First Broad Screening of Allelopathic Potential of Wild and Cultivated Plants in Turkey
Abstract
1. Introduction
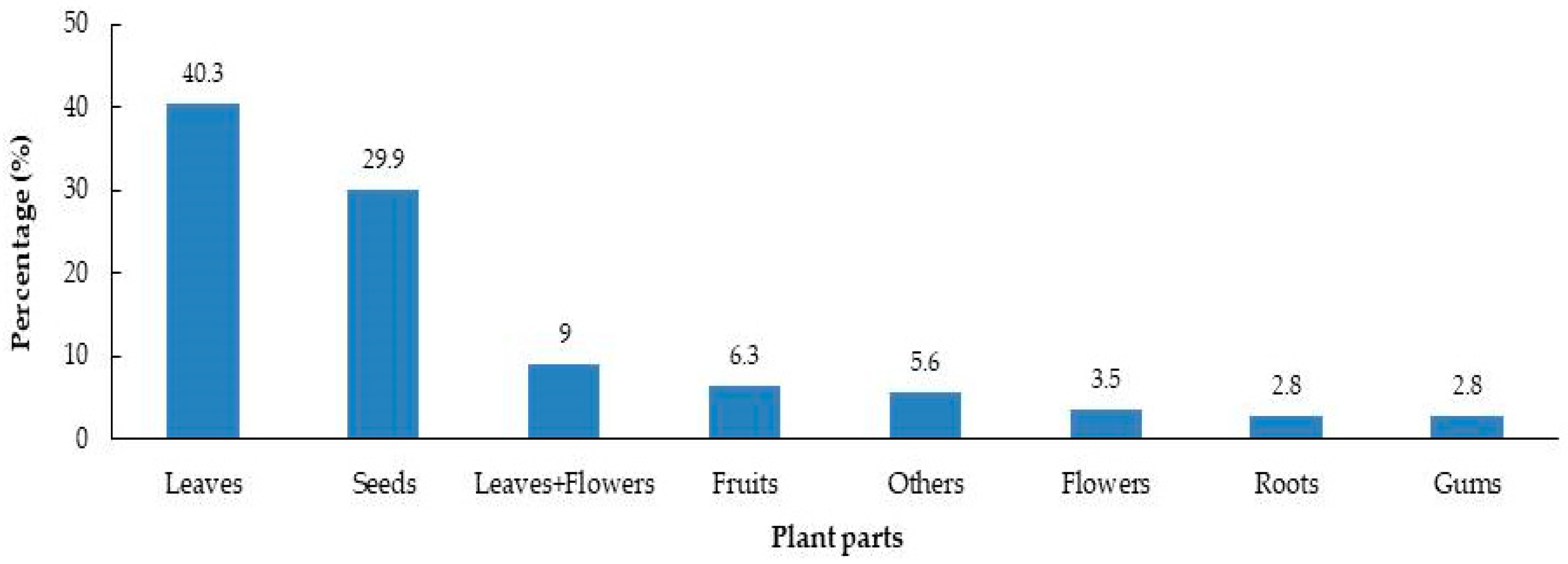
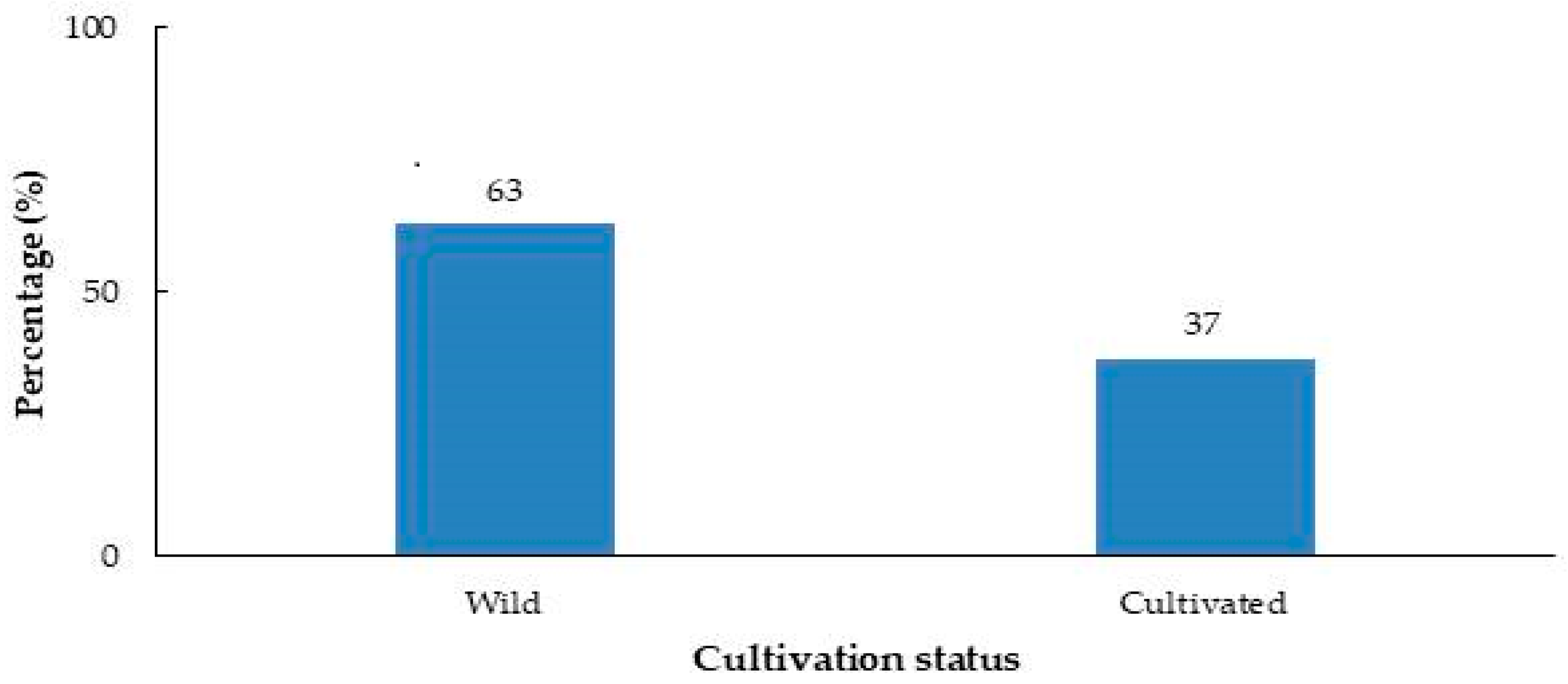
2. Results
3. Discussion
4. Materials and Methods
4.1. Material (Plant Samples)
4.2. Method (Sandwich Method)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gawronska, H.; Ciarka, D.; Bernat, W.; Gawronski, S.W. Sunflower-desired Allelopathic Crop for Sustainable and Organic Agriculture. In Allelopathy New Concept and Methodology; Fujii, Y., Hiradate, S., Eds.; Science Publishers: Enfield, CT, USA, 2007; pp. 185–210. [Google Scholar]
- Fujii, Y.; Parvez, S.S.; Parvez, M.M.; Ohmae, Y.; Iida, O. Screening of 239 medicinal plant species for allelopathic activity using the sandwich method. Weed Biol. Manag. 2003, 3, 233–241. [Google Scholar] [CrossRef]
- Iqbal, Z.; Nasir, H.; Fujii, Y. Allelopathic Activity of Buckwheat: A ground Cover Crop for Weed Control. In Allelopathy New Concept and Methodology; Fujii, Y., Hiradate, S., Eds.; Science Publishers: Enfield, CT, USA, 2007; pp. 173–183. [Google Scholar]
