Herbicide Resistance Management: Recent Developments and Trends
Abstract
:1. Introduction
2. Herbicide Discovery: Renewed Efforts
3. Herbicide-Resistant (HR) Crops: The Changing State of Trait Adoption
4. Increasing Prominence of Preemergence Herbicides
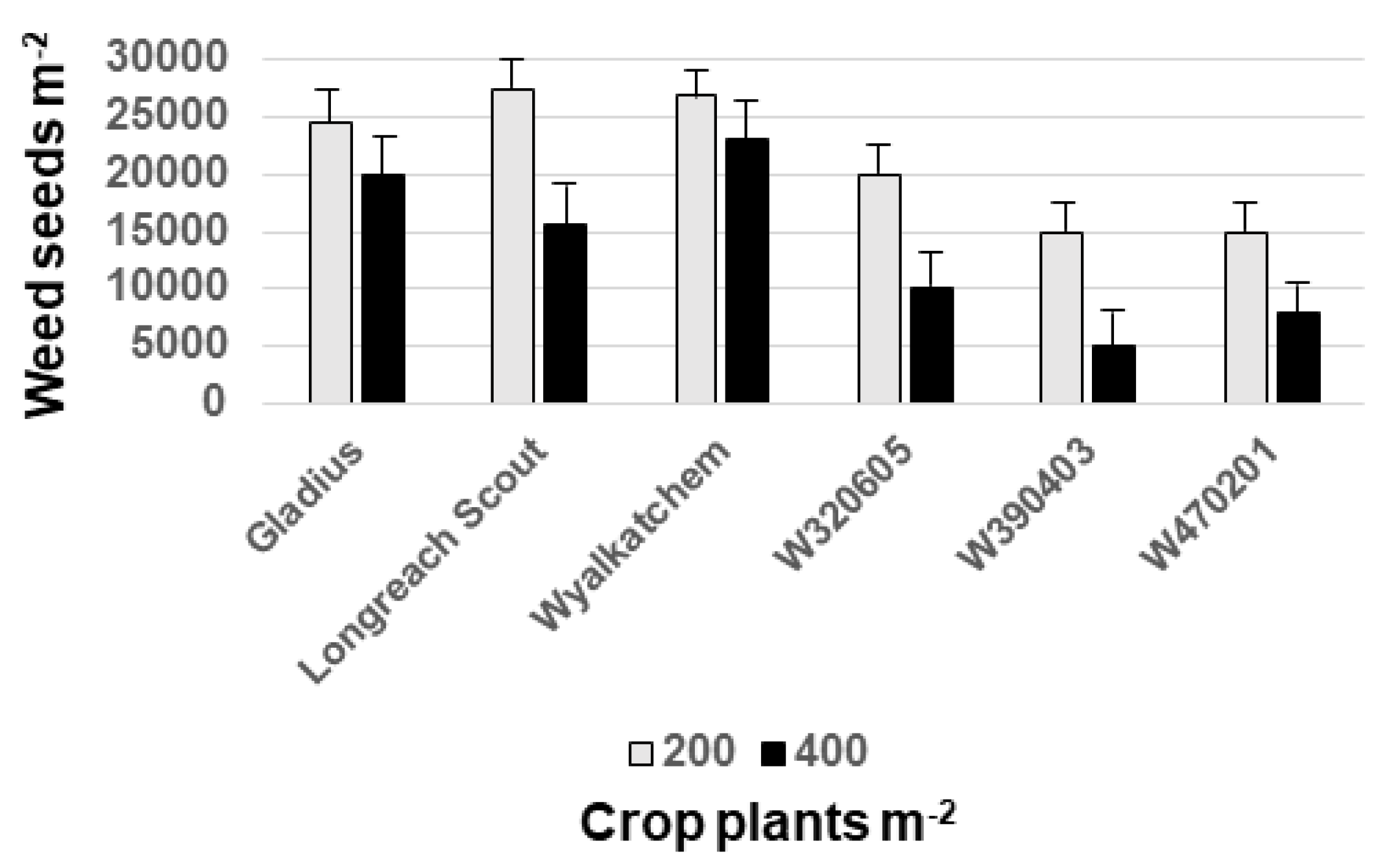
5. Plant Breeders Consider Weed Competitiveness
6. Harvest Weed Seed Control (HWSC) Gaining Momentum Globally
7. Is Site-Specific Weed Management (SSWM) in Agronomic Field Crops Set to Take Off?
8. Conclusions and Future Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Jones, R.E.; Medd, R.W. Economic thresholds and the case for longer term approaches to population management of weeds. Weed Technol. 2000, 14, 337–350. [Google Scholar] [CrossRef]
- Lacoste, M.; Powles, S. RIM: Anatomy of a weed management decision support system for adaptation and wider application. Weed Sci. 2015, 63, 676–689. [Google Scholar] [CrossRef]
- Somerville, G.J.; Powles, S.B.; Walsh, M.J.; Renton, M. How do spatial heterogeneity and dispersal in weed population models affect predictions of herbicide resistance evolution? Ecol. Model. 2017, 362, 37–53. [Google Scholar] [CrossRef]
- Barber, L.T.; Smith, K.L.; Scott, R.C.; Norsworthy, J.K.; Vangilder, A.M. Zero Tolerance: A Community-Based Program for Glyphosate-Resistant Palmer Amaranth Management; University of Arkansas Cooperative Extension Service Bulletin FSA2177: Fayetteville, AR, USA, 2015. [Google Scholar]
- Bayer CropScience. How a Zero Tolerance Approach can Mimimize Weed Resistance. 2019. Available online: https://www.cropscience.bayer.us/learning-center/articles/how-a-zero-tolerance-approach-can-minimize-weed-resistance (accessed on 1 May 2019).
- Green, J.M.; Owen, M.D.K. Herbicide-resistant crops: Utilities and limitations for herbicide-resistant weed management. J. Agric. Food Chem. 2011, 59, 5819–5829. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Cornejo, J.; Hallahan, C.; Nehring, R.; Wechsler, S. Conservation tillage, herbicide use, and genetically engineered crops in the United States: The case of soybeans. AgBioForum 2012, 15, 231–241. [Google Scholar]
- Katalin, T.-G.; Eniko, L.; István, T. Economic benefits of precision weed control and why its uptake is so slow. Stud. Agric. Econ. 2013, 115, 40–46. [Google Scholar]
- Heap, I.M. International Survey of Herbicide Resistant Weeds. 2019. Available online: http://www.weedscience.org (accessed on 1 May 2019).
