Scribbling the Cat: A Case of the “Miracle” Plant, Moringa oleifera
Abstract
1. Introduction
2. Phytochemicals in Moringa oleifera
2.1. Glucosinolates
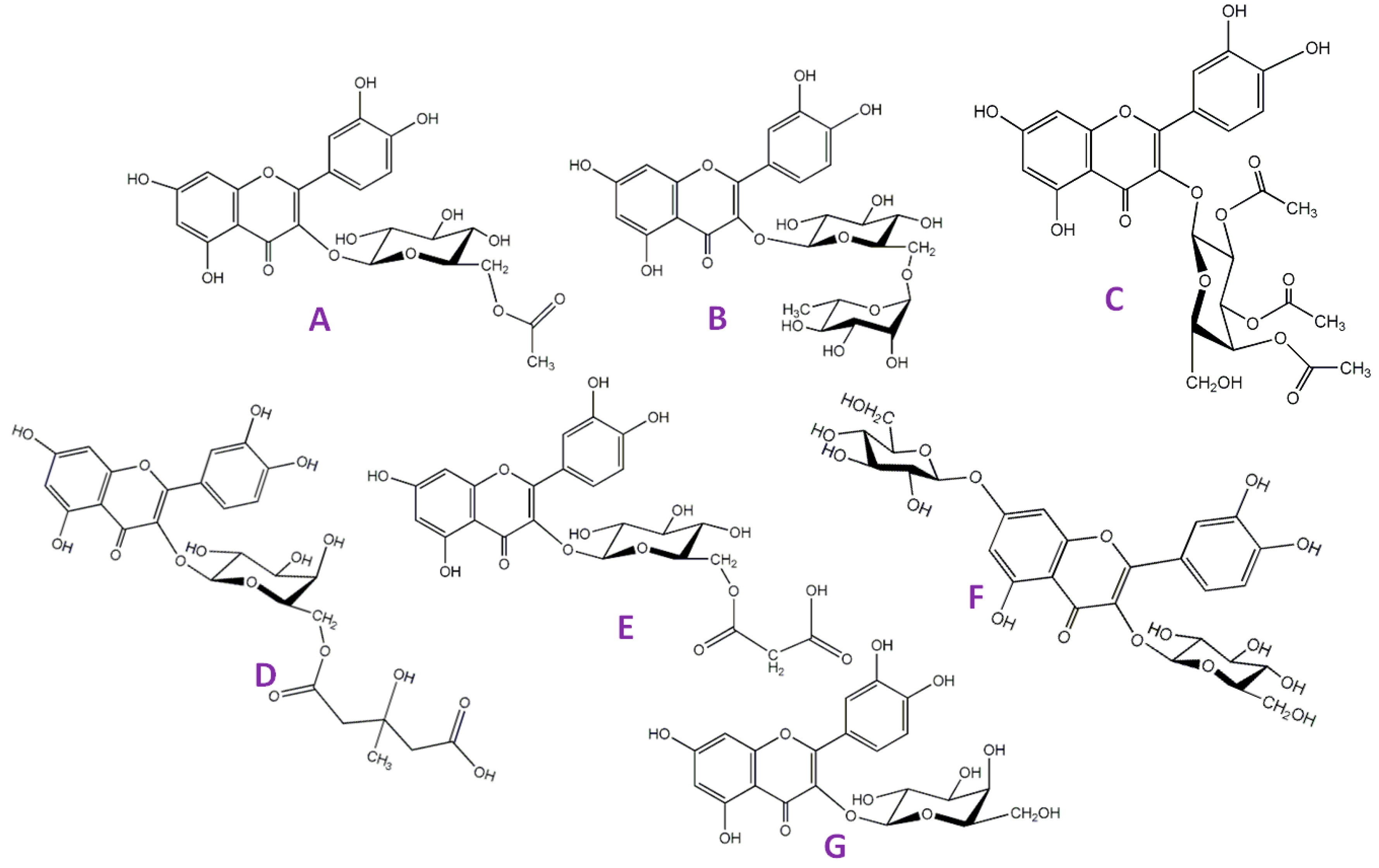
2.2. Flavonoids
2.3. Phenolic Acids
2.4. Vitamins and Minerals
2.5. Tannins
3. Nutritional Aspects of Moringa oleifera
3.1. In Humans
3.2. In Livestock
4. Medicinal Properties of Moringa oleifera
4.1. Antioxidant Properties
4.2. Anti-Inflammatory Properties
4.3. Anti-Diabetic Properties
4.4. Anti-Cancer Properties
5. Side Effects of Moringa oleifera
6. Contraindications of M. oleifera
7. Water Purification
8. Invasiveness and Allelopathy of Moringa oleifera
9. Conclusions
- Gram on gram, M. oleifera contains higher amounts of elemental nutrients than most conventional vegetable sources which makes it a potentially lucrative crop to combat food and nutritional insecurity.
- There are no scientifically proven side effects of M. oleifera to this date, despite the lack of safety information on its utility, particularly in humans.
- Based on available M. oleifera, it produces a chemically diverse range of phytochemicals which can be exploited for the development of pharmaceutical agents.
- Due to a pool of phytochemicals found in M. oleifera extracts, a number of medicinal properties have been reported to date.
- M. oleifera holds great potential both as a food supplement and medicine, however, more clinical trials are needed for the development of pharmaceutical agents.
- M. oleifera has shown some potential as a water treatment agent and can be a useful resource particularly in resource poor communities.
- There is no literature that suggests that M. oleifera could be an invasive plant species, although extreme caution has to be exercised when replanting, or introducing it, particularly in degraded lands.
