A Review on Si Uptake and Transport System
Abstract
:1. Introduction
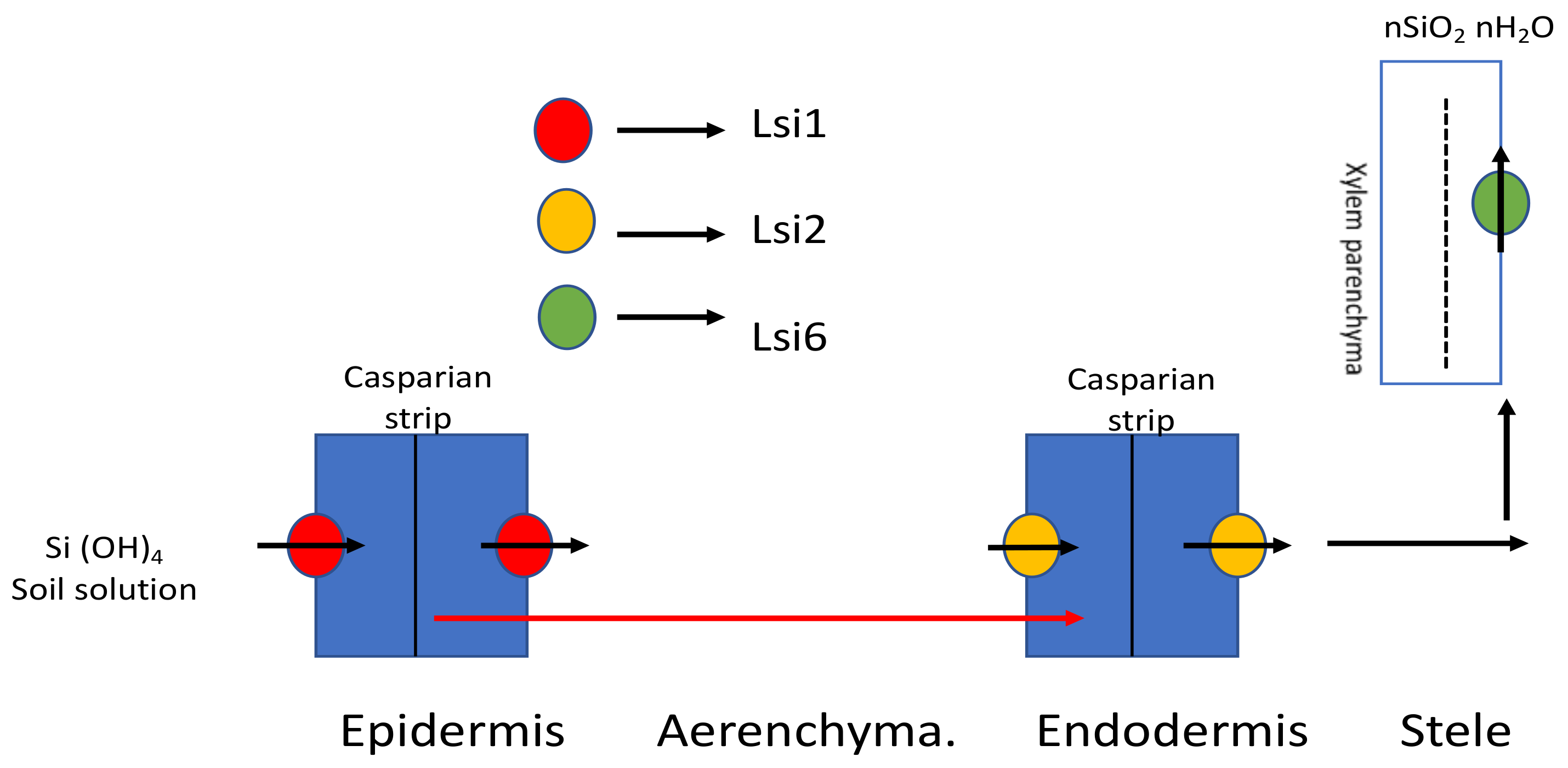
2. Mode of Cellular Si Uptake and Accumulation
3. Si Facilitation via Transporter Genes
3.1. Angiosperms
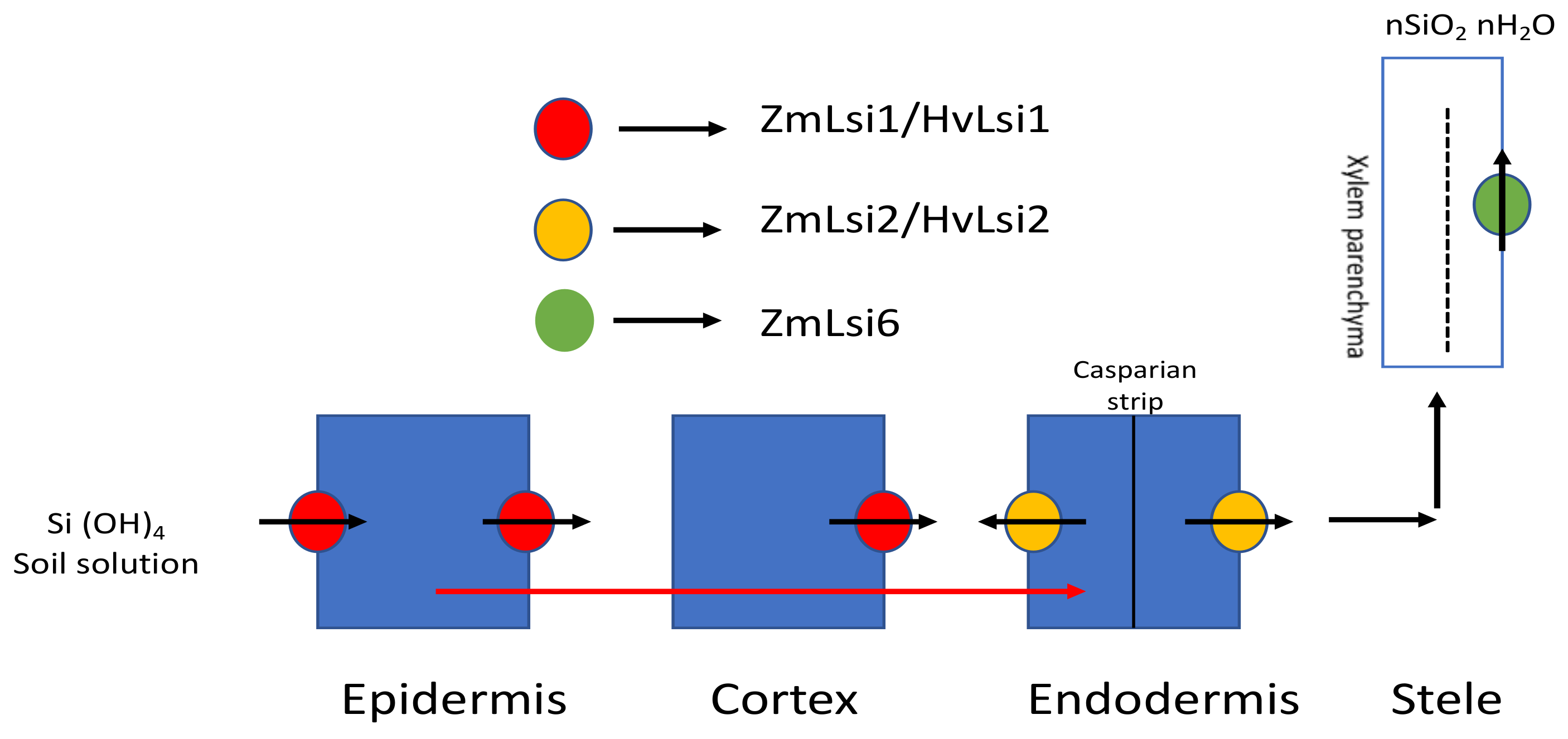
3.1.1. In Monocots
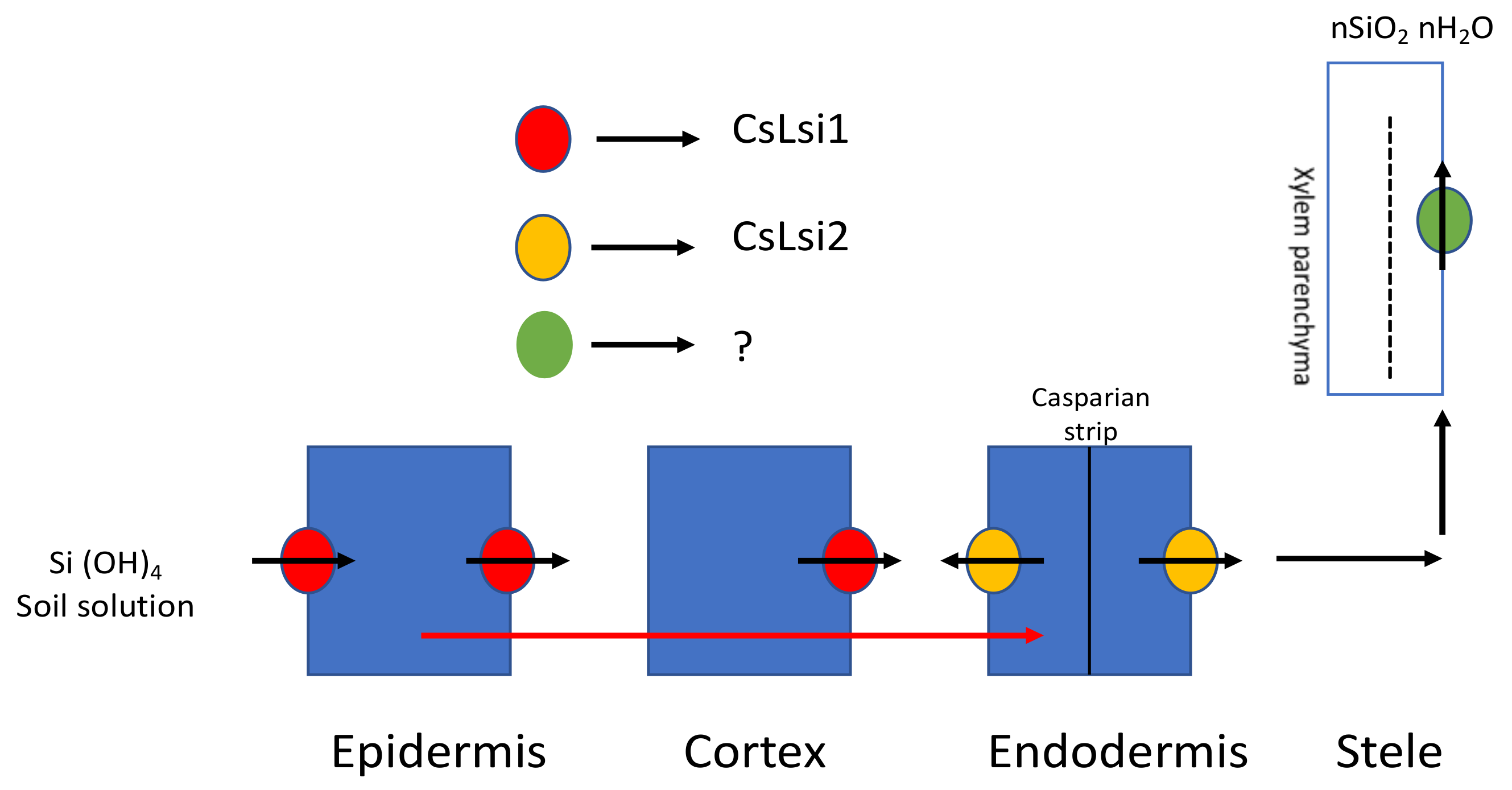
3.1.2. In Eudicots
3.2. Cryptogams
4. Conclusions and Future Prospects
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wedepohl, K.H. The composition of the continental crust. Geochim. Cosmochim. Acta 1995, 59, 1217–1232. [Google Scholar] [CrossRef]
- Pereira, C.E.D.B.; Felcman, J. Correlation between five minerals and the healing effect of Brazilian medicinal plants. Biol. Trace Elem. Res. 1998, 65, 251–259. [Google Scholar] [CrossRef] [PubMed]
- Savant, N.K.; Korndörfer, G.H.; Datnoff, L.E.; Snyder, G.H. Si nutrition and sugarcane production: A review. J. Plant Nutr. 1999, 22, 1853–1903. [Google Scholar] [CrossRef]
- Ma, J.F. Role of Si in enhancing the resistance of plants to biotic and abiotic stresses. Soil Sci. Plant Nutr. 2004, 50, 11–18. [Google Scholar] [CrossRef]
- Zhu, Y.; Gong, H. Beneficial effects of Si on salt and drought tolerance in plants. Agron. Sustain. Dev. 2014, 34, 455–472. [Google Scholar] [CrossRef]
- Adrees, M.; Ali, S.; Rizwan, M.; Zia-ur-Rehman, M.; Ibrahim, M.; Abbas, F. Mechanisms of Si-mediated alleviation of heavy metal toxicity in plants: A review. Ecotox. Environ. Saf. 2015, 119, 186–197. [Google Scholar] [CrossRef] [PubMed]
- Farooq, M.A.; Dietz, K.J. Silicon as versatile player in plant and human biology-Overlooked and poorly understood. Front. Plant Sci. 2015, 6, 994. [Google Scholar] [CrossRef]
- Deshmukh, R.; Belanger, R.R. Molecular evolution of aquaporins and Si influx in plants. The functional role of Si in plant Biology. Funct. Ecol. 2016, 30, 1277–1285. [Google Scholar] [CrossRef]
