Elevated Ozone Concentration Reduces Photosynthetic Carbon Gain but Does Not Alter Leaf Structural Traits, Nutrient Composition or Biomass in Switchgrass
Abstract
:1. Introduction
2. Results
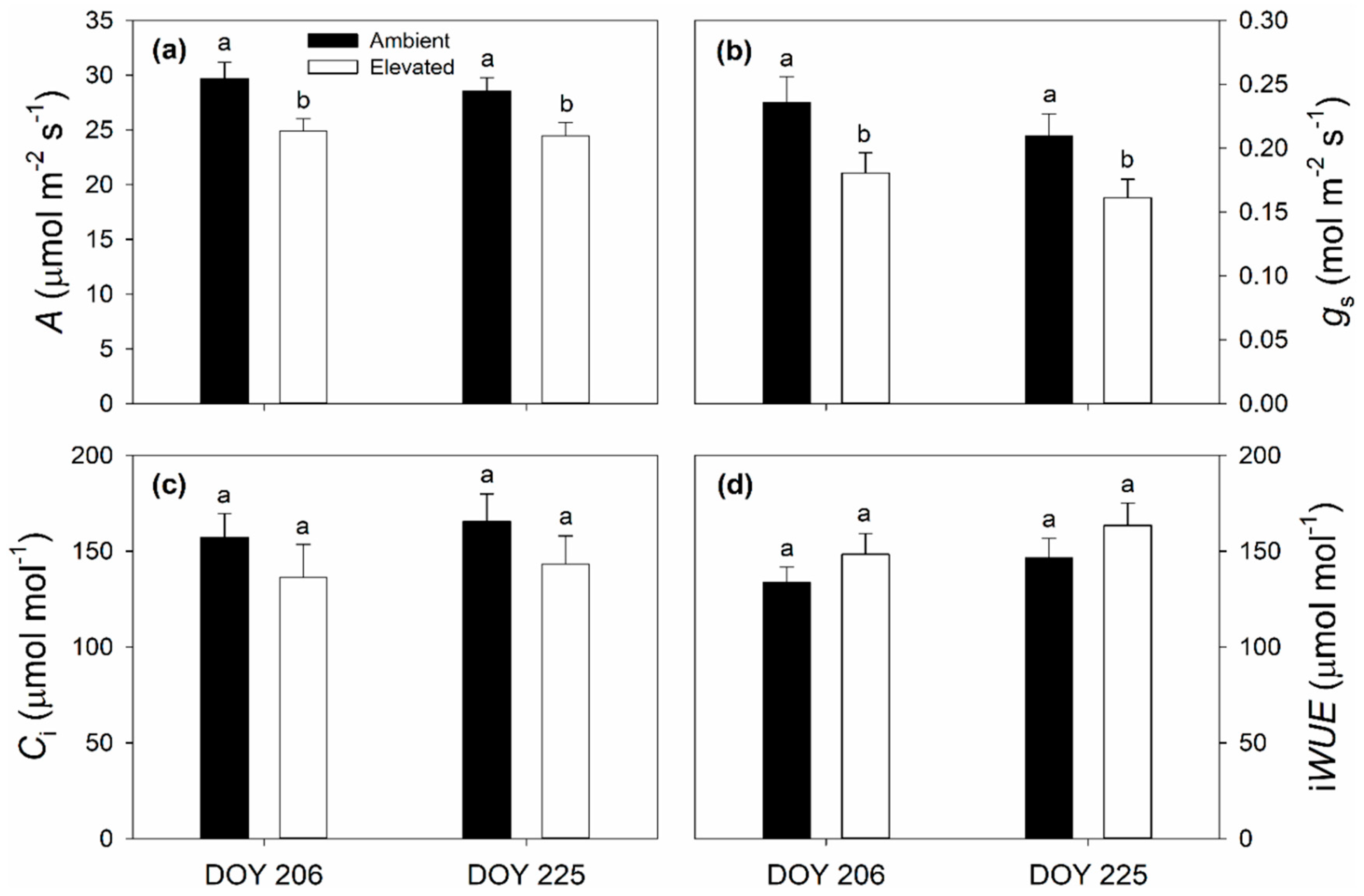
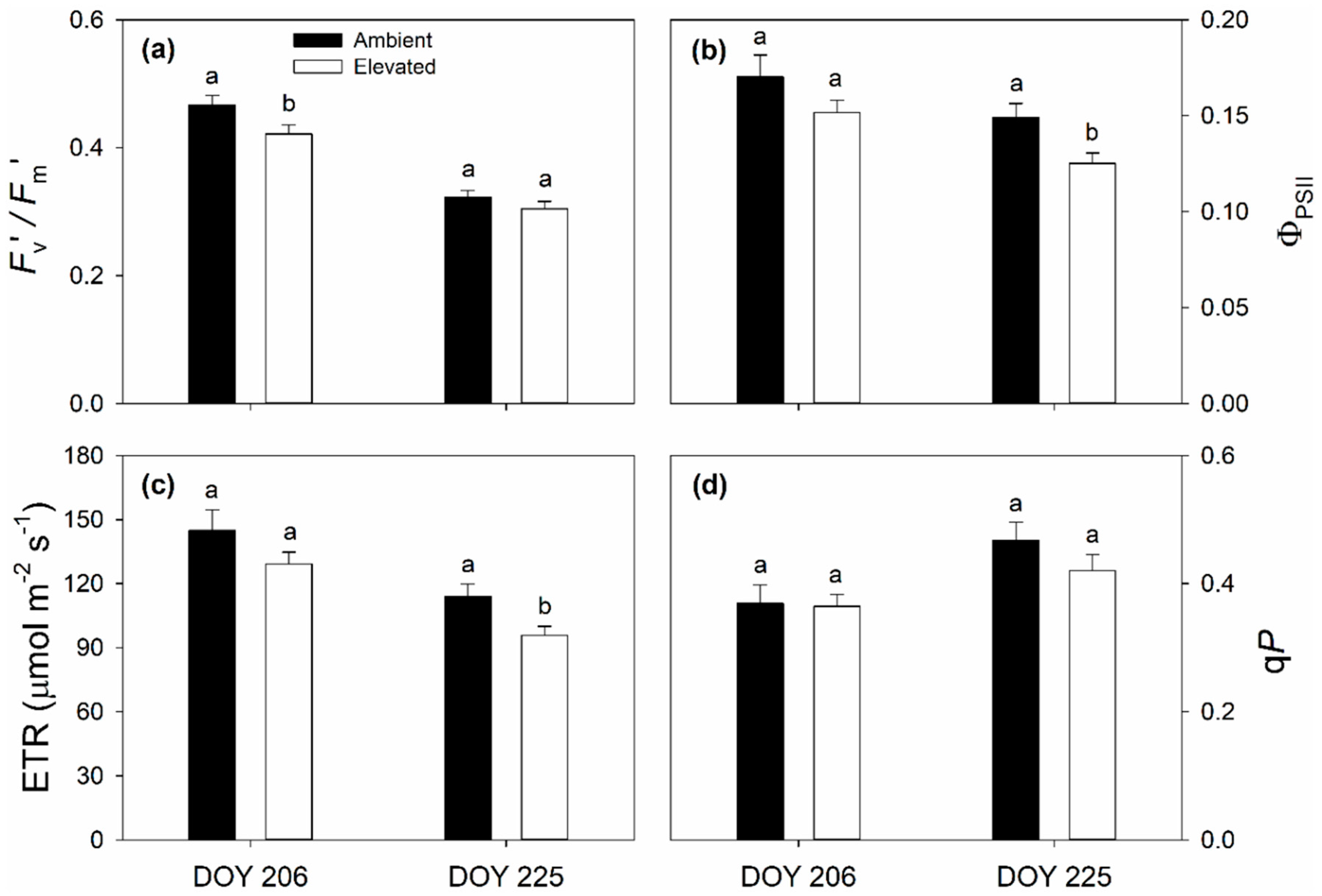
2.1. Leaf Photosynthetic and Chlorophyll Fluorescence Responses to Elevated O3
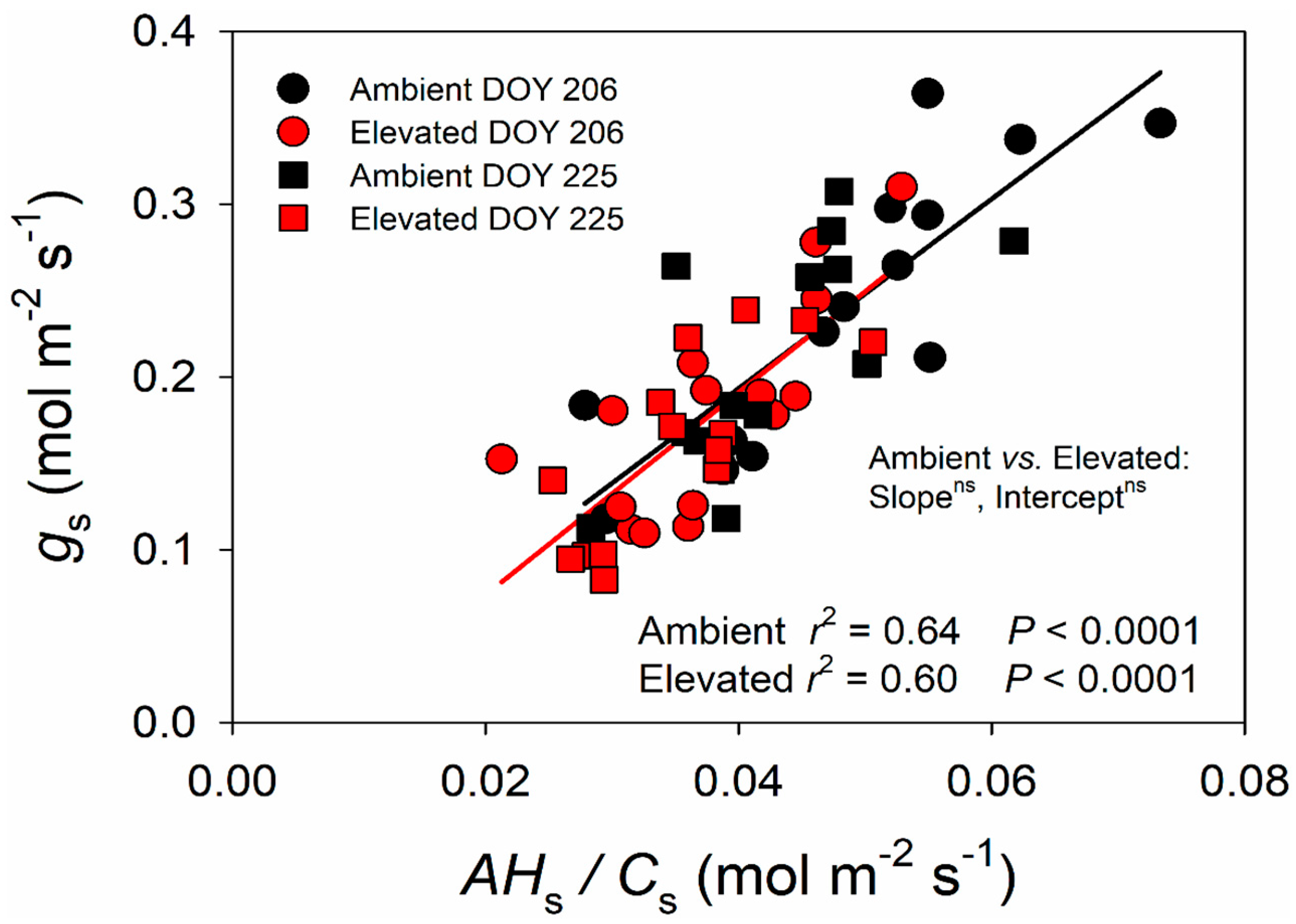
2.2. Changes in the BWB Relationship due to Elevated O3
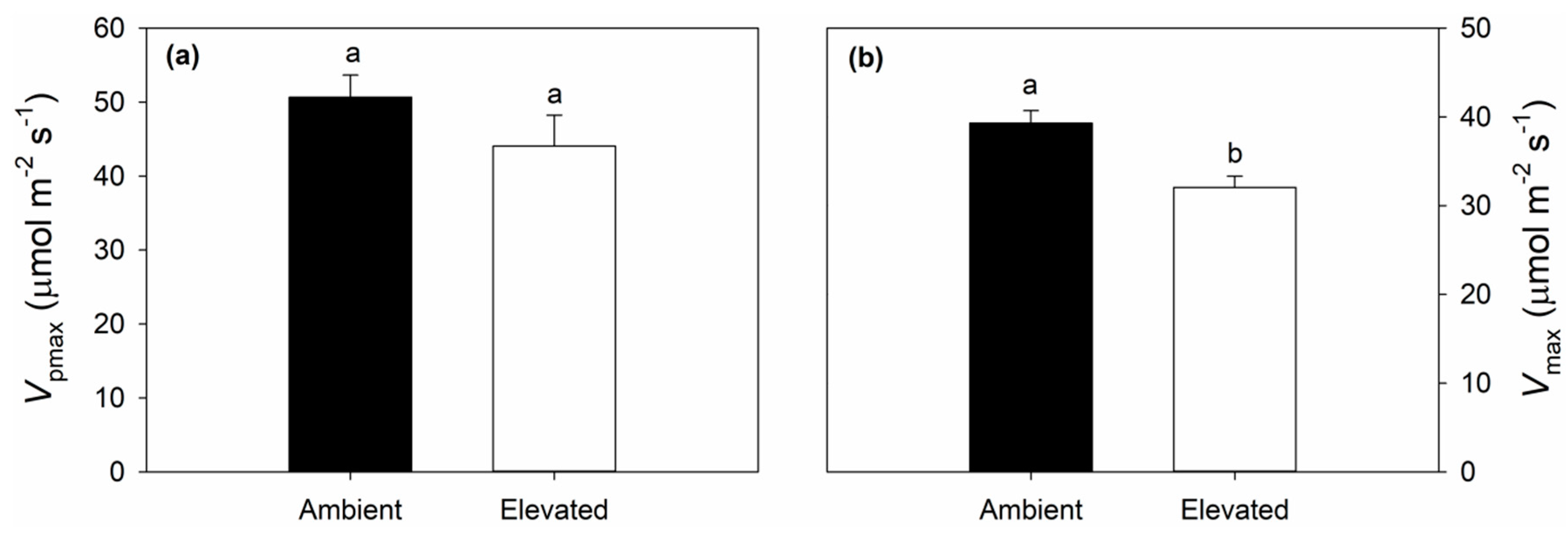
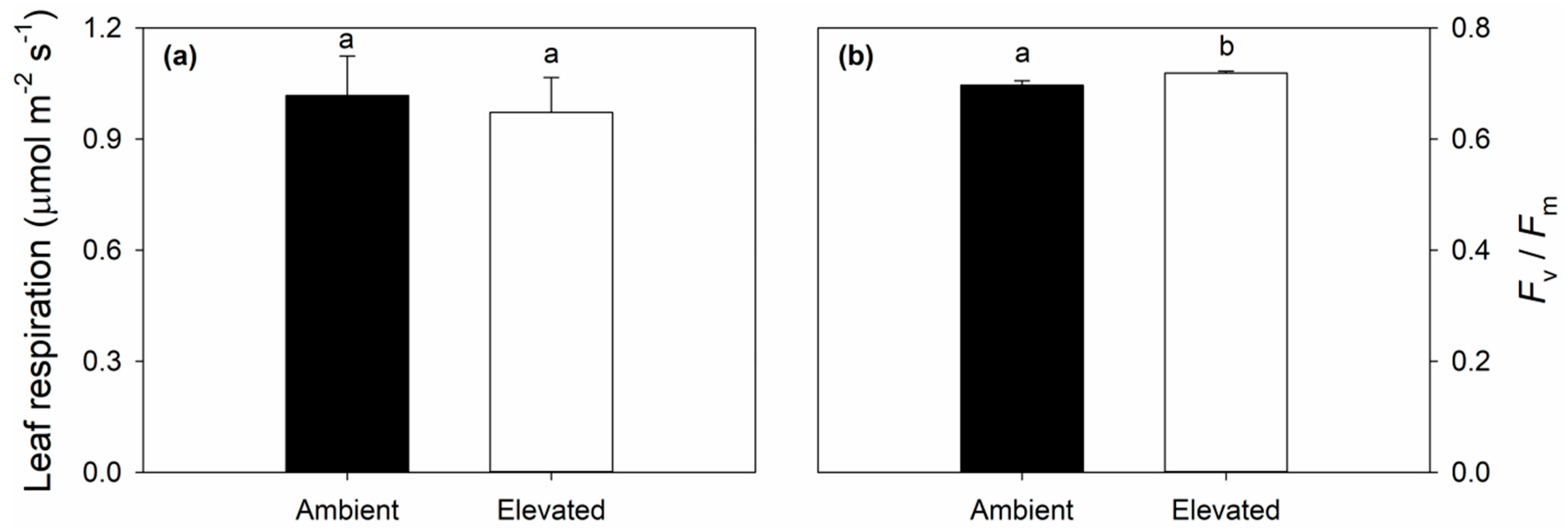
2.3. Leaf Respiration and Dark Adapted Chlorophyll Fluorescence Responses to Elevated O3
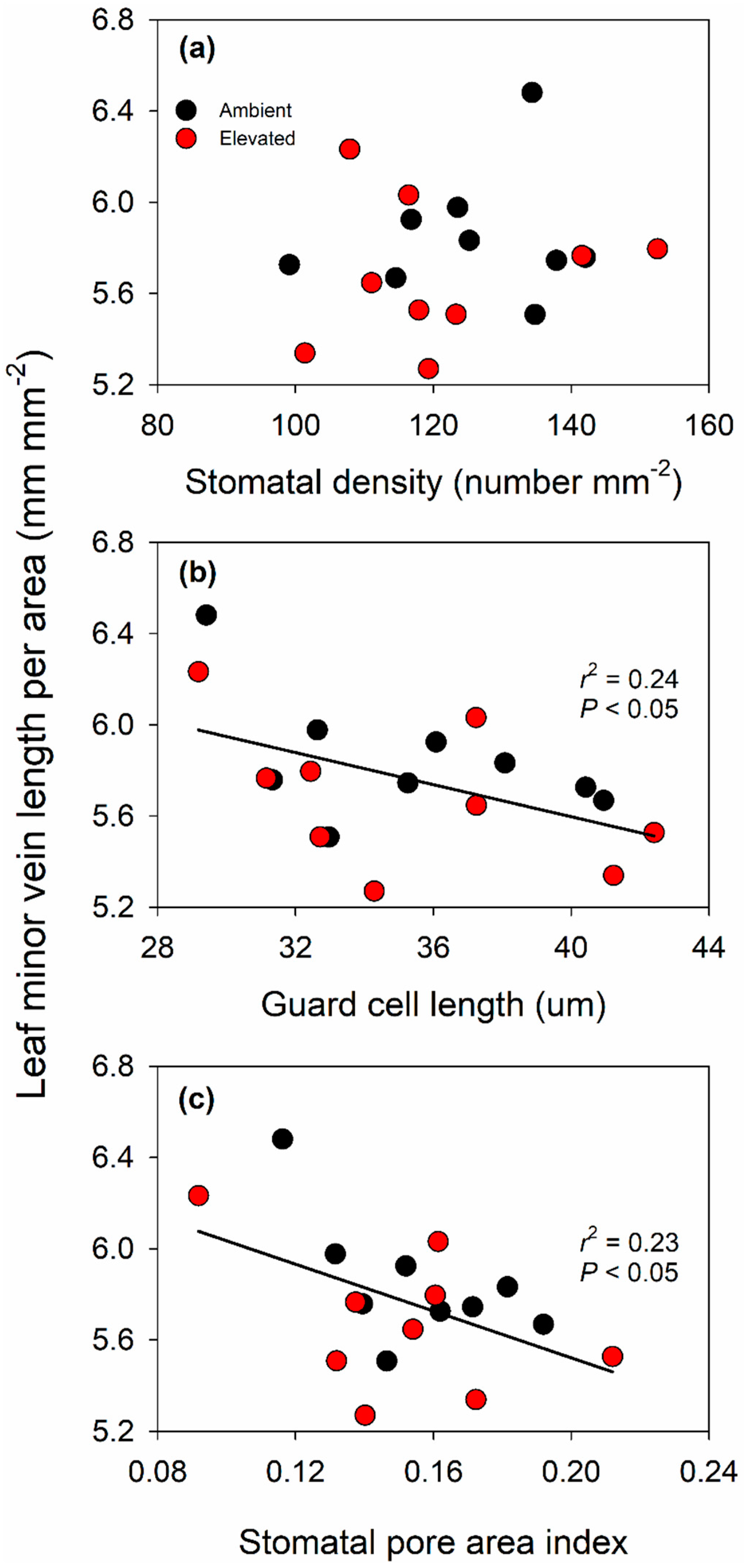
2.4. Leaf Morphology and Anatomy Were Not Altered by Elevated O3
2.5. No Changes in Biomass and Nutrient Composition between Ambient and Elevated O3
3. Discussion
3.1. Impact of Elevated O3 on Photosynthesis and Stomatal Conductance
3.2. Effect of Elevated O3 on Leaf Structure
3.3. Effect of Elevated O3 on Biomass and Nutrient Composition
3.4. Implications for Bioenergy Feedstock Development
4. Materials and Methods
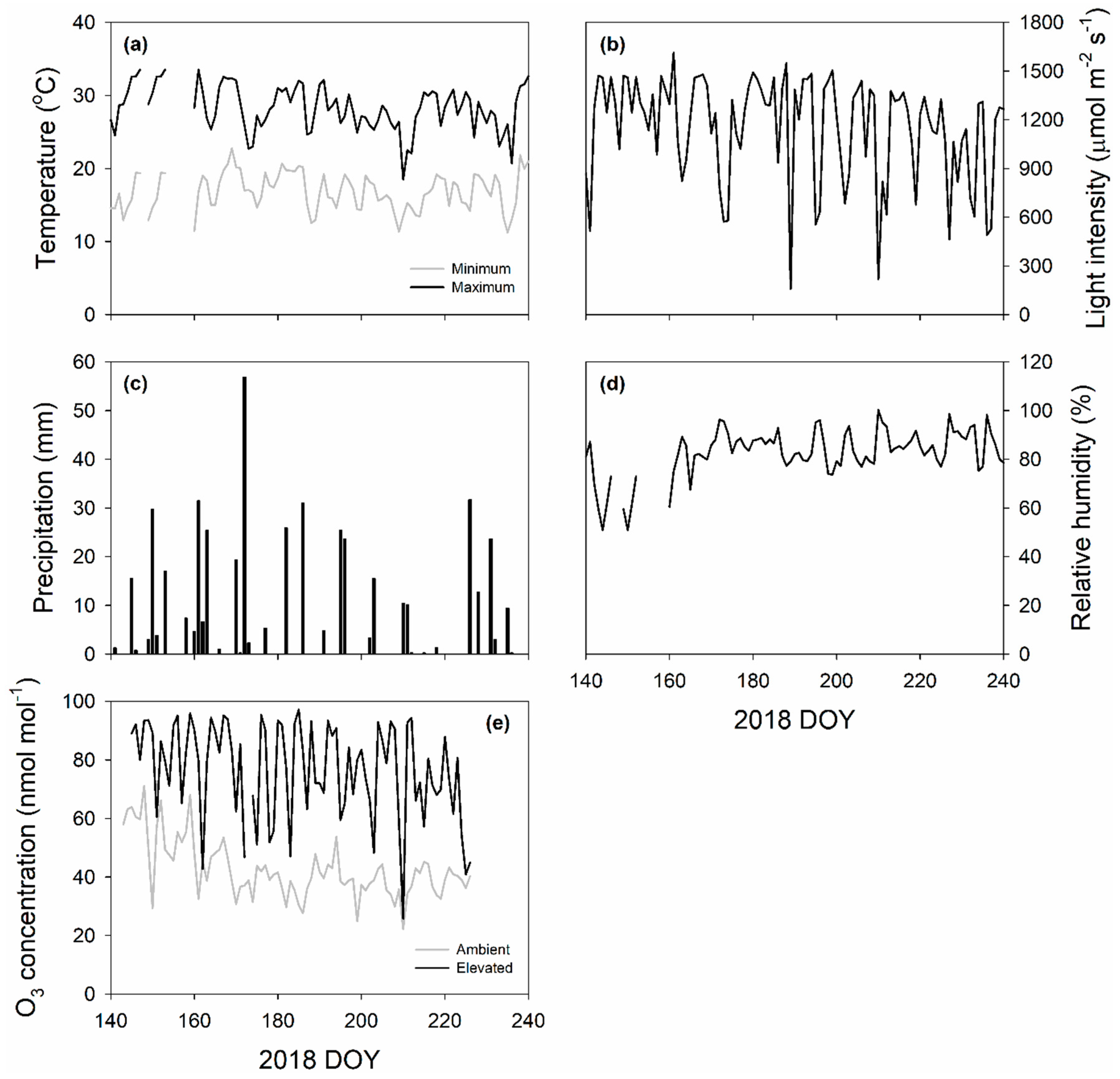
4.1. Field Site, Plant Material and Growth Condition
4.2. Leaf Midday Gas Exchange, Chlorophyll Fluorescence and A/Ci Curve
4.3. Ball–Woodrow–Berry Relationship
4.4. Dark Respiration and Dark-Adapted Chlorophyll Fluorescence
4.5. Leaf Anatomy
4.6. Biomass, C and N Content and Nutrient Composition Quantification
4.7. Statistical Analysis
Author Contributions
Acknowledgements
Conflicts of Interest
References
- McLaughlin, S.B.; De La Torre Ugarte, D.G.; Jr Garten, C.T.; Lynd, L.R.; Sanderson, M.A.; Tolbert, V.R.; Wolf, D.D. High-value renewable energy from prairie grasses. Environ. Sci. Technol. 2002, 36, 2122–2129. [Google Scholar] [CrossRef]
- Tilman, D.; Socolow, R.; Foley, J.A.; Hill, J.; Larson, E.; Lynd, L.; Pacala, S.; Reilly, J.; Searchinger, T.; Somerville, C.; et al. Beneficial biofuels-the food, energy, and environment trilemma. Science 2009, 325, 270–271. [Google Scholar] [CrossRef] [PubMed]
- Wullschleger, S.D.; Davis, E.B.; Borsuk, M.E.; Gunderson, C.A.; Lynd, L.R. Biomass production in switchgrass across the United States: Database description and determinants of yield. Agron. J. 2010, 102, 1158–1168. [Google Scholar] [CrossRef]
- Vadas, P.A.; Barnett, K.H.; Undersander, D.J. Economics and energy of ethanol production from alfalfa, corn, and switchgrass in the upper Midwest, USA. Bioenerg. Res. 2008, 1, 44–55. [Google Scholar] [CrossRef]
- U.S. Department of Energy. U.S. Billion-Ton Update: Biomass Supply for a Bioenergy and Bioproducts Industry. In ORNL/TM-2011/224; Perlack, R.D., Stokes, B.J., Eds.; Oak Ridge National Laboratory: Oak Ridge, TN, USA, 2011. [Google Scholar]
- Moser, L.E.; Vogel, K.P. Switchgrass, big bluestem, and indiangrass. In An Introduction to Grassland Agriculture, 5th ed.; Barnes, R.F., Miller, D.A., Nelson, C.J., Eds.; Iowa State Univ. Press: Ames, IA, USA, 1995; pp. 409–420. [Google Scholar]
- McLaughlin, S.B.; Kszos, L.A. Development of switchgrass (Panicum virgatum) as a bioenergy feedstock in the United States. Biomass Bioenergy 2005, 28, 515–535. [Google Scholar] [CrossRef]
- Schmer, M.R.; Vogel, K.P.; Mitchell, R.B.; Perrin, R.K. Net energy of cellulosic ethanol from switchgrass. Proc. Natl. Acad. Sci. USA 2008, 105, 464–469. [Google Scholar] [CrossRef]
- Vogel, K.P. Energy production from forages (or American agriculture-back to the future). J. Soil Water Conserv. 1996, 51, 137–139. [Google Scholar]