Author Contributions
Funding
Conflicts of Interest
References
- Ekor, M. The growing use of herbal medicines: Issues relating to adverse reactions and challenges in monitoring safety. Front. Pharmacol. 2014, 4, 177. [Google Scholar] [CrossRef]
- Bennett, R.N.; Wallsgrove, R.M. Secondary metabolites in plant defence mechanisms. New Phytol. 1994, 127, 617–633. [Google Scholar] [CrossRef]
- Ma, Z.F.; Ahmad, J.; Zhang, H.; Khan, I.; Muhammad, S. Evaluation of phytochemical and medicinal properties of Moringa (Moringa oleifera) as a potential functional food. S. Afr. J. Bot. 2019. (In press) [CrossRef]
- Leone, A.; Spada, A.; Battezzati, A.; Schiraldi, A.; Aristil, J.; Bertoli, S. Cultivation, Genetic, Ethnopharmacology, Phytochemistry and Pharmacology of Moringa oleifera Leaves: An Overview. Int. J. Mol. Sci. 2015, 16, 12791–12835. [Google Scholar] [CrossRef]
- Fahey, J.W. Moringa oleifera: A review of the medical evidence for its nutritional, therapeutic and prophylactic properties. Trees Life J. 2005, 1, 5. [Google Scholar]
- Folkard, G.K.; Sutherland, J.P. Moringa oleifera: A tree and a litany of Potential. Agrofor. Today 1996, 8, 5–8. [Google Scholar]
- Sharma, V.; Paliwal, R.; Sharma, P.; Sharma, S. Phytochemical analysis and evaluation of antioxidant activities of hydro-ethanolic extract of Moringa oleifera Lam. pods. J. Pharm. Res. 2011, 4, 554–557. [Google Scholar]
- Gopalakrishnan, L.; Doriya, K.; Kumar, D.S. Moringa oleifera: A review on nutritive importance and its medicinal application. Food Sci. Hum. Wellness 2016, 5, 49–56. [Google Scholar] [CrossRef]
- Shih, M.-C.; Chang, C.-M.; Kang, S.-M.; Tsai, M.-L. Effect of Different Parts (Leaf, Stem and Stalk) and Seasons (Summer and Winter) on the Chemical Compositions and Antioxidant Activity of Moringa oleifera. Int. J. Mol. Sci. 2011, 12, 6077–6088. [Google Scholar] [CrossRef] [PubMed]
- Ashfaq, M.; Basra, S.M.A.; Ashfaq, U. Moringa: A miracle plant of agroforestry. J. Agric. Soc. Sci. 2012, 8, 115–122. [Google Scholar]
- Daba, M. Miracle Tree: A Review on Multi-purposes of Moringa oleifera and Its Implication for Climate Change Mitigation. J. Earth Sci Clim. Chang. 2016, 7, 366. [Google Scholar] [CrossRef]
- Anwar, F.; Latif, S.; Ashraf, M.; Gilani, A.H. Moringa oleifera: A food plant with multiple medicinal uses. Phytother. Res. 2007, 21, 17–25. [Google Scholar] [CrossRef] [PubMed]
- Ganguly, S. Indian ayurvedic and traditional medicinal implications of indigenously available plants, herbs and fruits: A review. Int. J. Res. Ayurveda Pharm. 2013, 4, 623–625. [Google Scholar] [CrossRef]
- Kumar, S.; Gopal, K. Screening of plant species for inhibition of bacterial population of raw water. J. Environ. Sci. Health A Tox. Hazard. Subst. Environ. Eng. 1999, 34, 975–987. [Google Scholar] [CrossRef]
- Mofijur, M.; Masjuki, H.H.; Kalam, M.A.; Atabani, A.E.; Fattah, I.M.R.; Mobarak, H.M. Comparative evaluation of performance and emission characteristics of Moringa oleifera and Palm oil based biodiesel in a diesel engine. Ind. Crops Prod. 2014, 53, 78–84. [Google Scholar] [CrossRef]
- Karthy, E.S.; Ranjitha, P.; Mohankumar, A. Antimicrobial potential of plant seed extracts against Multidrug Resistant Methicillin Resistant Staphylococcus aureus (MDR—MRSA). Int. J. Biol. Sci. 2009, 1, 34–40. [Google Scholar] [CrossRef][Green Version]
- Ncube, B.; Finnie, J.F.; Van Staden, J. Quality from the field: The impact of environmental factors as quality determinants in medicinal plants. S. Afr. J. Bot. 2012, 82, 11–20. [Google Scholar] [CrossRef]
- Makita, C.; Chimuka, L.; Steenkamp, P.; Cukrowska, E.; Madala, E. Comparative analyses of flavonoid content in Moringa oleifera and Moringa ovalifolia with the aid of UHPLC-qTOF-MS fingerprinting. S. Afr. J. Bot. 2016, 105, 116–122. [Google Scholar] [CrossRef]
- Bennett, R.N.; Mellon, F.A.; Foidl, N.; Pratt, J.H.; DuPont, M.S.; Perkins, L.; Kroon, P.A. Profiling glucosinolates and phenolics in vegetative and reproductive tissues of the multi-purpose trees Moringa oleifera L. (Horseradish tree) and Moringa stenopetala L. J. Agric. Food Chem. 2003, 51, 3546–3553. [Google Scholar] [CrossRef]
- Ramabulana, T.; Mavunda, R.D.; Steenkamp, P.A.; Piater, L.A.; Dubery, I.A.; Madala, N.E. Perturbation of pharmacologically relevant polyphenolic compounds in Moringa oleifera against photo-oxidative damages imposed by gamma radiation. J. Photochem. Photobiol. B Biol. 2016, 156, 79–86. [Google Scholar] [CrossRef]
- Mithen, R. Glucosinolates-biochemistry, genetics and biological activity. Plant Growth Regul. 2001, 34, 91–103. [Google Scholar] [CrossRef]
- Fahey, J.W.; Zalcmann, A.T.; Talalay, P. The chemical diversity and distribution of glucosinolates and isothiocyanates among plants. Phytochemistry 2001, 56, 5–51. [Google Scholar] [CrossRef]
- Maldini, M.; Maksoud, S.A.; Natella, F.; Montoro, P.; Petretto, G.L.; Foddai, M.; De Nicola, G.R.; Chessa, M.; Pintore, G. Moringa oleifera: Study of phenolics and glucosinolates by mass spectrometry. J. Mass Spectrom. 2014, 49, 900–910. [Google Scholar] [CrossRef]
- Brown, P.D.; Tokuhisa, J.G.; Reichelt, M.; Gershenzon, J. Variation of glucosinolate accumulation among different organs and developmental stages of Arabidopsis thaliana. Phytochemistry 2003, 62, 471–481. [Google Scholar] [CrossRef]
- Clarke, D.B. Glucosinolates, structures and analysis in food. Anal. Chem. 2010, 9660, 310–325. [Google Scholar] [CrossRef]
- Amaglo, N.K.; Bennett, R.N.; Lo Curto, R.B.; Rosa, E.A.S.; Lo Turco, V.; Giuffrida, A.; Lo Curto, A.; Crea, F.; Timpo, G.M. Profiling selected phytochemicals and nutrients in different tissues of the multipurpose tree Moringa oleifera L., grown in Ghana. Food Chem. 2010, 122, 1047–1054. [Google Scholar] [CrossRef]
- Ramabulana, T.; Mavunda, R.D.; Steenkamp, P.A.; Piater, L.A.; Dubery, I.A.; Ndhlala, A.R.; Madala, N.E. Gamma radiation treatment activates glucomoringin synthesis in Moringa oleifera. Rev. Bras. Farmacogn. 2017, 27, 569–575. [Google Scholar] [CrossRef]
- Bones, A.M.; Rossiter, J.T. The enzymic and chemically induced decomposition of glucosinolates. Phytochemistry 2006, 67, 1053–1067. [Google Scholar] [CrossRef]
