rev. 2014, 35, 265–278. [Google Scholar] [CrossRef]
- AOAC. Association of Official Analytical Chemists. Official Methods of Analysis of the Association of Official Analytical Chemists International, 16th ed.; AOAC: Arlington, VA, USA, 1995. [Google Scholar]
- Folch, J.; Lees, M.; Sloane-Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar]
- Ryckebosch, Ε.; Muylaert, Κ.; Foubert, Ι. Optimization of an analytical procedure for extraction of lipids from microalgae. J. Am. Oil Chem. Soc. 2012, 89, 189–198. [Google Scholar] [CrossRef]
- Singleton, V.L.; Rossi, S.A. Colorimetry of total phenolics with phosphomolibdic-phosphotungestic acid reagents. J. Enol. Vitic. 1965, 16, 144. [Google Scholar]
- Dean, A.; Sigee, D.; Estrada, B.; Pittman, J. Using FTIR spectroscopy for rapid determination of lipid accumulation in response to nitrogen limitation in freshwater microalgae. Bioresour. Technol. 2010, 101, 4499–4507. [Google Scholar] [CrossRef]
- Vasileva, I.; Marinova, G.; Gigova, L. Effect of nitrogen source on the growth and biochemical composition of a new Bulgarian isolate of Scenedesmus sp. J. BioSci. Biotechnol. 2015, 125–129. [Google Scholar]
- Ördög, V.; Stirk, W.A.; Bálint, P.; Aremu, A.O.; Okem, A.; Lovász, C.; Molnár, Z.; Staden, J. Effect of temperature and nitrogen concentration on lipid productivity and fatty acid composition in three Chlorella strains. Algal Res. 2015, 16, 141–149. [Google Scholar] [CrossRef]
- Dayananda, C.; Sarada, R.; Usha, R.M.; Shamala, T.R.; Ravishankar, G.A. Autotrophic cultivation of Botryococcus braunii for the production of hydrocarbons and exopolysaccharides in various media. Biomass Bioenergy 2007, 31, 87–93. [Google Scholar] [CrossRef]
- Kitaya, Y.; Azuma, H.; Kiyota, M. Effects of temperature, CO2/O2 concentrations and light intensity on cellular multiplication of microalgae, Euglena gracilis. Adv. Space Res. 2005, 35, 1584–1588. [Google Scholar] [CrossRef]
- Singh, S.P.; Singh, P. Effect of temperature and light on the growth of algae species: A review. Renew. Sustain. Energy Rev. 2015, 50, 431–444. [Google Scholar] [CrossRef]
- Daliry, S.; Hallajisani, A.; Mohammadi, R.J.; Nouri, H.; Golzary, A. Investigation of optimal condition for Chlorella vulgaris microalgae growth. Glob. J. Environ. Sci. Manag. 2017, 3, 217–230. [Google Scholar] [CrossRef]
- Cheirsilp, B.; Torpee, S. Enhanced growth and lipid production of microalgae under mixotrophic culture condition: Effect of light intensity, glucose concentration and fed-batch cultivation. Bioresour. Technol. 2012, 110, 510–516. [Google Scholar] [CrossRef]
- Khoeyi, Z.A.; Seyfabadi, J.; Ramezanpour, Z. Effect of light intensity and photoperiod on biomass and fatty acid composition of the microalgae, Chlorella vulgaris. Aquac. Int. 2010, 20, 41–49. [Google Scholar] [CrossRef]