- Casler, M.D.; Vogel, K.P.; Taliaferro, C.M.; Wynia, R.L. Latitudinal adaptation of switchgrass populations. Crop Sci. 2004, 44, 293–303. [Google Scholar] [CrossRef]
- Mitchell, R.; Vogel, K.P.; Sarath, G. Managing and enhancing switchgrass as a bioenergy feedstock. Biofuels Bioprod. Bioref. 2008, 2, 530–539. [Google Scholar] [CrossRef]
- Stroup, J.A.; Sanderson, M.A.; Muir, J.P.; McFarland, M.J.; Reed, R.L. Comparison of growth and performance in upland and lowland switchgrass types to water and nitrogen stress. Bioresour. Technol. 2003, 86, 65–72. [Google Scholar] [CrossRef]
- Barney, J.N.; Jeremiah Mann, J.; Kyser, G.B.; Blumwald, E.; Van Deynze, A.; DiTomaso, J.M. Tolerance of switchgrass to extreme soil moisture stress: Ecological implications. Plant Sci. 2009, 177, 724–732. [Google Scholar] [CrossRef]
- Jiang, Y.; Yao, Y.; Wang, Y. Physiological response, cell wall components, and gene expression of switchgrass under short-term drought stress and recovery. Crop Sci. 2012, 52, 2718–2727. [Google Scholar] [CrossRef]
- Kim, S.; Rayburn, A.L.; Voigt, T.; Parrish, A.; Lee, D.K. Salinity effects on germination and plant growth of prairie cordgrass and switchgrass. Bioenerg. Res. 2012, 5, 225–235. [Google Scholar] [CrossRef]
- Sanderson, M.A.; Adler, P.R.; Boateng, A.A.; Casler, M.D.; Sarath, G. Switchgrass as a biofuels feedstoack in the USA. Can. J. Plant Sci. 2006, 86, 1315–1325. [Google Scholar] [CrossRef]
- Sillman, S. The relation between ozone, NOx and hydrocarbons in urban and polluted rural environments. Atmos. Environ. 1999, 33, 1821–1845. [Google Scholar] [CrossRef]
- Atkinson, R. Atmospheric chemistry of VOCs and NOx. Atmos. Environ. 2000, 34, 2063–2101. [Google Scholar] [CrossRef]
- Fowler, D.; Amann, M.; Anderson, F.; Ashmore, M.; Cox, P.; Depledge, M.; Derwent, D.; Grennfelt, P.; Hewitt, N.; Hov, O.; et al. Ground-Level Ozone in the 21st Century: Future Trends, Impacts and Policy Implications; The Royal Society: London, UK, 2008. [Google Scholar]
- Sharma, P.; Kuniyal, J.C.; Chand, K.; Guleria, R.P.; Dhyani, P.P.; Chauhau, C. Surface ozone concentration and its behaviour with aerosols in the northwestern Himalaya, India. Atmos. Environ. 2013, 71, 44–53. [Google Scholar] [CrossRef]
- Monks, P.S.; Archibald, A.T.; Colette, A.; Cooper, O.; Coyle, M.; Derwent, R.; Fowler, D.; Granier, C.; Law, K.S.; Mills, G.E.; et al. Tropospheric ozone and its precursors form the urban to the global scale from air quality to short-lived climate forcer. Atmos. Chem. Phys. 2015, 15, 8889–8973. [Google Scholar] [CrossRef]
- Yuan, X.; Calatayud, V.; Jiang, L.; Manning, W.J.; Hayes, F.; Tian, Y.; Feng, Z. Assessing the effects of ambient ozone in China on snap bean genotypes by using ethylenediurea (EDU). Environ. Pollut. 2015, 205, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Ainsworth, E.A. Understanding and improving global crop response to ozone pollution. Plant J. 2017, 90, 886–897. [Google Scholar] [CrossRef] [PubMed]
- Auvray, M.; Bey, I. Long-range transport to Europe: Seasonal variations and implications for the European ozone budget. J. Geophys. Res. 2005, 110, D11303. [Google Scholar] [CrossRef]
- Liao, K.J.; Hou, X.; Baker, D.R. Impacts of interstate transport of pollutants on high ozone events over the Mid-Atlantic United States. Atmos. Environ. 2014, 84, 100–112. [Google Scholar] [CrossRef]
- Weiss-Penzias, P.; Jaffe, D.A.; Jaegle´, L.; Liang, Q. Influence of long-range-transported pollution on the annual and diurnal cycles of carbon monoxide and ozone at Cheeka Peak Observatory. J. Geophys. Res. 2004, 109, D23S14. [Google Scholar] [CrossRef]
