Natural Compounds That Modulate the Development of the Fungus Botrytis cinerea and Protect Solanum lycopersicum
Abstract
1. Introduction
2. Results
2.1. Effects of Compounds on Several Growth Parameters
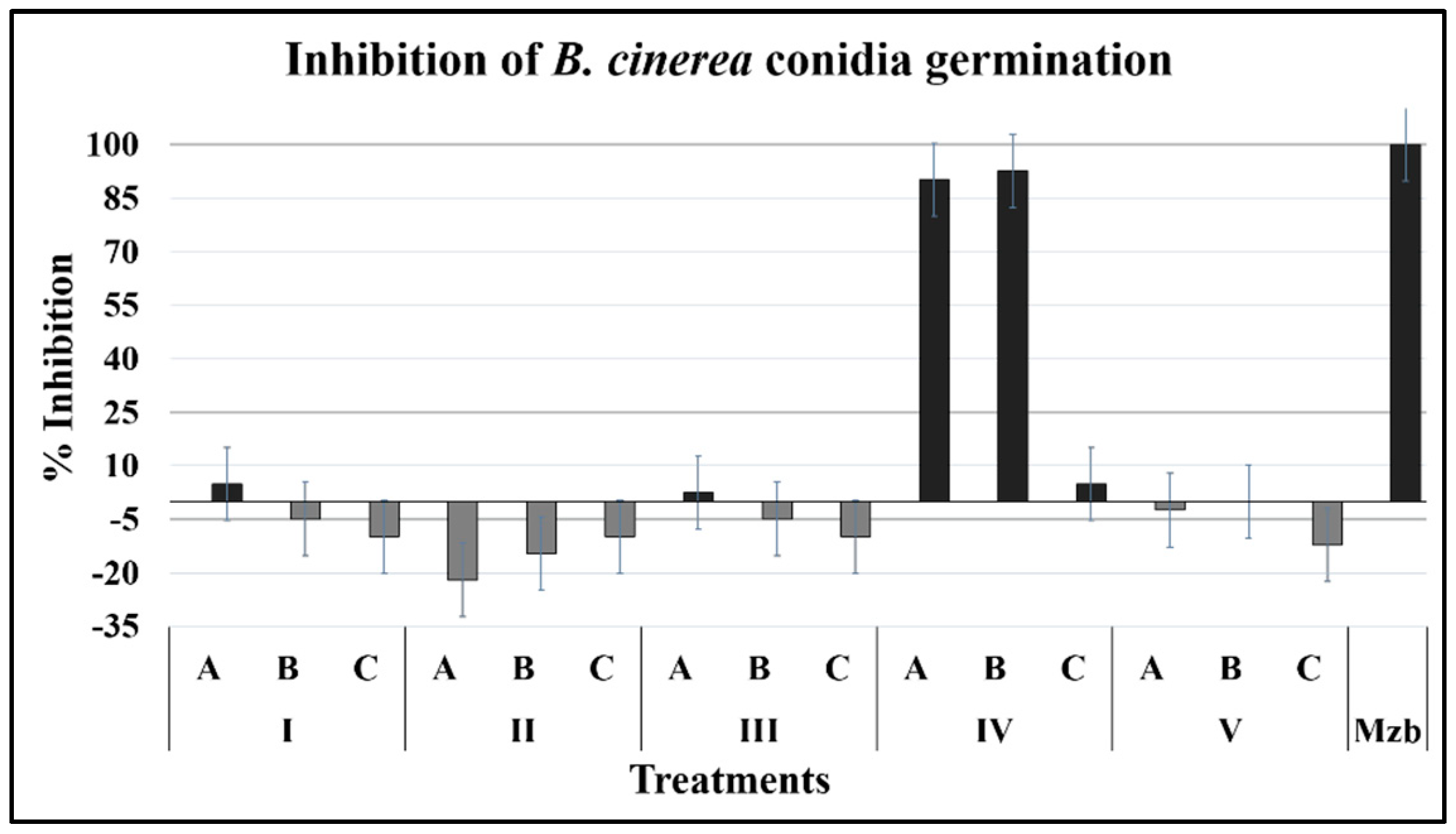
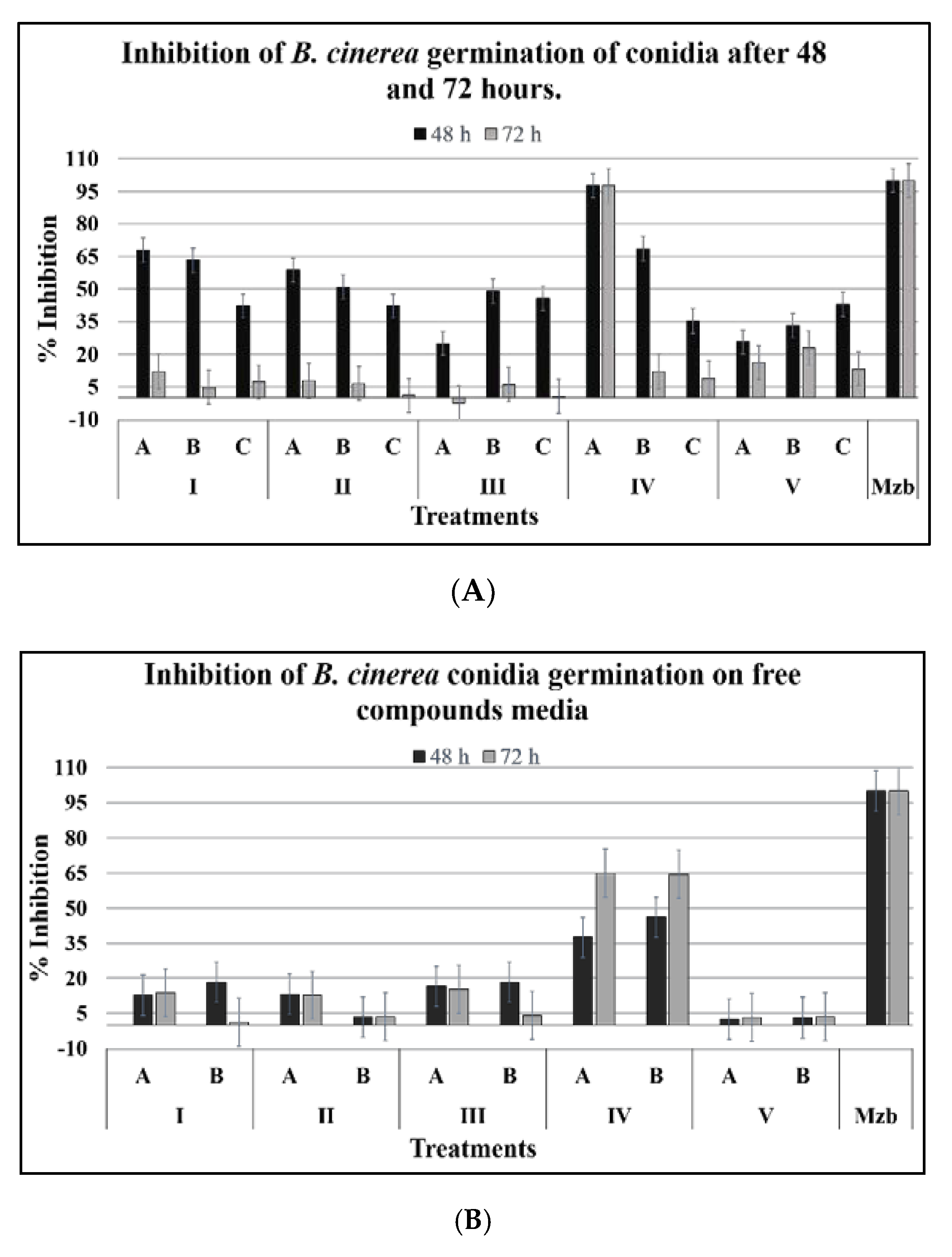
2.1.1. Conidia Germination
2.1.2. Development of Germ Tubes
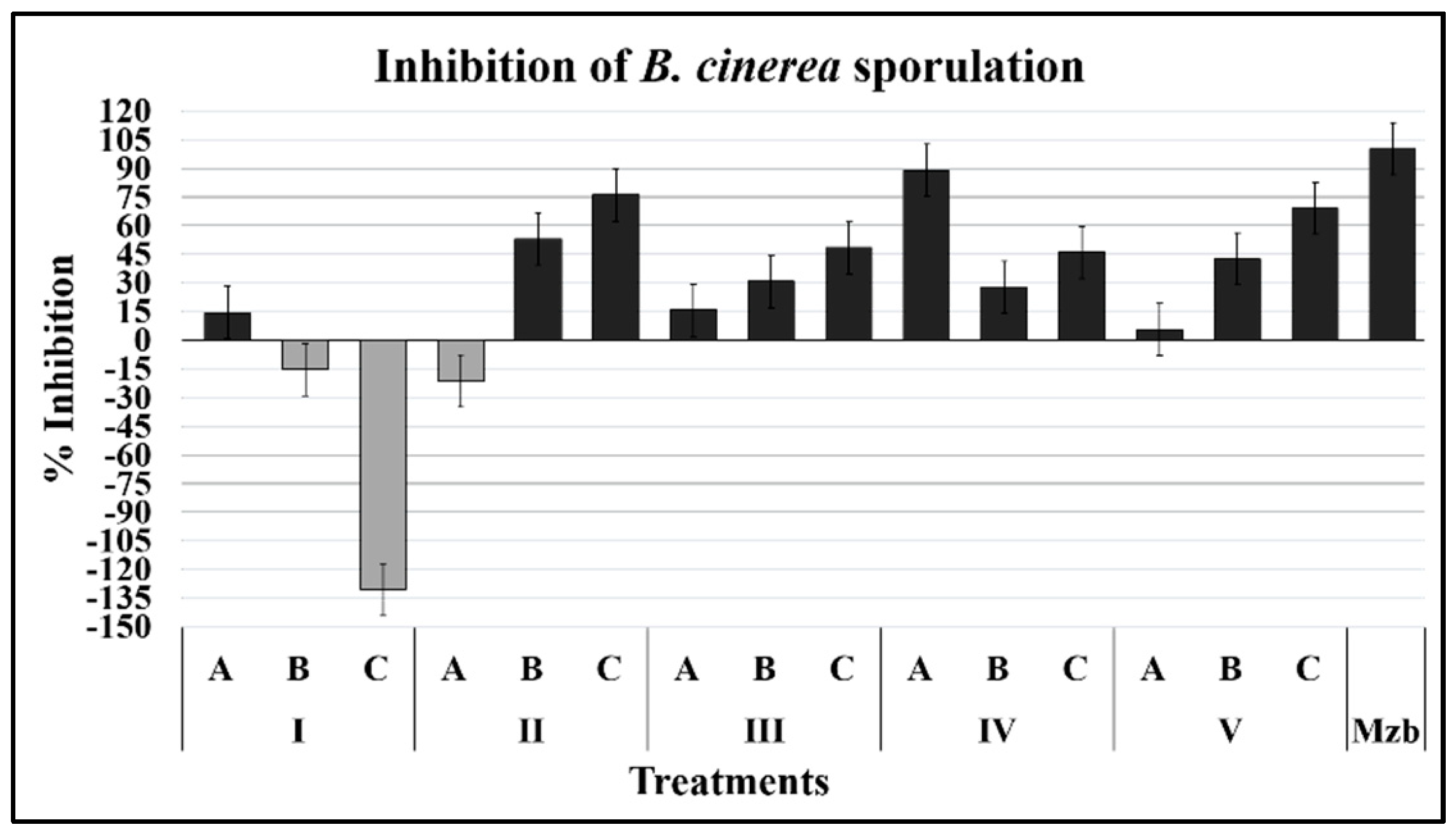
2.1.3. Sporulation
2.2. Phytopathological Tests
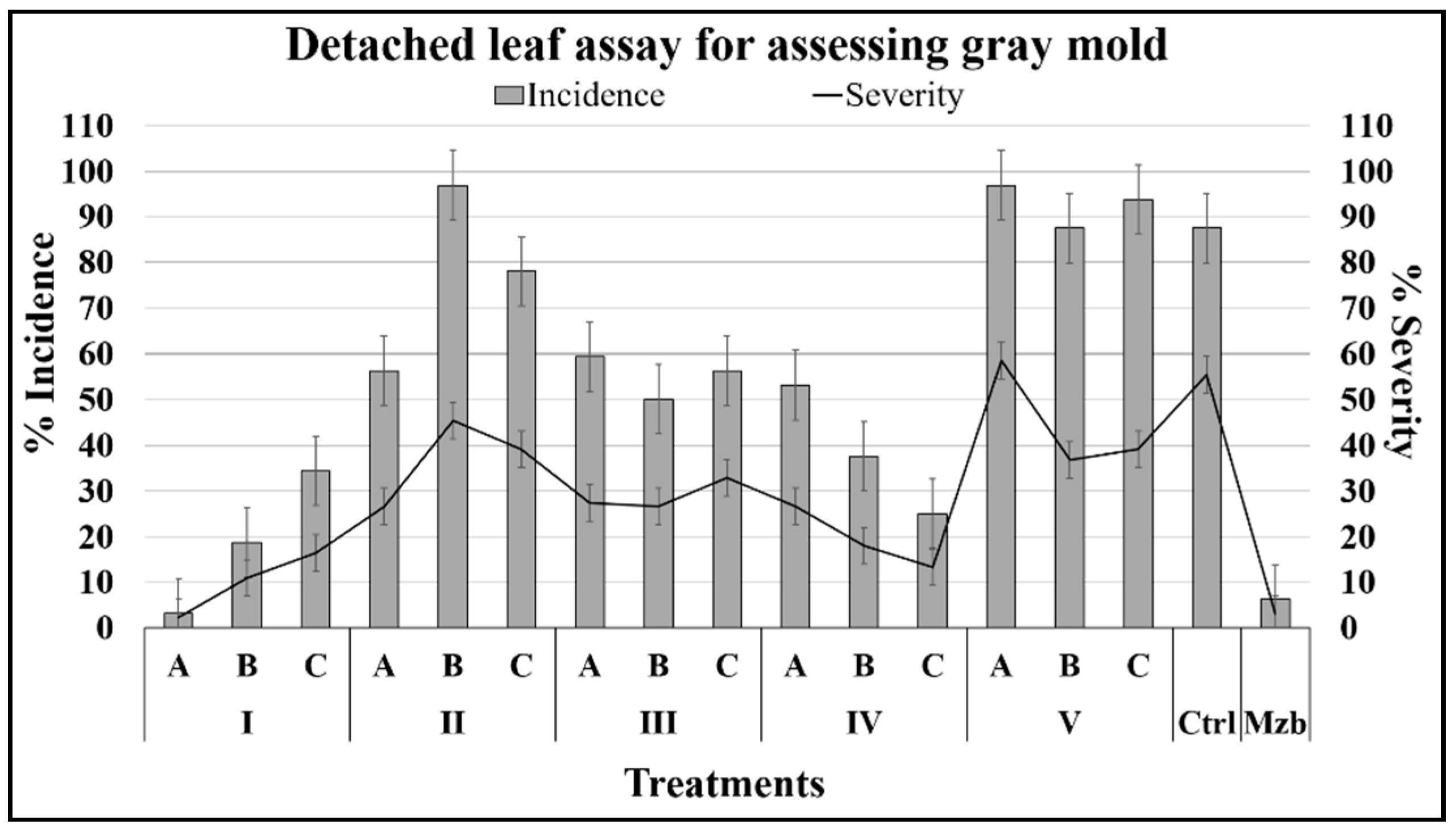
2.2.1. Effects on Foliage Infected with B. cinerea
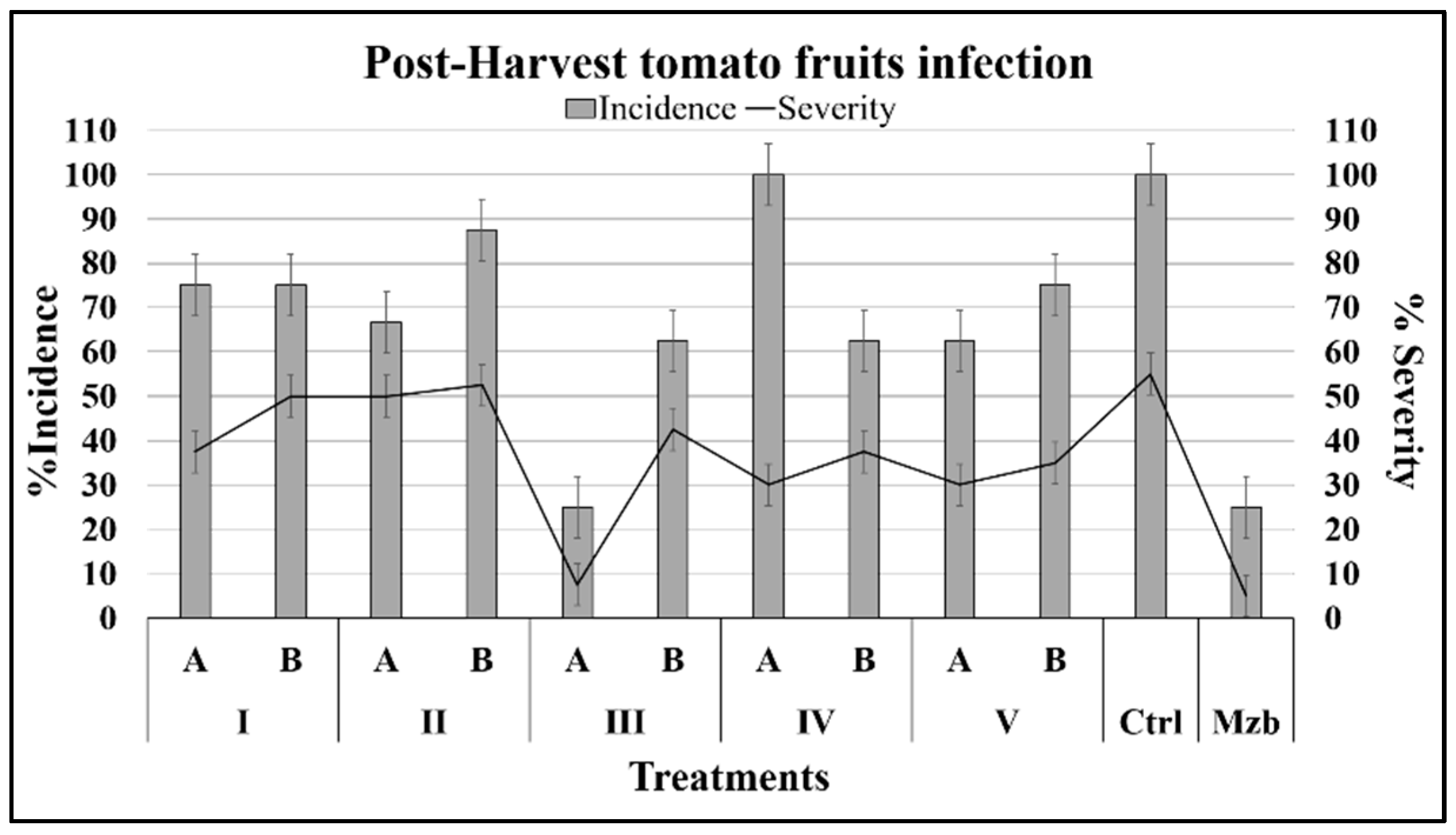
2.2.2. Effects on Fruits Infected with B. cinerea
3. Discussion
4. Materials and Methods
4.1. Equipment and Software
4.2. Fungal Strain
4.3. Plant Material
4.4. Treatments
4.5. Evaluation of Fungal Growth Parameters
4.5.1. Effect on Germination
4.5.2. Effect on Germ Tube Formation
4.5.3. Effect on Sporulation
4.6. Phytopathological Assays
4.6.1. Protection of Detached Leaves
4.6.2. Protection of Tomato Fruit
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Elad, Y.; Pertot, I.; Cotes Prado, A.M.; Stewart, A. Plant Hosts of Botrytis spp. In Botrytis—the Fungus, the Pathogen and Its Management in Agricultural Systems; Springer International Publishing: Cham, Switzerland, 2016; pp. 413–486. [Google Scholar]
- Breen, J.; Mur, L.; Sivakumaran, A.; Akinyemi, A.; Wilkinson, M.; Rodriguez Lopez, C.M. DNA methylation plays a role on in vitro culture induced loss of virulence in Botrytis cinerea. bioRxiv 2016. [Google Scholar] [CrossRef]
