Insect Antifeedant Components of Senecio fistulosus var. fistulosus—Hualtata
Abstract
1. Introduction
2. Results and Discussion
3. Materials and Methods
3.1. General
3.2. Extraction and Isolation
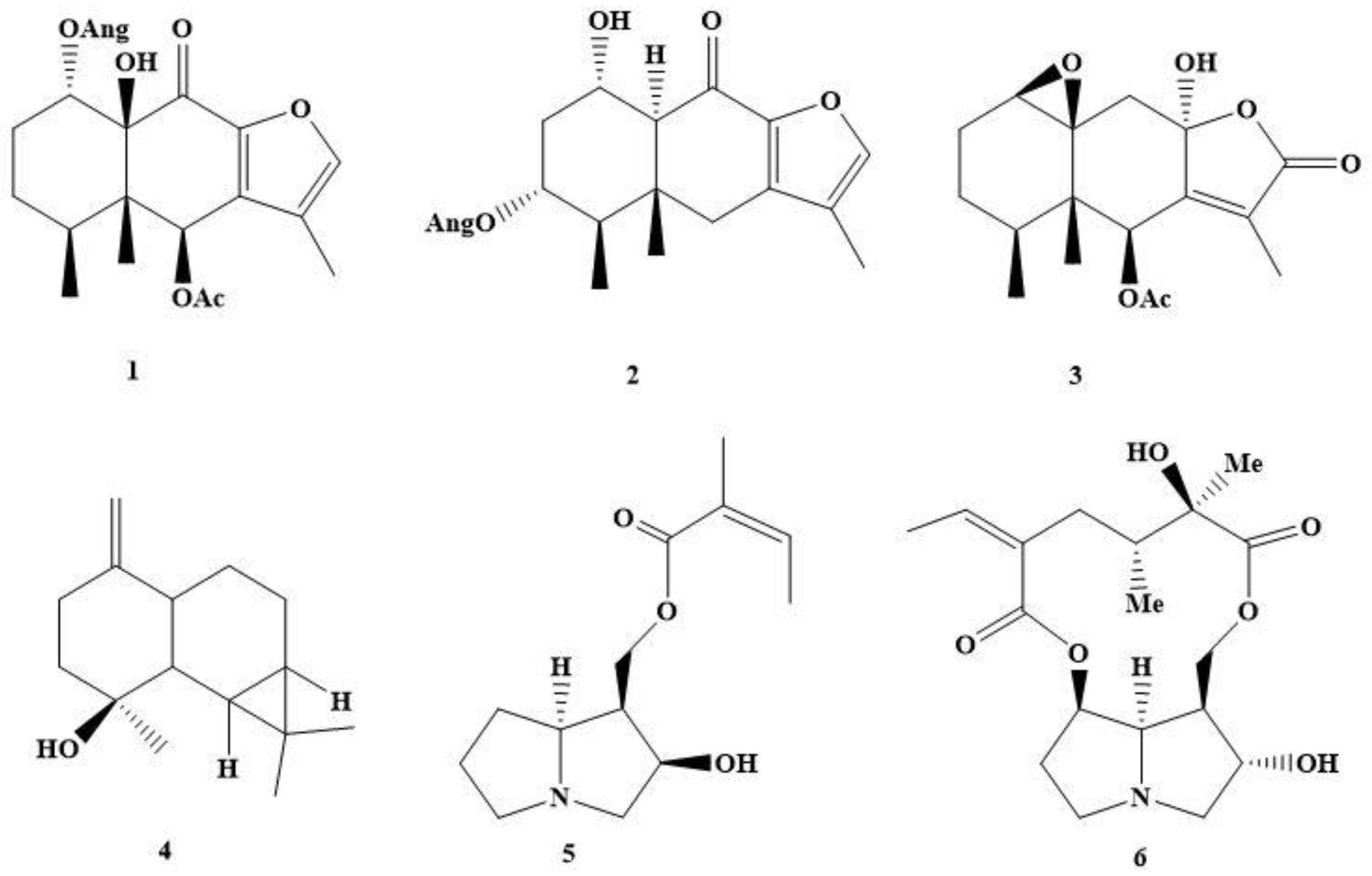
3.2.1. α-Angeloyloxy-6β-acetoxy-10β-hydroxy-9-oxo-furanoeremophilane (1)
3.2.2. α-hydroxy-3α-angeloyloxy-10αH-9-oxo-furanoeremophilane (2)
3.2.3. β,10β-epoxy-6β-acetoxy-8α-hydroxy-eremophil-7(11)-en-8β,12-olide (3)
3.2.4. (+)-1(14)-en-maaliol (4)
3.2.5. 9-O-angelylpetasinecine (hectorine) (5)
3.2.6. Rosmarinine (6)
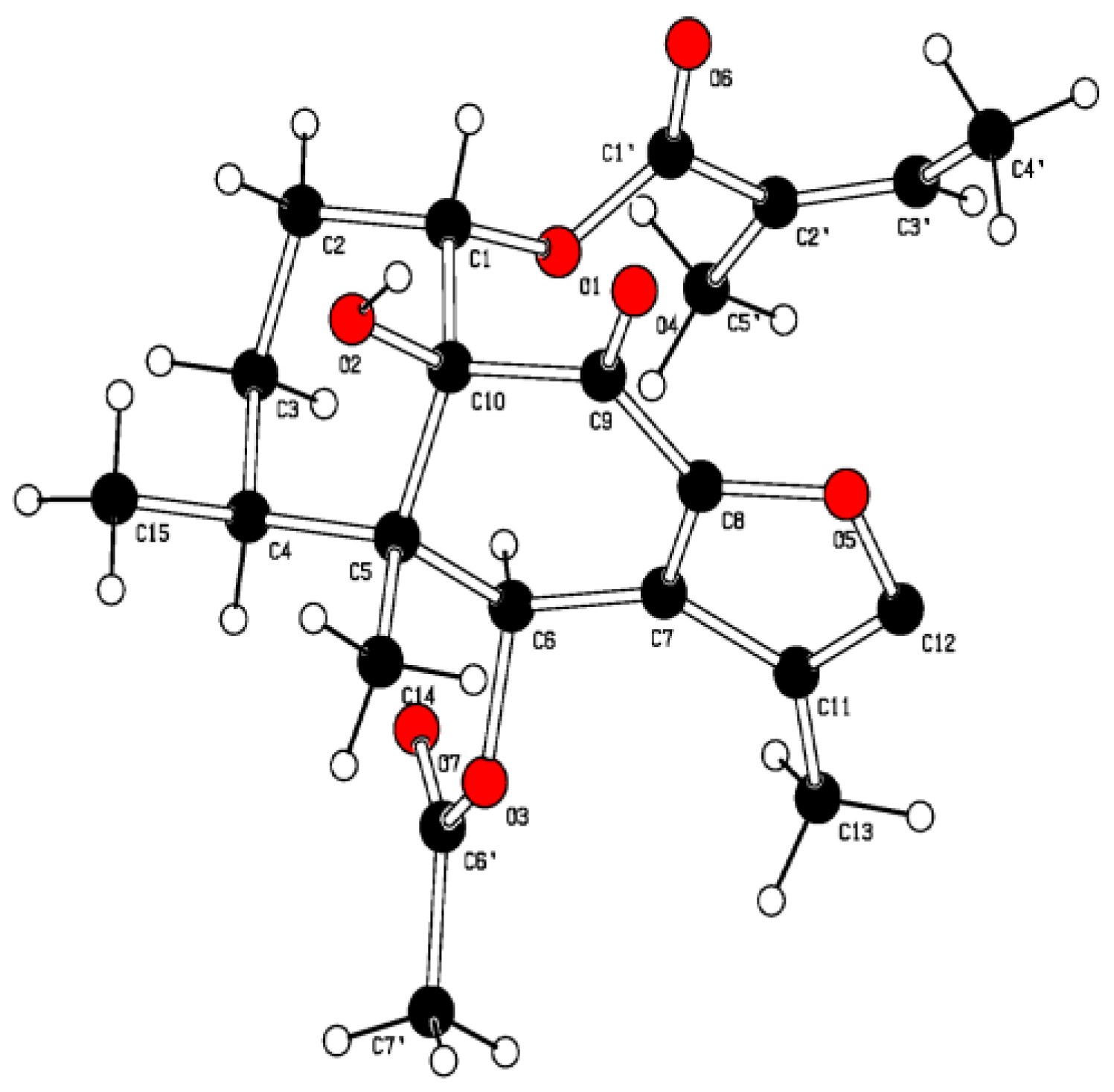
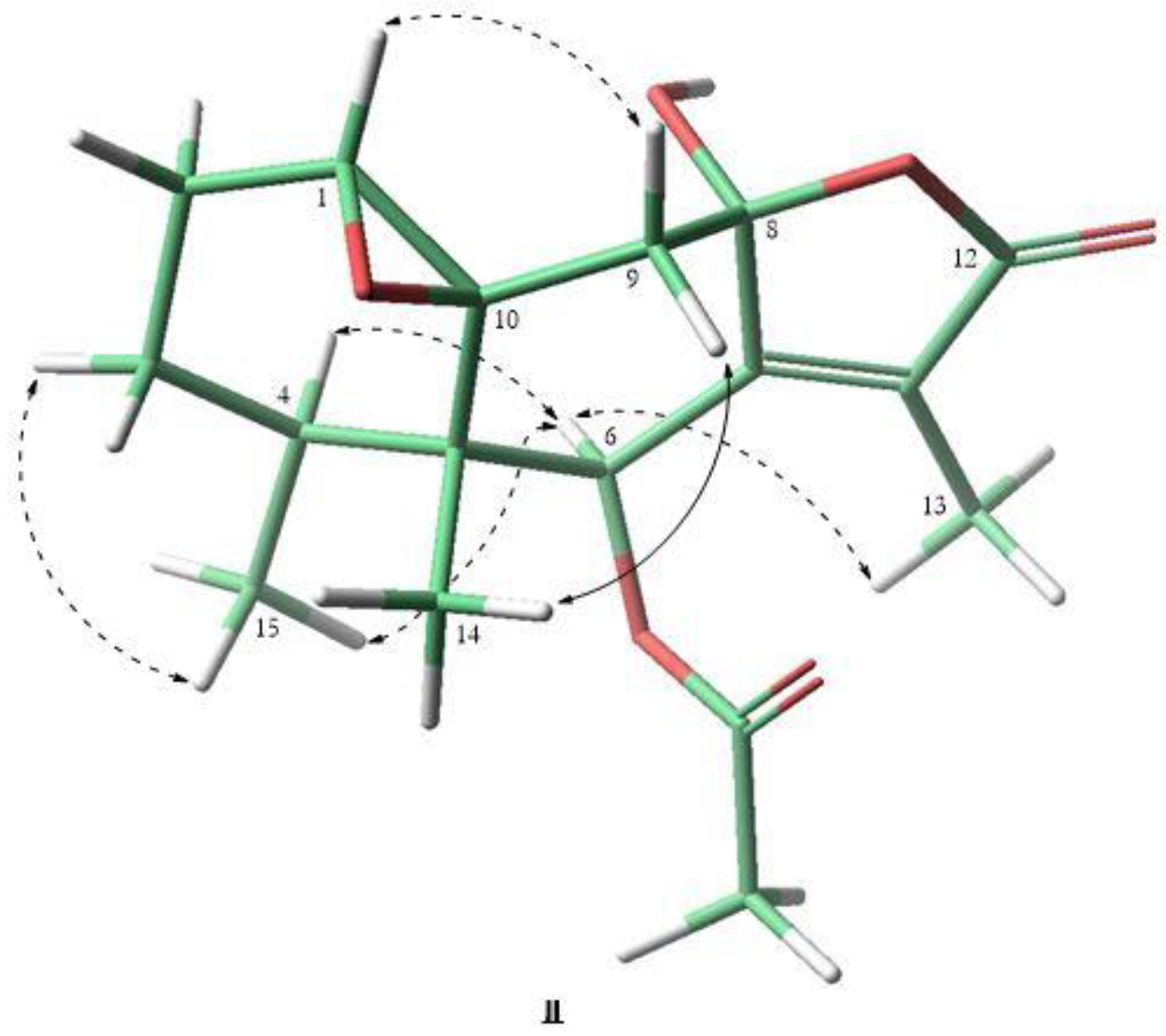
3.2.7. Crystal Structure Analysis
3.3. Insect Bioassays
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Portero, A.G.; González-Coloma, A.; Reina, M.; Díaz, C.E. Plant-defensive sesquiterpenoids from Senecio species with biopesticide potential. Phytochem. Rev. 2012, 11, 391–403. [Google Scholar] [CrossRef]
- Reina, M.; Santana, O.; Domínguez, D.M.; Villarroel, L.; Fajardo, V.; López-Rodríguez, M.; González-Coloma, A. Defensive sesquiterpenes from Senecio candidans and S. magellanicus, and their structure-activity relationships. Chem. Biodivers. 2012, 9, 625–643. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Vásquez, L.; Reina, M.; López-Rodríguez, M.; Giménez, C.; Cabrera, R.; Cuadra, P.; Fajardo, V.; González-Coloma, A. Sesquiterpenes, flavonoids, shikimic acid derivatives and pyrrolizidine alkaloids from Senecio kingii Hook. Phytochemistry 2015, 117, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Vásquez, L.; Olmeda, A.S.; Zuñiga, G.; Villarroel, L.; Echeverri, L.F.; González-Coloma, A.; Reina, M. Insect Antifeedant and Ixodicidal Compounds from Senecio adenotrichius. Chem. Biodivers. 2017, 14, e1600155. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Vásquez, L.; Ruiz Mesia, L.; Reina-Artiles, M.; López-Rodríguez, M.; González-Platas, J.; Giménez, C.; Cabrera, R.; González-Coloma, A. Benzofurans, benzoic acid derivatives, diterpenes and pyrrolizidine alkaloids from Peruvian Senecio. Phytochem. Lett. 2018, 28, 47–54. [Google Scholar] [CrossRef]
- Agullo-Ortuño, M.T.; Diaz, C.E.; Gonzalez-Coloma, A.; Reina, M. Structure-Dependent Cytotoxic Effects of Eremophilanolide Sesquiterpenes. Nat. Prod. Commun. 2017, 12, 663–665. [Google Scholar] [CrossRef] [PubMed]
- Henríquez, J.M.; Pisano, E.; Marticorena, C. Catálogo de la Flora Vascular de Magallanes (XII Región); Anales Instituto de la Patagonia, Sección Ciencias Naturales, Universidad de Magallanes: Punta Arenas, Chile, 1995; pp. 5–30. [Google Scholar]
- Arias Cassará, M.L.; Borkosky, S.A.; González Sierra, M.; Bardón, A.; Ybarra, M.I. Two new furanoeremophilanes from Senecio santelisis. Chem. Biodivers. 2010, 7, 1745–1753. [Google Scholar] [CrossRef] [PubMed]
- Bolzan, A.A.; Silva, C.M.; Francescato, L.N.; Murari, A.L.; Silva, G.N.; Heldwein, C.G.; Heinzmann, B. Espécies de Senecio na Medicina Popular da América Latina e Toxicidade Relacionada a sua Utilização. Lat. Am. J. Pharm. 2007, 26, 619–625. [Google Scholar]
- Villarroel, L.; Torres, R.J. A new furanoeremophilane from Senecio fistulosus. J. Nat. Prod. 1985, 48, 841–842. [Google Scholar] [CrossRef]
- Domínguez, D.M.; Reina, M.; Villarroel, L.; Fajardo, V.; González-Coloma, A. Bioactive Furanoeremophilanes from Senecio otites Kunze ex DC.Z. Naturforsch 2008, 63c, 837–842. [Google Scholar] [CrossRef]
- Bohlmann, F.; Zdero, C.; King, R.M.; Robinson, H. Furanoeremophilanes from Senecio smithii. Phytochemistry 1981, 20, 2389–2391. [Google Scholar] [CrossRef]