- Feng, Z.; Kobayashi, K.; Ainsworth, E.A. Impact of elevated ozone concentration on growth, physiology, and yield of wheat (Triticum aestivum L.): A meta-analysis. Glob. Chang. Biol. 2008, 14, 2696–2708. [Google Scholar]
- Wittig, V.E.; Ainsworth, E.A.; Naidu, A.L.; Karnosky, D.F.; Long, S.P. Quantifying the impact of current and future tropospheric ozone on the tree biomass, growth, physiology and biochemistry: A quantitative meta-analysis. Glob. Chang. Biol. 2009, 15, 396–424. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Yendrek, C.R.; Sitch, S.; Collins, W.J.; Emberson, L.D. The effects of tropospheric ozone on net primary productivity and implications for climate change. Annu. Rev. Plant. Biol. 2012, 63, 637–661. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, S.; Mills, G.; Illidge, R.; Davies, W.J. How is ozone pollution reducing our food supply? J. Exp. Bot. 2012, 63, 527–536. [Google Scholar] [CrossRef] [PubMed]
- McGrath, J.M.; Betzelberger, A.M.; Wang, S.; Shook, E.; Zhu, X.-G.; Long, S.P.; Ainsworth, E.A. An analysis of ozone damage to historical maize and soybean yields in the United States. Proc. Natl. Acad. Sci. USA 2015, 112, 14390–14395. [Google Scholar] [CrossRef] [PubMed]
- Rebouças, D.M.; De Sousa, Y.M.; Bagard, M.; Costa, J.H.; Jolivet, Y.; De Melo, D.F.; Repellin, A. Combined effects of ozone and drought on the physiology and membrane lipids of two cowpea (Vigna unguiculata (L.) Walp) cultivars. Plants 2017, 6, 14. [Google Scholar] [CrossRef] [PubMed]
- Mills, G.; Sharps, K.; Simpson, D.; Pleijel, H.; Broberg, M.; Uddling, J.; Jaramillo, F.; Davies, W.; Dentener, F.; Van den Berg, M.; et al. Ozone pollution will compromise efforts to increase global wheat production. Glob. Chang. Biol. 2018, 24, 3560–3574. [Google Scholar] [CrossRef]
- Ainsworth, E.A.; Lemonnier, P.; Wedow, J.M. The influence of rising tropospheric carbon dioxide and ozone on plant productivity. Plant Biol. 2019. [CrossRef]
- Heath, R.L. The Biochemistry of Ozone Attack on the Plasma Membrane of Plant Cells. In Phytochemical Effects of Environmental Compounds. Recent Advances in Phytochemistry; Saunders, J.A., Kosak-Channing, L., Conn, E.E., Eds.; Springer: Boston, MA, 1987; Volume 21. [Google Scholar]
- Long, S.P.; Naidu, S.L. Effect of oxidants at the biochemical, cell and physiological levels, with particular reference to ozone. In Air Pollution and Plant Life; Bell, J.N.B., Treshow, M., Eds.; John Wiley & Sons, Ltd.: West Sussex, UK, 2002; pp. 69–88. [Google Scholar]
- Pasqualini, S.; Piccioni, C.; Reale, L.; Ederli, L.; Della, T.G.; Ferranti, F. Ozone-induced cell death in tobacco cultivar Bel W3 plant. The role of programmed cell death in lesion formation. Plant Physiol. 2003, 133, 1122–1134. [Google Scholar] [CrossRef]
- Fiscus, E.L.; Booker, F.L.; Burkey, K.O. Crop responses to ozone: Uptake, models of action, carbon assimilation and partitioning. Plant Cell Environ. 2005, 28, 997–1011. [Google Scholar] [CrossRef]
- Li, S.; Harley, P.C.; Niinemets, Ü. Ozone-induced foliar damage and release of stress volatiles is highly dependent on stomatal openness and priming by low-level ozone exposure in Phaseolus vulgaris. Plant Cell Environ. 2017, 40, 1984–2003. [Google Scholar] [CrossRef]
- Flowers, M.D.; Fiscus, E.L.; Burkey, K.O.; Booker, F.L.; Dubois, J.-J.B. Photosynthesis, chlorophyll fluorescence, and yield of snap bean (Phaseolus vulgaris L.) genotypes differing in sensitivity to ozone. Environ. Exp. Bot. 2007, 61, 190–198. [Google Scholar] [CrossRef]
