A Polyamine Oxidase from Selaginella lepidophylla (SelPAO5) can Replace AtPAO5 in Arabidopsis through Converting Thermospermine to Norspermidine instead to Spermidine
Abstract
1. Introduction
2. Results
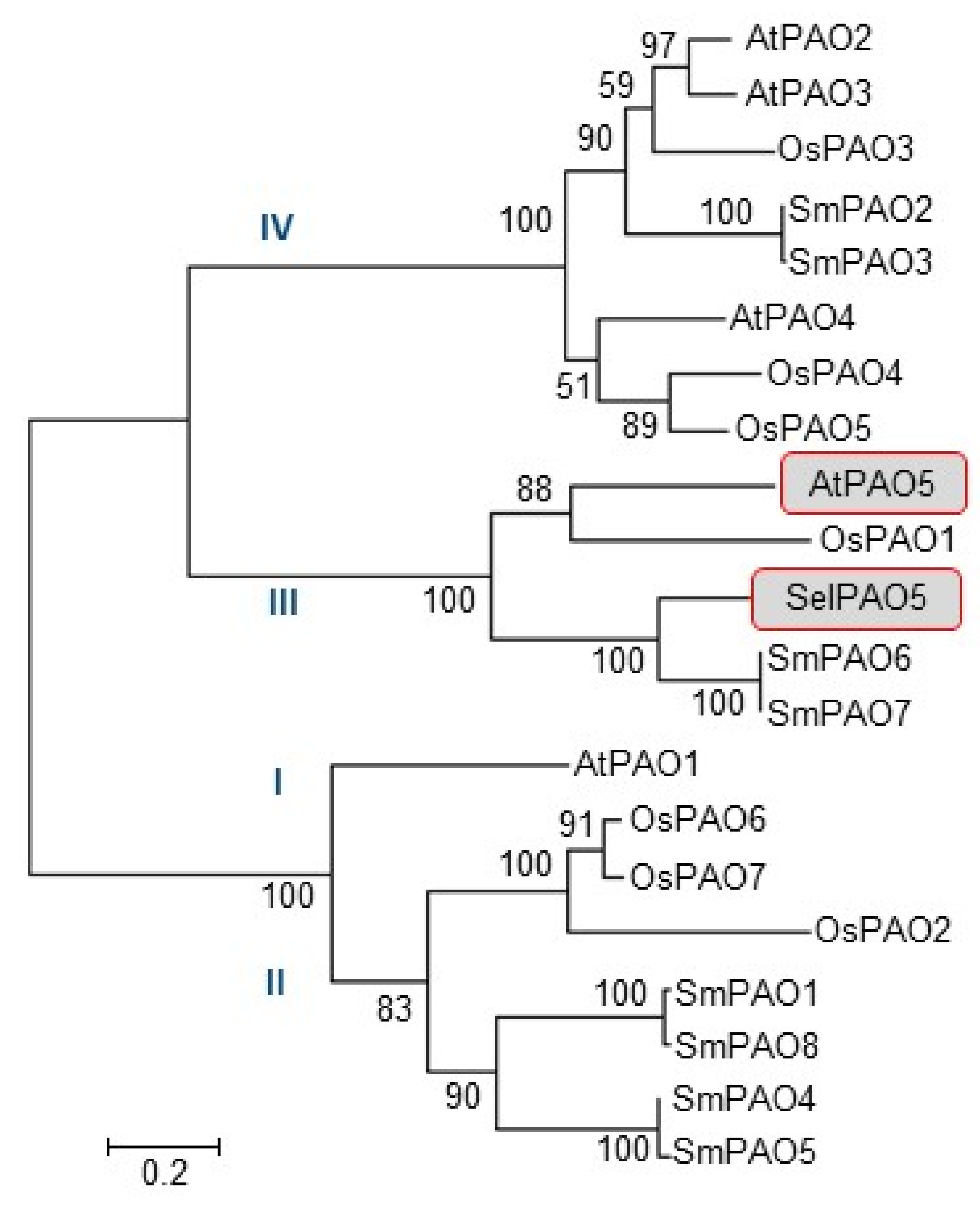
2.1. Phylogenetic Classification of SelPAO5 and Cellular Localization
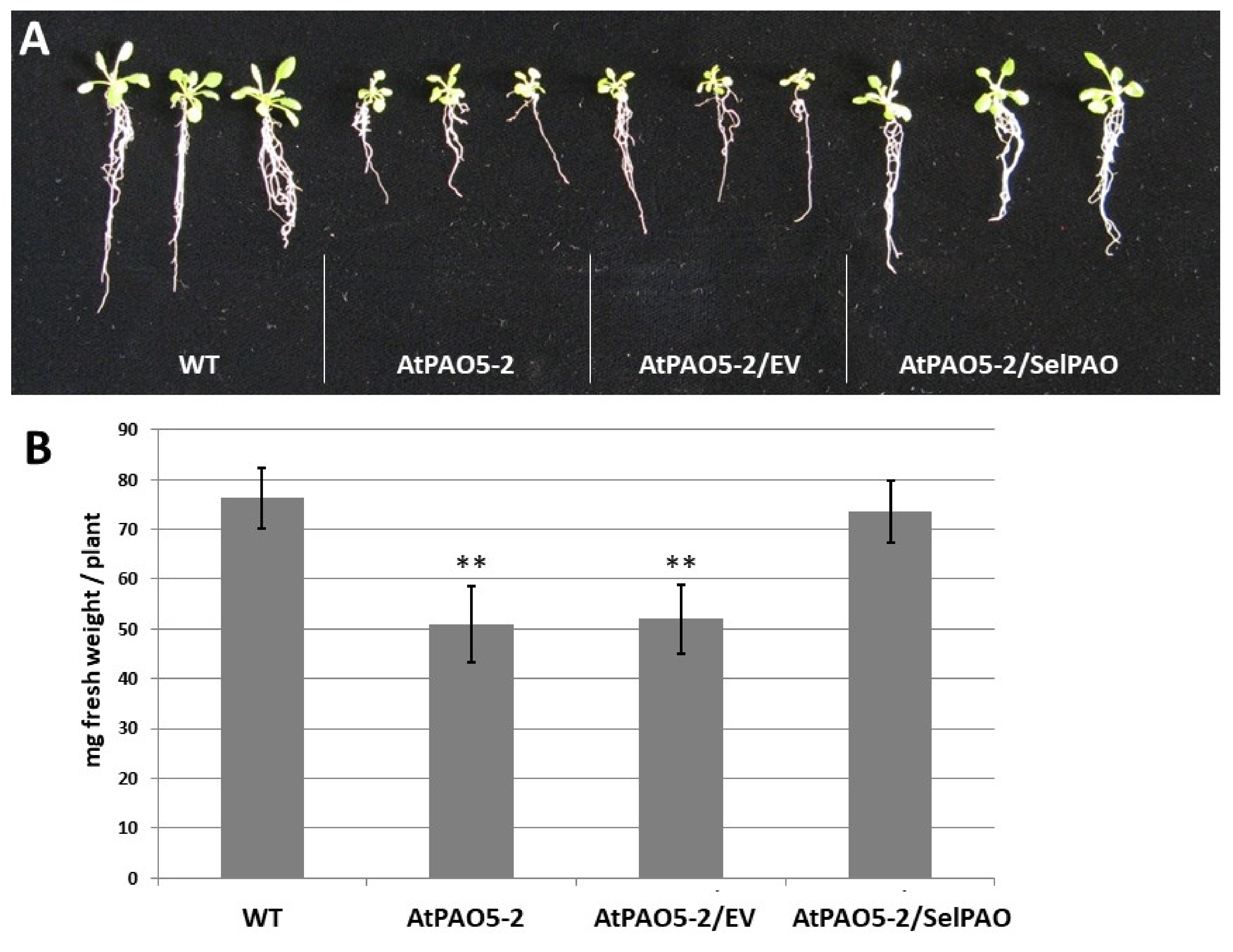
2.2. SelPAO5 Complementation of Atpao5-2 Mutant Rescues T-Spm-Induced Growth Inhibition
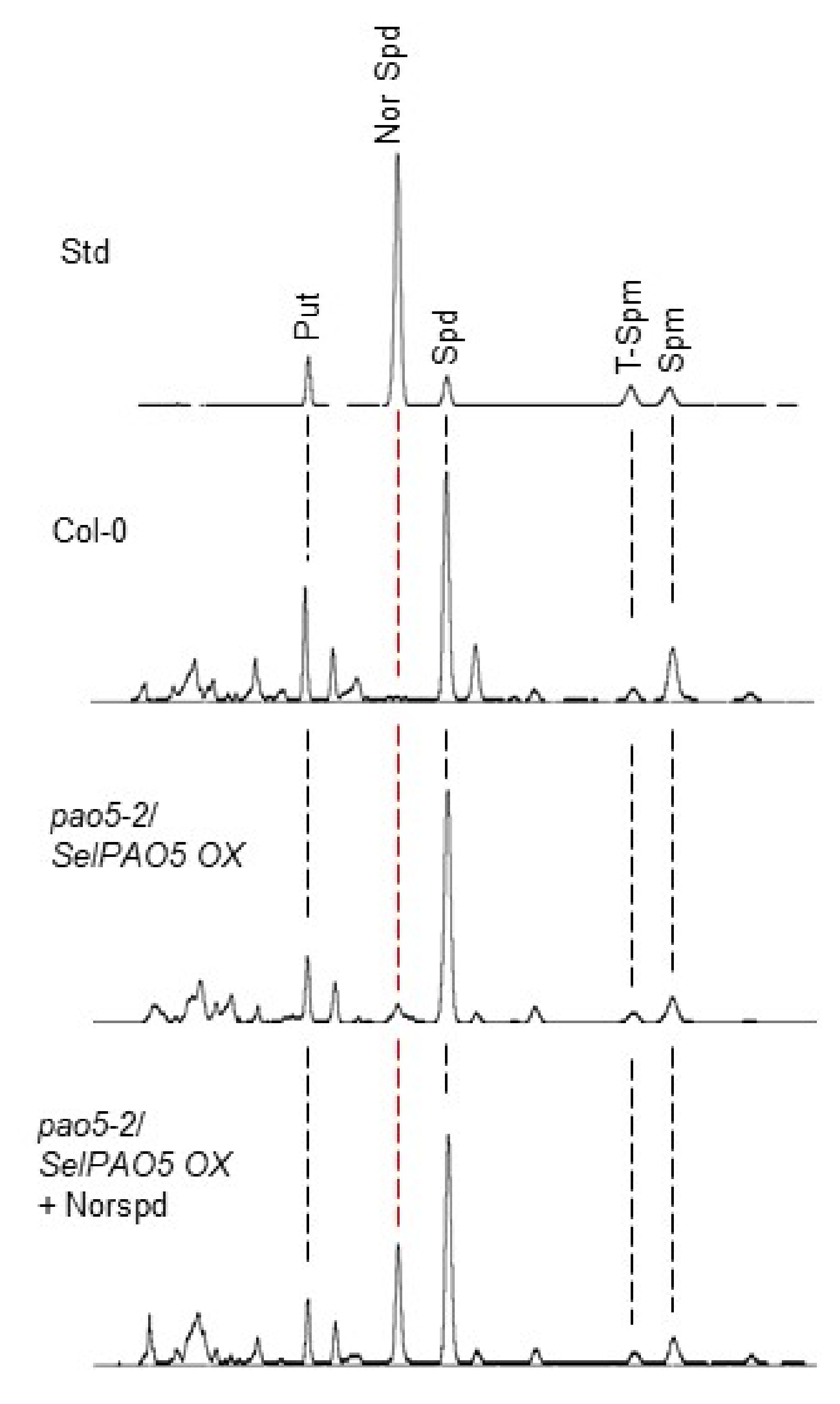
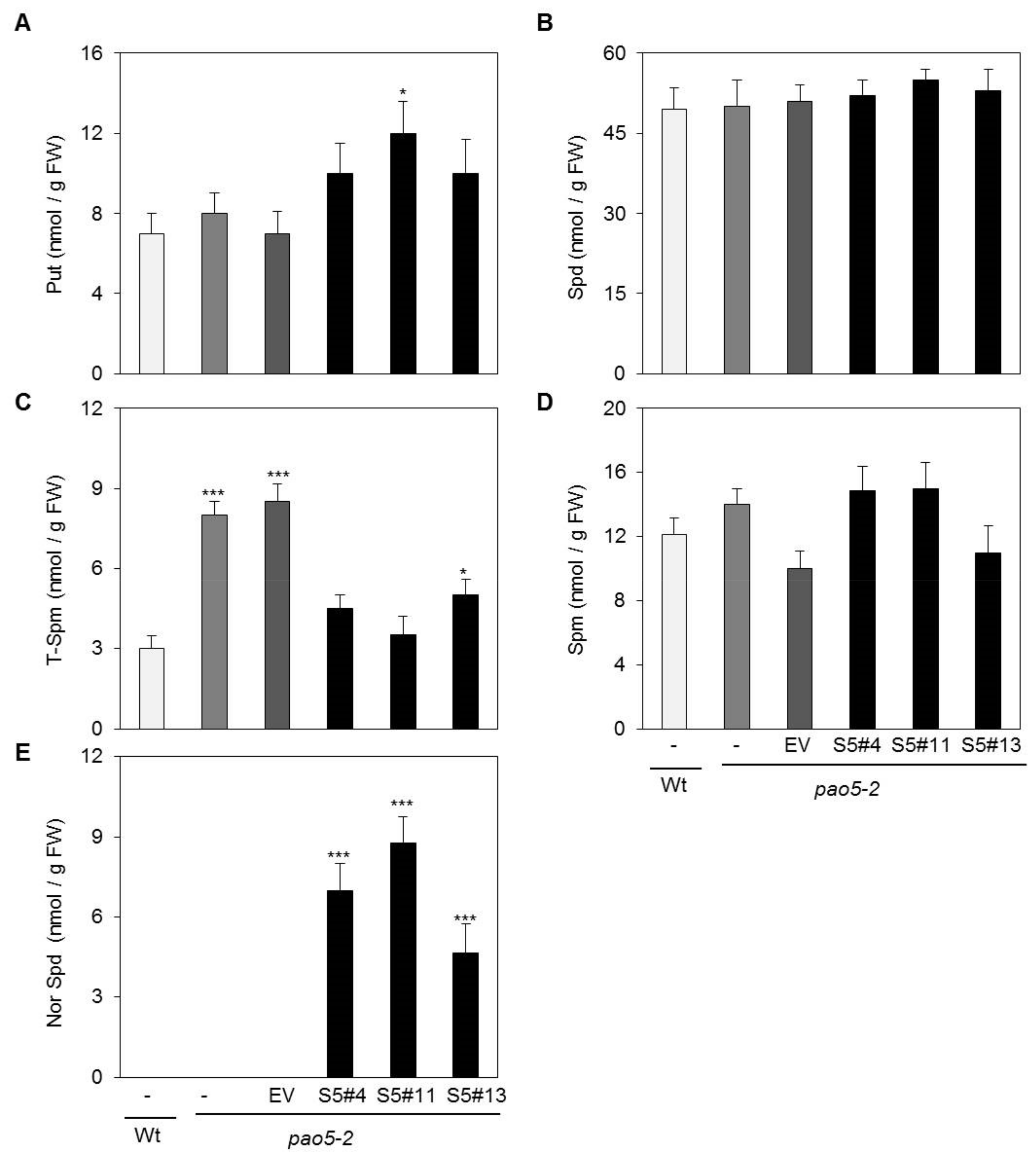
2.3. SelPAO Produces NorSpd in Arabidopsis Plants
3. Discussion
4. Materials and Methods
4.1. Plant Materials and Growth Conditions
4.2. Determination of Plant Fresh Weight
4.3. Chemicals
4.4. Generation of Arabidopsis pao5 T-DNA Insertion Mutant Transgenic Lines Expressing SelPAO5 ORF
4.5. RT-PCR Analysis
4.6. PA Analysis by High-Performance Liquid Chromatography (HPLC)
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Tabor, C.W.; Tabor, H. Polyamines. Annu. Rev. Biochem. 1984, 53, 749–790. [Google Scholar] [CrossRef] [PubMed]
- Cohen, S.S. A Guide to the Polyamines; Oxford University Press: Oxford, UK, 1998. [Google Scholar]
- Knott, J.M.; Römer, P.; Sumper, M. Putative spermine synthases from Thalassiosira pseudonana and Arabidopsis thaliana synthesize thermospermine rather than spermine. FEBS Lett. 2007, 581, 3081–3086. [Google Scholar] [CrossRef] [PubMed]
- Kakehi, J.; Kuwashiro, Y.; Niitsu, M.; Takahashi, T. Thermospermine is required for stem elongation in Arabidopsis thaliana. Plant Cell Physiol. 2008, 49, 1342–1349. [Google Scholar] [CrossRef] [PubMed]
