Arabidopsis Natural Accessions Display Adaptations in Inflorescence Growth and Vascular Anatomy to Withstand High Salinity during Reproductive Growth
Abstract
1. Introduction
2. Results
2.1. Experimental Scheme for Studying the Tolerance to High Salinity during the Reproductive Stage
2.2. Growth of Thellungiella salsuginea under High Salinity during the Reproductive Stage
2.3. Natural Variation in the Floral Stem Growth and in Sugar Content in Control Conditions
2.4. Diversity in the Arabidopsis Floral Stem Growth and Sugar Content in Response to High Salinity
2.5. Responses to High Salinity and Kinetics in the Stem Growth
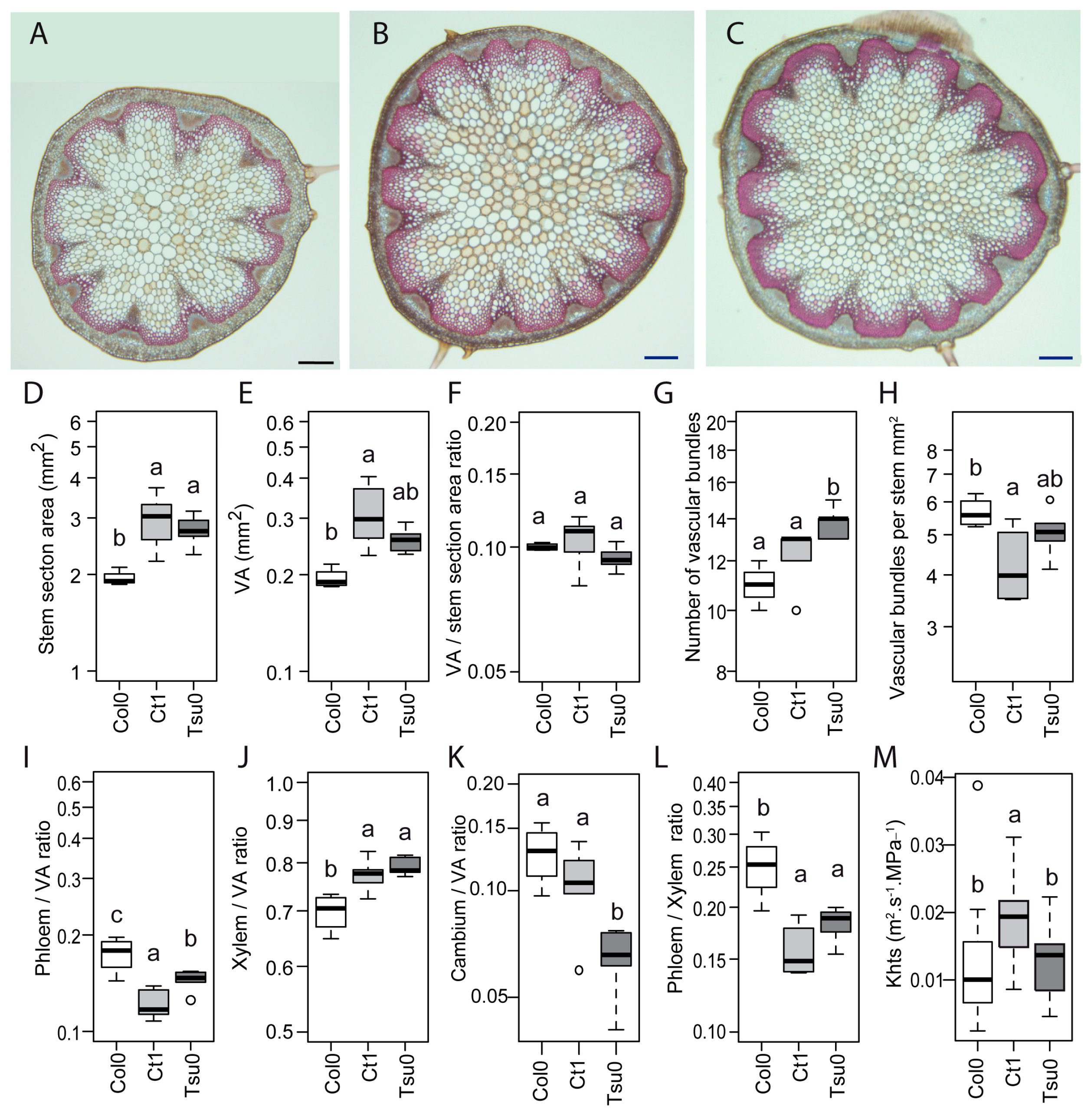
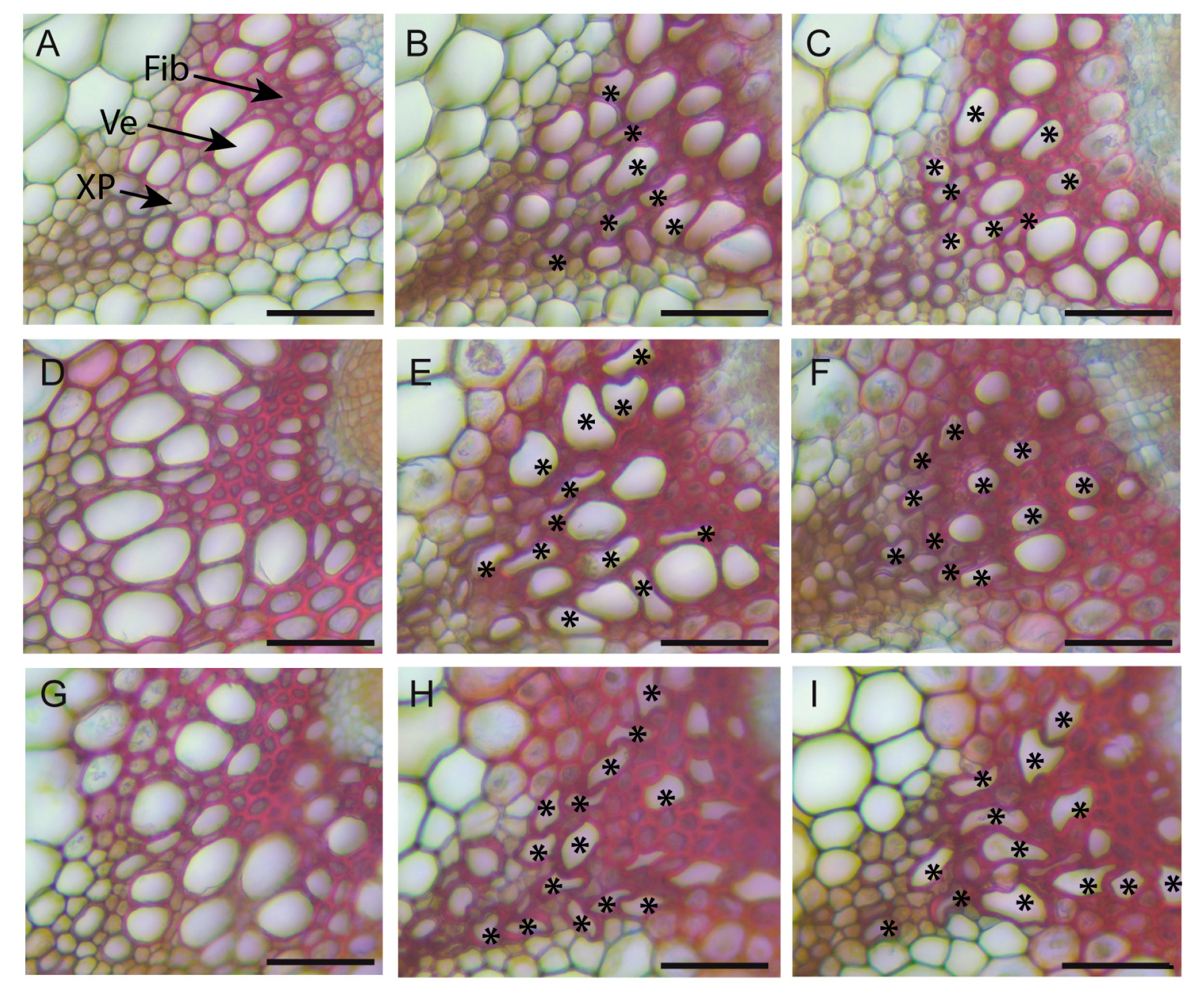
2.6. Natural Variation in the Anatomy of the Stem in Normal and High Salinity Conditions
2.7. Contrasted Responses of the Traits Related to Sugar Homeostasis and Anatomy in the Stem
3. Discussion
4. Materials and Methods
4.1. Plant Material and Growth Conditions
4.2. Salt Treatment and Pretreatment (Acclimation)
4.3. Growth and Physiological Parameters
4.4. Carbohydrate Content
4.5. Anatomy of Stem Sections
4.6. Stem Theoretical Hydraulic Specific Conductivity
4.7. Statistical Analyses
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Parihar, P.; Singh, S.; Singh, R. Effect of salinity stress on plants and its tolerance strategies: A review. Environ. Sci. Pollut. Res. 2015, 22, 4056–4075. [Google Scholar] [CrossRef] [PubMed]
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef]
- Huang, G.T.; Ma, S.L.; Bai, L.P.; Zhang, L.; Ma, H.; Jia, P.; Liu, J.; Zhong, M.; Guo, Z.F. Signal transduction during cold, salt, and drought stresses in plants. Mol. Biol. Rep. 2012, 39, 969–987. [Google Scholar] [CrossRef] [PubMed]
- Golldack, D.; Lüking, I.; Yang, O. Plant tolerance to drought and salinity: Stress regulating transcription factors and their functional significance in the cellular transcriptional network. Plant Cell Rep. 2011, 30, 1383–1391. [Google Scholar] [CrossRef] [PubMed]
- Golldack, D.; Li, C.; Mohan, H.; Probst, N. Tolerance to drought and salt stress in plants: Unraveling the signaling networks. Front. Plant Sci. 2014, 5, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Singh, M.; Kumar, J.; Singh, S.; Singh, V.; Prasad, S. Roles of osmoprotectants in improving salinity and drought tolerance in plants: A review. Rev. Environ. Sci. Bio/Technology 2015, 14, 407–426. [Google Scholar] [CrossRef]