- Golisz, A.; Lata, B.; Gawronski, S.W.; Fujii, Y. Specific and total activities of the allelochemicals identified in buckwheat. Weed Biol. Manag. 2007, 7, 164–171. [Google Scholar] [CrossRef]
- Haig, T. Allelochemicals in Plants. In Allelopathy in Sustainable Agriculture and Forestry; Zeng, R.S., Mallik, A.U., Luo, S.M., Eds.; Springer: New York, NY, USA, 2008; pp. 63–104. [Google Scholar]
- Jabran, K.; Farooq, M.; Hussain, M.; Ali, M.A. Wild Oat (Avena fatua L.) and Canary Grass (Phalaris minor Ritz.) Management through Allelopathy. J. Plant Prot. Res. 2010, 50, 41–44. [Google Scholar] [CrossRef]
- Wu, H.; Pratley, J.; Lemerle, D.; Haig, T.; An, M. Screening method for the evaluation of crop allelopathic potential. Bot. Rev. 2001, 67, 403–415. [Google Scholar] [CrossRef]
- Oerke, E.C. Crop Losses to Pests. J. Agric. Sci. 2006, 144, 31–43. [Google Scholar] [CrossRef]
- Weed Science Society of America (WSSA) Calculates Billions in Potential Economic Losses from Uncontrolled Weeds. Available online: http://wssa.net/2016/05/wssa-calculates-billions-in-potential-economic-losses-from-uncontrolled-weeds/ (accessed on 27 July 2019).