- Naka, Y.; Watanabe, K.; Sagor, G.H.M.; Niitsu, M.; Pillai, M.A.; Kusano, T.; Takahashi, Y. Quantitative analysis of plant polyamines including thermospermine during growth and salinity stress. Plant Physiol. Biochem. 2010, 48, 527–533. [Google Scholar] [CrossRef]
- Takano, A.; Kakehi, J.I.; Takahashi, T. Thermospermine is not a minor polyamine in the plant kingdom. Plant Cell Physiol. 2012, 53, 606–616. [Google Scholar] [CrossRef]
- Michael, A.J. Polyamines in eukaryotes, bacteria, and archea. J. Biol. Chem. 2016, 291, 14896–14903. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, T. Unique polyamines produced by an extreme thermophile, Thermus thermophilus. Amino Acids 2007, 33, 367–372. [Google Scholar] [CrossRef]
- Groppa, M.D.; Benavides, M.P. Polyamines and abiotic stress: Recent advances. Amino Acids 2008, 34, 35–45. [Google Scholar] [CrossRef]
- Kusano, T.; Berberich, T.; Tateda, C.; Takahashi, Y. Polyamines: Essential factors for growth and survival. Planta 2008, 228, 367–381. [Google Scholar] [CrossRef]
- Alcázar, R.; Altabella, T.; Marco, F.; Bortolotti, C.; Reymond, M.; Koncz, C.; Carrasco, P.; Tiburcio, A.F. Polyamines: Molecules with regulatory functions in plant abiotic stress tolerance. Planta 2010, 231, 1237–1249. [Google Scholar] [CrossRef]
- Mattoo, A.K.; Minocha, S.C.; Minocha, R.; Handa, A.K. Polyamines and cellular metabolism in plants: Transgenic approaches reveal different responses to diamine putrescine versus higher polyamines spermidine and spermine. Amino Acids 2010, 38, 405–413. [Google Scholar] [CrossRef]