- Deinlein, U.; Stephan, A.B.; Horie, T.; Luo, W.; Xu, G.; Schroeder, J.I. Plant salt-tolerance mechanisms. Trends Plant Sci. 2014, 19, 371–379. [Google Scholar] [CrossRef] [PubMed]
- Plett, D.C.; Møller, I.S. Na + transport in glycophytic plants: What we know and would like to know. Plant. Cell Environ. 2010, 33, 612–626. [Google Scholar] [CrossRef] [PubMed]
- Roy, S.J.; Negrão, S.; Tester, M. Salt resistant crop plants. Curr. Opin. Biotechnol. 2014, 26, 115–124. [Google Scholar] [CrossRef] [PubMed]
- Almeida, D.M.; Margarida Oliveira, M.; Saibo, N.J.M. Regulation of Na+and K+homeostasis in plants: Towards improved salt stress tolerance in crop plants. Genet. Mol. Biol. 2017, 40, 326–345. [Google Scholar] [CrossRef] [PubMed]
- Lemoine, R.; La Camera, S.; Atanassova, R.; Dédaldéchamp, F.; Allario, T.; Pourtau, N.; Bonnemain, J.-L.; Laloi, M.; Coutos-Thévenot, P.; Maurousset, L.; et al. Source-to-sink transport of sugar and regulation by environmental factors. Front. Plant Sci. 2013, 4, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Krasensky, J.; Jonak, C. Drought, salt, and temperature stress-induced metabolic rearrangements and regulatory networks. J. Exp. Bot. 2012, 63, 1593–1608. [Google Scholar] [CrossRef] [PubMed]
- Koch, K.E. Sucrose metabolism: Regulatory mechanisms and pivotal roles in suger sensing and plant development. Curr. Opin. Plant Biol. 2004, 7, 235–246. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yang, L.; Zheng, Z.; Grumet, R.; Loescher, W.; Zhu, J.-K.; Yang, P.; Hu, Y.; Chan, Z. Transcriptomic and Physiological Variations of Three Arabidopsis Ecotypes in Response to Salt Stress. PLoS ONE 2013, 8, e69036. [Google Scholar] [CrossRef]
- Sutka, M.; Li, G.; Boudet, J.; Boursiac, Y.; Doumas, P.; Maurel, C. Natural variation of root hydraulics in Arabidopsis grown in normal and salt-stressed conditions. Plant Physiol. 2011, 155, 1264–1276. [Google Scholar] [CrossRef] [PubMed]
- Julkowska, M.M.; Klei, K.; Fokkens, L.; Haring, M.A.; Schranz, M.E.; Testerink, C. Natural variation in rosette size under salt stress conditions corresponds to developmental differences between Arabidopsis accessions and allelic variation in the LRR-KISS gene. J. Exp. Bot. 2016, 67, 2127–2138. [Google Scholar] [CrossRef] [PubMed]
- Quesada, V.; García-Martínez, S.; Piqueras, P.; Ponce, M.R.; Micol, J.L. Genetic architecture of NaCl tolerance in Arabidopsis. Plant Physiol. 2002, 130, 951–963. [Google Scholar] [CrossRef] [PubMed]