- Duke, S.O. The history and current status of glyphosate. Pest Manag. Sci. 2018, 74, 1027–1034. [Google Scholar] [CrossRef]
- Benbrook, C.M. Trends in glyphosate herbicide use in the United States and globally. Environ. Sci. Eur. 2016, 28, 3. [Google Scholar] [CrossRef]
- Duke, S.O.; Stidham, M.A.; Dayan, F.E. A novel genomic approach to herbicide and herbicide mode of action discovery. Pest Manag. Sci. 2019, 75, 314–317. [Google Scholar] [CrossRef]
- Peters, B.; Strek, H.J. Herbicide discovery in light of rapidly spreading resistance and ever-increasing regulatory hurdles. Pest Manag. Sci. 2018, 74, 2211–2215. [Google Scholar] [CrossRef]
- Phillips McDougall. The Cost of New Agrochemical Product Discovery, Development and Registration in 1995, 2000, 2005–8 and 2010 to 2014—R&D Expenditure in 2014 and Expectations for 2019; R&D Report; Phillips McDougall: Pathhead, UK, 2016; 41p. [Google Scholar]
- Dickmann, R. Herbicide Discovery a Global Expedition; GroundCover Issue 135; Grains Research and Development Corporation: Canberra, Australia, 2018. [Google Scholar]
- Begemann, S. FMC to Introduce Two New Herbicide Modes of Action in Next Decade; AgProfessional Farm Journal: Lenexa, KS, USA, 2019. [Google Scholar]
- Westwood, J.H.; Charudattan, R.; Duke, S.O.; Fennimore, S.A.; Marrone, P.; Slaughter, D.C.; Swanton, C.; Zollinger, R. Weed management in 2050: Perspectives on the future of weed science. Weed Sci. 2017, 66, 275–285. [Google Scholar] [CrossRef]
- Dayan, F.E.; Duke, S.O. Natural compounds as next-generation herbicides. Plant Physiol. 2014, 166, 1090–1105. [Google Scholar] [CrossRef]
- Corral, M.G.; Leroux, J.; Stubbs, K.A.; Mylne, J.S. Herbicidal properties of antimalarial drugs. Sci. Rep. 2017, 7, e45871. [Google Scholar] [CrossRef]
- Kraehmer, H.; van Almsick, A.; Beffa, R.; Dietrich, H.; Eckes, P.; Hacker, E.; Hain, R.; Strek, H.J.; Stuebler, H.; Willms, L. Herbicides as weed control agents: State of the art: II. recent achievements. Plant Physiol. 2014, 166, 1132–1148. [Google Scholar] [CrossRef]
- [USDA-NASS] United States Department of Agriculture–National Agricultural Statistics Service. USDA Reports Soybean, Corn Acreage Down; 2018. Available online: https://www.nass.usda.gov/Newsroom/2018/06-29-2018.php (accessed on 1 May 2019).
- Wechsler, S.J. Trends in the Adoption of Genetically Engineered Corn, Cotton, and Soybeans. 2018. Available online: https://www.ers.usda.gov/amber-waves/2018/december/trends-in-the-adoption-of-genetically-engineered-corn-cotton-and-soybeans (accessed on 1 May 2019).