- Minocha, R.; Majumdar, R.; Minocha, S.C. Polyamines and abiotic stress in plants: A complex relationship. Front. Plant Sci. 2014, 5, 175. [Google Scholar] [CrossRef]
- Berberich, T.; Sagor, G.H.M.; Kusano, T. Polyamines in Plant Stress Response. In Polyamines, A Universal Molecular Nexus for Growth, Survival, and Specialized Metabolism; Kusano, T., Suzuki, H., Eds.; Springer: Tokyo, Japan, 2015; ISBN 978-4-431-55211-6. [Google Scholar]
- Takahashi, T.; Tong, W. Regulation and diversity of polyamine biosynthesis in plants. In Polyamines, A Universal Molecular Nexus for Growth, Survival, and Specialized Metabolism; Kusano, T., Suzuki, H., Eds.; Springer: Tokyo, Japan, 2015; ISBN 978-4-431-55211-6. [Google Scholar]
- Hamana, K.; Matsuzaki, S. Distinct difference in the polyamine compositions of Bryophyta and Pteridophyta. J. Biochem. 1985, 97, 1595–1601. [Google Scholar] [CrossRef]
- Kuehn, G.D.; Rodriguez-Garay, B.; Bagga, S.; Phillips, G.C. Novel occurrence of uncommon polyamines in higher plants. Plant Physiol. 1990, 94, 855–857. [Google Scholar] [CrossRef]
- Hamana, K.; Aizaki, T.; Arai, E.; Uchikata, K.; Ohnishi, H. Distribution of norspermidine as a cellular polyamine within micro green algae including non-photosynthetic achlorophyllous Polytoma, Polytomella, Prototheca and Helicosporidium. J. Gen. Appl. Microbiol. 2004, 50, 289–295. [Google Scholar] [CrossRef]
