Application of Loop-Mediated Isothermal Amplification in an Early Warning System for Epidemics of an Externally Sourced Plant Virus
Abstract
1. Introduction
2. Results
2.1. GPA-Specific LAMP Protocol Development and Validation
2.2. LAMP Protocol Field Validation
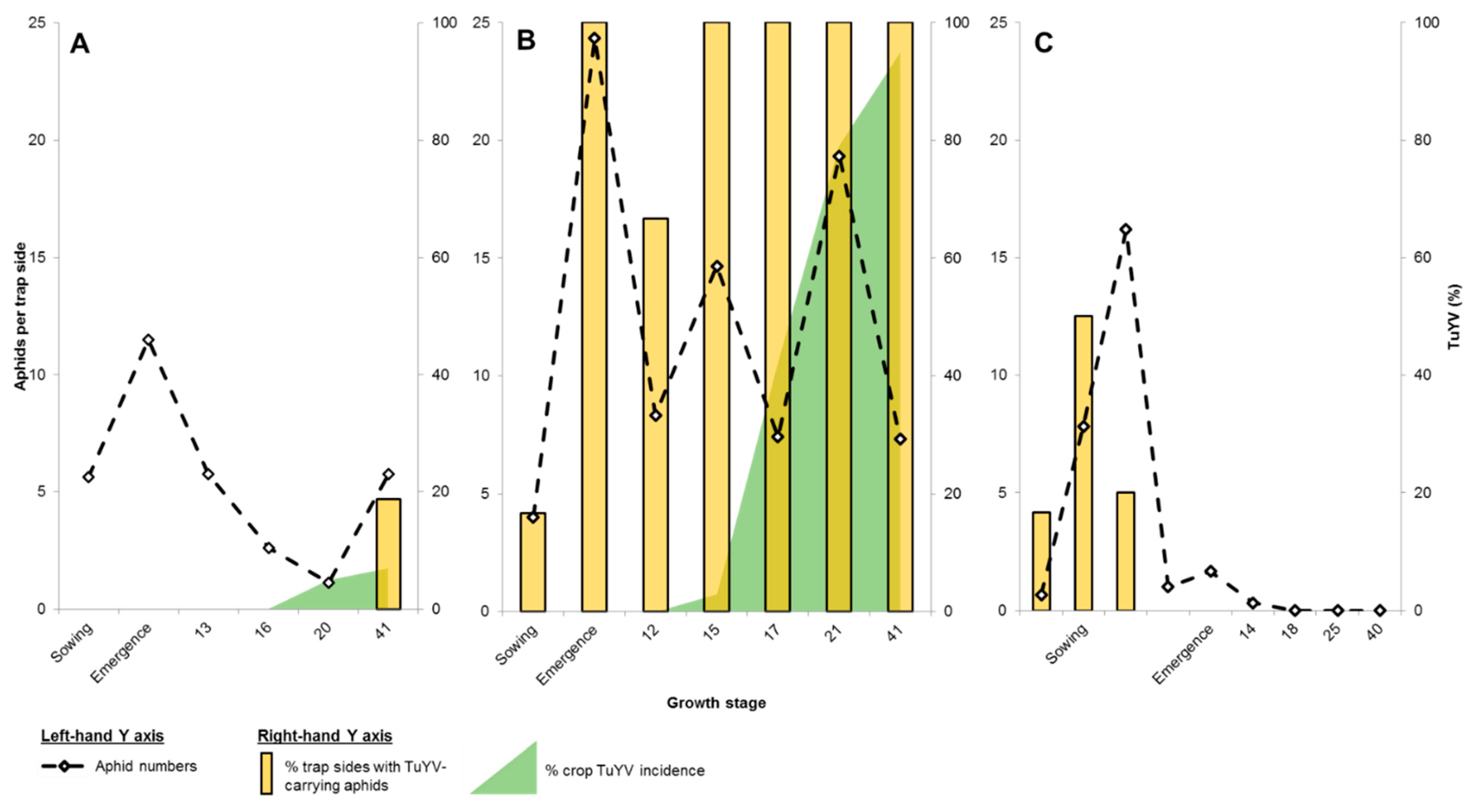
2.3. Aphid Numbers, LAMP Detection and Virus Incidence
2.3.1. 2017 Sites
2.3.2. 2018 Sites
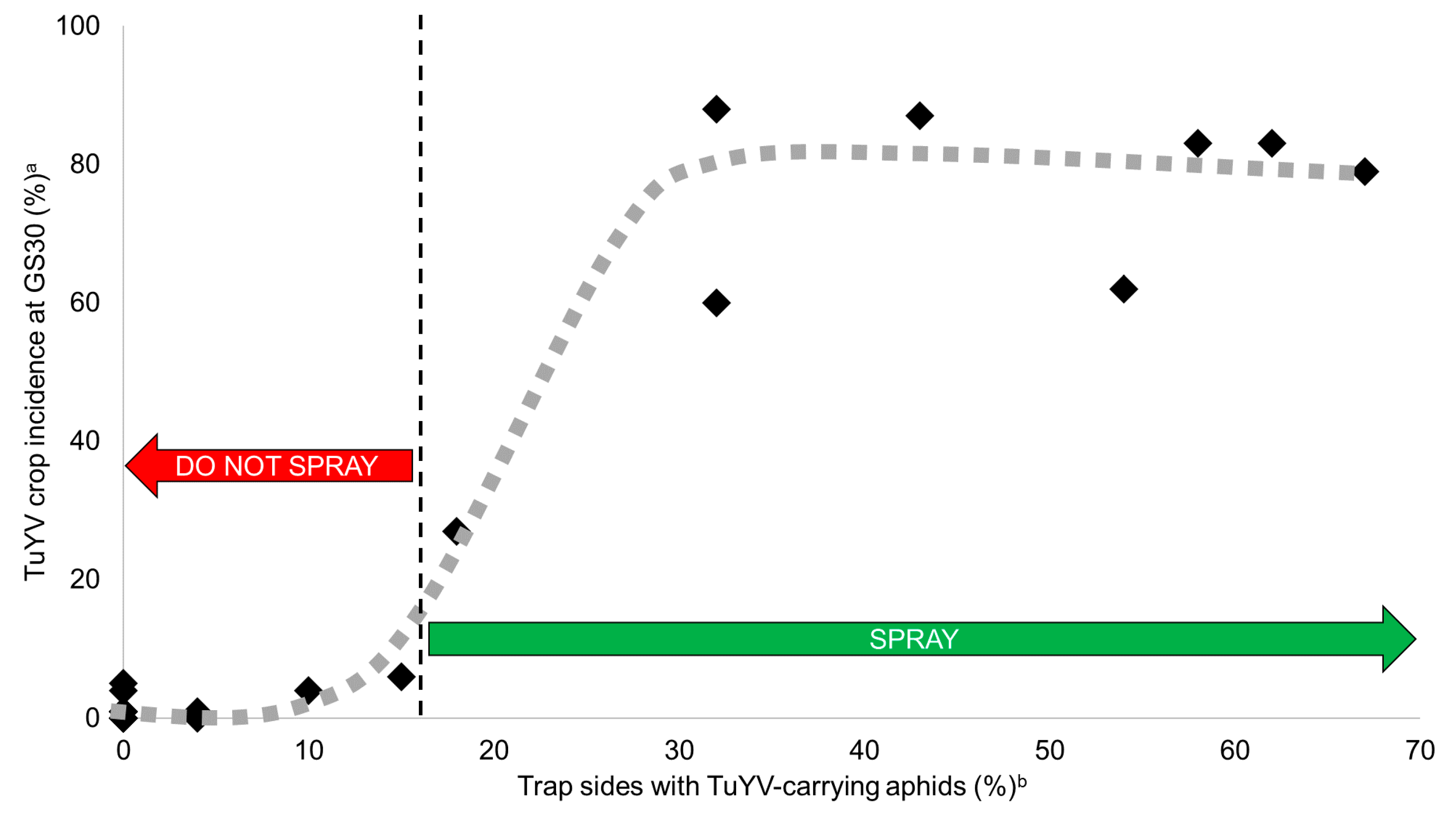