- Unglesbee, E. Soybean Herbicide Tolerant-Trait Systems for 2019—Breaking It Down. 2018. Available online: https://agfax.com/2018/10/03/soybean-seed-choices-for-2019-breaking-it-down-dtn (accessed on 1 May 2019).
- Peterson, M.A.; Collavo, A.; Ovejero, R.; Shivrain, V.; Walsh, M.J. The challenge of herbicide resistance around the world: A current summary. Pest Manag. Sci. 2018, 74, 2246–2259. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.J.; Fowler, T.M.; Crowe, B.; Ambe, T.; Powles, S.B. The potential for pyroxasulfone to selectively control resistant and susceptible rigid ryegrass (Lolium rigidum) biotypes in Australian grain crop production systems. Weed Technol. 2011, 25, 30–37. [Google Scholar] [CrossRef]
- Webster, T.M.; Sosnoskie, L.M. Loss of glyphosate efficacy: A changing weed spectrum in Georgia cotton. Weed Sci. 2010, 58, 73–79. [Google Scholar] [CrossRef]
- Beckie, H.J.; Reboud, X. Selecting for weed resistance: Herbicide rotation and mixture. Weed Technol. 2009, 23, 363–370. [Google Scholar] [CrossRef]
- Owen, M.D. Diverse approaches to herbicide-resistant weed management. Weed Sci. 2016, 64, 570–584. [Google Scholar] [CrossRef]
- Gill, G.S.; Holmes, J.E. Efficacy of cultural control methods for combating herbicide-resistant Lolium rigidum. Pestic. Sci. 1997, 51, 352–358. [Google Scholar] [CrossRef]
- Gomez-Macpherson, H.; Richards, R.A. Effect of sowing time on yield and agronomic characteristics of wheat in south-eastern Australia. Aust. J. Agric. Res. 1995, 46, 1381–1399. [Google Scholar] [CrossRef]
- Flower, K.; Crabtree, B.; Butler, G. No-till cropping systems in Australia. In No-Till Farming Systems; Goddard, T., Zoebisch, M., Gan, Y., Ellis, W., Watson, A., Sombatpanit, S., Eds.; Special Publication No. 3; World Association of Soil and Water Conservation: Bankok, Thailand, 2008; pp. 457–467. [Google Scholar]
- Ashworth, M.; Desbiolles, J.; Tola, E. Disc Seeding in Zero-Till Farming Systems—A Review of Technology and Paddock Issues; Western Australian No-Tillage Farmers Association: Northam, Australia, 2010; 226p. [Google Scholar]
- Beckie, H.J.; Harker, K.N. Our top 10 herbicide-resistant weed management practices. Pest Manag. Sci. 2017, 73, 1045–1052. [Google Scholar] [CrossRef] [PubMed]
- WeedSmart. The Big 6. 2019. Available online: http://www.weedsmart.org.au (accessed on 1 May 2019).