- Borges, Á.V.; Moreira Saraiva, R.; Maffia, L.A. Key factors to inoculate Botrytis cinerea in tomato plants. Summa Phytopathol. 2014, 40, 221–225. [Google Scholar] [CrossRef]
- Leroux, P. Chemical control of botrytis and its resistance to chemical fungicides. In Botrytis: Biology, Pathology and Control; Elad, Y., Williamson, B., Tudzynski, P., Kluwer, D.N., Eds.; Springer: Dordrecht, The Netherlands, 2007; pp. 195–222. [Google Scholar]
- Fillinger, S.; Walker, A.-S. Chemical control and resistance management of botrytis diseases. In Botrytis—The Fungus, the Pathogen and Its Management in Agricultural Systems; Springer International Publishing: Cham, Switzerland, 2016; pp. 189–216. [Google Scholar]
- Chen, S.N.; Luo, C.X.; Hu, M.J.; Schnabel, G. Fitness and competitive ability of Botrytis cinerea isolates with resistance to multiple chemical classes of fungicides. Phytopathology 2016, 106, 997–1005. [Google Scholar] [CrossRef] [PubMed]
- Vitale, A.; Panebianco, A.; Polizzi, G. Baseline sensitivity and efficacy of fluopyram against Botrytis cinerea from table grape in Italy. Ann. Appl. Biol. 2016, 169, 36–45. [Google Scholar] [CrossRef]
- Ahmed, F.A.; Sipes, B.S.; Alvarez, A.M. Natural products to control postharvest gray mold of tomato fruit-possible mechanisms. J. Plant Pathol. Microbiol. 2016, 7, 1–7. [Google Scholar] [CrossRef]
- Liu, P.; Cheng, Y.; Yang, M.; Liu, Y.; Chen, K.; Long, C.A.; Deng, X. Mechanisms of action for 2-phenylethanol isolated from Kloeckera apiculata in control of Penicillium molds of citrus fruits. BMC Microbiol. 2014, 14, 242. [Google Scholar] [CrossRef] [PubMed]