- Seal, A.N.; Pratley, J.E.; Haig, A. Can results from a laboratory bioassay be used as an indicator of field performance of rice cultivars with allelopathic potential against Damasonium minus (starfruit)? Aust. J. Agric. Res. 2008, 59, 183–188. [Google Scholar] [CrossRef]
- Tesio, F.; Weston, L.A.; Ferrero, A. Allelochemicals identified from Jerusalem artichoke (Helianthus tuberosus L.) residues and their potential inhibitory activity in the field and laboratory. Sci. Hortic. 2011, 129, 361–368. [Google Scholar] [CrossRef]
- Morikawa, C.I.O.; Miyaura, R.; Figueroa, M.L.T.; Salgado, E.L.R.; Fujii, Y. Screening of 170 Peruvian plant species for allelopathic activity by using the Sandwich Method. Weed Biol. Manag. 2012, 12, 1–11. [Google Scholar] [CrossRef]
- Appiah, K.S.; Li, Z.; Zeng, R.S.; Luo, S.; Oikawa, Y.; Fujii, Y. Determination of allelopathic potentials in plant species in Sino-Japanese floristic region by sandwich method and dish pack method. Int. J. Basic Appl. Sci. 2015, 4, 381–394. [Google Scholar] [CrossRef]
- Raihan, I.; Miyaura, R.; Baki, B.B.; Fujii, Y. Assessment of allelopathic potential of goniothalamin allelochemical from Malaysian plant Goniothalamus andersonii J. Sinclair by sandwich method. Allelopath. J. 2019, 46, 25–40. [Google Scholar] [CrossRef]
- Shiraishi, S.; Watanabe, I.; Kuno, K.; Fujii, Y. Allelopathic activity of leaching from dry leaves and exudate from roots of ground cover plants assayed on agar. Weed Biol. Manag. 2002, 2, 133–142. [Google Scholar] [CrossRef]
- Osivand, A.; Araya, H.; Appiah, S.K.; Mardani, H.; Ishizaki, T.; Fujii, Y. Allelopathy of Wild Mushrooms-An Important Factor for Assessing Forest Ecosystem in Japan. Forest 2018, 9, 773. [Google Scholar] [CrossRef]
- Begum, K.; Shammi, M.; Hasan, N.; Asaduzzaman, M.; Appiah, K.S.; Fujii, Y. Potential Allelopathic Candidates for Land Use and Possible Sustainable Weed Management in Asian Ecosystem. Sustainability 2019, 11, 2649. [Google Scholar] [CrossRef]
- Tan, A. Turkey: Country Report to the FAO international Technical Conference on Plant Genetic Resource, Leipzig, Germany 17–23 June 1996. Available online: http://www.fao.org/fileadmin/templates/agphome/documents/PGR/SoW1/east/TURKEY.pdf (accessed on 27 July 2019).
- Tan, A. Second National Report of TURKEY on Conservation and Sustainable Utilisation of Plant Genetic Resources for Food and Agriculture. 2009. Available online: http://www.fao.org/docrep/013/i1500e/Turkey.pdf (accessed on 27 July 2019).
- Piyatida, P.; Kato-Noguchi, H. Screening of Allelopathic Activity of Eleven Thai Medicinal Plants on Seedling Growth of Five Test Plant Species. Asian J. Plant Sci. 2010, 9, 486–491. [Google Scholar] [CrossRef][Green Version]
- Da-Costa-Rocha, I.; Bonnlaender, B.; Sievers, H.; Pischel, I.; Heinrich, M. Hibiscus sabdariffa L.—A phytochemical and pharmacological review. Food Chem. 2014, 165, 423–443. [Google Scholar]
- Carvajal-Zarrabal, O.; Barradas-Dermitz, D.M.; Orta-Flores, Z.; Hayward-Jones, P.M.; Nolasco-Hipólito, C.N.; Aguilar-Uscanga, M.G.; Miranda-Medina, A.; Bujang, K.B. Hibiscus sabdariffa L., roselle calyx, from ethnobotany to pharmacology. J. Exp. Pharmacol. 2012, 4, 25–39. [Google Scholar] [CrossRef]
- Hida, H.; Yamada, T.; Yamada, Y. Absolute Configuration of Hydroxycitric Acid Produced by Microorganisms. Biosci. Biotechnol. Biochem. 2006, 70, 1972–1974. [Google Scholar] [CrossRef][Green Version]
- Yamada, T.; Hida, H.; Yamada, Y. Chemistry, physiological properties, and microbial production of hydroxycitric acid. Appl. Microbiol. Biotechnol. 2007, 75, 977–982. [Google Scholar] [CrossRef]
- Zheoat, A.M.; Gray, A.I.; Igoli, J.O.; Kennedy, A.R.; Ferro, V.A. Crystal structures of hibiscus acid and hibiscus acid dimethyl ester isolated from Hibiscus sabdariffa (Malvaceae). Acta Cryst. 2017, 73, 1368–1371. [Google Scholar]
- U.S Food and Drug Administration. Food Additives Permitted for Direct Addition to Food for Human Consumption, Code of Federal Regulations Title 21, 172.510. Available online: https://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfCFR/CFRSearch.cfm?fr=172.510 (accessed on 27 July 2019).
- U.S Food and Drug Administration. Food Additive Status List. Available online: https://www.fda.gov/food/food-additives-petitions/food-additive-status-list#ftnR (accessed on 27 July 2019).
- Food and Agriculture Organization of the United Nations. HIBISCUS: Post-Production Management for Improved Market Access. Available online: http://www.fao.org/fileadmin/user_upload/inpho/docs/Post_Harvest_Compendium-Hibiscus.pdf (accessed on 27 July 2019).