- Venkata, M.S.; Devi, M.P. Salinity stress induced lipid synthesis to harness biodiesel during dual mode cultivation of mixotrophic microalgae. Bioresour. Technol. 2014, 165, 288–294. [Google Scholar] [CrossRef]
- Sibi, G.; Shetty, V.; Mokashi, K. Enhanced lipid productivity approaches in microalgae as an alternate for fossil fuels—A review. J. Energy Inst. 2016, 89, 330–334. [Google Scholar] [CrossRef]
- Gigova, L.; Ivanova, N.; Gacheva, G.; Andreeva, R.; Furnadzhieva, S. Response of Trachydiscus minutus (Xanthophyceae) to temperature and light. J. Phycol. 2012, 48, 85–93. [Google Scholar] [CrossRef]
- Liu, J.; Yuan, C.; Hu, G.; Li, F. Effects of light intensity on the growth and lipid accumulation of microalga Scenedesmus sp. 11-1 under nitrogen limitation. Appl. Biochem. Biotechnol. 2012, 166, 2127–2137. [Google Scholar] [CrossRef]
- Solovchenko, A.E.; Khozin-Goldberg, I.; Didi-Cohen, S.; Cohen, Z.; Merzlyak, M.N. Effects of light intensity and nitrogen starvation on growth, total fatty acids and arachidonic acid in the green microalga Parietochloris incisa. J. Appl. Phycol. 2008, 20, 245–251. [Google Scholar] [CrossRef]
- He, Q.; Yang, H.; Wu, L.; Hu, C. Effect of light intensity on physiological changes, carbon allocation and neutral lipid accumulation in oleaginous microalgae. Bioresour. Technol. 2015, 191, 219–228. [Google Scholar] [CrossRef]
- Seyfabad, J.; Ramezanpour, Z.; Khoeyi, Z.A. Protein, fatty acid, and pigment content of Chlorella vulgaris under different light regimes. J. Appl. Phycol. 2011, 23, 721–726. [Google Scholar] [CrossRef]
- Sharma, R.; Singh, G.P.; Sharma, V.K. Effects of culture conditions on growth and biochemical profile of Chlorella vulgaris. J. Plant. Pathol. Microbiol. 2012, 3, 5. [Google Scholar] [CrossRef]
- Song, X.; Linglin, W.; Aifen, L.; Min, S.; Chengwu, Z. Effects of nutrients and light intensity on the growth and biochemical composition of a marine microalga Odontella aurita. Chin. J. Oceanol. Limnol. 2013, 31, 1163–1173. [Google Scholar] [CrossRef]
- Maadane, A.; Merghoub, N.; Ainane, T.; Arroussi, H.E.; Benhima, R.; Amzazi, S.; Bakri, Y.; Wahby, I. Antioxidant activity of some Moroccan marine microalgae: Pufaprofiles, carotenoids and phenolic content. J. Biotechnol. 2015, 215, 13–19. [Google Scholar] [CrossRef]
- Goiris, K.; Muylaert, K.; Fraeye, I.; Foubert, I.; Brabanter, J.D.; Cooman, L.D. Antioxidant potential of microalgae in relation to their phenolic and carotenoid content. J. Appl. Phycol. 2012, 24, 1477–1486. [Google Scholar] [CrossRef]
- Cervantes-Garcia, D.; Troncoso, R.; Sánchez-Estrada, A.; González-Mendoza, D.; Grimaldo-Juarez, O. Production of phenolics and flavonoids compounds in Euglena gracilis under copper stress. J. Pure Appl. Microbiol. 2013, 7, 93–100. [Google Scholar]