- Fuell, C.; Elliot, K.A.; Hanfrey, C.C.; Franceschetti, M.; Michael, A.J. Polyamine biosynthetic diversity in plants and algae. Plant Physiol. Biochem. 2010, 48, 513–520. [Google Scholar] [CrossRef]
- Rodriguez-Garay, B.; Phillips, G.C.; Kuehn, G.D. Detection of norspermidine and norspermine in Medicago sativa, L. (alfalfa). Plant Physiol. 1989, 89, 525–529. [Google Scholar] [CrossRef]
- Koc, E.C.; Bagga, S.; Songstad, D.D.; Betz, S.R.; Kuehn, G.D.; Phillips, G.C. Occurrence of uncommon polyamines in cultured tissues of maize. In Vitro Cell Dev. Biol. Plant 1998, 34, 623–631. [Google Scholar] [CrossRef]
- Kusano, T.; Kim, D.W.; Liu, T.; Berberich, T. Polyamine catabolism in plants. In Polyamines, A Universal Molecular Nexus for Growth, Survival, and Specialized Metabolism; Kusano, T., Suzuki, H., Eds.; Springer: Tokyo, Japan, 2015; ISBN 978-4-431-55211-6. [Google Scholar]
- Federico, R.; Angelini, R.; Cona, A.; Niglio, A. Polyamine oxidase bound to cell walls from Zea mays seedlings. Phytochemistry 1992, 31, 2955–2957. [Google Scholar] [CrossRef]
- Tavladoraki, P.; Schininà, M.E.; Cecconi, F.; Di Agostino, S.; Manera, F.; Rea, G.; Mariottini, P.; Federico, R.; Angelini, R. Maize polyamine oxidase: Primary structure from protein and cDNA sequencing. FEBS Lett. 1998, 426, 62–66. [Google Scholar] [CrossRef]
- Radová, A.; Sebela, M.; Galuszka, P.; Frébort, I.; Jacobsen, S.; Faulhammer, H.G.; Pec, P. Barley polyamine oxidase: Characterisation and analysis of the cofactor and the N-terminal amino acid sequence. Phytochem. Anal. 2001, 12, 166–173. [Google Scholar] [CrossRef]