- Pukclai, P.; Kato-Noguchi, H. Evaluation of allelopathic activity of Hibiscus sabdariffa L. Adv. Biol. Res. 2011, 5, 366–372. [Google Scholar]
- Suwitchayanon, P.; Pukclai, P.; Ohno, O.; Suenaga, K.; Kato-Noguchi, H. Isolation and identification of an allelopathic substance from Hibiscus sabdariffa. Nat. Prod. Commun. 2015, 10, 765–766. [Google Scholar] [CrossRef] [PubMed]
- Piyatida, P.; Kimira, P.; Sato, M.; Kato-Noguchi, H. Isolation of β-sitosterol from Hibiscus sabdariffa L. Allelopath. J. 2013, 32, 289–300. [Google Scholar]
- Bolarinwa, I.F.; Orfila, C.; Morgan, M.R.A. Amygdalin content of seeds, kernels and food products commercially-available in the UK. Food Chem. 2014, 152, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Astaraei, A.R.; Sampietro, D.A. Allelopathic Effect of Almond on Cress and Fenugreek. Allelopath. J. 2008, 22, 239–243. [Google Scholar]
- Hassanshahi, M.; Hassani, M.R.; Sheibani, Z. Insecticidal effect of two plant extract seeds, on Agonoscena pistaciae (Hemiptera: Aphlaridae) under laboratory conditions. J. Entomol. Zool. 2016, 4, 445–448. [Google Scholar]
- Sezik, E.; Basaran, A. Turkish Mahalep and its oil. J. Fac. Pharm. Gazi Univ. 1985, 21, 121–127. [Google Scholar]
- Al-Said, M.S.; Hifnawy, M.S. Dihydrocoumarin and Certain Other Coumarins from Prunus mahaleb Seeds. J. Nat. Prod. 1986, 49, 721. [Google Scholar] [CrossRef]
- Ieri, F.; Pinelli, P.; Romani, A. Simultaneous determination of anthocyanins, coumarins and phenolic acids in fruits, kernels and liqueur of Prunus mahaleb L. Food Chem. 2012, 135, 2157–2162. [Google Scholar] [CrossRef]
- Ozcelik, B.; Kaya, D.A.; Sekeroglu, N. Evaluation of the in vitro Bioactivities of Mahaleb Cherry (Prunus mahaleb L.). Rom. Biotechnol. Lett. 2012, 17, 7863–7872. [Google Scholar]
- Haig, T.J.; Seal, A.N.; Pratley, J.E.; An, M.; Wu, H. Lavender as a source of novel plant compounds for the development of a natural herbicide. J. Chem. Ecol. 2009, 35, 1129–1136. [Google Scholar] [CrossRef]
- Nazemi, A.H.; Asad, G.A.; Ghorbani, R. Allelopathic Potential of Lavender′s Extract and Coumarin Applied as Pre-Plant Incorporated into Soil Under Agronomic Conditions. Planta Daninha 2018, 36, 1–5. [Google Scholar] [CrossRef]
- Herraiz, T.; Gonzalez, D.; Ancin-Azpilicueta, C.; Aran, V.J.; Guillen, H. Beta-carboline alkaloids in Peganum harmala and inhibition of human monoamine oxidase (MAO). Food Chem. Toxicol. 2010, 48, 839–845. [Google Scholar] [CrossRef] [PubMed]
- Sodaeizadeh, H.; Rafieiolhossaini, M.; Havlík, J.; Van Damme, P. Allelopathic activity of different plant parts of Peganum harmala L. and identification of their growth inhibitors substances. Plant Growth Regul. 2009, 5, 227. [Google Scholar] [CrossRef]
- Sodaeizadeh, H.; Rafieiolhossaini, M.; Van Damme, P. Herbicidal activity of a medicinal plant, Peganum harmala L., and decomposition dynamics of its phytotoxins in the soil. Ind. Crops Prod. 2010, 31, 385–394. [Google Scholar]
- Shao, H.; Huang, X.; Zhang, Y.; Zhang, C. Main Alkaloids of Peganum harmala L. and Their Different Effects on Dicot and Monocot Crops. Molecules 2013, 18, 2623–2634. [Google Scholar] [CrossRef] [PubMed]
- Biodiversity of Turkey: Contribution of Genetic Resources to Sustainable Agriculture and Food Systems. Available online: http://www.fao.org/3/ca1517en/CA1517EN.pdf (accessed on 27 July 2019).