- Bai, Y.; Benn, M.; Duke, N.; Gul, W.; Huangand, Y.Y.; Rüeger, H. The alkaloids of Brachyglottis hectori. Arkivoc 2006, 3, 34–42. [Google Scholar]
- Were, O.; Benn, M.; Munavu, R.M. The pyrrolizidine alkaloids of Senecio syringifolius and S. hadiensis from Kenya. Phytochemistry 1993, 32, 1595–1602. [Google Scholar] [CrossRef]
- Naya, K.; Nogi, N.; Makiyama, Y.; Takashima, H.; Imagawa, T. The Photosensitized Oxygenation of Furanoeremophilanes. II. The Preparation and Stereochemistry of the Isomeric Hydroperoxides and the Corresponding Lactones from Furanofukinin and Furanoeremophilane. Bull. Chem. Soc. Jpn. 1977, 50, 3002–3006. [Google Scholar] [CrossRef]
- Harrie, J.M.; Joannes, B.P.A.; Wijnberg, C.R.; Groom, A. Rearrangement reactions of aromadendrane derivatives. The synthesis of (+)-maaliol, starting from natural (+)-aromadendrene-IV. Tetrahedron 1994, 50, 4733–4744. [Google Scholar]
- De Pascual, T.; Urones, J.G.; Fernández, A. An aristolochic acid derivative from Aristolochia longa. Phytochemistry 1983, 22, 2753–2754. [Google Scholar] [CrossRef]
- Gutierrez, C.; Fereres, A.; Reina, M.; Cabrera, R.; González-Coloma, A. Behavioral and sublethal effects of structurally related lower terpenes on Myzus persicae. J. Chem. Ecol. 1997, 23, 1641–1650. [Google Scholar] [CrossRef]
- Duran-Peña, M.J.; Ares, J.M.B.; Hanson, J.R.; Collado, I.G.; Hernandez-Galan, R. Biological activity of natural sesquiterpenoids containg a gem-dimethylcyclopropane unit. Nat. Prod. Rep. 2015, 32, 1236–1248. [Google Scholar] [CrossRef]
- Hartmann, T. Chemical ecology of pyrrolizidine alkaloids. Planta 1999, 207, 483–495. [Google Scholar] [CrossRef]