- Katori, T.; Ikeda, A.; Iuchi, S.; Kobayashi, M.; Shinozaki, K.; Maehashi, K.; Sakata, Y.; Tanaka, S.; Taji, T. Dissecting the genetic control of natural variation in salt tolerance of Arabidopsis thaliana accessions. J. Exp. Bot. 2010, 61, 1125–1138. [Google Scholar] [CrossRef] [PubMed]
- Lugan, R.; Niogret, M.F.; Leport, L.; Guégan, J.P.; Larher, F.R.; Savouré, A.; Kopka, J.; Bouchereau, A. Metabolome and water homeostasis analysis of Thellungiella salsuginea suggests that dehydration tolerance is a key response to osmotic stress in this halophyte. Plant J. 2010, 64, 215–229. [Google Scholar] [CrossRef] [PubMed]
- Bartels, D.; Dinakar, C. Balancing salinity stress responses in halophytes and non-halophytes: A comparison between Thellungiella and Arabidopsis thaliana. Funct. Plant Biol. 2013, 40, 819–831. [Google Scholar] [CrossRef]
- Savi, T.; Casolo, V.; Luglio, J.; Bertuzzia, S.; Trifilo’, P.; Lo Gullo, M.A.; Nardinia, A. Species-specific reversal of stem xylem embolism after a prolonged drought correlates to endpoint concentration of soluble sugars. Plant Physiol. Biochem. J. 2016, 106, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Trifilò, P.; Casolo, V.; Raimondo, F.; Petrussa, E.; Boscutti, F.; Lo Gullo, M.A.; Nardini, A. Effects of prolonged drought on stem non-structural carbohydrates content and post-drought hydraulic recovery in Laurus nobilis L.: The possible link between carbon starvation and hydraulic failure. Plant Physiol. Biochem. 2017, 120, 232–241. [Google Scholar] [CrossRef] [PubMed]
- Antoun, M.; Ouellet, F. Growth temperature affects inflorescence architecture in Arabidopsis. Botany 2013, 91, 642–651. [Google Scholar] [CrossRef]
- Shafi, A.; Chauhan, R.; Gill, T.; Swarnkar, M.; Sreenivasulu, Y.; Kumar, S.; Kumar, N.; Shankar, R.; Ahuja, P.; Singh, A. Expression of SOD and APX genes positively regulates secondary cell wall biosynthesis and promotes plant growth and yield in Arabidopsis under salt stress. Plant Mol. Biol. 2015, 87, 615–631. [Google Scholar] [CrossRef] [PubMed]
- Sanchez, P.; Nehlin, L.; Greb, T. From thin to thick: Major transitions during stem development. Trends Plant Sci. 2012, 17, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Altamura, M.M.; Possenti, M.; Matteucci, A.; Baima, S.; Ruberti, I.; Morelli, G. Development of the vascular system in the inflorescence stem of Arabidopsis. New Phytol. 2001, 151, 381–389. [Google Scholar] [CrossRef]