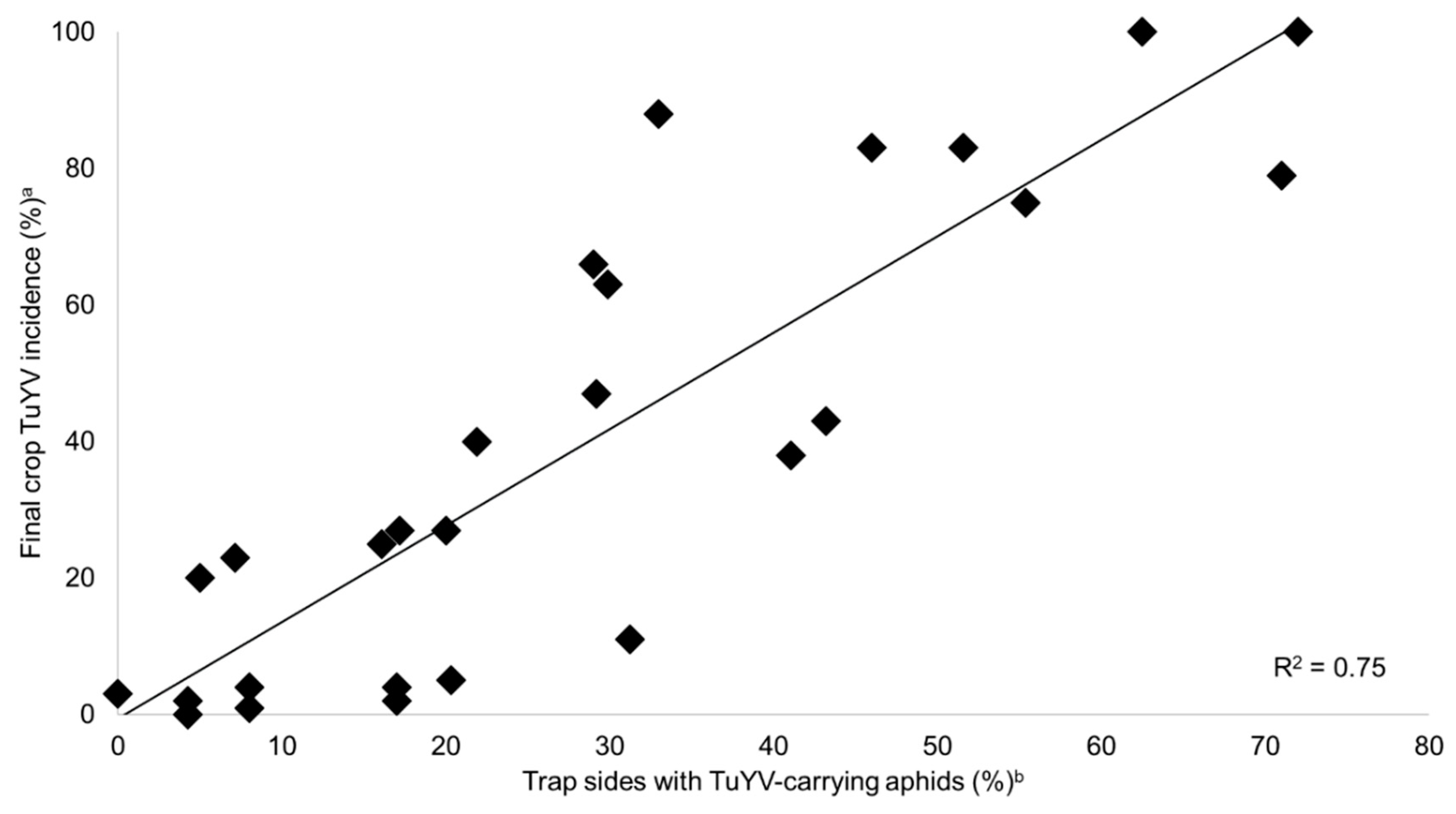
2.4. Predicting TuYV Epidemics
3. Discussion
4. Materials and Methods
4.1. Crude, Total RNA and Total DNA Extraction
4.2. LAMP
4.3. GPA-Specific LAMP Protocol Development and Validation
4.3.1. Primer Design
4.3.2. Specificity
4.3.3. Sensitivity
4.4. RT-PCR
4.5. ELISA
4.6. LAMP Protocol Field Validation
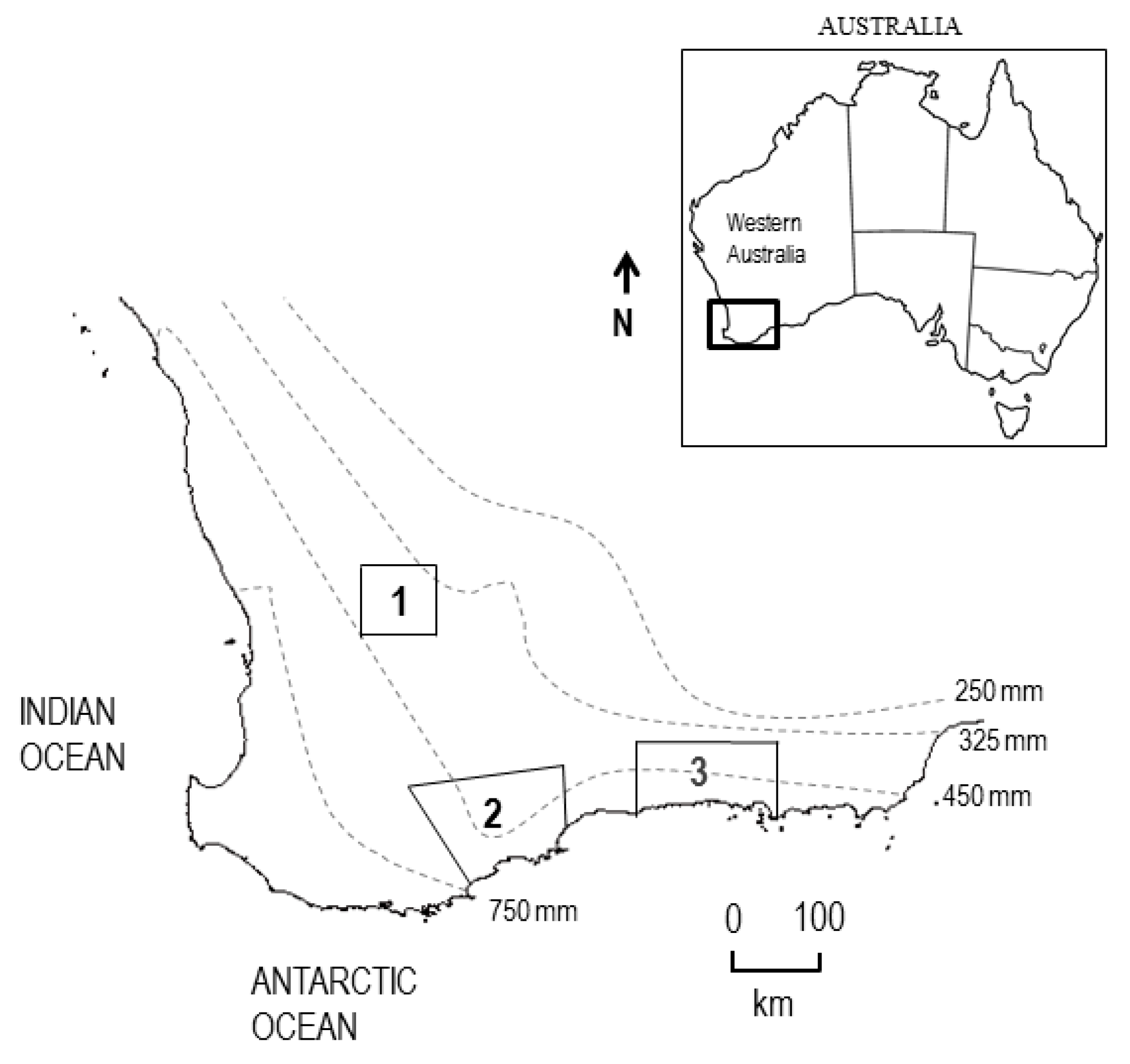
4.6.1. Site Location
4.6.2. Aphid Trapping and Testing
4.6.3. Green-Bridge Host and Canola Crop Testing
4.7. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Australian Bureau of Statistics. Value of Agricultural Commodities Produced; Cat. no. 7503.0; Commonwealth of Australia, Australian Bureau of Statistics: Belconnen, Australia, 2018. Available online: https://www.abs.gov.au/AUSSTATS/abs@.nsf/allprimarymainfeatures/58529ACD49B5ECE0CA2577A000154456?opendocument (accessed on 26 May 2019).