- Liebman, M.; Baraibar, B.; Buckley, Y.; Childs, D.; Christensen, S.; Cousens, R.; Eizenberg, H.; Heijting, S.; Loddo, D.; Morotto, A., Jr.; et al. Ecologically sustainable weed management: How do we get from proof-of-concept to adoption. Ecol. Appl. 2016, 26, 1352–1369. [Google Scholar] [CrossRef] [PubMed]
- Andrew, I.K.S.; Storkey, J.; Sparkes, D.L. A review of the potential for competitive cereal cultivars as a tool in integrated weed management. Weed Res. 2015, 55, 239–248. [Google Scholar] [CrossRef] [PubMed]
- Rebetzke, G. Weed-Competitive Plants on the Way. GroundCover Supplement Issue 127, Grains Research and Development Corporation, Canberra, Australia. 2017. Available online: https://grdc.com.au/resources-and-publications/groundcover/ground-cover-supplements/groundcover-issue-127-wheat-prebreeding/weed-competitive-plants-on-the-way (accessed on 1 May 2019).
- Worthington, M.; Reberg-Horton, C. Breeding cereal crops for enhanced weed suppression: Optimizing allelopathy and competitive ability. J. Chem. Ecol. 2013, 39, 213–231. [Google Scholar] [CrossRef] [PubMed]
- Widderick, M.; Lemerle, D.; Bell, K.; Storrie, A.; Hashem, A.; Taylor, C.; Osten, V.; Johansen, C.; Cook, T. Crop Competition Management Options for Herbicide-resistant Weeds: Literature Review, Meta-analysis, Gap Analysis and Trial Design Recommendations. GRDC Project DAQ00197. 2015. Available online: https://grdc.com.au/research/reports/report?id=6253 (accessed on 1 May 2019).
- Walsh, M.J.; Broster, J.C.; Schwartz-Lazaro, L.M.; Norsworthy, J.K.; Davis, A.S.; Tidemann, B.D.; Beckie, H.J.; Lyon, D.J.; Soni, N.; Neve, P.; et al. Opportunities and challenges for harvest weed seed control in global cropping systems. Pest Manag. Sci. 2018, 74, 2235–2245. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.J.; Harrington, R.B.; Powles, S.B. Harrington Seed Destructor: A new nonchemical weed control tool for global grain crops. Crop Sci. 2012, 52, 1343–1347. [Google Scholar] [CrossRef]
- Walsh, M.J.; Ouzman, J.; Newman, P.; Powles, S.; Llewellyn, R. High levels of adoption indicate that harvest weed seed control is now an established weed control practice in Australian cropping. Weed Technol. 2017, 31, 1–7. [Google Scholar] [CrossRef]
- Tidemann, B.D.; Hall, L.M.; Harker, K.N.; Beckie, H.J. Factors affecting weed seed devitalization with the Harrington Seed Destructor. Weed Sci. 2017, 65, 650–658. [Google Scholar] [CrossRef]
- Tidemann, B.D.; Hall, L.M.; Harker, K.N.; Beckie, H.J.; Johnson, E.N.; Stevenson, F.C. Suitability of wild oat (Avena fatua), false cleavers (Galium spurium), and volunteer canola (Brassica napus) for harvest weed seed control in western Canada. Weed Sci. 2017, 65, 769–777. [Google Scholar] [CrossRef]
- Beckie, H.J.; Blackshaw, R.E.; Harker, K.N.; Tidemann, B.D. Weed seed shatter in spring wheat in Alberta. Can. J. Plant Sci. 2018, 98, 107–114. [Google Scholar] [CrossRef]
- Schwartz-Lazaro, L.M.; Green, J.K.; Norsworthy, J.K. Seed retention of Palmer amaranth (Amaranthus palmeri) and barnyardgrass (Echinochloa crus-galli) in soybean. Weed Technol. 2017, 31, 617–622. [Google Scholar] [CrossRef]