- Witzany, G. (Ed.) Introduction: Keylevels of biocommunication in fungi. In Biocommunication of Fungi; Springer: Cham, Switzerland, 2012; pp. 1–18. ISBN 9789400742642. [Google Scholar]
- Nassr, S.; Barakat, R. Effect of factors on conidium germination of Botrytis cinerea in vitro. Int. J. Plant Soil Sci. 2013, 2, 41–54. [Google Scholar] [CrossRef]
- Bhatia, S.P.; Wellington, G.A.; Cocchiara, J.; Lalko, J.; Letizia, C.S.; Api, A.M. Fragrance material review on 3-phenyl-1-propanol. Food Chem. Toxicol. 2011, 49, S246–S251. [Google Scholar] [CrossRef]
- Lah, L.; Podobnik, B.; Novak, M.; Korošec, B.; Berne, S.; Vogelsang, M.; Kraševec, N.; Zupanec, N.; Stojan, J.; Bohlmann, J.; et al. The versatility of the fungal cytochrome P450 monooxygenase system is instrumental in xenobiotic detoxification. Mol. Microbiol. 2011, 81, 1374–1389. [Google Scholar] [CrossRef]
- Lester, G. Inhibition of growth, synthesis, and permeability in Neurospora crassa by Phenethyl Alcohol. J. Bacteriol. 1965, 90, 29–37. [Google Scholar]
- Lee, Y.S.; Kim, J.; Lee, S.G.; Oh, E.; Shin, S.C.; Park, I.K. Effects of plant essential oils and components from oriental sweetgum (Liquidambar orientalis) on growth and morphogenesis of three phytopathogenic fungi. Pestic. Biochem. Physiol. 2009, 93, 138–143. [Google Scholar] [CrossRef]