- Earley, E.J.; Ingland, B.; Winkler, J.; Tonsor, S.J. Inflorescences contribute more than rosettes to lifetime carbon gain in arabidopsis thaliana (Brassicaceae). Am. J. Bot. 2009, 96, 786–792. [Google Scholar] [CrossRef] [PubMed]
- Leonardos, E.D.; Rauf, S.A.; Weraduwage, S.M.; Marillia, E.F.; Taylor, D.C.; Micallef, B.J.; Grodzinski, B. Photosynthetic capacity of the inflorescence is a major contributor to daily-C-gain and the responsiveness of growth to elevated CO2in Arabidopsis thaliana with repressed expression of mitochondrial-pyruvate-dehydrogenase-kinase. Environ. Exp. Bot. 2014, 107, 84–97. [Google Scholar] [CrossRef]
- Gnan, S.; Marsh, T.; Kover, P.X. Inflorescence photosynthetic contribution to fitness releases Arabidopsis thaliana plants from trade-off constraints on early flowering. PLoS ONE 2017, 12, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Su, Z.; Ma, X.; Guo, H.; Sukiran, N.L.; Guo, B.; Assmann, S.M.; Ma, H. Flower development under drought stress: Morphological and transcriptomic analyses reveal acute responses and long-term acclimation in Arabidopsis. Plant Cell 2013, 25, 3785–3807. [Google Scholar] [CrossRef] [PubMed]
- M’rah, S.; Ouerghi, Z.; Eymery, F.; Rey, P.; Hajji, M.; Grignon, C.; Lachaâl, M. Efficiency of biochemical protection against toxic effects of accumulated salt differentiates Thellungiella halophila from Arabidopsis thaliana. J. Plant Physiol. 2007, 164, 375–384. [Google Scholar] [CrossRef] [PubMed]
- Rajendran, K.; Tester, M.; Roy, S.J. Quantifying the three main components of salinity tolerance in cereals. Plant Cell Environ. 2009, 32, 237–249. [Google Scholar] [CrossRef] [PubMed]
- Sulpice, R.; Nikoloski, Z.; Tschoep, H.; Antonio, C.; Kleessen, S.; Larhlimi, A.; Selbig, J.; Ishihara, H.; Gibon, Y.; Fernie, A.R.; et al. Impact of the carbon and nitrogen supply on relationships and connectivity between metabolism and biomass in a broad panel of Arabidopsis accessions. Plant Physiol. 2013, 162, 347–363. [Google Scholar] [CrossRef] [PubMed]
- Sulpice, R.; Trenkamp, S.; Steinfath, M.; Usadel, B.; Gibon, Y.; Witucka-Wall, H.; Pyl, E.; Tschoep, H.; Steinhauser, C.; Guenther, M.; et al. Network analysis of enzyme activities and metabolite levels and their relationship to biomass in a large panel of Arabidopsis accessions. Plant Cell 2010, 22, 2872–2893. [Google Scholar] [CrossRef] [PubMed]