- Ozcan, M.; Haciseferogullari, H.A. Condiment [Sumac (Rhus coriaria) Fruits]: Some Physio-chemical Properties. Bulg. J. Plantphysiol. 2004, 30, 74–84. [Google Scholar]
- Kosar, A.; Bozan, B.; Temelli, F.; Baser, K.H.C. Antioxidant activity and phenolic composition of sumac (Rhus coriaria L.) extracts. Food Chem. 2007, 103, 952–959. [Google Scholar] [CrossRef]
- Kossah, R.; Nsabimana, C.; Zhao, J.; Chen, H.; Tian, F.; Zhang, H.; Chen, W. Comparative Study on the Chemical Composition of Syrian Sumac (Rhus coriaria L.) and Chinese Sumac (Rhus typhina L.) Fruits. Pak. J. Nutr. 2009, 8, 1570–1574. [Google Scholar] [CrossRef]
- Romeo, F.V.; Ballistreri, G.; Fabroni, S.; Pangallo, S.; Nicosia, M.G.L.D.; Schena, L.; Rapisarda, P. Chemical Characterization of Different Sumac and Pomegranate Extracts Effective against Botrytis cinerea Rots. Molecules 2015, 20, 11941–11958. [Google Scholar] [CrossRef]
- Qasem, J.R. Allelopathic effects of selected medicinal plants on Amaranthus retroflexus and Chenopodium murale. Allelopath. J. 2002, 10, 105–122. [Google Scholar]
- Labruzzo, A.; Carrubba, A.; Di Marco, G.; Ebadi, M.T. Herbicidal potential of aqueous extracts from Melia azedarach L., Artemisia arborescens L., Rhus coriaria L. and Lantana camara L. Allelopath. J. 2017, 41, 81–89. [Google Scholar] [CrossRef]
- Schütz, K.; Carle, R.; Schieber, A. Taraxacum—A review on its phytochemical and pharmacological profile. J. Ethnopharmacol. 2006, 107, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Jankowska, J.; Ciepiela, G.A.; Sosnowski, J.; Kolczarek, R.; Jankowski, K. The Allelopathic Effect of Taraxacum officinale F.G. Wigg on the Seeds Germination and Initial Growth of Lolium westerwoldicum R.Br. Acta Agrobot. 2009, 62, 207–212. [Google Scholar] [CrossRef][Green Version]
- Jankowska, J.; Ciepiela, G.A.; Jankowski, K.; Kolczarek, R.; Sosnowski, J.; Wiśniewska-Kadżajan, B. The Allelopathic Influence of Taraxacum officinale on the initial Growth and Development of Festuca rubra L. J. Ecol. Eng. 2014, 15, 38–44. [Google Scholar]
- Riaz, M.; Khan, O.; Sherkheli, M.A. Chemical Constituents of Terminalia chebula. Nat. Prod. Ind. J. 2017, 13, 112. [Google Scholar]
- Upadhyay, A.; Agrahari, P.; Singh, D.K. A Review on the Pharmacological Aspects of Terminalia chebula. Int. J. Pharmacol. 2014, 10, 289–298. [Google Scholar]
- Manikadan, M.; Rejula, M. Identification of allelochemicals from Terminalia chebula. Afr. Res. Rev. 2008, 2, 306–314. [Google Scholar]
- Shinwari, M.I.; Iida, O.; Shinwari, M.I.; Fujii, Y. Evaluation of Phytodiversity for Allelopathic Activity and Application to Minimize Climate Change Impact: Japanese Medicinal Plants. Pak. J. Bot. 2017, 49, 139–144. [Google Scholar]
- Rao, O.P.; Saxena, A.K.; Singh, B.P. Allelopathic effects of certain agroforestry tree species on the germination of wheat, paddy and gram. Ann. For. 1994, 2, 60–64. [Google Scholar]