- Brunelli, D.; Tavecchio, M.; Falcioni, C.; Frapolli, R.; Erba, E.; Iori, R.; Rollin, P.; Barillari, J.; Manzotti, C.; Morazzoni, P.; et al. The isothiocyanate produced from glucomoringin inhibits NF-kB and reduces myeloma growth in nude mice in vivo. Biochem. Pharmacol. 2010, 79, 1141–1148. [Google Scholar] [CrossRef]
- Tumer, T.B.; Rojas-Silva, P.; Poulev, A.; Raskin, I.; Waterman, C. Direct and indirect antioxidant activity of polyphenol- and isothiocyanate-enriched fractions from moringa oleifera. J. Agric. Food Chem. 2015, 63, 1505–1513. [Google Scholar] [CrossRef]
- Jaafaru, M.S.; Nordin, N.; Shaari, K.; Rosli, R.; Abdull Razis, A.F. Isothiocyanate from Moringa oleifera seeds mitigates hydrogen peroxide-induced cytotoxicity and preserved morphological features of human neuronal cells. PLoS ONE 2018, 13, e0196403. [Google Scholar] [CrossRef] [PubMed]
- Cook, N.C.; Samman, S. Flavonoids-chemistry, metabolism, cardioprotective effects, and dietary sources. J. Nutr. Biochem. 1996, 7, 66–76. [Google Scholar] [CrossRef]
- Wedick, N.M.; Pan, A.; Cassidy, A.; Rimm, E.B.; Sampson, L.; Rosner, B.; Willett, W.; Hu, F.B.; Sun, Q.; van Dam, R.M. Dietary flavonoid intakes and risk of type 2 diabetes in US men and women. Am. J. Clin. Nutr. 2012, 95, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Yao, L.H.; Jiang, Y.M.; Shi, J.; Tomas-Barberan, F.A.; Datta, N.; Singanusong, R.; Chen, S.S. Flavonoids in food and their health benefits. Plant Foods Hum. Nutr. 2004, 59, 113–122. [Google Scholar] [CrossRef]
- Prasain, J.K.; Carlson, S.H.; Wyss, J.M. Flavonoids and age-related disease: Risk, benefits and critical windows. Maturitas 2010, 66, 163–171. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.Y.; Chen, Y.C. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013, 15, 2099–2107. [Google Scholar] [CrossRef] [PubMed]
- Antunes-Ricardo, M.; Gutiérrez-Uribe, J.A.; Martínez-Vitela, C.; Serna-Saldívar, S.O. Topical anti-inflammatory effects of isorhamnetin glycosides isolated from Opuntia ficus-indica. Biomed. Res. Int. 2015, 2015, 847320. [Google Scholar] [CrossRef]
- Allemann, I.B.; Baumann, L. Botanicals in skin care products. Int. J. Dermatol. 2009, 48, 923–934. [Google Scholar] [CrossRef]
- Lai, J.S.; Lin, C.; Chiang, T.M. Tyrosinase inhibitory activity and thermostability of the flavonoid complex from Sophora japonica L (Fabaceae) Trop. J. Pharm. Res. 2014, 13, 243–247. [Google Scholar]
- Rodriguez-Perez, C.; Quirantes-Pine, R.; Fernandez-Gutierrez, A.; Segura-Carretero, A. Optimization of extraction method to obtain a phenolic compounds-rich extract Moringa oleifera Lam leaves. Ind. Crops Prod. 2015, 66, 246–254. [Google Scholar] [CrossRef]
- Matshediso, P.G.; Cukrowska, E.; Chimuka, L. Development of pressurised hot water extraction (PHWE) for essential compounds from Moringa oleifera leaf extracts. Food Chem. 2015, 172, 423–427. [Google Scholar] [CrossRef]
- Masike, K.; Khoza, B.S.; Steenkamp, P.A.; Smit, E.; Dubery, I.A.; Madala, N.E. A metabolomics-guided exploration of the phytochemical constituents of Vernonia fastigiata with the aid of pressurized hot water extraction and liquid chromatography-mass spectrometry. Molecules 2017, 22, 1200. [Google Scholar] [CrossRef]
- Nagaprashantha, L.D.; Vatsyayan, R.; Singhal, J.; Fast, S.; Roby, R.; Awasthi, S.; Singhal, S.S. Anti-cancer effects of novel flavonoid vicenin-2 as a single agent and in synergistic combination with docetaxel in prostate cancer. Biochem. Pharmacol. 2011, 82, 1100–1109. [Google Scholar] [CrossRef]
- Habtemariam, S. The African Moringa is to change the lives of millions in Ethiopia and far beyond. Asian Pac. J. Trop. Biomed. 2016, 6, 355–356. [Google Scholar] [CrossRef]
- Makita, C.; Chimuka, L.; Cukrowska, E.; Steenkamp, P.A.; Kandawa-Schutz, M.; Ndhlala, A.R.; Madala, N.E. UPLC-qTOF-MS profiling of pharmacologically important chlorogenic acids and associated glycosides in Moringa ovalifolia leaf extracts. S. Afr. J. Bot. 2017, 108, 193–199. [Google Scholar] [CrossRef]
- Ncube, E.N.; Mhlongo, M.I.; Piater, L.A.; Steenkamp, P.A.; Dubery, I.A.; Madala, N.E. Analyses of chlorogenic acids and related cinnamic acid derivatives from Nicotiana tabacum tissues with the aid of UPLC-QTOF-MS/MS based on the in-source collision-induced dissociation method. Chem. Cent. J. 2014, 8, 66. [Google Scholar] [CrossRef]
- Deshpande, S.; Matei, M.F.; Jaiswal, R.; Bassil, B.S.; Kortz, U.; Kuhnert, N. Synthesis, structure, and tandem mass spectrometric characterization of the diastereomers of quinic acid. J. Agric. Food Chem. 2016, 64, 7298–7306. [Google Scholar] [CrossRef] [PubMed]
- Clifford, M.N.; Jaganath, I.B.; Ludwigc, I.A.; Crozier, A. Chlorogenic acids and the acyl-quinic acids: Discovery, biosynthesis, bioavailability and bioactivity. Nat. Prod. Rep. 2017, 34, 1391–1421. [Google Scholar] [CrossRef] [PubMed]
- Mhlongo, M.I.; Piater, L.A.; Steenkamp, P.A.; Madala, N.E.; Dubery, I.A. Metabolomic fingerprinting of primed tobacco cells provide the first evidence for the biological origin of cis-chlorogenic acid. Biotechnol. Lett. 2015, 37, 205–209. [Google Scholar] [CrossRef]
- Reddy, A.R.; Chaitanya, K.V.; Vivekanandan, M. Drought-induced responses of photosynthesis and antioxidant metabolism in higher plants. J. Plant Physiol. 2004, 161, 1189–1202. [Google Scholar] [CrossRef]
- Leone, A.; Fiorillo, G.; Criscuoli, F.; Ravasenghi, S.; Santagostini, L.; Fico, G.; Spadafranca, A.; Battezzati, A.; Schiraldi, A.; Pozzi, F.; et al. Nutritional Characterization and Phenolic Profiling of Moringa oleifera Leaves Grown in Chad, Sahrawi Refugee Camps, and Haiti. Int. J. Mol. Sci. 2015, 16, 18923–18937. [Google Scholar] [CrossRef] [PubMed]
- Tshabalala, T.; Ndhlala, A.R.; Ncube, B.; Abdelgadir, H.A.; Van Staden, J. Potential substitution of the root with the leaf in the use of Moringa oleifera for antimicrobial, antidiabetic and antioxidant properties. S. Afr. J. Bot. 2019. (In press) [CrossRef]
- Sreelatha, S.; Padma, P.R. Antioxidant activity and total phenolic content of Moringa oleifera leaves in two stages of maturity. Plant Foods Hum. Nutr. 2009, 64, 303–311. [Google Scholar] [CrossRef] [PubMed]