- Guidi, L.; Degl’Innocenti, E.; Martinelli, F.; Piras, M. Ozone effects on carbon metabolism in sensitive and insensitive Phaseolus cultivars. Environ. Exp. Bot. 2009, 66, 117–125. [Google Scholar] [CrossRef]
- Li, S.; Tosens, T.; Harley, P.C.; Jiang, Y.; Kanagendran, A.; Grosberg, M.; Jaamets, K.; Niinemets, Ü. Glandular trichomes as a barrier against atmospheric oxidative stress: Relationships with ozone uptake, leaf damage, and emission of LOX products across a diverse set of species. Plant Cell Environ. 2018, 40, 1263–1277. [Google Scholar] [CrossRef]
- Masutomi, Y.; Kinose, Y.; Takimoto, T.; Yonekura, T.; Oue, H.; Kobayashi, K. Ozone changes the linear relationship between photosynthesis and stomatal conductance and decreases water use efficiency in rice. Sci. Total Environ. 2019, 655, 1009–1016. [Google Scholar] [CrossRef]
- Paoletti, E.; Grulke, N.E. Does living in elevated CO2 ameliorate tree response to ozone? A review on stomatal responses. Environ. Pollut. 2005, 137, 483–493. [Google Scholar] [CrossRef]
- Vahisalu, T.; Puzõrjova, I.; Brosché, M.; Valk, E.; Lepiku, M.; Moldau, H.; Pechter, P.; Wang, Y.-S.; Lindgren, O.; Salojärvi, J.; et al. Ozone-triggered rapid stomatal response involves the production of reactive oxygen species, and is controlled by SLAC1 and OST1. Plant J. 2010, 62, 442–453. [Google Scholar] [CrossRef]
- Paoletti, E.; Grulke, N.E. Ozone exposure and stomatal sluggishness in different plant physiognomic classes. Environ. Pollut. 2010, 158, 2664–2671. [Google Scholar] [CrossRef]
- Hoshika, Y.; Omasa, K.; Paoletti, E. Both ozone exposure and soil water stress are able to induce stomatal sluggishness. Environ. Exp. Bot. 2013, 88, 19–23. [Google Scholar] [CrossRef]
- Ball, J.T.; Woodrow, I.E.; Berry, J.A. A model predicting stomatal conductance and its contribution to the control of photosynthesis under different environmental conditions. In Progress in Photosynthesis Research; Biggins, J., Ed.; Martinus Nijhoff Publishers: Dordrecht, The Netherlands, 1987; pp. 221–224. [Google Scholar]
- Wolz, K.J.; Wertin, T.M.; Abordo, M.; Wang, D.; Leakey, A.D.B. Diversity in stomatal function is integral to modelling plant carbon and water fluxes. Nat. Ecol. Evol. 2017, 1, 1292–1298. [Google Scholar] [CrossRef]
- Leitao, L.; Bethenod, O.; Biolley, J.-P. The impact of ozone on juvenile maize (Zea mays L.) plant photosynthesis: Effects on vegetative biomass, pigmentation, and carboxylases (PEPc and rubisco). Plant Biol. 2007, 9, 478–488. [Google Scholar] [CrossRef]
- Leitao, L.; Maoret, J.J.; Biolley, J.P. Changes in PEP carboxylase, rubisco and rubisco activase mRNA levels from maize (Zea mays) exposed to a chronic ozone stress. Biol. Res. 2007, 40, 137–153. [Google Scholar] [CrossRef]
- Singh, A.A.; Agrawal, S.B.; Shahi, J.P.; Agrawal, M. Assessment of growth and yield losses in two Zea mays L. cultivars (quality protein maize and nonquality protein maize) under projected levels of ozone. Environ. Sci. Pollut. Res. 2014, 21, 2628–2641. [Google Scholar] [CrossRef]
- Yendrek, C.R.; Erice, G.; Montes, C.M.; Tomaz, T.; Sorgini, C.A.; Brown, P.J.; McIntyre, L.M.; Leakey, A.D.B.; Ainsworth, E.A. Elevated ozone reduces photosynthetic carbon gain by accelerating leaf senescence of inbred and hybrid maize in a genotype-specific manner. Plant Cell Environ. 2017, 40, 3088–3100. [Google Scholar] [CrossRef]