- Mitani, N.; Ma, J.F. Uptake system of Si in different plant species. J. Exp. Bot. 2005, 56, 1255–1261. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Takahashi, E. Si-Accumulating Plants in the Plant Kingdom. In Soil, Fertilizer, and Plant Si Research in Japan; Elsevier Science: Amsterdam, The Netherlands, 2002; pp. 63–71. [Google Scholar]
- Hodson, M.; White, P.; Mead, A.; Broadley, M. Phylogenetic variation in the Si composition of plants. Ann. Bot. 2005, 96, 1027–1046. [Google Scholar] [CrossRef]
- Cornelis, J.T.; Delvaux, B.; Georg, R.B.; Lucas, Y.; Ranger, J.; Opfergelt, S. Tracing the origin of dissolved Si transferred from various soil plant systems towards rivers: A review. Biogeosciences 2011, 8, 89–112. [Google Scholar] [CrossRef]
- Epstein, E. The anomaly of Si in plant biology. Proc. Natl. Acad. Sci. USA 1994, 91, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Miyakeb, Y.; Takahashi, E. Si as a Beneficial Element for Crop Plants. In Si in Agriculture; Datnoff, L.E., Snyder, G.H., Korndörfer, G.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; Volume 8, pp. 17–39. [Google Scholar]
- Takahashi, E.; Ma, J.F.; Miyake, Y. The possibility of Si as an essential element for higher plants. Comments Agric. Food Chem. 1990, 2, 99–122. [Google Scholar]
- Liang, Y.; Si, J.; Römheld, V. Si uptake and transport is an active process in Cucumis sativus. New Phytol. 2005, 167, 797–804. [Google Scholar] [CrossRef] [PubMed]
- Marron, A.O.; Sarah, R.; Wheeler, G.L.; Goldstein, R.E.; King, N.; Not, F.; Vargas, C.D.; Richter, D.J. The evolution of Si transport in eukaryotes. Mol. Biol. Evol. 2016, 33, 3226–3248. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, M.; Nikolic, N.; Liang, Y.C.; Kirkby, E.A.; Römheld, V. Germanium-68 as an adequate tracer for Si transport in plants. Characterization of Si uptake in different crop species. Plant Physiol. 2007, 143, 495–503. [Google Scholar] [CrossRef]