- Castells, E.; Mulder, P.P.J.; Pérez-Trujillo, M. Diversity of pyrrolizidine alkaloids in native and invasive Senecio pterophorus (Asteraceae): Implications for toxicity. Phytochemistry 2014, 108, 137–146. [Google Scholar] [CrossRef]
- Culvenor, C.C.J.; Edgar, J.A.; Smith, L.W.; Jago, M.V.; Peterson, J.E. Active metabolites in the chronic hepatotoxicity of pyrrolizidine alkaloids, including otonecine esters. Nat. New Biol. 1971, 229, 255–256. [Google Scholar] [CrossRef]
- Styles, J.; Asbey, J.; Mattocks, A.R. Evaluation in vitro of several pyrrolizidine alkaloid carcinogens: Observations on the essential pyrrolic nucleus. Carcinogenesis 1980, 1, 161–164. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.H.; Kan, W.L.T.; Li, N.; Lin, G. Assessment of pyrrolizidine alkaloid induced toxicity in an in vitro screening model. J. Ethnopharmacol. 2013, 150, 560–567. [Google Scholar] [CrossRef] [PubMed]
- CrysAlis RED Version 1.171.32.5; Oxford Diffraction Ltd.: Abingdon, UK, 2007.
- Altomare, A.; Cascarano, G.; Giacovazzo, C.; Guagliardi, A.; Moliterni, A.G.G.; Burla, M.C.; Polidor, G.; Camalli, M. Spagna R. SIR97; University of Bari: Bari, Italy, 1997. [Google Scholar]
- Sheldrick, G.M. A short history of SHELX. Acta Cryst. 2008, A64, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Farrugia, L.J. WinGX suite for small-molecule single-crystal crystallography. J. Appl. Cryst. 1999, 32, 837–838. [Google Scholar] [CrossRef]
- Spek, A.L. Single-crystal structure validation with the program PLATON. J. Appl. Cryst. 2003, 36, 7–13. [Google Scholar] [CrossRef]
- Flack, H.D. On enantiomorph-polarity estimation. Acta Cryst. 1983, A39, 876–881. [Google Scholar] [CrossRef]
- Hooft, R.W.W.; Straver, L.H.; Spek, A.L. Determination of absolute structure using Bayesian statistics on Bijvoet differences. J. Appl. Cryst. 2008, 41, 96–103. [Google Scholar] [CrossRef]