- Seymour, M.; Kirkegaard, J.A.; Peoples, M.B.; White, P.F.; French, R.J. Break-crop benefits to wheat in Western Australia—Insights from over three decades of research. Crop Pasture Sci. 2012, 63, 1–16. [Google Scholar] [CrossRef]
- Coutts, B.A.; Jones, R.A.C. Viruses infecting canola (Brassica napus) in south-west Australia: incidence, distribution, spread and infection reservoir in wild radish (Raphanus raphinistrum). Aust. J. Agric. Res. 2000, 51, 925–936. [Google Scholar] [CrossRef]
- Lancashire, P.D.; Bleiholder, H.; Boom, T.V.D.; Langelüddeke, P.; Stauss, R.; Weber, E.; Witzenberger, A. A uniform decimal code for growth stages of crops and weeds. Ann. Appl. Biol. 1991, 119, 561–601. [Google Scholar] [CrossRef]
- Graichen, K.; Schliephake, E. Infestation of winter oilseed rape by turnip yellows luteovirus and its effect on yield in Germany. In Proceedings of the 10th International Rapeseed Congress—New Horizons for an Old Crop, Canberra, Australia, 26–29 September 1999; International Consultative Group for Research on Rapeseed: Paris, France; pp. 131–136. Available online: http://gcirc.org/fileadmin/documents/Proceedings/IRC1999Canberravol1/43.htm (accessed on 26 May 2019).
- Jay, C.N.; Rossall, S.; Smith, H.G. Effects of beet western yellows virus on growth and yield of oilseed rape (Brassica napus). J. Agric. Sci. 1999, 133, 131–139. [Google Scholar] [CrossRef]
- Jones, R.A.C.; Coutts, B.A.; Hawkes, J.R. Yield-limiting potential of Beet western yellows virus in Brassica napus. Aust. J. Agric. Res. 2007, 58, 788–801. [Google Scholar] [CrossRef]
- Congdon, B.S.; Matson, P.; Begum, F.; Dore, A.; Kehoe, M.A.; Coutts, B.A. Turnip yellows virus epidemic in 2018—Time to get one step ahead of the green peach aphid. In Proceedings of the 2019 GRDC Research Updates, Perth, Australia, 26 February 2019; Grains Research and Development Corporation: Barton, Australia; pp. 84–85. Available online: https://grdc.com.au/resources-and-publications/grdc-update-papers/tab-content/grdc-update-papers/2019/02/1-turnip-yellows-virus-epidemic-in-2018-time-to-get-one-step-ahead-of-the-green-peach-aphid. (accessed on 26 May 2019).