- Raven, J.A. Si Transport at the Cell and Tissue Level. In Si in Agriculture; Studies in Plant Science; Datnoff, L.E., Snyder, G.H., Korndörfer, G.H., Eds.; Elsevier: Amsterdam, The Netherlands, 2001; Volume 8, pp. 41–55. [Google Scholar]
- Reynolds, O.L.; Keeping, M.G.; Meyer, J.H. Si-augmented resistance of plants to herbivorous insects: A review. Ann. Appl. Biol. 2009, 155, 171–186. [Google Scholar] [CrossRef]
- Ma, J.F.; Yamaji, N. Si uptake and accumulation in higher plants. Trends Plant Sci. 2006, 11, 392–397. [Google Scholar] [CrossRef]
- Meunier, J.D.; Barboni, D.; Anwarul Haq, M.; Levard, C.; Chaurand, P.; Vidal, V.; Grauby, O.; Huc, R.; Laffont-Schwob, I.; Rabier, J.; et al. Effect of phytoliths for mitigating water stress in durum wheat. New Phytol. 2017, 215, 229–239. [Google Scholar] [CrossRef]
- Ma, J.F. Si Transporters in Higher Plants. In MIPs and Their Role in the Exchange of Metalloids. Advances in Experimental Medicine and Biology; Jahn, T.P., Bienert, G.P., Eds.; Springer: New York, NY, USA, 2010; Volume 679, pp. 99–109. [Google Scholar]
- Mitani, N.; Yamaji, N.; Ago, Y.; Iwasaki, K.; Ma, J.F. Isolation and functional characterization of an influx Si transporter in two pumpkin cultivars contrasting in Si accumulation. Plant J. 2011, 66, 231–240. [Google Scholar] [CrossRef] [PubMed]
- Montpetit, J.; Vivancos, J.; Mitani-Ueno, N.; Yamaji, N.; Rémus-Borel, W.; Belzile, F.; Ma, J.F.; Bélanger, R.R. Cloning, functional characterization and heterologous expression of TaLsi1, a wheat Si transporter gene. Plant Mol. Biol. 2012, 79, 35–46. [Google Scholar] [CrossRef]