- Poitout, S.; Bues, S. Elevage de plusieursespeces de Lepidopteres Noctuidae sur milleu artificiel simplifié. Ann. Zool. Ecol. Anim. 1970, 2, 79–91. [Google Scholar]
- Burgueño-Tapia, E.; Castillo, L.; González-Coloma, A.; Joseph-Nathan, P. Antifeedant and phytotoxic activity of the sesquiterpene p-benzoquinone perezone and some of its derivatives. J. Chem. Ecol. 2008, 34, 766–771. [Google Scholar] [CrossRef]

| Position | 1 | 2 | ||
|---|---|---|---|---|
| δH in ppm, Multiplicity, J (in Hz) | δC in ppm | δH in ppm, Multiplicity, J (in Hz) | δC in ppm | |
| 1β | 4.84 br s | 74.5 d | 4.25 ddd (4.8, 9.7, 11.7) | 65.5 d |
| 2α | 2.30 m | 20.7 t | 1.50 q (11.8) | 39.1 t |
| 2β | 1.64 m | 2.47 m | ||
| 3α | 1.40 m | 23.9 t | - | 71.4 d |
| 3β | 2.32 m | 4.90 dt (4.6, 11.5) | ||
| 4α | 1.65 m | 32.3 d | 1.84 m | 46.9 d |
| 5 | - | 50.2 s | - | 43.9 s |
| 6α | 7.03 s | 68.6 d | 2.52 d (16.6) | 35.9 t |
| 6β | - | 2.71 d (16.6) | ||
| 7 | - | 139.4 s | - | 138.1 s |
| 8 | - | 145.9 s | - | 146.5 s |
| 9 | - | 186.9 s | - | 188.7 s |
| 10β | - | 79.8 s | 2.42 d (9.8) | 60.6 d |
| 11 | - | 121.8 s | - | 121.7 s |
| 12 | 7.41 br s | 146.9 d | 7.41 s | 145.8 d |
| 13 | 1.91 d (1.1) | 8.3 q | 1.99 d (1.0) | 7.8 q |
| 14 | 0.98 s | 15.5 q | 0.88 s | 14.3 q |
| 15 | 1.17 d (7.5) | 16.0 q | 0.98 d (6.7) | 10.5 q |
| 1’ | - | 165.7 s | - | 167.6 s |
| 2’ | - | 126.6 s | - | 128.1 s |
| 3’ | 5.92 qq (1.5, 7.2) | 140.5 d | 6.05 qq (1.4, 7.2) | 137.8 d |
| 4’ | 1.88 dq (1.6, 7.4) | 15.8 q | 1.97 dq (1.5, 7.2) | 15.8 q |
| 5’ | 1.55 quint. (1.5) | 19.9 q | 1.88 quint. (1.5) | 20.6 q |
| OCOCH3 | 2.19 s | 20.9 q | - | - |
| OCOCH3 | - | 170.9 s | - | - |
| OH-10 | 3.98 br s | - | - | - |
| Position | 3 | 7 * | ||
|---|---|---|---|---|