- Yendrek, C.R.; Tomaz, T.; Montes, C.M.; Cao, Y.; Morse, A.M.; Brown, P.J.; McIntyre, L.M.; Leakey, A.D.B.; Ainsworth, E.A. High-throughput phenotyping of maize leaf physiological and biochemical traits using hyperspectral reflectance. Plant Physiol. 2017, 173, 614–626. [Google Scholar] [CrossRef]
- Grantz, D.A.; Vu, H.B. O3 sensitivity in a potential C4 bioenergy crop: Sugarcane in California. Crop Sci. 2009, 49, 643–650. [Google Scholar] [CrossRef]
- Grantz, D.A.; Vu, H.B.; Tew, T.L.; Veremis, J.C. Sensitivity of gas exchange parameters to ozone in diverse C4 sugarcane hybrids. Crop Sci. 2012, 52, 1270–1280. [Google Scholar] [CrossRef]
- Moura, B.B.; Hoshika, Y.; Ribeiro, R.V.; Paoletti, E. Exposure- and flux-based assessment of ozone risk to sugarcane plants. Atmos. Environ. 2018, 176, 252–260. [Google Scholar] [CrossRef]
- Bussotti, F. Functional leaf traits, plant communities and acclimationprocesses in relation to oxidative stress in trees: A critical overview. Glob. Chang. Biol. 2008, 14, 2727–2739. [Google Scholar]
- Grass Phylogeny Working Group. Phylogeny and subfamilial classification of the grasses (Poaceae). Ann. Mo. Bot. Gard. 2001, 88, 373–457. [Google Scholar] [CrossRef]
- Reich, P.B. Quantifying plant response to ozone: A unifying theory. Tree Physiol. 1987, 3, 63–91. [Google Scholar] [CrossRef]
- Hoshika, Y.; Katata, G.; Deushi, M.; Watanabe, M.; Koike, T.; Paoletti, E. Ozone-induced stomatal sluggishness changes carbon and water balance of temperate deciduous forests. Sci. Rep. 2015, 5, 09871. [Google Scholar] [CrossRef]
- Li, P.; Calatayud, V.; Gao, F.; Uddling, J.; Feng, Z. Differences in ozone sensitivity among woody species are related to leaf morphology and antioxidant levels. Tree Physiol. 2016, 36, 1105–1116. [Google Scholar] [CrossRef]
- Feng, Z.; Büker, P.; Pleijel, H.; Emberson, L.; Karlsson, P.E.; Uddling, J. A unifying explanation for variation in ozone sensitivity among woody plants. Glob. Chang. Biol. 2018, 24, 78–84. [Google Scholar] [CrossRef]
- Pignon, C.P.; Lundgren, M.R.; Osborne, C.P.; Long, S.P. Bundle sheath chloroplast volume can house sufficient Rubisco to avoid limiting C4 photosynthesis during chilling. J. Exp. Bot. 2019, 70, 357–365. [Google Scholar] [CrossRef]
- Zhang, W.-W.; Wang, M.; Wang, A.-Y.; Yin, X.-H.; Feng, Z.-Z.; Hao, G.-Y. Elevated ozone concentration decreases whole-plant hydraulic conductance and disturbs water use regulation in soybean plants. Physiol. Plant. 2018, 163, 183–195. [Google Scholar] [CrossRef]
- Hu, J.; Yang, Q.-Y.; Huang, W.; Zhang, S.B.; Hu, H. Effects of temperature on leaf hydraulic architecture of tobacco plants. Planta 2014, 240, 489–496. [Google Scholar] [CrossRef] [PubMed]
- Uhl, D.; Mosbrugger, V. Leaf venation density as a climate and environmental proxy: A critical review and new data. Palaeogeogr. Palaeoclimatol. Palaeoecol. 1999, 149, 15–26. [Google Scholar] [CrossRef]
- Kouwenberg, L.L.; Kürschner, W.M.; McElwain, J.C. Stomatal frequency change over altitudinal gradients: Prospects for paleoaltimetry. Rev. Mineral. Geochem. 2007, 66, 215–241. [Google Scholar] [CrossRef]
- Carins Murphy, M.R.; Jordan, G.J.; Brodribb, T.J. Acclimation to humidity modifies the link between leaf size and the density of veins and stomata. Plant Cell Environ. 2014, 37, 124–131. [Google Scholar] [CrossRef]
- Brodribb, T.J.; Jordan, G.J.; Carpenter, R.J. Unified changes in cell size permit coordinated lead evolution. New Phytol. 2013, 199, 559–570. [Google Scholar] [CrossRef]