- Abdulkadir, A.R.; Jahan, M.S.; Zawawi, D.D. Effect of chlorophyll content and maturity on total phenolic, total flavonoid contents and antioxidant activity of Moringa oleifera leaf (Miracle tree). J. Chem. Pharm. Res. 2015, 75, 1147–1152. [Google Scholar]
- Ndhlala, A.; Mulaudzi, R.; Ncube, B.; Abdelgadir, H.; du Plooy, C.; Van Staden, J. Antioxidant, Antimicrobial and Phytochemical Variations in Thirteen Moringa oleifera Lam. Cultivars. Molecules 2014, 19, 10480–10494. [Google Scholar] [CrossRef]
- Sangkitikomol, W.; Rocejanasaroj, A.; Tencomnao, T. Effect of Moringa oleifera on advanced glycation end-product formation and lipid metabolism gene expression in HepG2 cells. Genet. Mol. Res. 2014, 13, 723–735. [Google Scholar] [CrossRef]
- Valentão, P.; Fernandes, E.; Carvalho, F.; Andrade, P.B.; Seabra, R.M.; Bastos, M.L. Studies on the antioxidant activity of Lippia citriodora infusion: Scavenging effect on superoxide radical, hydroxyl radical and hypochlorous acid. Biol. Pharm. Bull. 2002, 25, 1324–1327. [Google Scholar] [CrossRef]
- Mahmood, K.T.; Mugal, T.; Haq, I.U. Moringa oleifera: A natural gift-A review. J. Pharm. Sci. Res. 2010, 2, 775–781. [Google Scholar]
- Ferreira, P.M.P.; Farias, D.F.; Oliveira, J.T.D.A.; Carvalho, A.D.F.U. Moringa oleifera: Bioactive compounds and nutritional potential. Rev. Nutr. 2008, 21, 431–437. [Google Scholar] [CrossRef]
- Saini, R.K.; Manoj, P.; Shetty, N.P.; Srinivasan, K.; Giridhar, P. Relative bioavailability of folate from the traditional food plant Moringa oleifera L. as evaluated in a rat model. J. Food Sci. Technol. 2016, 53, 511–520. [Google Scholar] [CrossRef]
- Joshi, P.; Mehta, D. Effect of dehydration on the nutritive value of drumstick leaves. J. Metabol. Syst. Biol. 2010, 1, 5–9. [Google Scholar]
- Kidmose, U.; Yang, R.Y.; Thilsted, S.H.; Christensen, L.P.; Brandt, K. Content of carotenoids in commonly consumed Asian vegetables and stability and extractability during frying. J. Food Compos. Anal. 2006, 19, 562–571. [Google Scholar] [CrossRef]
- Binstock, R.H. The war on anti-aging medicine. Gerontologist 2003, 43, 4–14. [Google Scholar] [CrossRef]
- Pong, K. Oxidative stress in neurodegenerative diseases: Therapeutic implications for superoxide dismutase mimetics. Expert Opin. Biol. Ther. 2003, 3, 127–139. [Google Scholar] [CrossRef]
- Ramachandran, C.; Peter, K.V.; Gopalakrishnan, P.K. Drumstick (Moringa oleifera): A multipurpose Indian vegetable. Econ. Bot. 1980, 34, 276–283. [Google Scholar] [CrossRef]
- Reed, J.D.; Soller, H.; Woodward, A. Fodder tree and straw diets for sheep: Intake, growth, digestibility and the effects of phenolics on nitrogen utilization. Anim. Feed Sci. Technol. 1990, 30, 39–50. [Google Scholar] [CrossRef]
- Bhatta, R.; Saravanan, M.; Baruah, L.; Sampath, K.T. Nutrient content, in vitro ruminal fermentation characteristics and methane reduction potential of tropical tannin-containing leaves. J. Sci. Food Agric. 2012, 92, 2929–2935. [Google Scholar] [CrossRef]
- Teixeira, E.M.B.; Carvalho, M.R.B.; Neves, V.A.; Silva, M.A.; Arantes-Pereira, L. Chemical characteristics and fractionation of proteins from Moringa oleifera Lam. leaves. Food Chem. 2014, 147, 51–54. [Google Scholar] [CrossRef]
- Moyo, B.; Masika, P.J.; Hugo, A.; Muchenje, V. Nutritional characterization of Moringa (Moringa oleifera Lam.) leaves. Afr. J. Biotechnol. 2011, 10, 12925–12933. [Google Scholar]
- Hagerman, A.E.; Riedl, K.M.; Jones, G.A.; Sovik, K.N.; Ritchard, N.T.; Hartzfeld, P.W.; Riechel, T.L. High molecular weight plant polyphenolics (tannins) as biological antioxidants. J. Agric. Food Chem. 1998, 46, 1887–1892. [Google Scholar] [CrossRef]
- Tshabalala, T.; Sikosana, J.L.N.; Chivandi, E. Nutrient intake, digestibility and nitrogen retention in indigenous goats fed on Acacia nilotica fruits treated for condensed tannins. S. Afr. J. Anim. Sci. 2013, 43, 457–463. [Google Scholar] [CrossRef]
- Lu, W.; Wang, J.; Zhang, H.J.; Wu, S.G.; Qi, G.H. Evaluation of Moringa oleifera leaf in laying hens: Effects on laying performance, egg quality, plasma biochemistry and organ histopathological indices. Ital. J. Anim. Sci. 2016, 15, 658–665. [Google Scholar] [CrossRef]
- Tesfaye, E.B.; Animut, G.M.; Urge, M.L.; Dessie, T.A. Cassava root chips and Moringa oleifera leaf meal as alternative feed ingredients in the layer ration1. J. Appl. Poult. Res. 2014, 23, 614–624. [Google Scholar] [CrossRef]
- Singh, A.K.; Rana, H.K.; Tshabalala, T.; Kumar, R.; Ndhlala, A.R.; Pandey, A.K. Phytochemical, nutraceutical and pharmacological attributes of a functional crop Moringa oleifera Lam: An overview. S. Afr. J. Bot. 2019. [Google Scholar] [CrossRef]
- Animashaun, J.O.; Williams, F.; Toye, A. Towards validating moringa′s nutraceutical benefits: An examination of consumers′ perspectives vis-a-vis health benefits efficacy and willingness to pay. J. Agris On-Line Pap. Econ. Inform. 2013, 5, 11. [Google Scholar]
- Mabapa, M.P.; Ayisi, K.K.; Mariga, I.K.; Mohlabi, R.C.; Chuene, R.S. Production and Utilization of Moringa by Farmers in Limpopo Province, South Africa. Agric. Res. 2017, 12, 160–171. [Google Scholar] [CrossRef]
- Saini, R.K.; Sivanesan, I.; Keum, Y.-S. Phytochemicals of Moringa oleifera: A review of their nutritional, therapeutic and industrial significance. 3 Biotech 2016, 6, 203. [Google Scholar] [CrossRef]
- Mohammed, A.; Lai, O.; Muhammad, S.; Long, K.; Ghazali, H. Moringa oleifera, potentially a new source of oleic acid-type oil for Malaysia. Investig. Innov. 2003, 3, 137–140. [Google Scholar]
- Frigolet, M.E.; Gutiérrez-Aguilar, R. The Role of the Novel Lipokine Palmitoleic Acid in Health and Disease. Adv. Nutr. 2017, 8, 173S–181S. [Google Scholar] [CrossRef]
- Jongrungruangchok, S.; Bunrathep, S.; Songsak, T. Nutrients and minerals content of eleven different samples of Moringa oleifera cultivated in Thailand. J. Health Res. 2010, 24, 123–127. [Google Scholar]
- Ntila, S.; Ndhlala, A.R.; Kolanisi, U.; Abdelgadir, H.; Siwela, M. Acceptability of a moringa-added complementary soft porridge to caregivers in Hammanskraal, Gauteng province and Lebowakgomo, Limpopo province, South Africa. S. Afr. J. Clin. Nutr. 2019, 32, 51–57. [Google Scholar] [CrossRef]