| δH in ppm, Multiplicity, J (in Hz) | δC in ppm | δH in ppm, Multiplicity, J (in Hz) | δC in ppm | |
| 1β | 3.18 d (4.6) | 62.7 d | 3.14 d (4.3) | 63.0 d |
| 2α | 1.96 dd (6.8, 10.8) | 20.3 t | 2.04 m | 21.0 t |
| 2β | 2.04 dd (5.9, 10.5) | 2.21 m | ||
| 3α | 1.36 dc (3.5, 9.4) | 23.9 t | 1.38 m | 24.2 t |
| 3β | 1.63 m | 1.61 m | ||
| 4α | 1.62 m | 32.5 d | 1.62 m | 33.0 d |
| 5 | - | 43.4 s | - | 43.5 s |
| 6α | 5.92 c (1.8) | 73.8 d | 5.69 t (1.7) | 74.3 d |
| 7 | - | 155.0 s | - | 153.9 s |
| 8 | - | 101.3 s | - | 104.4 s |
| 9α | 1.79 d (13.6) | 43.4 t | 1.80 d (13.6) | 43.6 t |
| 9β | 2.31 d (13.6) | 2.27 d (13.6) | ||
| 10 | - | 60.9 s | - | 61.0 s |
| 11 | - | 124.6 s | - | 126.8 s |
| 12 | - | 170.8 s | - | 170.9 s |
| 13 | 1.87 d (1.8) | 8.2 q | 1.92 d (1.2) | 8.6 q |
| 14 | 1.09 s | 14.5 q | 1.09 s | 14.5 q |
| 15 | 1.04 d (7.2) | 16.1 q | 1.03 d (7.0) | 16.5 q |
| OMe-8 | - | - | 3.23 s | 50.9 q |
| OCOCH3 | 2.20 s | 20.9 q | 2.21 s | 21.0 q |
| OCOCH3 | - | 170.6 s | - | 170.3 s |
| Position | δH in ppm, Multiplicity, J (in Hz) | δC in ppm | HMBC |
|---|---|---|---|
| 1 | - | 153.6 s | - |
| 2a | 2.42 ddd (1.3, 6.3, 13.3) | 39.1 t | C-1, C-4, C-10, C-14 |
| 2b | 2.05 m | ||
| 3a | 1.77 m | 41.9 t | C-2, C-4 |
| 3b | 1.56 d | ||
| 4 | - | 81.1 s | - |
| 5α | 1.32 m | 54.6 d | C-1, C-4, C-6, C-10, C-11 |
| 6β | 0.47 dd (9.6, 11.4) | 30.1 d | C-4, C-8, C-11, C-13 |
| 7β | 0.71 ddd (6.1, 9.5, 11.4) | 27.7 d | C-5, C-11, C-13 |
| 8a | 1.98 m | 24.9 t | - |
| 8b | 1.01 m | ||
| 9a | 1.90 m | 26.9 t | C-5, C-10 |
| 9b | 1.63 m | ||
| 10β | 2.20 m | 53.6 d | C-1, C-2, C-5, C-6, C-14 |
| 11 | - | 20.4 s | - |
| 12 | 1.06 s | 28.8 q | C-7, C-6, C-11, C-13 |
| 13 | 1.04 s | 16.5 q | C-7, C-6, C-11, C-12 |
| 14a | 4.69 t (1.6) | 106.4 t | C-2, C-10 |
| 14b | 4.63 q (1.7) | ||