- Leisner, C.P.; Ainsworth, E.A. Quantifying the effects of ozone on plant reproductive growth and development. Glob. Chang. Biol. 2012, 18, 606–616. [Google Scholar] [CrossRef]
- Yendrek, C.R.; Leisner, C.P.; Ainsworth, E.A. Chronic ozone exacerbates the reduction in photosynthesis and acceleration of senescence caused by limited N availability in Nicotiana sylvestris. Glob. Chang. Biol. 2013, 19, 3155–3166. [Google Scholar] [CrossRef] [PubMed]
- Longstreth, D.J.; Nobel, P.S. Nutrient influences on leaf photosynthesis. Plant Physiol. 1980, 65, 541–543. [Google Scholar] [CrossRef] [PubMed]
- Spiller, S.; Terry, N. Limiting factors in photosynthesis: II. iron stress diminishes photochemical capacity by reducing the number of photosynthetic units. Plant Physiol. 1980, 65, 121–125. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Oosterhuis, D.M.; Bednarz, C.W. Influence of potassium deficiency on photosynthesis, chlorophyll content, and chloroplast ultrastructure of cotton plants. Photosynthetica 2001, 39, 103–109. [Google Scholar] [CrossRef]
- Lewandowski, I.; Kicherer, A. Combustion quality of biomass: Practical relevance and experiments to modify the biomass quality of Miscanthus × giganteus. Eur. J. Agron. 1997, 6, 163–177. [Google Scholar] [CrossRef]
- Reich, P.B.; Schoettle, A.W. Effects of ozone and acid rain on white pine (Pinus strobus) seedlings grown in five soils. III Nutrient relations. Can. J. Bot. 1988, 66, 1517–1531. [Google Scholar] [CrossRef]
- Oksanen, E.; Riikonen, J.; Kaakinen, S.; Holopainen, T.; Vapaavuori, E. Structural characteristics and chemical composition of birch (Betula pendula) leaves are modified by increasing CO2 and ozone. Glob. Chang. Biol. 2005, 11, 732–748. [Google Scholar] [CrossRef]
- Thomas, V.F.D.; Braun, S.; Flückiger, W. Effects of simultaneous ozone exposure and nitrogen loads on carbohydrate concentrations, biomass, growth, and nutrient concentrations of young beech trees (Fagus sylvatica). Environ. Pollut. 2006, 143, 341–354. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, G.D.; O’Leary, M.H.; Berry, J.A. On the relationship between carbon isotope discrimination and the intercellular carbon dioxide concentration in leaves. Aust. J. Plant Physiol. 1982, 9, 121–137. [Google Scholar] [CrossRef]
- Farquhar, G.D.; Ehleringer, J.R.; Hubick, K.T. Carbon isotope discrimination and photosynthesis. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1989, 40, 503–537. [Google Scholar] [CrossRef]
- Farquhar, G.D.; Richards, R.A. Isotopic composition of plant carbon correlates with water-use efficiency of wheat genotypes. Aust. J. Plant Physiol. 1984, 11, 539–552. [Google Scholar] [CrossRef]
- Handley, L.L.; Raven, J.A. The use of natural abundance of nitrogen isotopes in plant physiology and ecology. Plant Cell Environ. 1992, 15, 965–985. [Google Scholar] [CrossRef]
- Evans, R.D. Physiological mechanisms influencing plant nitrogen isotope composition. Trends Plant Sci. 2001, 6, 121–126. [Google Scholar] [CrossRef]
- Robinson, D. δ15N as an integrator of the nitrogen cycle. Trends Ecol. Evol. 2001, 16, 153–162. [Google Scholar] [CrossRef]
- Morgan, P.B.; Bernacchi, C.J.; Ort, D.R.; Long, S.P. An in vivo analysis of the effect of season-long open-air elevation of ozone to anticipated 2050 levels on photosynthesis in soybean. Plant Physiol. 2004, 135, 2348–2357. [Google Scholar] [CrossRef]
- Naidu, S.L.; Long, S.P. Potential mechanisms of low-temperature tolerance of C4 photosynthesis in Miscanthus × giganteus: An in vivo analysis. Planta 2004, 220, 145–155. [Google Scholar] [CrossRef]