- Thapaliyal, S.; Bali, R.S.; Singh, B.; Todaria, N.P. Allelopathic Effects of Tree of Economic Importance on Germination and Growth of Food Crops. J. Herbs Spices Med. Plants 2008, 13, 11–23. [Google Scholar] [CrossRef]
- Fujii, Y. Allelopathy of Velvet bean: Determination and Identification of Allelopathic Substances. In Biologically Active Natural Products: Agrochemicals; Cutler, H.G., Cutler, S., Eds.; CRC Press: Boca Raton, FL, USA, 1999; pp. 33–47. [Google Scholar]
- Fujii, Y.; Shibuya, T.; Yasuda, T. L-3, 4-Dihydroxyphenylalanine as an allelochemical candidate from Mucuna pruriens (L.) DC.var.utilis. Agric. Biol. Chem. 1991, 55, 617–618. [Google Scholar]
- Fujii, Y. Screening and future exploitation of allelopathic plants as alternative herbicides with special reference to hairy vetch. J. Crop. Prod. 2001, 4, 257–275. [Google Scholar] [CrossRef]
- Horimoto, S.; Araki, H.; Itoh, K.; Fujii, Y. Weed suppression and rice yield in the hairy vetch introduced paddy field. Weed Sci. Technol. 2002, 47, 168–174. [Google Scholar] [CrossRef]
- Kamo, T.; Hiradate, S.; Fujii, Y. First isolation of natural cyanamide as a possible allelochemical from hairy vetch Vicia villosa. J. Chem. Ecol. 2003, 29, 275–283. [Google Scholar] [CrossRef]
- Iqbal, Z.; Nasir, Z.H.; Hiradate, S.; Fujii, Y. Plant growth inhibitory activity of Lycoris radiata Herb. and possible involvement of lycorine as an allelochemical. Weed Biol. Manag. 2006, 6, 221–227. [Google Scholar] [CrossRef]
- Fujii, Y.; Shibuya, T.; Yasuda, T. Survey of Japanese Weed and Crops, For the Detection of Water Extractable Allelopathic Chemicals Using Richards′ Function Fitted to Lettuce Germination Test. Weed Res. 1990, 35, 362–370. [Google Scholar]
- Appiah, K.S.; Mardani, H.K.; Osivand, A.; Kpabitey, S.; Amoatey, C.A.; Oikawa, Y.; Fujii, Y. Exploring Alternative Use of Medicinal Plants for Sustainable Weed Management. Sustainability 2017, 9, 1468. [Google Scholar] [CrossRef]
- Kato-Noguchi, H.; Hamada, N.; Morita, M.; Suenaga, K. A Novel Allelopathic Substance, 13-Epi-Orthosiphol N, in Orthosiphon stamineus. J. Plant Physiol. 2013, 170, 1–5. [Google Scholar] [CrossRef]
- Bich, T.T.N.; Kato-Noguchi, H. Isolation and Identification of Phytotoxic Substance from Emergent Macrophyt Centrostachys aquatica. Bot. Stud. 2014, 55, 1–5. [Google Scholar] [CrossRef]
- Kato-Noguchi, H.; Puklai, P.; Ohno, O.; Suenaga, K. Isolation and Identification of Plant Growth Inhibitor from Tinospora tuberculate Beumee. Acta Physiol. Plant 2014, 36, 1621–1626. [Google Scholar] [CrossRef]
- Fujii, Y.; Shibuya, T.; Nakata, K.; Itani, T.; Hiradate, S.; Parvez, M.M. Assessment Method for the Allelopathic Effect from Leaf Leachate. Weed Biol. Manag. 2004, 4, 19–23. [Google Scholar] [CrossRef]