- Ma, J.F.; Mitani, N.; Nagao, S.; Konishi, S.; Tamai, K.; Iwashita, T.; Yano, M. Characterization of Si uptake system and molecular mapping of Si transporter gene in rice. Plant Physiol. 2004, 136, 3284–3289. [Google Scholar] [CrossRef]
- Gomes, D.; Agasse, A.; Thiebaud, P.; Delrot, S.; Geros, H.; Chaumont, F. Aquaporins are multifunctional water and solute transporters highly divergent in living organisms. Biochim. Biophys. Acta Biomemb. 2009, 1788, 1213–1228. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Tamai, K.; Ichii, M.; Wu, G.F. A rice mutant defective in Si uptake. Plant Physiol. 2002, 130, 2111–2117. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Yamaji, N.; Mitani, N.; Tamai, K.; Konishi, S.; Fujiwara, T.; Katsuhara, M.; Yano, M. An efflux transporter of Si in rice. Nature 2007, 448, 209–212. [Google Scholar] [CrossRef]
- Yamaji, N.; Ma, J.F. Further characterization of a rice Si efflux transporter, Lsi2. Soil Sci. Plant Nutr. 2011, 57, 259–264. [Google Scholar] [CrossRef]
- Yamaji, N.; Mitani, N.; Ma, J.F. A transporter regulating Si distribution in rice shoots. Plant Cell 2008, 20, 1381–1389. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.F.; Yamaji, N.; Mitani-Ueno, N. Transport of Si from roots to panicles in plants. Proc. Jpn. Acad. Ser. B Phys. Biol. Sci. 2011, 87, 377. [Google Scholar] [CrossRef]
- Remus-Borel, W.; Menzies, J.; Belanger, R. Si induces antifungal compounds in powdery mildew-infected wheat. Physiol. Mol Plant Pathol. 2005, 66, 108–115. [Google Scholar] [CrossRef]
- Chiba, Y.; Mitani, N.; Yamaji, N.; Ma, J.F. HvLsi1 is a Si influx transporter in barley. Plant J. 2009, 57, 810–818. [Google Scholar] [CrossRef]
- Mitani, N.; Yamaji, N.; Ma, J.F. Identification of maize Si influx transporters. Plant Cell Physiol. 2009, 50, 5–12. [Google Scholar] [CrossRef] [PubMed]