- Minussi, R.C.; Rossi, M.; Bologna, L.; Cordi, L.; Rotilio, D.; Mpastore, G.; Durán, N. Phenolic compounds and total antioxidant potential of commercial wines. Food Chem. 2003, 82, 409–416. [Google Scholar] [CrossRef]
- Dambolena, J.S.; Zunino, M.P.; Lucini, E.I.; Olmedo, R.; Banchio, E.; Bima, P.J.; Zygadlo, J.A. Total phenolic content, radical scavenging properties, and essential oil composition of Origanum species from different populations. J. Agric. Food Chem. 2010, 58, 1115–1120. [Google Scholar] [CrossRef]

| Microalgae Species | October | Controlled Environment | March | June | |
|---|---|---|---|---|---|
| I | 6.6 ± 2.8 | 8.1 ± 1.7 | 15.9 ± 5.3 | 25.7 ± 3.3 | |
| T | 23.2 ± 2.5 | 24.0 ± 1.1 | 28.5 ± 2.9 | 36.1 ± 3.2 | |
| C. vulgaris | μexp | 0.25 ± 0.01 c | 0.24 ± 0.01 c | 0.32 ± 0.01 a | 0.30 ± 0.01 b |
| Pb | 0.47 ± 0.01 c | 0.49 ± 0.00 c | 1.10 ± 0.01 a | 0.89 ± 0.01 b | |
| B. braunii | μexp | 0.19 ± 0.01 bc | 0.22 ± 0.01 b | 0.24 ± 0.00 a | 0.17 ± 0.02 c |
| Pb | 0.54 ± 0.00 b | 0.40 ± 0.01 c | 0.95 ± 0.00 a | 0.30 ± 0.01 d | |
| C. reinhardtii | μexp | 0.16 ± 0.01 c | 0.14 ± 0.01 d | 0.20 ± 0.01 b | 0.29 ± 0.01 a |
| Pb | 0.33 ± 0.01 c | 0.26 ± 0.01 d | 0.80 ± 0.00 b | 0.86 ± 0.01 a | |
| N. oculata | μexp | 0.15 ± 0.01 d | 0.20 ± 0.01 c | 0.23 ± 0.01 b | 0.31 ± 0.01 a |
| Pb | 0.43 ± 0.01 d | 0.66 ± 0.00 c | 0.98 ± 0.00 b | 1.28 ± 0.01 a | |
| E. gracilis | μexp | 0.16 ± 0.01 c | 0.19 ± 0.01 b | 0.25 ± 0.01 a | 0.26 ± 0.01 a |
| Pb | 0.37 ± 0.01 c | 0.69 ± 0.01 b | 0.96 ± 0.01 a | 0.95 ± 0.01 a |
| Microalgae Species | Nutrient Content % | October | Controlled Environment | March | June |
|---|---|---|---|---|---|
| C. vulgaris | Moisture | 8.1 ± 0.2 a/A | 8.3 ± 0.1 a/AB | 8.2 ± 0.1 a/AB | 8.1 ± 0.2 a/A |
| Lipids | 8.2 ± 0.4 a/AB | 9.3 ± 0.3 b/A | 15.0 ± 0.4 c/A | 20.3 ± 0.3 d/A | |
| Proteins | 25.1 ± 0.2 a/A | 25.4 ± 0.3 a/A | 25.5 ± 0.4 a/A | 25.9 ± 0.2 a/A | |
| Ash | 12.2 ± 0.3 a/AB | 12.7 ± 0.3 a/A | 12.1 ± 0.2 a/A | 12.3 ± 0.2 a/AB | |
| Fiber | 8.9 ± 0.3 a/A | 8.5 ± 0.1 ab/A | 8.2 ± 0.2 b/A | 8.1 ± 0.4 b/A | |
| NFE | 37.5 ± 0.5 a/A | 35.8 ± 0.3 b/A | 31.0 ± 0.1 c/A | 25.3 ± 0.3 d/A | |
| B. braunii | Moisture | 8.4 ± 0.2 a/A | 8.6 ± 0.1 a/AB | 8.2 ± 0.1 a/AB | 8.3 ± 0.2 a/A |
| Lipids | 7.4 ± 0.4 a/A | 8.6 ± 0.3 b/AB | 11.6 ± 0.2 c/B | 16.6 ± 0.5 d/B | |
| Proteins | 23.7 ± 0.3 a/BC | 23.8 ± 0.3 a/BC | 23.7 ± 0.1 a/B | 23.4 ± 0.1 a/B | |
| Ash | 10.9 ± 0.4 a/C | 10.9 ± 0.1 a/B | 10.1 ± 0.2 b/B | 10.4 ± 0.1 ab/C | |
| Fiber | 11.5 ± 0.2 a/CD | 11.4 ± 0.2 a/B | 11.1 ± 0.4 a/B | 10.9 ± 0.2 a/BC | |
| NFE | 38.1 ± 0.4 a/A | 36.7 ± 0.2 b/A | 35.3 ± 0.2 c/B | 30.4 ± 0.1 d/B | |
| C. reinhardtii | Moisture | 8.3 ± 0.2 a/A | 8.8 ± 0.1 b/B | 8.2 ± 0.2 a/AB | 8.2 ± 0.2 a/A |
| Lipids | 8.3 ± 0.4 a/AB | 9.2 ± 0.5 a/AB | 13.8 ± 0.4 b/CD | 15.8 ± 0.4 c/B | |
| Proteins | 24.3 ± 0.4 a/AB | 24.0 ± 0.3 a/B | 24.3 ± 0.3 a/C | 24.5 ± 0.2 a/C | |