- Abd El-Hack, M.; Alagawany, M.; Elrys, A.; Desoky, E.S.; Tolba, H.; Elnahal, A.; Elnesr, S.; Swelum, A. Effect of Forage Moringa oleifera L.(moringa) on Animal Health and Nutrition and Its Beneficial Applications in Soil, Plants and Water Purification. Agriculture 2018, 8, 145. [Google Scholar] [CrossRef]
- Kakengi, A.; Shem, M.; Sarwatt, S.; Fujihara, T. Can Moringa oleifera be used as a protein supplement for ruminants? Asian-australas. J. Anim. Sci. 2005, 18, 42–47. [Google Scholar] [CrossRef]
- Rubanza, C.; Shem, M.; Otsyina, E.; Bakengesa, S.; Ichinohe, T.; Fujihara, T. Polyphenolics and tanninseffect on in vitro digestibility of selected Acacia species leaves. Anim. Feed Sci. Technol. 2005, 119, 129–142. [Google Scholar] [CrossRef]
- Anwar, F.; Rashid, U. Physico-chemical characteristics of Moringa oleifera seeds and seed oil from a wild provenance of Pakistan. Pak. J. Bot. 2007, 39, 1443–1453. [Google Scholar]
- Siddhuraju, P.; Becker, K. Antioxidant properties of various solvent extracts of total phenolic constituents from three different agroclimatic origins of drumstick tree (Moringa oleifera Lam.) leaves. J. Agric. Food Chem. 2003, 51, 2144–2155. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.R.; Vijayakumar, M.; Mathela, C.S.; Rao, C.V. In vitro and in vivo antioxidant properties of different fractions of Moringa oleifera leaves. Food Chem. Toxicol. 2009, 47, 2196–2201. [Google Scholar] [CrossRef] [PubMed]
- Lalas, S.; Tsaknis, J. Extraction and identification of natural antioxidant from the seeds of the Moringa oleifera tree variety of Malawi. J. Am. Oil Chem. Soc. 2002, 799, 677–683. [Google Scholar] [CrossRef]
- Fakurazi, S.; Sharifudin, S.A.; Arulselvan, P. Moringa oleifera hydroethanolic extracts effectively alleviate acetaminophen-induced hepatotoxicity in experimental rats through their antioxidant nature. Molecules 2012, 17, 8334–8350. [Google Scholar] [CrossRef]
- Singh, B.N.; Singh, B.; Singh, R.; Prakash, D.; Dhakarey, R.; Upadhyay, G.; Singh, H. Oxidative DNA damage protective activity, antioxidant and anti-quorum sensing potentials of Moringa oleifera. Food Chem. Toxicol. 2009, 47, 1109–1116. [Google Scholar] [CrossRef]
- Moyo, B.; Oyedemi, S.; Masika, P.; Muchenje, V. Polyphenolic content and antioxidant properties of Moringa oleifera leaf extracts and enzymatic activity of liver from goats supplemented with Moringa oleifera leaves/sunflower seed cake. Meat Sci. 2012, 91, 441–447. [Google Scholar] [CrossRef] [PubMed]
- Jayawardana, B.C.; Liyanage, R.; Lalantha, N.; Iddamalgoda, S.; Weththasinghe, P. Antioxidant and antimicrobial activity of drumstick (Moringa oleifera) leaves in herbal chicken sausages. LWT-Food Sci. Technol. 2015, 64, 1204–1208. [Google Scholar] [CrossRef]
- Hazra, S.; Biswas, S.; Bhattacharyya, D.; Das, S.K.; Khan, A. Quality of cooked ground buffalo meat treated with the crude extracts of Moringa oleifera (Lam.) leaves. J. Food Sci. Technol. 2012, 49, 240–245. [Google Scholar] [CrossRef] [PubMed]
- Das, A.K.; Rajkumar, V.; Verma, A.K.; Swarup, D. Moringa oleifera leaves extract: A natural antioxidant for retarding lipid oxidation in cooked goat meat patties. Int. J. Food Sci. Technol. 2012, 47, 585–591. [Google Scholar] [CrossRef]
- Qwele, K.; Hugo, A.; Oyedemi, S.; Moyo, B.; Masika, P.; Muchenje, V. Chemical composition, fatty acid content and antioxidant potential of meat from goats supplemented with Moringa (Moringa oleifera) leaves, sunflower cake and grass hay. Meat Sci. 2013, 93, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Feldmann, M.; Brennan, F.M.; Maini, R.N. Role of cytokines in rheumatoid arthritis. Annu. Rev. Immunol. 1996, 14, 397–440. [Google Scholar] [CrossRef]
- Graves, D.T.; Liu, R.; Oates, T.W. Diabetes enhanced inflammation and apoptosis–impact on periodontal pathosis. Periodontology 2000 2007, 45, 128–137. [Google Scholar] [CrossRef]
- Araújo, L.C.C.; Aguiar, J.S.; Napoleão, T.H.; Mota, F.V.B.; Barros, A.L.S.; Moura, M.C.; Coriolano, M.C.; Coelho, L.C.B.B.; Silva, T.G.; Paiva, P.M.G. Evaluation of cytotoxic and anti-inflammatory activities of extracts and lectins from Moringa oleifera seeds. PLoS ONE 2013, 8, e81973. [Google Scholar] [CrossRef]
- Cheenpracha, S.; Park, E.-J.; Yoshida, W.Y.; Barit, C.; Wall, M.; Pezzuto, J.M.; Chang, L.C. Potential anti-inflammatory phenolic glycosides from the medicinal plant Moringa oleifera fruits. Bioorg. Med. Chem. 2010, 18, 6598–6602. [Google Scholar] [CrossRef]
- Muangnoi, C.; Chingsuwanrote, P.; Praengamthanachoti, P.; Svasti, S.; Tuntipopipat, S. Moringa oleifera pod inhibits inflammatory mediator production by lipopolysaccharide-stimulated raw 264.7 murine macrophage cell lines. Inflammation 2012, 35, 445–455. [Google Scholar] [CrossRef]
- Sulaiman, M.R.; Zakaria, Z.; Bujarimin, A.; Somchit, M.; Israf, D.; Moin, S. Evaluation of Moringa oleifera aqueous extract for antinociceptive and anti-inflammatory activities in animal models. Pharm. Biol. 2008, 46, 838–845. [Google Scholar] [CrossRef]
- Udupa, S.; Udupa, A.; Kulkarni, D. Studies on the anti-inflammatory and wound healing properties of Moringa oleifera and Aegle marmelos. Fitoterapia 1994, 65, 119–123. [Google Scholar]
- Vasanth, K.; Minakshi, G.; Ilango, K.; Kumar, R.M.; Agrawal, A.; Dubey, G. Moringa oleifera attenuates the release of pro-inflammatory cytokines in lipopolysaccharide stimulated human monocytic cell line. Ind. Crops Prod. 2015, 77, 44–50. [Google Scholar] [CrossRef]
- Chuang, H.Y.; Lee, E.; Liu, Y.T.; Lee, D.; Ideker, T. Network based classification of breast cancer metastasis. Mol. Syst. Biol. 2007, 3, 140. [Google Scholar] [CrossRef] [PubMed]
- Faizi, M.S.H.; Hussain, S. Dichlorido (n, n-diethyl-4-{[(quinolin-2-yl) methylidene] amino-κ2n, n′} aniline) mercury (ii). Acta Cryst. 2014, 70, m197. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, P.; Patel, N.K.; Bhutani, K.K. Synthesis of new heterocyclic lupeol derivatives as nitric oxide and pro-inflammatory cytokine inhibitors. Bioorg. Med. Chem. Lett. 2014, 24, 3596–3599. [Google Scholar] [CrossRef] [PubMed]