- Ashworth, M.B.; Walsh, M.J.; Flower, K.C.; Vila-Aiub, M.M.; Powles, S.B. Directional selection for flowering time leads to adaptive evolution in Raphanus raphanistrum (wild radish). Evol. Appl. 2016, 9, 619–629. [Google Scholar] [CrossRef] [PubMed]
- Walsh, M.J.; Aves, C.; Powles, S.B. Harvest weed seed control systems are similarly effective on rigid ryegrass. Weed Technol. 2017, 31, 178–183. [Google Scholar] [CrossRef]
- Young, S.L.; Pierce, F.J. Automation: The Future of Weed Control in Cropping Systems; Springer: New York, NY, USA, 2014; 265p. [Google Scholar]
- Fernández-Quintanilla, C.; Peña, J.M.; Andújar, D.; Dorado, J.; Ribeiro, A.; López-Granados, F. Is the current state of the art of weed monitoring suitable for site-specific weed management in arable crops? Weed Res. 2018, 58, 259–272. [Google Scholar] [CrossRef]
- Cameron, J.; Storrie, A. Summer Fallow Weed Management: A Reference Manual for Grain Growers and Advisers in the Southern and Western Grains Regions of Australia. GRDC Publ. ICN00012. 2014. Available online: https://grdc.com.au/__data/assets/pdf_file/0028/98632/summer-fallow-weed management-manual.pdf.pdf (accessed on 1 May 2019).
- Partel, V.; Kakarla, S.C.; Ampatzidis, Y. Development and evaluation of a low-cost and smart technology for precision weed management utilizing artificial intelligence. Comput. Electron. Agric. 2019, 157, 339–350. [Google Scholar] [CrossRef]
- Van Evert, F.K.; Fountas, S.; Jakovetic, D.; Crnojevic, V.; Travlos, I.; Kempenaar, C. Big data for weed control and crop protection. Weed Res. 2017, 57, 218–233. [Google Scholar] [CrossRef]
- Rew, L.J.; Cousens, R.D. Spatial distributions of weeds in arable crops: Are current sampling and analytical methods appropriate? Weed Res. 2001, 41, 1–18. [Google Scholar] [CrossRef]
- Somerville, G.J.; Powles, S.B.; Walsh, M.J.; Renton, M. Modeling the impact of harvest weed seed control on herbicide-resistance evolution. Weed Sci. 2018, 66, 395–403. [Google Scholar] [CrossRef]
- Castillejo-González, I.L.; Peña-Barragán, J.M.; Jurado-Expósito, M.; Mesas-Carrascosa, F.J.; López-Granados, F. Evaluation of pixel- and object-based approaches for mapping wild oat (Avena sterilis) weed patches in wheat fields using QuickBird imagery for site-specific management. Eur. J. Agron. 2014, 59, 57–66. [Google Scholar] [CrossRef]
- Huang, Y.; Reddy, K.N.; Fletcher, R.S.; Pennington, D. UAV low-altitude remote sensing for precision weed management. Weed Technol. 2017, 32, 2–6. [Google Scholar] [CrossRef]
- Beckie, H.J. Herbicide-resistant weeds: Management tactics and practices. Weed Technol. 2006, 20, 793–814. [Google Scholar] [CrossRef]
- Moss, S. Integrated weed management (IWM): Why are farmers reluctant to adopt non-chemical alternatives to herbicides? Pest. Manag. Sci. 2019, 75, 1205–1211. [Google Scholar] [CrossRef] [PubMed]
- Davis, A.S.; Frisvold, G.B. Are herbicides a once in a century method of weed control? Pest Manag. Sci. 2017, 73, 2209–2220. [Google Scholar] [CrossRef]