- Mitani-Ueno, N.; Yamaji, N.; Ma, J.F. Si efflux transporters isolated from two pumpkin cultivars contrasting in Si uptake. Plant Signal. Behav. 2011, 6, 991–994. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Guo, J.; Duan, Y.; Zhang, T.; Huo, H.; Gong, H. Isolation and functional characterization of CsLsi1, a Si transporter gene in Cucumis sativus. Physiol. Plant 2017, 159, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Duan, Y.; Qi, X.; Zhang, L.; Huo, H.; Gong, H. Isolation and characterization of CsLsi2, a cucumber Si efflux transport gene. Ann. Bot. 2018, 122. [Google Scholar] [CrossRef] [PubMed]
- Deshmukh, R.K.; Vivabcos, J.; Guerin, V.; Sonah, H.; Labbe, C.; Belzile, F.; Belanger, R.R. Identification and functional characterization of Si transporters in soybean using comparative genomics of major intrinsic proteins in Arabidopsis and rice. Plant Mol. Biol. 2013, 83, 303–315. [Google Scholar] [CrossRef]
- Vulavala, V.K.R.; Elbaum, R.; Yermiyahu, U.; Fogelman, E.; Kumar, A.; Ginzberg, I. Si fertilization of potato: Expression of putative transporters and tuber skin quality. Planta 2016, 243, 217–229. [Google Scholar] [CrossRef] [PubMed]
- Gregoire, C.; Remus-Borel, W.; Vivancos, J.; Labbe, C.; Belzile, F.; Belanger, R.R. Discovery of a multigene family of aquaporin Si transporters in the primitive plants Equisetum arvense. Plant J. 2012, 72, 320–330. [Google Scholar] [CrossRef]
- Vivancos, J.; Deshmukh, R.; Gregoire, C.; Remus-Borel, W.; Belzile, F.; Belanger, R.R. Identification and characterization of Si efflux transporters in horsetail (Equisteum arvense). J. Plant Physiol. 2016, 200, 82–89. [Google Scholar] [CrossRef]