- Pandith, A.A.; Shah, Z.A.; Siddiqi, M.A. Oncogenic Role of Fibroblast Growth Factor Receptor 3 in Tumorigenesis of Urinary Bladder Cancer; Elsevier: Amsterdam, The Netherlands, 2013. [Google Scholar]
- Valerio, M.; Awad, A.B. Β-sitosterol down-regulates some pro-inflammatory signal transduction pathways by increasing the activity of tyrosine phosphatase SHP-1 in J774a.1 murine macrophages. Int. Immunopharmacol. 2011, 11, 1012–1017. [Google Scholar] [CrossRef]
- Cavallari, I.; Cannon, C.P.; Braunwald, E.; Goodrich, E.L.; Im, K.; Lukas, M.A.; O’donoghue, M.L. Metabolic syndrome and the risk of adverse cardiovascular events after an acute coronary syndrome. Eur. J. Prev. Cardiol. 2018, 25, 830–838. [Google Scholar] [CrossRef]
- Grundy, S.M. Metabolic syndrome update. Trends Cardiovasc. Med. 2016, 26, 364–373. [Google Scholar] [CrossRef]
- O′neill, S.; O′driscoll, L. Metabolic syndrome: A closer look at the growing epidemic and its associated pathologies. Obes Res. 2015, 16, 1–12. [Google Scholar] [CrossRef]
- López, M.; Ríos-Silva, M.; Huerta, M.; Cárdenas, Y.; Bricio-Barrios, J.A.; Díaz-Reval, M.I.; Urzúa, Z.; Huerta-Trujillo, M.; López-Quezada, K.; Trujillo, X. Effects of Moringa oleifera leaf powder on metabolic syndrome induced in male Wistar rats: A preliminary study. J. Int. Med. Res. 2018, 46, 3327–3336. [Google Scholar] [CrossRef] [PubMed]
- Villarruel-López, A.; López-de la Mora, D.; Vázquez-Paulino, O.; Puebla-Mora, A.; Torres-Vitela, M.R.; Guerrero-Quiroz, L.; Nuño, K. Effect of Moringa oleifera consumption on diabetic rats. BMC Complement. Altern. Med. 2018, 18, 127. [Google Scholar] [CrossRef] [PubMed]
- Divi, S.; Bellamkonda, R.; Dasireddy, S.K. Evaluation of antidiabetic and antihyperlipedemic potential of aqueous extract of Moringa oleifera in fructose fed insulin resistant and STZ induced diabetic wistar rats: A comparative study. Asian J. Pharm. Clin. Res. 2012, 5, 67–72. [Google Scholar]
- Sugunabai, J.; Jayaraj, M.; Karpagam, T.; Varalakshmi, B. Antidiabetic efficiency of Moringa oleifera and Solanum nigrum. Int. J. Pharm. Pharm. Sci. 2014, 6, 40–42. [Google Scholar]
- Jaiswal, D.; Rai, P.K.; Kumar, A.; Mehta, S.; Watal, G. Effect of Moringa oleifera Lam.leaves aqueous extract therapy on hyperglycemic rats. J. Ethnopharmacol. 2009, 123, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Park, A. Park′s Textbook of Social and Preventive Medicine; Banarasidas Bhanot Publishers: Jabalpur, India, 2007. [Google Scholar]
- WHO. World Health Organisation, Study Group on Diabetes Mellitus; WHO: Geneva, Switzerland, 1994; pp. 78–79. [Google Scholar]
- Francis, J.A.; Jayaprakasam, B.; Olson, L.K.; Nair, M. Insulin secretagogues from Moringa oleifera with cyclooxygenase enzyme and lipid peroxidation inhibiting activities. Helv. Chim. Acta 2004, 87, 317–326. [Google Scholar] [CrossRef]
- Yassa, H.D.; Tohamy, A.F. Extract of Moringa oleifera leaves ameliorates streptozotocin-induced diabetes mellitus in adult rats. Acta Histochem. 2014, 116, 844–854. [Google Scholar] [CrossRef]
- Adeeyo, A.; Adefule, A.; Ofusori, D.; Aderinola, A.; Caxton-Martins, E. Antihyperglycemic effects of aqueous leaf extracts of mistletoe and Moringa oleifera in streptozotocin-induced diabetes wistar rats. Diabetol. Croat. 2013, 42, 81–88. [Google Scholar]
- Rudge, M.V.; Damasceno, D.C.; Volpato, G.T.; Almeida, F.C.; Calderon, I.M.; Lemonica, I.P. Effect of Ginkgo biloba on the reproductive outcome and oxidative stress biomarkers of streptozotocin-induced diabetic rats. Braz. J. Med. Biol. Res. 2007, 40, 1095–1099. [Google Scholar] [CrossRef]
- Ghiridhari, V.V.A.; Malhati, D.; Geetha, K. Anti-diabetic properties of drumstick (Moringa oleifera) leaf tablets. Int. J. Health Nutr. 2011, 2, 1–5. [Google Scholar]
- Gupta, R.; Sharma, A.K.; Dobhal, M.; Sharma, M.; Gupta, R. Antidiabetic and antioxidant potential of β-sitosterol in streptozotocin induced experimental hyperglycemia. J. Diabetes 2011, 3, 29–37. [Google Scholar] [CrossRef] [PubMed]
- Bernal-Mizrachi, E.; Wen, W.; Stahlhut, S.; Welling, C.M.; Permutt, M.A. Islet β cell expression of constitutively active akt1/pkbα induces striking hypertrophy, hyperplasia, and hyperinsulinemia. J. Clin. Investig. 2001, 108, 1631–1638. [Google Scholar] [CrossRef] [PubMed]
- Ross, M.H.; Pawlina, W. Histology: A Text and Atlas, with Correlated Cell and Molecular Biology; Lippincott Williams and Wilkins: Philadelphia, PA, USA, 2001; pp. 647–664. [Google Scholar]
- Jaiswal, D.; Rai, P.; Mehta, S.; Chatterji, S.; Shukla, S.; Rai, D.; Sharma, G.; Sharma, B.; Watal, G. Role of Moringa oleifera in regulation of diabetes-induced oxidative stress. Asian Pac. J. Trop. Med. 2013, 6, 426–432. [Google Scholar] [CrossRef]
- Kumari, D.J. Hypoglycaemic effect of Moringa oleifera and Azadirachta indica in type 2 diabetes mellitus. Bioscan 2010, 5, 211–214. [Google Scholar]
- Ndong, M.; Uehara, M.; Katsumata, S.; Suzuki, K. Effects of oral administration of Moringa oleifera Lam on glucose tolerance in goto-kakizaki and wistar rats. J. Clin. Biochem. Nutr. 2007, 40, 229–233. [Google Scholar] [CrossRef]
- Khan, H.A.; Alhomida, A.S. A review of the logistic role of l-carnitine in the management of radiation toxicity and radiotherapy side effects. J. Appl. Toxicol. 2011, 31, 707–713. [Google Scholar] [CrossRef]
- Guevara, A.; Vargas, C.; Sakurai, H.; Fujiwara, Y.; Hashimoto, K.; Maoka, T.; Kozuka, M.; Ito, Y.; Tokuda, H.; Nishino, H. An antitumor promoter from Moringa oleifera Lam. Mutat Res. Genet. Toxicol Environ. Mutagen. 1999, 440, 181–188. [Google Scholar] [CrossRef]
- Murakami, A.; Kitazono, Y.; Jiwajinda, S.; Koshimizu, K.; Ohigashi, H. Niaziminin, a thiocarbamate from the leaves of Moringa oleifera, holds a strict structural requirement for inhibition of tumor-promoter-induced epstein-barr virus activation. Planta Med. 1998, 64, 319–323. [Google Scholar] [CrossRef]
- Parvathy, M.; Umamaheshwari, A. Cytotoxic effect of Moringa oleifera leaf extracts on human multiple myeloma cell lines. Trends Med. Res. 2007, 2, 44–50. [Google Scholar]
- Jung, I.L. Soluble Extract from Moringa oleifera Leaves with a New Anticancer Activity. PLoS ONE 2014, 9, e95492. [Google Scholar] [CrossRef]