- Weber, G. Genetic variability in host plant adaptation of the green peach aphid, Myzus persicae. Entomol. Exp. Appl. 1985, 38, 49–56. [Google Scholar] [CrossRef]
- Mathers, T.C.; Chen, Y.; Kaithakottil, G.; Legeai, F.; Mugford, S.T.; Baa-Puyoulet, P.; Bretaudeau, A.; Clavijo, B.; Colella, S.; Collin, O.; et al. Rapid transcriptional plasticity of duplicated gene clusters enables a clonally reproducing aphid to colonise diverse plant species. Genome Biol. 2017, 18, 27. [Google Scholar] [CrossRef]
- Schliephake, E.; Graichen, K.; Rabenstein, F. Investigations on the vector transmission of the Beet mild yellowing virus (BMYV) and the Turnip yellows virus (TuYV). J. Plant Dis. Protect. 2000, 107, 81–87. [Google Scholar]
- Anstead, J.A.; Williamson, M.S.; Denholm, I. Evidence for multiple origins of identical insecticide resistance mutations in the aphid Myzus persicae. Insect Mol. Biol. 2005, 35, 249–256. [Google Scholar] [CrossRef]
- Umina, P.A.; Edwards, O.; Carson, P.; Van Rooyen, A.; Anderson, A. High levels of resistance to carbamate and pyrethroid chemicals widespread in Australian Myzus persicae (Hemiptera: Aphididae) populations. J. Econ. Entomol. 2014, 107, 1626–1638. [Google Scholar] [CrossRef] [PubMed]
- Severtson, D.; Flower, K.; Nansen, C. Nonrandom distribution of cabbage aphids (Hemiptera: Aphididae) in dryland canola (Brassicales: Brassicaceae). Environ. Entomol. 2015, 44, 767–779. [Google Scholar] [CrossRef]
- Coutts, B.A.; Hawkes, J.R.; Jones, R.A.C. Occurrence of Beet western yellows virus and its aphid vectors in over-summering broad-leafed weeds and volunteer crop plants in the grainbelt region of south-western Australia. Aust. J. Agric. Res. 2006, 57, 975–982. [Google Scholar] [CrossRef]
- Latham, L.J.; Smith, L.J.; Jones, R.A.C. Incidence of three viruses in vegetable brassica plantings and wild radish weeds in south-west Australia. Australas. Plant Pathol. 2003, 32, 387–391. [Google Scholar] [CrossRef]
- Maling, T.; Diggle, A.J.; Thackray, D.J.; Siddique, K.H.M.; Jones, R.A.C. An epidemiological model for externally acquired vector-borne viruses applied to Beet western yellows virus in Brassica napus crops in a Mediterranean-type environment. Crop Pasture Sci. 2010, 61, 132–144. [Google Scholar] [CrossRef]
- Coutts, B.A.; Webster, C.G.; Jones, R.A.C. Control of Beet western yellows virus in Brassica napus crops: Infection resistance in Australian genotypes and effectiveness of imidacloprid seed dressing. Crop Pasture Sci. 2010, 61, 321–330. [Google Scholar] [CrossRef]
- Sekulic, G.; Rempel, C.B. Evaluating the role of seed treatments in canola/oilseed Rape production: integrated pest management, pollinator health, and biodiversity. Plants 2016, 5, 32. [Google Scholar] [CrossRef]
- Stamm, M.D.; Heng-Moss, T.M.; Baxendale, F.P.; Siegfried, B.D.; Blankenship, E.E.; Nauen, R. Uptake and translocation of imidacloprid, clothianidin and flupyradifurone in seed-treated soybeans. Pest Manag. Sci. 2016, 72, 1099–1109. [Google Scholar] [CrossRef]
- De Little, S.C.; Edwards, O.; van Rooyen, A.R.; Weeks, A.; Umina, P.A. Discovery of metabolic resistance to neonicotinoids in green peach aphids (Myzus persicae) in Australia. Pest Manag. Sci. 2017, 73, 1611–1617. [Google Scholar] [CrossRef]
- Sparks, T.C.; Watson, G.B.; Loso, M.R.; Geng, C.; Babcock, J.M.; Thomas, J.D. Sulfoxaflor and the sulfoximine insecticides: Chemistry, mode of action and basis for efficacy on resistant insects. Pestic. Biochem. Phys. 2013, 107, 1–7. [Google Scholar] [CrossRef]
- Annetts, R.; Elias, N.; Corr, I. Transform™ Insecticide (sulfoxaflor) for control of aphids in canola. In Proceedings of the 16th Agronomy Conference 2012, Armidale, Australia, 14 October 2012; Agronomy Australia: Australia. Available online: http://agronomyaustraliaproceedings.org/images/sampledata/2012/8476_5_annetts.pdf. (accessed on 26 May 2019).