- Cervelli, M.; Cona, A.; Angelini, R.; Polticelli, F.; Federico, R.; Mariottini, P. A barley polyamine oxidase isoform with distinct structural features and subcellular localization. Eur. J. Biochem. 2001, 268, 3816–3830. [Google Scholar] [CrossRef]
- Cervelli, M.; Di Caro, O.; Di Penta, A.; Angelini, R.; Federico, R.; Vitale, A.; Mariottini, P. A novel C-terminal sequence from barley polyamine oxidase is a vacuolar sorting signal. Plant J. 2004, 40, 410–418. [Google Scholar] [CrossRef]
- Cona, A.; Rea, G.; Angelini, R.; Federico, R.; Tavladoraki, P. Functions of amine oxidases in plant development and defence. Trends Plant Sci. 2006, 11, 80–88. [Google Scholar] [CrossRef]
- Angelini, R.; Cona, A.; Federico, R.; Fincato, P.; Tavladoraki, P.; Tisi, A. Plant amine oxidases “on the move”: An update. Plant Physiol. Biochem. 2010, 48, 560–564. [Google Scholar] [CrossRef]
- Tavladoraki, P.; Rossi, M.N.; Saccuti, G.; Perez-Amador, M.A.; Polticelli, F.; Angelini, R.; Federico, R. Heterologous expression and biochemical characterization of a polyamine oxidase from Arabidopsis involved in polyamine back conversion. Plant Physiol. 2006, 141, 1519–1532. [Google Scholar] [CrossRef]
- Moschou, P.N.; Sanmartin, M.; Andriopoulou, A.H.; Rojo, E.; Sanchez-Serrano, J.J.; Roubelakis-Angelakis, K.A. Bridging the gap between plant and mammalian polyamine catabolism: A novel peroxisomal polyamine oxidase responsible for a full back-conversion pathway in Arabidopsis. Plant Physiol. 2008, 147, 1845–1857. [Google Scholar] [CrossRef]