- Jung, I.L.; Lee, J.H.; Kang, S.C. A potential oral anticancer drug candidate, Moringa oleifera leaf extract, induces the apoptosis of human hepatocellular carcinoma cells. Oncol. Lett. 2015, 10, 1597–1604. [Google Scholar] [CrossRef] [PubMed]
- Muyibi, S.A.; Evison, L.M. Optimizing physical parameters affecting coagulation of Turbid water with Moringa oleifera seeds. Water Research 1995, 29, 2689–2695. [Google Scholar] [CrossRef]
- Vasanth, K.; Ilango, K.; MohanKumar, R.; Agrawal, A.; Dubey, G.P. Anticancer activity of Moringa oleifera mediated silver nanoparticles on human cervical carcinoma cells by apoptosis induction. Colloids Surf. B 2014, 117, 354–359. [Google Scholar] [CrossRef] [PubMed]
- Tahiliani, P.; Kar, A. Role of Moringa oleifera leaf extract in the regulation of thyroid hormone status in adult male and female rats. Pharmacol. Res. 2000, 41, 319–323. [Google Scholar] [CrossRef] [PubMed]
- Das, N.; Sikder, K.; Ghosh, S.; Fromenty, B.; Dey, S. Moringa oleifera Lam. leaf extract prevents early liver injury and restores antioxidant status in mice fed with high-fat diet. Indian J. Exp. Biol. 2012, 50, 404–412. [Google Scholar] [PubMed]
- Kelly, G. Peripheral metabolism of thyroid hormones: A review. Altern. Med. Rev. 2000, 5, 306. [Google Scholar]
- Sileshi, T.; Makonnen, E.; Debella, A.; Tesfaye, B. Antihyperglycemic and subchronic toxicity study of Moringa stenopetala leaves in mice. J. Coast. Life Med. 2014, 2, 214–221. [Google Scholar]
- Gholap, S.; Kar, A. Hypoglycaemic effects of some plant extracts are possibly mediated through inhibition in corticosteroid concentration. Pharmazie 2004, 59, 876–878. [Google Scholar]
- Barichella, M.; Pezzoli, G.; Faierman, S.A.; Raspini, B.; Rimoldi, M.; Cassani, E.; Bertoli, S.; Battezzati, A.; Leone, A.; Iorio, L. Nutritional characterisation of Zambian Moringa oleifera: Acceptability and safety of short-term daily supplementation in a group of malnourished girls. Int. J. Food Sci. Nutr. 2019, 70, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Stohs, S.J.; Hartman, M.J. Review of the safety and efficacy of Moringa oleifera. Phytother. Res. 2015, 29, 796–804. [Google Scholar] [CrossRef]
- Annongu, A.; Karim, O.; Toye, A.; Sola-Ojo, F.; Kayode, R.; Badmos, A.; Alli, O.; Adeyemi, K. Geo-Assessment of chemical composition and nutritional Evaluation of Moringa oleifera seeds in nutrition of Broilers. J. Agric. Sci. 2014, 6, 119. [Google Scholar] [CrossRef]
- Maizuwo, A.I.; Hassan, A.S.; Momoh, H.; Muhammad, J.A. Phytochemical Constituents, Biological Activities, Therapeutic Potentials and Nutritional Values of Moringa oleifera (Zogale): A Review. J. Drug Des. Med. 2017, 3, 60. [Google Scholar]
- Dutta, A.K. Moringa oleifera: A Review on its importance and medicinal applications in recent age. World J. Pharm. Pharm. Sci. 2017, 6, 1829–1843. [Google Scholar]
- Adedapo, A.; Mogbojuri, O.; Emikpe, B. Safety evaluations of the aqueous extract of the leaves of Moringa oleifera in rats. J. Med. Plants Res. 2009, 3, 586–591. [Google Scholar]
- Pritchard, M.; Mkandawire, T.; Edmondson, A.; O′neill, J.G.; Kululanga, G. Potential of using plant extracts for purification of shallow well water in Malawi. Phys. Chem. Earth Parts A B C 2009, 34, 799–805. [Google Scholar] [CrossRef]
- Amagloh, F.K.; Benang, A. Effectiveness of Moringa oleifera seed as coagulant for water purification. Afr. J. Agric. Res. 2009, 4, 119–123. [Google Scholar]
- Sotheeswaran, S.; Nand, V.; Matakite, M.; Kanayathu, K. Moringa oleifera and other local seeds in water purification in developing countries. Res. J. Chem. Environ. 2011, 15, 135–138. [Google Scholar]
- Muyibi, S.A.; Megat, J.M.M.N.; Lam, H.L.; Tan, K.L. Effects of oil extraction from Moringa oleifera seeds on coagulation of turbid water. Int. J. Environ. Stud. 2002, 59, 243–254. [Google Scholar] [CrossRef]
- Ndabigengesere, A.; Narasiah, K.S.; Talbot, B.G. Active agent and mechanism of coagulation of turbid waters using Moringa oleifera. Water Res 1995, 2, 703–710. [Google Scholar] [CrossRef]
- Pritchard, M.; Craven, T.; Mkandawire, T.; Edmondson, A.S.; O′neill, J.G. A study of the parameters affecting the effectiveness of Moringa oleifera in drinking water purification. Phys. Chem. Earth Parts A B C 2010, 35, 791–797. [Google Scholar] [CrossRef]
- Mangale, S.M.; Chonde, S.G.; Jadhav, A.S.; Raut, P.D. Study of Moringa oleifera (drumstick) seed as natural absorbent and antimicrobial agent for river water treatment. J. Nat. Prod. Plant Resour. 2012, 2, 89–100. [Google Scholar]
- Bakare, B.F. An Investigation of Moringa oleifera Seed Extract as a Natural Coagulant in Water Treatment. In Proceedings of the World Congress on Engineering and Computer Science, San Francisco, CA, USA, 19–21 October 2016. [Google Scholar]
- Schwarz, D. Water clarification using Moringa olifera. Technol. Inform. 2000, 1, 17–20. [Google Scholar] [CrossRef]
- Scholes, R.J.; Archer, S.R. Tree-grass interactions in savannas. Annu. Rev. Ecol. Evol. Syst. 1997, 28, 517–544. [Google Scholar] [CrossRef]
- Tshabalala, T.; Ncube, B.; Moyo, H.P.; Abdel-Rahman, E.M.; Mutanga, O.; Ndhlala, A.R. Predicting the spatial suitability distribution of Moringa oleifera cultivation using analytical hierarchical process modelling. S. Afr. J. Bot. 2019. (In press) [CrossRef]
- Zaku, S.G.; Emmanuel, S.; Tukur, A.A.; Kabir, A. Moringa oleifera: An underutilized tree in Nigeria with amazing versatility: A review. Afr. J. Food Sci. 2015, 9, 456–461. [Google Scholar]
- Bridgemohan, P.; Bridgemohan, R.S.H. Invasive weed risk assesment of three potential bioenerygy fuel species. Int. J. Biodivers. Conserv. 2014, 6, 790–796. [Google Scholar]
- Nahar, S.; Faisal, F.M.; Iqbal, J.; Rahman, M.; Yusuf, A. Antiobesity activity of Moringa oleifera leaves against high fat diet-induced obesity in rats. Int. J. Basic Clin. Pharmacol. 2016, 5, 1263–1268. [Google Scholar] [CrossRef]
- Navie, S.; Csurhes, S. Invasive Plant Risk Assessment: Moringa Oleifera; Department of Agriculture and Fisheries Biosecurity Queensland: Biosecurity Queensland: Brisbane, Queensland, Australia, 2016.
- CABI. Invasive Species Compendium. Available online: https://www.cabi.org/isc/datasheet/34868 (accessed on 22 June 2018).