| Ash | 11.5 ± 0.4 ab/BC | 11.7 ± 0.2 b/C | 11.1 ± 0.1 a/C | 11.4 ± 0.1 ab/D | |
| Fiber | 12.1 ± 0.4 a/D | 11.8 ± 0.1 ab/B | 11.7 ± 0.3 ab/B | 11.5 ± 0.1 b/C | |
| NFE | 35.5 ± 0.2 a/B | 34.5 ± 0.6 a/B | 30.9 ± 0.6 b/A | 28.6 ± 0.1 c/C | |
| N. oculata | Moisture | 8.2 ± 0.3 a/A | 8.1 ± 0.2 a/AB | 8.4 ± 0.2 a/A | 8.3 ± 0.2 a/A |
| Lipids | 8.5 ± 0.2 a/B | 11.5 ± 0.4 b/C | 14.8 ± 0.5 c/AD | 18.8 ± 0.4 d/C | |
| Proteins | 19.3 ± 0.5 a/D | 19.4 ± 0.2 a/D | 19.6 ± 0.1 a/D | 19.7 ± 0.2 a/D | |
| Ash | 12.3 ± 0.3 a/AB | 12.5 ± 0.2 a/A | 12.8 ± 0.3 a/D | 12.6 ± 0.1 a/A | |
| Fiber | 10.3 ± 0.4 a/B | 10.1 ± 0.1 a/C | 10.2 ± 0.2 a/C | 10.4 ± 0.3 a/B | |
| NFE | 41.4 ± 0.5 a/C | 38.4 ± 0.3 b/C | 34.2 ± 0.1 c/C | 30.2 ± 0.2 d/B | |
| E. gracilis | Moisture | 8.1 ± 0.2 a/A | 7.8 ± 0.1 a/A | 7.9 ± 0.1 aB | 7.8 ± 0.2 a/A |
| Lipids | 7.4 ± 0.4 a/A | 8.3 ± 0.3 b/B | 13.1 ± 0.4 c/C | 17.8 ± 0.7 d/C | |
| Proteins | 23.0 ± 0.2 a/C | 23.2 ± 0.3 a/C | 23.9 ± 0.7 b/BC | 23.3 ± 0.1 a/B | |
| Ash | 12.7 ± 0.3 ab/A | 12.9 ± 0.2 b/A | 12.3 ± 0.3 ab/AD | 12.2 ± 0.2 a/B | |
| Fiber | 11.1 ± 0.3 a/BC | 11.4 ± 0.3 a/B | 11.2 ± 0.2 a/B | 10.8 ± 0.2 a/B | |
| NFE | 37.7 ± 0.4 a | 36.4 ± 0.5 b/A | 31.6 ± 0.3 c/A | 28.1 ± 0.8 d/D |
| Microalgae Species | Total Phenolic Content (TPC) (μg GAE g−1 dry biomass) | |||
|---|---|---|---|---|
| October | Controlled Environment | March | June | |
| C. vulgaris | 3629 ± 21 a | 2703 ± 24 b | 3024 ± 31 c | 2469 ± 21 d |
| C. reinhardtii | 2406 ± 16 a | 2469 ± 20 b | 2124 ± 19 c | 2476 ± 12 b |
| B. braunii | 2342 ± 11 a | 2775 ± 13 b | 2144 ± 23 c | 2321 ± 13 a |
| N. oculata | 1318 ± 26 a | 1115 ± 14 b | 912 ± 14 c | 2906 ± 16 d |
| E. gracilis | 5806 ± 10 a | 5741 ± 18 b | 4166 ± 15 c | 2815 ± 18 d |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Metsoviti, M.N.; Papapolymerou, G.; Karapanagiotidis, I.T.; Katsoulas, N. Comparison of Growth Rate and Nutrient Content of Five Microalgae Species Cultivated in Greenhouses. Plants 2019, 8, 279. https://doi.org/10.3390/plants8080279
Metsoviti MN, Papapolymerou G, Karapanagiotidis IT, Katsoulas N. Comparison of Growth Rate and Nutrient Content of Five Microalgae Species Cultivated in Greenhouses. Plants. 2019; 8(8):279. https://doi.org/10.3390/plants8080279
Chicago/Turabian StyleMetsoviti, Maria N., George Papapolymerou, Ioannis T. Karapanagiotidis, and Nikolaos Katsoulas. 2019. "Comparison of Growth Rate and Nutrient Content of Five Microalgae Species Cultivated in Greenhouses" Plants 8, no. 8: 279. https://doi.org/10.3390/plants8080279
APA StyleMetsoviti, M. N., Papapolymerou, G., Karapanagiotidis, I. T., & Katsoulas, N. (2019). Comparison of Growth Rate and Nutrient Content of Five Microalgae Species Cultivated in Greenhouses. Plants, 8(8), 279. https://doi.org/10.3390/plants8080279