- Kamada-Nobusada, T.; Hayashi, M.; Fukazawa, M.; Sakakibara, H.; Nishimura, M. A putative peroxisomal polyamine oxidase AtPAO4 is involved in polyamine catabolism in Arabidopsis thaliana. Plant Cell Physiol. 2008, 49, 1272–1282. [Google Scholar] [CrossRef] [PubMed]
- Fincato, P.; Moschou, P.N.; Spedaletti, V.; Tavazza, R.; Angelini, R.; Federico, R.; Roubelakis-Angelakis, K.A.; Tavladoraki, P. Functional diversity inside the Arabidopsis polyamine oxidase gene family. J. Exp. Bot. 2011, 62, 1155–1168. [Google Scholar] [CrossRef]
- Kim, D.W.; Watanabe, K.; Murayama, C.; Izawa, S.; Niitsu, M.; Michael, A.J.; Berberich, T.; Kusano, T. Polyamine oxidase 5 regulates Arabidopsis thaliana growth through a thermospermine oxidase activity. Plant Physiol. 2014, 165, 1575–1590. [Google Scholar] [CrossRef] [PubMed]
- Sagor, G.H.M.; Inoue, M.; Kim, D.W.; Kojima, S.; Niitsu, M.; Berberich, T.; Kusano, T. The polyamine oxidase from lycophyte Selaginella lepidophylla (SelPAO5), unlike that of angiosperms, back-converts thermospermine to norspermidine. FEBS Lett. 2015, 589, 3071–3078. [Google Scholar] [CrossRef]
- Liu, T.; Kim, D.W.; Niitsu, M.; Berberich, T.; Kusano, T. Oryza sativa polyamine oxidase 1 back-converts tetraamines, spermine and thermospermine, to spermidine. Plant Cell Rep. 2014, 33, 143–151. [Google Scholar] [CrossRef]
- Banks, J.A.; Nishiyama, T.; Hasebe, M.; Bowman, J.L.; Gribskov, M.; dePamphilis, C.; Albert, V.A.; Aono, N.; Aoyama, T.; Ambrose, B.A.; et al. The Selaginella genome identifies genetic changes associated with the evolution of vascular plants. Science 2012, 332, 960–963. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef]