- Kfoury, M.; Lounès-Hadj Sahraoui, A.; Bourdon, N.; Laruelle, F.; Fontaine, J.; Auezova, L.; Greige-Gerges, H.; Fourmentin, S. Solubility, photostability and antifungal activity of phenylpropanoids encapsulated in cyclodextrins. Food Chem. 2016, 196, 518–525. [Google Scholar] [CrossRef]
- Johansen, P.; Jespersen, L. Impact of quorum sensing on the quality of fermented foods. Curr. Opin. Food Sci. 2017, 13, 16–25. [Google Scholar] [CrossRef]
- Krom, B.P.; Levy, N.; Meijler, M.M.; Jabra-Rizk, M.A. Farnesol and Candida albicans: Quorum Sensing or Not Quorum Sensing? Isr. J. Chem. 2016, 56, 295–301. [Google Scholar] [CrossRef]
- Verheecke, C.; Choque, E.; Mathieu, F. Application of fungal metabolites against mycotoxins production. In Fungal Metabolites; Springer International Publishing: Cham, Switzerland, 2016; pp. 1–37. ISBN 978-3-319-19456-1. [Google Scholar]
- Mirata, M.A.; Wüst, M.; Mosandl, A.; Schrader, J. Fungal biotransformation of (±)-linalool. J. Agric. Food Chem. 2008, 56, 3287–3296. [Google Scholar] [CrossRef]
- Kanchiswamy, C.N.; Malnoy, M.; Maffei, M.E. Chemical diversity of microbial volatiles and their potential for plant growth and productivity. Front. Plant Sci. 2015, 6, 151. [Google Scholar] [CrossRef]
- Padder, S.A.; Prasad, R.; Shah, A.H. Quorum sensing: A less known mode of communication among fungi. Microbiol. Res. 2018, 210, 51–58. [Google Scholar] [CrossRef]
- Macko, V.; Staples, R.C.; Allen, P.J.; Renwick, J.A.A. Identification of the germination self-inhibitor from wheat stem rust uredospores. Science 1971, 173, 835–836. [Google Scholar] [CrossRef]