- Leakey, A.D.B.; Uribelarrea, M.; Ainsworth, E.A.; Naidu, S.L.; Rogers, A.; Ort, D.R.; Long, S.P. Photosynthesis, productivity, and yield of maize are not affected by open-air elevation of CO2 concentration in the absence of drought. Plant Physiol. 2006, 140, 779–790. [Google Scholar] [CrossRef]
- Nardini, A.; Gortan, E.; Ramani, M.; Salleo, S. Heterogeneity of gas exchange rates over the leaf surface in tobacco: An effect of hydraulic architecture? Plant Cell Environ. 2008, 31, 804–812. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Zhang, Y.J.; Sack, L.; Scoffoni, C.; Ishida, A.; Chen, Y.-J.; Cao, K.-F. Heterogeneity and spatial pattering of structure and physiology across the leaf surface in giant leaves of Alocasia macrorrhiza. PLoS ONE 2013, 8, e66016. [Google Scholar]
- Bernacchi, C.J.; Morgan, P.B.; Ort, D.R.; Long, S.P. The growth of soybean under free air [CO2] enrichment (FACE) stimulates photosynthesis while decreasing in vivo Rubisco capacity. Planta 2005, 220, 434–446. [Google Scholar] [CrossRef] [PubMed]
- Farquhar, G.D.; von Caemmere, S.; Berry, J.A. A biochemical model of photosynthetic CO2 fixation in C3 species. Planta 1980, 149, 178–190. [Google Scholar] [CrossRef]
- Von Caemmerer, S. Biochemcial models of leaf photosynthesis; CSIRO Publishing: Collingwood, Australia, 2000. [Google Scholar]
- Markelz, R.J.C.; Strellner, R.S.; Leakey, A.D.B. Impairment of C4 photosynthesis by drought is exacerbated by limiting nitrogen and ameliorated by elevated [CO2] in maize. J. Exp. Bot. 2011, 62, 3235–3246. [Google Scholar] [CrossRef]
- Twidwell, E.K.; Johnson, K.D.; Patterson, J.A.; Cherney, J.H.; Bracker, C.E. Degradation of switchgrass anatomical tissue by rumen microorganisms. Crop Sci. 1991, 30, 1321–1328. [Google Scholar] [CrossRef]
- Christin, P.-A.; Osborne, C.P.; Chatelet, D.S.; Columbus, J.T.; Besnard, G.; Hodkinson, T.R.; Garrison, L.M.; Vorontsove, M.S.; Edwards, E.J. Anatomical enablers and the evolution of C4 photosynthesis in grasses. Proc. Natl. Acad. Sci. USA 2013, 110, 1381–1386. [Google Scholar] [CrossRef]
- Sack, L.; Cowan, P.D.; Jaikumar, N.; Holbrook, N.M. The hydrology of leaves: Co-ordination of structure and function in temperate woody species. Plant Cell Environ. 2003, 26, 1343–1356. [Google Scholar] [CrossRef]
- Maillard, A.; Etienne, P.; Diquélou, S.; Trouverie, J.; Billard, V.; Yvin, J.C.; Ourry, A. Nutrient deficiencies modify the ionomic composition of plant tissues: A focus on cross-talk between molybdenum and other nutrients in Brassica napus. J. Exp. Bot. 2016, 67, 5631–5641. [Google Scholar] [CrossRef] [PubMed]
- Warton, D.I.; Wright, I.J.; Falster, D.S.; Westoby, M. Bivariate line-fitting methods for allometry. Biol. Rev. 2006, 81, 259–291. [Google Scholar] [CrossRef] [PubMed]
| Ambient O3 | Elevated O3 | |
|---|---|---|
| Bundle sheath density (number mm−1) | 5.63 ± 0.13 (a) | 5.70 ± 0.11 (a) |
| Distance between secondary vein (mm) | 1.08 ± 0.056 (a) | 0.97 ± 0.025 (a) |
| Leaf thickness (µm) | 203.3 ± 4.99 (a) | 195.9 ± 5.81 (a) |
| Interveinal distance (IVD, µm) | 175.2 ± 3.56 (a) | 173.1 ± 2.65 (a) |
| Conduit diameter (µm) | 32.5 ± 1.09 (a) | 31.6 ± 1.22 (a) |
| Conduit size (µm2) | 781.3 ± 43.3 (a) | 723.8 ± 50.5 (a) |
| Out bundle sheath size (µm2) | 20885.2 ± 891.6 (a) | 20315.1 ± 945.2 (a) |
| Inner bundle sheath size (µm2) | 12443.0 ± 515.7 (a) | 11928.3 ± 625.7 (a) |
| Vein size (µm2) | 4263.3 ± 248.1 (a) | 4034.5 ± 289.0 (a) |
| Colorless cell size (µm2) | 336.2 ± 21.4 (a) | 364.2 ± 33.1 (a) |