- Fuse, T.; Sasaki, T.; Yano, M. Ti-plasmid vectors useful for functional analysis of rice genes. Plant Biotech. 2001, 18, 219–222. [Google Scholar] [CrossRef]
- Bordenave, C.D.; Granados Mendoza, C.; Jiménez Bremont, J.F.; Gárriz, A.; Rodríguez, A.A. Defining novel plant polyamine oxidase subfamilies through molecular modeling and sequence analysis. BMC Evol. Biol. 2019, 19, 28. [Google Scholar] [CrossRef]
- Hanzawa, Y.; Takahashi, T.; Komeda, Y. ACL5: An Arabidopsis gene required for internodal elongation after flowering. Plant J. 1997, 12, 863–874. [Google Scholar] [CrossRef]
- Hamana, K.; Niitsu, M.; Samejima, K. Unusual polyamines in aquatic plants: The occurrence of homospermidine, norspermidine, thermospermine, norspermine, aminopropylhomospermidine, bis(aminopropyl)ethanediamine, and methylspermidine. Can. J. Bot. 1998, 76, 130–133. [Google Scholar]
- Hamana, K.; Matsuzaki, S. Widespread occurrence of norspermidine and norspermine in eukaryotic algae. J. Biochem. 1982, 91, 1321–1328. [Google Scholar] [CrossRef]
- Bagga, S.; Rochford, J.; Klaene, Z.; Kuehn, G.D.; Phillips, G.C. Putrescine aminopropyltransferase is responsible for biosynthesis of spermidine, spermine, and multiple uncommon polyamines in osmotic stress-tolerant Alfalfa. Plant Physiol. 1997, 114, 445–454. [Google Scholar] [CrossRef][Green Version]