- Fujii, Y. Screening of allelopathic candidates by new specific discrimination, assessment methods for allelopathy, and the inhibition of L-DOPA as the allelopathic substance from the most promising velvet bean (Mucuna pruriens). Bull. Nat. Inst. Agro-Environ. Sci. 1994, 10, 115–218. [Google Scholar]
- Fujii, Y.; Matsuyama, M.; Hiradate, S.; Shimozawa, H. Dish pack method: A new bioassay for volatile allelopathy. In Proceedings of the 4th World Congress on Allelopathy, Establishing the Scientific Base, Fourth World Congress on Allelopathy, Wagga Wagga, Australia, 21–26 August 2005; pp. 493–497. [Google Scholar]
- Fujii, Y.; Pariasca, D.; Shibuya, T.; Yasuda, T.; Kahn, B.; Waller, G.R. Plant-box Method: A Specific Bioassay to Evaluate Allelopathy through Root Exudates. In Allelopathy New Concept and Methodology; Fujii, Y., Hiradate, S., Eds.; Science Publishers: Enfield, CT, USA, 2007; pp. 39–56. [Google Scholar]
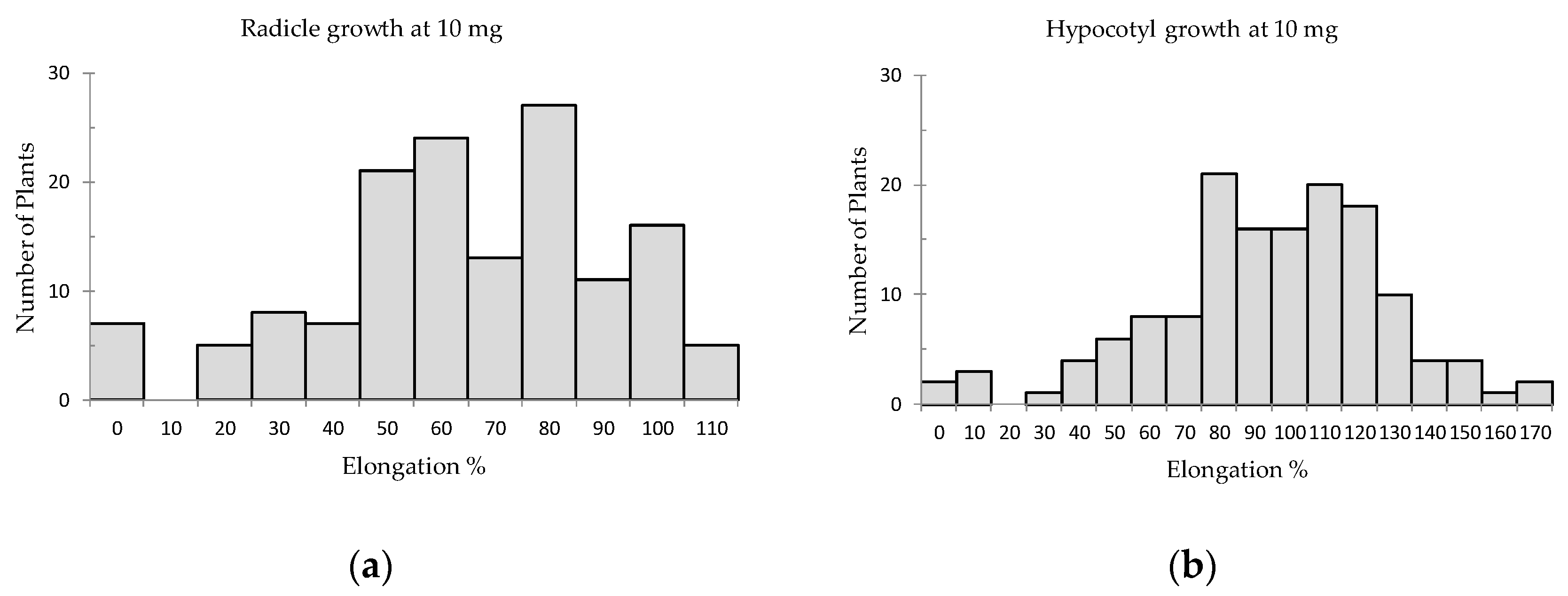
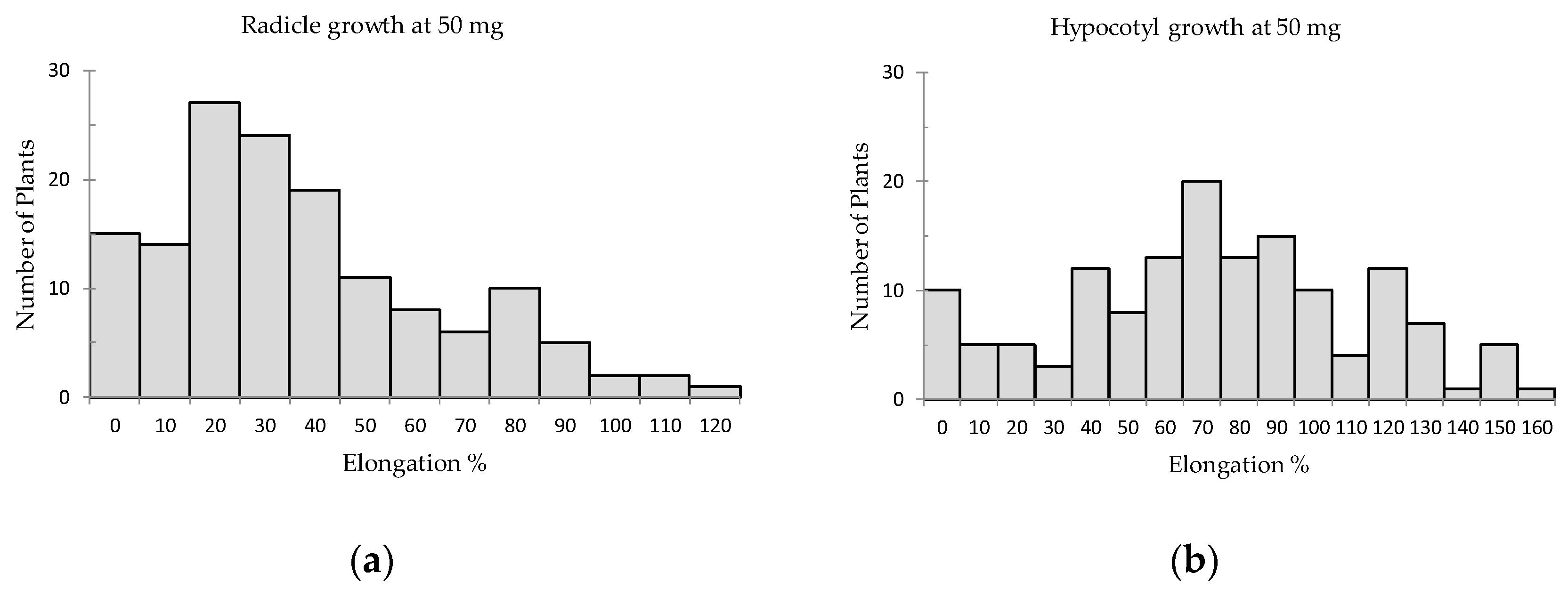
| Species [Used Part *] | Radicle Elongation (% of Control) | Hypocotyl Elongation (% of Control) | Total Score of * | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 10 mg | Crit. | 50 mg | Crit. | 10 mg | Crit. | 50 mg | Crit. | ||
| Hibiscus sabdariffa L. [C] | 3.2 | ***** | 5.1 | ** | 6.6 | ***** | 3.3 | *** | 15 |
| Prunus dulcis (Mill.) D.A. Webb [S] | 5.7 | **** | 0.0 | *** | 6.7 | ***** | 0.0 | *** | 15 |
| Taraxacum officinale (L.) Weber [S] | 7.1 | **** | 4.0 | ** | 45.5 | *** | 11.9 | *** | 12 |
| Terminalia chebula Retz. [S] | 7.1 | **** | 5.7 | ** | 41.0 | *** | 13.0 | *** | 12 |
| Rhus coriaria L. [Fr] | 7.4 | **** | 3.2 | ** | 33.1 | **** | 10.6 | *** | 13 |
| Prunus mahaleb L. [S] | 7.8 | **** | 3.1 | ** | 11.6 | ***** | 0.0 | *** | 14 |
| Peganum harmala L. [S] | 8.6 | **** | 4.2 | ** | 16.4 | ***** | 4.9 | *** | 14 |
| Punica granatum L. [Fl) | 24.5 | *** | 8.3 | ** | 56.4 | ** | 45.3 | * | 8 |
| Alcea rosea L. [L, Fl] | 27.3 | *** | 16.4 | * | 57.3 | ** | 43.6 | * | 7 |
| Lavandula stoechas L. [L, Fl] | 27.4 | *** | 1.9 | ** | 81.5 | * | 0.5 | *** | 9 |
| Silybum marianum (L.) Gaertn. [S] | 27.7 | *** | 29.9 | 60.8 | ** | 91.0 | 5 | ||
| Nigella sativa L. [S] | 27.8 | *** | 14.0 | ** | 15.1 | ***** | 11.7 | *** | 13 |
| Pinus brutia Tenore [L] | 31.4 | ** | 9.0 | ** | 92.7 | 50.0 | * | 5 | |
| Vitis vinifera L. [L] | 32.9 | ** | 11.7 | ** | 66.6 | ** | 44.5 | * | 7 |
| Liquidambar orientalis L. [G] | 36.0 | ** | 3.8 | ** | 40.7 | *** | 1.2 | *** | 10 |
| Camellia sinensis (L.) Kuntze [L] | 36.2 | ** | 13.5 | ** | 87.9 | 53.6 | * | 5 | |
| Malva sylvestris L. [L, Fl] | 37.0 | ** | 15.4 | * | 83.8 | 63.9 | 3 | ||
| Liquidambar orientalis L [B] | 37.5 | ** | 6.1 | ** | 46.8 | *** | 16.8 | *** | 10 |