- Pradhan, S.; Flores, F.J.; Melouk, H.; Walker, N.R.; Molineros, J.E.; Garzon, C.D. Chemical hormesis on plant pathogenic fungi and Oomycetes. In Pesticide Dose: Effects on the Environment and Target and Non-Target Organisms; Duke, S.O., Kudsk, P., Solomon, K., Eds.; ACS Publications: Washington, DC, USA, 2017; pp. 121–133. [Google Scholar]
- Deryabin, D.G.; Inchagova, K.S. Subinhibitory concentrations of the penicillin antibiotics induce quorum-dependent violacein synthesis in Chromobacterium violaceum. Microbiology 2017, 86, 463–468. [Google Scholar] [CrossRef]
- Bernier, S.P.; Surette, M.G. Concentration-dependent activity of antibiotics in natural environments. Front. Microbiol. 2013, 4, 20. [Google Scholar] [CrossRef]
- Barry, D. Development of Novel Image Analysis Methods for the Morphological Quantification of Filamentous Fungi. Ph.D. Thesis, Dublin Institute of Technology, Dublin, Ireland, 2010. [Google Scholar]
- Villasanti, C. El Cultivo de Tomate con Buenas Prácticas Agrícolas en la Agricultura Urbana y Periurbana; Organización de las Naciones Unidas para la Alimentación y la Agricultura: Asunción, Paraguay, 2013; ISBN 9789253077793. [Google Scholar]
- Slawecki, R.A.; Ryan, E.P.; Young, D.H. Novel fungitoxicity assays for inhibition of germination-associated adhesion of Botrytis cinerea and Puccinia recondita spores. Appl. Environ. Microbiol. 2002, 68, 597–601. [Google Scholar] [CrossRef]