- Frisvold, G.B. How low can you go? Estimating impacts of reduced pesticide use. Pest Manag. Sci. 2019, 75, 1223–1233. [Google Scholar] [CrossRef] [PubMed]



| Decade | Site of Action * | Example | WSSA | HRAC | Australia |
|---|---|---|---|---|---|
| 1930s | Uncouplers (membrane disruption) | Dinoterb | 24 | M | Z |
| 1940s | Synthetic auxins | 2,4-D | 4 | O | I |
| Auxin transport inhibitors | Diflufenzopyr | 19 | P | P | |
| Mitosis inhibitors | Propham | 23 | K2 | E | |
| 1950s | Microtubule assembly inhibitors | Trifluralin | 3 | K1 | D |
| PS-II inhibitors | Atrazine | 5 | C1 | C | |
| PS-II inhibitors (ureas and amides) | Chlorotoluron | 7 | C2 | C | |
| Lipid inhibitors | Triallate | 8 | N | J | |
| Carotenoid biosynthesis inhibitors | Amitrole | 11 | F3 | Q | |
| Nucleic acid inhibitors | MSMA | 17 | Z | Z | |
| PS-I electron diverters | Paraquat | 22 | D | L | |
| 1960s | PS-II inhibitors (nitriles) | Bromoxynil | 6 | C3 | C |
| PPO inhibitors | Oxyfluorfen | 14 | E | G | |
| VLCFA inhibitors | Metolachlor | 15 | K3 | K | |
| Lipid inhibitors | Ethofumesate | 16 | N | J | |
| DHP synthase inhibitors | Asulam | 18 | I | R | |
| Cellulose inhibitors | Dichlobenil | 20 | L | I,O,Z | |
| 1970s | ACCase inhibitors | Diclofop | 1 | A | A |
| ALS inhibitors | Chlorsulfuron | 2 | B | B | |
| Cell elongtion inhibitors | Difenzoquat | 8 | Z | Z | |
| EPSPS inhibitors | Glyphosate | 9 | G | M | |
| Glutamine synthase inhibitors | Glufosinate | 10 | H | N | |
| Carotenoid biosynthesis inhibitors (PDS) | Diflufenican | 12 | F1 | F | |
| Antimicrotubule mitotic disrupters | Flamprop | 25 | Z | Z | |
| 1980s | DOXP inhibitors | Clomazone | 13 | F4 | Q |
| Cellulose inhibitors | Dichlobenil | 21,26 | L | I,O,Z | |
| HPPD inhibitors | Isoxaflutole | 27 | F2 | H |
| HR Trait | Soybean | Maize | Cotton | Rice | Canola | Wheat |
|---|---|---|---|---|---|---|
| ACCase inhibitor | X | X | X | |||
| ALS inhibitor | X | X | X | X | ||
| Triazine * | X | |||||
| Glyphosate | X | X | X | X | ||
| Glufosinate | X | X | X | X | ||
| Glyphosate+glufosinate | X | X | X | |||
| Glyphosate+triazine* | X | |||||
| Glyphosate+dicamba | X | X | ||||
| Glyphosate+2,4-D+APP (ACCase) | X | |||||
| Glyphosate+isoxaflutole | X | |||||
| Glyphosate+glufosinate+dicamba | X | X | ||||
| Glyphosate+glufosinate+2,4-D | X | X | ||||
| Glyphosate+isoxaflutole+glufosinate | X |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Beckie, H.J.; Ashworth, M.B.; Flower, K.C. Herbicide Resistance Management: Recent Developments and Trends. Plants 2019, 8, 161. https://doi.org/10.3390/plants8060161
Beckie HJ, Ashworth MB, Flower KC. Herbicide Resistance Management: Recent Developments and Trends. Plants. 2019; 8(6):161. https://doi.org/10.3390/plants8060161
Chicago/Turabian StyleBeckie, Hugh J., Michael B. Ashworth, and Ken C. Flower. 2019. "Herbicide Resistance Management: Recent Developments and Trends" Plants 8, no. 6: 161. https://doi.org/10.3390/plants8060161
APA StyleBeckie, H. J., Ashworth, M. B., & Flower, K. C. (2019). Herbicide Resistance Management: Recent Developments and Trends. Plants, 8(6), 161. https://doi.org/10.3390/plants8060161