- Wobus, U.; Weber, H. Sugars as signal molecules in plant seed development. Biol. Chem 1999, 380, 937–944. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.; Mohnen, D. A review of xylan and lignin biosynthesis: Foundation for studying Arabidopsis irregular xylem mutants with pleiotropic phenotypes. Crit. Rev. Biochem. Mol. Biol. 2014, 49, 212–241. [Google Scholar] [CrossRef] [PubMed]
- Grime, J.P. Evidence for the existence of three primary strategies in plants and its relevance to ecological and evolutionary theory. Am. Nat. 1977, 111, 1169–1194. [Google Scholar] [CrossRef]
- Sibout, R.; Plantegenet, S.; Hardtke, C.S. Flowering as a condition for xylem expansion in Arabidopsis hypocotyl and root. Curr. Biol. 2008, 18, 458–463. [Google Scholar] [CrossRef] [PubMed]
- Meng, D.; Fricke, W. Changes in root hydraulic conductivity facilitate the overall hydraulic response of rice (Oryza sativa L.) cultivars to salt and osmotic stress. Plant Physiol. Biochem. 2017, 113, 64–77. [Google Scholar] [CrossRef] [PubMed]
- Qian, Z.J.; Song, J.J.; Chaumont, F.; Ye, Q. Differential responses of plasma membrane aquaporins in mediating water transport of cucumber seedlings under osmotic and salt stresses. Plant Cell Environ. 2015, 38, 461–473. [Google Scholar] [CrossRef] [PubMed]
- Junghans, U.; Polle, A.; Düchting, P.; Weiler, E.; Kuhlman, B.; Gruber, F.; Teichmann, T. Adaptation to high salinity in poplar involves changes in xylem anatomy and auxin physiology. Plant. Cell Environ. 2006, 29, 1519–1531. [Google Scholar] [CrossRef] [PubMed]
- Escalante-Pérez, M.; Lautner, S.; Nehls, U.; Selle, A.; Teuber, M.; Schnitzler, J.P.; Teichmann, T.; Fayyaz, P.; Hartung, W.; Polle, A.; et al. Salt stress affects xylem differentiation of grey poplar (Populus × canescens). Planta 2009, 229, 299–309. [Google Scholar] [CrossRef] [PubMed]
- Zubrinich, T.M.; Loveys, B.; Gallasch, S.; Seekamp, J.V.; Tyerman, S.D. Tolerance of salinized floodplain condition in a naturally occurring Eucalyptus hybrid related to lowered plant water potential. Tree Physiol. 2000, 20, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Lemaître, T.; Gaufichon, L.; Boutet-Mercey, S.; Christ, A.; Masclaux-Daubresse, C. Enzymatic and metabolic diagnostic of nitrogen deficiency in Arabidopsis thaliana Wassileskija accession. Plant Cell Physiol. 2008, 49, 1056–1065. [Google Scholar] [CrossRef] [PubMed]