- Bass, C.; Puinean, A.M.; Andrews, M.; Cutler, P.; Daniels, M.; Elias, J.; Paul, V.L.; Crossthwaite, A.J.; Denholm, I.; Field, L.M.; et al. Mutation of a nicotinic acetylcholine receptor β subunit is associated with resistance to neonicotinoid insecticides in the aphid Myzus persicae. BMC Neurosci. 2011, 12, 51. [Google Scholar] [CrossRef]
- Bass, C.; Denholm, I.; Williamson, M.S.; Nauen, R. The global status of insect resistance to neonicotinoid insecticides. Pestic. Biochem. Phys. 2015, 121, 78–87. [Google Scholar] [CrossRef] [PubMed]
- Bass, C.; Puinean, A.M.; Zimmer, C.T.; Denholm, I.; Field, L.M.; Foster, S.P.; Gutbrod, O.; Nauen, R.; Slater, R.; Williamson, M.S. The evolution of insecticide resistance in the peach potato aphid, Myzus persicae. Insect Biochem. Mol. Biol. 2014, 51, 41–51. [Google Scholar] [CrossRef]
- Congdon, B.S.; Kehoe, M.A.; Filardo, F.F.; Coutts, B.A. In-field capable loop-mediated isothermal amplification detection of Turnip yellows virus in plants and its principal aphid vector Myzus persicae. J. Virol. Methods 2019, 265, 15–21. [Google Scholar] [CrossRef]
- Fabre, F.; Kervarrec, C.; Mieuzet, L.; Riault, G.; Vialatte, A.; Jacquot, E. Improvement of Barley yellow dwarf virus-PAV detection in single aphids using a fluorescent real time RT-PCR. J. Virol. Methods 2003, 110, 51–60. [Google Scholar] [CrossRef]
- Marroquín, C.; Olmos, A.; Teresa Gorris, M.A.; Bertolini, E.; Carmen Martínez, M.; Carbonell, E.A.; Hermoso de Mendoza, A.; Cambra, M. Estimation of the number of aphids carrying Citrus tristeza virus that visit adult citrus trees. Virus Res. 2004, 100, 101–108. [Google Scholar] [CrossRef]
- Cambra, M.; Bertolini, E.; Olmos, A.; Capote, N. Molecular methods for detection and quantitation of virus in aphids. In Virus Disease and Crop Biosecurity; Cooper, J.I., Kuehne, T., Polischuk, V., Eds.; NATO series C; Environmental Security, Springer: Dordecht, The Netherlands, 2006; pp. 81–88. [Google Scholar]
- Fabre, F.; Plantegenest, M.; Mieuzet, L.; Dedryver, C.A.; Leterrier, J.-L.; Jacquot, E. Effects of climate and land use on the occurrence of viruliferous aphids and the epidemiology of barley yellow dwarf disease. Agric. Ecosyst. Environ. 2005, 106, 49–55. [Google Scholar] [CrossRef]
- Jones, R.A.C. Control of plant virus diseases. Adv. Virus Res. 2006, 67, 205–244. [Google Scholar]
- Maling, T.; Diggle, A.J.; Thackray, D.J.; Siddique, K.H.M.; Jones, R.A.C. An epidemiological model for externally sourced vector-borne viruses applied to Bean yellow mosaic virus in lupin crops in a Mediterranean-type environment. Phytopathology 2008, 98, 1280–1290. [Google Scholar] [CrossRef]
- Thackray, D.J.; Diggle, A.J.; Berlandier, F.A.; Jones, R.A.C. Forecasting aphid outbreaks and epidemics of Cucumber mosaic virus in lupin crops in a Mediterranean-type environment. Virus Res. 2004, 100, 67–82. [Google Scholar] [CrossRef]
- Farré, I.; Robertson, M.J.; Walton, G.H.; Asseng, S. Simulating phenology and yield response of canola to sowing date in Western Australia using the APSIM model. Aust. J. Agric. Res. 2002, 53, 1155–1164. [Google Scholar] [CrossRef]
- Severtson, D.; Congdon, B.S.; Valentine, C. Apps, traps and LAMP’s: ‘Smart’ improvements to pest and disease management. In Proceedings of the 2018 Grains Research Updates, Perth, Western Australia, 26 February 2018; Grains Research and Development Corporation: Barton, Australia; p. 71. [Google Scholar]
- Coeur d’acier, A.; Cruaud, A.; Artige, E.; Genson, G.; Clamens, A.-L.; Pierre, E.; Hudaverdian, S.; Simon, J.-C.; Jousselin, E.; Rasplus, J.-Y. DNA barcoding and the associated PhylAphidB@se website for the identification of european aphids (Insecta: Hemiptera: Aphididae). PLoS ONE 2014, 9, e97620. [Google Scholar] [CrossRef] [PubMed]
- Clark, M.F.; Adams, A.N. Characteristics of the microplate method of enzyme-linked immunosorbent assay for the detection of plant viruses. J. Gen. Virol. 1977, 34, 475–483. [Google Scholar] [CrossRef] [PubMed]