- Trembath-Reichert, E.; Wilson, J.P.; McGlynn, S.E.; Fischer, W.W. Four hundred million years of silica bio mineralization in land plants. Proc. Natl. Acad. Sci. USA 2015, 112, 5449–5454. [Google Scholar] [CrossRef]
| Group | Species | Si Accumulation |
|---|---|---|
| Angiosperms | Acoraceae Acorus calamus | - |
| Asteraceae Anaphalis margarita | - | |
| Helianthus annuus | + | |
| Helianthus maximilianii, | + | |
| Helianthus atrorubens | + | |
| Inula helenium, Inula viscosa | ± | |
| Lactuca serriola | ± | |
| Cyperaceae (Carex cinica) | + | |
| Poaceae Agrostis spp. | + | |
| Andropogon scoparius | + | |
| Arundinaria gigantean | + | |
| Bouteloua hirsuta | + | |
| Brachypodium sylvaticum | + | |
| Chasmanthium latifolium | + | |
| Ctenium aromaticum | + | |
| Echinochloa colona | ± | |
| Elymus molli | ± | |
| Oryza sativa | + | |
| Zea mays | ± | |
| Cucurbitaceae Benincasa hispida | ± | |
| Citrullus lanatus | ± | |
| Ecballium elaterium | ± | |
| Equisetophyta | Equisetaceae Equisetum arvense | + |
| Equisetum hyemale | + |
| Plant Species | Si Transporters |
|---|---|
| Angiosperms (monocots and a few eudicots) | + |
| Equisetopsida | + |
| Gymnosperms | - |
| Lycophytes | - |
| Bryophytes (Liverworts, Mosses, Hornworts) | - |
| Gnetophytes | - |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaur, H.; Greger, M. A Review on Si Uptake and Transport System. Plants 2019, 8, 81. https://doi.org/10.3390/plants8040081
Kaur H, Greger M. A Review on Si Uptake and Transport System. Plants. 2019; 8(4):81. https://doi.org/10.3390/plants8040081
Chicago/Turabian StyleKaur, Harmanjit, and Maria Greger. 2019. "A Review on Si Uptake and Transport System" Plants 8, no. 4: 81. https://doi.org/10.3390/plants8040081
APA StyleKaur, H., & Greger, M. (2019). A Review on Si Uptake and Transport System. Plants, 8(4), 81. https://doi.org/10.3390/plants8040081