- Pinedo, C.; Moraga, J.; Barua, J.; Gonzalez-Rodriguez, V.E.; Aleu, J.; Duran-Patron, R.; Macias-Sanchez, A.J.; Hanson, J.R.; Viaud, M.; Hernandez-Galan, R.; et al. Chemically induced cryptic sesquiterpenoids and expression of sesquiterpene cyclases in Botrytis cinerea revealed new sporogenic (+)-4-epieremophil-9-en-11-ols. ACS Chem. Biol. 2016, 11, 1391–1400. [Google Scholar] [CrossRef]
- Mouekouba, L.D.O.; Kehinde Olajide, E.; Zhang, Z.-Z.; Wang, A.-J.; Wang, A.-X. Biological control of botrytis cinerea in tomato leaves. Int. Proc. Chem. Biol. Environ. Eng. 2013, 60, 64. [Google Scholar]


| Compound | Name * | Micromolar Equivalents | Molecular Structure | |
|---|---|---|---|---|
| [ppm] | [µM] | |||
| Isoeugenol | I | A (1000) | 121.8 | 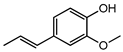 |
| B (500) | 60.9 | |||
| C (100) | 12.9 | |||
| D (50) | 6.09 | |||
| 2-(3-hydroxyphenyl)-ethanol | II | A (1000) | 144.8 | 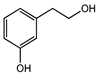 |
| B (500) | 72.4 | |||
| C (100) | 14.5 | |||
| D (50) | 7.24 | |||
| 1-Phenylethanol | III | A (1000) | 163.7 | 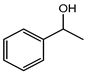 |
| B (500) | 81.8 | |||
| C (100) | 16.4 | |||
| D (50) | 8.2 | |||
| 3-Phenyl-1-propanol | IV | A (1000) | 146.8 | 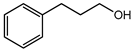 |
| B (500) | 73.4 | |||
| C (100) | 14.7 | |||
| D (50) | 7.34 | |||
| 2´-Hydroxyphenylacetic acid | V | A (1000) | 131.4 | 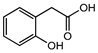 |
| B (500) | 65.7 | |||
| C (100) | 13.14 | |||
| D (50) | 6.6 | |||
| Mancozeb (chemical control) | Mzb | 2957.4 † | 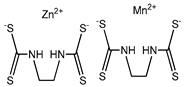 | |
| Treatments and Nomenclature | Parameters Evaluated; Percentage of Inhibition (%) | Tomato Assays | |||||