| Citrus sinensis (L.) Osbeck [P] | 37.9 | ** | 28.8 | 56.3 | ** | 43.7 | * | 5 | |
| Tribulus terrestis L. [L] | 39.5 | ** | 18.6 | * | 104 | 91.9 | 3 | ||
| Capsella bursa-pastoris (L.) Medik. [L] | 40.7 | ** | 26.5 | * | 82.3 | * | 74.9 | 4 | |
| Papaver rhoeas L. [L] | 41.9 | ** | 28.8 | 128 | 102.3 | 2 | |||
| Raphanus sativus L. [S] | 42.1 | ** | 26.5 | * | 63.5 | ** | 84.1 | 5 | |
| Matricaria chamomilla L. [L, Fl] | 42.3 | ** | 23.0 | * | 109 | 93.6 | 3 | ||
| Capsicum annum L. [Fr] | 44.5 | * | 18.9 | * | 85.9 | 70.5 | 2 | ||
| Corylus avellana L. [L] | 44.5 | * | 44.4 | 83.9 | 90.1 | 1 | |||
| Brassica napobrassica (L.) Mill. [S] | 49.8 | * | 24.7 | * | 65.2 | ** | 41.5 | * | 5 |
| Ocimum basilicum L. [L] | 50.2 | * | 29.7 | 98.3 | 78.3 | 1 | |||
| Valeriana officinalis L. [R] | 50.3 | * | 6.5 | ** | 75.0 | * | 24.7 | ** | 6 |
| Artemisia absinthium L. [L] | 52.1 | * | 37.3 | 102 | 86.3 | 1 | |||
| Mean, M | 69.5 | 42.3 | 99.6 | ||||||
| Standard Deviation, SD | 26.0 | 27.9 | 32.8 | ||||||
| M−0.5 × SD | 56.5 | * | 28.4 | * | 83.2 | * | 57.8 | * | |
| M−1.0 × SD | 43.5 | ** | 14.5 | ** | 66.8 | ** | 38.0 | ** | |
| M−1.5 × SD | 30.5 | *** | 0.6 | *** | 50.4 | *** | 18.2 | *** | |
| M−2.0 × SD | 17.5 | **** | 34.0 | **** | |||||
| M−2.5 × SD | 4.5 | ***** | 17.6 | ***** | |||||
| Plant Family | Number of Species | Percent (%) of Species | Number of Samples | Percent (%) of Samples |
|---|---|---|---|---|
| Lamiaceae | 15 | 11.9 | 15 | 10.4 |
| Asteraceae | 14 | 11.1 | 14 | 9.7 |
| Rosaceae | 11 | 8.7 | 13 | 9.0 |
| Apiaceae | 9 | 7.1 | 11 | 7.6 |
| Brassicaceae | 5 | 4.0 | 5 | 3.5 |
| Fabaceae | 5 | 4.0 | 6 | 4.2 |
| Malvaceae | 5 | 4.0 | 5 | 3.5 |
| Cupressaceae | 3 | 2.4 | 3 | 2.1 |
| Papaveraceae | 3 | 2.4 | 3 | 2.1 |
| Poaceae | 3 | 2.4 | 5 | 3.5 |
| Other 45 * | 53 | 29.4 | 64 | 44.4 |
| Total | 126 | 100 | 144 | 100 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Isin Ozkan, T.G.; Akalin Urusak, E.; Appiah, K.S.; Fujii, Y. First Broad Screening of Allelopathic Potential of Wild and Cultivated Plants in Turkey. Plants 2019, 8, 532. https://doi.org/10.3390/plants8120532
Isin Ozkan TG, Akalin Urusak E, Appiah KS, Fujii Y. First Broad Screening of Allelopathic Potential of Wild and Cultivated Plants in Turkey. Plants. 2019; 8(12):532. https://doi.org/10.3390/plants8120532
Chicago/Turabian StyleIsin Ozkan, Tugba Gonca, Emine Akalin Urusak, Kwame Sarpong Appiah, and Yoshiharu Fujii. 2019. "First Broad Screening of Allelopathic Potential of Wild and Cultivated Plants in Turkey" Plants 8, no. 12: 532. https://doi.org/10.3390/plants8120532
APA StyleIsin Ozkan, T. G., Akalin Urusak, E., Appiah, K. S., & Fujii, Y. (2019). First Broad Screening of Allelopathic Potential of Wild and Cultivated Plants in Turkey. Plants, 8(12), 532. https://doi.org/10.3390/plants8120532