- Tassoni, A.; Awad, N.; Griffiths, G. Effect of ornithine decarboxylase and norspermidine in modulating cell division in the green alga Chlamydomonas reinhardtii. Plant Physiol. Biochem. 2018, 123, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Niitsu, M.; Samejima, K. Synthesis of a series of linear pentaamines with three and four methylene chain intervals. Chem. Pharm. Bull. 1986, 34, 1032–1038. [Google Scholar] [CrossRef][Green Version]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 1998, 16, 735–743. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sagor, G.H.M.; Kusano, T.; Berberich, T. A Polyamine Oxidase from Selaginella lepidophylla (SelPAO5) can Replace AtPAO5 in Arabidopsis through Converting Thermospermine to Norspermidine instead to Spermidine. Plants 2019, 8, 99. https://doi.org/10.3390/plants8040099
Sagor GHM, Kusano T, Berberich T. A Polyamine Oxidase from Selaginella lepidophylla (SelPAO5) can Replace AtPAO5 in Arabidopsis through Converting Thermospermine to Norspermidine instead to Spermidine. Plants. 2019; 8(4):99. https://doi.org/10.3390/plants8040099
Chicago/Turabian StyleSagor, G. H. M., Tomonobu Kusano, and Thomas Berberich. 2019. "A Polyamine Oxidase from Selaginella lepidophylla (SelPAO5) can Replace AtPAO5 in Arabidopsis through Converting Thermospermine to Norspermidine instead to Spermidine" Plants 8, no. 4: 99. https://doi.org/10.3390/plants8040099
APA StyleSagor, G. H. M., Kusano, T., & Berberich, T. (2019). A Polyamine Oxidase from Selaginella lepidophylla (SelPAO5) can Replace AtPAO5 in Arabidopsis through Converting Thermospermine to Norspermidine instead to Spermidine. Plants, 8(4), 99. https://doi.org/10.3390/plants8040099