- Gibbs, A.J.; Gower, J.C. The use of a multiple-transfer method in plant virus transmission studies—Some statistical points arising in the analysis of results. Ann. Appl. Biol. 1960, 48, 75–83. [Google Scholar] [CrossRef]
| Dilution | Positives | Mean Time to Positive (min) |
|---|---|---|
| 1/1 a | 4/4 b | 19.6 ± 0.9 c |
| 1/10 | 4/4 | 19.2 ± 0.7 |
| 1/20 | 4/4 | 17.3 ± 0.6 |
| 1/50 | 4/4 | 19.1 ± 0.5 |
| 1/100 | 4/4 | 21.5 ± 0.7 |
| 0/100 | 4/4 | - |
| Negative | 0/4 | - |
| Positive | 4/4 | 17.7 ± 0.5 |
| Location | Year | Zone a | TuYV Detection in Broad-Leaf Weeds b | Mean Aphids Per Trap Side Pre-Emergence to GS15 c | Trap Sides with TuYV-Carrying Aphids Pre-Emergence to GS15 (%) | Trap Sides with GPA Pre-Emergence to GS15 (%) | Crop TuYV Incidence at GS30 (%) d | Crop TuYV Incidence at GS75 (%) |
|---|---|---|---|---|---|---|---|---|
| Irish Town | 2017 | 1 | 100% VC | 5 | 32 | 32 | 60 | 100 |
| Kojaneerup | 2017 | 2 | Nil | 2 | 18 | 25 | 27 | 47 |
| Coomalbidgup | 2017 | 3 | Nil | 8 | 10 | 21 | 5 | 83 |
| Jerramungup | 2017 | 2 | Nil | 2 | 0 | 22 | 4 | 38 |
| Kendenup | 2017 | 2 | Nil | 2 | 0 | 9 | 4 | 27 |
| Wongamine | 2017 | 1 | Nil | 2 | 0 | 19 | 1 | 41 |
| Munglinup | 2017 | 3 | Nil | 4 | 0 | 25 | 1 | 27 |
| South Stirlings | 2017 | 2 | <1% WR | 2 | 4 | 0 | 1 | 25 |
| Mount Barker | 2017 | 2 | Nil | 5 | 0 | 29 | 1 | 23 |
| Esperance Downs | 2017 | 3 | Nil | 9 | 0 | 8 | 1 | 12 |
| Nunile | 2017 | 1 | <1% WR | 2 | 0 | 6 | 1 | 11 |
| Gairdner | 2017 | 2 | Nil | 3 | 0 | 0 | 0 | 75 |
| Wellstead | 2017 | 2 | Nil | 2 | 4 | 0 | 0 | 43 |
| Gibson | 2017 | 3 | Nil | 5 | 0 | 10 | 0 | 20 |
| Esperance Downs | 2018 | 3 | Nil | 7 | 32 | 39 | 88 | 100 |
| Munglinup | 2018 | 3 | Nil | 10 | 43 | 32 | 87 | 100 |
| Gibson | 2018 | 3 | <1% SC | 14 | 62 | 33 | 83 | 100 |
| Dalyup | 2018 | 3 | - | 11 | 58 | 33 | 83 | 100 |
| Coomalbidgup | 2018 | 3 | Nil | 11 | 67 | 14 | 79 | 100 |
| Grass Patch | 2018 | 3 | Nil | 6 | 54 | 29 | 62 | 100 |
| Jerramungup | 2018 | 2 | Nil | 1 | 15 | 15 | 6 | 63 |
| Gairdner | 2018 | 2 | Nil | 0 | 0 | 0 | 0 | - |
| Nunile | 2018 | 1 | Nil | 2 | 0 | 8 | 0 | 4 |
| Wongamine | 2018 | 1 | Nil | 1 | 0 | 0 | 0 | 4 |
| South Stirlings | 2018 | 2 | Nil | 1 | 0 | 0 | 0 | 3 |
| Bejoording | 2018 | 1 | Nil | 1 | 0 | 4 | 0 | 2 |
| Mount Barker | 2018 | 2 | Nil | 0 | 0 | 0 | 0 | 2 |
| Coondle | 2018 | 1 | - | 0 | 0 | 0 | 0 | 1 |
| Tenterdon | 2018 | 2 | Nil | 0 | 0 | 0 | 0 | 1 |
| Kendenup | 2018 | 2 | Nil | 0 | 0 | 0 | 0 | 0 |
| Primer | Type | Position on Genome a | Length (nt) | Sequence 5′-3′ |
|---|---|---|---|---|
| F3 | Forward outer | 897-914 | 18 | TGATGTCACCCTCCTCCG |
| B3 | Backward outer | 1084-1102 | 19 | AGTGTCCTCCTTCCGTGTG |
| FIP | Forward inner | 970-991 and 926-945 | 42 | TGCATTTTGCTAGGTTGGCAGCATTGGGAAGGACTGTTAGGC |
| BIP | Backward inner | 1019-1040 and 1064-1083 | 42 | ATGGCTGGGTTAGCGGTTATGCGCTCAGGACCATAACATCGG |
| LF2 | Loop forward outer | 946-964 | 19 | TGACGTTGGCCGCTTTACA |
| LB2 | Loop backward outer | 1041-1062 | 22 | CGAGATTGTAGGCTCAGAAGGT |
| Primer | Type | Position on Gene a | Length (nt) | Sequence 5′-3′ |
|---|---|---|---|---|
| F3 | Forward outer | 339-356 | 18 | TACAGCCGTCAGCAAGGA |
| B3 | Backward outer | 534-553 | 20 | CAGTCTGATCAGAAGGCGAG |
| FIP | Forward inner | 406-426 and 362-281 | 41 | TAAGTTACGGCCGGTGTCCGTCCAGGGATTTCATGGCAGTG |
| BIP | Backward inner | 435-455 and 495-514 | 41 | CGATGTTACCAAGTGGCCCGCGTACCAAAGCCAATCCTCGG |
| LF2 | Loop forward outer | 382-403 | 22 | GATCCCTGACTACATCTGGGAA |
| LB2 | Loop backward outer | 456-479 | 24 | AAAGCTGTTGCAATACAATGTGCC |
| Year | Zone a | Location | February to April Rainfall (mm) | Cultivar | Neonicotinoid Seed Treatment | Sowing Date | Date Aphid Trap First Deployed b | Green-Bridge Species Tested (Number of Plants) c |
|---|---|---|---|---|---|---|---|---|
| 2017 | 1 | Irish Town | 123 | ATR Bonito | No | 1-May | 8-Apr | VC (20) |