|---|---|---|---|---|---|---|---|
| Germination | Germ Tubes | Sporulation | % Incidence; Leaves | % Severity; Leaves | % Incidence; Fruits | % Severity; Fruits | |
| Isoeugenol 1000 ppm (IA) | 19.5 | 76.7 | 14.5 | 3.1 | 2.3 | 75 | 37.5 |
| Isoeugenol 500 ppm (IB) | 14.6 | 74.3 | −15.4 | 18.8 | 10.9 | 75 | 50 |
| Isoeugenol 100 ppm (IC) | 4.9 | 31.5 | −130.4 | 34.4 | 16.4 | 100 | 55 |
| Isoeugenol 50 ppm (ID) | 24.4 | −50.7 | −85 | 43.8 | 23.4 | 100 | 54.8 |
| 2-(3-Hydroxyphenyl) ethanol) 1000 ppm (IIA) | 43.9 | −72.5 | −21.4 | 56.3 | 25.5 | 66.7 | 50 |
| 2-(3-Hydroxyphenyl) ethanol) 500 ppm (IIB) | 7.3 | −82.7 | 53 | 96.9 | 45.3 | 87.5 | 52.5 |
| 2-(3-HYdroxyphenyl) ethanol) 100 ppm (IIC) | 29.3 | −96.3 | 76 | 78.1 | 39.1 | 100 | 56 |
| 2-(3-Hydroxyphenyl) ethanol) 50 ppm (IID) | 26.8 | −60.9 | 30.6 | 87.5 | 35.2 | 100 | 55.3 |
| 1-Phenylethanol 1000 ppm (IIIA) | 31.7 | 67.4 | 15.7 | 59.4 | 27.3 | 25 | 7.5 |
| 1-Phenylethanol 500 ppm (IIIB) | 19.5 | 9 | 30.7 | 50 | 26.6 | 62.5 | 42.5 |
| 1-Phenylethanol 100 ppm (IIIC) | 12.2 | −28.5 | 48.4 | 56.3 | 32.8 | 100 | 55.6 |
| 1-Phenylethanol 50 ppm (IIID) | −4.9 | −15.4 | 48 | 21.9 | 8.6 | 100 | 54.3 |
| 3-Phenyl-1-propanol 1000 ppm (IVA) | 87.8 | 95.7 | 88.9 | 53.1 | 25.6 | 100 | 30 |
| 3-Phenyl-1-propanol 500 ppm (IVB) | 80.5 | 96.5 | 27.8 | 37.5 | 18 | 62.5 | 37.5 |
| 3-Phenyl-1-propanol 100 ppm (IVC) | 17.1 | −32.2 | 46.1 | 25 | 13.3 | 100 | 52.5 |
| 3-Phenyl-1-propanol 50 ppm (IVD) | 14.6 | −27.9 | 68.3 | 84.4 | 46.1 | 100 | 55.3 |