- Le Hir, R.; Spinner, L.; Klemens, P.A.W.; Chakraborti, D.; de Marco, F.; Vilaine, F.; Wolff, N.; Lemoine, R.; Porcheron, B.; Géry, C.; et al. Disruption of the sugar transporters AtSWEET11 and AtSWEET12 affects vascular development and freezing tolerance in Arabidopsis. Mol. Plant 2015, 8, 1687–1690. [Google Scholar] [CrossRef] [PubMed]
- Tolivia, D.; Tolivia, J. Fasga: A new polychromatic method for simultaneous and differential staining of plant tissues. J. Microsc. 1987, 148, 113–117. [Google Scholar] [CrossRef]
- Tixier, A.; Cochard, H.; Badel, E.; Dusotoit-Coucaud, A.; Jansen, S.; Herbette, S. Arabidopsis thaliana as a model species for xylem hydraulics: Does size matter? J. Exp. Bot. 2013, 64, 2295–2305. [Google Scholar] [CrossRef] [PubMed]
- Christman, M.A.; Sperry, J.S. Single-vessel flow measurements indicate scalariform perforation plates confer higher flow resistance than previously estimated. Plant Cell Environ. 2010, 33, 431–443. [Google Scholar] [CrossRef] [PubMed]








© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sellami, S.; Le Hir, R.; Thorpe, M.R.; Aubry, E.; Wolff, N.; Vilaine, F.; Brini, F.; Dinant, S. Arabidopsis Natural Accessions Display Adaptations in Inflorescence Growth and Vascular Anatomy to Withstand High Salinity during Reproductive Growth. Plants 2019, 8, 61. https://doi.org/10.3390/plants8030061
Sellami S, Le Hir R, Thorpe MR, Aubry E, Wolff N, Vilaine F, Brini F, Dinant S. Arabidopsis Natural Accessions Display Adaptations in Inflorescence Growth and Vascular Anatomy to Withstand High Salinity during Reproductive Growth. Plants. 2019; 8(3):61. https://doi.org/10.3390/plants8030061
Chicago/Turabian StyleSellami, Sahar, Rozenn Le Hir, Michael R. Thorpe, Emilie Aubry, Nelly Wolff, Françoise Vilaine, Faiçal Brini, and Sylvie Dinant. 2019. "Arabidopsis Natural Accessions Display Adaptations in Inflorescence Growth and Vascular Anatomy to Withstand High Salinity during Reproductive Growth" Plants 8, no. 3: 61. https://doi.org/10.3390/plants8030061
APA StyleSellami, S., Le Hir, R., Thorpe, M. R., Aubry, E., Wolff, N., Vilaine, F., Brini, F., & Dinant, S. (2019). Arabidopsis Natural Accessions Display Adaptations in Inflorescence Growth and Vascular Anatomy to Withstand High Salinity during Reproductive Growth. Plants, 8(3), 61. https://doi.org/10.3390/plants8030061