| 15 | 1.28 s | 26.2 q | C-3, C-4, C-5 |
| Compound | S. littoralis | EC50 (μg/cm2) b | M. persicae | EC50 (μg/cm2) b |
|---|---|---|---|---|
| %FI (50 μg/cm2) a | %SI b (50 μg/cm2) a | |||
| 1 | 65 ± 6 * | 52 ± 7 | ||
| 2 | 83 ± 6 * | 0.64 (0.36-1.16) | 52 ± 7 | |
| 4 | 64 ± 7 * | 90 ± 3 * | 0.97 (0.71–1.32) |
| Compound | Substituent | S. littoralis | M. persicae | |||
|---|---|---|---|---|---|---|
| C-1 | C-3 | C-6 | C-10 | %FI c (EC50) d | %SI c (EC50) d | |
| 1 | α-OAng | H2 | β-OAc | β-OH | 65.0 | 52.0 |
| 2 | α-OH | α-OAng | H2 | α-H | 83.0 (0.64) | 52.0 |
| 8 a | Δ1 | H2 | β-OAng | Δ10 | 32.0 | 67.0 |
| 9 a | Δ1 | H2 | β-OH | Δ10 | 62.0 | 71.0 |
| 10 a | Δ1 | H2 | β-OCOCH2CH3 | Δ10 | 45.0 | 75.0 |
| 11 b | H2 | H2 | β-OAc | α-H | 51.0 | 74.0 (21.9) |
| 12 b | H2 | H2 | β-OTigl | α-H | 65.0 | 74.0 (12.2) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruiz-Vásquez, L.; Reina, M.; Fajardo, V.; López, M.; González-Coloma, A. Insect Antifeedant Components of Senecio fistulosus var. fistulosus—Hualtata. Plants 2019, 8, 176. https://doi.org/10.3390/plants8060176
Ruiz-Vásquez L, Reina M, Fajardo V, López M, González-Coloma A. Insect Antifeedant Components of Senecio fistulosus var. fistulosus—Hualtata. Plants. 2019; 8(6):176. https://doi.org/10.3390/plants8060176
Chicago/Turabian StyleRuiz-Vásquez, Liliana, Matías Reina, Víctor Fajardo, Matías López, and Azucena González-Coloma. 2019. "Insect Antifeedant Components of Senecio fistulosus var. fistulosus—Hualtata" Plants 8, no. 6: 176. https://doi.org/10.3390/plants8060176
APA StyleRuiz-Vásquez, L., Reina, M., Fajardo, V., López, M., & González-Coloma, A. (2019). Insect Antifeedant Components of Senecio fistulosus var. fistulosus—Hualtata. Plants, 8(6), 176. https://doi.org/10.3390/plants8060176