| 2-Hydroxy phenyl acetic acid 1000 ppm (VA) | 56.1 | −16.9 | 5.6 | 96.9 | 53.6 | 62.5 | 30 |
| 2-Hydroxy phenyl acetic acid 500 ppm (VB) | 41.5 | −35.6 | 42.5 | 87.5 | 36.7 | 75 | 35 |
| 2-Hydroxy phenyl acetic acid 100 ppm (VC) | 22 | −12.6 | 69 | 93.8 | 39.1 | 100 | 54.5 |
| 2-Hydroxy phenyl acetic acid 50 ppm (VD) | 19.5 | −12.4 | 54.6 | 90.6 | 32 | 100 | 52.5 |
| Control | - | - | - | 87.5 | 55.5 | 100 | 55 |
| Mancozeb | 100 | - | - | 6.3 | 3.1 | 25 | 5 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rosero-Hernández, E.D.; Moraga, J.; Collado, I.G.; Echeverri, F. Natural Compounds That Modulate the Development of the Fungus Botrytis cinerea and Protect Solanum lycopersicum. Plants 2019, 8, 111. https://doi.org/10.3390/plants8050111
Rosero-Hernández ED, Moraga J, Collado IG, Echeverri F. Natural Compounds That Modulate the Development of the Fungus Botrytis cinerea and Protect Solanum lycopersicum. Plants. 2019; 8(5):111. https://doi.org/10.3390/plants8050111
Chicago/Turabian StyleRosero-Hernández, Esteban D., Javier Moraga, Isidro G. Collado, and Fernando Echeverri. 2019. "Natural Compounds That Modulate the Development of the Fungus Botrytis cinerea and Protect Solanum lycopersicum" Plants 8, no. 5: 111. https://doi.org/10.3390/plants8050111
APA StyleRosero-Hernández, E. D., Moraga, J., Collado, I. G., & Echeverri, F. (2019). Natural Compounds That Modulate the Development of the Fungus Botrytis cinerea and Protect Solanum lycopersicum. Plants, 8(5), 111. https://doi.org/10.3390/plants8050111