| 2017 | 1 | Nunile | 123 | InVigor T4510 | Yes | 16-May | 8-Apr | AM (100), BN (100), CG (100), WR (100) |
| 2017 | 1 | Wongamine | 123 | Pioneer 43Y23 | Yes | 12-May | 8-Apr | AM (100), CG (100), VC (100), WR (100) |
| 2017 | 2 | Gairdner | 160 | ATR Bonito | No | 24-Apr | 23-Mar | AM (100), BN (100), SC (100), Se (100), VC (100), WR (100) |
| 2017 | 2 | Jerramungup | 131 | ATR Bonito | Yes | 20-Apr | 23-Mar | BN (100), CG (100), SC (100), VC (100), WR (100) |
| 2017 | 2 | Kendenup | 102 | ATR Mako | Yes | 8-May | 23-Mar | BN (100), FF (100) |
| 2017 | 2 | Kojaneerup | 137 | ATR Mako | No | 13-Jun | 23-Mar | AM (100), BN (100), CG (100), SC (100), Se (100), VC (100), WR (100) |
| 2017 | 2 | Mount Barker | 185 | ATR Mako | Yes | 1-May | 23-Mar | Nil |
| 2017 | 2 | Wellstead | 243 | Thumper TT | Yes | 20-Apr | 23-Mar | BN (100), CG (100), Se (100), SC (100) |
| 2017 | 2 | South Stirlings | 137 | Nuseed GT-53 | Yes | 22-Apr | 23-Mar | WR (100) |
| 2017 | 3 | Coomalbidgup | 256 | ATR Wahoo | Yes | 25-Apr | 30-Mar | BN (100), CG (100), VC (100), SC (100) |
| 2017 | 3 | Esperance Downs | 221 | ATR Mako | Yes | 30-Apr | 30-Mar | Se (100) |
| 2017 | 3 | Gibson | 221 | ATR Bonito | Yes | 28-Apr | 30-Mar | WR (100) |
| 2017 | 3 | Munglinup | 256 | Hyola 559TT | Yes | 18-Apr | 30-Mar | BN (100), WR (100) |
| 2018 | 1 | Bejoording | 16 | Nuseed GT-53 | Yes | 21-Apr | 20-Mar | AM (3), WR (62) |
| 2018 | 1 | Coondle | 16 | ATR Bonito | Yes | 14-May | 10-Jul | - |
| 2018 | 1 | Nunile | 16 | Pioneer 44Y27 | Yes | 26-Apr | 20-Mar | CST (10), So (100), WR (30) |
| 2018 | 1 | Wongamine | 16 | Pioneer 44Y27 | Yes | 25-Apr | 20-Mar | So (50), WR (6) |
| 2018 | 2 | Gairdner | 38 | ATR Mako | No | 10-Apr | 27-Mar | Nil |
| 2018 | 2 | Jerramungup | 42 | ATR Bonito | No | 10-Apr | 27-Mar | AM (6), BN (2), CG (2), CST (6), FF (2), MM (15), SC (2), WR (8) |
| 2018 | 2 | Kendenup | 39 | InVigor T4510 | Yes | 23-May | 27-Mar | AM (10), WR (15) |
| 2018 | 2 | Mount Barker | 39 | InVigor T4510 | Yes | 11-May | 27-Mar | AM (10), SC (5), VC (20), WR (20) |
| 2018 | 2 | Tenterdon | 36 | InVigor T4510 | Yes | 3-May | 27-Mar | Nil |
| 2018 | 2 | South Stirlings | 44 | Pioneer 45Y25 | Yes | 16-Apr | 27-Mar | Nil |
| 2018 | 3 | Coomalbidgup | 78 | Hyola 404RR | Yes | 5-May | 27-Mar | AM (5), BN (3), CG (100), CST (16), WR (45) |
| 2018 | 3 | Dalyup | 78 | Pioneer 44Y27 | Yes | 15-May | 19-Jun | - |
| 2018 | 3 | Esperance Downs | 146 | ATR Bonito | Yes | 21-May | 27-Mar | AM (4), SC (20), WR (7), VC (7) |
| 2018 | 3 | Gibson | 146 | ATR Bonito | Yes | 28-Apr | 27-Mar | MM (30), SC (65), WR (30), |
| 2018 | 3 | Grass Patch | 186 | ATR Bonito | No | 7-Apr | 27-Mar | MM (10), WR (18) |
| 2018 | 3 | Munglinup | 78 | Hyola 559TT | Yes | 20-Apr | 27-Mar | CST (10), VC (130), WR (95) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Congdon, B.; Matson, P.; Begum, F.; Kehoe, M.; Coutts, B. Application of Loop-Mediated Isothermal Amplification in an Early Warning System for Epidemics of an Externally Sourced Plant Virus. Plants 2019, 8, 139. https://doi.org/10.3390/plants8050139
Congdon B, Matson P, Begum F, Kehoe M, Coutts B. Application of Loop-Mediated Isothermal Amplification in an Early Warning System for Epidemics of an Externally Sourced Plant Virus. Plants. 2019; 8(5):139. https://doi.org/10.3390/plants8050139
Chicago/Turabian StyleCongdon, Benjamin, Paul Matson, Farhana Begum, Monica Kehoe, and Brenda Coutts. 2019. "Application of Loop-Mediated Isothermal Amplification in an Early Warning System for Epidemics of an Externally Sourced Plant Virus" Plants 8, no. 5: 139. https://doi.org/10.3390/plants8050139
APA StyleCongdon, B., Matson, P., Begum, F., Kehoe, M., & Coutts, B. (2019). Application of Loop-Mediated Isothermal Amplification in an Early Warning System for Epidemics of an Externally Sourced Plant Virus. Plants, 8(5), 139. https://doi.org/10.3390/plants8050139