| Upper epidermis cell size (µm2) | 141.0 ± 5.52 (a) | 143.9 ± 6.87 (a) |
| Lower epidermis cell size (µm2) | 205.9 ± 11.4 (a) | 200.9 ± 14.0 (a) |
| Motor cell size (µm2) | 767.9 ± 39.6 (a) | 794.9 ± 50.7 (a) |
| Sclerenchyma size (µm2) | 755.4 ± 43.2 (a) | 657.5 ± 50.4 (a) |
| Stomatal density (mm−2) | 125.3 ± 4.55 (a) | 121.1 ± 5.42 (a) |
| Guard cell length (µm) | 35.2 ± 1.34 (a) | 35.3 ± 1.51 (a) |
| Stomatal pore area index (SPI, ×10−2) | 0.15 ± 0.0081 (a) | 0.15 ± 0.011 (a) |
| Vein density (mm mm−2) | 5.85 ± 0.092 (a) | 5.68 ± 0.10 (a) |
| Ambient O3 | Elevated O3 | |
|---|---|---|
| Leaf area (cm2 plant−1) | 5624.5 ± 659.6 (a) | 6681.1 ± 875.3 (a) |
| Leaf biomass (g plant−1) | 36.4 ± 4.28 (a) | 44.1 ± 4.58 (a) |
| Leaf mass per area (LMA, g m−2) | 64.8 ± 1.12 (a) | 69.4 ± 2.60 (a) |
| Tiller number | 31.0 ± 2.96 (a) | 32.4 ± 2.45 (a) |
| Tiller biomass (g plant−1) | 75.2 ± 8.89 (a) | 97.0 ± 13.12 (a) |
| Leaf area per tiller (cm2 branch−1) | 178.8 ± 13.99 (a) | 198.6 ± 13.37 (a) |
| Leaf mass per tiller (g plant−1) | 1.15 ± 0.086 (a) | 1.36 ± 0.054 (a) |
| Average tiller mass (g) | 2.39 ± 0.20 (a) | 2.87 ± 0.21 (a) |
| Leaf N (%) | 2.53 ± 0.045 (a) | 2.49 ± 0.035 (a) |
| Leaf C:N | 17.8 ± 0.29 (a) | 18.1 ± 0.24 (a) |
| Stem N (%) | 1.36 ± 0.057 (a) | 1.39 ± 0.049 (a) |
| Stem C:N | 33.0 ± 1.49 (a) | 32.2 ± 1.23 (a) |
| Ambient O3 | Elevated O3 | |
|---|---|---|
| Mg (mg kg−1) | 5079.1 ± 229.8 (a) | 5617.5 ± 285.2 (a) |
| P (mg kg−1) | 2466.8 ± 65.9 (a) | 2732.1 ± 146.9 (a) |
| S (mg kg−1) | 2326.0 ± 77.0 (a) | 2316.1 ± 86.1 (a) |
| K (mg kg−1) | 18695.6 ± 737.5 (a) | 16245.0 ± 688.3 (b) |
| Ca (mg kg−1) | 5994.7 ± 387.8 (a) | 7590.4 ± 685.4 (a) |
| B (mg kg−1) | 3.43 ± 0.10 (a) | 3.71 ± 0.086 (a) |
| Mn (mg kg−1) | 65.1 ± 10.7 (a) | 52.5 ± 9.91 (a) |
| Fe (mg kg−1) | 459.7 ± 61.1 (a) | 457.2 ± 43.2 (a) |
| Ni (mg kg-1) | 3.18 ± 0.26 (a) | 2.80 ± 0.23 (a) |
| Cu (mg kg−1) | 9.78 ± 0.43 (a) | 9.07 ± 0.33 (a) |
| Zn (mg kg−1) | 27.6 ± 2.18 (a) | 27.4 ± 2.21 (a) |
| Mo (mg kg−1) | 1.17 ± 0.069 (a) | 1.27 ± 0.24 (a) |
| Na (mg kg−1) | 106.3 ± 11.5 (a) | 142.7 ± 23.5 (a) |
| V (mg kg−1) | 0.36 ± 0.021 (a) | 0.36 ± 0.014 (a) |
| Co (mg kg−1) | 0.13 ± 0.012 (a) | 0.11 ± 0.0080 (a) |
| δ13C (‰) | −12.22 ± 0.060 (a) | −12.49 ± 0.080 (b) |
| δ15N (‰) | 4.04 ± 0.46 (a) | 5.35 ± 0.78 (a) |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, S.; Courbet, G.; Ourry, A.; Ainsworth, E.A. Elevated Ozone Concentration Reduces Photosynthetic Carbon Gain but Does Not Alter Leaf Structural Traits, Nutrient Composition or Biomass in Switchgrass. Plants 2019, 8, 85. https://doi.org/10.3390/plants8040085
Li S, Courbet G, Ourry A, Ainsworth EA. Elevated Ozone Concentration Reduces Photosynthetic Carbon Gain but Does Not Alter Leaf Structural Traits, Nutrient Composition or Biomass in Switchgrass. Plants. 2019; 8(4):85. https://doi.org/10.3390/plants8040085
Chicago/Turabian StyleLi, Shuai, Galatéa Courbet, Alain Ourry, and Elizabeth A. Ainsworth. 2019. "Elevated Ozone Concentration Reduces Photosynthetic Carbon Gain but Does Not Alter Leaf Structural Traits, Nutrient Composition or Biomass in Switchgrass" Plants 8, no. 4: 85. https://doi.org/10.3390/plants8040085
APA StyleLi, S., Courbet, G., Ourry, A., & Ainsworth, E. A. (2019). Elevated Ozone Concentration Reduces Photosynthetic Carbon Gain but Does Not Alter Leaf Structural Traits, Nutrient Composition or Biomass in Switchgrass. Plants, 8(4), 85. https://doi.org/10.3390/plants8040085