- Fuglie, L.J. New Uses of Moringa Studied in Nica Ragua: ECHO′s Technical Network Site-Networking Global Hunger Solutions; ECHO: North Fort Myers, FL, USA, 2010. [Google Scholar]
- Mehboob, W.; Rehman, H.; Basra, S.M.A.; Afzal, I. Role of seed priming in improving performance of spring maize. In Proceedings of the International Seminar on Crop Management: Issues and Options, University of Agriculture, Faisalabad, Pakistan, 1–2 June 2011; p. 55. [Google Scholar]
- Soliman, M.H.; Ahlam, H.H.; Hamdah, A.G.; Shroug, S. Allelopathic Effect of Moringa oleifera Leaves Extract on Seed Germination and Early Seedling Growth of Faba Bean (Vicia faba L.). Int. J. Agric. Technol. 2017, 13, 105–117. [Google Scholar]
- Iqbal, M.A. Role of moringa, brassica and sorghum water extracts in increasing crops growth and yield: A Review. Am. Eurasian J. Agric. Environ. Sci. 2014, 14, 1150–1158. [Google Scholar]
- Nouman, W.; Siddiqui, M.T.; Basra, S.M.A. Moringa oleifera leaf extract: An innovative priming tool for rangeland grasses. Turk. J. Agric. For. 2012, 35, 65–75. [Google Scholar]
- Phiri, C. Influence of Moringa oleifera leaf extracts on germination and early seedling development of major cereals. Agric. Biol. J. N Am. 2010, 1, 774–777. [Google Scholar] [CrossRef]
- Hossain, M.M.; Miah, G.; Ahamed, T.; Sarmin, N.S. Study on allelopathic effect of Moringa oleifera on the growth and productivity of mungbean. Int. J. Agric. Crop. Sci. 2012, 4, 1122–1128. [Google Scholar]
- Tahir, N.A.; Qader, K.O.; Azeez, H.A.; Rashid, J.S. Inhibitory allelopathic effects of Moringa oleifera Lamk plant extracts on wheat and Sinapis arvensis L. Allelopath. J. 2018, 44, 35–48. [Google Scholar] [CrossRef]
- Hussain, M.; Farooq, M.; Basra, S.M.; Lee, D.J. Application of Moringa allelopathy in crop sciences. In Allelopathy; Springer: Berlin/Heidelberg, Germany, 2013; pp. 469–483. [Google Scholar]
- Iqbal, M.A. Managing sunflower (Helianthus annuus L.) nutrition with foliar application of moringa (Moringa oleifera Lam.) leaf extract. Am. Eurasian J. Agric. Environ. Sci. 2014, 14, 1339–1345. [Google Scholar]
| Bioactive Compound | Specific compound | Properties | References |
|---|---|---|---|
| Vitamin A | Retinol, Retinal and Retinoic acids | Leaves contain 11,300–23,000 IU (international unit) of vitamin A. | [5,59] |
| Vitamin B | Folates, such as 5-Formyl-5,6,7,8-tetrahydrofolic acid, 5,6,7,8-tetrahydrofolic acid and 5-Methyl-5,6,7,8-tetrahydrofolic acid | Involved in DNA synthesis and cell division. | [60] |
| Carotenoids | β-carotene | Ranges from 6.63 mg/100 g in fresh leaves to about 39.6 mg/100 g in air-dried leaves. | [4,61,62] |
| Vitamin C | Ascorbic acid | Found in amounts of about 200 mg/100 g (greater than in orange fruits). Acts as an antioxidant. | [63,64,65] |
| Minerals | Potassium (K), Iron (Fe), Calcium (Ca) and Magnesium (Mg). | Contains more calcium, iron and potassium than in milk, spinach and bananas respectively. Vegetative parts and immature fruits contain the most potassium. | [5,8,26] |
| Antioxidant Model Used | Candidate Compounds | Solvent | Ref. |
|---|---|---|---|
| Leaf | |||
| 1,1-diphenyl 2-picrylhydrazyl (DPPH) | Crude extracts, quercetin, kaempferol, gallic, chlorogenic, ellagic, ferulic acid, rutin, gallic acid, vanillin | Water, 70% ethnol, 80% ethanol, 80% methanol. Chloroform, acetone | [80,82,84,85,86,87,89,90] |
| β-carotene-linoleic acid | Quercetin, kaempferol, gallic, chlorogenic, ellagic, ferulic acid, rutin, gallic acid, vanillin | Water, 70% ethnol, 80% methanol, chloroform | [80,82,85] |
| Superoxide radical scavenging | Quercetin, kaempferol | Water, 70% ethnol, 80% methanol | [80] |
| Liposome Peroxidation | Quercetin, kaempferol | Water, 70% ethnol, 80% methanol, | [80] |
| Enzymatic Lipid Peroxidation of Microsomes Induced by NADPH/ADP/Fe3+ | Crude extracts, quercetin, kaempferol, gallic, chlorogenic, ellagic, ferulic acid, rutin | Water, 70% ethnol, 80% methanol, acetone, chloroform | [80,82,86] |
| Linoleic Acid Peroxidation System | Quercetin, kaempferol | Water, 70% ethnol, 80% methanol | [80] |
| Superoxide dismutase (SOD) | Crude extracts, quercetin, kaempferol, gallic, chlorogenic, ellagic, ferulic acid, rutin | Chloroform, water, 80% ethanol, acetone | [82,84,86,90] |
| Catalase | Crude extracts, quercetin, kaempferol, gallic, chlorogenic, ellagic, ferulic acid, rutin | Chloroform, water, 80% ethanol, acetone | [82,84,86,90] |
| Glutathione peroxidase | Crude extracts | Water, acetone | [86,90] |
| Nitric oxide (NO) radical scavenging | Crude extracts | Water, acetone | [86] |
| 2,2′-azino-bis-3-ethylbenzothiazoline-6-sulphonic acid (ABTS) | Crude extracts | Water, acetone | [86] |
| Ferric Reducing Iron Power (FRAP) | Crude extract | Water, 80% ethanol, acetone | [85,86] |
| Seed | |||
| UV accelerated method | Crude extract fractions | Chloroform/methanol (1:1), diethylether, n-butanol, and water | [83] |
| DPPH | Crude extract, gallic acid, chlorogenic acid, ellagic acid, ferulic acid, kaempferol, quercetin, vanillin. | Water, 80% ethanol | [84,85] |
| FRAP | Crude extract, gallic acid, chlorogenic acid, ellagic acid, ferulic acid, kaempferol, quercetin, vanillin | Water, 80% ethanol | [84,85] |
| SOD | Crude extract | 80% ethanol | [84] |
| Catalase | Crude extract | 80% ethanol | [84] |
| β-carotene-linoleic acid | gallic acid, chlorogenic acid, ellagic acid, ferulic acid, kaempferol, quercetin, vanillin | Water | [85] |
| Lipid Peroxidation | Crude extracts, gallic acid, chlorogenic acid, ellagic acid, ferulic acid, kaempferol, quercetin, vanillin | Water | [85,88,90] |
| Flower | |||
| DPPH | Crude extract | 80% ethanol | [84] |
| FRAP | Crude extract | 80% ethanol | [84] |
| Superoxide dismutase (SOD) | Crude extract | 80% ethanol | [84] |
| Catalase | Crude extract | 80% ethanol | [84] |
| Pod | |||
| DPPH | Crude extract, gallic acid, chlorogenic acid, ellagic acid, ferulic acid, kaempferol, quercetin, vanillin | Water, 80% ethanol | [84,85] |
| FRAP | Crude extract, gallic acid, chlorogenic acid, ellagic acid, ferulic acid, kaempferol, quercetin, vanillin | Water, 80% ethanol | [84,85] |
| SOD | Crude extract | 80% ethanol | [84] |
| Catalase | Crude extract | 80% ethanol | [84] |
| β-carotene-linoleic acid | Gallic acid, chlorogenic acid, ellagic acid, ferulic acid, kaempferol, quercetin, vanillin | Water | [85] |
| Lipid Peroxidation | Gallic acid, chlorogenic acid, ellagic acid, ferulic acid, kaempferol, quercetin, vanillin | Water | [85] |
| Stem | |||
| DPPH | Crude extract | 80% ethanol | [84] |
| FRAP | Crude extract | 80% ethanol | [84] |
| SOD | Crude extract | 80% ethanol | [84] |
| Catalase | Crude extract | 80% ethanol | [84] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tshabalala, T.; Ncube, B.; Madala, N.E.; Nyakudya, T.T.; Moyo, H.P.; Sibanda, M.; Ndhlala, A.R. Scribbling the Cat: A Case of the “Miracle” Plant, Moringa oleifera. Plants 2019, 8, 510. https://doi.org/10.3390/plants8110510
Tshabalala T, Ncube B, Madala NE, Nyakudya TT, Moyo HP, Sibanda M, Ndhlala AR. Scribbling the Cat: A Case of the “Miracle” Plant, Moringa oleifera. Plants. 2019; 8(11):510. https://doi.org/10.3390/plants8110510
Chicago/Turabian StyleTshabalala, Thulani, Bhekumthetho Ncube, Ntakadzeni Edwin Madala, Trevor Tapiwa Nyakudya, Hloniphani Peter Moyo, Mbulisi Sibanda, and Ashwell Rungano Ndhlala. 2019. "Scribbling the Cat: A Case of the “Miracle” Plant, Moringa oleifera" Plants 8, no. 11: 510. https://doi.org/10.3390/plants8110510
APA StyleTshabalala, T., Ncube, B., Madala, N. E., Nyakudya, T. T., Moyo, H. P., Sibanda, M., & Ndhlala, A. R. (2019). Scribbling the Cat: A Case of the “Miracle” Plant, Moringa oleifera. Plants, 8(11), 510. https://doi.org/10.3390/plants8110510





